Effect of Magnetized Water on the Mechanical and Durability Properties of Concrete Block Pavers
Abstract
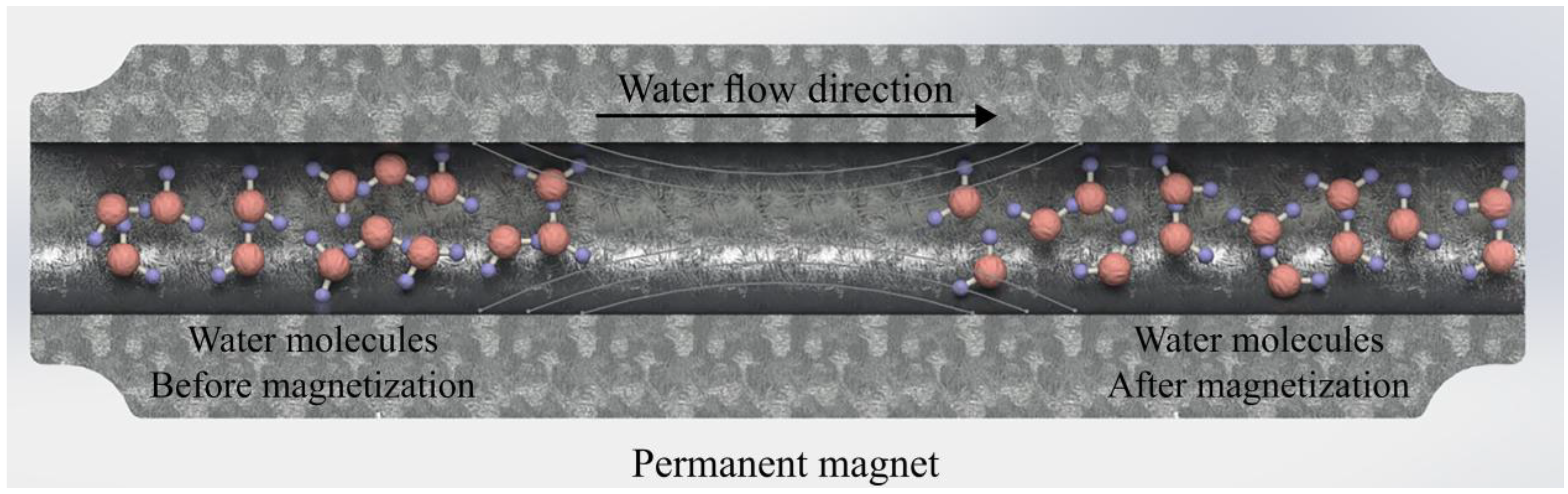
:1. Introduction
2. Materials and Sample Preparation
2.1. Materials
2.2. Experimental Design
2.3. Specimen Preparation
2.4. Test Methods
2.4.1. Compressive Strength
2.4.2. Splitting Tensile Strength
2.4.3. Flexural Strength
2.4.4. Mass Loss
2.4.5. Water Absorption
3. Results and Discussion
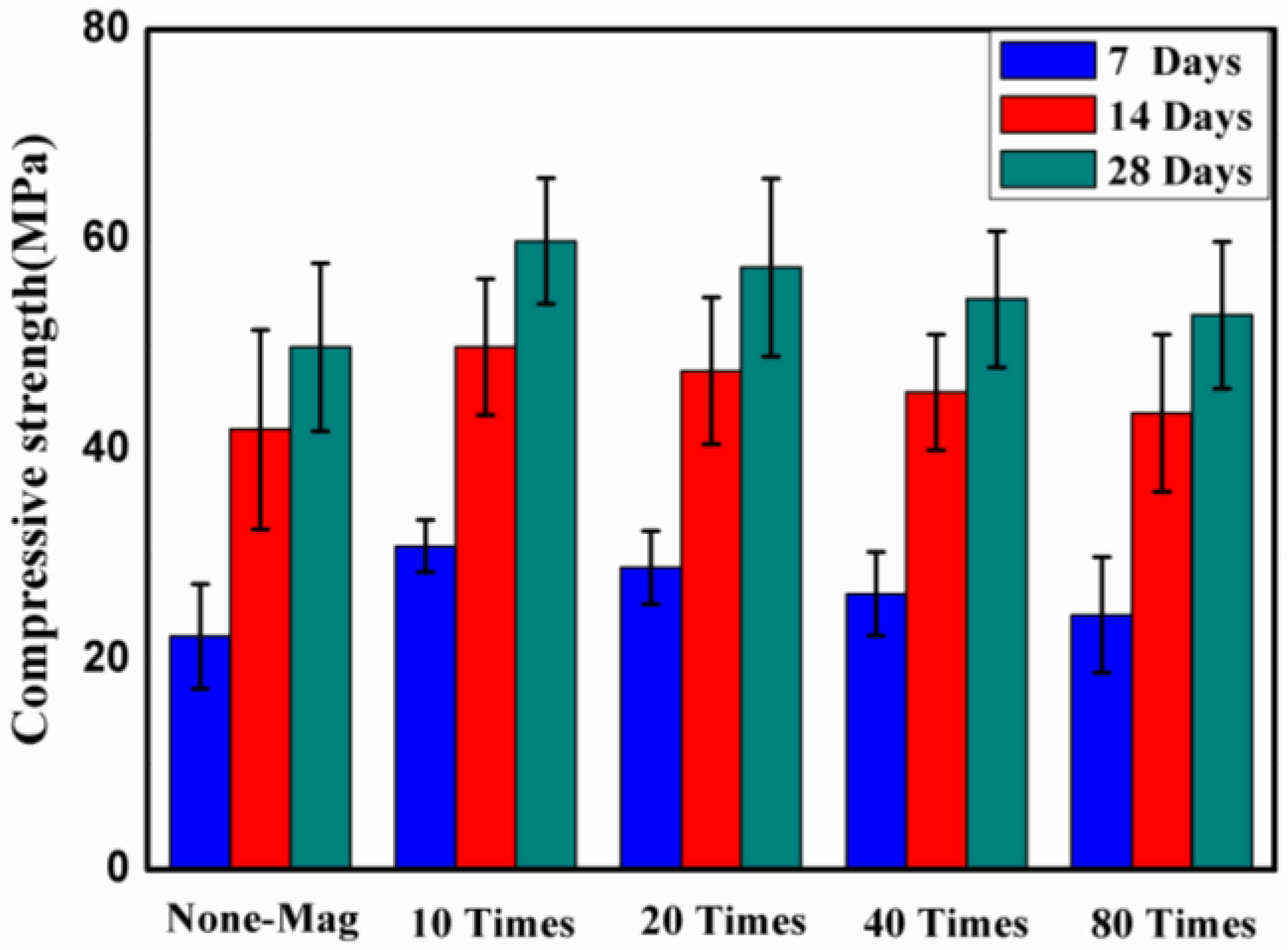
3.1. Effect of Magnetized Water on Compressive Strength
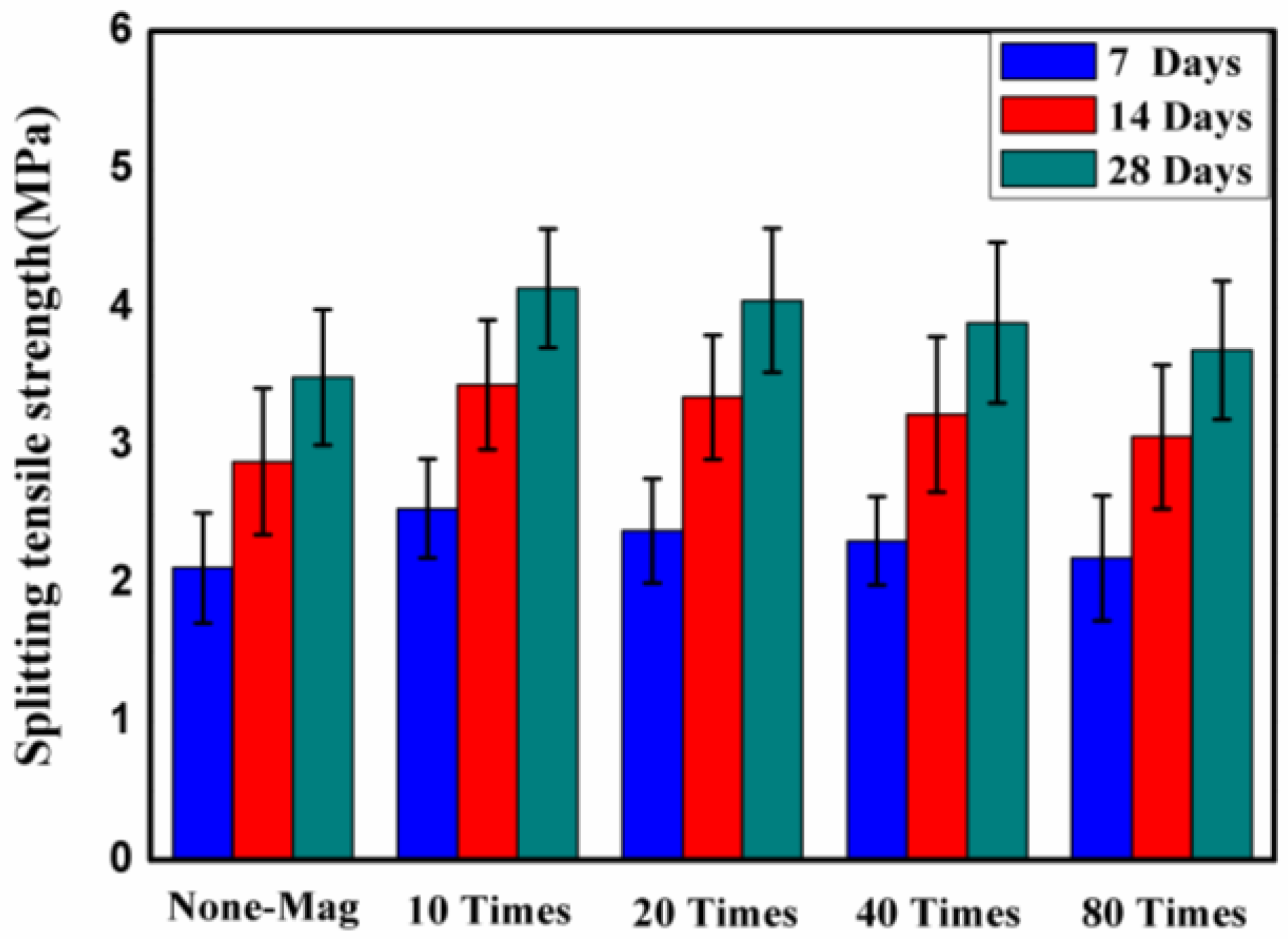
3.2. Effect of Magnetized Water on Splitting Tensile Strength
3.3. Effect of Magnetized Water on Flexural Strength
3.4. Effects of Sulfuric Acid Immersion
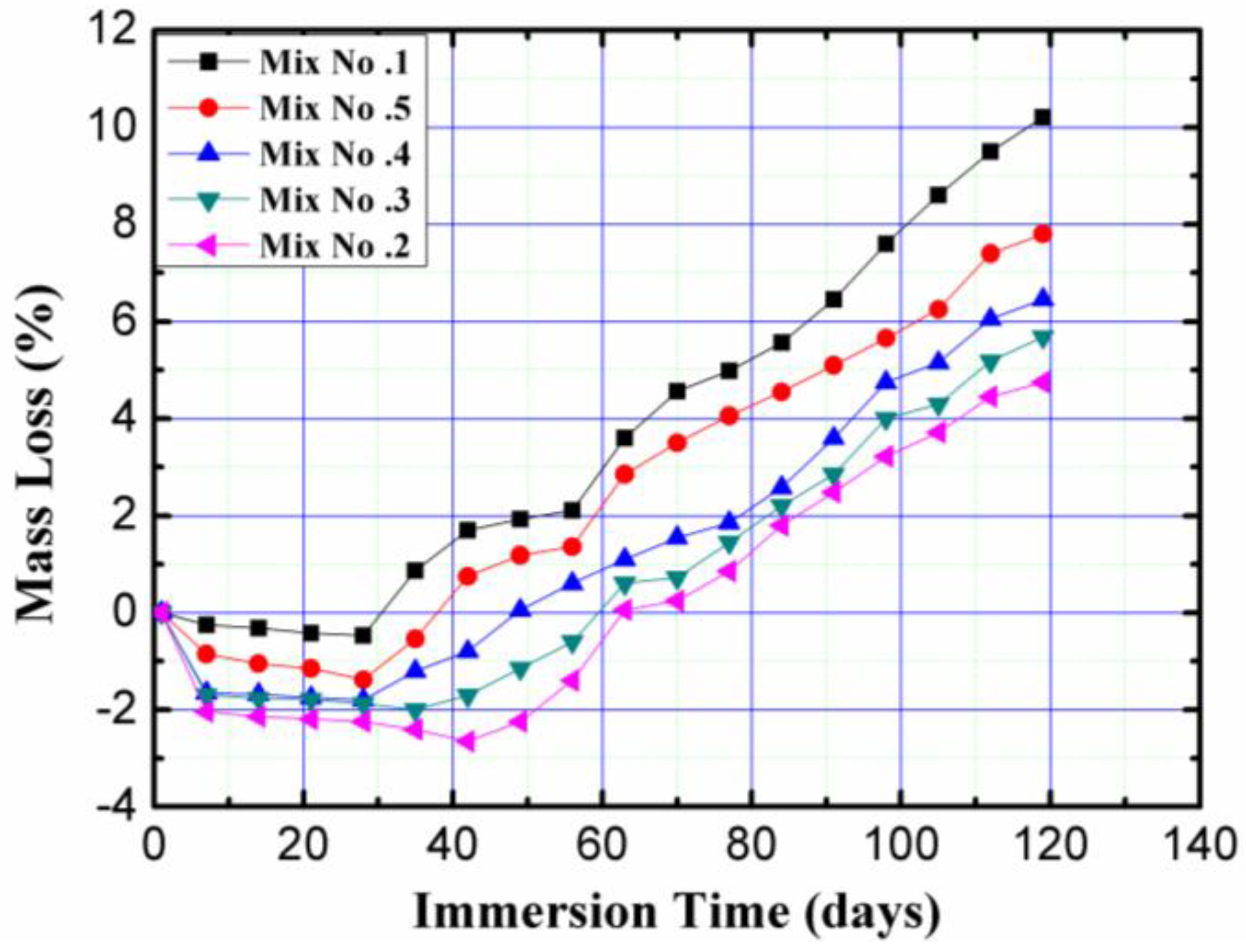
3.4.1. Mass Loss
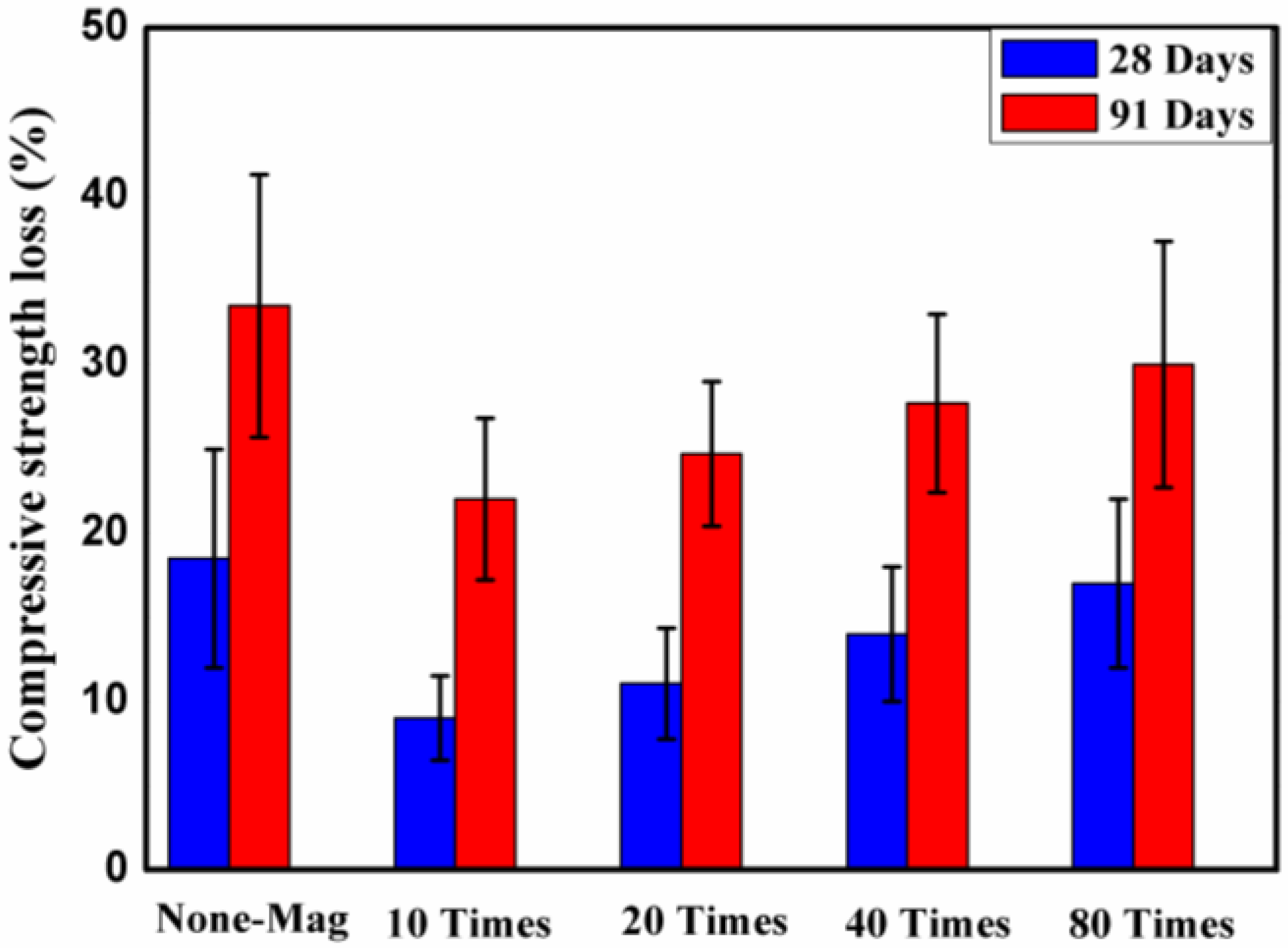
3.4.2. Compressive Strength Loss
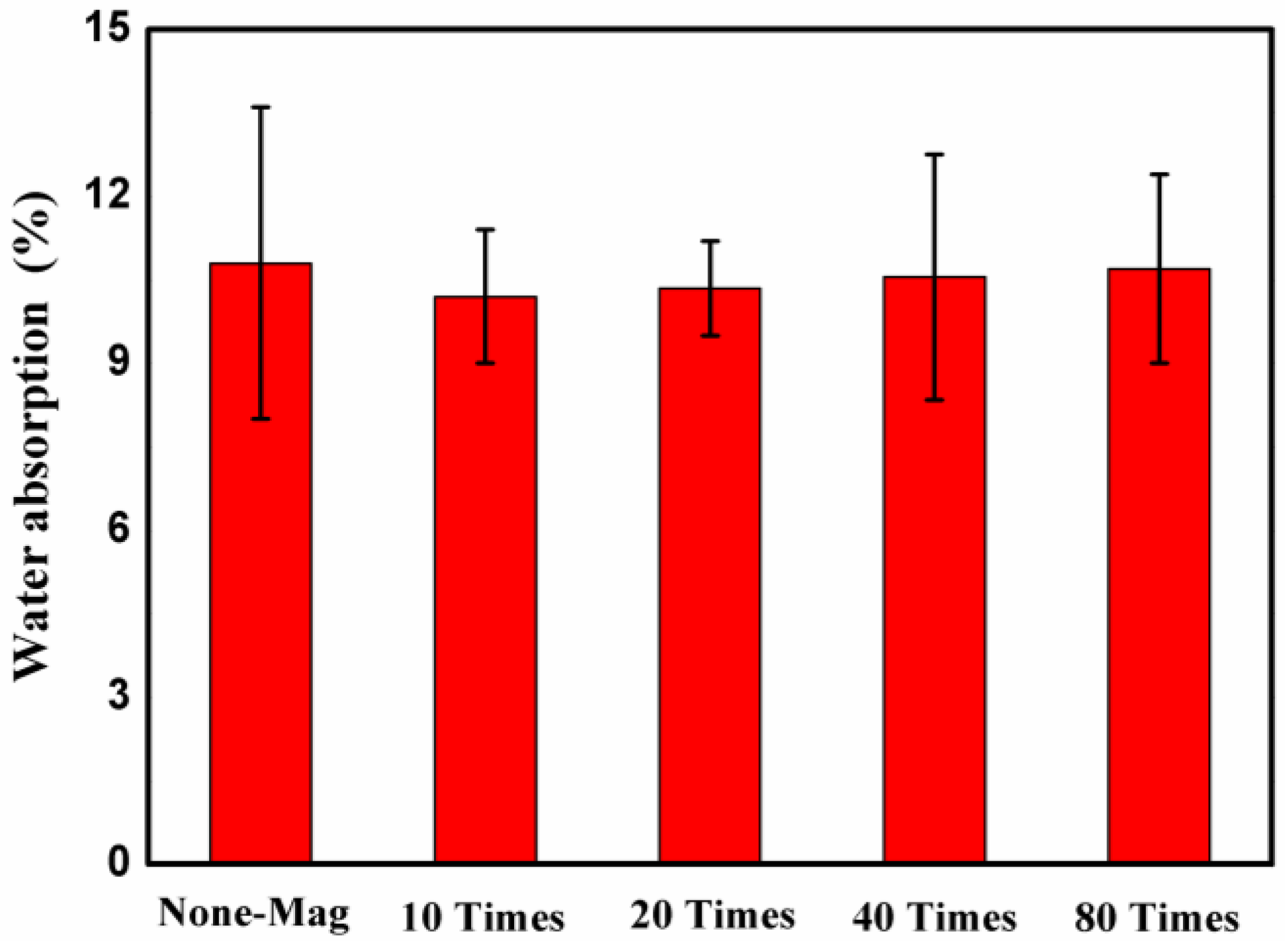
3.5. Effect of Magnetized Water on Water Absorption
3.6. Effect of Magnetized Water on Microstructure of Concrete
4. Conclusions
- The mechanical performance of concrete showed an improvement due to using magnetized water instead of regular tap water: relative to the control mix, an average improvement of 12.5%, 13%, and 9% after 28 days of water curing was registered for the compressive strength, splitting tensile strength, and flexural strength, respectively;
