Hydrogen Peroxide Diffusion through Enamel and Dentin
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Selection
2.2. Sample Preservation and Preparation
2.3. Preparation of the Experimental Reservoir
2.4. Products Used and Bleaching Protocol
2.5. Measurement of HP Diffusion
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bowles, W.H.; Ugwuneri, Z. Pulp chamber penetration by hydrogen peroxide following vital bleaching procedures. J. Endod. 1987, 13, 375–377. [Google Scholar] [CrossRef]
- Camargo, S.E.; Cardoso, P.E.; Valera, M.C.; de Araújo, M.A.; Kojima, A.N. Penetration of 35% hydrogen peroxide into the pulp chamber in bovine teeth after LED or Nd: YAG laser activation. Eur. J. Estht. Dent. 2009, 4, 82–88. [Google Scholar]
- Sulieman, M. An overview of bleaching techniques: I. History, chemistry, safety and legal aspects. Dent. Updat. 2004, 31, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, M.Q. Tooth-bleaching procedures and their controversial effects: A literature review. Saudi Dent. J. 2014, 26, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Joiner, A. The bleaching of teeth: A review of the literature. J. Dent. 2006, 34, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Minoux, M.; Serfaty, R. Vital tooth bleaching: Biologic adverse effects––A review. Quintessence Int. 2008, 39, 645–659. [Google Scholar] [PubMed]
- Ubaldini, A.L.; Baesso, M.L.; Medina-Neto, A.; Sato, F.; Bento, A.C.; Pascotto, R.C. Hydrogen peroxide diffusion dynamics in dental tissues. J. Dent. Res. 2013, 92, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Li, Q.; Wang, Y. Effects of pH values of hydrogen peroxide bleaching agents on enamel surface properties. Oper. Dent. 2011, 36, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Al-Qunaian, T.A.; Matis, B.A.; Cochra, M.A. In vivo kinetics of bleaching gel with three-percent hydrogen peroxide within the first hour. Oper. Dent. 2003, 28, 236–241. [Google Scholar] [PubMed]
- Matis, B.A.; Gaiao, U.; Blackman, D.; Schultz, F.A.; Eckert, G.J. In vivo degradation of bleaching gel used in whitening teeth. J. Am. Dent. Assoc. 