Study on the Synergetic Fire-Retardant Effect of Nano-Sb2O3 in PBT Matrix
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of Nanocomposites
2.3. Characterization
3. Result and Discussion
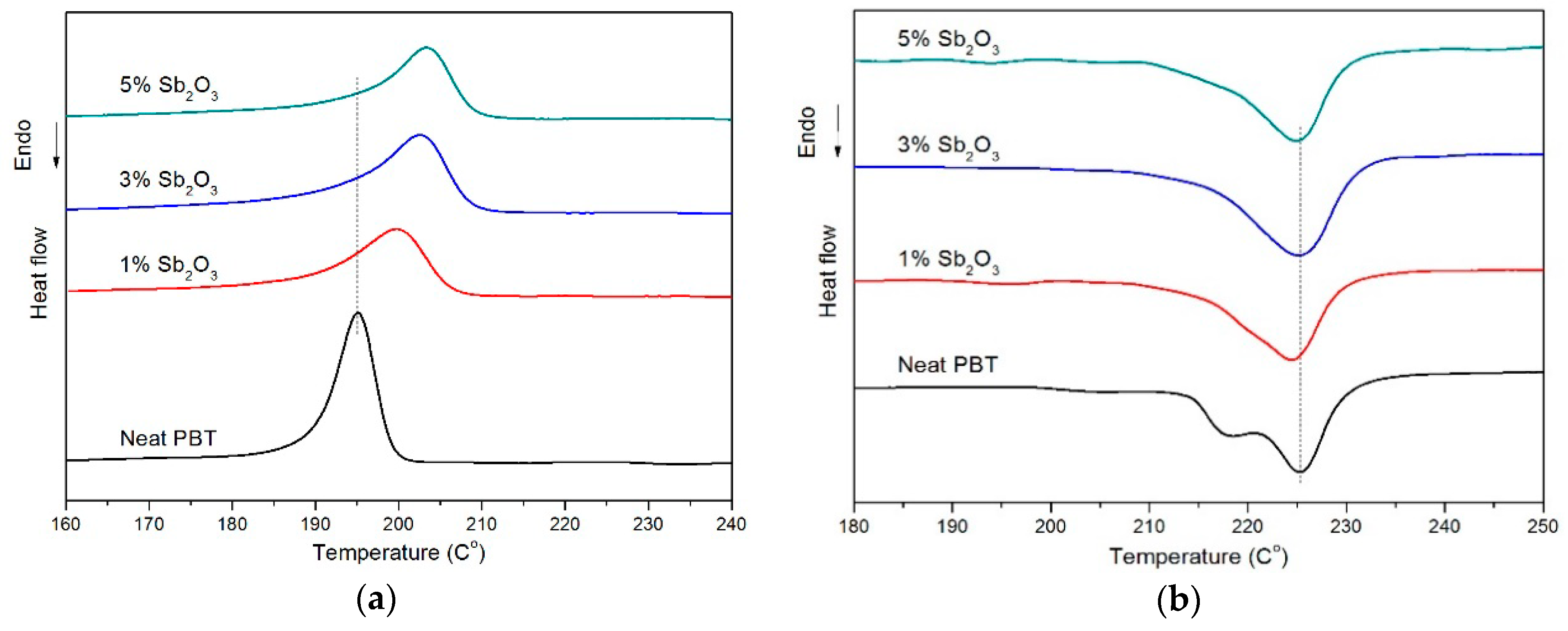
3.1. Crystallization and Melting Behavior
3.2. Mechanical Properties
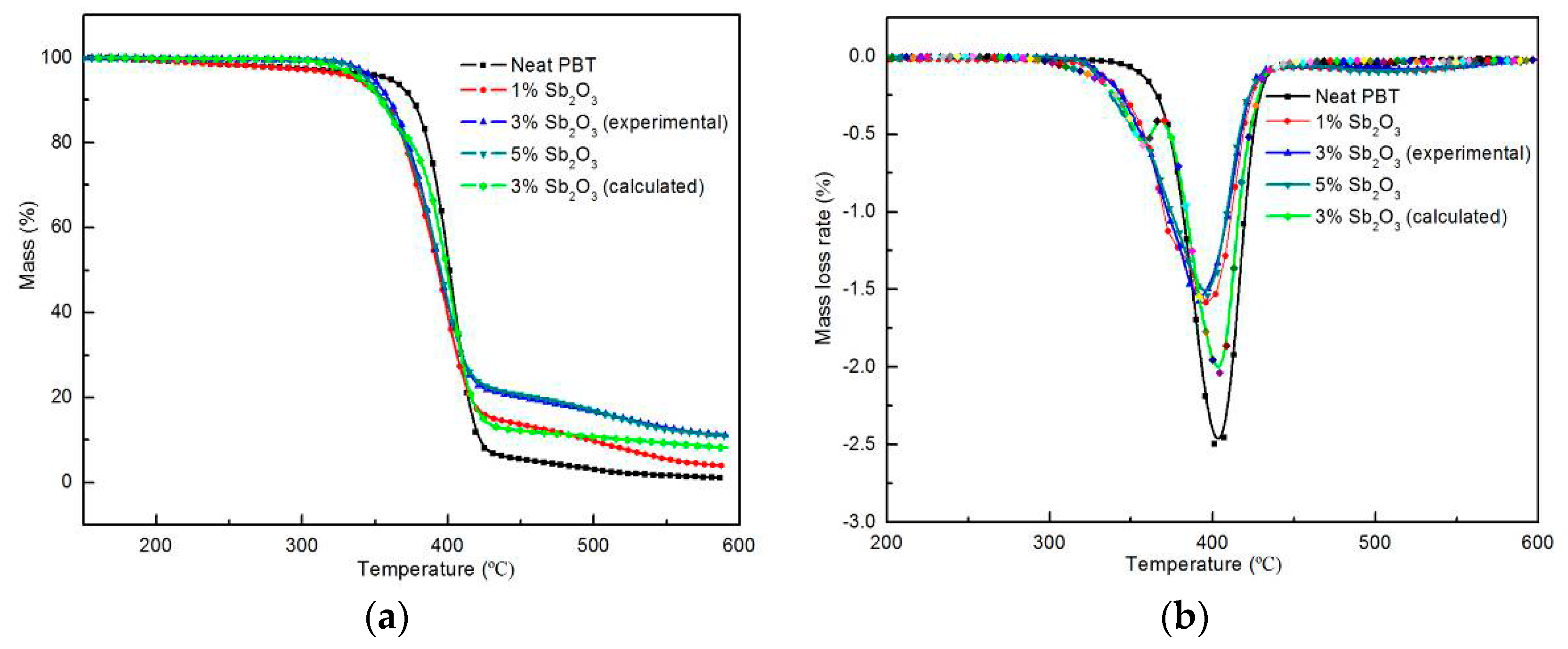
3.3. Thermal Decomposition Behaviors
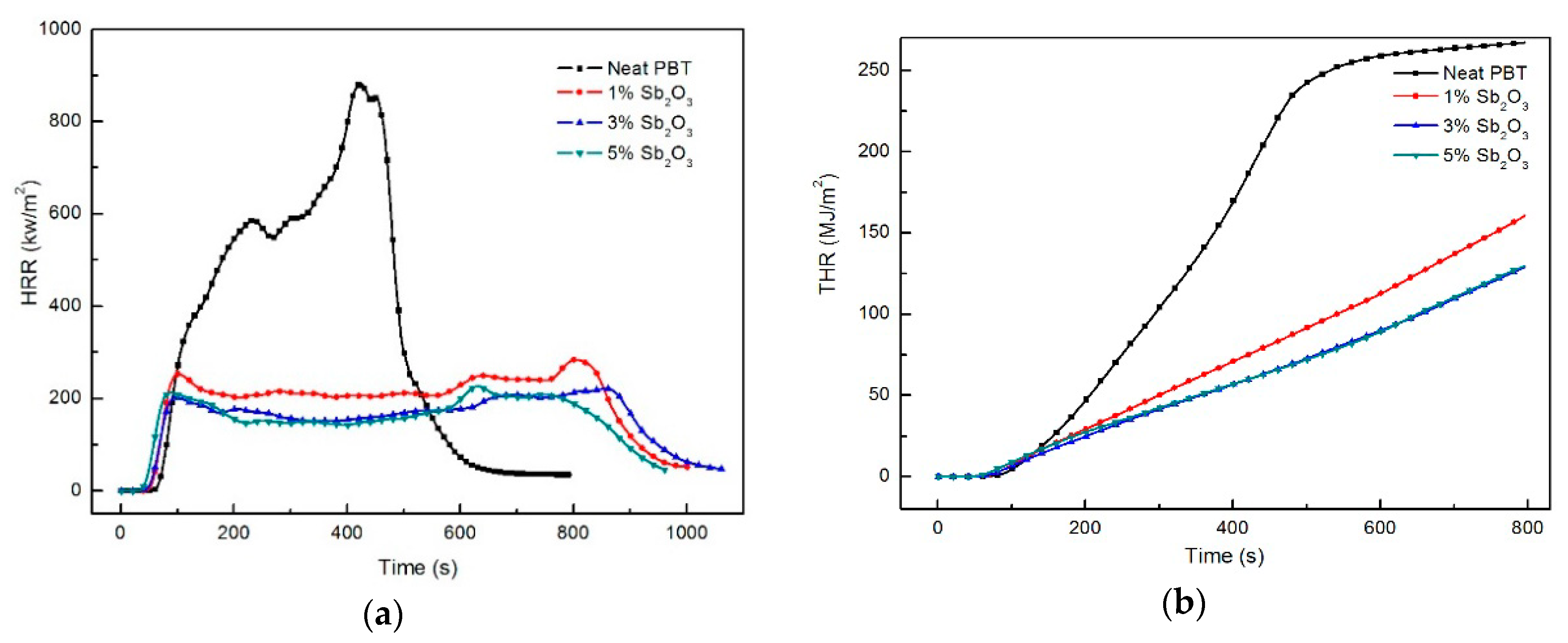
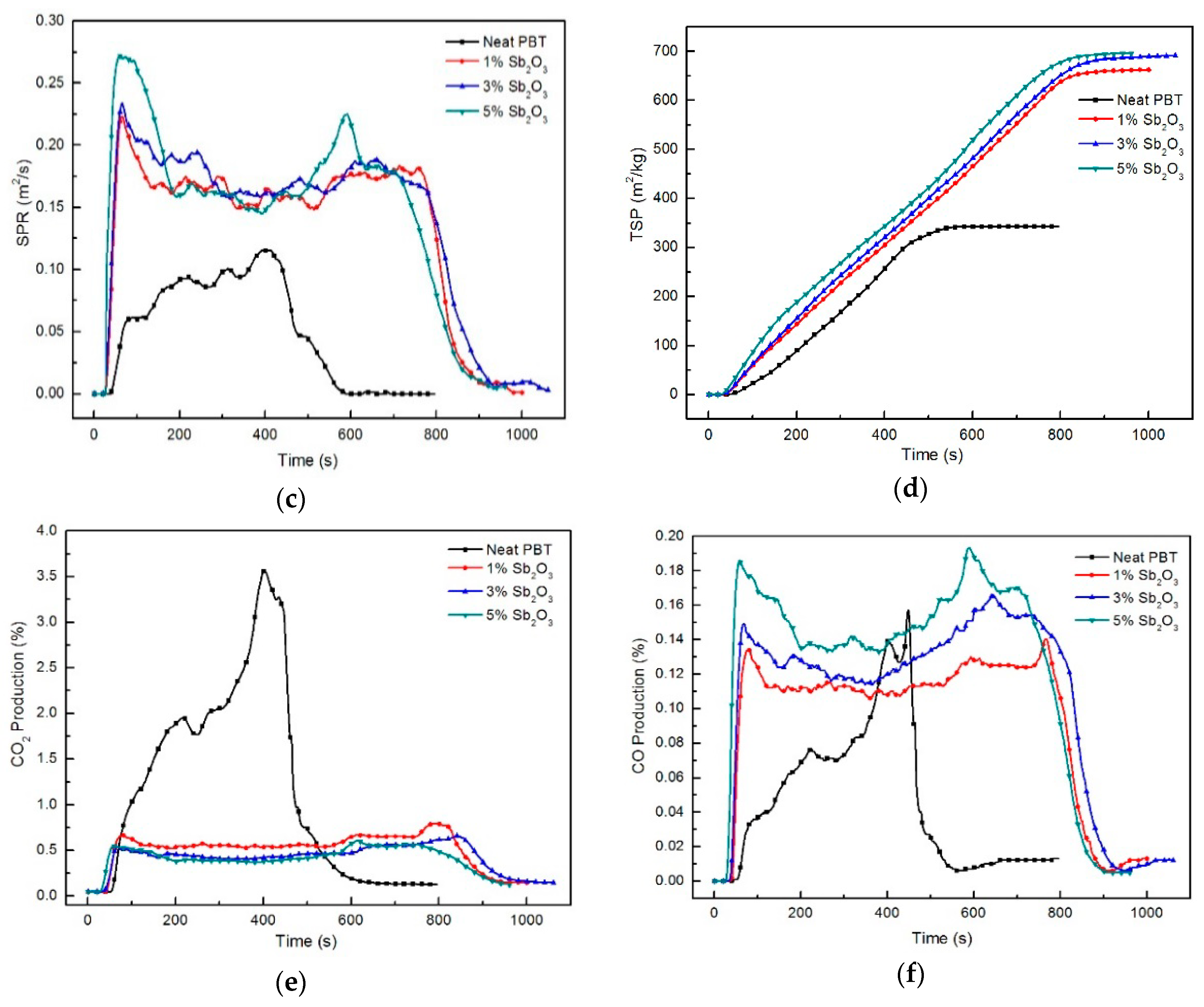
3.4. Fire Behaviour: Forced Flaming Combustion (Conecalorimeter)
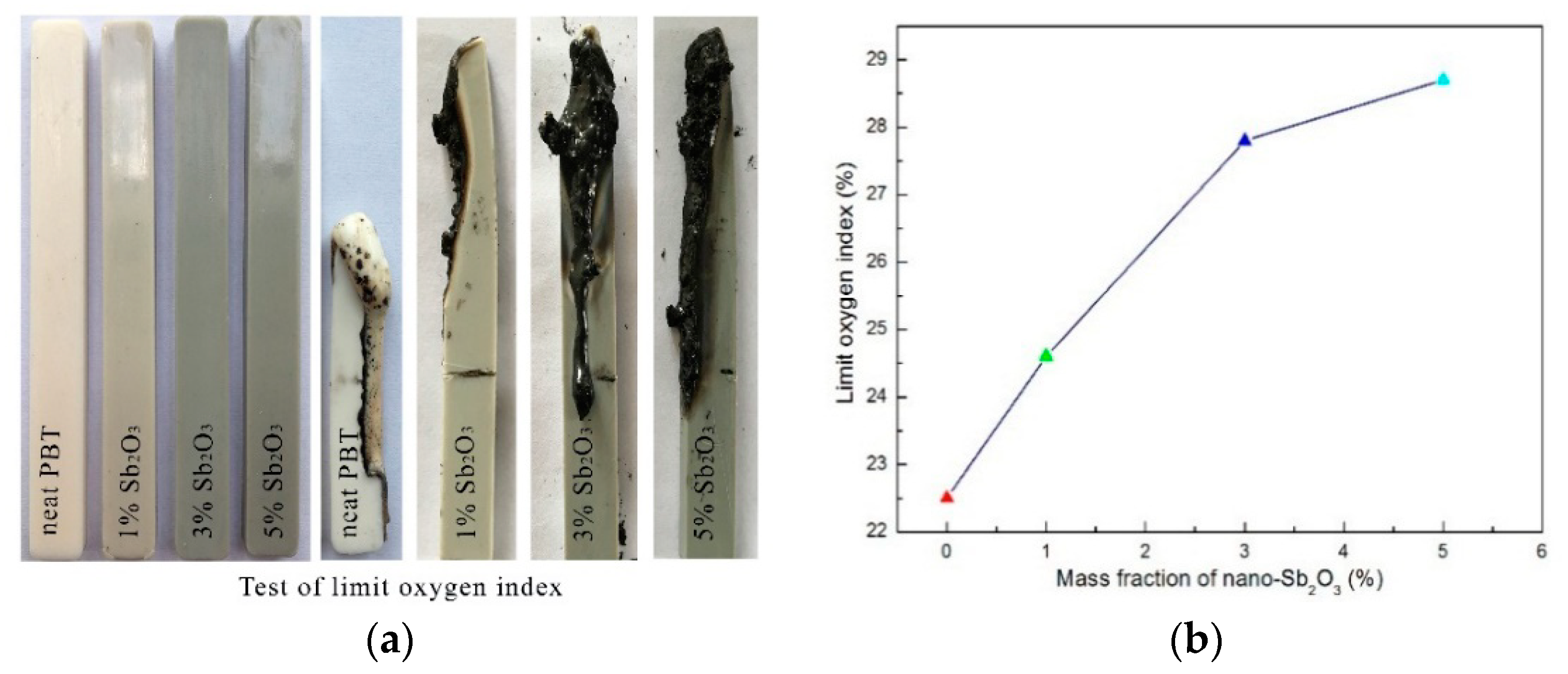
3.5. Flammability
3.6. Discussion of the Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yang, L.; Chen, H.; Jia, S.; Lu, X.; Huang, J. Influences of ethylene-butylacrylate-glycidyl methacrylate on morphology and mechanical properties of poly(butylene erephthalate)/polyolefin elastomer blends. J. Appl. Polym. Sci. 2014, 131, 1107–1117. [Google Scholar] [CrossRef]
- Zhang, D.H.; He, M.; He, W.D.; Zhou, Y.Q.; Shu, H.; Yu, J. Influence of thermo-oxidative ageing on the thermal and dynamical mechanical properties of long glass fibre-reinforced poly(butylene terephthalate) composites filled with DOPO. Materials 2017, 10, 500. