Ni(OH)2 and NiO Based Composites: Battery Type Electrode Materials for Hybrid Supercapacitor Devices
Abstract
1. Introduction
- Capacitive storage implies an electrostatic surface reaction via the capacitive adsorption of ions in the electrochemical double layer. Indeed, when a potential is applied at the electrode surface, the charge accumulated in the solid is compensated by an electrolyte ion adsorption. The amount of charge depends of the applied potential and the stored energy varies linearly as a function of the potential. In this configuration, the ability to store energy is characterized by its capacitance and is expressed in Farads, i.e., C·V−1 (Equation (1)), or F. It must be pointed out that the value of the capacitance must remain constant all over the given potential window for ideal capacitive storage. The capacitance unit can be used to easily compare various capacitors, but such value always needs to be accompanied with the corresponding cell voltage in which the capacitor can be safely operated [5]. The same goes for a single electrode: the capacitance value measured in a three-electrode cell needs to be quoted with the potential window in which the electrode can be safely used to store charge.
- Faradaic charge storage involves redox processes which imply an electronic transfer between the electrode and the electrolyte ions, thus leading to a change of oxidation state within the bulk of the electrode. The stored energy occurs at a quasi-constant potential and the charge stored depends of the quantity of electrons exchanged during the reaction. In this configuration, the stored charge (Q) is known as capacity and is expressed in C·g−1 or most commonly in mAh·g−1 in order to compare various types of batteries (Equation (2)).where n is the number of exchanged electron, F the Faraday constant and M the molar mass of the reactant species.
