Optimization of Micro and Nano Palm Oil Fuel Ash to Determine the Carbonation Resistance of the Concrete in Accelerated Condition
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Binders
2.1.2. High-Energy Ball Mill
2.1.3. Aggregates
2.1.4. Superplasticizer
2.2. Concrete Mix Proportions and Specimens Preparation
2.3. Testing Procedures
2.3.1. X-ray Fluorescence (XRF)
2.3.2. X-ray Diffraction (XRD)
2.3.3. Scanning Electron Microscopy (SEM)
2.3.4. Slump Test
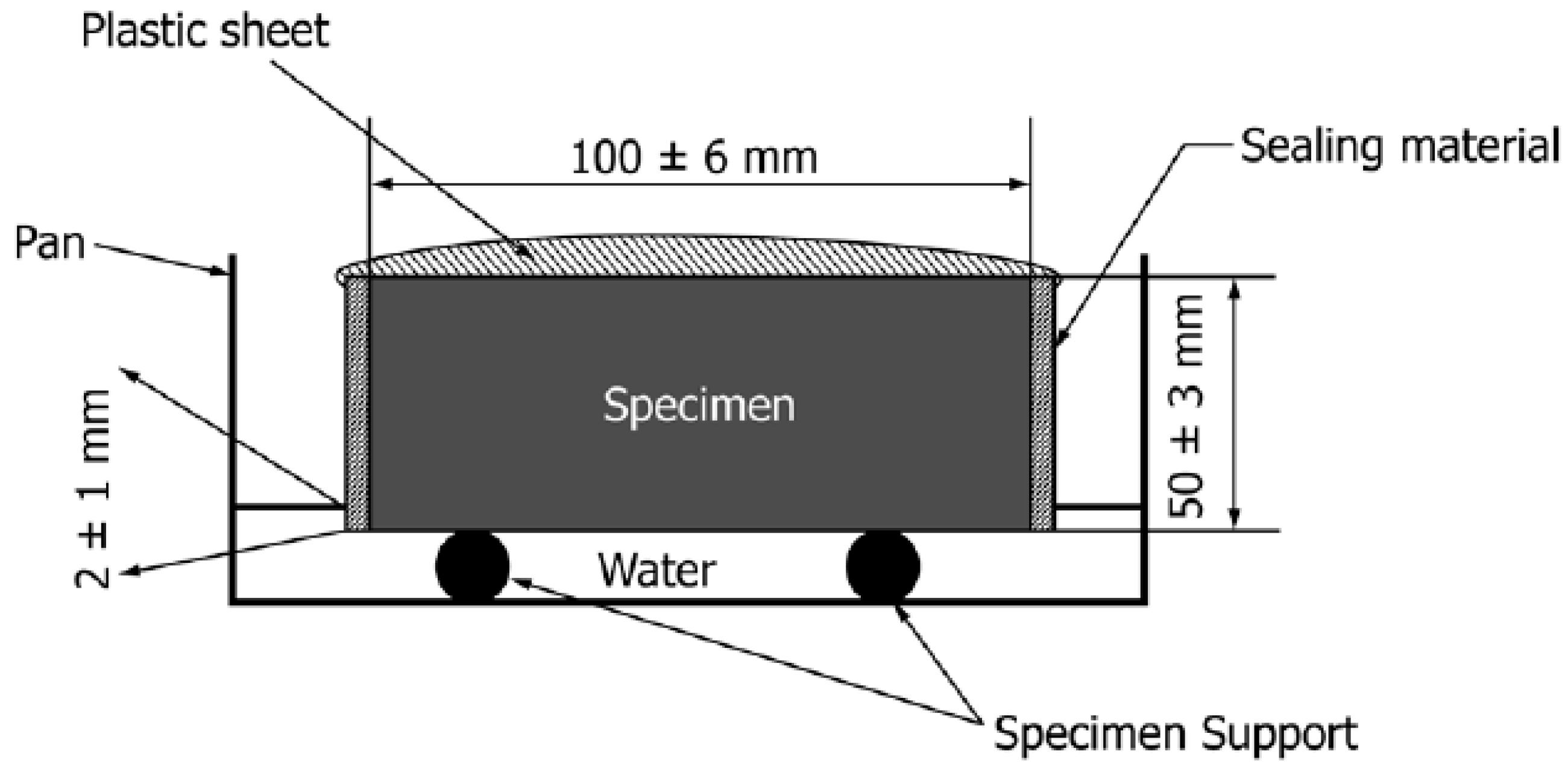
2.3.5. Carbonation Experiment: Accelerated Method
2.3.6. Sorptivity Test
3. Test Results and Discussion
3.1. Characterization of POFA
3.1.1. X-ray Fluorescence
3.1.2. X-Ray Diffraction
