Effect of Thermal Vacancy on Thermodynamic Behaviors in BCC W Close to Melting Point: A Thermodynamic Study
Abstract
:1. Introduction
2. Thermodynamic Model for bcc W with Thermal Vacancy
2.1. Expression for GW
2.2. Expression for Gva
2.3. Expression for Interaction Parameter Ω
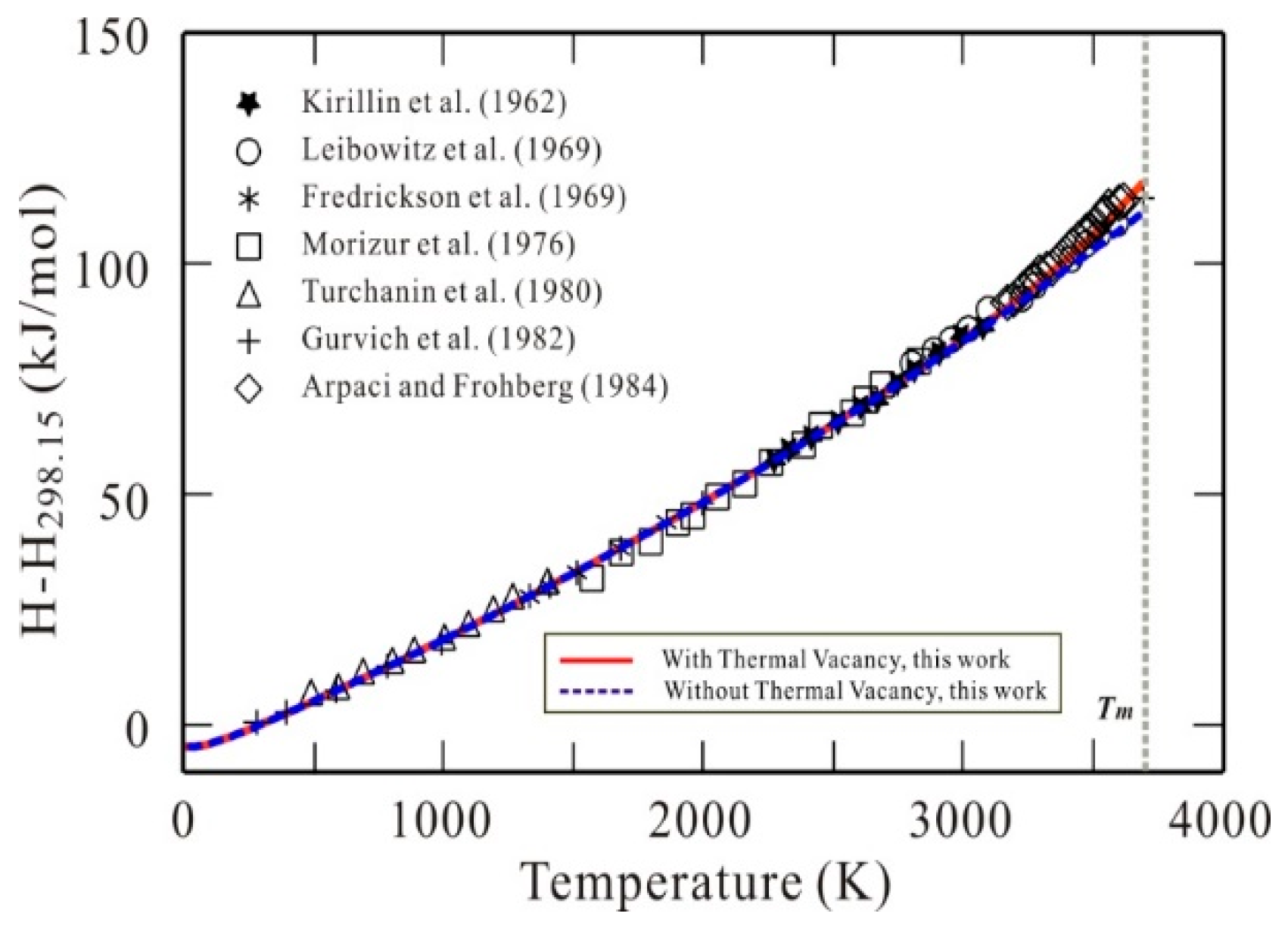
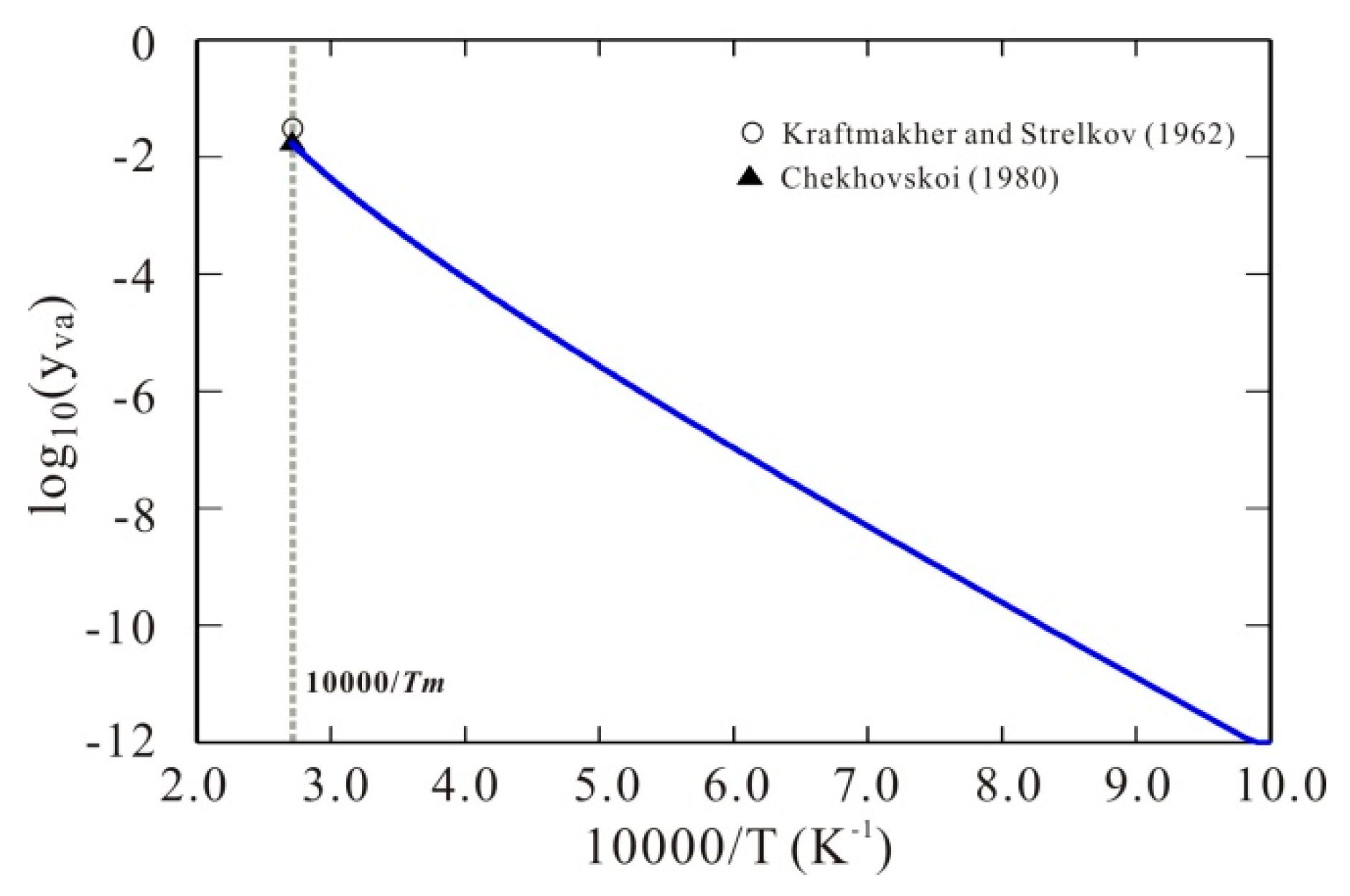
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ven, A.V.; Yu, H.C.; Ceder, G.; Thornton, K. Vacancy mediated substitutional diffusion in binary crystalline solids. Prog. Mater. Sci. 2010, 55, 61–105. [Google Scholar]
- Protik, N.H.; Carrete, J.; Katcho, N.A.; Mingo, N.; Broido, D. Ab initio study of the effect of vacancies on the thermal conductivity of boron arsenide. Phys. Rev. B Condens. Matter 2016, 94, 045207. [Google Scholar] [CrossRef]
- Kraftmakher, Y. Equilibrium vacancies and thermophysical properties of metals. Phys. Rep. 1998, 299, 79–188. [Google Scholar] [CrossRef]
- Causey, R.A.; Venhaus, T.J. The use of tungsten in fusion reactors: A review of the hydrogen retention and migration properties. Phys. Scr. 2001, T94, 9–15. [Google Scholar] [CrossRef]
- Abernethy, R.G. Predicting the performance of tungsten in a fusion environment: A literature review. Mater. Sci. Technol. 2017, 33, 388–399. [Google Scholar] [CrossRef]
- Chekhovskoi, V.Y. The enthalpy and heat capacity of tungsten in the temperature range 400-3500 K. Teplofiz. Vys. Temp. 1980, 18, 1191. [Google Scholar]
- Kraftmakher, Y.A.; Strelkov, P.G. Energy of formation and concentration of vacancies in tungsten. Phys. Solid State 1962, 4, 1662–1664. [Google Scholar]
- Worthing, A.G. Atomic heats of tungsten and of carbon at incandescent temperatures. Phys. Rev. 1918, 12, 199–225. [Google Scholar] [CrossRef]
- Jaeger, F.M.; Rosenbohm, E. The exact measurement of the specific heats of solid substances at high temperatures. III. The specific heats of palladium and of tungsten. Konicki. Ned. Akad. Wetensch. Proc. Ser. B 1930, 33, 457–472. [Google Scholar]
- Hoch, M.; Johnston, H.L. A high temperature drop calorimeter: The heat capacities of tantalum and tungsten between 1000 and 3000 K. J. Chem. Phys. 1961, 65, 855–860. [Google Scholar] [CrossRef]