1999, 130, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Marson, F.C.; Gonçalves, R.S.; dos Santos, P.H.; Cintra, L.T.; Pascotto, R.C.; Santos, P.H.; Briso, A.L. Penetration of hydrogen peroxide and degradation rate of different bleaching product. Oper. Dent. 2015, 40, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Sulieman, M.; Addy, M.; MacDonald, E.; Rees, J.S. The effect of hydrogen peroxide concentration on the outcome of tooth whitening: An in vitro study. J. Dent. 2004, 32, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, J.B.; McPharlin, R.; Paravina, R.D.; Ferracane, J.L. Comparison of at-home and in-office tooth whitening using a novel shade guide. Oper. Dent. 2010, 35, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Matis, B.A.; Cochran, M.A.; Franco, M.; Al-Ammar, W.; Eckert, G.J.; Stropes, M. Eight in-office tooth whitening systems evaluated in vivo: A pilot study. Oper. Dent. 2007, 32, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.G.; Basso, F.G.; Pontes, E.C.; da FR Garcia, L.; Hebling, J.; de Souza Costa, C.A. Effective tooth-bleaching protocols capable of reducing H(2)O(2) diffusion through enamel and dentine. J. Dent. 2014, 42, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Llena, C.; Forner, L.; Vazquez, M. Hydrogen peroxide diffusion with and without light activation. Int. J. Esthet. Dent. 2016, 11, 430–441. [Google Scholar] [PubMed]
- Kwon, S.R.; Li, Y.; Oyoyo, U.; Aprecio, R.M. Dynamic model of hydrogen peroxide diffusion kinetics into the pulp cavity. J. Contemp. Dent. Pract. 2012, 13, 440–445. [Google Scholar] [PubMed]
- Mottola, H.A.; Simpson, B.E.; Gorin, G. Absorptiometric determination of hydrogen peroxide in submicrogram amounts with leuco crystal violet and peroxidase as catalyst. Anal. Chem. 1970, 42, 410–411. [Google Scholar] [CrossRef]
- Soares, D.G.; Ribeiro, A.P.; da Silveira Vargas, F.; Hebling, J.; de Souza Costa, C.A. Efficacy and cytotoxicity of a bleaching gel after short application times on dental enamel. Clin. Oral. Investig. 2013, 17, 1901–1909. [Google Scholar] [CrossRef] [PubMed]
- Duque, C.C.; Soares, D.G.; Basso, F.G.; de Souza Costa, C.A. Bleaching effectiveness, hydrogen peroxide diffusion, and cytotoxicity of a chemically activated bleaching gel. Clin. Oral. Investig. 2014, 18, 1631–1637. [Google Scholar] [PubMed]
- Yazaki, K.; Kawada, E.; Oda, Y. An evaluation of the penetration of peroxide from tooth-whitener. Biomed. Res. 2003, 24, 325–330. [Google Scholar] [CrossRef]
- Barja de Quiroga, C. Mitochondrial oxygen radical generation and leak: Sites of produccion in states 4 and 3, organ specificity and relation to aging and longevity. J. Bioenerg. Biomembr. 1999, 31, 347–366. [Google Scholar] [CrossRef]
