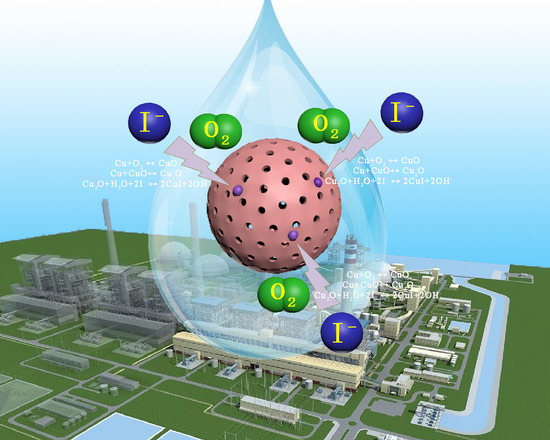
Enhanced Uptake of Iodide from Solutions by Hollow Cu-Based Adsorbents
Abstract
:1. Introduction
2. Experimental Section
2.1. Synthesis of Hollow Cu-Based Adsorbents
2.2. Characterization
2.3. Batch Experiments
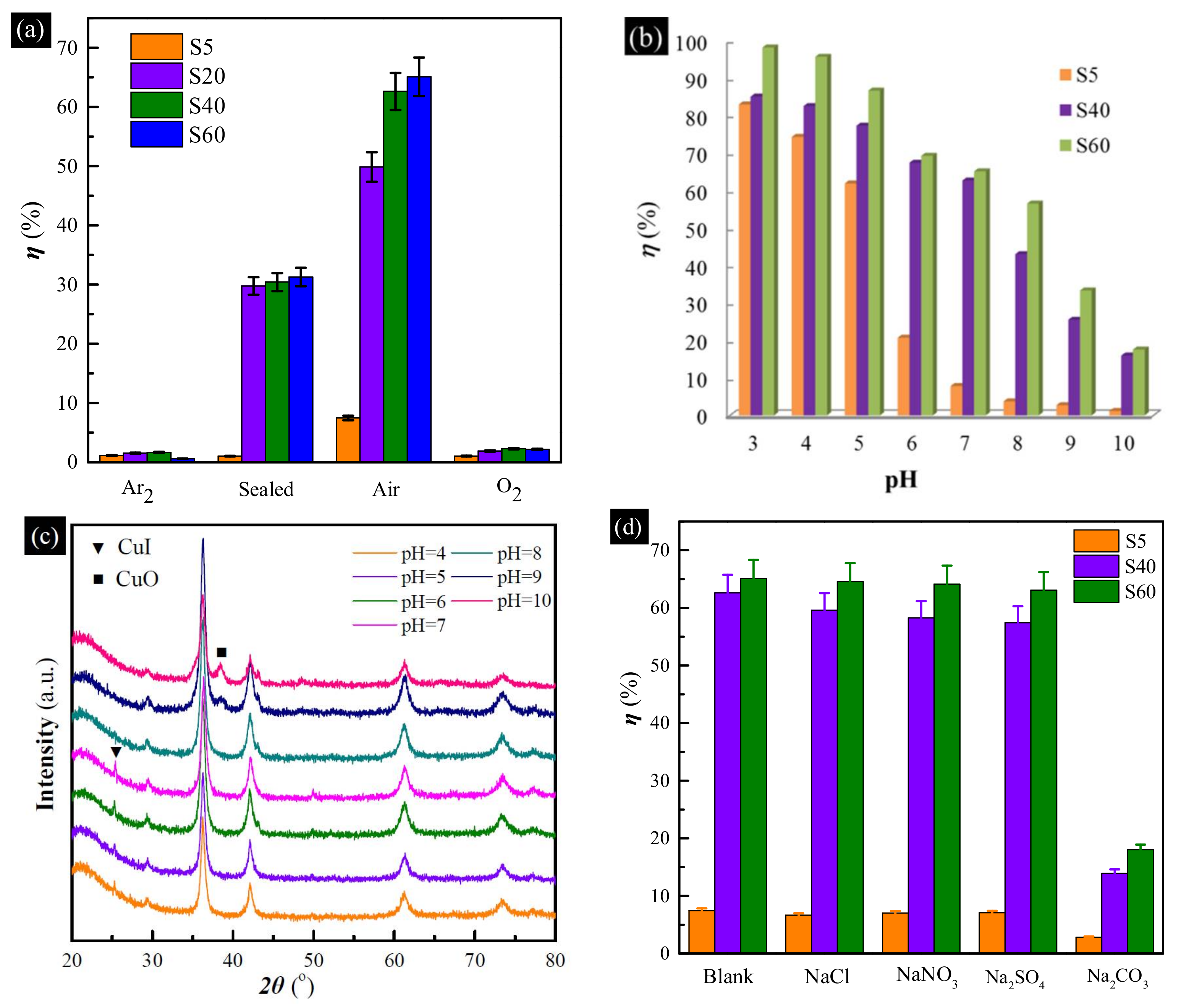
3. Results and Discussion
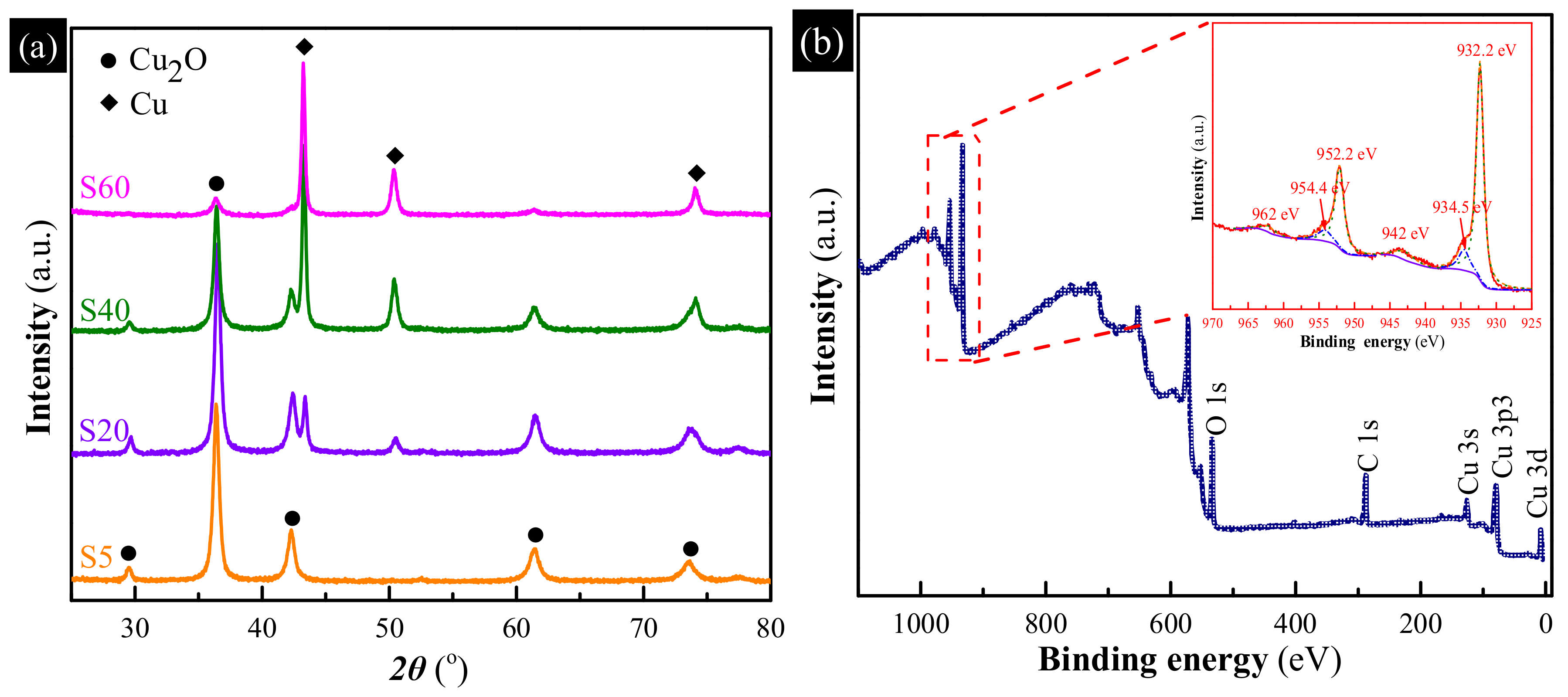
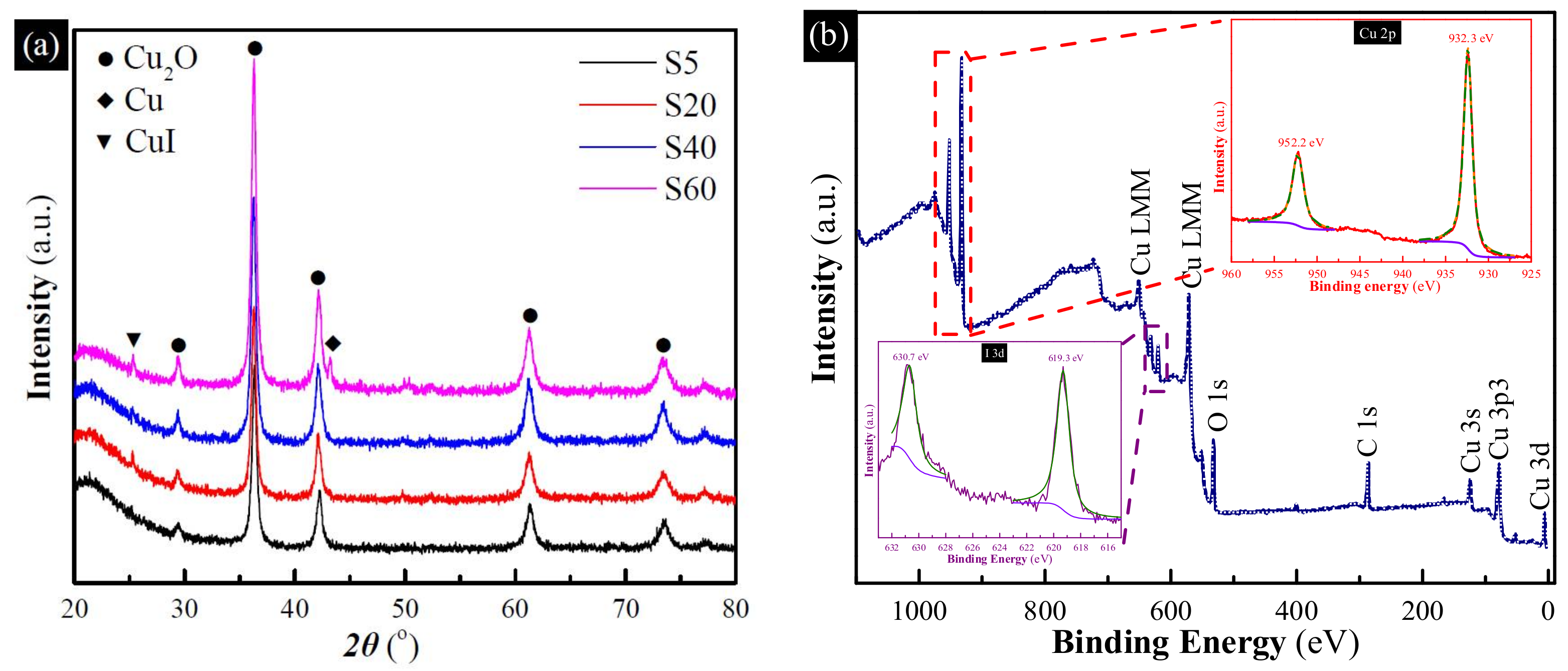
3.1. Characterization
3.2. Uptake Performance Studies
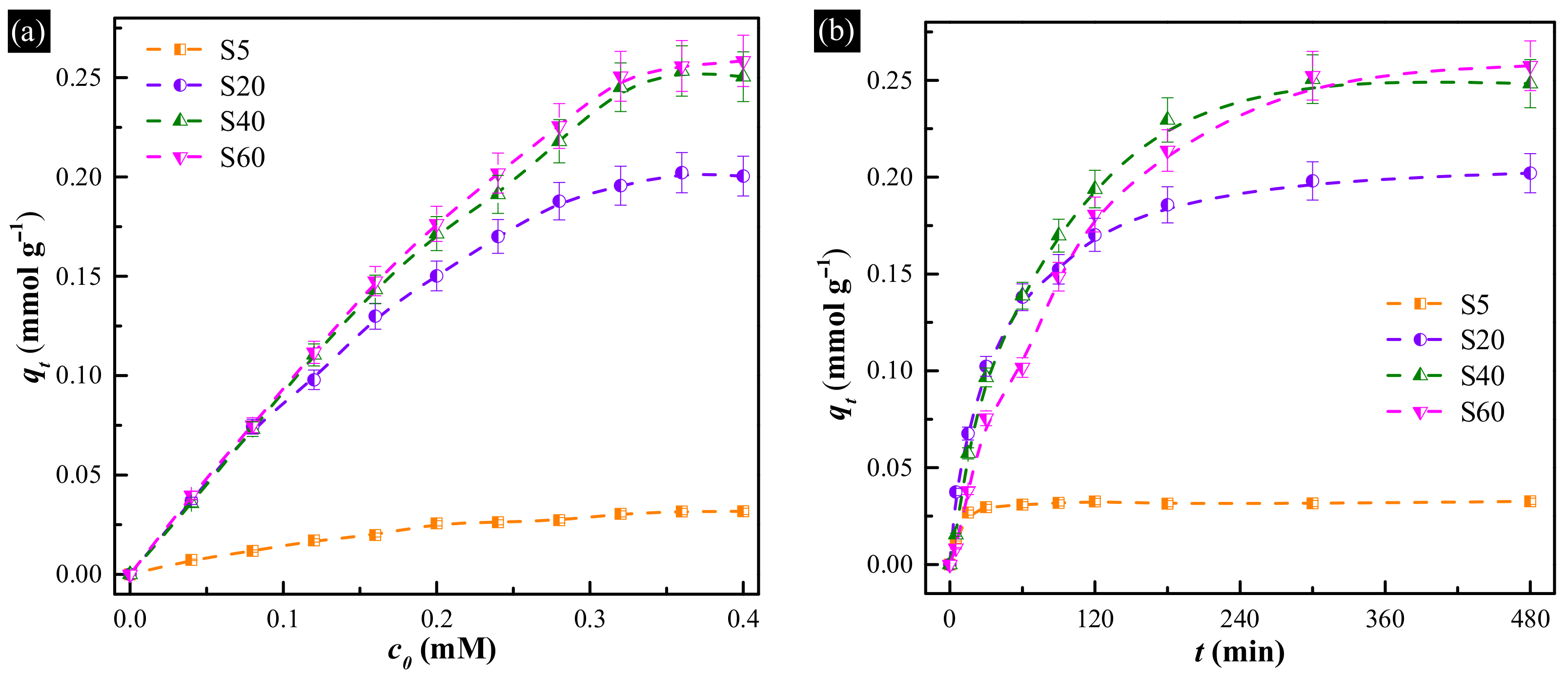
3.2.1. Uptake Equilibrium Isotherms
3.2.2. Uptake Kinetics
3.3. Uptake Mechanism
3.4. Effect of Factors
3.4.1. Effect of pH
3.4.2. Selective Uptake
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rohde, R.A.; Muller, R.A. Air Pollution in China: Mapping of Concentrations and Sources. PLoS ONE 2015, 10, e0135749. [Google Scholar] [CrossRef] [PubMed]
- Kan, H.; Chen, B.; Hong, C. Health impact of outdoor air pollution in China: Current knowledge and future research needs. Environ. Health Perspect. 2009, 117, A187. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Gao, Y.; Xu, Z.; Wong, K.P.; Lai, L.L.; Xue, Y.; Dong, Z.Y.; Hill, D.J. Powering China’s sustainable development with renewable energies: Current status and future trend. Electr. Power Compon. Syst. 2015, 43, 1193–1204. [Google Scholar] [CrossRef]
