A Study of the Stability Mechanism of the Dispersed Particle Gel Three-Phase Foam Using the Interfacial Dilational Rheology Method
Abstract
:1. Introduction
2. Experimental Section
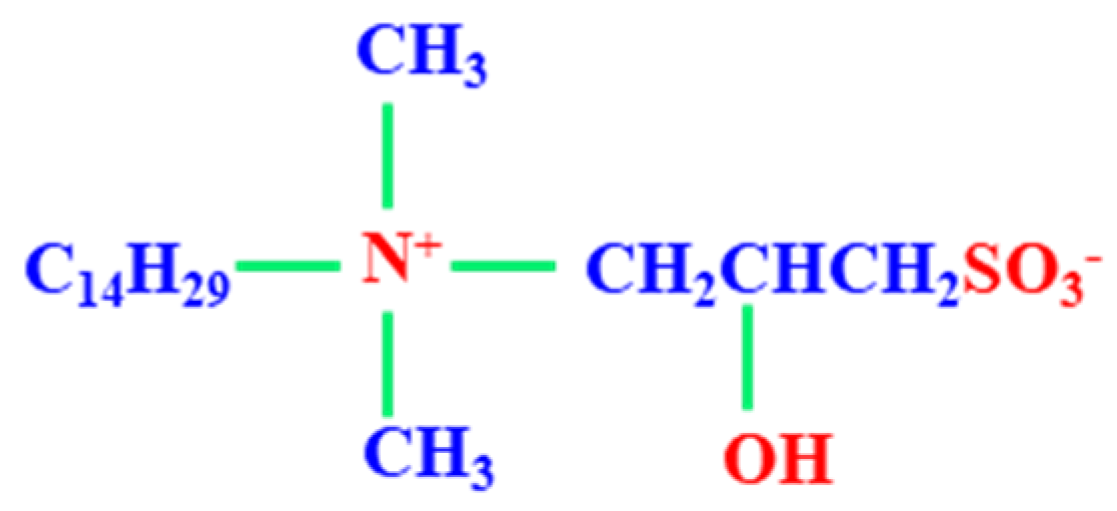
2.1. Materials
2.2. Preparation of the DPG Three-Phase Foam
