Boron Nitride Nanosheets/PNIPAM Hydrogels with Improved Thermo-Responsive Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
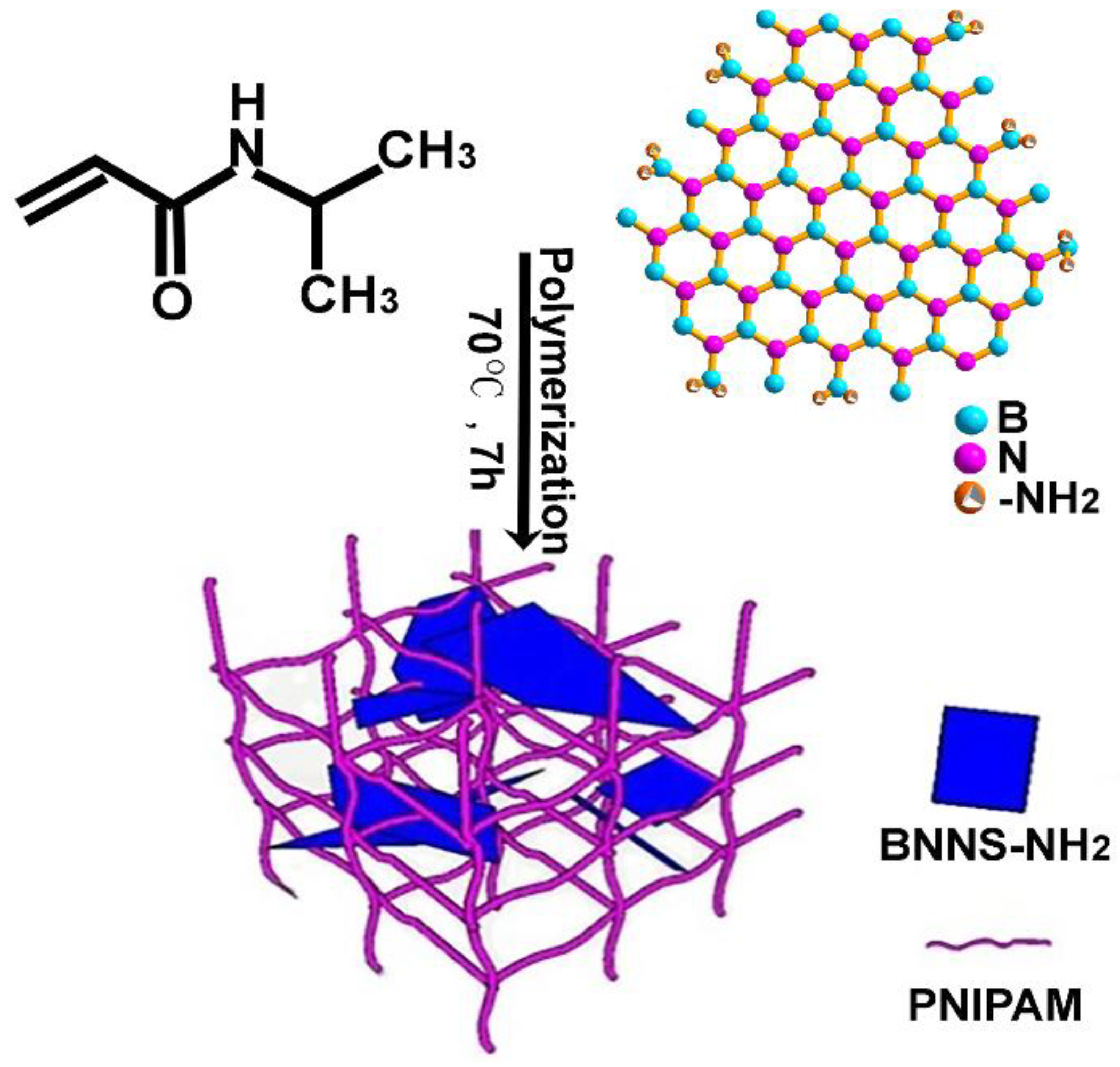
2.2. Synthesis of the PNIPAM Hydrogels Incorporated with BNNS-NH2
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chang, H.C.; Qin, J.L.; Xiao, P.S.; Yang, Y.; Zhang, T.F.; Ma, Y.F.; Huang, Y.; Chen, Y.S. Highly reversible and recyclable absorption under both hydrophobic and hydrophilic conditions using a reduced bulk graphene oxide material. Adv. Mater. 2016, 28, 3504–3509. [Google Scholar] [CrossRef] [PubMed]
- Cai, P.Q.; Leow, W.R.; Wang, X.Y.; Wu, Y.L.; Chen, X.D. Programmable nano-bio interfaces for functional biointegrated devices. Adv. Mater. 2017, 29, 1605529. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Li, Z.; Wang, X.; Zheng, Z. Facile functionalization of a tetrahedron-like PEG macromonomer-based fluorescent hydrogel with high strength and its heavy metal ion detection. J. Mater. Chem. A 2015, 3, 1158–1663. [Google Scholar] [CrossRef]
- White, E.M.; Yatvin, J.; Grubbs, J.B.; Bilbrey, J.A.; Locklin, J. Advances in smart materials: Stimuli-responsive hydrogel thin films. J. Polym. Sci. Part B Polym. Phys. 2013, 5, 1084–1099. [Google Scholar] [CrossRef]
- Kang, D.H.; King, S.M.; Lee, B.; Yoon, H.; Suh, K.Y. Stimuli-responsive hydrogel patterns for smart microfluidics and microarrays. Analyst 2013, 138, 6230–6242. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.X.; Liu, K.L.; Li, J. A Thermoresponsive hydrogel formed from a star-star supramolecular architecture. Angew. Chem. Int. Ed. 2013, 52, 6180–6184. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.N.; Liu, S.X.; Wang, H.M.; Tian, R.J. Synthesis and micellization of P(NIPAM-co-HMAM)-b-PEO-b-P(NIPAM-co-HMAM) triblock copolymers. Polym. Res. 2012, 19, 9989. [Google Scholar] [CrossRef]
- Stetsyshyn, Y.; Fornal, K.; Raczkowska, J.; Zemla, J.; Kostruba, A.; Ohar, H.; Ohar, M.; Donchak, V.; Harhay, K.; Awsiuk, K.; et al. Temperature and pH dual-responsive POEGMA-based coatings for protein adsorption. J. Colloid Interface Sci. 2013, 411, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.L.; Tang, D.Y.; Guo, Y.D. The fabrication and self-flocculation effect of hybrid TiO2 nanoparticles grafted with poly(N-isopropylacrylamide) at ambient temperature via surface-initiated atom transfer radical polymerization. J. Mater. Chem. 2012, 22, 16872–16879. [Google Scholar] [CrossRef]
- Huang, C.J.; Chang, F.C. Polypeptide diblock copolymers: Syntheses and properties of poly(N-isopropylacrylamide)-b-polylysine. Macromolecules 2008, 41, 7041–7052. [Google Scholar] [CrossRef]
- Lu, X.J.; Zhang, L.F.; Meng, L.Z.; Liu, Y.H. Synthesis of poly(N-isopropylacrylamide) by ATRP using a fluorescein-based initiator. Polym. Bull. 2007, 59, 195–206. [Google Scholar] [CrossRef]
