Synthesis and Irreversible Thermochromic Sensor Applications of Manganese Violet
Abstract
1. Introduction
2. Materials and Methods
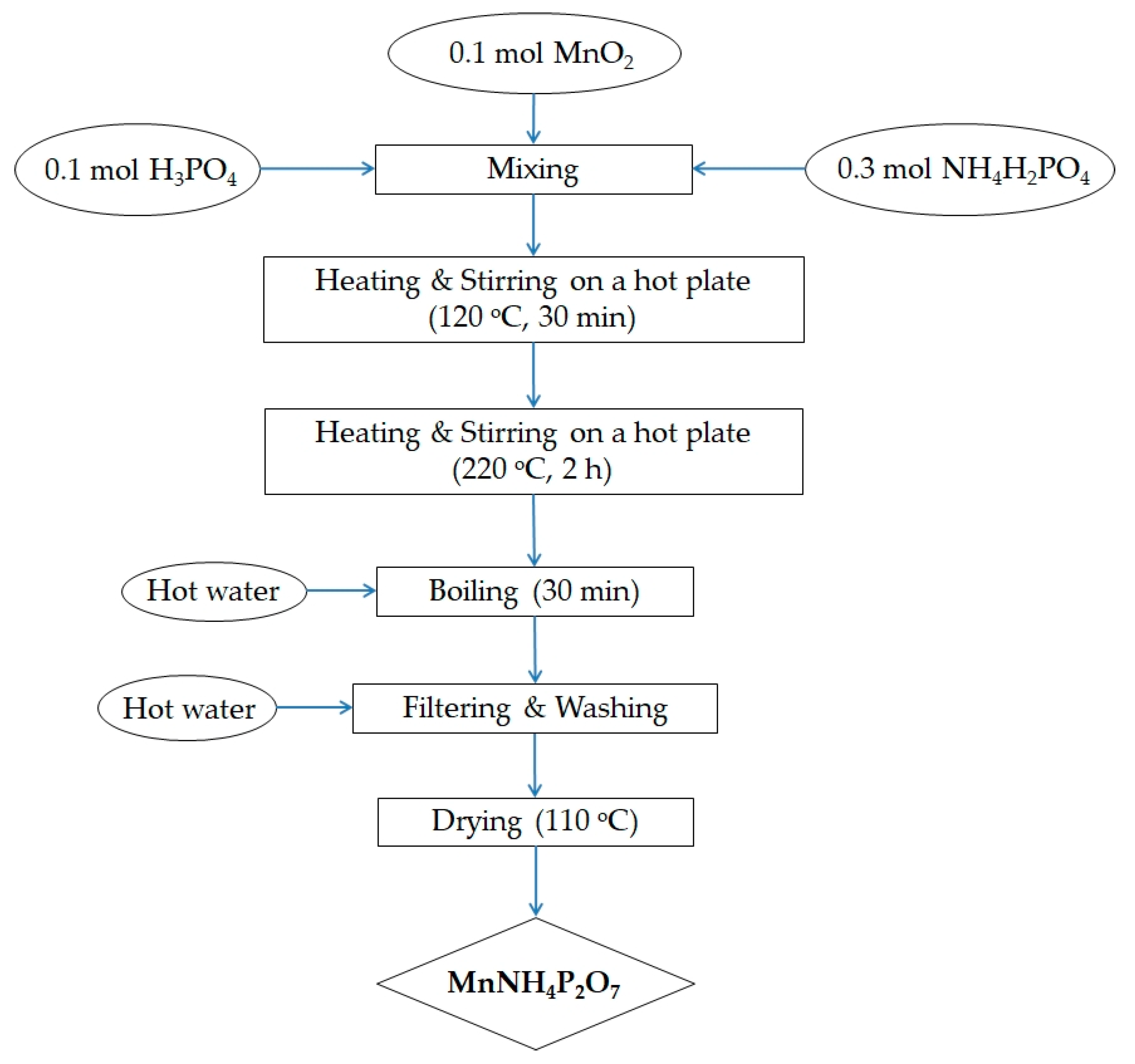
2.1. Synthesis
2.2. Characterization Techniques
2.3. Thermochromic Sensor Fabrication
3. Results and Discussion
3.1. Structure and Properties of Manganese Violet
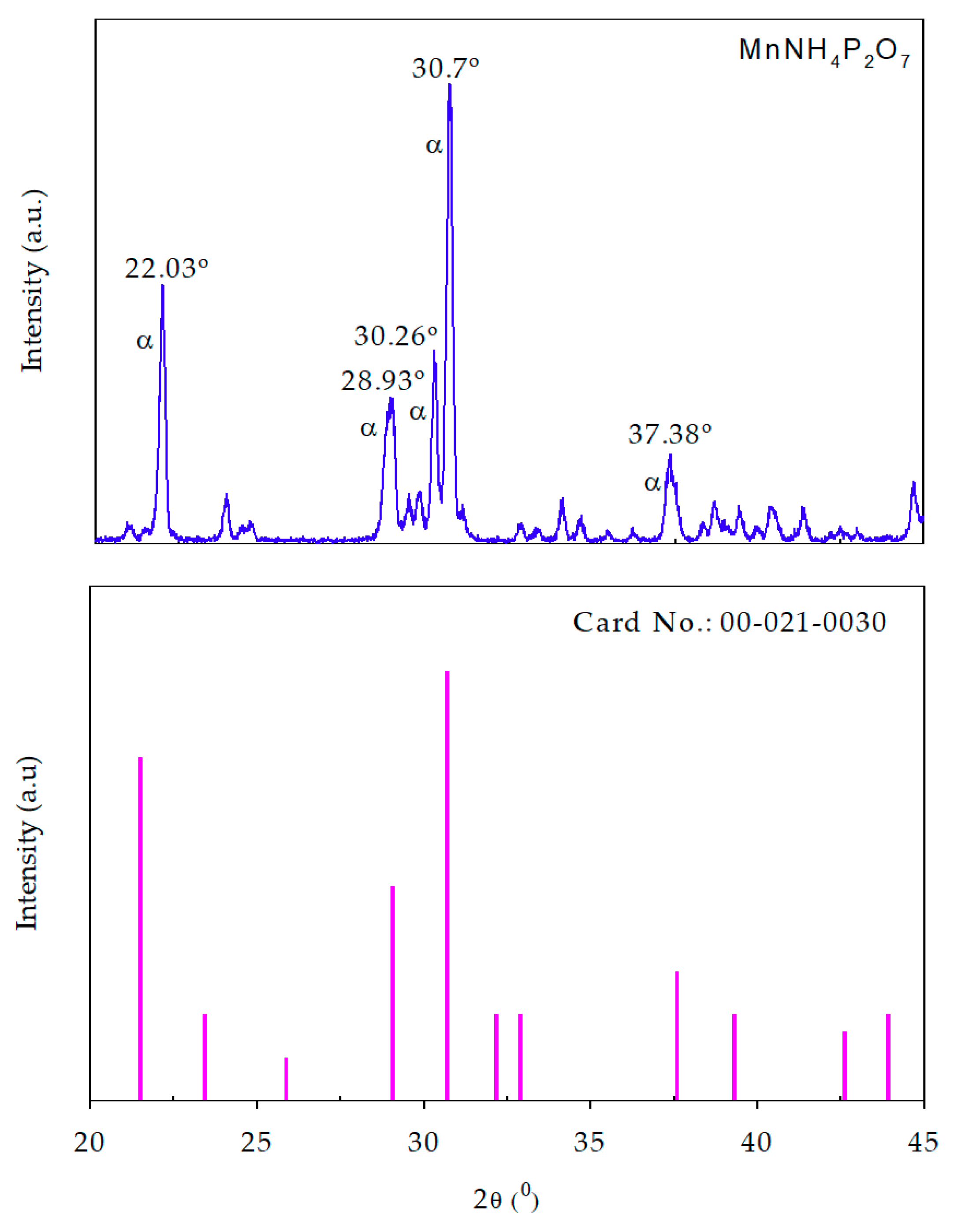
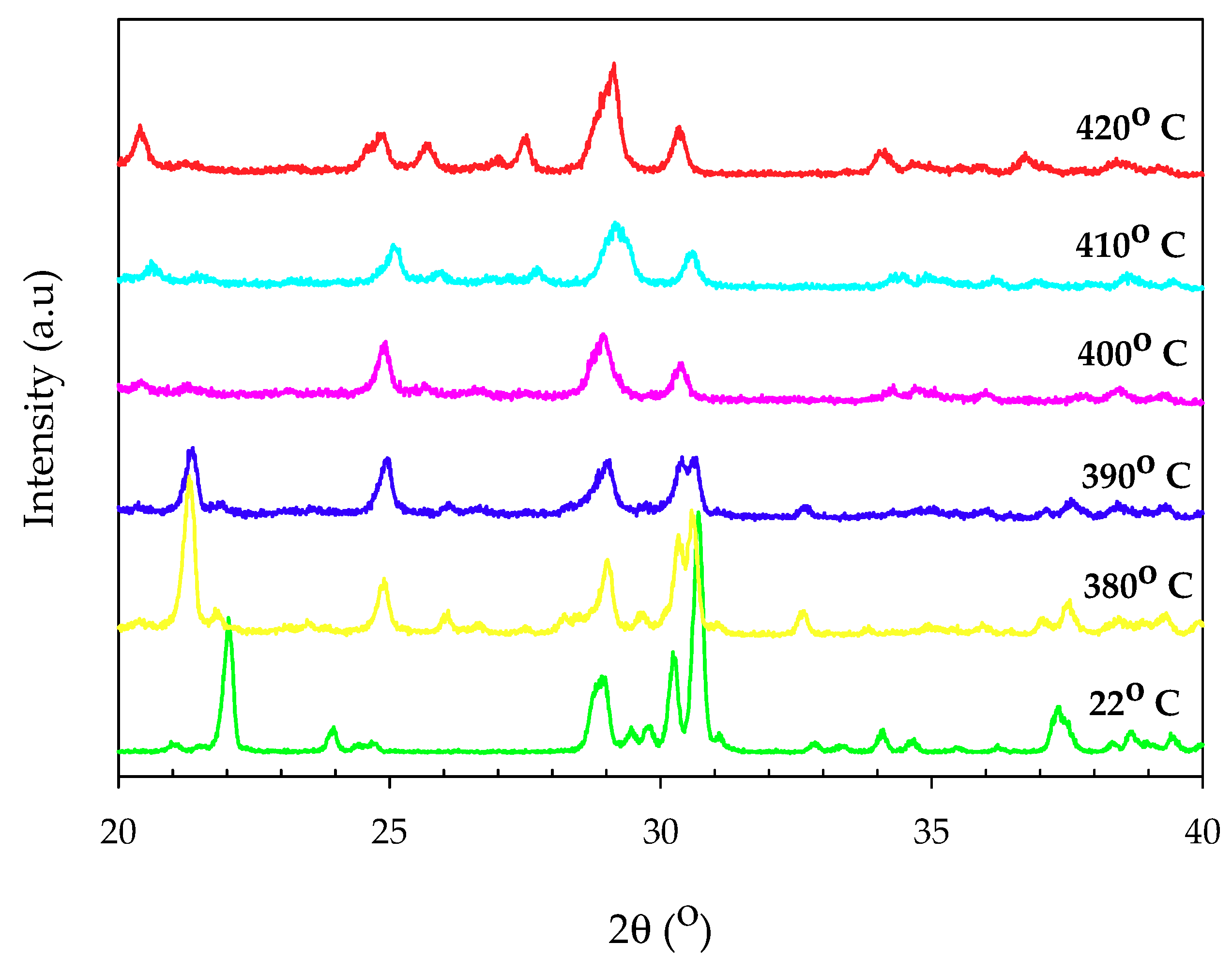
3.1.1. X-ray Diffraction Spectra
3.1.2. SEM and Spot-Chemical Analysis
3.2.3. FT-IR Spectra
3.2. Thermal Properties
3.3. UV-Vis Spectra
3.4. Thermochromism of Synthesized Pigment Powders
3.5. Manganese Violet-Based Irreversible Thermochromic Sensors
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Freddi, S.; Sironi, L.; D’Antuono, R.; Morone, D.; Dona, A.; Cabrini, E.; D’Alfonso, L.; Collini, M.; Pallavicini, P.; Baldi, G.; et al. A molecular thermometer for nanoparticles for optical hyperthermia. Nano Lett. 2013, 13, 2004–2010. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Li, Y.; Liu, T.; Zhang, G.; Liu, S. Intracellular cascade FRET for temperature imaging of living cells with polymeric ratiometric fluorescent thermometers. ACS Appl. Mater. Interfaces 2015, 7, 15551–15560. [Google Scholar] [CrossRef] [PubMed]
- Abram, C.; Fond, B.; Heyes, A.L.; Beyrau, F. High-speed planar thermometry and velocimetry using thermographic phosphor particles. Appl. Phys. B-Lasers Opt. 2013, 111, 155–160. [Google Scholar] [CrossRef]