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.L.; Hu, Y.; Chen, X.F.; Shi, Y.Q.; Niu, Y.; Zhang, Y.; He, S.; Dai, H.M. Dual modification of graphene by polymeric flame retardant and Ni(OH)2 nanosheets for improving flame retardancy of polypropylene. Compos. Part A 2017, 100, 106–117. [Google Scholar] [CrossRef]
- Qiu, S.L.; Wang, X.; Yu, B.; Feng, X.M.; Mu, X.W.; Richard, K.K.; Hu, Y. Flame-retardant-wrapped polyphosphazene nanotubes: A novelstrategy for enhancing the flame retardancy and smoke toxicity suppression of epoxy resins. J. Hazard. Mater. 2017, 325, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Niroumand, J.S.; Peighambardoust, S.J.; Shenavar, A. Polystyrene-based composites and nanocomposites with reduced brominated-flame retardant. Iran. Polym. J. 2016, 25, 607–614. [Google Scholar] [CrossRef]
- Samyn, F.; Bourbigo, S.; Jama, C.; Bellayer, S.; Nazare, S.; Hull, R.; Fina, A.; Castrovinci, A.; Camino, G. haracterisation of the dispersion in polymer flame retarded nanocomposites. Eur. Polym. J. 2008, 44, 1631–1641. [Google Scholar] [CrossRef]
