Chemical Component Optimization Based on Thermodynamic Calculation of Fe-1.93Mn-0.07Ni-1.96Cr-0.35Mo Ultra-High Strength Steel
Abstract
1. Introduction
2. Thermodynamic Calculation Model
2.1. Microstructure and Phase Calculation Model
2.2. Yield Strength Calculation Model
2.3. Tensile Strength Calculation Model
2.4. Phase Volume and Performance Calculation Model
3. Experimental Materials and Method
3.1. Chemical Composition Design and Optimization
3.2. Mechanical Properties Analysis
4. Results and Discuss
4.1. Phase and Chemical Composition Optimization
4.1.1. Equilibrium Phase Diagram of Original Bainitic Steel
4.1.2. Effect of Ni and Mo Contents on Equilibrium Phase
4.1.3. Effect of Cr and W Content on Equilibrium Precipitation Phase
4.1.4. Effect of the Optimized Chemical Component System on Balanced Phase Diagram
4.2. Effect of Alloy Content on Mechanical Properties
5. Conclusions
- (1).
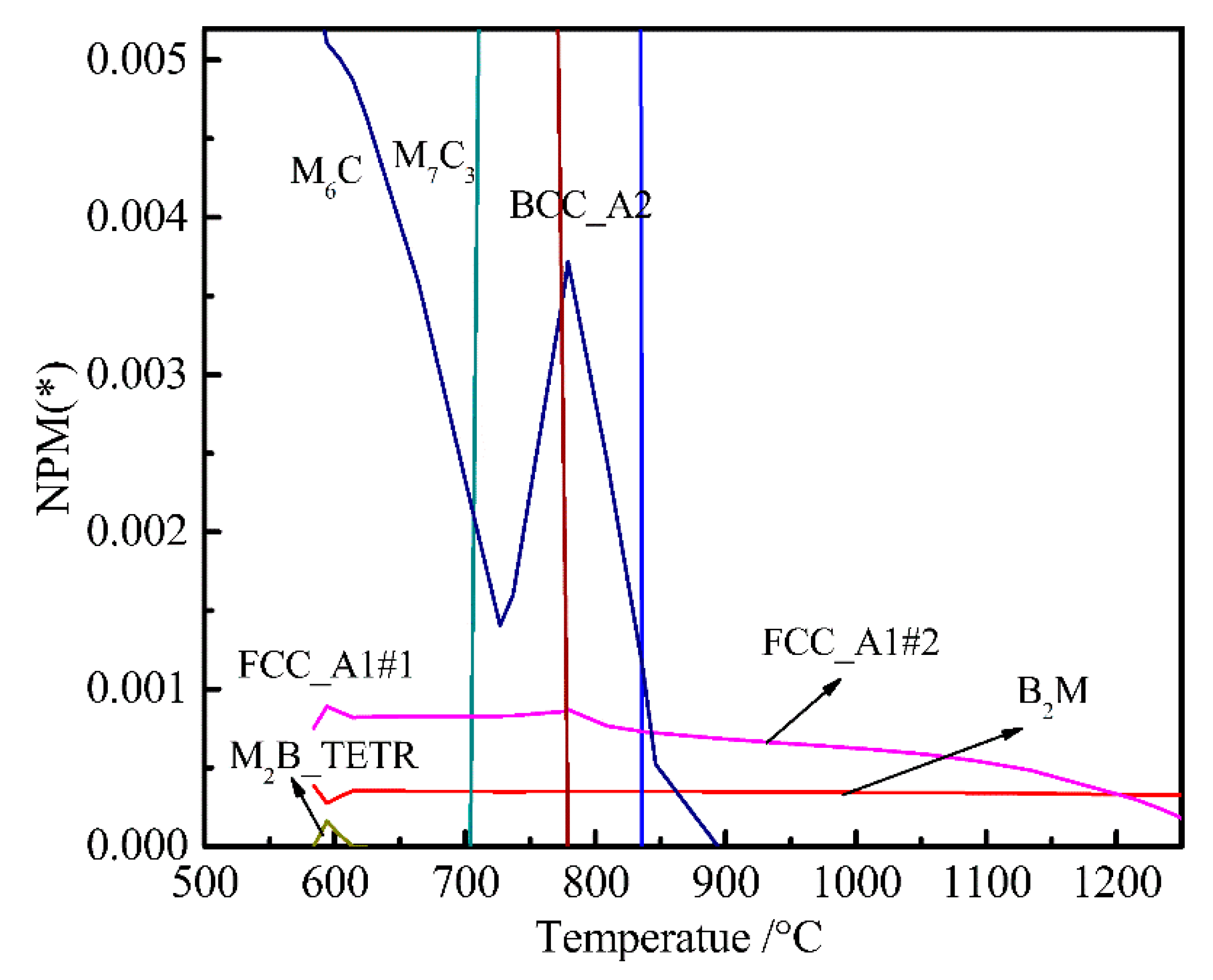
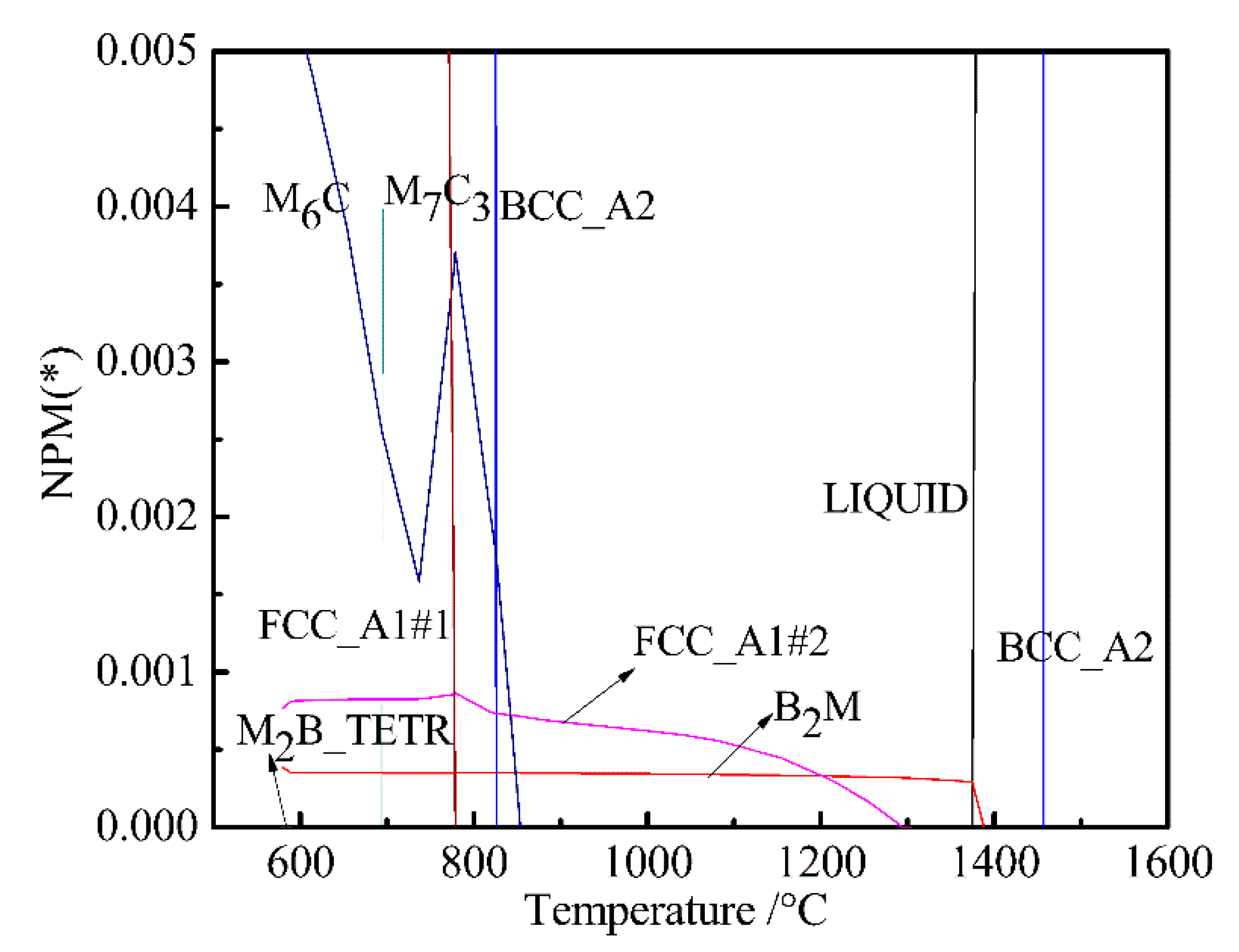