- Guo, X.; Guo, X. Nuclear power development in China after the restart of new nuclear construction and approval: A system dynamics analysis. Renew. Sustain. Energy Rev. 2016, 57, 999–1007. [Google Scholar] [CrossRef]
- Theiss, F.L.; Ayoko, G.A.; Frost, R.L. Iodide removal using LDH technology. Chem. Eng. J. 2016, 296, 300–309. [Google Scholar] [CrossRef]
- Inoue, H. Transport of 125I and 36Cl across an anion-exchange paper membrane. Appl. Radiat. Isot. 2002, 56, 659–665. [Google Scholar] [CrossRef]
- Inoue, H. Radioactive iodine and chloride transport across a paper membrane bearing trimethylhydroxypropylammonium anion exchange groups. J. Membr. Sci. 2003, 222, 53–57. [Google Scholar] [CrossRef]
- Inoue, H. Effects of co-ions on transport of iodide ions through a non-conventional anion exchange paper membrane. J. Membr. Sci. 2004, 228, 209–215. [Google Scholar] [CrossRef]
- Liu, S.; Kang, S.; Wang, H.; Wang, G.; Zhao, H.; Cai, W. Nanosheets-built flowerlike micro/nanostructured Bi2O2.33 and its highly efficient iodine removal performances. Chem. Eng. J. 2016, 289, 219–230. [Google Scholar] [CrossRef]
- Liu, L.; Liu, W.; Zhao, X.; Chen, D.; Cai, R.; Yang, W.; Komarneni, S.; Yang, D. Selective capture of iodide from solutions by microrosette-like δ-Bi2O3. ACS Appl. Mater. Interfaces 2014, 6, 16082–16090. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Qi, B.; Liu, Y.; Zhao, L.; Jiao, Y.; Zhang, Y.; Jiang, Z.; Li, Q.; Wang, J.; Chen, S. AgII doped MIL-101 and its adsorption of iodine with high speed in solution. J. Solid State Chem. 2016, 237, 274–283. [Google Scholar] [CrossRef]