- Bakirtzis, D.; Ramani, A.; Delichatsios, M.A.; Zhang, J. Structure of the condensed phase and char of fire-retarded PBT nanocomposites by TGA/ATR in N2. Fire Saf. J. 2009, 44, 1023–1029. [Google Scholar] [CrossRef]
- Matzen, M.; Kandola, B.; Huth, C.; Schartel, B. Influence of Flame Retardants on the Melt Dripping Behaviour of Thermoplastic Polymers. Materials 2015, 8, 5621–5646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grause, G.; Ishibashi, J.; Kameda, T.; Bhaskar, T.; Yoshioka, T. Kinetic studies of the decomposition of flame retardant containing high-impact polystyrene. Polym. Degrad. Stab. 2010, 95, 1129–1137. [Google Scholar] [CrossRef]
- Yang, W.; Kan, Y.C.; Song, L.; Richard, K. Effect of organo-modified montmorillonite on flame retardant poly(1,4-butyleneterephthalate) composites. Polym. Adv. Technol. 2011, 22, 2564–2570. [Google Scholar] [CrossRef]
- Gallo, E.; Braun, U.; Schartel, B.; Russo, P.; Aciernoa, D. Halogen-free flame retardedpoly(butylenes terephthalate) (PBT) using metal oxides/PBT nanocompositesin combination with aluminium phosphinate. Polym. Degrad. Stab. 2009, 94, 1245–1253. [Google Scholar] [CrossRef]