- The results showed that as the curing age increases, the compressive strength, splitting tensile strength, and flexural strength of all of the mixes increases, as expected. However, the rate of increase varies for different mixes;
- The mass and compressive strength loss and water absorption results showed that magnetized water had a positive effect on the resistance to sulfuric attack and water absorption of the concrete mixes. The improvement grew as the number of times that water passed through the permanent magnetic field decreased;
- For the same mix proportions, concrete mixes with magnetized water will have a higher compressive strength, splitting tensile strength, and flexural strength, and a lower mass/compressive strength loss under acid attack and water absorption than control mix specimens, due to their greater density and more efficient degree of cement hydration;
- The SEM images showed that using magnetized water instead of regular tap water led to a significant improvement of the microstructure of the corresponding concrete mixes and resulted in a denser structure compared to the control mix;
- The cost of magnetizing water is very low because of the simple devices used. In this study, the following devices were used, with a total a cost of approximately ($600 USD): (a) an electric pump, (b) two water tanks, and (c) one permanent magnetic field. The cost would have to be adapted to the scale of the work involved. The time needed to pass 10 L of regular tap water through the permanent magnet in this study was about 28 s. This time would decrease as the strength of the electric pump increased.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Soutsos, M.N.; Tang, K.; Khalid, H.A.; Millard, S.G. The effect of construction pattern and unit interlock on the structural behaviour of block pavements. Constr. Build. Mater. 2011, 25, 3832–3840. [Google Scholar] [CrossRef]
- Holt, E.; Raivio, P. Use of gasification residues in compacted concrete paving blocks. Cem. Concr. Res. 2006, 36, 441–448. [Google Scholar] [CrossRef]
- Gencel, O.; Ozel, C.; Koksal, F.; Erdogmus, E.; Martínez-Barrera, G.; Brostow, W. Properties of concrete paving blocks made with waste marble. J. Clean. Prod. 2012, 21, 62–70. [Google Scholar] [CrossRef]
- Algin, H.M. The Behaviour of Flexibly Bedded Concrete Paver Pavements. Ph.D. Thesis, Newcastle University, Newcastle upon Tyne, UK, 1996. [Google Scholar]
- Navya, G.; Rao, J.V. Experimental investigation on properties concrete paver block with the inclusion of natural fibers. Int. J. Eng. Res. Appl. 2014, 8, 34–38. [Google Scholar]
- Wei, H.; Wang, Y.; Luo, J. Influence of magnetic water on early-age shrinkage cracking of concrete. Constr. Build. Mater. 2017, 147, 91–100. [Google Scholar] [CrossRef]