- Lutz, J.F.; Akdemir, O.; Hoth, A. Point by point comparison of two thermosensitive polymers exhibiting a similar LCST: Is the age of poly(NIPAM) over? J. Am. Chem. Soc. 2006, 128, 13046–13047. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wu, D.Q.; Li, Q.; Chang, C.; Zhou, J.P.; Zhang, X.Z.; Zhuo, R.X. Preparation of shell cross-linked thermoresponsive micelles as well as hollow spheres based on P(NIPAAm-co-HMAAm-co-MPMA)-b-PCL. J. Phys. Chem. C 2008, 112, 15329–15334. [Google Scholar] [CrossRef]
- Lei, W.W.; Mochalin, V.N.; Liu, D.; Qin, S.; Gogotsi, Y.; Chen, Y. Boron nitride colloidal solutions, ultralight aerogels and freestanding membranes through one-step exfoliation and functionalization. Nat. Commun. 2015, 6, 8849. [Google Scholar] [CrossRef] [PubMed]
- Marsh, K.L.; Souliman, M.; Kaner, R.B. Co-solvent exfoliation and suspension of hexagonal boron nitride. Chem. Commun. 2015, 51, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Weng, Q.H.; Wang, B.J.; Wang, X.B.; Hanagata, N.; Li, X.; Liu, D.; Wang, X.; Jiang, X.F.; Bando, Y.; Golberg, D. Highly water-soluble, porous, and biocompatible boron nitrides for anticancer drug delivery. ACS Nano 2014, 8, 6123–6130. [Google Scholar] [CrossRef] [PubMed]
- Lei, W.W.; Zhang, H.; Wu, Y.; Zhang, B.; Liu, D.; Qin, S.; Liu, Z.W.; Liu, L.M.; Ma, Y.M.; Chen, Y. Oxygen-doped boron nitride nanosheets with excellent performance in hydrogen storage. Nano Energy 2014, 6, 219–224. [Google Scholar] [CrossRef]
- Chen, Z.G.; Zhou, J. Field emitters: Ultrathin BN nanosheets protruded from BN fibers. J. Mater. Chem. 2011, 21, 1191–1195. [Google Scholar] [CrossRef]
- Cong, H.P.; Qiu, J.H.; Yu, S.H. Thermoresponsive poly(N-isopropylacrylamide)/Graphene/Au nanocomposite hydrogel for water treatment by a laser-assisted approach. Small 2015, 11, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.Z.; Bao, H.Q.; Sahoo, N.G.; Wu, T.F.; Li, L. Water-soluble poly(N-isopropylacrylamide) and ash; graphene sheets synthesized via click chemistry for drug delivery. Adv. Funct. Mater. 2011, 21, 2754–2763. [Google Scholar] [CrossRef]
- Yang, K.; Feng, L.Z.; Shi, X.Z.; Liu, Z. Nano-graphene in biomedicine: Theranostic applications. Chem. Soc. Rev. 2013, 42, 530–547. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Du, P.; Xu, D.Y.; Li, Y.; Peng, W.C.; Zhang, G.L.; Zhang, F.B.; Fan, X.B. Near-infrared responsive MoS2/poly(N-isopropylacrylamide) hydrogels for remote light-controlled microvalves. Ind. Eng. Chem. Res. 2016, 55, 4526–4531. [Google Scholar] [CrossRef]
- Cui, Z.H.; Martinez, A.P.; Adamson, D.H. PMMA functionalized boron nitride sheets as nanofillers. Nanoscale 2015, 7, 10193–10197. [Google Scholar] [CrossRef] [PubMed]