- Kirillin, V.A.; Sheindlin, A.E.; Chekhovskoi, V.Y.; Petrov, V.A. Thermodynamic properties of tungsten. Russ. J. Phys. Chem. 1961, 37, 1212. [Google Scholar]
- Lowenthal, G.C. The specific heat of metals between 1200 K and 2400 K. Aust. J. Phys. 1963, 16, 47–67. [Google Scholar]
- Hein, R.A.; Flagella, P.N. Enthalpy Measurements of UO2 and Tungsten to 3260 K; GE (General Electric) Report GEMP-578; General Electric: Boston, MA, USA, February 1968. [Google Scholar]
- Leibowitz, L.; Chasanov, M.G.; Mishler, L.W. The enthalpy of solid tungsten from 2800 K to its melting point. Trans. Metall. AIME 1969, 245, 981–984. [Google Scholar]
- Yakunkin, M.M. Specific-heat of tungsten by periodic pulsed heating. High Temp. 1983, 21, 848–853. [Google Scholar]
- Senchenko, V.N.; Sheindlin, M.A. Experimental investigation of the caloric properties of tungsten and graphite near their melting-points. High Temp. 1987, 25, 364–368. [Google Scholar]
- Righini, F.; Spisiak, J.; Bussolino, G.C.; Rosso, A.; Haidar, J. Measurement of thermophysical properties by a pulse-heating method: Thoriated tungsten in the range 1200 to 3600 K. Int. J. Thermophys. 1994, 15, 1311–1322. [Google Scholar] [CrossRef]
- Cezairliyan, A.; McClure, J.L. High-speed electrical (subsecond) measurement of heat capacity, resistivity, and thermal radiation properties of tungsten in the range 2000 to 3600 K. J. Res. Nat. Stand. Sec. A 1971, 75, 283–290. [Google Scholar] [CrossRef]
- Magnus, A.; Holzmann, H. Untersuchungen über die Spezifischen Wärme von Tantal, Wolfram und Beryllium zwischen 100 und 900 °C. Ann. Phys. 1929, 3, 585–612. [Google Scholar] [CrossRef]
- Jaeger, F.M.; Rosenbohm, E. La détermination exacte des chaleurs spécifiques vraies du tungstèn, du rhodium, du palladium, du ruthénium, delosmiumet de l’osmium et l’iridium à des temperatures entre 0 et 1625 °C. Recl. Trav. Chim. Pays-Bas 1932, 51, 1–46. [Google Scholar] [CrossRef]
- Waiters, T.R.; Craig, R.S.; Wallace, W.E. Heat Capacity of Tungsten between 4 and 15 K. Phys. Rev. 1956, 104, 1240–1241. [Google Scholar]
- DeSorbo, W. Low temperature heat capacity of bismuth and tungsten. J. Phys. Chem. 1958, 62, 965–967. [Google Scholar] [CrossRef]
