Environmental Screening of Electrode Materials for a Rechargeable Aluminum Battery with an AlCl3/EMIMCl Electrolyte
Abstract
1. Introduction
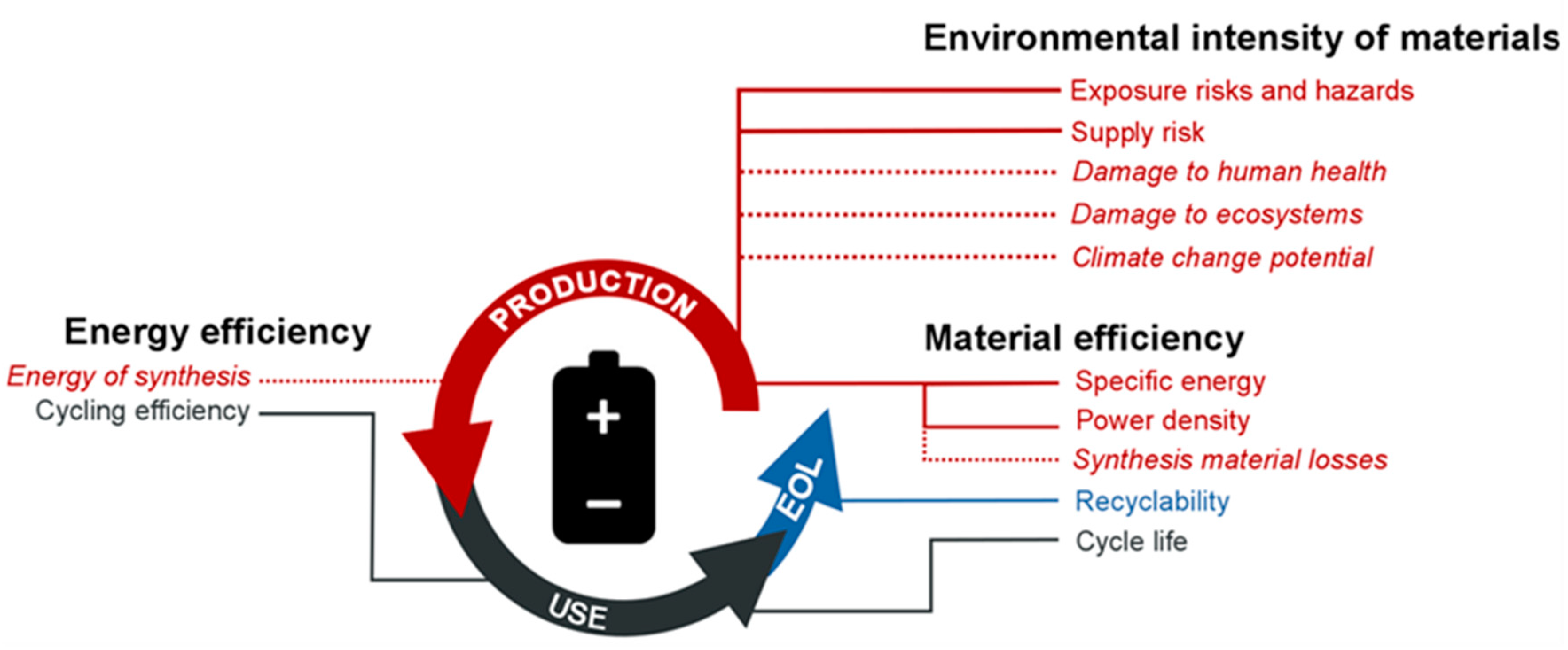
2. Materials and Methods
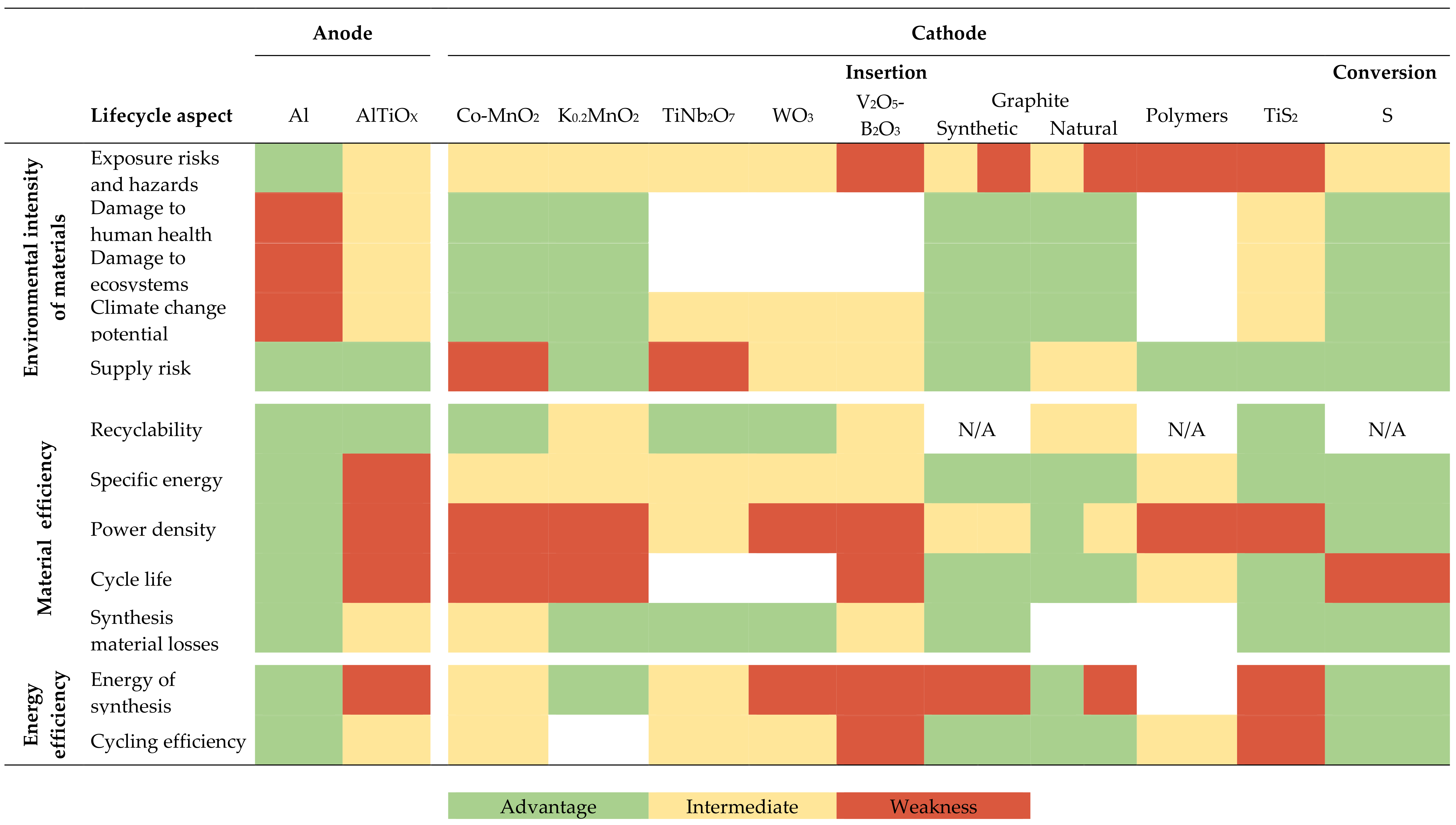
3. Results
3.1. Anode Materials
3.1.1. Pure Aluminum
3.1.2. Aluminum-Titanate Oxide Insertion Material
3.2. Cathode Materials
3.2.1. Manganese Oxides
3.2.2. Other Transition Metal Oxides
3.2.3. Graphitic Materials
3.2.4. Conductive Polymers
3.2.5. Titanium Sulfide
3.2.6. Sulfur
4. Discussion
4.1. Discussion on Aluminium Battery Electrode Materials
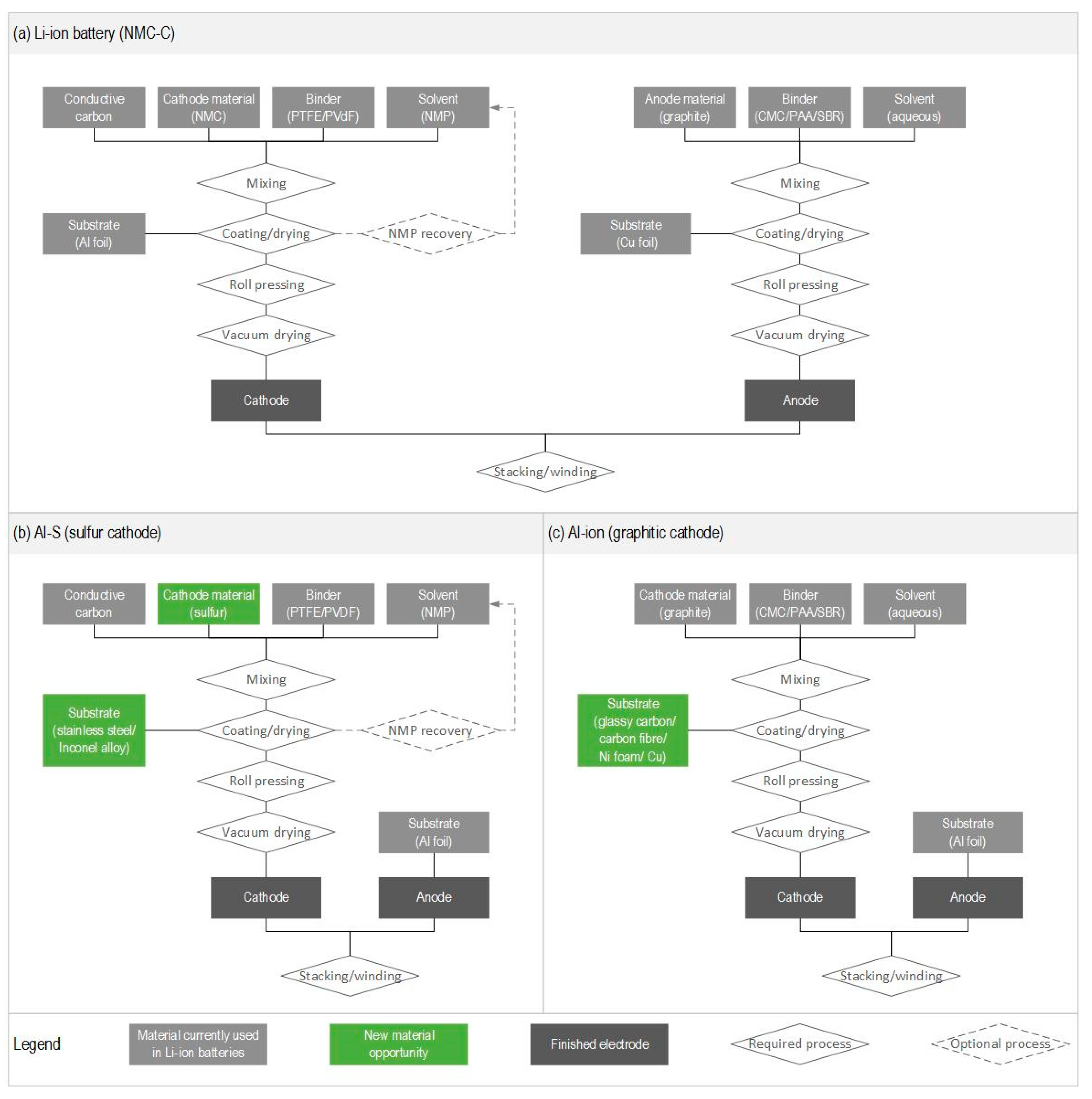
4.2. Comparison with Li-Ion Batteries
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. General Considerations for the Environmental Screening
Appendix A.2. Exposure Risks and Hazards
| Green | Yellow | Red |
|---|---|---|
| Aluminium | Aluminium titanium oxide | Cobalt |
| PVDF | Aluminium oxide nanopowder | Graphene oxide |
| Boron oxide | Potassium | |
| Carbon foam | Pyrrole | |
| Manganese dioxide | Thiophene | |
| Niobium oxide nanopowder | Titanate sulphide | |
| Pyrolytic graphite | Vanadium pentoxide | |
| Titanium oxide nanopowder | ||
| Tungsten trioxide nanopowder | ||
| Sulphur |
Appendix A.3. Potential Damage to Human Health, Ecosystems, and Climate from Material Production