- Saleh, F.; Rivera, R.; Salehi, S.; Teodoriu, C.; Ghalambor, A. How does mixing water quality affect cement properties? In Proceedings of the SPE International Conference and Exhibition on Formation Damage Control, Lafayette, LA, USA, 7–9 February 2018; Society of Petroleum Engineers: Richardson, TX, USA, 2018. [Google Scholar]
- Esfahani, A.R.; Reisi, M.; Mohr, B. Magnetized water effect on compressive strength and dosage of superplasticizers and water in self-compacting concrete. J. Mater. Civ. Eng. 2018, 30, 04018008. [Google Scholar] [CrossRef]
- Hover, K.C. The influence of water on the performance of concrete. Constr. Build. Mater. 2011, 25, 3003–3013. [Google Scholar] [CrossRef]
- Esmaeilnezhad, E.; Choi, H.J.; Schaffie, M.; Gholizadeh, M.; Ranjbar, M. Characteristics and applications of magnetized water as a green technology. J. Clean. Prod. 2017, 161, 908–921. [Google Scholar] [CrossRef]
- Toledo, E.J.L.; Ramalho, T.C.; Magriotis, Z.M. Influence of magnetic field on physical–chemical properties of the liquid water: Insights from experimental and theoretical models. J. Mol. Struct. 2008, 888, 409–415. [Google Scholar] [CrossRef]
- Zhou, K.; Lu, G.; Zhou, Q.; Song, J.; Jiang, S.; Xia, H. Monte carlo simulation of liquid water in a magnetic field. J. Appl. Phys. 2000, 88, 1802–1805. [Google Scholar] [CrossRef]
- Inaba, H.; Saitou, T.; Tozaki, K.-I.; Hayashi, H. Effect of the magnetic field on the melting transition of H2O and D2O measured by a high resolution and supersensitive differential scanning calorimeter. J. Appl. Phys. 2004, 96, 6127–6132. [Google Scholar] [CrossRef]
