1. Introduction
As a result of the use of plastics in a wide range of products, a huge amount of plastic waste is being generated every year. The consumption of plastics is readily increasing due to their various advantages such as versatility, low cost and high chemical stability [
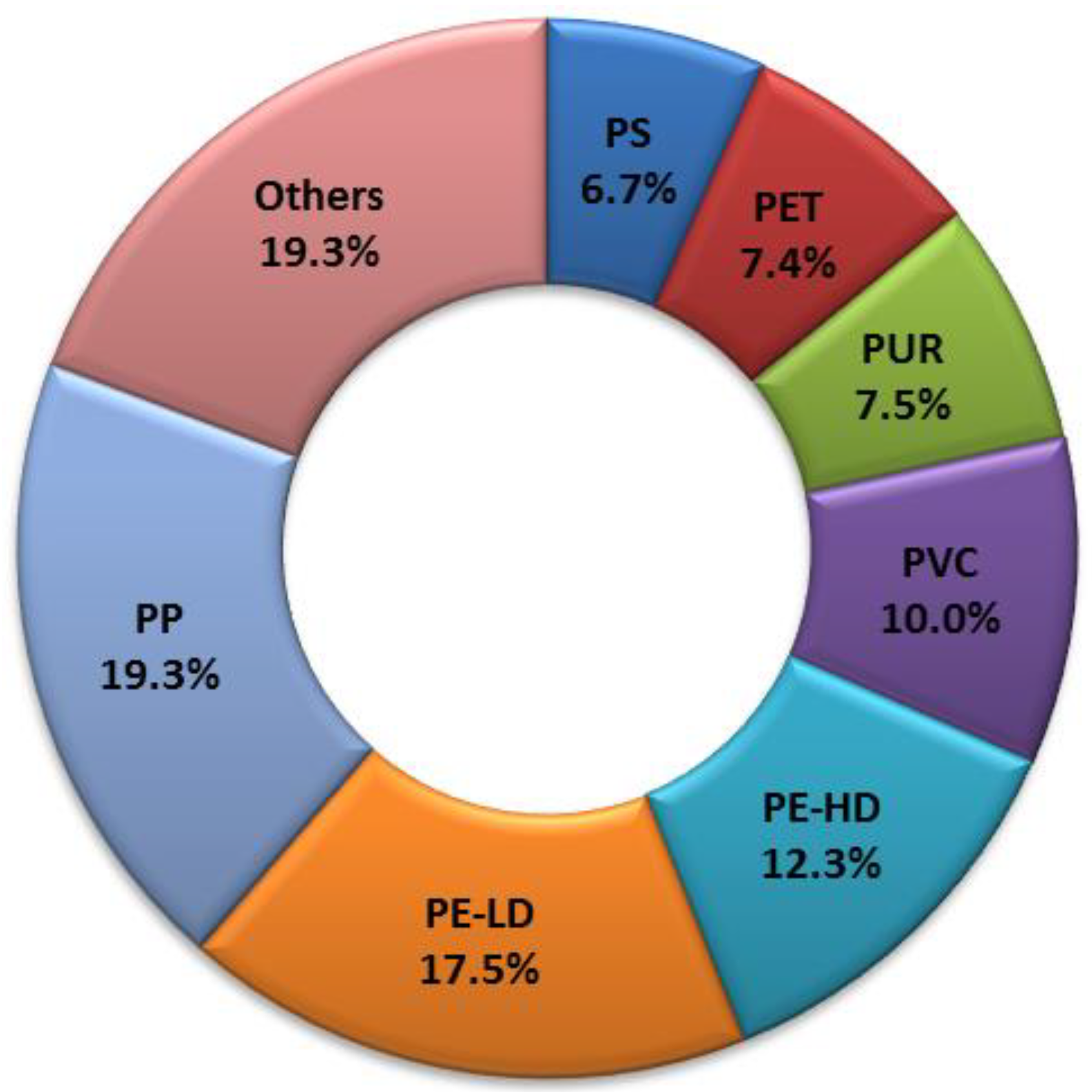
1]. Plastic production data from 2016 show the global production of 335 Mt and the European production of 60 Mt. Considering the types of polymers that make up the bulk of the collected plastic waste, a fair idea can be obtained by looking at the plastic demand for new products. In terms of plastic demand by polymer type (
Figure 1), Europe was the leader in 2016 with respect to polypropylene (PP), with 19.3% [
2,
3].
