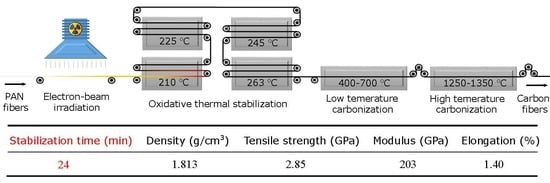
Rapid and Continuous Preparation of Polyacrylonitrile-Based Carbon Fibers with Electron-Beam Irradiation Pretreatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
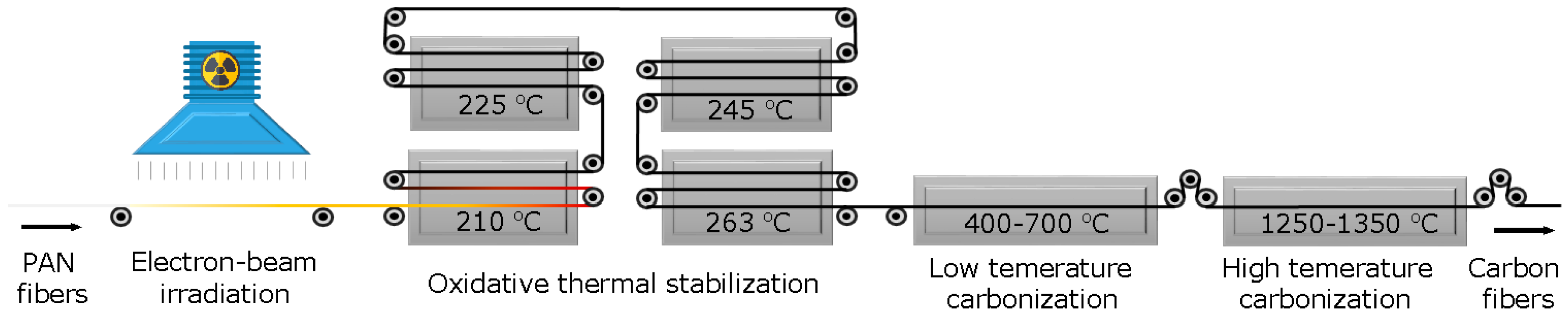
2.2. Electron Beam Irradiation of PAN Fibers
2.3. Stabilization and Carbonization
2.4. Characterizations
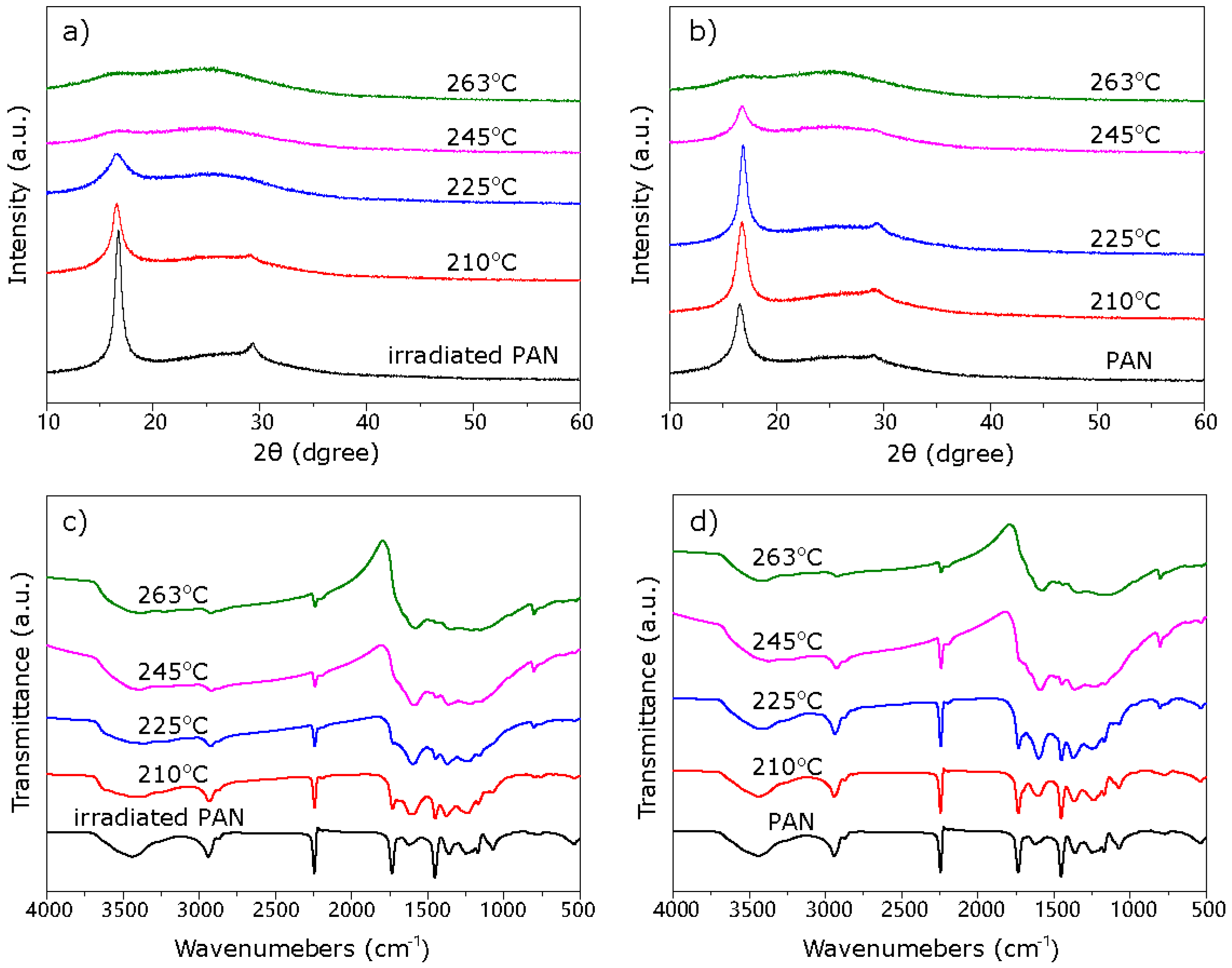
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Frank, E.; Steudle, L.M.; Ingildeev, D.; Spörl, J.M.; Buchmeiser, M.R. Carbon fibers: Precursor systems, processing, structure, and properties. Angew. Chem. Int. Ed. 2014, 53, 5262–5298. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.; Liu, Y.; Kumar, S. Recent progress in fabrication, structure, and properties of carbon fibers. Polym. Rev. 2012, 34, 234–258. [Google Scholar] [CrossRef]
- Salim, N.V.; Blight, S.; Creighton, C.; Nunna, S.; Atkiss, S.; Razal, J.M. The role of tension and temperature for efficient carbonization of polyacrylonitrile fibers: Toward low cost carbon fibers. Ind. Eng. Chem. Res. 2018, 57, 4268–4276. [Google Scholar] [CrossRef]
- Huang, X. Fabrication and properties of carbon fibers. Materials 2009, 2, 2369–2403. [Google Scholar] [CrossRef]
- Sullivan, R.A. Automotive carbon fiber: Opportunities and challenges. JOM 2006, 58, 77–79. [Google Scholar] [CrossRef]