2.3. Interfacial Dilational Rheology
3. Results and Discussion
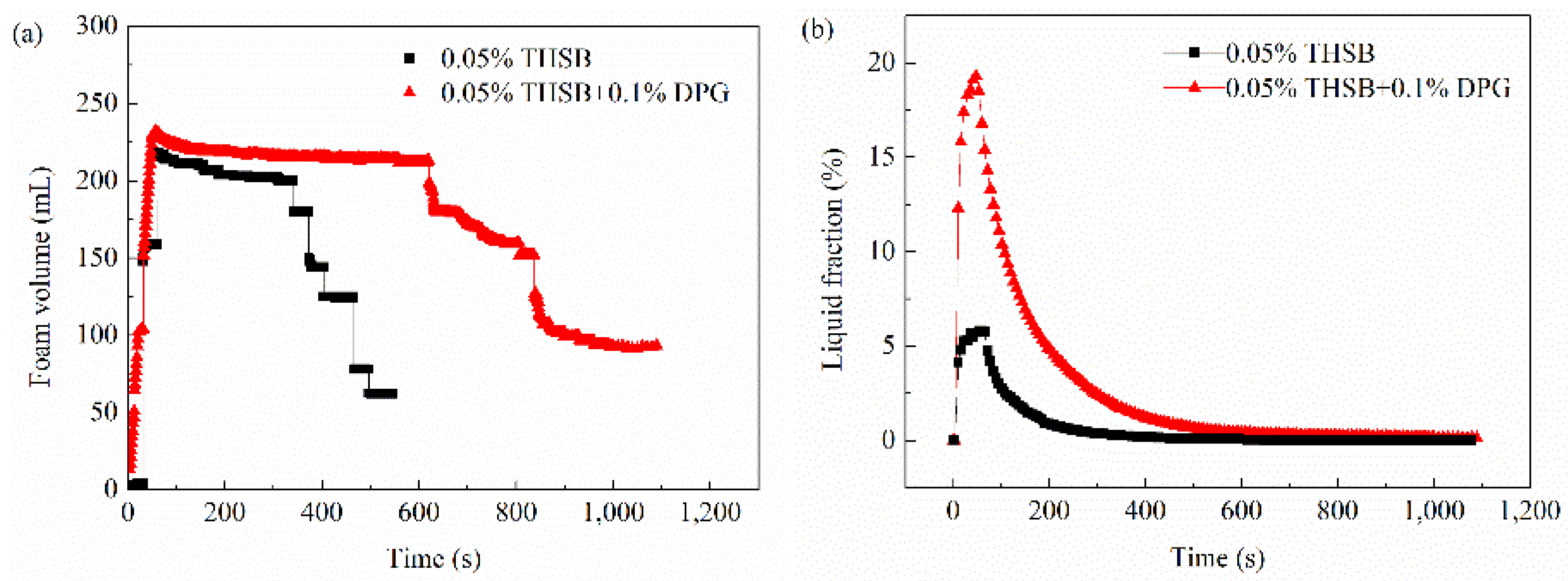
3.1. Evaluation of Foam Stability