- Lei, W.W.; Portehault, D.; Liu, D.; Qin, S.; Chen, Y. Porous boron nitride nanosheets for effective water cleaning. Nat. Commun. 2013, 4, 1777. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Sina, N.; Gilberto, C.; Majharul, H.K.; Tomas, K.; Lei, J.; Liu, H.K.; Li, H.J.; Huang, Z.G. Edge-hydroxylated boron nitride nanosheets as an effective additive to improve the thermal response of hydrogels. Adv. Mater. 2015, 27, 7196–7203. [Google Scholar]
- Wu, X.; Liu, H.; Tang, Z.H.; Guo, B.C. Scalable fabrication of thermally conductive elastomer/boron nitride nanosheets composites by slurry compounding. Compos. Sci. Technol. 2016, 123, 179–186. [Google Scholar] [CrossRef]
- Wang, X.B.; Zhi, C.Y.; Weng, Q.H.; Bando, Y.; Golberg, D. Boron nitride nanosheets: Novel syntheses and applications in polymeric composites. J. Phys. Conf. Ser. 2013, 471, 012003. [Google Scholar] [CrossRef]
- Liu, D.; Lei, W.W.; Klika, K.D.; Kong, L.X.; Chen, Y. Multifunctional polymer/porous boron nitride nanosheet membranes for superior trapping emulsified oils and organic molecules. Adv. Mater. Interfaces 2015, 2, 1500228. [Google Scholar] [CrossRef]
- Lee, D.J.; Lee, B.; Park, K.H.; Ryu, H.J.; Jeon, S.; Hong, S.H. Scalable exfoliation process for highly soluble boron nitride nanoplatelets by hydroxide-assisted ball milling. Nano Lett. 2015, 15, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.P.; Xue, S.S.; Yang, H.; Zhang, H.Y.; Zhang, T.; Gou, S.H. Polymerization-induced phase separation for the fabrication of magnetic sponges for oil spill reclamation. Chem. Eng. J. 2017, 328, 639–644. [Google Scholar] [CrossRef]
- Mo, J.; Xu, N.; Xiao, C.; Cheng, B.; Han, X. Polymethacrylate-based fiber containing an interpenetrating polymer network and its use for oil absorption. Fibers Polym. 2015, 16, 8–16. [Google Scholar] [CrossRef]





© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, S.; Wu, Y.; Wang, J.; Guo, M.; Liu, D.; Lei, W. Boron Nitride Nanosheets/PNIPAM Hydrogels with Improved Thermo-Responsive Performance. Materials 2018, 11, 1069. https://doi.org/10.3390/ma11071069
Xue S, Wu Y, Wang J, Guo M, Liu D, Lei W. Boron Nitride Nanosheets/PNIPAM Hydrogels with Improved Thermo-Responsive Performance. Materials. 2018; 11(7):1069. https://doi.org/10.3390/ma11071069
Chicago/Turabian StyleXue, Shishan, Yuanpeng Wu, Jiemin Wang, Meiling Guo, Dan Liu, and Weiwei Lei. 2018. "Boron Nitride Nanosheets/PNIPAM Hydrogels with Improved Thermo-Responsive Performance" Materials 11, no. 7: 1069. https://doi.org/10.3390/ma11071069
APA StyleXue, S., Wu, Y., Wang, J., Guo, M., Liu, D., & Lei, W. (2018). Boron Nitride Nanosheets/PNIPAM Hydrogels with Improved Thermo-Responsive Performance. Materials, 11(7), 1069. https://doi.org/10.3390/ma11071069