3.1.3. Scanning Electron Microscope
3.2. Workability of Concretes
3.3. Microstructure of Concretes
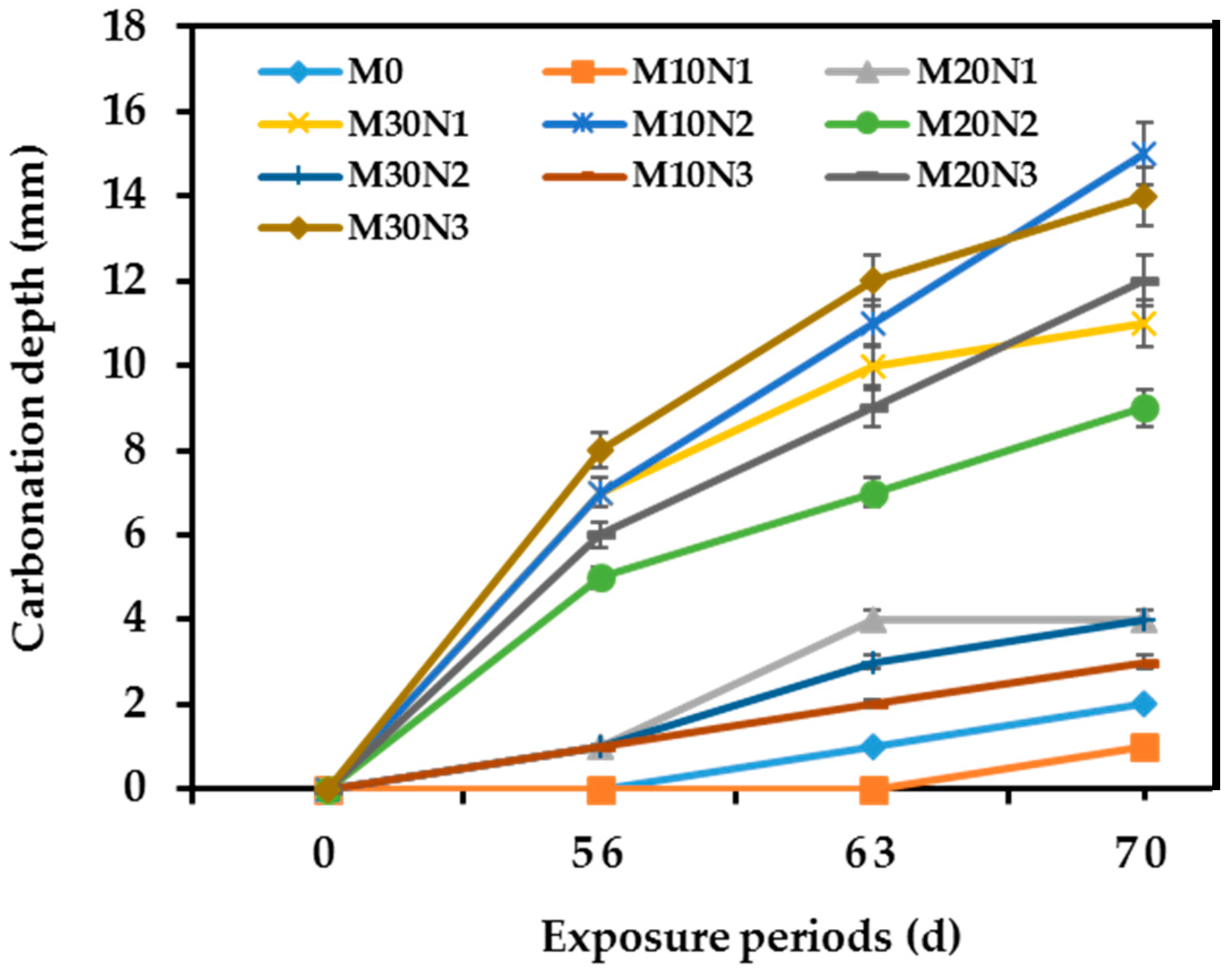
3.4. Carbonation Depth
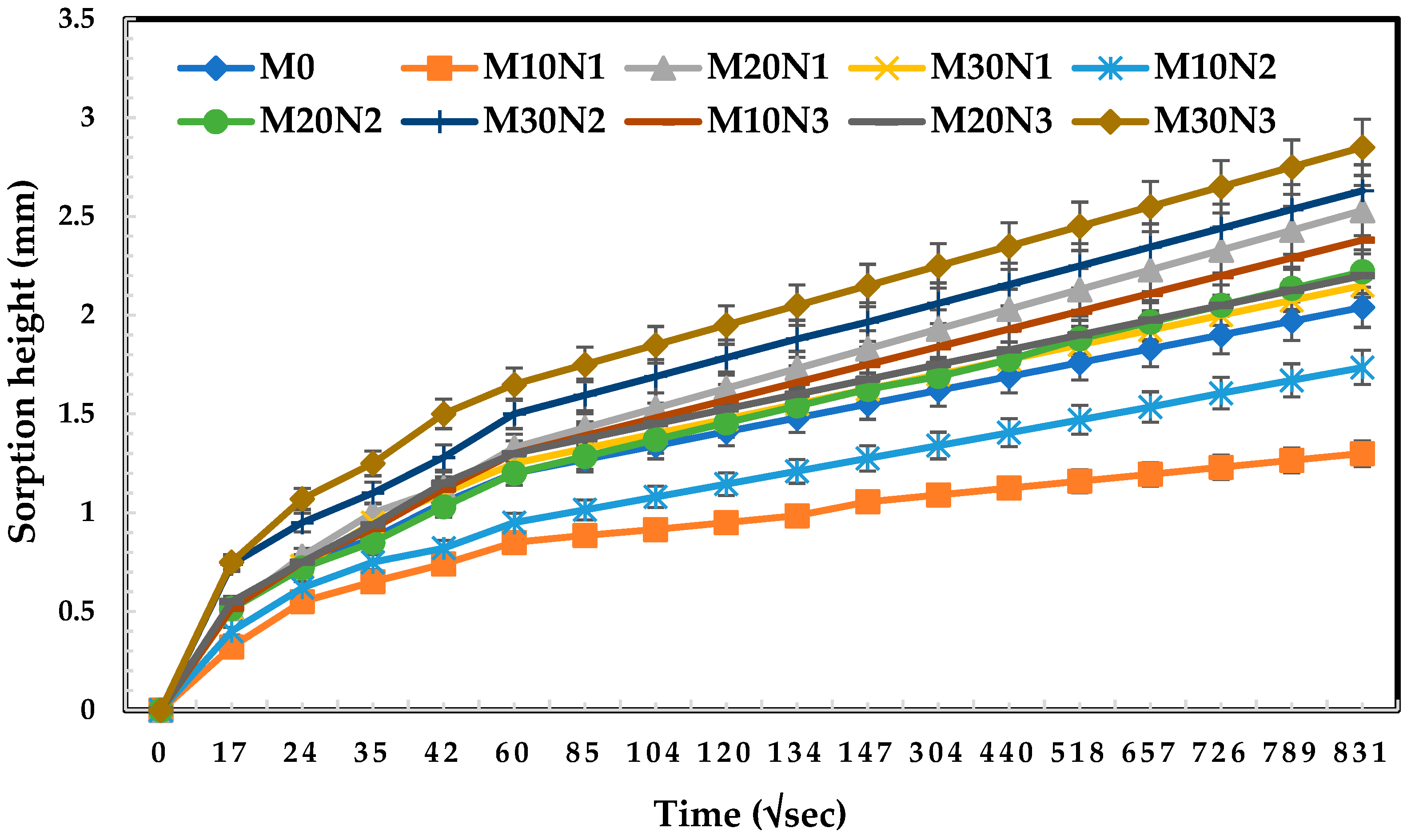
3.5. Water Sorption
4. Conclusions
- (a)
- XRF results show that POFA has higher SiO2 as well as SiO2 + Al2O3 + Fe2O3 compared with OPC.