The preferred option in waste management is recycling. In recycling, new raw materials are obtained via a mechanical or chemical pathway. If polymer waste cannot be recycled, energy recovery is the preferred option. Landfills, the least preferred option, should be avoided at all cost [
3]. In Europe, in ten years (2006–2016), plastic waste recycling has increased by almost 80%, and for the first time, in 2016, more plastic waste was recycled (31.1%) than landfilled (27.3%). However, due to the ban on landfilling of plastic waste in some European countries, recycling is unevenly distributed among the states of Europe [
2]. Furthermore, every type of plastic waste is recycled to a different extent. As we can see, PP represents the largest part of plastic production, but its recycling ratio is very low. For example, in the United States in 2012, when PP in plastic production reached second place with 22.6%, its recycling ratio was only 0.6%, which makes it one of the least recycled postconsumer plastics [
4].
In 2010, buildings accounted for 32% of global final energy use. Increase in energy consumption in buildings in the last twenty years has been modest in US and EU countries, due to the improvement of energy efficiency and the promotion of renewable energy sources. The ambitious EU 2030 Climate & Energy package fixed the reduction of greenhouse gas emissions at 40% from 1990 levels, the share for renewable energy at 27% and improvement in energy efficiency at 27% [
5]. Special attention is devoted to buildings due to the large amount both of energy consumption and estimated energy savings of this sector in EU member states. Technology innovations offer new solutions for buildings and smart energy concepts that enable more dynamic and interactive buildings, where technologies are used in conjunction with optimum design techniques [
6,
7,
8]. On the other hand, as the economy, urbanization, and population of developing countries continue to grow, a significant increase in building energy consumption in these countries is expected. Given the current global demand trends, building energy saving is highly critical [
9,
10]. In order to design buildings with preferable thermal attributes and optimum energy performance, an important element is the envelope design, and the use of materials with effective thermal insulation properties should be taken into consideration [
10,
11].
Among the various types of recycling management approaches, the reuse of waste and recycled plastic material in the construction industry is considered an ideal method for disposing plastic waste [
8]. The great advantages of this method include that the recycled plastics substitute the use of virgin construction materials (with related environmental and energy impacts) and moreover, in terms of energy saving, the composites with plastic aggregates exhibit improved thermal insulation performance. The use of recycled plastic materials in conventional cement mortar and concrete has been researched extensively [
8,
9,
10,
11,
12,
13,
14]. On the other hand, no study has reported the use of waste plastic-based aggregates in MOC composites to date.
As an air-dried magnesia-based cementing material, magnesium oxychloride cement (MOC) was developed by a French scientist, Sorel, shortly after the invention of Portland cement in 1867 [
15,
16,
17]. It is obtained by mixing magnesium oxide (MgO) powder with a concentrated solution of magnesium chloride (MgCl
2) and is one of the main representative chemically bonded cements [
18,
19,
20]. Through a solution reaction, two chemical composition phases, namely, phase 3 (3Mg(OH)
2·MgCl
2·8H
2O) and phase 5 (5Mg(OH)
2·MgCl
2·8H
2O), can be formed at ambient temperature and provide the bulk of the mechanical integrity [
21]. The production of lightly burnt MgO used in MOC requires a much lower calcination temperature compared to that for Portland cement, thus reducing the vast amount of energy consumption [
22].