- Pseudocapacitive electrodes, such as those made of RuO2 or MnO2, store energy via fast redox processes limited to the electrode surface [6,7] but they have the same electrochemical signature than that of a capacitive electrode, i.e., exhibiting a linear dependence of the charge stored with voltage within the potential window range. Thus, a constant capacitance value is often provided to compare pseudocapacitive materials.
2. Methods
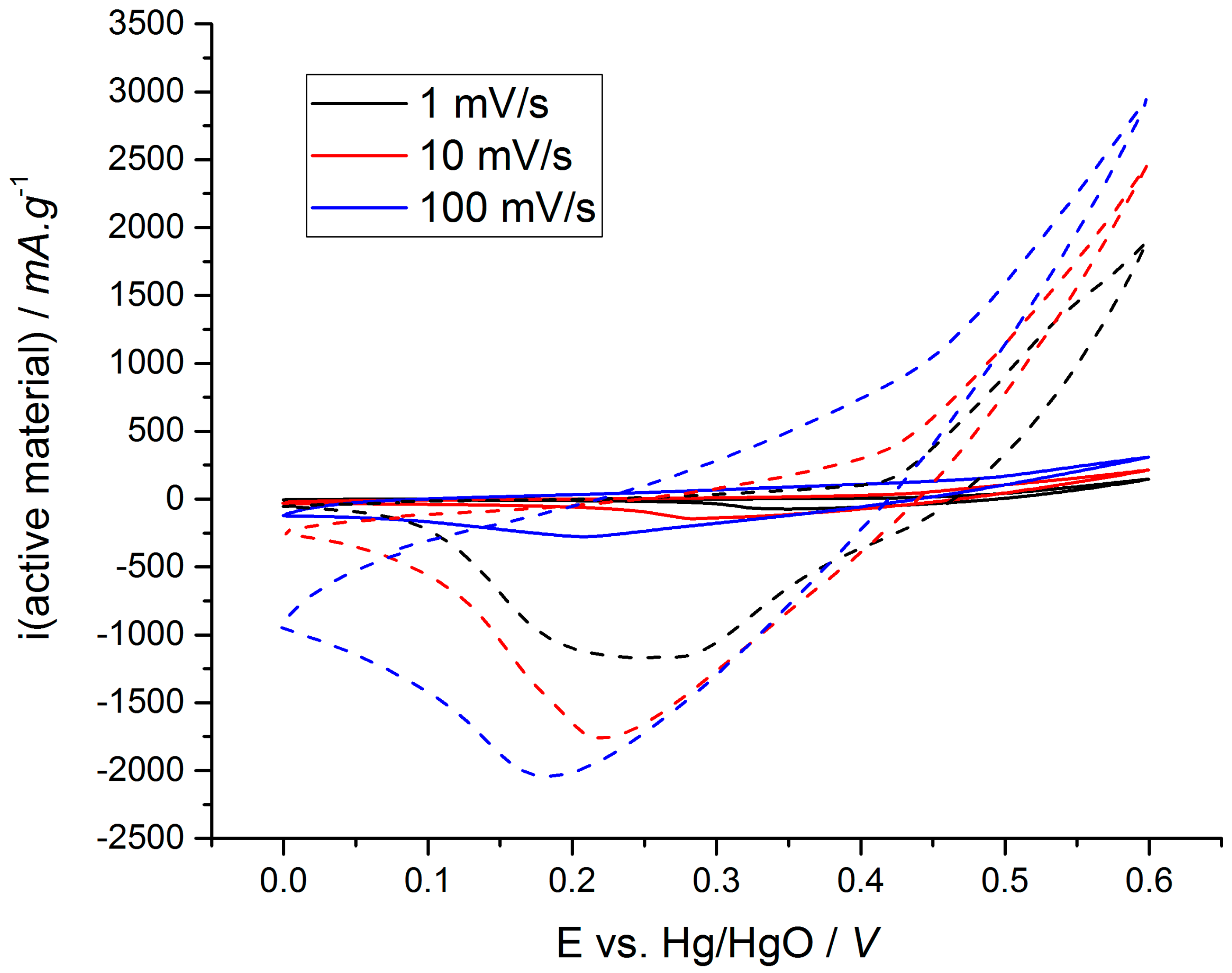
3. Results and Discussion
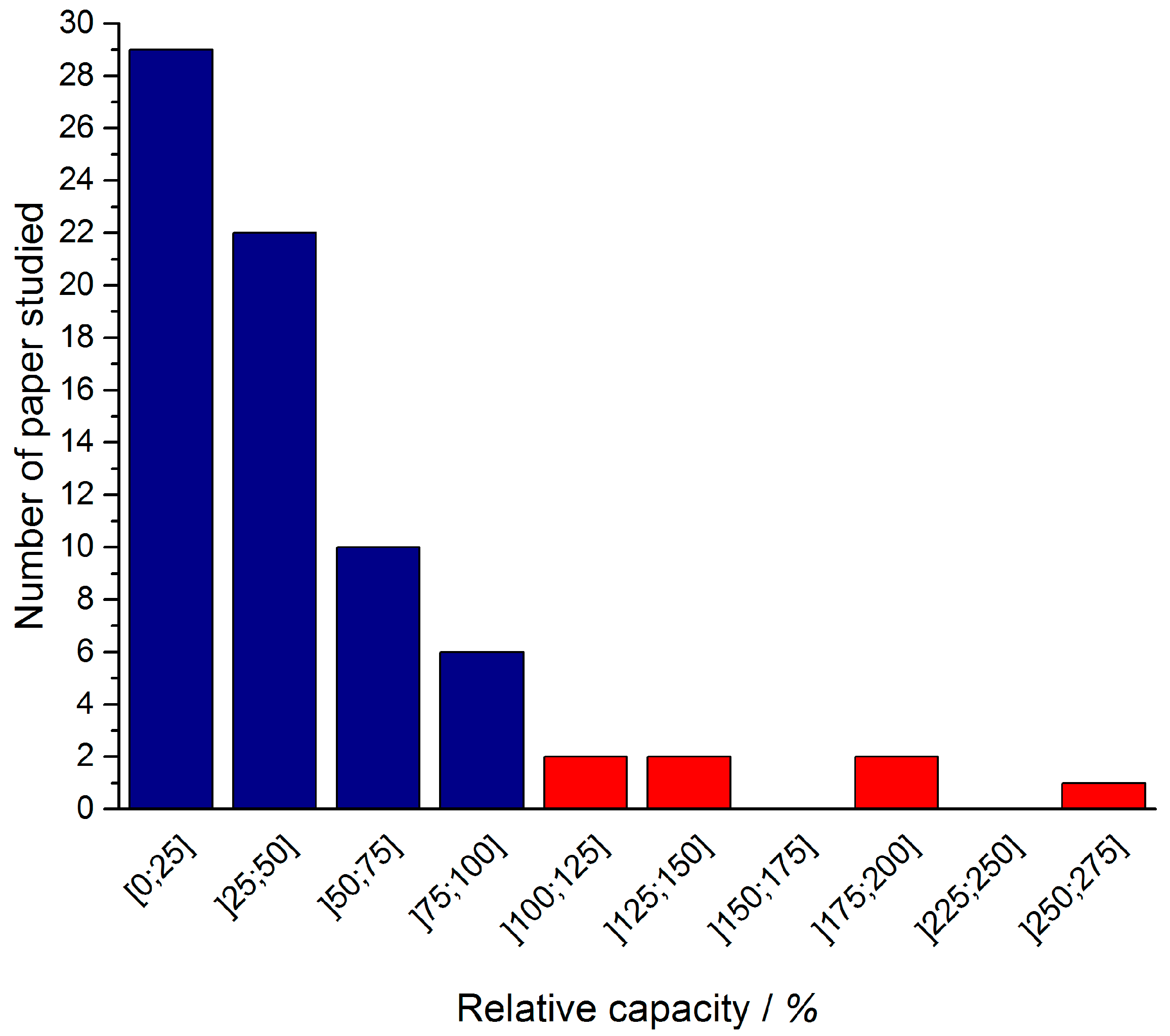
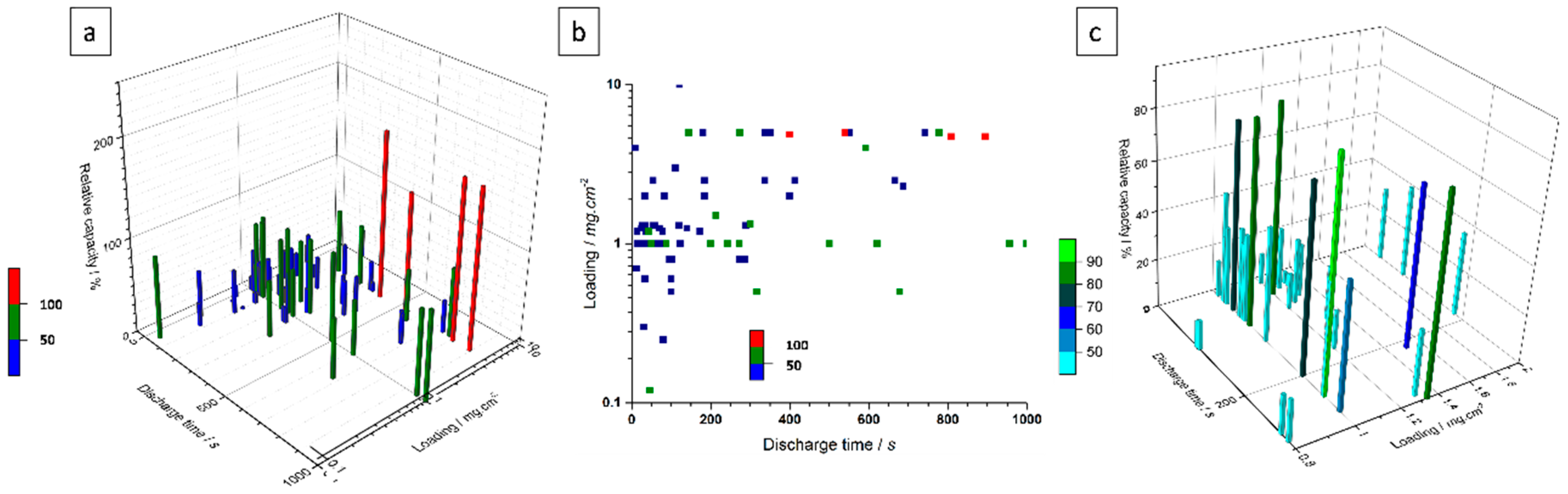
3.1. Litterature Findings
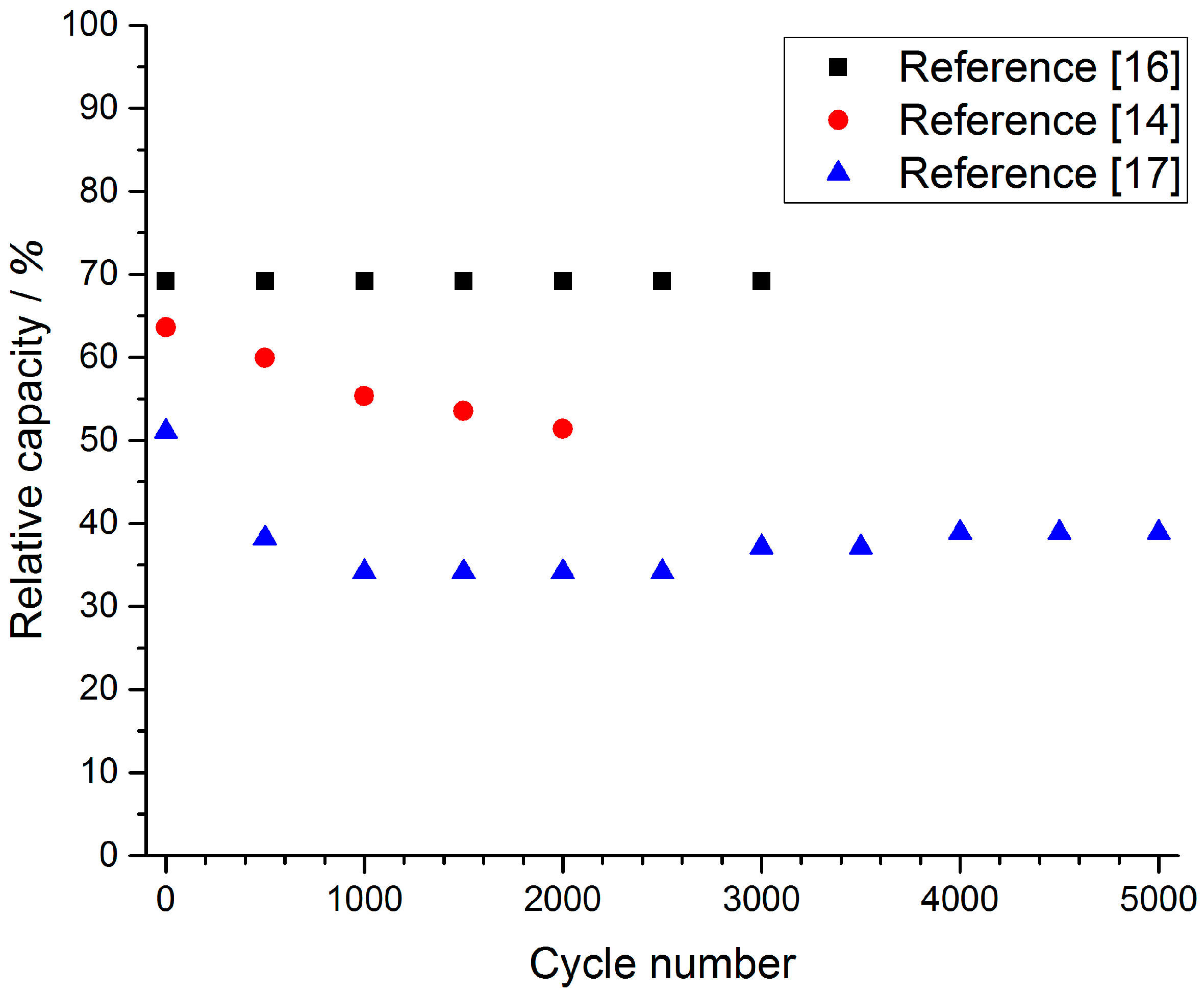
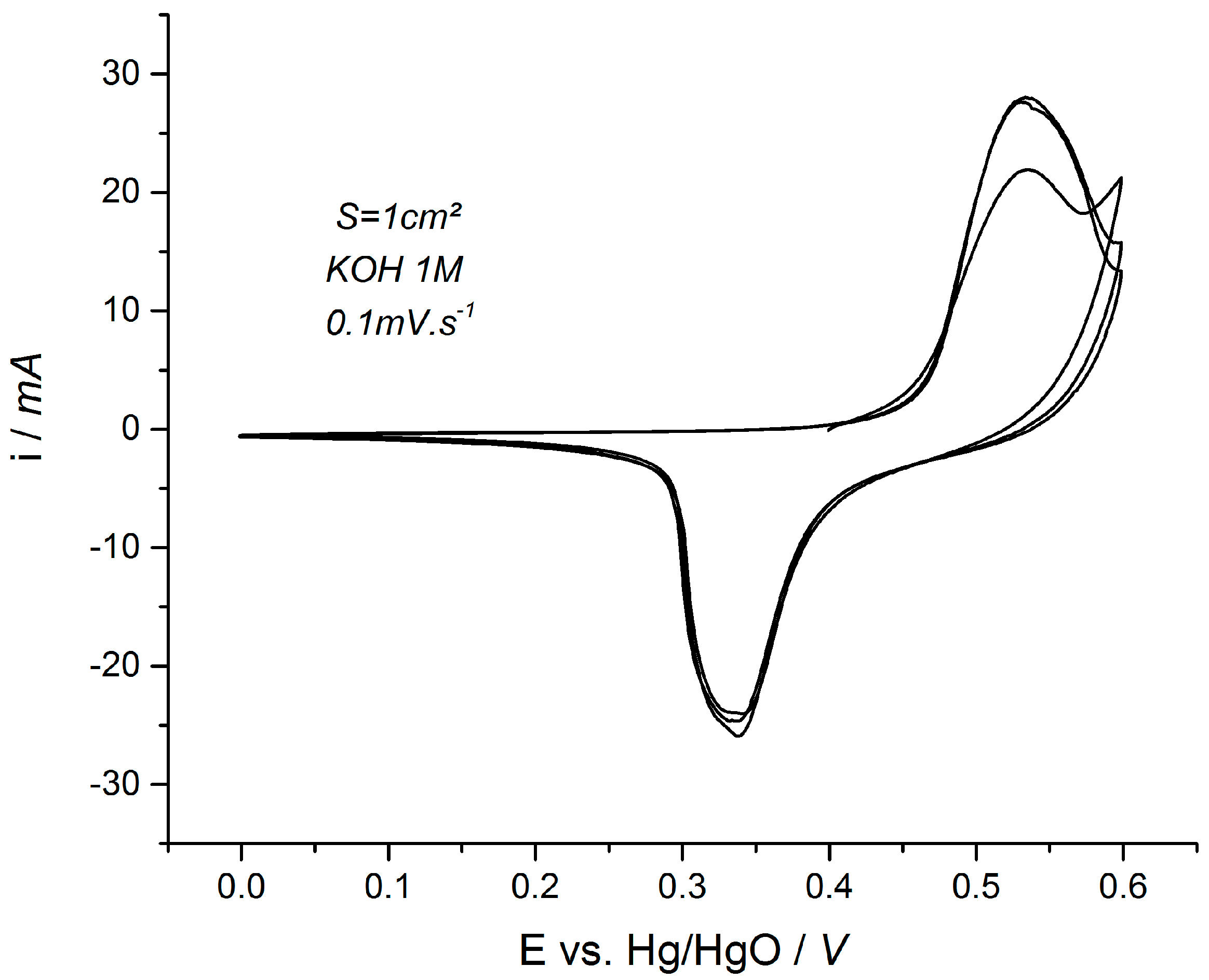
3.2. Cycling Ability of Ni(OH)2 Based Electrodes
4. Conclusions
- Firstly, the preparation routes are often more complex than standard Ni(OH)2 battery-type electrodes, unlike what most of the authors are claiming.