- Cai, R.; Yang, H.; He, J.; Zhu, W. The effects of magnetic fields on water molecular hydrogen bonds. J. Mol. Struct. 2009, 938, 15–19. [Google Scholar] [CrossRef]
- Ahmed, H.I. Behavior of magnetic concrete incorporated with Egyptian nano alumina. Constr. Build. Mater. 2017, 150, 404–408. [Google Scholar] [CrossRef]
- Su, N.; Wu, Y.H.; Mar, C.Y. Effect of magnetic water on the engineering properties of concrete containing granulated blast-furnace slag. Cem. Concr. Res. 2000, 30, 599–605. [Google Scholar] [CrossRef]
- Gholhaki, M.; Kheyroddin, A.; Hajforoush, M.; Kazemi, M. An investigation on the fresh and hardened properties of self-compacting concrete incorporating magnetic water with various pozzolanic materials. Constr. Build. Mater. 2018, 158, 173–180. [Google Scholar] [CrossRef]
- Su, N.; Wu, C.F. Effect of magnetic field treated water on mortar and concrete containing fly ash. Cem. Concr. Compos. 2003, 25, 681–688. [Google Scholar] [CrossRef]
- Faris, A.S.; Al-Mahaidi, R.; Jadooe, A. Implementation of magnetized water to improve the properties of concrete. Int. J. Civ. Eng. Technol. 2014, 5, 43–57. [Google Scholar]
- Colic, M.; Morse, D. The elusive mechanism of the magnetic ‘memory’of water. Colloid Surf. A 1999, 154, 167–174. [Google Scholar] [CrossRef]