Appendix A.4. Supply Risk
Appendix A.5. Specific Energy, Power Density, Cycle Life, and Cycling Efficiency
| Specific Energy (mWh·g−1) | Specific Power (W·g−1) | Cycle Life and Cycling Efficiency |
|---|---|---|
| Capacity (mA·h·g−1) | Capacity (mA·h·g−1) | Cycle number |
| Discharge voltage (V) | Specific current (mA·g−1) | Capacity loss |
| Discharge voltage (V) | Columbic efficiency | |
| Voltage hysteresis |
Appendix A.6. Recyclability
Appendix A.7. Electrode Material Synthesis
Appendix A.7.1. Materials Synthesized within the ALION Project
Appendix A.7.2. Materials Synthesized in Previous Studies
Appendix A.7.3. Materials Ready for Use
Appendix A.7.4. Synthesis Material Losses
Appendix A.7.5. Energy of Synthesis
| Green | Yellow | Red |
|---|---|---|
| Dry ball milling | Annealing (450 °C for 2 h and 600 °C for 2 h) | Annealing (2850 °C for 30 min) |
| Dry mixing of constituent materials | Calcination (750 °C for 1 h) | Ceramic sintering (1100 °C for 4 h) |
| Hydrothermal | Jet milling (75 min) | Chemical vapor deposition |
| Foil immersion in aqueous solution at 60 °C | Ceramic sintering (800 °C for 2 h) | Solid state with thermal treatment (450 °C for 24 h and 600 °C for 3 days) |
| Micro bead milling | Flame spray pyrolysis | |
| Modified Hummer’s method | ||