- Torres, C.R.; Souza, C.S.; Borges, A.G.; Huhtala, M.F.; Caneppele, T.M. Influence of concentration and activation on hydrogen peroxide diffusion through dental tissues in vitro. Sci. World J. 2013, 18, 193–241. [Google Scholar]
- Hairul Nizam, B.R.; Lim, C.T.; Chng, H.K.; Yap, A.U.J. Nanoindentation study of human premolars subjected to bleaching agent. J. Biomech. 2005, 38, 2204–2211. [Google Scholar] [CrossRef] [PubMed]
- Chng, H.K.; Ramli, H.N.; Yap, A.U.J.; Lim, C.T. Effect of hydrogen peroxide on intertubular dentine. J. Dent. 2007, 33, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, B.; Datko, L.; Cupelli, M.; Alapati, S.; Dean, D.; Kennedy, M. Alteration of dentin-enamel mechanical properties due to dental whitening treatments. J. Mech. Behav. Biomed. Mater. 2010, 3, 339–346. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, L.C.; Soares, D.G.; Gallinari, M.O.; de Souza Costa, C.A.; Dos Santos, P.H.; Briso, A.L. Color alteration, hydrogen peroxide diffusion, and cytotoxicity caused by in-office bleaching protocols. Clin. Oral. Investig. 2005, 19, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Gökay, O.; Müjdeci, A.; Algn, E. Peroxide penetration into the pulp from whitening strips. J. Endod. 2004, 30, 887–889. [Google Scholar] [CrossRef] [PubMed]
- Hannig, C.; Weinhold, H.C.; Becker, K.; Attin, T. Diffusion of peroxides through dentine in vitro with and without prior use of a desensitizing varnish. Clin. Oral. Investig. 2011, 15, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Kalia, Y.N.; Guy, R.H. Modeling transdermal drug release. Adv. Drug. Deliv. Rev. 2001, 48, 159–172. [Google Scholar] [CrossRef]
- Soares, D.G.; Marcomini, N.; Basso, F.G.; Pansani, T.N.; Hebling, J.; de Souza Costa, C.A. Indirect cytocompatibility of a low-concentration hydrogen peroxide bleaching gel to odontoblast-like cells. Int. Endod. J. 2016, 49, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Dietschi, D.; Rossier, S.; Krejci, I. In vitro colorimetric evaluation of the efficacy of various bleaching methods and products. Quintessence Int. 2006, 37, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.R.; Oyoyo, U.; Li, Y. Effect of light activation on tooth whitening efficacy and hydrogen peroxide penetration: An in vitro study. J. Dent. 2013, 41, e39–e45. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.G.; Basso, F.G.; Hebling, J.; de Souza Costa, C.A. Concentrations of and application protocols for hydrogen peroxide bleaching gels: Effects on pulp cell viability and whitening efficacy. J. Dent. 2014, 42, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Llena, C.; Collado-González, M.; Tomás-Catalá, C.J.; García-Bernal, D.; Oñate-Sánchez, R.E.; Rodríguez-Lozano, F.J.; Forner, L. Human Dental Pulp Stem Cells Exhibit Different Biological Behaviours in Response to Commercial Bleaching Products. Materials 2018, 11, 1098. [Google Scholar] [CrossRef] [PubMed]
- Mena-Serrano, A.P.; Parreiras, S.O.; do Nascimento, E.M.; Berger, S.B.; Loguercio, A.D.; Reis, A. Effects of the concentration and composition of in-office bleaching gels on hydrogen peroxide penetration into the pulp chamber. Oper. Dent. 2015, 40, E76–E82. [Google Scholar] [CrossRef] [PubMed]
- Berger, S.B.; Tabchoury, C.P.; Ambrosano, G.M.; Giannini, M. Hydrogen peroxide penetration into the pulp chamber and dental permeability after bleaching. Gen. Dent. 2013, 61, e21–e25. [Google Scholar] [PubMed]
- Buzoglu, H.D.; Gümüsderelioğlu, M.; Rotstein, I. Effect of bleaching agents on surface free energy parameters of resin composite coated with saliva biofilm. Am. J. Dent. 2009, 22, 223–227. [Google Scholar] [PubMed]
- Kwon, S.R.; Pallavi, F.; Shi, Y.; Oyoyo, U.; Mohraz, A.; Li, Y. Effect of bleaching gel viscosity on tooth whitening efficacy and pulp chamber penetration: An in vitro study. Oper. Dent. 2018, 43, 326–334. [Google Scholar] [CrossRef] [PubMed]
| Product | Without Light Activation | With Light Activation | ||
|---|---|---|---|---|
| Opalescence Boost, HP 40% Ultradent, South Jordan, UT, USA (BT) | 3 × 10 min | 1 × 30 min | 3 × (3 min activ. + 5 min without activ.) | 1 × (3 min activ. + 25 min without activ.) |
| NorBlanc Office Automix, HP 37.5%—Normon, Madrid, Spain (NO) | 3 × 10 min | 1 × 30 min | 3 × (3 min activ. + 5 min without activ.) | 1 × (3 min activ. + 25 min without activ.) |
| Pola Office +, HP 37.5%—SDI, Bayswater, Victoria, Australia (PO) | 3 × 10 min | 1 × 30 min | 3 × (3 min activ. + 5 min without activ.) | 1 × (3 min activ. + 25 min without activ.) |
| Perfect Bleach, HP 35%—Voco, Cuxhaven, Germany (PB) | 3 × 10 min | 1 × 30 min | 3 × (3 min activ. + 5 min without activ.) | 1 × (3 min activ. + 25 min without activ.) |