- Baker, D.A.; Rials, T.G. Recent advances in low-cost carbon fiber manufacture from lignin. J. Appl. Polym. Sci. 2013, 130, 713–728. [Google Scholar] [CrossRef]
- Rahaman, M.S.A.; Ismail, A.F.; Mustafa, A. A review of heat treatment on polyacrylonitrile fiber. Polym. Degrad. Stab. 2007, 92, 1421–1432. [Google Scholar] [CrossRef]
- Tsai, J.-S.; Lin, C.-H. Effect of comonomer composition on the properties of polyacrylonitrile precursor and resulting carbon fiber. J. Appl. Polym. Sci. 1991, 43, 679–685. [Google Scholar] [CrossRef]
- Dietrich, J.; Hirt, P.; Herlinger, H. Electron-beam-induced cyclization to obtain C-fiber precursors from polyacrylonitrile homopolymers. Eur. Polym. J. 1996, 32, 617–623. [Google Scholar] [CrossRef]
- Kim, S.Y.; Lee, S.; Park, S.; Jo, S.M.; Lee, H.S.; Joh, H.I. Continuous and rapid stabilization of polyacrylonitrile fiber bundles assisted by atmospheric pressure plasma for fabricating large-tow carbon fibers. Carbon 2015, 94, 617–623. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, P.; Zhang, L.; Ma, Z.; Liang, J.; Fong, H. Thermo-chemical reactions occurring during the oxidative stabilization of electrospun polyacrylonitrile precursor nanofibers and the resulting structural conversions. Carbon 2009, 47, 1087–1095. [Google Scholar] [CrossRef]
- Xue, Y.; Liu, J.; Liang, J. Correlative study of critical reactions in polyacrylonitrile based carbon fiber precursors during thermal-oxidative stabilization. Polym. Degrad. Stab. 2013, 98, 219–229. [Google Scholar] [CrossRef]