- (b)
- The LOI value of treated POFA is greatly reduced compared with raw POFA. After the treatment of raw POFA, the LOI value is reduced by 28.75% and 33.33% for mPOFA and nPOFA, respectively.
- (c)
- XRD results reveal that mPOFA and nPOFA contain two major phases, namely Quartz and Cristobalite, along with amorphous silica, which participated in pozzolanic reaction.
- (d)
- SEM results show that mPOFA had crushed or irregular-shaped particles and found to be bigger in size compared with nPOFA.
- (e)
- Inclusion of nPOFA can reduce the size of pores in the concrete matrix due to its better micro-filling ability than mPOFA.
- (f)
- The concrete mixture with 10% mPOFA and 0.5% nPOFA, designated as M10N1, can have higher carbonation resistance and lesser sorptivity compared with OPC and other mixtures. However, a higher amount of micro- and nano-POFA has detrimental and negative effects on carbonation resistance and water sorptivity results.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Keulemans, G. The Problem with Reinforced Concrete. The Conversation AU. 2016. Available online: http://theconversation.com/the-problem-with-reinforced-concrete-56078 (accessed on 17 June 2016).
- Hussain, S.; Bhunia, D.; Singh, S.B. Comparative Study of Accelerated Carbonation of Plain Cement and Fly-Ash Concrete. J. Build. Eng. 2017, 10, 26–31. [Google Scholar] [CrossRef]
- Talukdar, S.; Banthia, N. Carbonation in Concrete Infrastructure in the Context of Global Climate Change: Development of a Service Lifespan Model. Constr. Build. Mater. 2013, 40, 775–782. [Google Scholar] [CrossRef]
- Hussain, S.; Bhunia, D.; Singh, S.B. Assessment of Carbonation Depth under Natural and Accelerated Carbonation Conditions. Indian Concr. J. 2016, 57–64. [Google Scholar]
- Mobin, T.R.; Muthupriya, P. Determination of Concrete Carbonation Depth by Experimental Investigation. Int. J. Eng. Sci. Invent. Res. Dev. 2016, 2, 534–541. [Google Scholar]
- Pham, S.T.; Prince, W. Effects of Carbonation on the Microstructure of Cement Materials: Influence of Measuring Methods and Types of Cement. Int. J. Concr. Struct. Mater. 2014, 8, 327–333. [Google Scholar] [CrossRef]
- Collado, J.; Go, J.; Rosanto, M.; Tan, J.L.; Jesus, R.D. Determination of Carbonation Depth of Structures in Intramuros Using Artificial Neural Network. Proc. DLSU Res. Congr. 2015, 3, 1–7. Available online: http://www.dlsu.edu.ph/conferences/dlsu_research_congress/2015/proceedings/SEE/006-SEE_Go_JA.pdf (accessed on 2 March 2015).
- Bouchaala, F.; Payan, C.; Garnier, V.; Balayssac, J.P. Carbonation Assessment in Concrete by Nonlinear Ultrasound. Cem. Concr. Res. 2011, 41, 557–559. [Google Scholar] [CrossRef]
- Gopi, S.P.; Subramanian, V.K.; Palanisamy, K. Aragonite–Calcite–Vaterite: A Temperature Influenced Sequential Polymorphic Transformation of CaCO3 in the Presence of DTPA. Mater. Res. Bull. 2013, 48, 1906–1912. [Google Scholar] [CrossRef]
- López-Arce, P.; Gómez-Villalba, L.S.; Martínez-Ramírez, S.; de Buergo, M.Á.; Fort, R. Influence of Relative Humidity on the Carbonation of Calcium Hydroxide Nanoparticles and the Formation of Calcium Carbonate Polymorphs. Powder Technol. 2011, 205, 263–269. [Google Scholar] [CrossRef]
- Uddin, M.T.; Islam, M.N.; Sutradhar, S.K.; Chowdhury, M.H.R.; Hasnat, A.; Khatib, J.M. Carbonation Coefficient of Concrete in Dhaka City. In Proceedings of the 3rd International Conference on Sustainable Construction Materials and Technologies, Kyoto, Japan, 18–22 August 2013. [Google Scholar]
- Lee, H.M.; Lee, H.S.; Singh, J.K. Prediction Model for the Carbonation of Post-Repair Materials in Carbonated RC Structures. Materials 2017, 10, 492. [Google Scholar]
- Lee, H.-M.; Lee, H.-S.; Min, S.-H.; Lim, S.; Singh, J.K. Carbonation-Induced Corrosion Initiation Probability of Rebars in Concrete with/without Finishing Materials. Sustainability 2018, 10, 3814. [Google Scholar] [CrossRef]