- Clusius, K.; Franzosini, P. Ergebnisse der Tieftemperaturforschung-XXIII Atom- und Elektronenwärme des Molybdäns und Wolframs zwischen 10 °K und 273 °K. Z. Naturforsch. Teil A 1959, 149, 99–105. [Google Scholar]
- Williams, M.E. Ab Initio Elastic and Thermodynamic Properties of High-Temperature Cubic Intermetallics at Finite. Master Thesis, Texas A&M University, College Station, TX, USA, May 2008. [Google Scholar]
- Mundy, J.N.; Rothman, S.J.; Lam, N.Q.; Hoff, H.A.; Nowicki, L.J. Self-diffusion in tungsten. Phys. Rev. B Condens. Matter 1978, 18, 6566. [Google Scholar] [CrossRef]
- Koning, M.; Debiaggi, S.R.; Monti, A.M. Vacancy-formation thermodynamics in aluminium and nickel: A computational study. Defect Diffus. Forum 2004, 224, 59–74. [Google Scholar] [CrossRef]
- Glensk, A.; Grabowski, B.; Hickel, T.; Neugebauer, J. Breakdown of the Arrhenius law in describing vacancy formation energies: The importance of local anharmonicity revealed by Ab initio thermodynamics. Phys. Rev. X 2014, 4, 011018. [Google Scholar]
- Franke, P. Modeling of thermal vacancies in metals within the framework of the compound energy formalism. J. Phase Equilib. Diffus. 2014, 35, 780–787. [Google Scholar] [CrossRef]
- Hillert, M. The compound energy formalism. J. Alloys Compd. 2001, 35, 161–176. [Google Scholar] [CrossRef]
- Chase, M.W.; Ansara, I.; Dinsdale, A.; Eriksson, G.; Grimvall, G.; Hoglund, L.; Yokokawa, H. Thermodynamic models and data for pure elements and others endmembers of solutions. Calphad 1995, 19, 437–447. [Google Scholar]
- Born, M.; Huang, K. Dynamical Theory of Crystal Lattices; Oxford University Press: Oxford, UK, 1954. [Google Scholar]
- Grimvall, G. Thermophysical Properties of Materials; Elsevier Science: Amsterdam, The Netherland, 1986. [Google Scholar]
- Dinsdale, A.T.; Khvan, A.V.; Watson, A. Critical assessment 5: Thermodynamic data for vacancies. Mater. Sci. Technol. 2014, 30, 1715–1718. [Google Scholar] [CrossRef]
- Kirillin, V.A.; Sheindlin, A.E.; Chekhovskoi, V.Y. Enthalpy and specific heat of tungsten in the 0−2400 °C temperature range. Dokl. Akad. Nauk SSSR 1962, 142, 1323–1326. [Google Scholar]
- Fredrickson, D.R.; Kleb, R.; Nuttall, R.L.; Hubbard, W.N. A drop calorimeter with an electron beam heated furnace. Rev. Sci. Instrum. 1969, 40, 1022–1025. [Google Scholar] [CrossRef]