- Shin, H.K.; Park, M.; Kim, H.-Y.; Park, S.-J. An overview of new oxidation methods for polyacrylonitrile-based carbon fibers. Carbon Lett. 2015, 16, 11–18. [Google Scholar] [CrossRef]
- Ozur, G.E.; Proskurovsky, D.I.; Rotshtein, V.P.; Markov, A.B. Production and application of low-energy high-current electron beams. Laser Part. Beams 2003, 21, 157–174. [Google Scholar] [CrossRef]
- Yuan, H.; Wang, Y.; Liu, P.; Yu, H.; Ge, B.; Mei, Y. Effect of electron beam irradiation on polyacrylonitrile precursor fibers and stabilization process. J. Appl. Polym. Sci. 2011, 122, 90–96. [Google Scholar] [CrossRef]
- Shin, H.K.; Park, M.; Kang, P.H.; Choi, H.-S.; Park, S.-J. Preparation and characterization of polyacrylonitrile-based carbon fibers produced by electron beam irradiation pretreatment. J. Ind. Eng. Chem. 2014, 20, 3789–3792. [Google Scholar] [CrossRef]
- Yu, H.; Yuan, H.; Wang, Y.; Wei, Z. The characterization of different doses of electron beam irradiation on the structure and properties of PAN precursor fibers. Adv. Mater. Res. 2011, 328, 1594–1597. [Google Scholar] [CrossRef]
- Shin, H.K.; Park, M.; Kim, H.-Y.; Park, S.-J. Influence of orientation on ordered microstructure of PAN-based fibers during electron beam irradiation stabilization. J. Ind. Eng. Chem. 2015, 32, 120–122. [Google Scholar] [CrossRef]