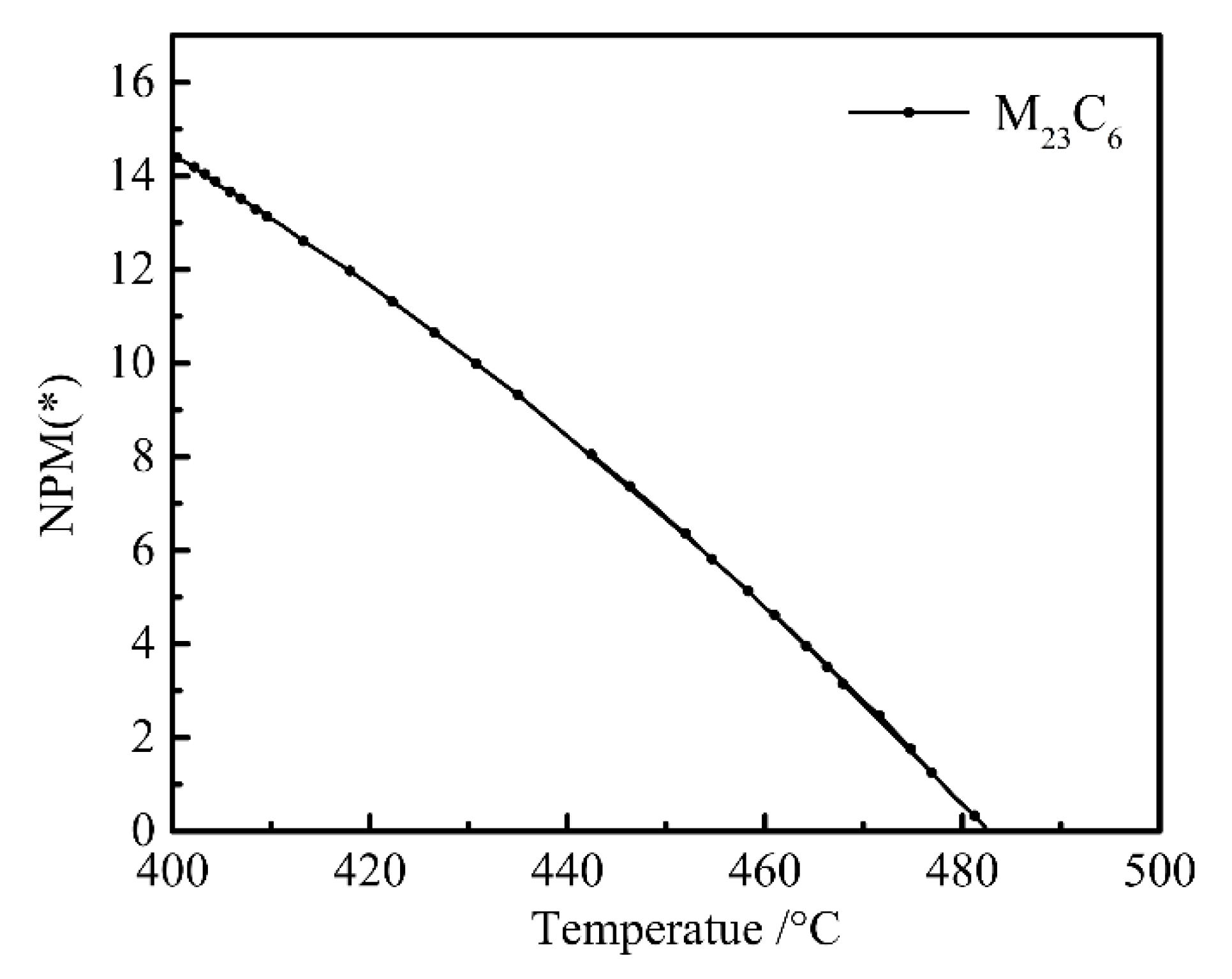
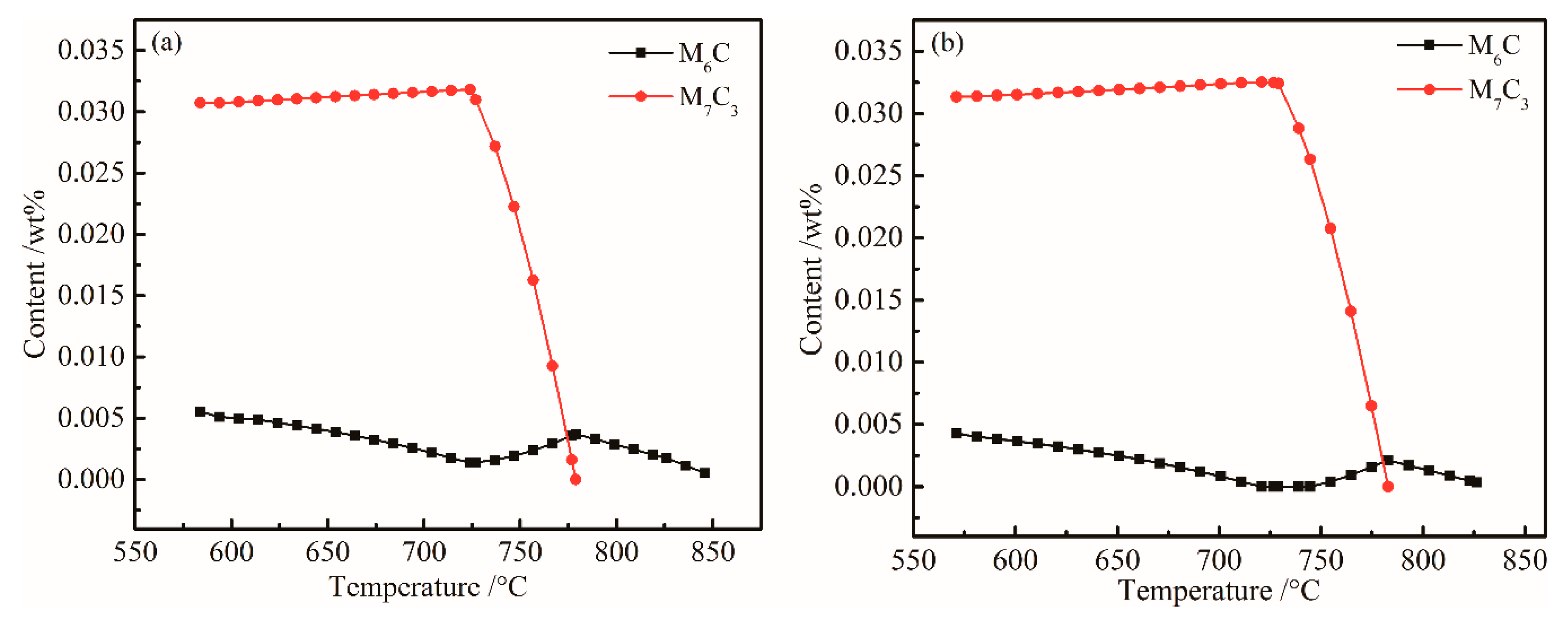
- The equilibrium phases in steel are B2M, BCC_A2, FCC_A1#1, FCCA1#2, M6C, M7C3, and M2B_TETR. B2M is a compound mainly composed of Ti and B. FCC_A1#2 is a compound mainly composed of Ti, C, Cr, and Nb. M6C is a compound mainly composed of Mo, Fe, Si, C, and Cr.
- (2).