| Spray drying | ||
| Wet ball milling |
References
- Bruckner, T.; Bashmakov, I.A.; Mulugetta, Y.; Chum, H.; de la Vega Navarro, A.; Edmonds, J.; Faaij, A.; Fungtammasan, B.; Garg, A.; Hertwich, E.; et al. Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; pp. 511–598. [Google Scholar]
- IEA. World Energy Outlook 2016; International Energy Agency: Paris, France, 2016; pp. 1–684. [Google Scholar]
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects and future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Manthiram, A.; Chung, S.-H.; Zu, C. Lithium–Sulfur Batteries: Progress and Prospects. Adv. Mater. 2015, 27, 1980–2006. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, M.M.; Wolverton, C.; Isaacs, E.D. Electrical energy storage for transportation—Approaching the limits of, and going beyond, lithium-ion batteries. Energy Environ. Sci. 2012, 5, 7854–7863. [Google Scholar] [CrossRef]
- Dunn, B.; Kamath, H.; Tarascon, J. Electrical energy storage for the grid: A Battery of choices. Science 2011, 334, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Armand, M.; Tarascon, J.-M. Building better batteries. Nature 2008, 451, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Ge, M.; Rong, J.; Fang, X.; Zhou, C. Porous doped silicon nanowires for lithium ion battery anode with long cycle life. Nano Lett. 2012, 12, 2318–2323. [Google Scholar] [CrossRef] [PubMed]