| PolaDay CP, CP 35%—SDI, Bayswater, Victoria, Australia (PD) | 3 × 10 min | 1 × 30 min | 3 × (3 min activ. + 5 min without activ.) | 1 × (3 min activ. + 25 min without activ.) |
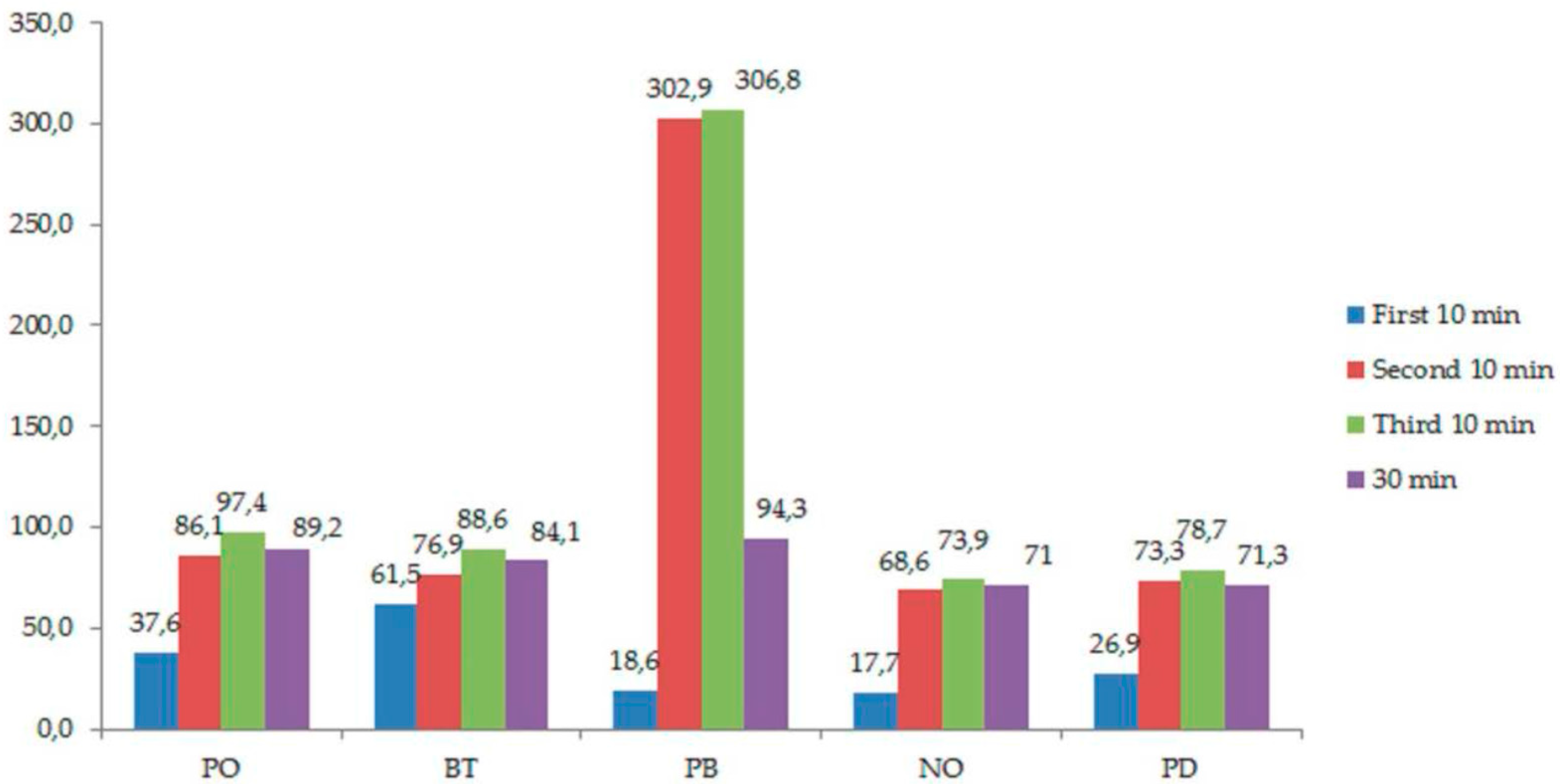
| Without Light Activation | |||||
|---|---|---|---|---|---|
| Pola Office + (PO) | Boost (BT) | Perfect Bleach (PB) | NorBlanc Office Automix (NO) | PolaDay CP (PD) | |
| First 10 min | 37.6 (31.4–43.7) | 61.5 (53.9–69) | 18.6 (7.4–29.6) a | 17.7 (11.2–24.0) a1 | 26.9 (19.3–34.5) a |
| Second 10 min | 86.1 (69.7–102.3) a | 76.9 (74.5–79.2) ab | 302.9 (264.9–340.9) 1 | 68.6 (60.2–76.8) b2 | 73.3 (64.3–82.1) a1 |
| Third 10 min | 97.4 (76.5–118.2) a | 88.6 (80–97.1) a | 306.8 (259–354.6) 2 | 73.9 (65.8–82) b3 | 78.7 (67.3–90) b2 |
| 30 min | 89.2 (83.6–99.4) a | 84.1 (79.3–96.2) a | 94.3 (88.46–100) | 71.0 (59.3–83.3) b4 | 71.3 (58.3–84.6) b3 |
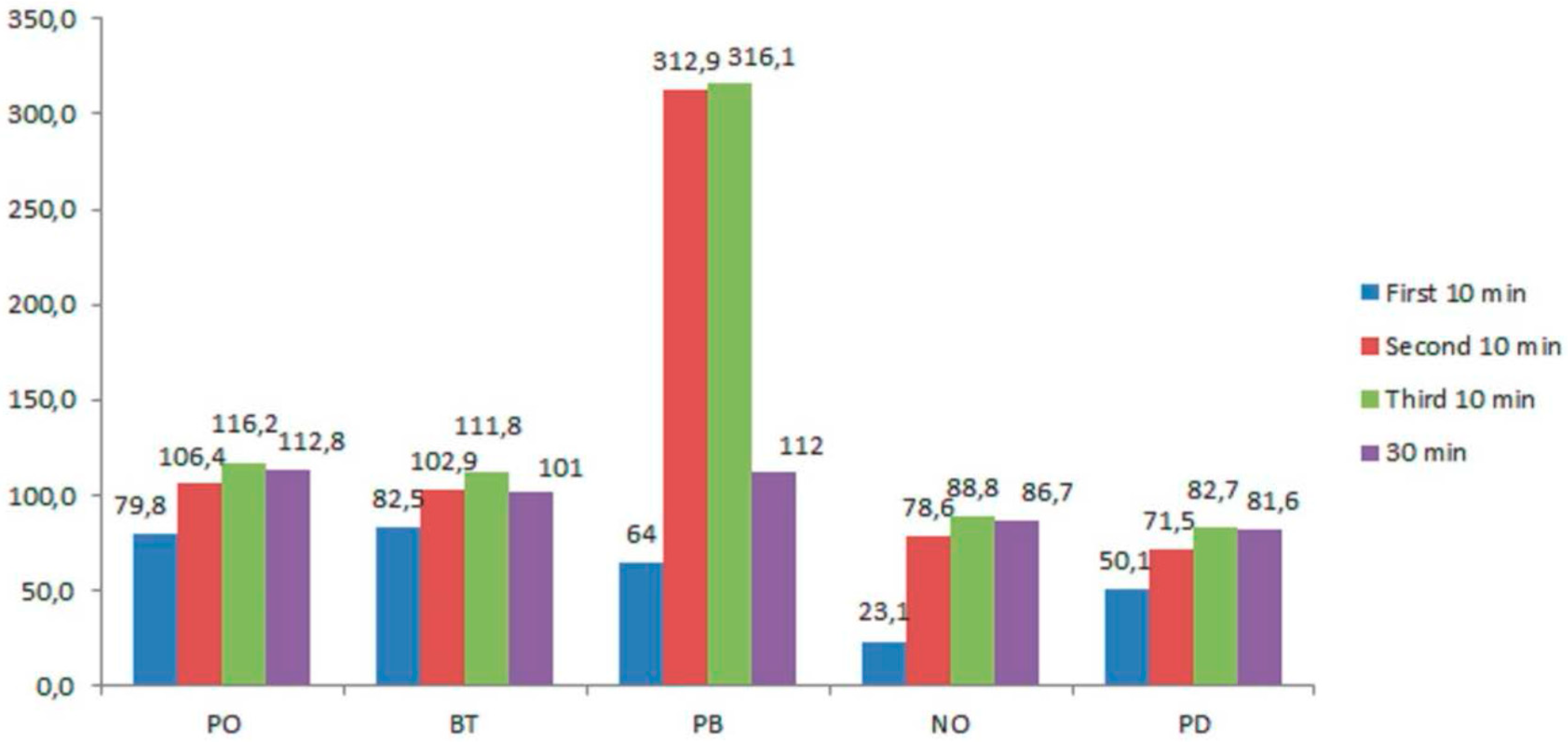
| With Light Activation | |||||
| First 10 min | 79.8 (163.2–96.4) b | 82.5 (63.9–100.9) b | 64 (52.1–75.9) a | 23.1 (11–35) 1 | 50.1 (39.9–60.2) a |
| Second 10 min | 106.4 (93.3–119.4) b | 102.9 (187.7–1) b | 312.90 (281.4–344.2) 1 | 78.6 (69.2–87.8) a2 | 71.5 (65.6–77.5) a1 |
| Third 10 min | 116.2 (102.2–130.1) b | 111.8 (97.7–125.9) b | 316.15 (286.8–345.3) 2 | 88.8 (75–102.6) a3 | 82.7 (78.9–86.4) a2 |
| 30 min | 112.8 (98.2–126.3) b | 101 (93.7–113.8) b | 112.01 (98.6–123.7) b | 86.7 (65.3–98.6) a4 | 81.6 (69.4–96.7) a3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llena, C.; Martínez-Galdón, O.; Forner, L.; Gimeno-Mallench, L.; Rodríguez-Lozano, F.J.; Gambini, J. Hydrogen Peroxide Diffusion through Enamel and Dentin. Materials 2018, 11, 1694. https://doi.org/10.3390/ma11091694
Llena C, Martínez-Galdón O, Forner L, Gimeno-Mallench L, Rodríguez-Lozano FJ, Gambini J. Hydrogen Peroxide Diffusion through Enamel and Dentin. Materials. 2018; 11(9):1694. https://doi.org/10.3390/ma11091694
Chicago/Turabian StyleLlena, Carmen, Oreto Martínez-Galdón, Leopoldo Forner, Lucía Gimeno-Mallench, Francisco J. Rodríguez-Lozano, and Juan Gambini. 2018. "Hydrogen Peroxide Diffusion through Enamel and Dentin" Materials 11, no. 9: 1694. https://doi.org/10.3390/ma11091694
APA StyleLlena, C., Martínez-Galdón, O., Forner, L., Gimeno-Mallench, L., Rodríguez-Lozano, F. J., & Gambini, J. (2018). Hydrogen Peroxide Diffusion through Enamel and Dentin. Materials, 11(9), 1694. https://doi.org/10.3390/ma11091694