MOC has been reported with many superior properties over Portland cement, such as high mechanical strength [
22], good fire resistance [
23,
24,
25], low thermal conductivity [
21,
26], resistance to abrasion [
20,
27] and especially remarkable high bonding ability to different types of fillers [
19,
21,
22]. By virtue of these advantages, MOC has been well utilized for making floors, fire protection panels, thermal insulation composites, decoration panels, grinding wheels, etc. [
21,
22].
Taking into account all the above-mentioned facts and knowledge gaps, the present study is aimed at evaluation of the use of waste-expanded polypropylene aggregate as a full replacement of natural aggregate for the development of MOC composite. In terms of both waste management and the improvement of material thermal resistance, the main objective was to produce an MOC composite incorporating as much waste plastic aggregate as possible, to obtain material with enhanced thermal properties that still meets the requirements for workability and strength. Based on literature analysis, such modification of MOC composite composition has not been reported, and the present study can therefore be considered as an initial attempt in this manner. Investigation of an MOC composite with incorporated waste-expanded polypropylene was performed in order to determine its mechanical, physical and thermal properties and water resistance behavior. This new developed material with enhanced thermal insulation performance can find application in the floor and wall structural systems of buildings, can prevent energy loss and will enable the reuse of a large amount of the studied type of plastic waste.
3. Results and Discussion
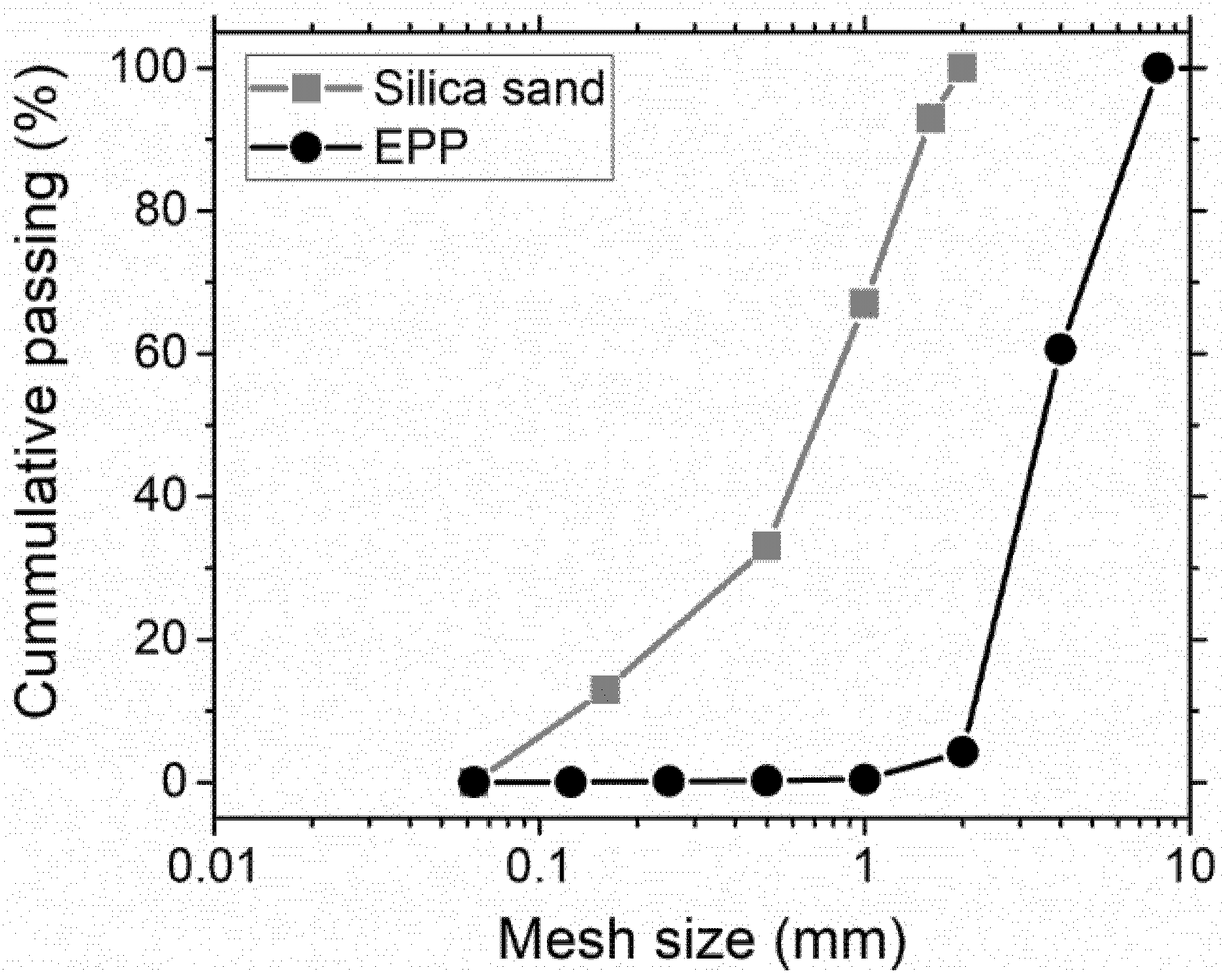
Figure 4 shows the grain-size curves of both natural and EPP aggregates. We can see that all EPP particles were smaller than 4 mm, which is suitable for their use as a silica sand replacement.
The specific density value of the EPP aggregate was 105 kg/m