- Glinicki, M.A.; Józwiak-Niedzwiedzka, D.; Gibas, K.; Dabrowski, M. Influence of Blended Cements with Calcareous Fly Ash on Chloride Ion Migration and Carbonation Resistance of Concrete for Durable Structures. Materials 2016, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Lange, L.C.; Hills, C.D.; Poole, A.B. Effect of Carbonation on Properties of Blended and Non-Blended Cement Solidified Waste Forms. J. Hazard. Mater. 1997, 52, 193–212. [Google Scholar] [CrossRef]
- Thomas, B.S.; Kumar, S.; Arel, H.S. Sustainable Concrete Containing Palm Oil Fuel Ash as a Supplementary Cementitious Material—A Review. Renew. Sustain. Energy Rev. 2017, 80, 550–561. [Google Scholar] [CrossRef]
- Kroehong, W.; Sinsiri, T.; Jaturapitakkul, C.; Chindaprasirt, P. Effect of Palm Oil Fuel Ash Fineness on the Microstructure of Blended Cement Paste. Constr. Build. Mater. 2011, 25, 4095–4104. [Google Scholar] [CrossRef]
- Islam, U.; Momeen, M.; Mo, K.H.; Alengaram, U.J.; Jumaat, M.Z. Durability Properties of Sustainable Concrete Containing High Volume Palm Oil Waste Materials. J. Clean. Prod. 2016, 137, 167–177. [Google Scholar] [CrossRef]
- Abdul Awal, A.S.M.; Hussin, M.W. The Effectiveness of Palm Oil Fuel Ash in Preventing Expansion Due to Alkali–Silica Reaction. Cem. Concr. Compos. 1997, 19, 367–372. [Google Scholar] [CrossRef]
- Tay, J.H. Ash from Oil-Palm Waste as Concrete Material. J. Mater. Civ. Eng. 1990, 2, 94–105. [Google Scholar] [CrossRef]
- Chindaprasirt, P.; Homwuttiwong, S.; Jaturapitakkul, C. Strength and Water Permeability of Concrete Containing Palm Oil Fuel Ash and Rice Husk-Bark Ash. Constr. Build. Mater. 2007, 21, 1492–1499. [Google Scholar] [CrossRef]
- Sata, V.; Jaturapitakkul, C.; Kiattikomol, K. Utilization of Palm Oil Fuel Ash in High-Strength Concrete. J. Mater. Civ. Eng. 2004, 16, 623–628. [Google Scholar] [CrossRef]
- Tangchirapat, W.; Khamklai, S.; Jaturapitakkul, C. Use of Ground Palm Oil Fuel Ash to Improve Strength, Sulfate Resistance, and Water Permeability of Concrete Containing High Amount of Recycled Concrete Aggregates. Mater. Des. 2012, 41, 150–157. [Google Scholar] [CrossRef]
- Altwair, N.M.; Johari, M.A.M.; Hashim, S.F.S. Strength Activity Index and Microstructural Characteristics of Treated Palm Oil Fuel Ash. Int. J. Civ. Environ. Eng. 2011, 11, 85–92. [Google Scholar]
- Tangchirapat, W.; Jaturapitakkul, C.; Chindaprasirt, P. Use of Palm Oil Fuel Ash as a Supplementary Cementitious Material for Producing High-Strength Concrete. Constr. Build. Mater. 2009, 23, 2641–2646. [Google Scholar] [CrossRef]
- Sata, V.; Jaturapitakkul, C.; Kiattikomol, K. Influence of Pozzolan from Various By-product Materials on Mechanical Properties of High-Strength Concrete. Constr. Build. Mater. 2007, 21, 1589–1598. [Google Scholar] [CrossRef]
- Johari, M.A.M.; Zeyad, A.M.; Bunnori, N.M.; Ariffin, K.S. Engineering and Transport Properties of High-Strength Green Concrete Containing High Volume of Ultrafine Palm Oil Fuel Ash. Constr. Buil. Mater. 2012, 30, 281–288. [Google Scholar] [CrossRef]
- Hassan, W.N.F.W.; Ismail, M.A.; Lee, H.-S.; Hussin, M.W.; Ismail, M.A.; Singh, J.K. Utilization of Nano Agricultural Waste to Improve the Workability and Early Strength of Concrete. Int. J. Sus. Build. Technol. Urban Dev. 2017, 8, 316–331. [Google Scholar]
- Rajak, M.A.A.; Majid, Z.A.; Ismail, M. Morphological Characteristics of Hardened Cement Pastes Incorporating Nano-Palm Oil Fuel Ash. Proced. Manuf. 2015, 2, 512–518. [Google Scholar] [CrossRef]
- Zeyad, A.M.; Johari, M.A.M.; Tayeh, B.A.; Yusuf, M.O. Pozzolanic Reactivity of Ultrafine Palm Oil Fuel Ash Waste on Strength and Durability Performances of High Strength Concrete. J. Clean. Prod. 2017, 144, 511–522. [Google Scholar] [CrossRef]
- BS 1881-210:2013, Determination of the Potential Carbonation Resistance of Concrete–Accelerated Carbonation Method; British Standards: London, UK, 2013.