- Omar, N.; Daowd, M.; van den Bossche, P.; Hegazy, O.; Smekens, J.; Coosemans, T.; van Mierlo, J. Rechargeable energy storage systems for plug-in hybrid electric vehicles-assessment of electrical characteristics. Energies 2012, 5, 2952–2988. [Google Scholar] [CrossRef]
- Larcher, D.; Tarascon, J.-M. Toward greener and more sustainable batteries for electrical energy storage. Nat. Chem. 2014, 7, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Koketsu, T.; Ma, J.; Morgan, B.J.; Body, M.; Legein, C.; Dachraoui, W.; Giannini, M.; Demortière, A.; Salanne, M.; Dardoize, F.; et al. Reversible magnesium and aluminium ions insertion in cation-deficient anatase TiO2. Nat. Mater. 2017, 16, 1142–1148. [Google Scholar] [CrossRef] [PubMed]
- Kushnir, D.; Sandén, B.A. The time dimension and lithium resource constraints for electric vehicles. Resour. Policy 2012, 37, 93–103. [Google Scholar] [CrossRef]
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.-M. Li–O2 and Li–S batteries with high energy storage. Nat. Mater. 2011, 11, 172–172. [Google Scholar] [CrossRef]
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions: on the 2017 list of Critical Raw Materials for the EU; European Commission: Brussels, Belgium, 2017.