3. The EPP aggregate therefore belongs to artificial ultra-lightweight (density of less than 300 kg/m
3) non-absorbent aggregates as given by Babu and Babu [
37]. The thermophysical parameters of the EPP aggregate and silica sand as well as their corresponding powder density are summarized in
Table 4.
The EPP aggregate exhibited significantly lower heat transport than silica sand typically used in mortars and fine-grained concretes. The volumetric heat capacity of silica sand was observed to be markedly higher in comparison with EPP due to its high density and mineral origin. With increasing time of compaction, the thermal conductivity and volumetric heat capacity increased due to the elimination of air gaps between the particles. This aggregate behavior can be expected during preparation of MOC composites with incorporated EPP aggregate.
Figure 5 displays images from the optical microscopy investigation under two different resolutions. The EPP foam consisted of closed-cell beads of high porosity that is linked to its low weight, high strength-to-weight ratio, low thermal conductivity and sound insulation properties. As the EPP aggregate used in this study comes from the crushing of waste EPP material, the obtained crushed particles has different colors, shapes and morphologies, but their skin texture is homogeneous and strongly oriented as apparent from the left microphotograph.
As expected, the use of the EPP aggregate led to a reduction in workability from a flow diameter of 235 mm (MOC-R) to 140 mm (MOC-EPP). Although there is no literature on MOC composites containing recycled plastic aggregates, the fact that the incorporation of plastic aggregates leads to the decrease of workability is reported by many authors for composites with Portland cement used as a binder [
13,
38]. However, the resulting workability of the developed MOC-EPP composite is still suitable for the intended application of the developed material in floor structure, sandwich insulation panels, facade panels, etc.