- Cellini, F.; Peterson, S.D.; Porfiri, M. Flow velocity and temperature sensing using thermosensitive fluorescent polymer seed particles in water. Int. J. Smart Nano Mater. 2017, 8, 232–252. [Google Scholar] [CrossRef]
- Barbieri, M.; Cellini, F.; Cacciotti, I.; Peterson, S.D.; Porfiri, M. In situ temperature sensing with fluorescent chitosan-coated PNIPAAm/alginate beads. J. Mater. Sci. 2017, 52, 12506–12512. [Google Scholar] [CrossRef]
- Popescu, M.; Serban, L.; Popescu, M. Thermo-indicating paint for damage warning. J. Therm. Anal. 1996, 46, 317–321. [Google Scholar] [CrossRef]
- Lempereur, C.; Andral, R.; Prudhomme, J.Y. Surface temperature measurement on engine components by means of irreversible thermal coating. Meas. Sci. Technol. 2008, 19, 105501–105512. [Google Scholar] [CrossRef]
- Pelvich, C.W.; Foulk, D.L.; Polec, T.W. Method of Sensing High Surface Temperature in an Aircraft. Patent EP1959246 A2, 19 June 2008. [Google Scholar]
- Watson, H.M.L. An Irreversible Temperature Indicating Paint. Patent EP1614724A2, 11 January 2006. [Google Scholar]
- Efremov, A.M.; Mikhailov, M.D. Thermochromic Material. Patent EP1405890 B1, 17 October 2012. [Google Scholar]