- Zhu, S.E.; Wang, L.L.; Chen, H.; Yang, W.; Yue, A.C.; Chen, T.B.; Luo, C.; Bin, W.M. Comparative Studies on Thermal, Mechanical, and Flame Retardant Properties of PBT Nanocomposites via Different Oxidation State Phosphorus-Containing Agents Modified Amino-CNTs. Nanomaterials 2018, 8, 70. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.L.; Zhou, S.G.; Niu, L.; Wen, C. Effect of Sb2O3 Modified by Various Surface Active Agents on Flame Retardant Properties of PVC Composite. J. Mater. Eng. 2016, 44, 64–69. [Google Scholar] [CrossRef]
- Xu, C.H.; Shi, S.Q.; Surya, C.; Woo, C.H. Synthesis of antimony oxide nano-particles by vapor transport and condensation. J. Mater. Sci. 2007, 42, 9855–9858. [Google Scholar] [CrossRef]
- Xu, J.L.; Zhang, L.; Guo, Q.; Feng, C. Research on the preparation of antimony nanoparticles by mechanical ball milling. Key Eng. Mater. 2014, 609, 244–249. [Google Scholar] [CrossRef]
- Yang, W.H.; Xu, J.L.; Niu, L.; Kang, C.H.; Ma, B.X. Analysis of agglomeration and interfacial properties in PBT/nano-Sb2O3 composites. J. Adhes. Sci. Technol. 2017, 32, 739–752. [Google Scholar] [CrossRef]