- Jayaprakash, N.; Das, S.K.; Archer, L.A. The rechargeable aluminum-ion battery. Chem. Commun. 2011, 47, 12610–12612. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jiang, B.; Xiong, W.; Sun, H.; Lin, Z.; Hu, L.; Tu, J.; Hou, J.; Zhu, H.; Jiao, S. A new cathode material for super-valent battery based on aluminium ion intercalation and deintercalation. Sci. Rep. 2013, 3, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.C.; Gong, M.; Lu, B.; Wu, Y.; Wang, D.Y.; Guan, M.; Angell, M.; Chen, C.; Yang, J.; Hwang, B.J.; et al. An ultrafast rechargeable aluminium-ion battery. Nature 2015, 520, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Egan, D.; de Leon, C.P.; Wood, R.J.K.; Jones, R.L.; Stokes, K.R.; Walsh, F.C. Developments in electrode materials and electrolytes for aluminiumeair batteries. J. Power Sources 2013, 236, 293–310. [Google Scholar] [CrossRef]
- Mori, R. A new structured aluminium–air secondary battery with a ceramic aluminium ion conductor. RSC Adv. 2013, 3, 11547–11551. [Google Scholar] [CrossRef]
- Mori, R. A novel aluminium–air secondary battery with long-term stability. RSC Adv. 2014, 4, 1982–1987. [Google Scholar] [CrossRef]
- Mori, R. Electrochemical properties of a rechargeable aluminum–air battery with a metal–organic framework as air cathode material. RSC Adv. 2017, 7, 6389–6395. [Google Scholar] [CrossRef]
- Bogolowski, N.; Drillet, J. Activity of different AlCl 3-based electrolytes for the electrically rechargeable aluminium-air battery. Electrochem. Acta 2018, 274, 353–358. [Google Scholar] [CrossRef]
- Elia, G.A.; Hasa, I.; Greco, G.; Diemant, T.; Marquardt, K.; Hoeppner, K.; Behm, R.J.; Hoell, A.; Passerini, S.; Hahn, R. Insights into the reversibility of aluminum graphite batteries. J. Mater. Chem. A 2017, 5, 9682–9690. [Google Scholar] [CrossRef]
- Angell, M.; Pan, C.; Rong, Y.; Yuan, C.; Lin, M.; Hwang, B.; Dai, H. High Coulombic efficiency aluminum-ion battery using an AlCl3 -urea ionic liquid analog electrolyte. Proc. Natl. Acad. Sci. USA 2017, 114, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Mahapatra, S.; Lahan, H. Aluminium-ion batteries: developments and challenges. J. Mater. Chem. A 2017, 5, 6347–6367. [Google Scholar] [CrossRef]
- Wang, H.; Gu, S.; Bai, Y.; Chen, S.; Wu, F.; Wu, C. High-Voltage and Noncorrosive Ionic Liquid Electrolyte Used in Rechargeable Aluminum Battery. ACS Appl. Mater. Interfaces 2016, 8, 27444–27448. [Google Scholar] [CrossRef] [PubMed]
- Elia, G.A.; Marquardt, K.; Hoeppner, K.; Fantini, S.; Lin, R.; Knipping, E.; Peters, W.; Drillet, J.-F.; Passerini, S.; Hahn, R. An Overview and Future Perspectives of Aluminum Batteries. Adv. Mater. 2016, 28, 7564–7579. [Google Scholar] [CrossRef] [PubMed]
- Hetherington, A.C.; Borrion, A.L.; Griffiths, O.G.; McManus, M.C. Use of LCA as a development tool within early research: Challenges and issues across different sectors. Int. J. Life Cycle Assess. 2014, 19, 130–143. [Google Scholar] [CrossRef]
- Hung, C.R.; Ellingsen, L.A.-W.; Majeau-Bettez, G. A framework for early stage lifecycle screening of emerging technologies. J. Ind. Ecol. 2018. in review. [Google Scholar]
- Gavankar, S.; Suh, S.; Keller, A.A. The Role of Scale and Technology Maturity in Life Cycle Assessment of Emerging Technologies: A Case Study on Carbon Nanotubes. J. Ind. Ecol. 2015, 19, 51–60. [Google Scholar] [CrossRef]
- Ellingsen, L.A.; Hung, C.R.; Majeau-Bettez, G.; Singh, B.; Chen, Z.; Whittingham, M.S.; Strømman, A.H. Nanotechnology for environmentally sustainable electromobility. Nat. Nanotechnol. 2016, 11, 1039–1051. [Google Scholar] [CrossRef] [PubMed]
- Bourgault, G. Implementation of Impact Assessment Methods in Ecoinvent Version 3.4, Zürich, Switzerland. 2017.
- Bach, V.; Finogenova, N.; Berger, M.; Winter, L.; Finkbeiner, M. Enhancing the assessment of critical resource use at the country level with the SCARCE method—Case study of Germany. Resour. Policy 2017, 53, 283–299. [Google Scholar] [CrossRef]
- ReCiPe. ReCiPe Mid/Endpoint Method, Version 1.11; Ministry of Housing, Spatial Planning and the Environment (VROM): The Hague, The Netherlands, 2013.
- Ecoinvent Centre. Ecoinvent Data and Reports 3.4; Ecoinvent Centre: Zurich, Switzerland, 2017. [Google Scholar]
- Bakkar, A.; Neubert, V. Electrodeposition and corrosion characterisation of micro- and nano-crystalline aluminium from AlCl3/1-ethyl-3-methylimidazolium chloride ionic liquid. Electrochim. Acta 2013, 103, 211–218. [Google Scholar] [CrossRef]
- Jiang, T.; Brym, M.J.C.; Dubé, G.; Lasia, A.; Brisard, G.M. Electrodeposition of aluminium from ionic liquids: Part I—electrodeposition and surface morphology of aluminium from aluminium chloride (AlCl3)-1-ethyl-3-methylimidazolium chloride ([EMIm]Cl) ionic liquids. Surf. Coat. Technol. 2006, 201, 1–9. [Google Scholar] [CrossRef]
- Hanisch, C.; Diekmann, J.; Stieger, A.; Haselrieder, W.; Kwade, A. Handbook of Clean Energy Systems; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; Volume 5, pp. 1–24. [Google Scholar]
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- European Commission. Raw Materials Information System (RMIS)—Raw Materials Profiles. EU Sci. HUB. Available online: http://rmis.jrc.ec.europa.eu/?page=rm-profiles-a1cdc1#/ (accessed on 27 February 2018).