- Secondly, only a few studies are using reasonable mass loadings (≥10 mg·cm−2) which are necessary to validate the gravimetric values reported for capacity, energy and power densities. There is a need to investigate electrodes with such reasonable loadings, especially with regards to the capacity and the rate capability that can be achieved in a real-life hybrid device. The use of standard electrode preparation processes such as tape casting, doctor blade or bar coating will provide such mass loadings.
- Thirdly, the cyclability is also a key parameter for Ni(OH)2 or NiO-based electrodes for supercapacitors application, and high cycling stability (≥7000 cycles) must be demonstrated depending upon the targeted application. Again, a sufficient mass loading must be used for these studies. Moreover, this parameter has to be evaluated not only for a single electrode but also when implemented in optimized full devices.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Le Comte, A.; Brousse, T.; Bélanger, D. New generation of hybrid carbon/Ni(OH)2 electrochemical capacitor using functionalized carbon electrode. J. Power Sources 2016, 326, 702–710. [Google Scholar] [CrossRef]
- Amatucci, G.G.; Badway, F.; Du Pasquier, A.; Zheng, T. An Asymmetric Hybrid Nonaqueous Energy Storage Cell. J. Electrochem. Soc. 2001, 148, A930–A939. [Google Scholar] [CrossRef]
- Zheng, J.P. The Limitations of Energy Density of Battery/Double-Layer Capacitor Asymmetric Cells. J. Electrochem. Soc. 2003, 150, A484–A492. [Google Scholar] [CrossRef]
- Pell, W.G.; Conway, B.E. Peculiarities and requirements of asymmetric capacitor devices based on combination of capacitor and battery-type electrodes. J. Power Sources 2004, 136, 334–345. [Google Scholar] [CrossRef]
- Balducci, A.; Belanger, D.; Brousse, T.; Long, J.W.; Sugimoto, W. Perspective—A Guideline for Reporting Performance Metrics with Electrochemical Capacitors: From Electrode Materials to Full Devices. J. Electrochem. Soc. 2017, 164, A1487–A1488. [Google Scholar] [CrossRef]
- Lee, H.Y.; Goodenough, J.B. Supercapacitor Behavior with KCl Electrolyte. J. Solid State Chem. 1999, 144, 220–223. [Google Scholar] [CrossRef]
- Brousse, T.; Toupin, M.; Dugas, R.; Athouël, L.; Crosnier, O.; Bélanger, D. Crystalline MnO2 as Possible Alternatives to Amorphous Compounds in Electrochemical Supercapacitors. J. Electrochem. Soc. 2006, 153, A2171–A2181. [Google Scholar] [CrossRef]
- Tessier, C.; Haumesser, P.H.; Bernard, P.; Delmas, C. The Structure of Ni(OH)2: From the Ideal Material to the Electrochemically Active One. J. Electrochem. Soc. 1999, 146, 2059–2061. [Google Scholar] [CrossRef]
- Oliva, P.; Leonardi, J.; Laurent, J.F.; Figlarz, M.; Fievet, F.; Delmas, C.; Braconnier, J.J. Review of the structure and the electrochemistry of nickel hydroxydes and oxy-hydroxides. J. Electrochem. Soc. 1982, 8, 229–255. [Google Scholar] [CrossRef]
- Brousse, T.; Belanger, D.; Long, J.W. To be or not to be pseudocapacitive? J. Electrochem. Soc. 2015, 162, A5185–A5189. [Google Scholar] [CrossRef]
- Frackowiak, E.; Béguin, F. Supercapacitors: Materials, Systems and Applications; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar]
- Yi, H.; Wang, H.; Jing, Y.; Peng, T.; Wang, Y.; Guo, J.; He, Q.; Guo, Z.; Wang, X. Advanced asymmetric supercapacitors based on CNT@Ni(OH)2 core–shell composites and 3D graphene networks. J. Mater. Chem. A 2015, 3, 19545–19555. [Google Scholar] [CrossRef]
- Bag, S.; Raj, C.R. Layered inorganic–organic hybrid material based on reduced graphene oxide and α-Ni(OH)2 for high performance supercapacitor electrodes. J. Mater. Chem. A 2014, 2, 17848–17856. [Google Scholar] [CrossRef]
- Fang, D.-L.; Chen, Z.-D.; Liu, X.; Wu, Z.-F.; Zheng, C.-H. Homogeneous growth of nano-sized β-Ni(OH)2 on reduced graphene oxide for high-performance supercapacitors. Electrochim. Acta 2012, 81, 321–329. [Google Scholar] [CrossRef]
- Wang, H.; Casalongue, H.S.; Liang, Y.; Dai, H. Ni(OH)2 Nanoplates Grown on Graphene as Advanced Electrochemical Pseudocapacitor Materials. J. Am. Chem. Soc. 2010, 132, 7472–7477. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, Z.; Sui, Z.; Liu, Z.; Zhou, J.; Zhou, X. Ni(OH)2 nanowires/graphite foam composite as an advanced supercapacitor electrode with improved cycle performance. Int. J. Hydrog. Energy 2016, 41, 12136–12145. [Google Scholar] [CrossRef]
- Cai, G.; Wang, X.; Cui, M.; Darmawan, P.; Wang, J.; Eh, A.L.-S.; Lee, P.S. Electrochromo-supercapacitor based on direct growth of NiO nanoparticles. Nano Energy 2015, 12, 258–267. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, L.; Xia, Y. Electrochemical Capacitance Performance of Hybrid Supercapacitors Based on Ni(OH)2/Carbon Nanotube Composites and Activated Carbon. J. Electrochem. Soc. 2006, 153, A743–A748. [Google Scholar] [CrossRef]