- Liu, S.; Wang, N.; Zhang, Y.; Li, Y.; Han, Z.; Na, P. Efficient removal of radioactive iodide ions from water by three-dimensional Ag2O-Ag/TiO2 composites under visible light irradiation. J. Hazard. Mater. 2015, 284, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre, G.; Walcarius, A.; Ehrhardt, J.J.; Bessière, J. Sorption of iodide on cuprite (Cu2O). Langmuir 2000, 16, 4519–4527. [Google Scholar] [CrossRef]
- Mao, P.; Liu, Y.; Jiao, Y.; Chen, S.; Yang, Y. Enhanced uptake of iodide on Ag@Cu2O nanoparticles. Chemosphere 2016, 164, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Qi, L.; Liu, X.; Liu, Y.; Jiao, Y.; Chen, S.; Yang, Y. Synthesis of Cu/Cu2O hydrides for enhanced removal of iodide from water. J. Hazard. Mater. 2017, 328, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Liu, Y.; Liu, X.; Wang, Y.; Liang, J.; Zhou, Q.; Dai, Y.; Jiao, Y.; Chen, S.; Yang, Y. Bimetallic AgCu/Cu2O hybrid for the synergetic adsorption of iodide from solution. Chemosphere 2017, 180, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Chen, M.; Fang, X.; Wu, L. Fabrication and application of inorganic hollow spheres. Chem. Soc. Rev. 2011, 40, 5472–5491. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, W. Template Synthesis of Multishelled Cu2O Hollow Spheres with a Single-Crystalline Shell Wall. Angew. Chem. Int. Ed. 2007, 46, 1489–1492. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, S.; Wang, H.; Yu, H.; Fang, Y.; Peng, F. Facile synthesis of self-assembled mesoporous CuO nanospheres and hollow Cu2O microspheres with excellent adsorption performance. RSC Adv. 2014, 4, 43024–43028. [Google Scholar] [CrossRef]
- Gao, J.; Li, Q.; Zhao, H.; Li, L.; Liu, C.; Gong, Q.; Qi, L. One-Pot Synthesis of Uniform Cu2O and CuS Hollow Spheres and Their Optical Limiting Properties. Chem. Mater. 2008, 20, 6263–6269. [Google Scholar] [CrossRef]
- Hung, L.-I.; Tsung, C.-K.; Huang, W.; Yang, P. Room-Temperature Formation of Hollow Cu2O Nanoparticles. Adv. Mater. 2010, 22, 1910–1914. [Google Scholar] [CrossRef] [PubMed]
- Oscarson, D.W.; Miller, H.G.; Watson, R.L. The potential effectiveness of mercury minerals in decreasing the level of iodine-129 in a nuclear fuel waste disposal vault. Nucl. Chem. Waste Manag. 1986, 6, 151–157. [Google Scholar] [CrossRef]
- Yao, W.; Li, F.-L.; Li, H.-X.; Lang, J.-P. Fabrication of hollow Cu2O@CuO-supported Au-Pd alloy nanoparticles with high catalytic activity through the galvanic replacement reaction. J. Mater. Chem. A 2015, 3, 4578–4585. [Google Scholar] [CrossRef]