- United States Geological Survey, Commodity Statistics and Information—Mineral Commodity Summaries. 2018. Available online: https://minerals.usgs.gov/minerals/pubs/commodity/ (accessed on 27 February 2018).
- Umicore (Brussels, Belgium). Personal communication, 2016.
- Accurec (Krefeld, Germany). Personal communication, 2017.
- Netherlands Organisation for Applied Scientific Research, “23. Potash” 2017. Available online: http://rmis.jrc.ec.europa.eu/uploads/rmprofiles/Potash.pdf (accessed on 27 February 2018).
- Wu, Z.; Li, W.; Xia, Y.; Webley, P.; Zhao, D. Ordered mesoporous graphitized pyrolytic carbon materials: synthesis, graphitization, and electrochemical properties. J. Mater. Chem. 2012, 22, 8835–8845. [Google Scholar] [CrossRef]
- Seo, D.H.; Pineda, S.; Fang, J.; Gozukara, Y.; Yick, S.; Bendavid, A.; Lam, S.K.H.; Murdock, A.T.; Murphy, A.B.; Han, Z.J.; et al. Single-step ambient-air synthesis of graphene from renewable precursors as electrochemical genosensor. Nat. Commun. 2017, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Samdarshi, S.K. A green precursor for carbon nanotube synthesis. New Carbon Mater. 2011, 26, 85–88. [Google Scholar] [CrossRef]
- Deloitte Sustainability; British Geological Survey; Bureau de Recherches Géologiques et Minières; Netherlands Organisation for Applied Scientific Research. Study on the Review of the List of Critical Raw Materials; European Commission: Brussel, Belgium, 2017. [CrossRef]
- American Elements, Safety Data Sheet, Carbon Foam. 2015. Available online: https://www.americanelements.com/carbon-foam-7440-44-0 (accessed on 5 February 2018).
- ESPI Metals, Material Safety Data Sheets. Available online: http://www.espimetals.com/index.php/msds (accessed on 5 February 2018).
- Science Lab. Material Safety Data Sheet, Graphite, 1–6; ScienceLab: Houston, TX, USA, 2005. [Google Scholar]
- US Research Nanomaterials Inc. Safety Data Sheet—Carbon Nanomaterials; US Research Nanomaterials Inc.: Houston, TX, USA, 2017; Available online: www.us-nano.com (accessed on 5 February 2018).
- ACS Material LCC. Safety Data Sheet—Pyrolytic Graphite Powder. Version 1.2. 2017. Available online: https://www.acsmaterial.com/media/catalog/product/s/d/sds_pyrolytic_graphite_powder.pdf (accessed on 5 February 2018).