- Bharath, S.; Subraja, S.; Kumar, P.A. Influence of magnetized water on concrete by replacing cement partially with copper slag. J. Chem. Pharm. Sci. 2016, 9, 2791. [Google Scholar]
- Ghods, A. A survey on the mechanical properties of magnetic self-compacting concrete containing nanosilica. Int. Res. J. Appl. Basic Sci. 2014, 8, 413–418. [Google Scholar]
- Patil, S.V.; Pathak, N.J. The experimental study on compressive strength of concrete using AR glass fibers and partial replacement of cement with GGBS with effect of magnetic water. Int. J. Eng. Technol. Manag. Appl. Sci. 2016, 4, 21. [Google Scholar]
- Reddy, B.S.K.; Ghorpade, V.G.; Rao, H.S. Influence of magnetic water on strength properties of concrete. Indian J. Sci. Technol. 2014, 7, 14–18. [Google Scholar]
- Chau, Z.J. The New Construction Method of Concrete; The Publishing House of Chinese Architectural Industry: Beijing, China, 1996. (In Chinese) [Google Scholar]
- Fu, W.; Wang, Z.B. The New Technology of Concrete Engineering; The Publishing House of Chinese Architectural Industry: Beijing, China, 1994. (In Chinese) [Google Scholar]
- Afshin, H.; Gholizadeh, M.; Khorshidi, N. Improving mechanical properties of high strength concrete by magnetic water technology. Sci. Iran. Trans. A Civ. Eng. 2010, 17, 74–79. [Google Scholar]
- Araghi, H.J.; Nikbin, I.M.; Reskati, S.R.; Rahmani, E.; Allahyari, H. An experimental investigation on the erosion resistance of concrete containing various pet particles percentages against sulfuric acid attack. Constr. Build. Mater. 2015, 77, 461–471. [Google Scholar] [CrossRef]