- Yan, F.; Ma, R.; Ma, X.; Cui, K.; Wu, K.; Chen, M.; Li, Y. Ethanolysis of Kraft lignin to platform chemicals on a MoC1-x/Cu-MgAlOz catalyst. Appl. Catal. B 2017, 202, 305–313. [Google Scholar] [CrossRef]
- Mohamed, R.M.; Aazam, E.S. Preparation and characterization of core-shell polyaniline/mesoporous Cu2O nanocomposites for the photocatalytic oxidation of thiophene. Appl. Catal. A 2014, 480, 100–107. [Google Scholar] [CrossRef]
- Mamba, G.; Pulgarin, C.; Kiwi, J.; Bensimon, M.; Rtimi, S. Synchronic coupling of Cu2O(p)/CuO(n) semiconductors leading to Norfloxacin degradation under visible light: Kinetics, mechanism and film surface properties. J. Catal. 2017, 353, 133–140. [Google Scholar] [CrossRef]
- Haq, Z.; Bancroft, G.M.; Fyfe, W.S.; Bird, G.; Lopata, V.J. Sorption of iodide on copper. Environ. Sci. Technol. 1980, 14, 1106–1110. [Google Scholar] [CrossRef]
- Lefèvre, G.; Bessière, J.; Ehrhardt, J.-J.; Walcarius, A. Immobilization of iodide on copper(I) sulfide minerals. J. Environ. Radioact. 2003, 70, 73–83. [Google Scholar] [CrossRef]
- Liu, Y.; Gu, P.; Jia, L.; Zhang, G. An investigation into the use of cuprous chloride for the removal of radioactive iodide from aqueous solutions. J. Hazard. Mater. 2016, 302, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, Q.; Nadeem, R.; Iqbal, M.; Saeed, R.; Ansari, T.M. Organic acids pretreatment effect on Rosa bourbonia phyto-biomass for removal of Pb(II) and Cu(II) from aqueous media. Bioresour. Technol. 