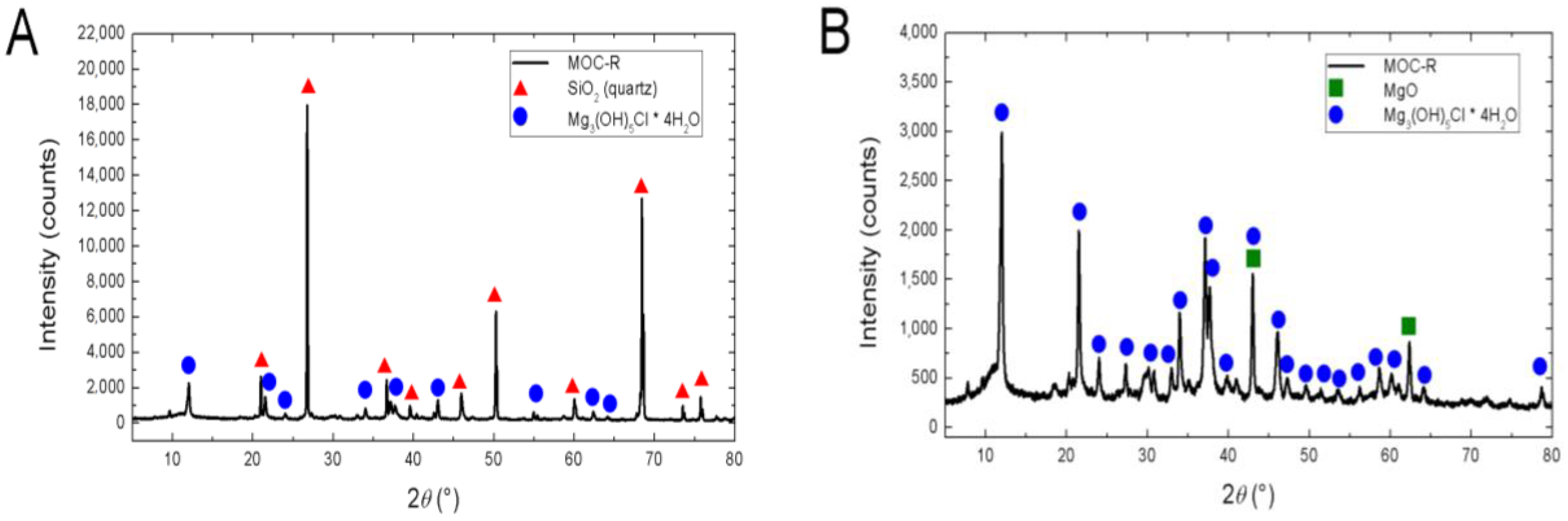
According to XRD (
Figure 6A), phase composition of the sample MOC-R contained two phases: strongly diffracting quartz (JCPDS 01-085-0865) and Mg
3(OH)
5Cl·4H
2O (JCPDS 00-007-0420). Also, a very low amount of MgO (JCPDS 01-077-2364) was detected (presence of MgO is not visible in the XRD diffractogram). Phase Mg
3(OH)
5Cl·4H
2O can be also expressed as 5Mg(OH)
2·MgCl
2·8H
2O. The second sample, MOC-EPP (
Figure 6B), contained as a major phase Mg
3(OH)
5Cl·4H
2O and also a minor phase of unreacted MgO (JCPDS 01-077-2364). The content of unreacted MgO was very low and can be assigned to the mix inhomogeneity and concentration of the MgCl
2 solution that affected the mix workability. A similar small amount of residual MgO was observed, e.g., Yanni et al. [
16].
Scanning electron microscopy (SEM) was used to analyze the morphology and binding conditions between the polymer and matrix (see
Figure 7). Measurement was performed on cut surfaces after gold sputtering. In the case of MOC-R, a relatively flat surface without any cracks or major defects was examined. On the other hand, sample MOC-EPP was not entirely flat because plastic deformation occurred when cutting the polymer and now the polymer is above the surface of the matrix. Moreover, even gold sputtering in combination with low accelerating voltage of the electron beam was not sufficient to remove the charge from the polymer, thus the polymer is much brighter in comparison to the matrix. However, it can be seen that the polymer was retained very tightly in the matrix; even the cutting did not break the bond with the matrix.
EDS maps are shown in
Figure 8. For both samples, carbon, oxygen, magnesium, chlorine and calcium were detected. Silicon was also obtained for MOC-R; however, the silicon content in MOC-EPP was negligible. The obtained results confirmed the presence of 5Mg(OH)
2·MgCl2·8H
2O and quartz in MOC-R, which is in good agreement with XRD. In sample MOC-EPP, the chemical composition of the matrix is very similar in comparison to MOC-R; only SiO
2 (quartz) was not present. According to the carbon elemental map, we can clearly recognize the polymer in the left upper corner in MOC-EPP.
Matrix density, bulk density and porosity data of the developed MOC composites are presented in
Table 5. The presented data represent average values from 5 measured samples.