- Si, M.M.; Feng, J.; Hao, J.W.; Xu, L.S.; Du, J.X. Synergistic flame retardant effects and mechanisms of nano-Sb2O3 in combination with aluminum phosphinate in poly(ethylene terephthalate). Polym. Degrad. Stab. 2014, 100, 70–78. [Google Scholar] [CrossRef]
- Huang, G.B.; Song, P.A.; Liu, L.N. Fabrication of multifunctional graphene decorated with bromine and nano-Sb2O3 towards high-performance polymer nanocomposites. Carbon 2016, 98, 689–701. [Google Scholar] [CrossRef]
- Mirdamadian, Z.; Ghanbari, D. Synergistic Effect between Sb2O3 Nanoparticles–Trichloromelamine and Carbon Nanotube on the Flame Retardancy and Thermal Stability of the Cellulose Acetate. J. Clust. Sci. 2014, 25, 925–936. [Google Scholar] [CrossRef]
- Yang, W.H.; Xu, J.L.; Niu, L.; Kang, C.H.; Ma, B.X. Effects of high energy ball milling on mechanicaland interfacial properties of PBT/nano-Sb2O3 composites. J. Adhes. Sci. Technol. 2018, 32, 291–301. [Google Scholar] [CrossRef]
- Plastics-Determination of Burning Behaviour by Oxygen Index-Part 2: Ambient-Temperature Test; GB/T 2406.2-2009; Standardization Administration of the People’s Republic of China: Beijing, China, 2009.