- Kim, D.-Y.; Shin, H.-K.; Jeun, J.-P.; Kim, H.-B.; Oh, S.-H.; Kang, P.-H. Characterization of polyacrylonitrile based carbon nanofiber mats via electron beam processing. J. Nanosci. Nanotechnol. 2012, 12, 6120–6124. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Yoo, S.H.; Kang, H.R.; Jo, S.M.; Joh, H.I.; Lee, S. Comprehensive stabilization mechanism of electron-beam irradiated polyacrylonitrile fibers to shorten the conventional thermal treatment. Sci. Rep. 2016, 6, 27330. [Google Scholar] [CrossRef] [PubMed]
- Fitzer, E.; Frohs, W.; Heine, M. Optimization of stabilization and carbonization treatment of PAN fibres and structural characterization of the resulting carbon fibres. Carbon 1986, 24, 387–395. [Google Scholar] [CrossRef]
- Shin, H.K.; Jeun, J.P.; Kang, P.H. The characterization of polyacrylonitrile fibers stabilized by electron beam irradiation. Fibers Polym. 2012, 13, 724–728. [Google Scholar] [CrossRef]
- Lian, F.; Liu, J.; Ma, Z.; Liang, J. Stretching-induced deformation of polyacrylonitrile chains both in quasicrystals and in amorphous regions during the in situ thermal modification of fibers prior to oxidative stabilization. Carbon 2012, 50, 488–499. [Google Scholar] [CrossRef]
- Houtz, R.C. “Orlon” acrylic fiber: Chemistry and properties. Text. Res. J. 1950, 20, 786–801. [Google Scholar] [CrossRef]