- Lataste, E.; Demourgues, A.; Salmi, J.; Naporea, C.; Gaudon, M. Thermochromic behaviour (400 < T °C < 1200 °C) of barium carbonate/binary metal oxide mixtures. Dyes Pigm. 2011, 91, 396–403. [Google Scholar]
- Khiem, N.D.; Heesoo, L.; In-Tae, K. Synthesis and thermochromic properties of Cr-doped Al2O3 for a reversible thermochromic sensor. Materials 2017, 10, 476. [Google Scholar] [CrossRef]
- Seeboth, A. Thermochromic and Thermotropic Materials; CRC Press: Singapore, 2014. [Google Scholar]
- Begum, Y.; Wright, A.J. Relating highly distorted Jahn-Teller MnO6 to colouration in manganese violet pigments. J. Mater. Chem. 2012, 22, 21110–21116. [Google Scholar] [CrossRef]
- Lee, J.D.; Browne, L.S. The nature and properties of manganese violet. J. Chem. Soc. A 1968. [Google Scholar] [CrossRef]
- Chemberlain, J.R. Temperature Indicating Paint and Method of Preparing a Specimen with the Same. Patent GB2204874A, 23 November 1988. [Google Scholar]
- Anselmi, C.; Vagnini, M.; Cartechini, L.; Grazia, C.; Vivani, R.; Romani, A.; Rosi, F.; Sgamellotti, A.; Miliani, C. Molecular and structure characterization of some violet phosphate pigments for their non-invasive identification in modern paintings. Spectrochim. Acta A 2017, 173, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Brouzi, K.; Ennaciri, A.; Harcharras, M.; Capitelli, F. Structure and vibrational spectra of a new trihydrate diphosphate MnNH4NaP2O7·3H2O. J. Ram. Spectr. 2004, 35, 41–46. [Google Scholar] [CrossRef]