We can see that incorporation of ultra-lightweight EPP aggregates into the MOC matrix led to a significant decrease in both matrix and bulk densities. The low bulk density of the MOC-EPP composite made it possible to classify it as lightweight concrete in class LC 1.0 according to EN 206-1 [
39]. On the contrary, the presence of plastic particles caused a corresponding increase in porosity. These obtained physical property results are similar to those reported for Portland cement-based concrete containing plastic aggregates [
40]. Gu and Ozbakkaloglu [
12] in their comprehensive review stated that with increasing plastic substitution, the density of concretes decreased regardless of the type or size of used plastic aggregates because plastic aggregates are generally lighter than natural aggregates.
Table 6 shows data on the mechanical resistance of the tested MOC composites. Within the flexural and Young’s modulus tests, 5 samples were measured. The compressive strength was measured on the 8 halves of broken prisms from the flexural strength test. Similar to Misra and Mathur [
41], we observed for both MOC-R and MOC-EPP samples higher flexural strength and higher elastic modulus than would be expected when using Portland cement concrete of the same compressive strength. In our case, the flexural/compressive strength ratio is approx. 0.31 for MOC-R and 0.76 for MOC-EPP samples. For OPC concrete, the flexural/compressive strength ratio is usually 0.1–0.2, as reported, e.g., in [
42,
43]. The high flexural/compressive strength ratio of MOC-based composites can potentially obviate the need of fiber reinforcement and permits reduction in thickness slab, panels, etc. made of MOC. The mixture containing EPP particles exhibits significantly lower mechanical resistance than that with silica sand, nevertheless, this mechanical performance is quite sufficient for its intended use as a non-bearing material for building components. The worsening of mechanical properties of Portland cement-based concrete with varying amounts and types of plastic aggregates was observed widely [
12,
13,
43,
44,
45,
46,
47]. It is quite apparent that the mechanical strength of concrete is highly influenced by the strength of the used aggregate. Brooks et al. [