- Nunna, S.; Naebe, M.; Hameed, N.; Creighton, C.; Naghashian, S.; Jennings, M.J.; Atkiss, S.; Setty, M.; Fox, B.L. Investigation of progress of reactions and evolution of radial heterogeneity in the initial stage of thermal stabilization of PAN precursor fibres. Polym. Degrad. Stab. 2016, 125, 105–114. [Google Scholar] [CrossRef]
- Liu, J.; Xiao, S.; Shen, Z.; Xu, L.; Zhang, L.; Peng, J. Study on the oxidative stabilization of polyacrylonitrile fibers by microwave heating. Polym. Degrad. Stab. 2018, 150, 86–91. [Google Scholar] [CrossRef]
- Pethkar, S.; Dharmadhikari, J.A.; Athawale, A.A.; Aiyer, R.C.; Vijayamohanan, K. Evidence for second-order optical nonlinearity in γ-ray induced partially cross-linked polyacrylonitrile. J. Phys. Chem. B 2001, 105, 5110–5113. [Google Scholar] [CrossRef]
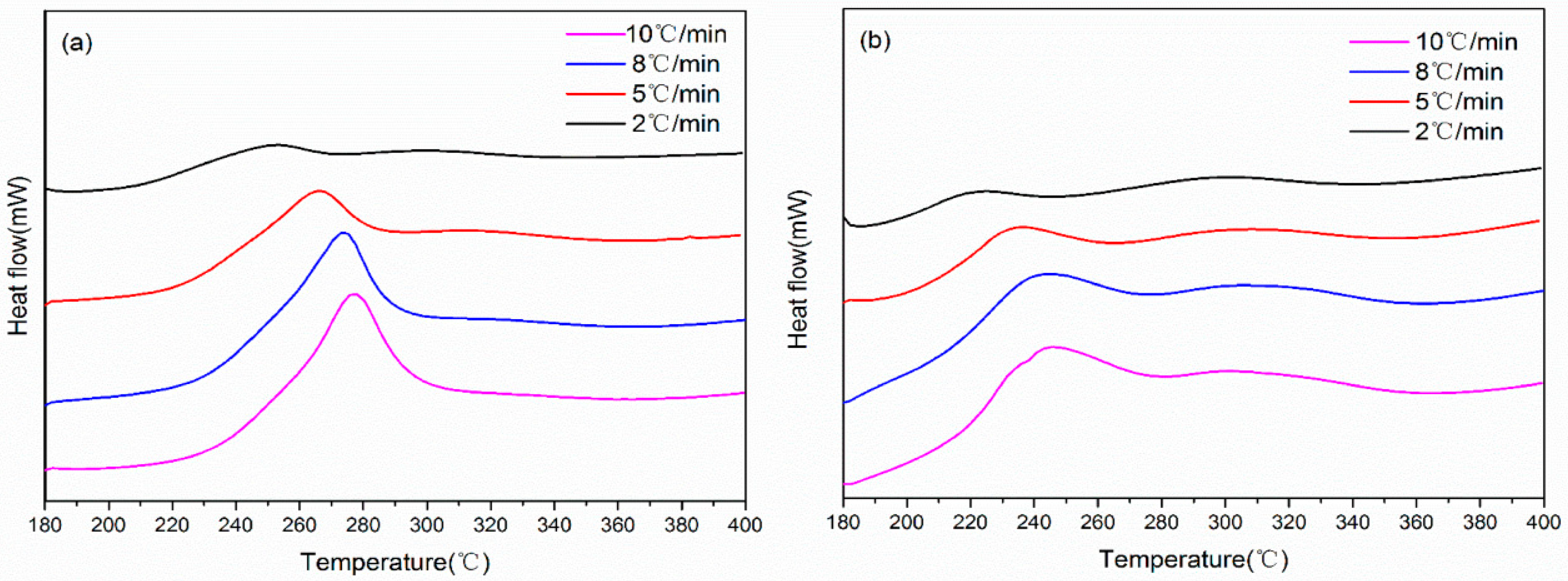

| Heating Rate (°C/min) | PAN Fibers | Irradiated PAN Fibers | ||||
|---|---|---|---|---|---|---|
| Tonset (°C) | Tp.1 (°C) | ΔH (J/g) | Tonset (°C) | Tp.1 (°C) | ΔH (J/g) | |
| 2 | 193.7 | 252.9 | 3199 | 186.8 | 224.2 | 1666 |
| 5 | 204.8 | 267.8 | 2363 | 189.9 | 235.1 | 1258 |
| 8 | 208.3 | 276.6 | 1972 | 181.3 | 242.6 | 1584 |
| 10 | 212.6 | 279.3 | 1616 | 182.6 | 244.9 | 1309 |
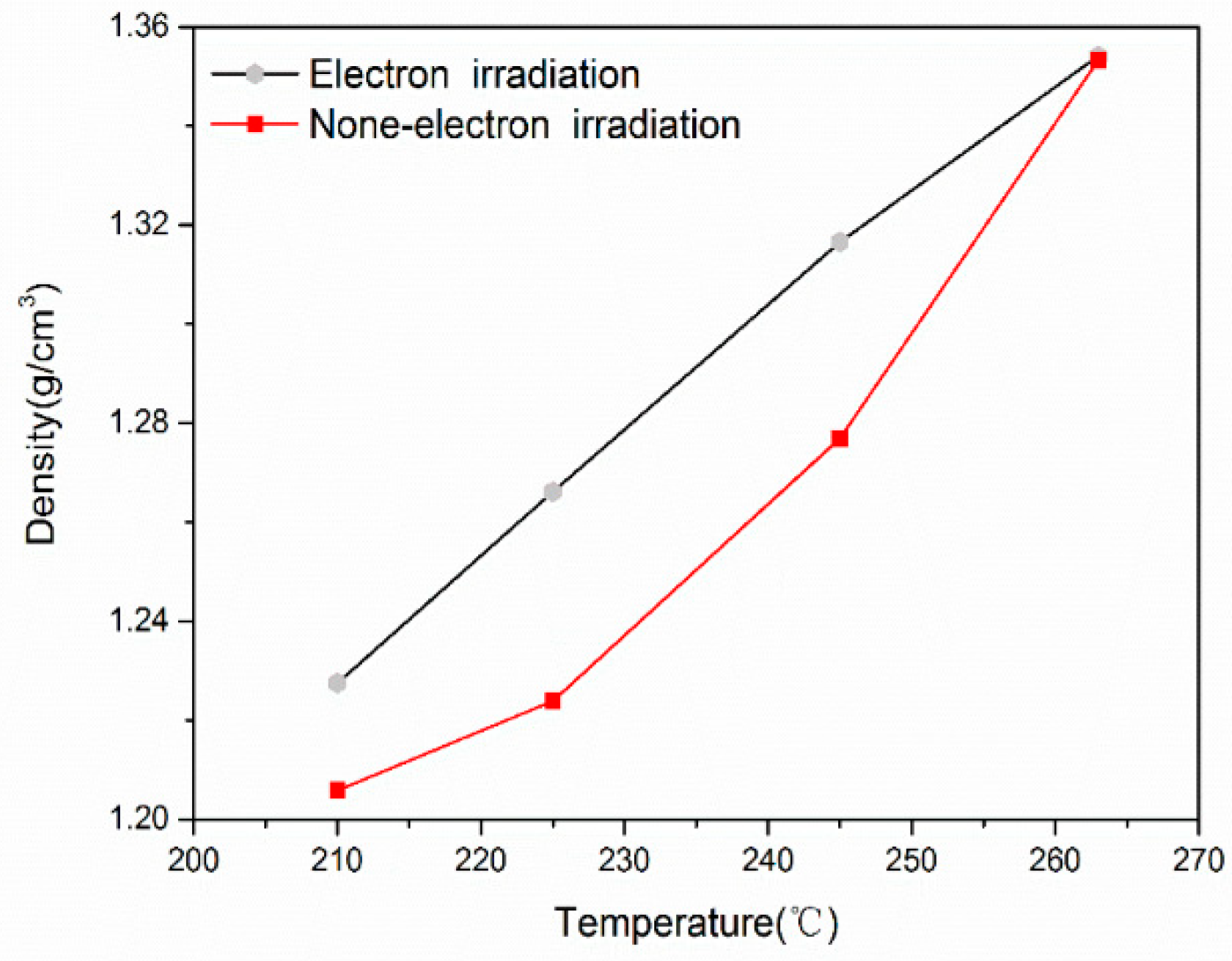
| RCIs | |||||
|---|---|---|---|---|---|
| 25 °C | 210 °C | 225 °C | 245 °C | 263 °C | |
| Irradiated PAN fibers | 18.7% | 38.3% | 61.1% | 71.3% | 77.5% |