- Wi, K.; Lee, H.-S.; Lim, S.; Song, H.; Hussin, M.W.; Ismail, M.A. Use of an Agricultural By-product, Nano Sized Palm Oil Fuel Ash as a Supplementary Cementitious Material. Constr. Build. Mater. 2018, 183, 139–149. [Google Scholar] [CrossRef]
- Al-mulali, M.Z.; Awang, H.; Khalil, H.P.S.A.; Aljoumaily, Z.S. The Incorporation of Oil Palm Ash in Concrete as a Means of Recycling: A Review. Cem. Concr. Comp. 2015, 55, 129–138. [Google Scholar] [CrossRef]
- Zeyad, A.M.; Johari, M.A.M.; Bunnori, N.M.; Ariffin, K.S.; Aktwair, N.M. Characteristics of Treated Palm Oil Fuel and Its Effects on Properties of High Strength Concrete. Adv. Mater. Res. 2013, 626, 152–156. [Google Scholar] [CrossRef]
- Chandara, C.; Azizli, K.A.M.; Ahmad, Z.A.; Hashim, S.F.S.; Sakai, E. Heat of Hydration of Blended Cement Containing Treated Ground Palm Oil Fuel Ash. Constr. Build. Mater. 2012, 27, 78–81. [Google Scholar] [CrossRef]
- Lim, N.H.A.S.; Ismail, M.A.; Lee, H.S.; Hussin, M.W.; Sam, A.R.M.; Samadi, M. The Effects of High Volume Nano Palm Oil Fuel Ash on Microstructure Properties and Hydration Temperature of Mortar. Constr. Building Mater. 2015, 93, 29–34. [Google Scholar] [CrossRef]
- Tammy, R.; Andrew, R.; Claire, R. The Capco Pascall Catalogue. Ball Mills. 2006. Available online: http://www.celsiuslab.co.za/pascall_catalogue%20-%20CAPCO.pdf (accessed on 15 July 2017).
- Rizlan, Z.; Mamat, O. Process Parameters Optimization of Silica Sand Nanoparticles Production Using Low Speed Ball Milling Method. Chin. J. Eng. 2014, 802459. [Google Scholar] [CrossRef]
- Guide for Selecting Proportions for High-Strength Concrete with Portland Cement and Fly Ash; ACI 211.4R-08; American Concrete Institute: Farmington Hills, MI, USA, 2008.
- ASTM C 33-16, Standards Specification for Concrete Aggregates. Annual Book of ASTM Standard; American Society for Testing and Materials: West Conshohocken, PA, USA, 2016.
- ASTM C 494-16, Standards Specification for Chemical Admixtures for Concrete. Annual Book of ASTM Standard; American Society for Testing and Materials: West Conshohocken, PA, USA, 2016.
- ASTM C143, Standard Test Method for Slump of Hydraulic Cement Concrete. Annual Book of ASTM Standard; American Society for Testing and Materials: West Conshohocken, PA, USA, 2015.
- McGrath, P.F. A Simple Chamber for Accelerated Carbonation Testing of Concrete. Available online: Available online: https://www.xypex.com.au/file/10310/284 (accessed on 15 August 2018).