- Capitelli, F.; Khaoulaf, R.; Harcharras, M.; Ennaciri, A.; Habyby, S.H.; Valentini, V.; Mattei, G.; Bertolasi, V. Crystal structure and vibrational spectroscopy of the new acidic diphosphate (NH4)2Zn(H2P2O7)2·2H2O. Z. Kristallogr. 2005, 220, 25–30. [Google Scholar]
- Brouzi, K.; Ennaciri, A.; Capitelli, F.; Valentini, V.; Mattei, G.; Harcharas, M. Vibrational study of manganese ammonium dihydrogendiphosphate hydrated Mn0.5NH4H2P2O7·H2O. Phosphorus Sulfur Silicon 2005, 180, 545–553. [Google Scholar] [CrossRef]
- Jegatheesan, A.; Murugan, J.; Neelakantaprasad, B.; Rajarajan, G. FTIR, XRD, SEM, TGA investigations of ammonium dihydrogen phosphate (ADP) single crystal. Int. J. Comput. Appl. 2012, 53, 15–18. [Google Scholar] [CrossRef]
- Philip, D.B.; Lizbeth, B.; Aruldhas, G. IR and polarized raman spectra of Na4P2O7.10 H2O. J. Raman Spectrosc. 1990, 21, 523–524. [Google Scholar] [CrossRef]
- Harcharras, M.; Ennaciri, A.; Rulmont, A.; Gilbert, B. Vibrational spectra and structures of double diphosphates M2CdP2O7 (M = Li, Na, K, Rb, Cs). Spectrochim. Acta A 1997, 53, 345–352. [Google Scholar] [CrossRef]
- Sherman, D.M.; Vergo, N. Optical spectrum, site occupancy, and oxidation state of Mn in montmorillonite. Am. Mineral. 1988, 73, 140–144. [Google Scholar]