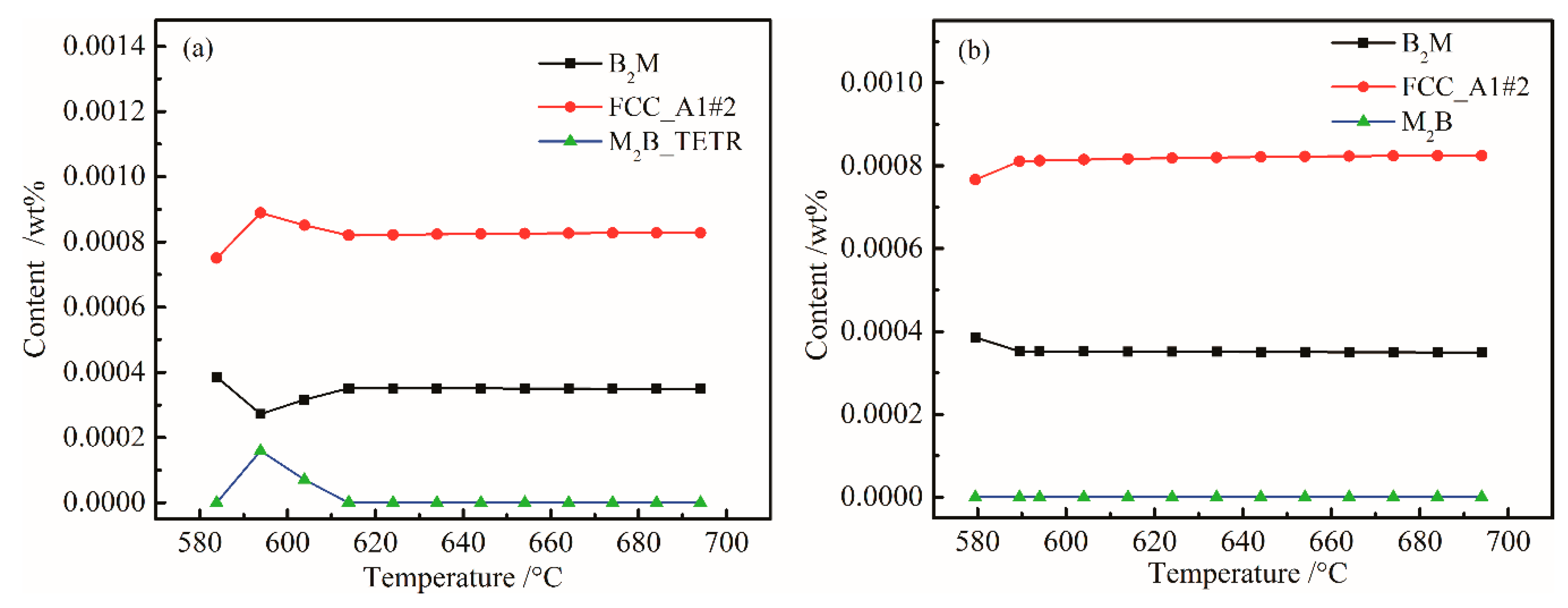
- When Ni content increased from 0 to 0.3%, M6C precision temperature increased from 555 and 575 °C, and the Ni content had little effect on FCC_A1#2 and M6C. Mo is a strong carbide element and forms M6C and M7C3 type carbides in UHSS. Mo content should not be too low, otherwise the strength of UHSS will decrease. M6C carbide with Cr increased with increasing Cr content. Below 800 °C, M7C3 carbide with Cr-rich gradually dissolved into the matrix. W mainly formed M2B_TETR borides. M2B_TETR can be converted with FCC_A1#2 and B2M in the temperature zone around 580 °C.
- (3).
- The chemical component system was optimized by reducing Ni, reducing Mo, removing W, and increasing Cr, then we obtained the same phase diagram as obtained with the origin content. The optimized composition is C 0.23, Si 1.96, Mn 1.93, Ni 0.07, Cr 1.96, Mo 0.35, Nb + V + Ti + Al + Cu + B ≤ 0.15, Fe bal. (wt %). With a cooling rate of 10 °C/s, the optimized alloying system fully performed its strengthening role in the steel, and the chemical components were in the optimal range. The thermodynamic models and our conclusions have the potential to be generalized for many other materials and process configurations without requiring extensive material testing. However, a lack of a real experiment is unfortunate, and the limitations and practicality of this methodology will be verified in future experiments.