48] reported a decrease in the compressive strength for Portland cement composites containing expanded polystyrene (EPS), from 39.3 MPa for reference sample to 12.5 MPa for the sample containing 28 vol. % of EPS; the decrease in strength the authors assigned primarily to the introduction of the weak aggregate phase, which promotes stress cracks to initiate and propagate within the materials under mechanical loading. Similar material performance under a mechanical load can also be anticipated in our case.
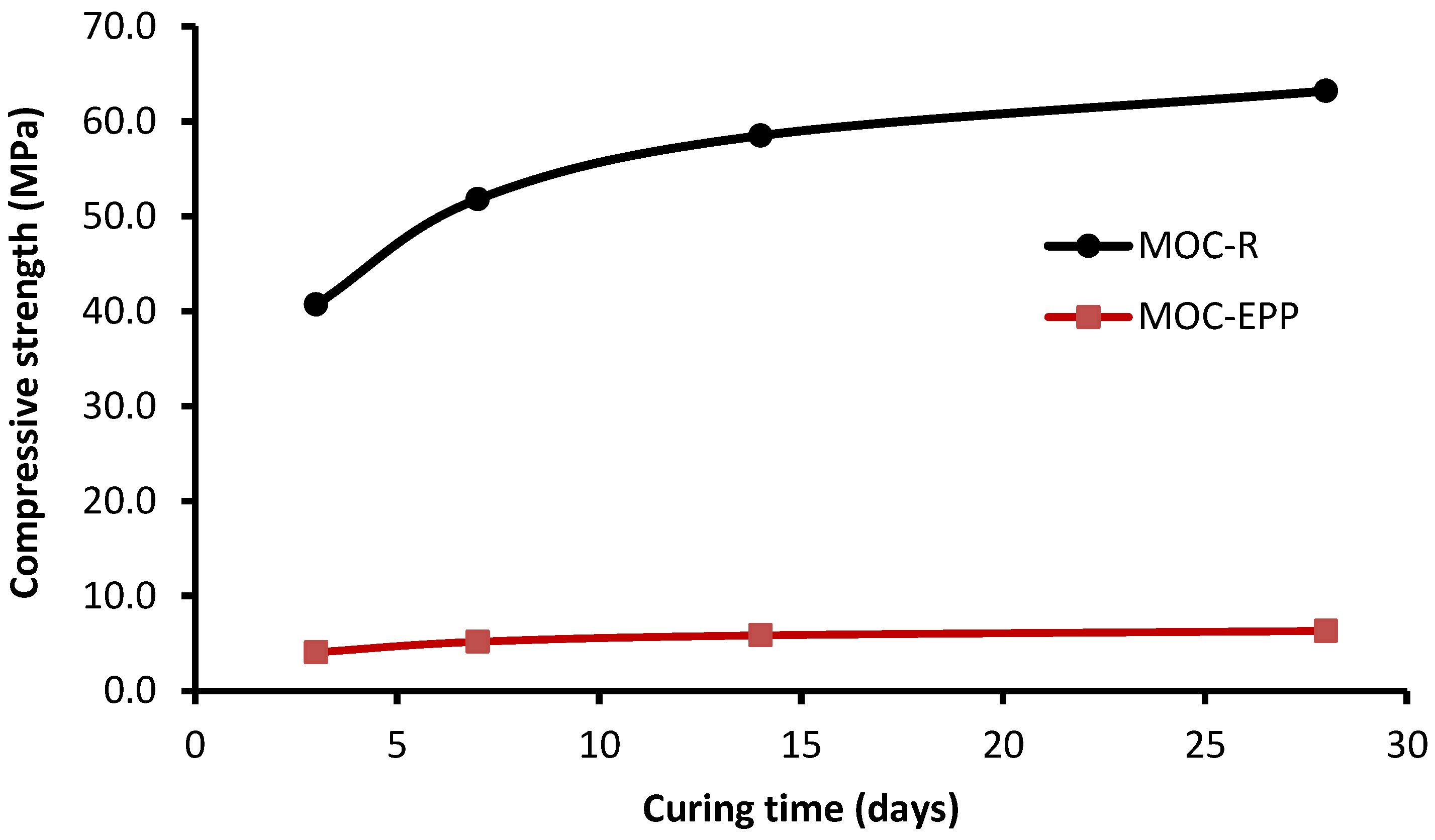
The compressive strength development of MOC-R and MOC-EPP in time is shown in
Figure 9. We can see rapid development of the compressive strength of both studied materials at the early curing age. From the 14
th day of curing, the compressive strength increased only slowly. This observation is consistent with the findings of Xu et al. [
20].
The thermal properties of MOC-based materials air cured for 28 days are summarized in
Table 7. The data present average values from 5 samples tested.
Thermal conductivity can be regarded as one of the most important properties for construction materials considered for using in building envelopes for energy savings. The resulting thermal properties of MOC composites are mainly influenced by the composition and reactivity of the magnesium oxide powder, concentration of magnesium chloride solution, and the quantity and character of the aggregates used. As can be seen in
Table 6, the use of EPP aggregates led to a great reduction in the thermal conductivity value that is almost 82% lower than for the reference sample. The contribution of the EPP aggregate with better thermal parameters compared to silica sand and increased porosity of MOC-EPP composite can be attributed as the main reasons for the observed thermal resistance improvement. The observed thermal conductivity of the MOC-R composite is consistent with the results of Xu et al. [
20]. Brooks et al. [
48] reported a decrease in thermal conductivity for Portland cement-based composites with incorporated EPS from 2.45 W/mK for the reference sample to 0.86 W/mK for the sample with 28 vol. % of EPS. Wang et al. [
49] stated that the thermal conductivity of mortar with Portland cement as a binder containing high impact polystyrene (HIPS) was reduced to 87%, 69%, and 44% of the reference mortar when the HIPS ratio was 10%, 20%, and 50% by volume, respectively.