- Lin, P.; She, Q.; Hong, B.; Liu, X.; Shi, Y.; Shi, Z.; Zheng, M.; Dong, Q. The Nickel Oxide/CNT Composites with High Capacitance for Supercapacitor. J. Electrochem. Soc. 2010, 157, A818–A823. [Google Scholar] [CrossRef]
- Yeager, M.P.; Su, D.; Marinkovic, N.S.; Teng, X. Pseudocapacitive NiO Fine Nanoparticles for Supercapacitor Reactions. J. Electrochem. Soc. 2012, 159, A1598–A1603. [Google Scholar] [CrossRef]
- Bu, Y.; Wang, S.; Jin, H.; Zhang, W.; Lin, J.; Wang, J. Synthesis of Porous NiO/Reduced Graphene Oxide Composites for Supercapacitors. J. Electrochem. Soc. 2012, 159, A990–A994. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, D.; Zhao, D.; Hou, M.; Wang, C.; Xia, Y. High Performance Hybrid Supercapacitor Based on Graphene-Supported Ni(OH)2-Nanowires and Ordered Mesoporous Carbon CMK-5. J. Electrochem. Soc. 2012, 160, A98–A104. [Google Scholar] [CrossRef]
- Li, H.B.; Yu, M.H.; Wang, F.X.; Liu, P.; Liang, Y.; Xiao, J.; Wang, C.X.; Tong, Y.X.; Yang, G.W. Amorphous nickel hydroxide nanospheres with ultrahigh capacitance and energy density as electrochemical pseudocapacitor materials. Nat. Commun. 2013, 4, 1894–1901. [Google Scholar] [CrossRef] [PubMed]
- Salunkhe, R.R.; Lin, J.; Malgras, V.; Dou, S.X.; Kim, J.H.; Yamauchi, Y. Large-scale synthesis of coaxial carbon nanotube/Ni(OH)2 composites for asymmetric supercapacitor application. Nano Energy 2015, 11, 211–218. [Google Scholar] [CrossRef]
- Tang, Z.; Tang, C.; Gong, H. A High Energy Density Asymmetric Supercapacitor from Nano-architectured Ni(OH)2/Carbon Nanotube Electrodes. Adv. Funct. Mater. 2012, 22, 1272–1278. [Google Scholar] [CrossRef]
- Dubal, D.P.; Gund, G.S.; Lokhande, C.D.; Holze, R. Decoration of Spongelike Ni(OH)2 Nanoparticles onto MWCNTs Using an Easily Manipulated Chemical Protocol for Supercapacitors. ACS Appl. Mater. Interfaces 2013, 5, 2446–2454. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Huang, X.; Lin, Z.; Zhong, X.; Huang, Y.; Duan, X. One-step strategy to graphene/Ni(OH)2 composite hydrogels as advanced three-dimensional supercapacitor electrode materials. Nano Res. 2013, 6, 65–76. [Google Scholar] [CrossRef]
- Yan, J.; Fan, Z.; Sun, W.; Ning, G.; Wei, T.; Zhang, Q.; Zhang, R.; Zhi, L.; Wei, F. Advanced Asymmetric Supercapacitors Based on Ni(OH)2/Graphene and Porous Graphene Electrodes with High Energy Density. Adv. Funct. Mater. 2012, 22, 2632–2641. [Google Scholar] [CrossRef]
- Lu, Q.; Lattanzi, M.W.; Chen, Y.; Kou, X.; Li, W.; Fan, X.; Unruh, K.M.; Chen, J.G.; Xiao, J.Q. Supercapacitor Electrodes with High-Energy and Power Densities Prepared from Monolithic NiO/Ni Nanocomposites. Angew. Chem. Int. Ed. 2011, 50, 6847–6850. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Tu, J.; Wang, X.; Gu, C.; Zhao, X. Hierarchically porous NiO film grown by chemical bath deposition via a colloidal crystal template as an electrochemical pseudocapacitor material. J. Mater. Chem. 2011, 21, 671–679. [Google Scholar] [CrossRef]
- Yuan, C.; Zhang, X.; Su, L.; Gao, B.; Shen, L. Facile synthesis and self-assembly of hierarchical porous NiO nano/micro spherical superstructures for high performance supercapacitors. J. Mater. Chem. 2009, 19, 5772–5777. [Google Scholar] [CrossRef]
- Liang, K.; Tang, X.; Hu, W. High-performance three-dimensional nanoporous NiO film as a supercapacitor electrode. J. Mater. Chem. 2012, 22, 11062–11067. [Google Scholar] [CrossRef]
- Meher, S.K.; Justin, P.; Ranga Rao, G. Microwave-Mediated Synthesis for Improved Morphology and Pseudocapacitance Performance of Nickel Oxide. ACS Appl. Mater. Interfaces 2011, 3, 2063–2073. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-I.; Lee, J.-S.; Ahn, H.-J.; Song, H.-K.; Jang, J.-H. Facile Route to an Efficient NiO Supercapacitor with a Three-Dimensional Nanonetwork Morphology. ACS Appl. Mater. Interfaces 2013, 5, 1596–1603. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, S.; Nagamuthu, S.; Muralidharan, G. Supercapacitor Studies on NiO Nanoflakes Synthesized Through a Microwave Route. ACS Appl. Mater. Interfaces 2013, 5, 2188–2196. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K.R.; Miura, N. Electrochemically deposited nanowhiskers of nickel oxide as a high-power pseudocapacitive electrode. Appl. Phys. Lett. 2004, 85, 4199–4201. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, J.; Guo, H.; Tong, X. Hierarchically porous nickel oxide nanoflake arrays grown on carbon cloth by chemical bath deposition as superior flexible electrode for supercapacitors. Mater. Lett. 2014, 136, 198–201. [Google Scholar] [CrossRef]
- Wang, L.; Chen, H.; Cai, F.; Chen, M. Hierarchical carbon nanotube/α-Ni(OH)2 nanosheet composite paper with enhanced electrochemical capacitance. Mater. Lett. 2014, 115, 168–171. [Google Scholar] [CrossRef]