3.2. Interfacial Rheology and Factors Influencing Foam Stability
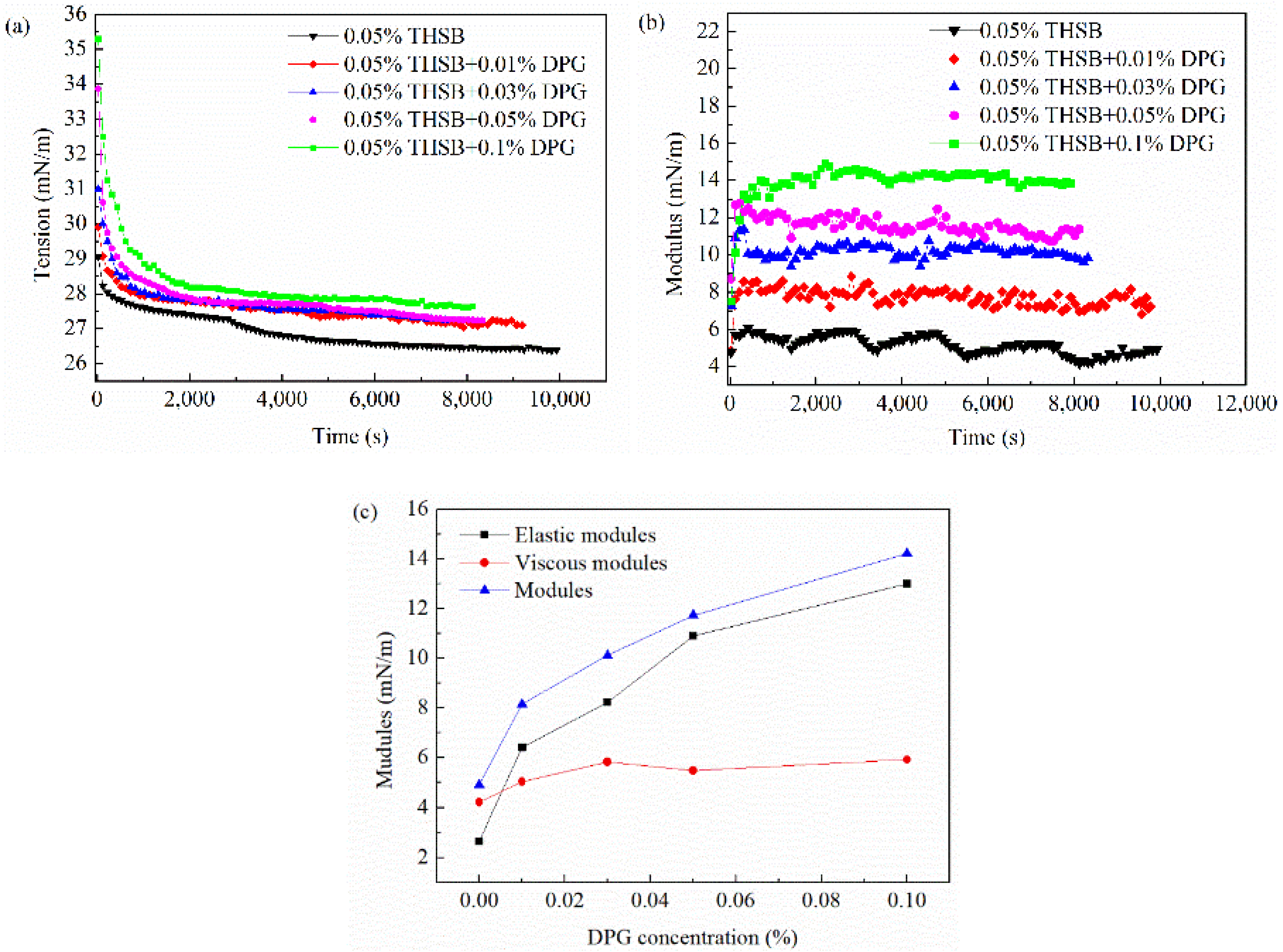
3.2.1. Influence of the DPG Concentration