| PAN fibers | 7.85% | 22.2% | 43.5% | 62.7% | 76.9% |
| Density (g/cm3) | Tensile Strength (GPa) | CV (%) | Modulus (GPa) | CV (%) | Elongation (%) | CV (%) | |
|---|---|---|---|---|---|---|---|
| CFs | 1.805 | 3.56 | 2.05 | 228 | 2.32 | 1.51 | 5.70 |
| i-CFs | 1.813 | 2.85 | 1.69 | 203 | 1.47 | 1.40 | 3.47 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Liu, Y.; Liu, J.; Shen, Z.; Liang, J.; Wang, X. Rapid and Continuous Preparation of Polyacrylonitrile-Based Carbon Fibers with Electron-Beam Irradiation Pretreatment. Materials 2018, 11, 1270. https://doi.org/10.3390/ma11081270
Yang J, Liu Y, Liu J, Shen Z, Liang J, Wang X. Rapid and Continuous Preparation of Polyacrylonitrile-Based Carbon Fibers with Electron-Beam Irradiation Pretreatment. Materials. 2018; 11(8):1270. https://doi.org/10.3390/ma11081270
Chicago/Turabian StyleYang, Jia, Yuchen Liu, Jie Liu, Zhigang Shen, Jieying Liang, and Xiaoxu Wang. 2018. "Rapid and Continuous Preparation of Polyacrylonitrile-Based Carbon Fibers with Electron-Beam Irradiation Pretreatment" Materials 11, no. 8: 1270. https://doi.org/10.3390/ma11081270
APA StyleYang, J., Liu, Y., Liu, J., Shen, Z., Liang, J., & Wang, X. (2018). Rapid and Continuous Preparation of Polyacrylonitrile-Based Carbon Fibers with Electron-Beam Irradiation Pretreatment. Materials, 11(8), 1270. https://doi.org/10.3390/ma11081270