Author Contributions
Funding
Conflicts of Interest
References
- Chang, L.C.; Bhadeshia, H.K.D.H. Carbon content of austenite in isothermally transformed 300 M steel. Mater. Sci. Eng. A 1994, 184, 17–19. [Google Scholar] [CrossRef]
- Sule, Y.; Sirin, K.; Kaluc, E. Effect of the ion nitriding surface hardening process on fatigue behavior of AISI 4340 steel. Mater. Charact. 2007, 59, 351–358. [Google Scholar]
- Wen, Z.H.; Kuang-Nian, H.E.; Hua, L.I.; Liao, W.T. Development Progress and Process Analysis of Ultra High-strength Plate. Steel Rolling 2012, 29, 43–45. [Google Scholar]
- Matsuda, H.; Mizuno, R.; Funakawa, Y.; Seto, K.; Matsuoka, S.; Tanaka, Y. Effects of auto-tempering behaviour of martensite on mechanical properties of ultra high strength steel sheets. J. Alloys Compd. 2013, 577, 661–667. [Google Scholar] [CrossRef]
- Abe, Y.; Kato, T.; Mori, K.I.; Nishino, S. Mechanical clinching of ultra-high strength steel sheets and strength of joints. J. Mater. Process. Technol. 2014, 214, 2112–2118. [Google Scholar] [CrossRef]
- Little, C.D.; Machmeier, P.M. High Strength Fracture Resistant Weldable Steels. U.S. Patent 4,076,525, 28 February 1978. [Google Scholar]
- Jahazi, M.; Egbali, B. The influence of hot rolling parameters on the microstructure and mechanical properties of an ultra-high strength steel. J. Mater. Process. Technol. 2000, 103, 276–279. [Google Scholar] [CrossRef]
- Hillert, M.; Staffansson, L.I.; Hillert, M.; Staffansson, L.I. The Regular Solution Model for Stoichiometric Phases and Ionic Melts. Acta Chem. Scand. 1970, 24, 3618–3626. [Google Scholar] [CrossRef]
- Sundman, B.; Ågren, J. A regular solution model for phases with several components and sublattices, suitable for computer applications. J. Phys. Chem. Solids 1981, 42, 297–301. [Google Scholar] [CrossRef]
- Porter, D.A.; Easterling, K.E. Phase Transformations In Metals and Alloys. Ann. Rev. Mater. Res. 1992, 1, 213–218. [Google Scholar]
- Nieh, T.G.; Wadsworth, J. Hall-Petch Relation in Nanocrystalline Solids. Scr. Metall. Mater. 1991, 25, 955–958. [Google Scholar] [CrossRef]
- Kirkaldy, J.S. Prediction of alloy hardenability from thermodynamic and kinetic data. Metall. Mater. Trans. B 1973, 4, 2327–2333. [Google Scholar] [CrossRef]
- Doane, D.V.; Kirkaldy, J.S. Hardenability Concepts with Applications to Steel. In Proceedings of the Symposium Held at the Sheraton-Chicago Hotel, Chicago, IL, USA, 24–26 October 1977. [Google Scholar]
- International, A.; Davis, J.R.; Committee, A.I.H. Properties and Selection: Irons, Steels and High-Performance Alloys; ASM International: Materials Park, OH, USA, 2001. [Google Scholar]
- Predel, E.H.C.B.; Hoch, E.M.; Pool, E.M. Effect of Diffusion on Phase Transformations; Springer: Berlin, Germany, 2004. [Google Scholar]
- Burke, J. The Kinetics of Phase Transformations in Metals; Pergamon Press: Long Island City, NY, USA, 1965. [Google Scholar]
- Porter, D.A.; Easterling, K.E.; Sherif, M.Y. Phase Transformations In Metals and Alloys; Chapman & Hall: London, UK, 1992. [Google Scholar]
- Committee, A.I.H.; Davis, J.R.; Abel, L.A. Properties and Selection: Irons, Steels, and High-Performance Alloys; ASM International: Materials Park, OH, USA, 1995. [Google Scholar]
- Egea, A.J.S.; Rojas, H.A.G.; Celentano, D.J.; Perio, J.J.; Cao, J. Thermomechanical Analysis of an Electrically Assisted Wire Drawing Process. J. Manuf. Sci. Eng.-Trans. ASME 2017, 139, 7. [Google Scholar] [CrossRef]
- Egea, A.J.S.; Rojas, H.A.G.; Celentano, D.J.; Peiro, J.J. Mechanical and metallurgical changes on 308L wires drawn by electropulses. Mater. Des. 2016, 90, 1159–1169. [Google Scholar] [CrossRef]