- Nambiar, E.K.; Ramamurthy, K. Sorption characteristics of foam concrete. Cem. Concr. Res. 2007, 37, 1341–1347. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, B.; Gong, Z.; Gao, K.; Ou, Y.; Zhang, J. The effect of a static magnetic field on the hydrogen bonding in water using frictional experiments. J. Mol. Struct. 2013, 1052, 102–104. [Google Scholar] [CrossRef]
- Jeffrey, G.A. An Introduction to Hydrogen Bonding; Oxford University Press: New York, NY, USA, 1997. [Google Scholar]
- Li, R.; Jiang, Z.; Yang, H.; Guan, Y. Effects of ions in natural water on the 17O NMR chemical shift of water and their relationship to water cluster. J. Mol. Liq. 2006, 126, 14–18. [Google Scholar] [CrossRef]
- Mohseni, E.; Tang, W.; Cui, H. Chloride diffusion and acid resistance of concrete containing zeolite and tuff as partial replacements of cement and sand. Materials 2017, 10, 372. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.J.; Leung, C.; Xi, Y.P. Structural Renovation in Concrete; CRC Press: London, UK, 2009. [Google Scholar]
- Siad, H.; Lachemi, M.; Sahmaran, M.; Hossain, K.M.A. Effect of glass powder on sulfuric acid resistance of cementitious materials. Constr. Build. Mater. 2016, 113, 163–173. [Google Scholar] [CrossRef]
- Allahverdi, A.; ŠKVÁRA, F. Acidic corrosion of hydrated cement based materials. Ceram.−Silikaty 2000, 44, 152–160. [Google Scholar]