- Tai, Y.-L.; Teng, H. Modification of porous carbon with nickel oxide impregnation to enhance the electrochemical capacitance and conductivity. Carbon 2004, 42, 2335–2338. [Google Scholar] [CrossRef]
- Shahid, M.; Liu, J.; Shakir, I.; Warsi, M.F.; Nadeem, M.; Kwon, Y.-U. Facile approach to synthesize Ni(OH)2 nanoflakes on MWCNTs for high performance electrochemical supercapacitors. Electrochim. Acta 2012, 85, 243–247. [Google Scholar] [CrossRef]
- Yuan, B.; Xu, C.; Deng, D.; Xing, Y.; Liu, L.; Pang, H.; Zhang, D. Graphene oxide/nickel oxide modified glassy carbon electrode for supercapacitor and nonenzymatic glucose sensor. Electrochim. Acta 2013, 88, 708–712. [Google Scholar] [CrossRef]
- Min, S.; Zhao, C.; Chen, G.; Qian, X. One-pot hydrothermal synthesis of reduced graphene oxide/Ni(OH)2 films on nickel foam for high performance supercapacitors. Electrochim. Acta 2014, 115, 155–164. [Google Scholar] [CrossRef]
- Liu, W.; Ju, C.; Jiang, D.; Xu, L.; Mao, H.; Wang, K. Ionic liquid-assisted grown of beta-nickel hydroxide nanowires on reduced graphene oxide for high-performance supercapacitors. Electrochim. Acta 2014, 143, 135–142. [Google Scholar] [CrossRef]
- Wang, L.; Tian, H.; Wang, D.; Qin, X.; Shao, G. Preparation and electrochemical characteristic of porous NiO supported by sulfonated graphene for supercapacitors. Electrochim. Acta 2015, 151, 407–414. [Google Scholar] [CrossRef]
- Yan, H.; Bai, J.; Wang, B.; Yu, L.; Zhao, L.; Wang, J.; Liu, Q.; Liu, J.; Li, Z. Electrochemical reduction approach-based 3D graphene/Ni(OH)2 electrode for high-performance supercapacitors. Electrochim. Acta 2015, 154, 9–16. [Google Scholar] [CrossRef]
- Cao, P.; Wang, L.; Xu, Y.; Fu, Y.; Ma, X. Facile hydrothermal synthesis of mesoporous nickel oxide/reduced graphene oxide composites for high performance electrochemical supercapacitor. Electrochim. Acta 2015, 157, 359–368. [Google Scholar] [CrossRef]
- Sui, L.; Tang, S.; Chen, Y.; Dai, Z.; Huangfu, H.; Zhu, Z.; Qin, X.; Deng, Y.; Haarberg, G.M. An asymmetric supercapacitor with good electrochemical performances based on Ni(OH)2/AC/CNT and AC. Electrochim. Acta 2015, 182, 1159–1165. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, T.; Jiang, L.; Zhang, K.; Yuen, M.M.F.; Xu, J.-B.; Fu, X.-Z.; Sun, R.; Wong, C.-P. NiO mesoporous nanowalls grown on RGO coated nickel foam as high performance electrodes for supercapacitors and biosensors. Electrochim. Acta 2016, 192, 205–215. [Google Scholar] [CrossRef]
- Mao, L.; Guan, C.; Huang, X.; Ke, Q.; Zhang, Y.; Wang, J. 3D Graphene-Nickel Hydroxide Hydrogel Electrode for High-Performance Supercapacitor. Electrochim. Acta 2016, 196, 653–660. [Google Scholar] [CrossRef]
- Dai, K.; Liang, C.; Dai, J.; Lu, L.; Zhu, G.; Liu, Z.; Liu, Q.; Zhang, Y. High-yield synthesis of carbon nanotube–porous nickel oxide nanosheet hybrid and its electrochemical capacitance performance. Mater. Chem. Phys. 2014, 143, 1344–1351. [Google Scholar] [CrossRef]
- Wen, Z.B.; Yu, F.; You, T.; Zhu, L.; Zhang, L.; Wu, Y.P. A core–shell structured nanocomposite of NiO with carbon nanotubes as positive electrode material of high capacitance for supercapacitors. Mater. Res. Bull. 2016, 74, 241–247. [Google Scholar] [CrossRef]
- Huang, M.L.; Gu, C.D.; Ge, X.; Wang, X.L.; Tu, J.P. NiO nanoflakes grown on porous graphene frameworks as advanced electrochemical pseudocapacitor materials. J. Power Sources 2014, 259, 98–105. [Google Scholar] [CrossRef]
- Liu, C.-G.; Lee, Y.-S.; Kim, Y.-J.; Song, I.-C.; Kim, J.-H. Electrochemical characteristics of hydrothermally deposited nickel hydroxide on multi-walled carbon nanotube for supercapacitor electrode. Synth. Met. 2009, 159, 2009–2012. [Google Scholar] [CrossRef]
- Yuan, C. NiO loaded on hydrothermally treated mesocarbon microbeads (h-MCMB) and their supercapacitive behaviors. Solid State Ion. 2008, 178, 1859–1866. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, J.; Xu, D.; Zhang, B.; Shi, L.; Huang, L.; Tan, S. In situ formation of Ni(OH)2 nanoparticle on nitrogen-doped reduced graphene oxide nanosheet for high-performance supercapacitor electrode material. Appl. Surf. Sci. 2014, 317, 370–377. [Google Scholar] [CrossRef]
- Jiang, L.; Qiu, Y.; Luo, P.; Yu, Y. Nickel hydroxide-impregnated and -coated carbon nanotubes using an easily manipulated solvothermal route for supercapacitors. Ceram. Int. 2016, 42, 11634–11639. [Google Scholar] [CrossRef]
- Wu, M.; Gao, J.; Zhang, S.; Chen, A. Comparative studies of nickel oxide films on different substrates for electrochemical supercapacitors. J. Power Sources 2006, 159, 365–369. [Google Scholar] [CrossRef]
- Wang, D.-W.; Li, F.; Cheng, H.-M. Hierarchical porous nickel oxide and carbon as electrode materials for asymmetric supercapacitor. J. Power Sources 2008, 185, 1563–1568. [Google Scholar] [CrossRef]
- Zhu, X.; Dai, H.; Hu, J.; Ding, L.; Jiang, L. Reduced graphene oxide–nickel oxide composite as high performance electrode materials for supercapacitors. J. Power Sources 2012, 203, 243–249. [Google Scholar] [CrossRef]