- Reaction-to-Fire Test-Heat Release, Smoke Production and Mass Loss Rate; BS ISO 5660 standard; BSI Standards Publication: London, UK, 2015.
- Chen, S.J.; Jin, J.; Zhang, J. Non-isothermal crystallization behaviors of poly(4-methyl-pentene-1). J. Therm. Anal. Calorim. 2011, 103, 229–236. [Google Scholar] [CrossRef]
- Yang, W.; Zhou, H.; Yang, B.H.; Lu, H.D.; Song, L.; Hu, Y. Facile preparation of modified carbon nanotube-reinforced PBT nanocomposites with enhanced thermal, flame retardancy, and mechanical properties. Polym. Compos. 2016, 37, 1812–1820. [Google Scholar] [CrossRef]
- Huang, T.; Li, J.L.; Yang, J.H.; Zhang, N.; Wang, Y.; Zhou, Z.W. Carbon nanotubes induced microstructure and property changes ofpolycarbonate/poly(butylene terephthalate) blend. Compos. Part B 2018, 133, 177–184. [Google Scholar] [CrossRef]
- Yin, H.J.; Dittrich, B.; Farooq, M.; Schartel, B. Carbon-based nanofillers/Poly(butylene terephthalate): Thermal, dielectric, electrical and rheological properties. J. Polym. Res. 2015, 22, 140. [Google Scholar] [CrossRef]
- Righetti, M.C.; Di Lorenzo, M.L. Melting process of poly (butylene terephthalate) analyzed by temperature-modulated differential scanning calorimetry. J. Polym. Sci. Part B 2004, 42, 2191–2201. [Google Scholar] [CrossRef]
- Wu, D.F.; Wu, L.C.; Yu, G.C.; Xu, B.; Zhang, M. Crystallization and thermal behavior of multiwalled carbon nanotube/poly(butylenes terephthalate) composites. Polym. Eng. Sci. 2008, 48, 1057–1067. [Google Scholar] [CrossRef]
- Deshmukh, G.S.; Peshwe, D.R.; Pathak, S.U.; Ekhe, J. A study on effect of mineral additions on the mechanical, thermal, and structural properties of poly(butylene terephthalate) (PBT) composites. J. Polym. Res. 2011, 18, 1081–1090. [Google Scholar] [CrossRef]
- Abe, H. Thermal Degradation of Environmentally Degradable Poly(hydroxyalkanoic acid)s. Macromol. Biosci. 2006, 6, 469–486. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.D.; Wilkie, C. Fire performance of flame retardant polypropylene and polystyrene composites screened with microscale combustion calorimetry. Polym. Adv. Technol. 2011, 22, 14–21. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Q.G.; Zhou, K.Q.; Yang, W.; Hu, Y.; Gong, X.L. The influence of manganese–cobalt oxide/graphene on reducing firehazards of poly(butylene terephthalate). J. Hazard. Mater. 