- ASTM C 1585-13, Standards Specification for Measurement of Rate of Absorption of Water by Hydraulic Cement Concretes. Annual Book of ASTM Standard; American Society for Testing and Materials: West Conshohocken, PA, USA, 2016.
- Qiang, X.; Zhan, S.; Xu, B.; Yang, H.; Qian, X.; Ding, X. Effect of Isobutyl-Triethoxy-Silane Penetrative Protective Agent on the Carbonation Resistance of Concrete. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2016, 31, 139–145. [Google Scholar] [CrossRef]
- Aprianti, S.E. A Huge Number of Artificial Waste Material Can Be Supplementary Cementitious Material (Scm) for Concrete Production—A Review Part II. J. Clean. Prod. 2017, 142, 4178–4194. [Google Scholar] [CrossRef]
- Altwair, N.M.; Johari, M.A.M.; Hashim, S.F.S. Influence of Calcination Temperature on Characteristics and Pozzolanic Activity of Palm Oil Waste Ash. Aust. J. Basic Appl. Sci. 2011, 5, 1010–1018. [Google Scholar]
- Alsubari, B.; Shafigh, P.; Jumaat, M. Development of Self-Consolidating High Strength Concrete Incorporating Treated Palm Oil Fuel Ash. Materials 2015, 8, 2154–2173. [Google Scholar] [CrossRef]
- Safiuddin, M.; Abdus Salam, M.; Jumaat, M.Z. Utilization of palm oil fuel ash in concrete: A review. J. Civ. Eng. Manag. 2011, 17, 234–247. [Google Scholar] [CrossRef]
- Tay, J.-H.; Show, K.-Y. Use of Ash Derived from Oil-Palm Waste Incineration as a Cement Replacement Material. Resour. Conserv. Recycl. 1995, 13, 27–36. [Google Scholar] [CrossRef]
- Safiuddin, M.; Jumaat, M. Fresh Properties of Self-Consolidating Concrete Incorporating Palm Oil Fuel Ash as a Supplementary Cementing Material. Chiang Mai J. Sci. 2011, 38, 389–404. [Google Scholar]
- Alsubari, B.; Shafigh, P.; Jumaat, M.Z.; Alengaram, U.J. Palm Oil Fuel Ash as a Partial Cement Replacement for Producing Durable Self-Consolidating High-Strength Concrete. Arab. J. Sci. Eng. 2014, 39, 8507–8516. [Google Scholar] [CrossRef]
- Chandara, C.; Sakai, E.; Azizli, K.A.M.; Ahmad, Z.A.; Hashim, S.F.S. The Effect of Unburned Carbon in Palm Oil Fuel Ash on Fluidity of Cement Pastes Containing Superplasticizer. Constr. Build. Mater. 2010, 24, 1590–1593. [Google Scholar] [CrossRef]
- Alsubari, B.; Shafigh, P.; Ibrahim, Z.; Jumaat, M.Z. Heat-Treated Palm Oil Fuel Ash as an Effective Supplementary Cementitious Material Originating from Agriculture Waste. Constr. Buil. Mater. 2018, 167, 44–54. [Google Scholar] [CrossRef]







| Mixture | OPC | mPOFA | nPOFA | Mixture | OPC | mPOFA | nPOFA |
|---|---|---|---|---|---|---|---|
| M0 | 100% | 0% | 0% | M20N2 | 79.0% | 20% | 1.0% |
| M10N1 | 89.5% | 10% | 0.5% | M30N2 | 69.0% | 30% | 1.0% |
| M20N1 | 79.5% | 20% | 0.5% | M10N3 | 88.5% | 10% | 1.5% |
| M30N1 | 69.5% | 30% | 0.5% | M20N3 | 78.5% | 20% | 1.5% |
| M10N2 | 89.0% | 10% | 1.0% | M30N3 | 68.5% | 30% | 1.5% |
| Mixture | Cement | POFA | Coarse Aggregates | Quarry Dust | Sand | Water | SP Dosage (0.2% of Binder *) | ||
|---|---|---|---|---|---|---|---|---|---|
| Micro | Nano | Wet | Dry | ||||||