The water resistance of MOC composites was evaluated on the basis of the strength measurement. The water resistance coefficient was calculated from the cubic compressive strength of 28-day air-cured samples and from the retained compressive strength of the samples with 28 days prolonged exposure to water. The calculation was performed according to Equation (1). Within the water resistance tests, 5 samples of both studied MOC-based composites were analyzed. The obtained
αw value was 0.68 for MOC-R and 0.28 for MOC-EPP sample. The lower
αw value for the MOC-EPP material compared to the value measured for MOC-R can be assigned to the higher porosity of MOC-EPP samples and thus their higher water absorption capacity. The accessed water resistance coefficients were obtained for samples having a moisture content by volume of 5.9% (MOC-R) and 8.5% (MOC-EPP). Xu et al. [
20] studied the influence of cenospheres on the properties of MOC-based composites and observed for reference samples a water resistance coefficient value 0.62. For samples with the addition of 15 and 20 mass % of cenospheres, the
αw value decreased to 0.55 and 0.41, respectively. They assigned this strength degradation to the decomposition of phase 5 to brucite as a result of the hydrolysis reaction [
50,
51], which is known to have limited binding ability. As a similar degradation mechanism was anticipated in our case, after the leaching experiment, samples were analyzed using XRD. It was confirmed that unreacted MgO reacted with water and Mg(OH)
2 was formed (brucite, JCPDS 00-001-1169). In addition, the more porous structure of the MOC composite with EPP facilitated water penetration in the composite matrix, which apparently led to an easier formation of brucite, and thus to more significant strength degradation.
4. Conclusions
In this study, the influence of waste-based expanded polypropylene aggregate on the structural, mechanical, thermal and water resistance properties of magnesium oxychloride cement composites was investigated and evaluated. On the basis of obtained experimental results, the following conclusions can be drawn:
The MOC matrix can incorporate a large amount of waste-based EPP aggregate, equivalent to 150% by volume of silica sand used in the reference composite, to still be usable in construction industry. Materials on the basis of MOC cement do not require water curing.
(1) The use of the EPP aggregate led to a 40.4% decrease in workability in comparison with the reference MOC-based material. Nevertheless, MOC-EPP exhibits sufficient rheological properties with good workability and placing characteristics.
(2) By incorporation of EPP, both the bulk and matrix densities of the hardened MOC composite decreased and porosity increased.
(3) Although compressive and flexural strength as well as Young’s modulus have been reduced by application of EPP aggregates, the MOC-EPP composite still maintains adequate mechanical parameters for intended use for non-structural thermal insulating applications. Generally, MOC composites have higher flexural strength and Young’s modulus as compared to Portland cement-based composites of the same compressive strength, even for composites containing plastic aggregates. MOC composites exhibit high compressive strength already at an early curing age.
(4) In comparison with silica sand, the EPP aggregate is characterized by significantly lower thermal conductivity. Consequently, for MOC-EPP composites, there was a great decrease of about 82% in the thermal conductivity value in comparison with the reference sample.
(5) The obtained water resistance coefficient values confirmed the commonly recognized poor water resistance of MOC-based materials. Although MOC is sensitive to water, it can be exploited as a binder by adding chemical additives to improve its water resistance and thus was used also in applications, where contact with water cannot be avoided.
It can be concluded that the application of waste-expanded polypropylene in magnesium oxychloride cement composites or/and MOC-based lightweight concretes can be considered as a promising option for the utilization of a large amount of this type of plastic waste. The developed MOC-EPP composite with improved thermal insulation properties can serve as a part of floor, ceiling, roof, wall or envelope components to reduce the energy consumption of buildings. Moreover, the developed MOC-EPP composite represent an environmentally friendly alternative to construction materials on a Portland cement basis. It should be also noted that MOC composites belong to air-dried materials; thus, appropriate design of building elements containing these composites must take into consideration their poor water resistance; possibly, additives improving MOC resistance against water must be used.