2014, 278, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Nazare, S.; Kandola, B.K.; Horrocks, A.R. Smoke, CO, and CO2 Measurements and Evaluationusing Different Fire Testing Techniques for Flame Retardant Unsaturated Polyester Resin Formulations. J. Fire Sci. 2008, 26, 215–242. [Google Scholar] [CrossRef]
- Guo, X.; Wang, H.; Ma, D.L.; He, J.N.; Lei, Z.Q. Synthesis of a novel, multifunctional inorganic curing agent and its effect on the flame-retardant and mechanical properties of intrinsically flame retardant epoxy resin. J. Appl. Polym. Sci. 2018, 135, 46410. [Google Scholar] [CrossRef]
- Zhang, J.L.; Li, G.Y.; Wu, Q.L.; Li, M.C.; Sun, X.X.; Ring, D. Synergistic influence of halogenated flame retardants and nanoclay on flame performance of high density polyethylene and wood flour composites. RSC Adv. 2017, 40, 24895–24902. [Google Scholar] [CrossRef]
- Babushok, V.; Deglmann, P.; Krämer, R.; Linteris, G.T. Influence of Antimony-Halogen Additives on Flame Propagation. Combust. Sci. Technol. 2017, 189, 290–311. [Google Scholar] [CrossRef] [PubMed]
- Gallo, E.; Schartel, B.; Acierno, D.; Russo, P. Flame retardant biocomposites: Synergism between phosphinate and nanometric metal oxides. Eur. Polym. J. 2011, 47, 1390–1401. [Google Scholar] [CrossRef]


| Sample No. | PBT | BEO | Nano-Sb2O3 |
|---|---|---|---|
| Neat PBT | 100 | 0 | 0 |
| PBT/BEO/nano-Sb2O3 1% | 83 | 16 | 1 |
| PBT/BEO/nano-Sb2O3 3% | 81 | 16 | 3 |
| PBT/BEO/nano-Sb2O3 5% | 79 | 16 | 5 |
| Sample No. | Tonset (°C) | Tc (°C) | Tonset-Tc/°C | ∆Hm (J·g−1) | NE (%) | Xc (%) |
|---|---|---|---|---|---|---|
| Neat PBT | 204.3 | 195.2 | 9.1 | 31.6 | 0 | 22.6 |
| PBT/BEO/nano-Sb2O3 1% | 205.9 | 199.7 | 6.2 | 31.9 | 49.5 | 27.5 |
| PBT/BEO/nano-Sb2O3 3% | 211.1 | 202.6 | 8.5 | 34.7 | 81.3 | 30.6 |
| PBT/BEO/nano-Sb2O3 5% | 213.1 | 203.4 | 9.7 | 29.6 | 90.1 | 26.8 |
| Sample No. | Tensile Strength (MPa) | Young’s Modulus (GPa) |
|---|---|---|
| Neat PBT | 54.6 ± 0.5 | 1.8 ± 0.1 |
| PBT/BEO/nano-Sb2O3 1% | 61.1 ± 0.8 | 2.1 ± 0.1 |
| PBT/BEO/nano-Sb2O3 3% | 61.7 ± 0.7 | 2.3 ± 0.1 |
| PBT/BEO/nano-Sb2O3 5% | 58.5 ± 0.7 | 2.2 ± 0.1 |
| Sample No. | T10% (°C) | TPeak% (°C) | Char at 600 °C (%) |
|---|---|---|---|
| Neat PBT | 372 | 407 | 1.1 |
| PBT/BEO/nano-Sb2O3 1% | 355 | 396 | 2.9 |