- Jena, A.; Munichandraiah, N.; Shivashankar, S.A. Carbonaceous nickel oxide nano-composites: As electrode materials in electrochemical capacitor applications. J. Power Sources 2013, 237, 156–166. [Google Scholar] [CrossRef]
- Huang, J.; Xu, P.; Cao, D.; Zhou, X.; Yang, S.; Li, Y.; Wang, G. Asymmetric supercapacitors based on β-Ni(OH)2 nanosheets and activated carbon with high energy density. J. Power Sources 2014, 246, 371–376. [Google Scholar] [CrossRef]
- Min, S.; Zhao, C.; Zhang, Z.; Chen, G.; Qian, X.; Guo, Z. Synthesis of Ni(OH)2/RGO pseudocomposite on nickel foam for supercapacitors with superior performance. J. Mater. Chem. A 2015, 3, 3641–3650. [Google Scholar] [CrossRef]
- Paravannoor, A.; Nair, S.V.; Pattathil, P.; Manca, M.; Balakrishnan, A. High voltage supercapacitors based on carbon-grafted NiO nanowires interfaced with an aprotic ionic liquid. Chem. Commun. 2015, 51, 6092–6095. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Zhou, J.; Wang, L.; Li, Y.; Huang, Z.; Chen, S.; Ma, Y.; Wang, L.; Yan, X. Synthesis of shape-controlled NiO-graphene nanocomposites with enhanced supercapacitive properties. New J. Chem. 2015, 39, 4026–4034. [Google Scholar] [CrossRef]
- Cheng, H.; Duong, H.M. Three dimensional carbon nanotube/nickel hydroxide gels for advanced supercapacitors. RSC Adv. 2015, 5, 30260–30267. [Google Scholar] [CrossRef]
- Tang, S.; Sui, L.; Dai, Z.; Zhu, Z.; Huangfu, H. High supercapacitive performance of Ni(OH)2/XC-72 composite prepared by microwave-assisted method. RSC Adv. 2015, 5, 43164–43171. [Google Scholar] [CrossRef]
- Lin, L.; Liu, T.; Liu, J.; Ji, K.; Sun, R.; Zeng, W.; Wang, Z. Synthesis of carbon fiber@nickel oxide nanosheet core–shells for high-performance supercapacitors. RSC Adv. 2015, 5, 84238–84244. [Google Scholar] [CrossRef]
- Xia, Y.; Wang, B.; Wang, G.; Wang, H. Easy access to nitrogen-doped mesoporous interlinked carbon/NiO nanosheet for application in lithium-ion batteries and supercapacitors. RSC Adv. 2015, 5, 98740–98746. [Google Scholar] [CrossRef]
- Xu, J.; Xue, Y.; Cao, J.; Wang, G.; Li, Y.; Wang, W.; Chen, Z. Carbon quantum dots/nickel oxide (CQDs/NiO) nanorods with high capacitance for supercapacitors. RSC Adv. 2016, 6, 5541–5546. [Google Scholar] [CrossRef]
- Guan, C.; Wang, Y.; Hu, Y.; Liu, J.; Ho, K.H.; Zhao, W.; Fan, Z.; Shen, Z.; Zhang, H.; Wang, J. Conformally deposited NiO on a hierarchical carbon support for high-power and durable asymmetric supercapacitors. J. Mater. Chem. A 2015, 3, 23283–23288. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, H.; Zhang, Y.; Bai, Y.; Wang, Y. Novel peapod NiO nanoparticles encapsulated in carbon fibers for high-efficiency supercapacitors and lithium-ion batteries. J. Mater. Chem. A 2016, 4, 3267–3277. [Google Scholar] [CrossRef]
- Yu, L.; Wang, G.; Wan, G.; Wang, G.; Lin, S.; Li, X.; Wang, K.; Bai, Z.; Xiang, Y. Highly effective synthesis of NiO/CNT nanohybrids by atomic layer deposition for high-rate and long-life supercapacitors. Dalton Trans. 2016, 45, 13779–13786. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Singh, R.K.; Savu, R.; Dubey, P.K.; Kumar, P.; Moshkalev, S.A. Microwave-assisted synthesis of void-induced graphene-wrapped nickel oxide hybrids for supercapacitor applications. RSC Adv. 2016, 6, 26612–26620. [Google Scholar] [CrossRef]
- Yu, L.; Wan, G.; Peng, X.; Dou, Z.; Li, X.; Wang, K.; Lin, S.; Wang, G. Fabrication of carbon-coated NiO supported on graphene for high performance supercapacitors. RSC Adv. 2016, 6, 14199–14204. [Google Scholar] [CrossRef]
- Boruah, B.D.; Misra, A. Nickel hydroxide coated carbon nanoparticles mediated hybrid three-dimensional graphene foam assembly for supercapacitor. RSC Adv. 2016, 6, 36307–36313. [Google Scholar] [CrossRef]
- Li, Q.; Wei, Q.; Xie, L.; Chen, C.; Lu, C.; Su, F.-Y.; Zhou, P. Layered NiO/reduced graphene oxide composites by heterogeneous assembly with enhanced performance as high-performance asymmetric supercapacitor cathode. RSC Adv. 2016, 6, 46548–46557. [Google Scholar] [CrossRef]
- Chen, S.; Duan, J.; Tang, Y.; Zhang, Q.S. Hybrid Hydrogels of Porous Graphene and Nickel Hydroxide as Advanced Supercapacitor Materials. Chem. Eur. J. 2013, 19, 7118–7124. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Bu, Y.; Jin, H.; Wang, J.; Zhang, W.; Wang, S.; Wang, J. The Preparation of Hierarchical Flowerlike NiO/Reduced Graphene Oxide Composites for High Performance Supercapacitor Applications. Energy Fuel 2013, 27, 6304–6310. [Google Scholar] [CrossRef]