2013, 132, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Ullah, I.; Nadeem, R.; Iqbal, M.; Manzoor, Q. Biosorption of chromium onto native and immobilized sugarcane bagasse waste biomass. Ecol. Eng. 2013, 60, 99–107. [Google Scholar] [CrossRef]
- Iqbal, M.; Khera, R.A. Adsorption of copper and lead in single and binary metal system onto Fumaria indica biomass. Chem. Int. 2015, 1, 157b–163b. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Gao, S.; Yang, J.; Li, Z.; Jia, X.; Chen, Y. Bioinspired synthesis of hierarchically micro/nano-structured CuI tetrahedron and its potential application as adsorbent for Cd(II) with high removal capacity. J. Hazard. Mater. 2012, 211, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Sankapal, B.R.; Ennaoui, A.; Guminskaya, T.; Dittrich, T.; Bohne, W.; Röhrich, J.; Strub, E.; Lux-Steiner, M.C. Characterization of p-CuI prepared by the SILAR technique on Cu-tape/n-CuInS2 for solar cells. Thin Solid Films 2005, 480–481, 142–146. [Google Scholar] [CrossRef]
- Park, J.C.; Kim, J.; Kwon, H.; Song, H. Gram-Scale Synthesis of Cu2O Nanocubes and Subsequent Oxidation to CuO Hollow Nanostructures for Lithium-Ion Battery Anode Materials. Adv. Mater. 2009, 21, 803–807. [Google Scholar] [CrossRef]