- Morizur, G.; Radenac, A.; Cretenet, J.C. Drop calorimetry at 3000 K. High Temp. High Press. 1976, 8, 113–120. [Google Scholar]
- Turchanin, A.G.; Babenko, S.A.; Ekimov, S.E. Device for exact determination of solids enthalpy at high temperatures. Tungsten enthalpy in 400–1400 K range. Teplofizika Vysokikh Temperatur 1980, 18, 995–1001. [Google Scholar]
- Gurvich, L.V.; Veits, I.V.; Medvedev, V.A. Calculations of Thermodynamic Properties; Nauka: Moscow, 1982. [Google Scholar]
- Arpaci, E.; Frohberg, M.G. Enthalpy measurements on solid and liquid tungsten by levitation calorimetry. Z. Metallkd. 1984, 75, 614–618. [Google Scholar]
- Walford, L.K. The X-ray debye temperature of tungsten. Mater. Res. Bull. 1969, 4, 137–142. [Google Scholar] [CrossRef]
- Cacciamani, G.; Chang, Y.A.; Grimvall, G.; Franke, P.; Kaufman, L.; Miodownik, P.; Sanchez, J.M.; Schalin, M.; Sigli, C. Thermodynamic modelling of solutions and alloys. Group 3: Order-disorder phase diagrams. Calphad 1997, 21, 219–246. [Google Scholar]
- Wang, Y.; Curtarolo, S.; Jiang, C.; Arroyave, R.; Wang, T.; Cederc, G.; Chen, L.; Liu, Z. Ab initio lattice stability in comparison with CALPHAD lattice stability. Calphad 2004, 28, 79–90. [Google Scholar] [CrossRef]

| Parameters | Values (Gibbs Energy in J/mol-atom; T in Kelvin) |
|---|---|
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, Y.; Zhang, L. Effect of Thermal Vacancy on Thermodynamic Behaviors in BCC W Close to Melting Point: A Thermodynamic Study. Materials 2018, 11, 1648. https://doi.org/10.3390/ma11091648
Tang Y, Zhang L. Effect of Thermal Vacancy on Thermodynamic Behaviors in BCC W Close to Melting Point: A Thermodynamic Study. Materials. 2018; 11(9):1648. https://doi.org/10.3390/ma11091648
Chicago/Turabian StyleTang, Ying, and Lijun Zhang. 2018. "Effect of Thermal Vacancy on Thermodynamic Behaviors in BCC W Close to Melting Point: A Thermodynamic Study" Materials 11, no. 9: 1648. https://doi.org/10.3390/ma11091648
APA StyleTang, Y., & Zhang, L. (2018). Effect of Thermal Vacancy on Thermodynamic Behaviors in BCC W Close to Melting Point: A Thermodynamic Study. Materials, 11(9), 1648. https://doi.org/10.3390/ma11091648