- Sun, Z.; Lu, X. A Solid-State Reaction Route to Anchoring Ni(OH)2 Nanoparticles on Reduced Graphene Oxide Sheets for Supercapacitors. Ind. Eng. Chem. Res. 2012, 51, 9973–9979. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.; Zhang, D.; Sun, X.; Lin, H.; Wang, C.; Ma, Y. One-Step Electrophoretic Deposition of Reduced Graphene Oxide and Ni(OH)2 Composite Films for Controlled Syntheses Supercapacitor Electrodes. J. Phys. Chem. B 2013, 117, 1616–1627. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Cheng, Y.; Varanasi, C.V.; Liu, J. Influence of the Nickel Oxide Nanostructure Morphology on the Effectiveness of Reduced Graphene Oxide Coating in Supercapacitor Electrodes. J. Phys. Chem. C 2014, 118, 2281–2286. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, Y.; Guo, W.; Chen, T.; Wang, H.; Yu, S.; Gao, F. Highly Oxidized Graphene Anchored Ni(OH)2 Nanoflakes as Pseudocapacitor Materials for Ultrahigh Loading Electrode with High Areal Specific Capacitance. J. Phys. Chem. C 2014, 118, 24866–24876. [Google Scholar] [CrossRef]
- Justin, P.; Meher, S.K.; Rao, G.R. Tuning of Capacitance Behavior of NiO Using Anionic, Cationic, and Nonionic Surfactants by Hydrothermal Synthesis. J. Phys. Chem. C 2010, 114, 5203–5210. [Google Scholar] [CrossRef]
- Madhu, R.; Veeramani, V.; Chen, S.-M.; Veerakumar, P.; Liu, S.-B. Functional Porous Carbon/Nickel Oxide Nanocomposites as Binder-Free Electrodes for Supercapacitors. Chem. Eur. J. 2015, 21, 8200–8206. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, S.; Nagamuthu, S.; Muralidharan, G. Porous NiO/C Nanocomposites as Electrode Material for Electrochemical Supercapacitors. ACS Sustain. Chem. Eng. 2013, 1, 1110–1118. [Google Scholar] [CrossRef]
- Wu, Z.; Huang, X.-L.; Wang, Z.-L.; Xu, J.-J.; Wang, H.-G.; Zhang, X.-B. Electrostatic Induced Stretch Growth of Homogeneous β-Ni(OH)2 on Graphene with Enhanced High-Rate Cycling for Supercapacitors. Sci. Rep. 2015, 4, 3669. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, R.; Yan, X. Synergistic Effect between Ultra-Small Nickel Hydroxide Nanoparticles and Reduced Graphene Oxide sheets for the Application in High-Performance Asymmetric Supercapacitor. Sci. Rep. 2015, 5, 11095. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Tu, J.; Mai, Y.; Chen, R.; Wang, X.; Gu, C.; Zhao, X. Graphene Sheet/Porous NiO Hybrid Film for Supercapacitor Applications. Chem. Eur. J. 2011, 17, 10898–10905. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Yang, S. Nanocomposites of Ni(OH)2/Reduced Graphene Oxides with Controllable Composition, Size, and Morphology: Performance Variations as Pseudocapacitor Electrodes. ChemPlusChem 2012, 77, 807–816. [Google Scholar] [CrossRef]
- Xu, J.; Li, L.; He, F.; Lv, R.; Yang, P. A Novel double-shelled C@NiO hollow microsphere: Synthesis and application for electrochemical capacitor. Electrochim. Acta 2014, 148, 211–219. [Google Scholar] [CrossRef]
- Yi, H.; Wang, H.; Jing, Y.; Peng, T.; Wang, X. Asymmetric supercapacitors based on carbon nanotubes@NiO ultrathin nanosheets core-shell composites and MOF-derived porous carbon polyhedrons with super-long cycle life. J. Power Sources 2015, 285, 281–290. [Google Scholar] [CrossRef]
- Lee, E.J.; Bang, J.H. Mesoporous Ni(OH)2 tubes on carbon fiber paper for pseudocapacitor electrode. Mater. Lett. 2013, 105, 28–31. [Google Scholar] [CrossRef]
- Gogotsi, Y.; Simon, P. True Performance Metrics in Electrochemical Energy Storage. Science 2011, 334, 917–918. [Google Scholar] [CrossRef] [PubMed]
- Toupin, M.; Bélanger, D.; Hill, I.R.; Quinn, D. Performance of experimental carbon blacks in aqueous supercapacitors. J. Power Sources 2005, 140, 203–210. [Google Scholar] [CrossRef]
- Yan, S.; Meng, T.; Young, K.-H.; Nei, J. A Ni/MH Pouch Cell with High-Capacity Ni(OH)2. Batteries 2017, 3, 38. [Google Scholar] [CrossRef]
- Cericola, D.; Ruch, P.W.; Kötz, R.; Novák, P.; Wokaun, A. Characterization of bi-material electrodes for electrochemical hybrid energy storage devices. Electrochem. Commun. 2010, 12, 812–815. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brisse, A.-L.; Stevens, P.; Toussaint, G.; Crosnier, O.; Brousse, T. Ni(OH)2 and NiO Based Composites: Battery Type Electrode Materials for Hybrid Supercapacitor Devices. Materials 2018, 11, 1178. https://doi.org/10.3390/ma11071178
Brisse A-L, Stevens P, Toussaint G, Crosnier O, Brousse T. Ni(OH)2 and NiO Based Composites: Battery Type Electrode Materials for Hybrid Supercapacitor Devices. Materials. 2018; 11(7):1178. https://doi.org/10.3390/ma11071178
Chicago/Turabian StyleBrisse, Anne-Lise, Philippe Stevens, Gwenaëlle Toussaint, Olivier Crosnier, and Thierry Brousse. 2018. "Ni(OH)2 and NiO Based Composites: Battery Type Electrode Materials for Hybrid Supercapacitor Devices" Materials 11, no. 7: 1178. https://doi.org/10.3390/ma11071178
APA StyleBrisse, A.-L., Stevens, P., Toussaint, G., Crosnier, O., & Brousse, T. (2018). Ni(OH)2 and NiO Based Composites: Battery Type Electrode Materials for Hybrid Supercapacitor Devices. Materials, 11(7), 1178. https://doi.org/10.3390/ma11071178