- Wang, D.; Wei, C.; Lin, M.; Pan, C.; Chou, H.; Chen, H.; Gong, M.; Wu, Y.; Yuan, C.; Angell, M.; et al. Advanced rechargeable aluminium ion battery with a high-quality natural graphite cathode. Nat. Commun. 2017, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xu, H.; Wang, S.; Huang, T.; Xi, J.; Cai, S.; Guo, F.; Xu, Z. Ultrafast all-climate aluminum-graphene battery with quarter-million cycle life. Sci. Adv. 2017, 3, eaao7233. [Google Scholar] [CrossRef] [PubMed]
- EPRI. Electric Energy Storage Technology Options: A White Paper Primer on Applications, Costs and Benefits; EPRI: Palo Alto, CA, USA, 2010; pp. 1–170. [Google Scholar]
- Charitidis, C.A.; Georgiou, P.; Koklioti, M.A.; Trompeta, A.-F.; Markakis, V. Manufacturing nanomaterials: from research to industry. Manuf. Rev. 2014, 1, 1–11. [Google Scholar] [CrossRef]
- Şengül, H.; Theis, T.L.; Ghosh, S. Toward Sustainable Nanoproducts. J. Ind. Ecol. 2008, 12, 329–359. [Google Scholar] [CrossRef]
- Hudak, N.S. Chloroaluminate-doped conducting polymers as positive electrodes in rechargeable aluminum batteries. J. Phys. Chem. C 2014, 118, 5203–5215. [Google Scholar] [CrossRef]
- Whittingham, M.S. Electrical Energy Storage and Intercalation Chemistry. Science 1976, 192, 1126–1127. [Google Scholar] [CrossRef] [PubMed]
- Geng, L.; Scheifers, J.; Fu, C.; Zhang, J.; Fokwa, B.P.T.; Guo, J. Titanium Sulfides as Intercalation-Type Cathode Materials for Rechargeable Aluminum Batteries. ACS Appl. Mater. Interfaces 2017, 9, 21251–21257. [Google Scholar] [CrossRef] [PubMed]
- Cohn, G.; Ma, L.; Archer, L.A. A novel non-aqueous aluminum sulfur battery. J. Power Sources 2015, 283, 416–422. [Google Scholar] [CrossRef]
- Gao, T.; Li, X.; Wang, X.; Hu, J.; Han, F.; Fan, X.; Suo, L.; Pearse, A.J.; Lee, S.B.; Rubloff, G.W.; et al. A Rechargeable Al/S Battery with an Ionic-Liquid Electrolyte. Angew. Chem. 2016, 55, 9898–9901. [Google Scholar] [CrossRef] [PubMed]
- Ellingsen, L.A.; Majeau-Bettez, G.; Singh, B.; Srivastava, A.K.; Valøen, L.O.; Strømman, A.H. Life cycle assessment of a lithium-ion battery vehicle pack. J. Ind. Ecol. 2014, 18, 113–124. [Google Scholar] [CrossRef]
- Yuan, C.; Deng, Y.; Li, T.; Yang, F. Manufacturing energy analysis of lithium ion battery pack for electric vehicles. CIRP Ann. 2017, 66, 53–56. [Google Scholar] [CrossRef]
- Wood, D.L.; Li, J.; Daniel, C. Prospects for reducing the processing cost of lithium ion batteries. J. Power Sources 2015, 275, 234–242. [Google Scholar] [CrossRef]
- Wood, D.L.; Quass, J.D.; Li, J.; Ahmed, S.; Ventola, D.; Daniel, C. Technical and economic analysis of solvent-based lithium-ion electrode drying with water and NMP. Dry. Technol. 2017, 36, 234–244. [Google Scholar] [CrossRef]
- Majeau-Bettez, G.; Hawkins, T.R.; Strømman, A.H. Life cycle environmental assessment of lithium-ion and nickel metal hydride batteries for plug-in hybrid and battery electric vehicles. Environ. Sci. Technol. 2011, 45, 4548–4554. [Google Scholar] [CrossRef] [PubMed]
- Khan, F. An initial seed selection algorithm for k-means clustering of georeferenced data to improve replicability of cluster assignments for mapping application. Appl. Soft Comput. J. 2012, 12, 3698–3700. [Google Scholar] [CrossRef]
- Science Lab. Material Safety Data Sheet—Tungsten Oxide; ScienceLab: Hourston, TX, USA, 2013. [Google Scholar]
- LTS Chemical, SDS|LTS. Available online: https://www.ltschem.com/msds/ (accessed on 5 February 2018).
- American Elements, Safety Data Sheet—Aluminum Oxide Nanopowder. 2015. Available online: https://www.americanelements.com/aluminum-oxide-nanoparticles-nanopowder-1344-28-1 (accessed on 5 February 2018).
- Science Lab. Material Safety Data Sheet—Cobalt; ScienceLab: Houston, TX, USA, 2013. [Google Scholar]
- Science Lab. Material Safety Data Sheet—Potassium; ScienceLab: Houston, TX, USA, 2013. [Google Scholar]
- Science Lab. Material Safety Data Sheet—Titanium Dioxide; ScienceLab: Houston, TX, USA, 2013; Available online: http://www.sciencelab.com/msds.php?msdsId=9925268 (accessed on 5 February 2018).
- American Elements, Safety Data Sheet—Niobium Oxide Nanopowder. 2015. Available online: https://www.americanelements.com/niobium-oxide-nanoparticles-nanopowder-1313-96-8#section-safety (accessed on 5 February 2018).
- Wernet, G.; Bauer, C.; Steubing, B.; Reinhard, J.; Moreno-Ruiz, E.; Weidema, B. The ecoinvent database version 3 (part I): overview and methodology. Int. J. Life Cycle Assess. 2016, 21, 1218–1230. [Google Scholar]
- Nuss, P.; Eckelman, M.J. Life cycle assessment of metals: A scientific synthesis. PLoS ONE 2014, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Netherlands Organisation for Applied Scientific Research, “29. Sulphur”. 2017. Available online: http://rmis.jrc.ec.europa.eu/uploads/rmprofiles/Sulphur.pdf (accessed on 26 February 2018).