- Allen, R.M.; Toth, L.S.; Oppedal, A.L.; El Kadiri, H. Crystal Plasticity Modeling of Anisotropic Hardening and Texture Due to Dislocation Transmutation in Twinning. Materials 2018, 11, 1855. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Kouadri-Henni, A.; Gavrus, A. Numerical simulation and experimental investigation on the residual stresses in a laser beam welded dual phase DP600 steel plate: Thermo-mechanical material plasticity model. Int. J. Mech. Sci. 2017, 122, 235–243. [Google Scholar] [CrossRef]
- Wang, Z.Q.; Denlinger, E.; Michaleris, P.; Stoica, A.D.; Ma, D.; Beese, A.M. Residual stress mapping in Inconel 625 fabricated through additive manufacturing: Method for neutron diffraction measurements to validate thermomechanical model predictions. Mater. Des. 2017, 113, 169–177. [Google Scholar] [CrossRef]
- Porter, D.A.; Easterling, K.E.; Sherif, M. Phase Transformations in Metals and Alloys; Revised Reprint; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Hall, E.O. The Deformation and Ageing of Mild Steel: III Discussion of Results. Proc. Phys. Soc. Sect. B 1951, 64, 747–751. [Google Scholar] [CrossRef]
- Petch, N.J. The Cleavage Strength of Polycrystals. J. Iron Steel Inst. 1953, 174, 25–28. [Google Scholar]
- Gladman, T. On the Theory of the Effect of Precipitate Particles on Grain Growth in Metals. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1966, 294, 298–309. [Google Scholar]
- Doane, D.V. Hardenability Concepts with Applications to Steel; American Institute of Mining: Centennial, CO, USA, 1978. [Google Scholar]
- Kattner, U.R.; Boettinger, W.J.; Coriell, S.R. Application of Lukas’ phase diagram programs to solidification calculations of multicomponent alloys. Z. Metallk. 1996, 87, 522–528. [Google Scholar]
- Predel, B.; Hoch, M.; Pool, M. Effect of diffusion on Phase Transformations. In Phase Diagrams and Heterogeneous Equilibria; Predel, B., Hoch, M., Pool, M., Eds.; Springer: Berlin, Germany, 2004. [Google Scholar]
- Christian, J.W. The Theory of Transformations in Metals and Alloys; Elsevier Science Ltd.: Kidlington, Oxford, UK, 2002. [Google Scholar]
- Caballero, F.G.; Santofimia, M.J.; Garcia-Mateo, C.; Chao, J.; de Andres, C.G. Theoretical design and advanced microstructure in super high strength steels. Mater. Des. 2009, 30, 2077–2083. [Google Scholar] [CrossRef]
- Aggen, G.; Allen, M. ASM Handbook Volume I Properties and Selection: Irons, Steels, and High-Performance Alloys; ASM International: Materials Park, OH, USA, 2018. [Google Scholar]
- Li, L. Precipitation of Carbonitrides Containing V and Nb in Steel and their Stability. Shanghai Met. 2005, 27, 1–3. [Google Scholar]
- Thomson, R.C.; Miller, M.K. Carbide precipitation in martensite during the early stages of tempering Cr- andMo-containing low alloy steels. Acta Mater. 1998, 46, 2203–2213. [Google Scholar] [CrossRef]
- Yang, S.; He, X.; Chen, M.; Dang, Z.; Jun, K. Strain Induced Precipitation at High Temperature in (Nb, B.) Microalloyed Steel. Mater. Sci. Eng. 1994, 12, 49–65. [Google Scholar]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Zhou, X.; Huang, J. Chemical Component Optimization Based on Thermodynamic Calculation of Fe-1.93Mn-0.07Ni-1.96Cr-0.35Mo Ultra-High Strength Steel. Materials 2019, 12, 65. https://doi.org/10.3390/ma12010065
Chen Y, Zhou X, Huang J. Chemical Component Optimization Based on Thermodynamic Calculation of Fe-1.93Mn-0.07Ni-1.96Cr-0.35Mo Ultra-High Strength Steel. Materials. 2019; 12(1):65. https://doi.org/10.3390/ma12010065
Chicago/Turabian StyleChen, Yongli, Xuejiao Zhou, and Jianguo Huang. 2019. "Chemical Component Optimization Based on Thermodynamic Calculation of Fe-1.93Mn-0.07Ni-1.96Cr-0.35Mo Ultra-High Strength Steel" Materials 12, no. 1: 65. https://doi.org/10.3390/ma12010065
APA StyleChen, Y., Zhou, X., & Huang, J. (2019). Chemical Component Optimization Based on Thermodynamic Calculation of Fe-1.93Mn-0.07Ni-1.96Cr-0.35Mo Ultra-High Strength Steel. Materials, 12(1), 65. https://doi.org/10.3390/ma12010065