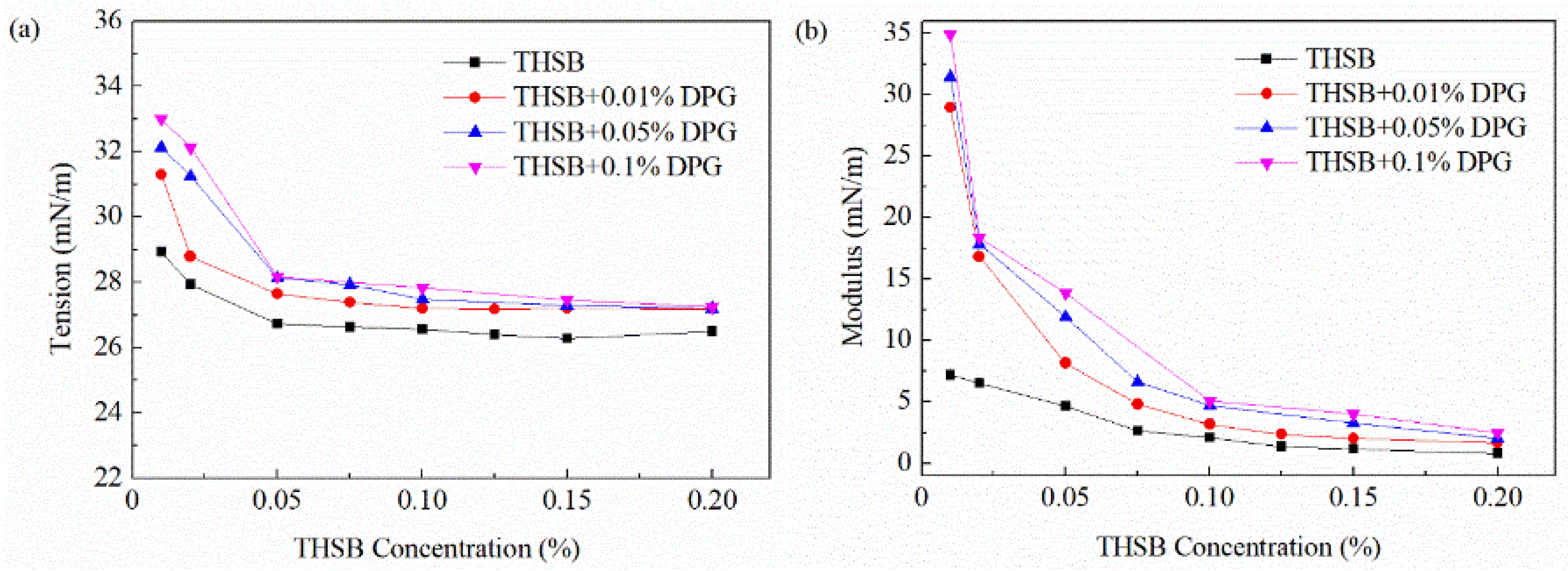
3.2.2. Influence of the Surfactant Concentration
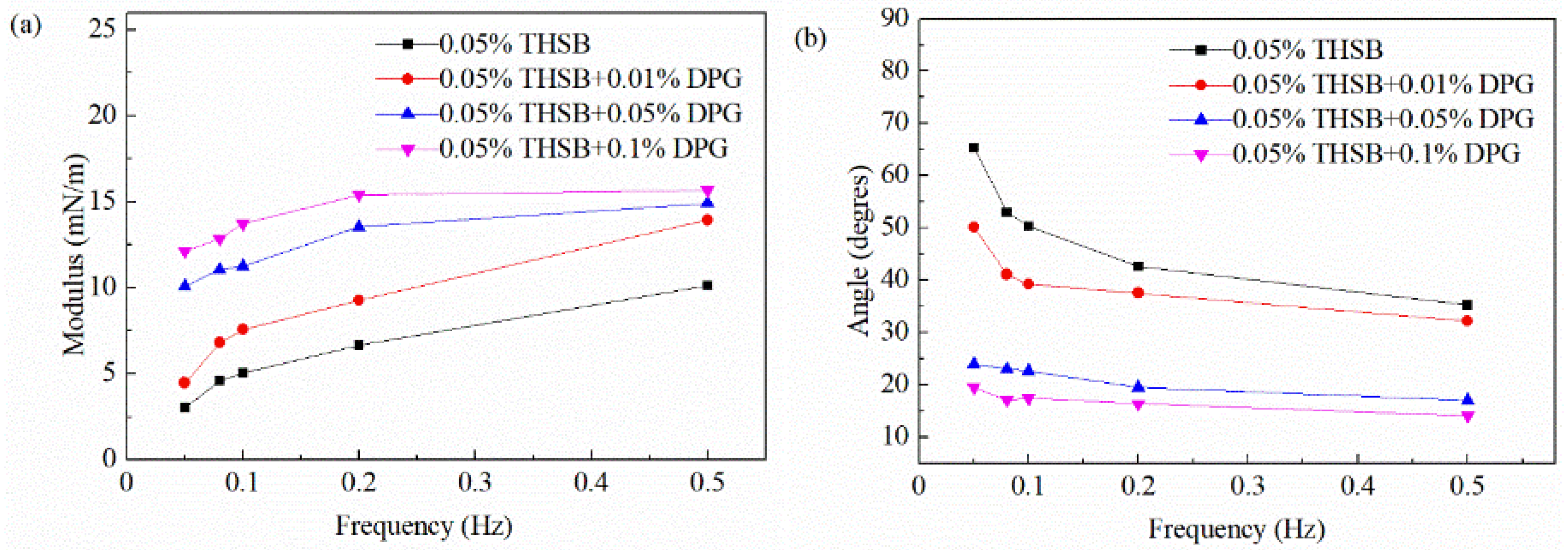