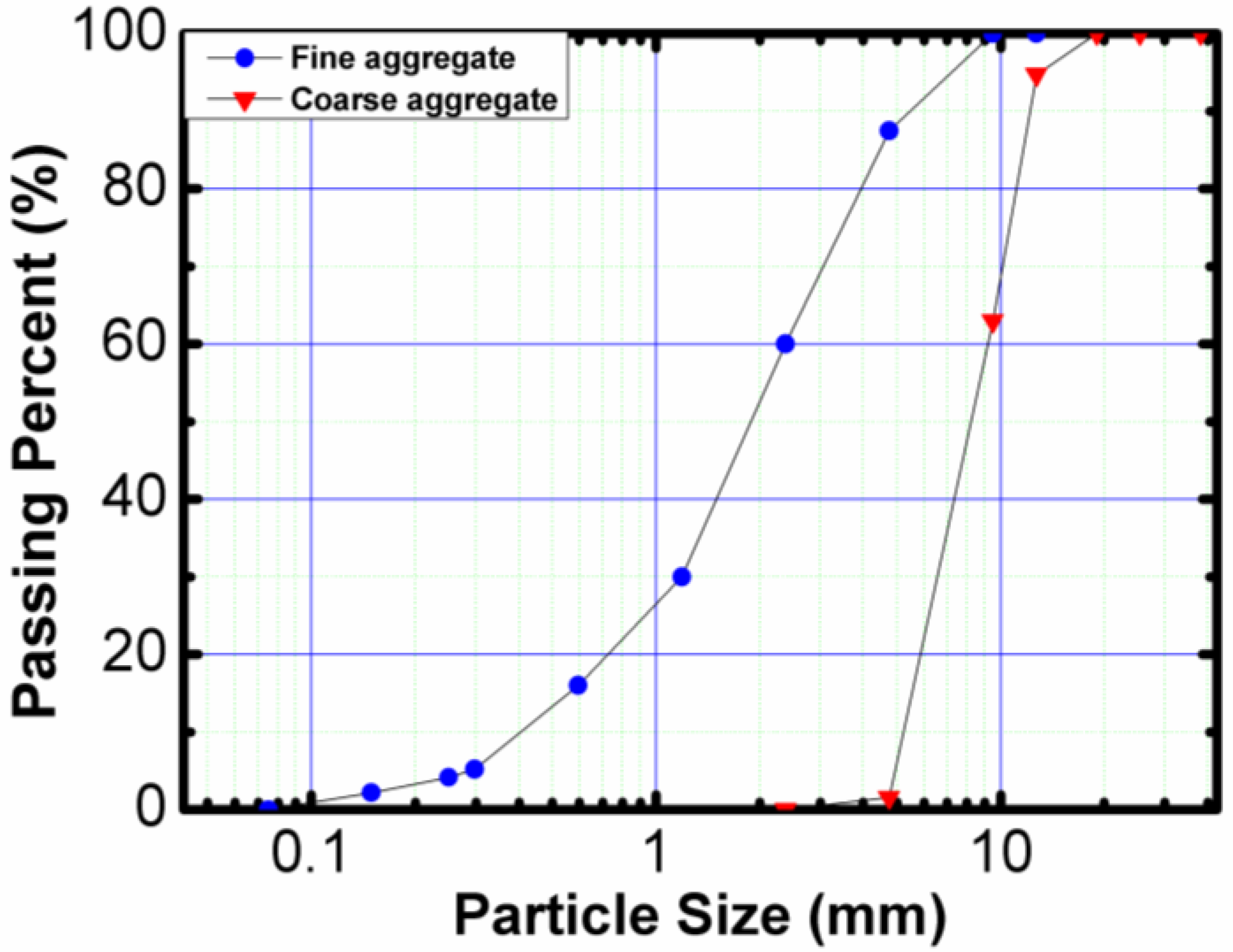





| Material | Chemical Composition (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | CaO | Al2O3 | Fe2O3 | MgO | SO3 | K2O | Na2O | CL * | LOI ** | |
| Cement (Type II) | 21.65 | 63.25 | 4.3 | 3.45 | 2.8 | 2.05 | 0.6 | 0.5 | 0.07 | 1.35 |
| Properties | Fine Aggregates | Coarse Aggregates |
|---|---|---|
| Water absorption (%) | 4.15 | 1.63 |
| Existent moisture (%) | 3.8 | 0.51 |
| Modulus of fineness | - | 4.79 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghorbani, S.; Gholizadeh, M.; De Brito, J. Effect of Magnetized Water on the Mechanical and Durability Properties of Concrete Block Pavers. Materials 2018, 11, 1647. https://doi.org/10.3390/ma11091647
Ghorbani S, Gholizadeh M, De Brito J. Effect of Magnetized Water on the Mechanical and Durability Properties of Concrete Block Pavers. Materials. 2018; 11(9):1647. https://doi.org/10.3390/ma11091647
Chicago/Turabian StyleGhorbani, Saeid, Mostafa Gholizadeh, and Jorge De Brito. 2018. "Effect of Magnetized Water on the Mechanical and Durability Properties of Concrete Block Pavers" Materials 11, no. 9: 1647. https://doi.org/10.3390/ma11091647
APA StyleGhorbani, S., Gholizadeh, M., & De Brito, J. (2018). Effect of Magnetized Water on the Mechanical and Durability Properties of Concrete Block Pavers. Materials, 11(9), 1647. https://doi.org/10.3390/ma11091647