- Majeau-bettez, G.; Wood, R.; Strømman, A.H. Unified theory of allocations and constructs in life cycle assessment and input-output. J. Ind. Ecol. 2014, 18, 747–770. [Google Scholar] [CrossRef]
- SCARCE—Methode zur Bewertung der Kritikalität von Abiotischen Ressourcen auf Regionaler Ebene. 2018. Available online: http://www.see.tu-berlin.de/menue/forschung/ergebnisse/scarce_method_enhancing_the_assessment_of_critical_resource_use_on_country_level (accessed on 25 February 2018).
- Kumar, M.; Ando, Y. Chemical Vapor Deposition of Carbon Nanotubes: A Review on Growth Mechanism and Mass Production. J. Nanosci. Nanotechnol. 2010, 10, 3739–3758. [Google Scholar] [CrossRef] [PubMed]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef] [PubMed]
- Elazari, R.; Salitra, G.; Garsuch, A.; Panchenko, A.; Aurbach, D. Sulfur-impregnated activated carbon fiber cloth as a binder-free cathode for rechargeable Li-S batteries. Adv. Mater. 2011, 23, 5641–5644. [Google Scholar] [CrossRef] [PubMed]
- Kozen, A.C.; Lin, C.; Pearse, A.J.; Schroeder, M.A.; Han, X.; Hu, L.; Lee, S.; Rubloff, G.W.; Noked, M. Next-Generation Lithium Metal Anode Engineering via Atomic Layer Deposition. ACS Nano 2015, 9, 5884–5892. [Google Scholar] [CrossRef] [PubMed]
- Goodfellow Catalogue, Aluminium-Foil. 2018, pp. 8–9. Available online: http://www.goodfellow.com/E/Aluminium-Foil.html (accessed on 26 February 2018).
- Fitzer, E.; Kochling, K.-H.; Boehm, H.P.; Marsh, H. Recommended Terminology for the Description of Carbon as a Solid. Pure Appl. Chem. 1995, 67, 473–506. [Google Scholar] [CrossRef]
- European Carbon and Graphite Association. Graphite Production & Further Processing; European Carbon and Graphite Association: Bruxelles, Belgium; Available online: http://www.carbonandgraphite.org/pdf/graphite_production.pdf (accessed on 26 February 2018).
- Nakamura, S.; Miyafuji, D.; Fujii, T.; Matsui, T.; Fukuyama, H. Low temperature transport properties of pyrolytic graphite sheet. Cryogenics 2017, 86, 118–122. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ellingsen, L.A.-W.; Holland, A.; Drillet, J.-F.; Peters, W.; Eckert, M.; Concepcion, C.; Ruiz, O.; Colin, J.-F.; Knipping, E.; Pan, Q.; et al. Environmental Screening of Electrode Materials for a Rechargeable Aluminum Battery with an AlCl3/EMIMCl Electrolyte. Materials 2018, 11, 936. https://doi.org/10.3390/ma11060936
Ellingsen LA-W, Holland A, Drillet J-F, Peters W, Eckert M, Concepcion C, Ruiz O, Colin J-F, Knipping E, Pan Q, et al. Environmental Screening of Electrode Materials for a Rechargeable Aluminum Battery with an AlCl3/EMIMCl Electrolyte. Materials. 2018; 11(6):936. https://doi.org/10.3390/ma11060936
Chicago/Turabian StyleEllingsen, Linda Ager-Wick, Alex Holland, Jean-Francois Drillet, Willi Peters, Martin Eckert, Carlos Concepcion, Oscar Ruiz, Jean-François Colin, Etienne Knipping, Qiaoyan Pan, and et al. 2018. "Environmental Screening of Electrode Materials for a Rechargeable Aluminum Battery with an AlCl3/EMIMCl Electrolyte" Materials 11, no. 6: 936. https://doi.org/10.3390/ma11060936
APA StyleEllingsen, L. A.-W., Holland, A., Drillet, J.-F., Peters, W., Eckert, M., Concepcion, C., Ruiz, O., Colin, J.-F., Knipping, E., Pan, Q., Wills, R. G. A., & Majeau-Bettez, G. (2018). Environmental Screening of Electrode Materials for a Rechargeable Aluminum Battery with an AlCl3/EMIMCl Electrolyte. Materials, 11(6), 936. https://doi.org/10.3390/ma11060936