3.2.3. Influence of Oscillation Frequency
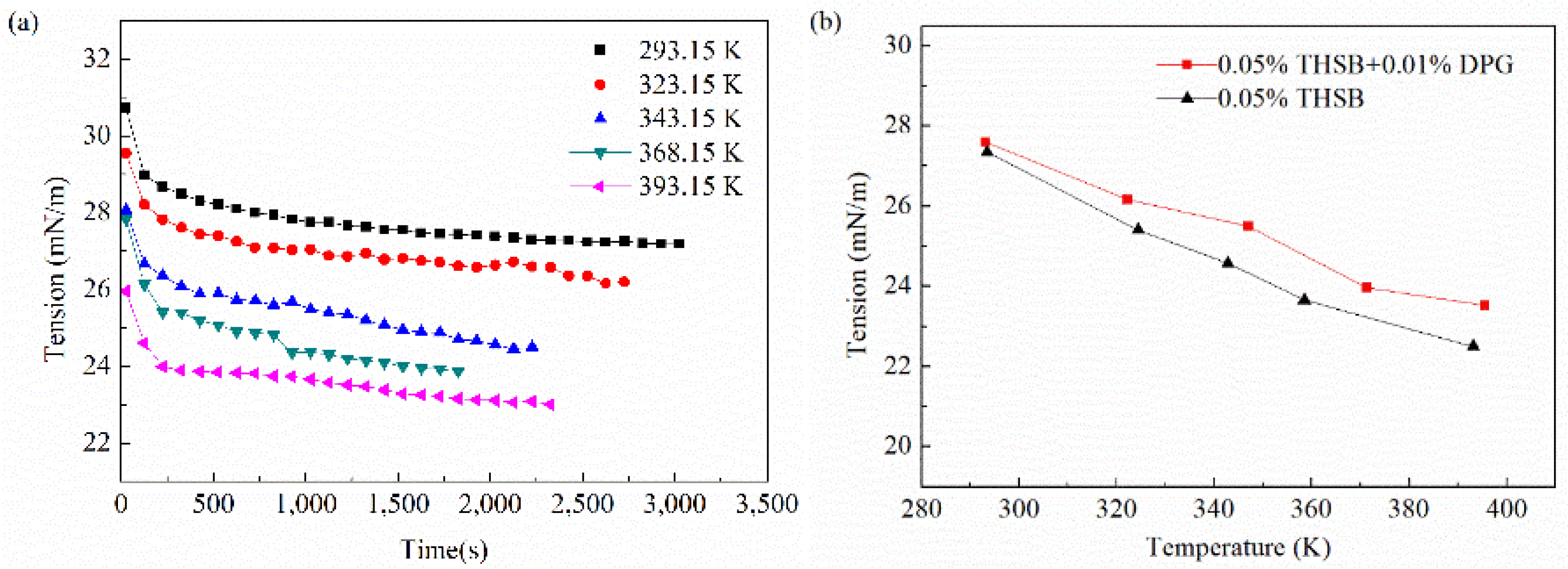
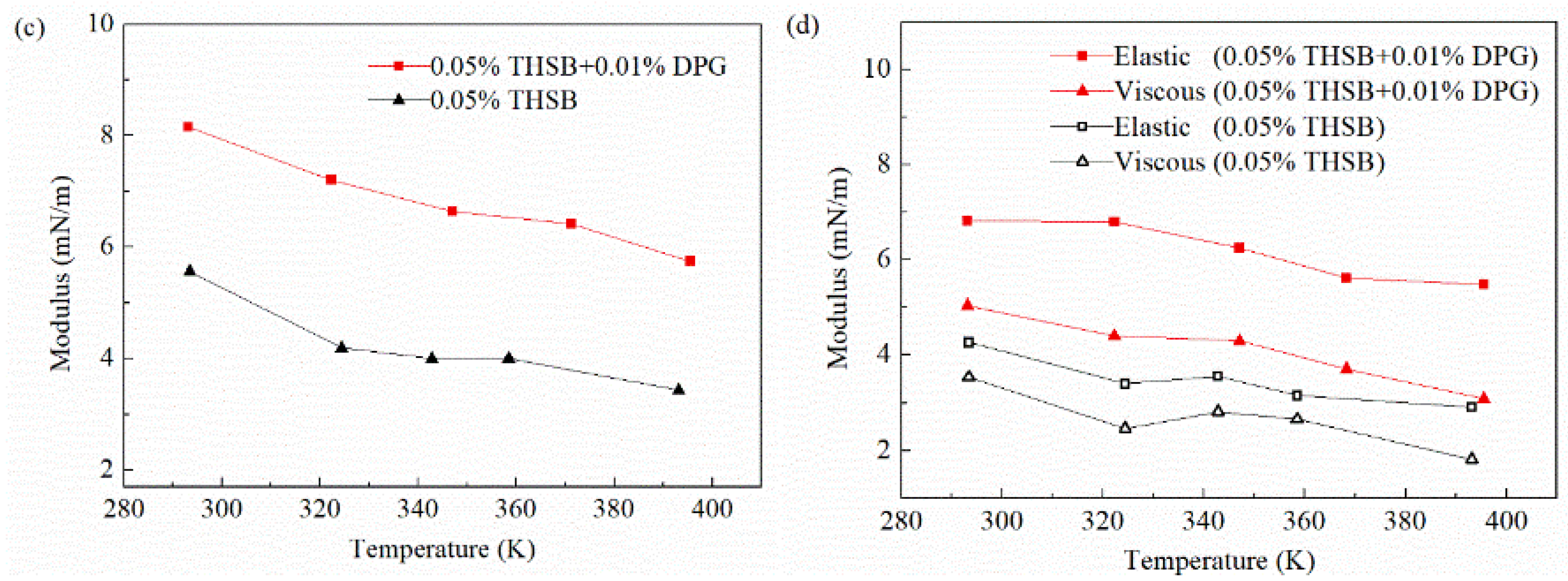
3.2.4. Influence of Temperature