| Sample | pH Value | Temperature (°C) | Removal Capacity (mmol g−1) | Ref. |
|---|---|---|---|---|
| S5 (Cu2O) | 7 | 25 | 0.03 | This work |
| S20 (24.2%-Cu/Cu2O) | 7 | 25 | 0.20 | This work |
| S40 (60.6%-Cu/Cu2O) | 7 | 25 | 0.25 | This work |
| S60 (Cu) | 7 | 25 | 0.26 | This work |
| Cu2O a | 6.7 | 25 | 0.002 | Ref. [13] |
| 31.8%-Cu/Cu2O | 7 | 25 | 0.18 | Ref. [14,15] |
| Cu2O | 7 | 25 | 0.02 | Ref. [14,15] |
| 1.0%-Ag@Cu2O | 7 | 25 | 0.20 | Ref. [14] |
| Cu | 7 | 22 | 0.05 | Ref. [27] |
| CuO | 7 | 22 | 0.002 | Ref. [27] |
| Cu2O | 7 | 22 | 0.016 | Ref. [27] |
| Cu2S a | 7 | 25 | 0.048 | Ref. [28] |
| CuCl b | 7 | 25 | 2.0 | Ref. [29] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mao, P.; Jiang, J.; Pan, Y.; Duanmu, C.; Chen, S.; Yang, Y.; Zhang, S.; Chen, Y. Enhanced Uptake of Iodide from Solutions by Hollow Cu-Based Adsorbents. Materials 2018, 11, 769. https://doi.org/10.3390/ma11050769
Mao P, Jiang J, Pan Y, Duanmu C, Chen S, Yang Y, Zhang S, Chen Y. Enhanced Uptake of Iodide from Solutions by Hollow Cu-Based Adsorbents. Materials. 2018; 11(5):769. https://doi.org/10.3390/ma11050769
Chicago/Turabian StyleMao, Ping, Jinlong Jiang, Yichang Pan, Chuansong Duanmu, Shouwen Chen, Yi Yang, Songlan Zhang, and Yonghao Chen. 2018. "Enhanced Uptake of Iodide from Solutions by Hollow Cu-Based Adsorbents" Materials 11, no. 5: 769. https://doi.org/10.3390/ma11050769