| M0 | 2.9 | 0 | 0 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M10N1 | 2.6 | 0.288 | 0.0144 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M20N1 | 2.31 | 0.58 | 0.0144 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M30N1 | 2.02 | 0.871 | 0.0144 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M10N2 | 2.58 | 0.288 | 0.0288 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M20N2 | 2.3 | 0.58 | 0.0288 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M30N2 | 2 | 0.871 | 0.0288 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M10N3 | 2.57 | 0.288 | 0.0432 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M20N3 | 2.28 | 0.58 | 0.0432 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| M30N3 | 1.99 | 0.871 | 0.0432 | 5.40 | 1.33 | 0.266 | 1.06 | 0.90 | 0.0058 |
| Time | 1 min | 5 min | 10 min | 20 min | 30 min | 60 min |
| Tolerance | 2 s | 10 s | 2 min | 2 min | 2 min | 2 min |
| Chemical Composition (%) | OPC | Raw POFA | mPOFA | nPOFA |
|---|---|---|---|---|
| SiO2 | 16.40 | 59.1 | 69.19 | 68.07 |
| Al2O3 | 4.24 | 4.5 | 3.34 | 3.71 |
| Fe2O3 | 3.53 | 6.5 | 3.19 | 3.24 |
| CaO | 68.30 | 8.6 | 6.70 | 7.41 |
| MgO | 2.39 | 2.6 | 4.65 | 5.10 |
| SO3 | 4.39 | 2.7 | 0.605 | 0.626 |
| SiO2 + Al2O3 + Fe2O3 | 24.17 | 70.1 | 75.19 | 75.02 |
| LOI | 2.40 | 10.5 | 1.71 | 1.60 |
| Mixture | Slump Value (mm) | Slump Loss (mm) | Mixture | Slump Value (mm) | Slump Loss (mm) |
|---|---|---|---|---|---|
| M0 | 140 | 0 | M20N2 | 65 | 75 |
| M10N1 | 125 | 15 | M30N2 | 45 | 95 |
| M20N1 | 50 | 90 | M10N3 | 145 | −5 |
| M30N1 | 40 | 100 | M20N3 | 80 | 60 |
| M10N2 | 140 | 0 | M30N3 | 65 | 75 |
| Mixture | Crystal Size (nm) | ||
|---|---|---|---|
| CH | C-S-H | Ettringite | |
| M0 | 560 | - | - |
| M10N3 | 249.6 | 128 | 219.2 |
| M30N1 | 600 | 192 | - |
| M30N3 | - | 144 | - |
| M10N1 | 216 | 167 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, W.L.; Lee, H.-S.; Vimonsatit, V.; Htut, T.; Singh, J.K.; Wan Hassan, W.N.F.; Ismail, M.A.; Seikh, A.H.; Alharthi, N. Optimization of Micro and Nano Palm Oil Fuel Ash to Determine the Carbonation Resistance of the Concrete in Accelerated Condition. Materials 2019, 12, 130. https://doi.org/10.3390/ma12010130
Tang WL, Lee H-S, Vimonsatit V, Htut T, Singh JK, Wan Hassan WNF, Ismail MA, Seikh AH, Alharthi N. Optimization of Micro and Nano Palm Oil Fuel Ash to Determine the Carbonation Resistance of the Concrete in Accelerated Condition. Materials. 2019; 12(1):130. https://doi.org/10.3390/ma12010130
Chicago/Turabian StyleTang, Wei Le, Han-Seung Lee, Vanissorn Vimonsatit, Trevor Htut, Jitendra Kumar Singh, Wan Nur Firdaus Wan Hassan, Mohamed A. Ismail, Asiful H. Seikh, and Nabeel Alharthi. 2019. "Optimization of Micro and Nano Palm Oil Fuel Ash to Determine the Carbonation Resistance of the Concrete in Accelerated Condition" Materials 12, no. 1: 130. https://doi.org/10.3390/ma12010130
APA StyleTang, W. L., Lee, H.-S., Vimonsatit, V., Htut, T., Singh, J. K., Wan Hassan, W. N. F., Ismail, M. A., Seikh, A. H., & Alharthi, N. (2019). Optimization of Micro and Nano Palm Oil Fuel Ash to Determine the Carbonation Resistance of the Concrete in Accelerated Condition. Materials, 12(1), 130. https://doi.org/10.3390/ma12010130