| PBT/BEO/nano-Sb2O3 3% (experimental) | 357 | 391 | 11.8 |
| PBT/BEO/nano-Sb2O3 3% (calculated) | 353 | 401 | 8.0 |
| PBT/BEO/nano-Sb2O3 5% | 356 | 397 | 10.1 |
| Sample No. | TTI (s) | HRR (kW/m2) | PHRR (kW/m2) | THR (MJ/m2) | PSPR (m2/s) | TSP (m2/kg) | PCO2P (kg/kg) | PCOP (kg/kg) | Residue (wt %) |
|---|---|---|---|---|---|---|---|---|---|
| Error | ±2 | ±10 | ±10 | ±1 | ±0.01 | ±30 | ±0.01 | ±0.002 | ±0.5 |
| Neat PBT | 36 | 375.1 | 917.5 (451 s) a | 265.6 | 0.073 | 342 | 18.91 | 1.058 | 3.6 |
| PBT/BEO/nano-Sb2O3 1% | 25 | 203.2 | 289.0 (789 s) a | 189.4 | 0.210 | 662 | 6.48 | 0.683 | 7.7 |
| PBT/BEO/nano-Sb2O3 3% | 22 | 161.1 | 224.5 (841 s) a | 154.2 | 0.241 | 691 | 6.06 | 0.965 | 11.3 |
| PBT/BEO/nano-Sb2O3 5% | 20 | 159.4 | 230.6 (616 s) a | 149.5 | 0.330 | 698 | 4.41 | 0.951 | 10.5 |
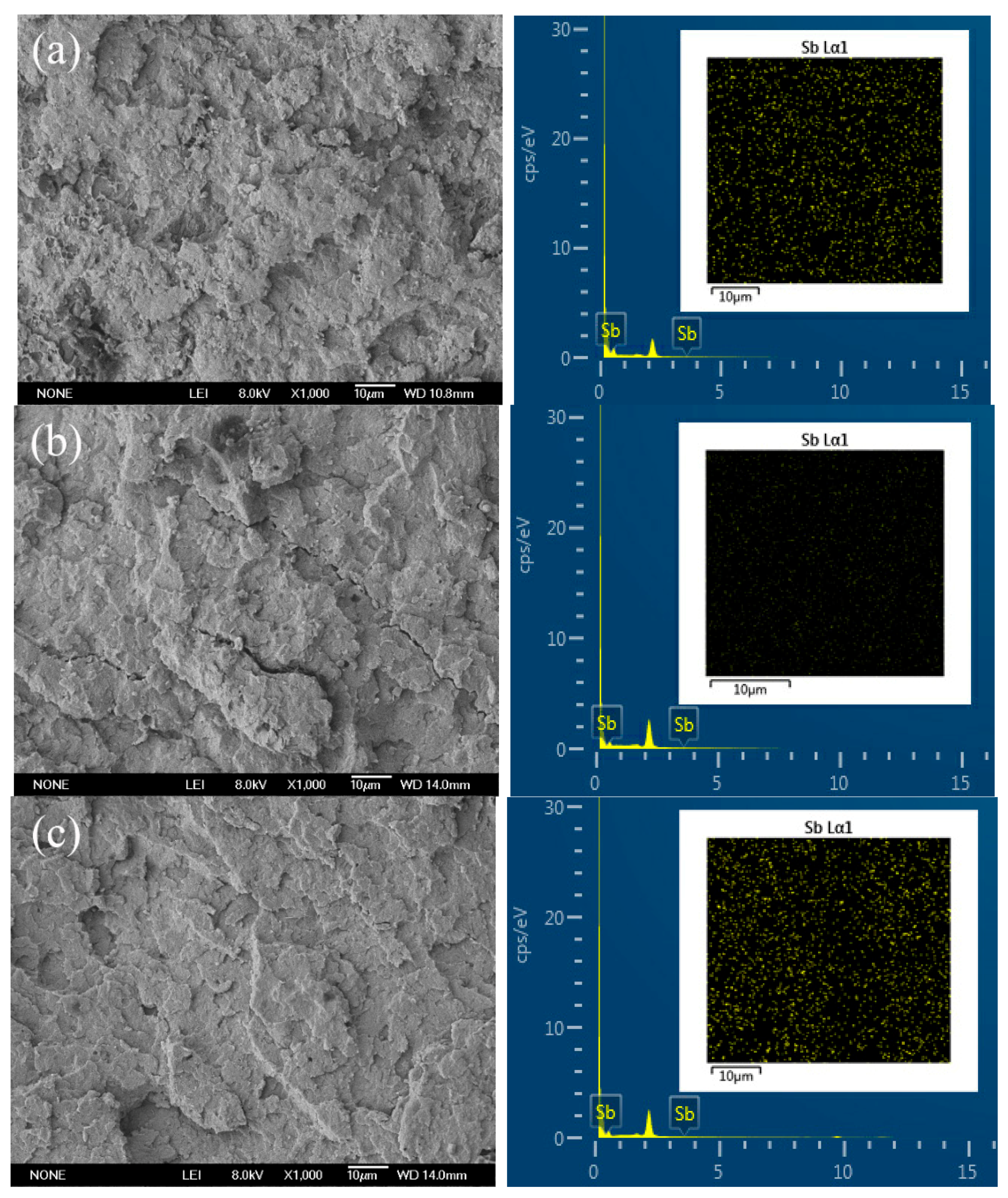
| Sample No. | Element Content (wt %) | ||
|---|---|---|---|
| C | O | Sb | |
| Neat PBT | 95.69 | 4.31 | 0 |
| PBT/BEO/Sb2O3 1% | 95.91 | 4.09 | 0 |
| PBT/BEO/Sb2O3 3% | 100 | 0 | 0 |
| PBT/BEO/Sb2O3 5% | 100 | 0 | 0 |
| Sample | LOI (%) | UL94, 4.0 mm Bar | ||
|---|---|---|---|---|
| t1/t2 a (s) | Dripping | Rating | ||
| Neat PBT | 21.8 ± 1 | BC b | Yes | NR c |
| PBT/BEO/nano-Sb2O3 1% | 24.6 ± 1 | 11.7/13.8 | No | V-1 |
| PBT/BEO/nano-Sb2O33% | 27.8 ± 1 | 5.1/7.6 | No | V-0 |
| PBT/BEO/nano-Sb2O3 5% | 28.7 ± 1 | 3.8/5.6 | No | V-0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, L.; Xu, J.; Yang, W.; Ma, J.; Zhao, J.; Kang, C.; Su, J. Study on the Synergetic Fire-Retardant Effect of Nano-Sb2O3 in PBT Matrix. Materials 2018, 11, 1060. https://doi.org/10.3390/ma11071060
Niu L, Xu J, Yang W, Ma J, Zhao J, Kang C, Su J. Study on the Synergetic Fire-Retardant Effect of Nano-Sb2O3 in PBT Matrix. Materials. 2018; 11(7):1060. https://doi.org/10.3390/ma11071060
Chicago/Turabian StyleNiu, Lei, Jianlin Xu, Wenlong Yang, Jiqiang Ma, Jinqiang Zhao, Chenghu Kang, and Jiaqiang Su. 2018. "Study on the Synergetic Fire-Retardant Effect of Nano-Sb2O3 in PBT Matrix" Materials 11, no. 7: 1060. https://doi.org/10.3390/ma11071060
APA StyleNiu, L., Xu, J., Yang, W., Ma, J., Zhao, J., Kang, C., & Su, J. (2018). Study on the Synergetic Fire-Retardant Effect of Nano-Sb2O3 in PBT Matrix. Materials, 11(7), 1060. https://doi.org/10.3390/ma11071060