| Thermochromic Materials | Color Change | Transition Temperature (°C) |
|---|---|---|
| [NH2(C2H5)2]2CuCl4 | deep green ↔ yellow | 38 |
| Ag2(HgI4) | yellow ↔ orange | 50 |
| CuI | gray-tan → orange | 60–62 |
| Cu2(HgI4) | red ↔ brown | 70 |
| HgI2 | red ↔ yellow | 127 |
| 2Cu(CNS)2·2pyridine | green → yellow | 135 |
| yellow → black | 220 | |
| NH4VO3 | white → brown | 150 |
| brown → black | 170 | |
| CoCO2 | violet → black | 330 |
| MnNH4P2O7 | violet → white | 400 |
| NiC2O4 | light blue → black | 410 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, D.K.; Bach, Q.-V.; Lee, J.-H.; Kim, I.-T. Synthesis and Irreversible Thermochromic Sensor Applications of Manganese Violet. Materials 2018, 11, 1693. https://doi.org/10.3390/ma11091693
Nguyen DK, Bach Q-V, Lee J-H, Kim I-T. Synthesis and Irreversible Thermochromic Sensor Applications of Manganese Violet. Materials. 2018; 11(9):1693. https://doi.org/10.3390/ma11091693
Chicago/Turabian StyleNguyen, Duy Khiem, Quang-Vu Bach, Jong-Han Lee, and In-Tae Kim. 2018. "Synthesis and Irreversible Thermochromic Sensor Applications of Manganese Violet" Materials 11, no. 9: 1693. https://doi.org/10.3390/ma11091693
APA StyleNguyen, D. K., Bach, Q.-V., Lee, J.-H., & Kim, I.-T. (2018). Synthesis and Irreversible Thermochromic Sensor Applications of Manganese Violet. Materials, 11(9), 1693. https://doi.org/10.3390/ma11091693