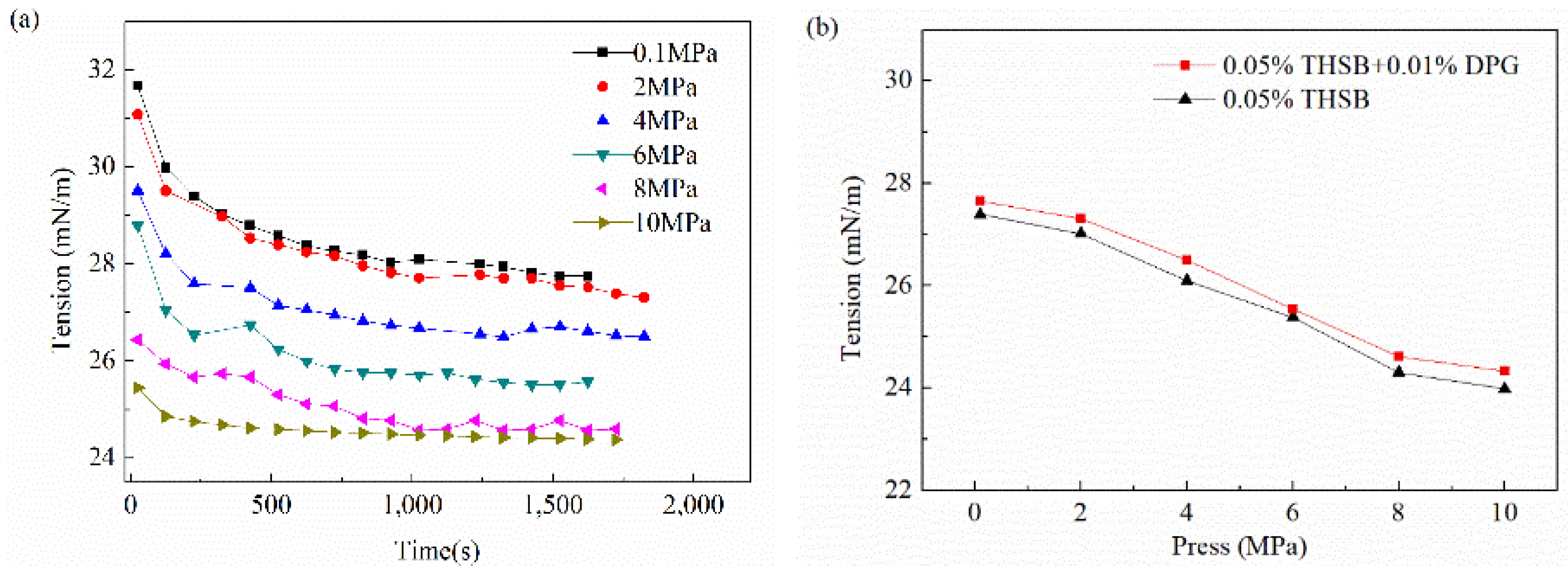
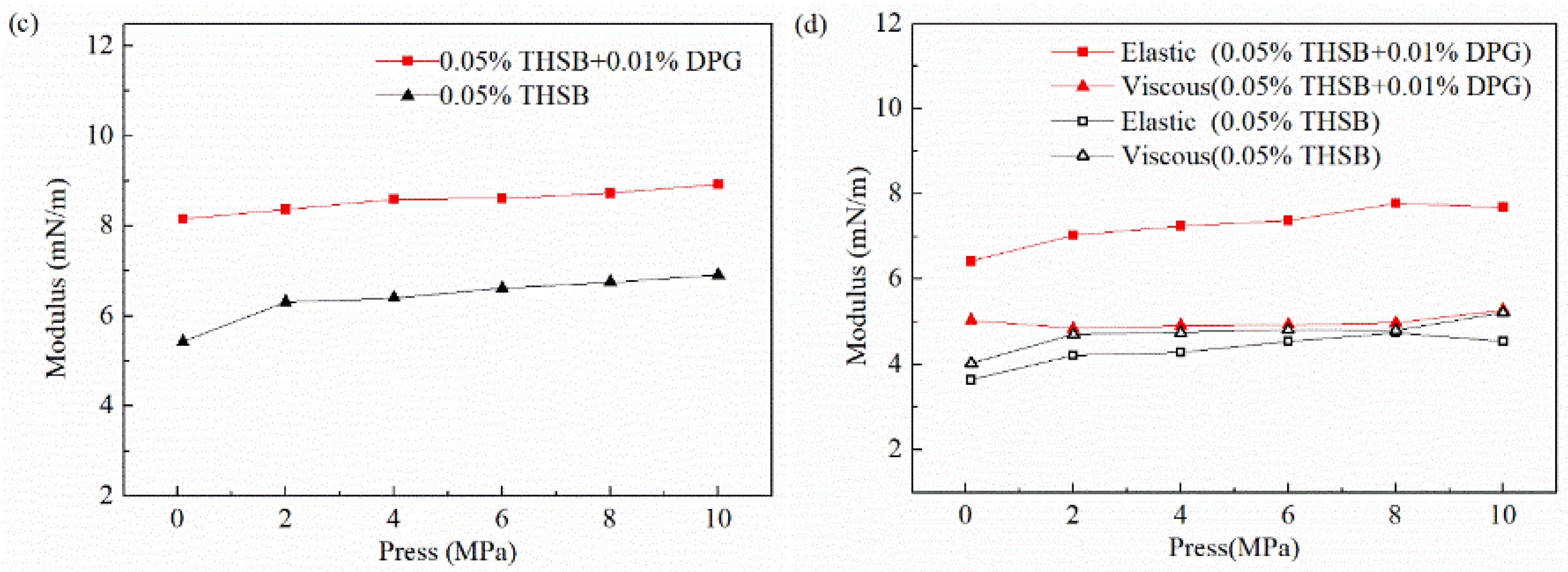
3.2.5. Influence of Pressure
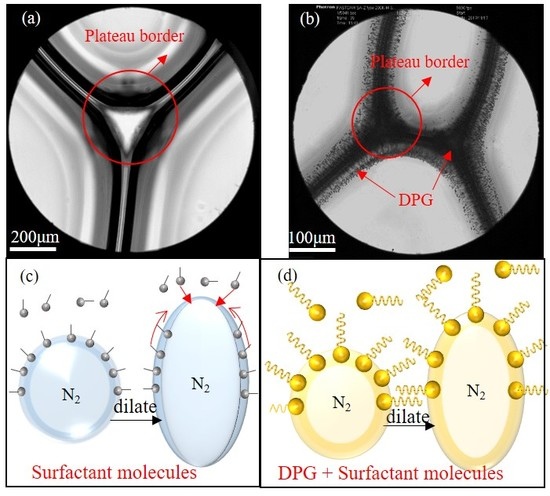
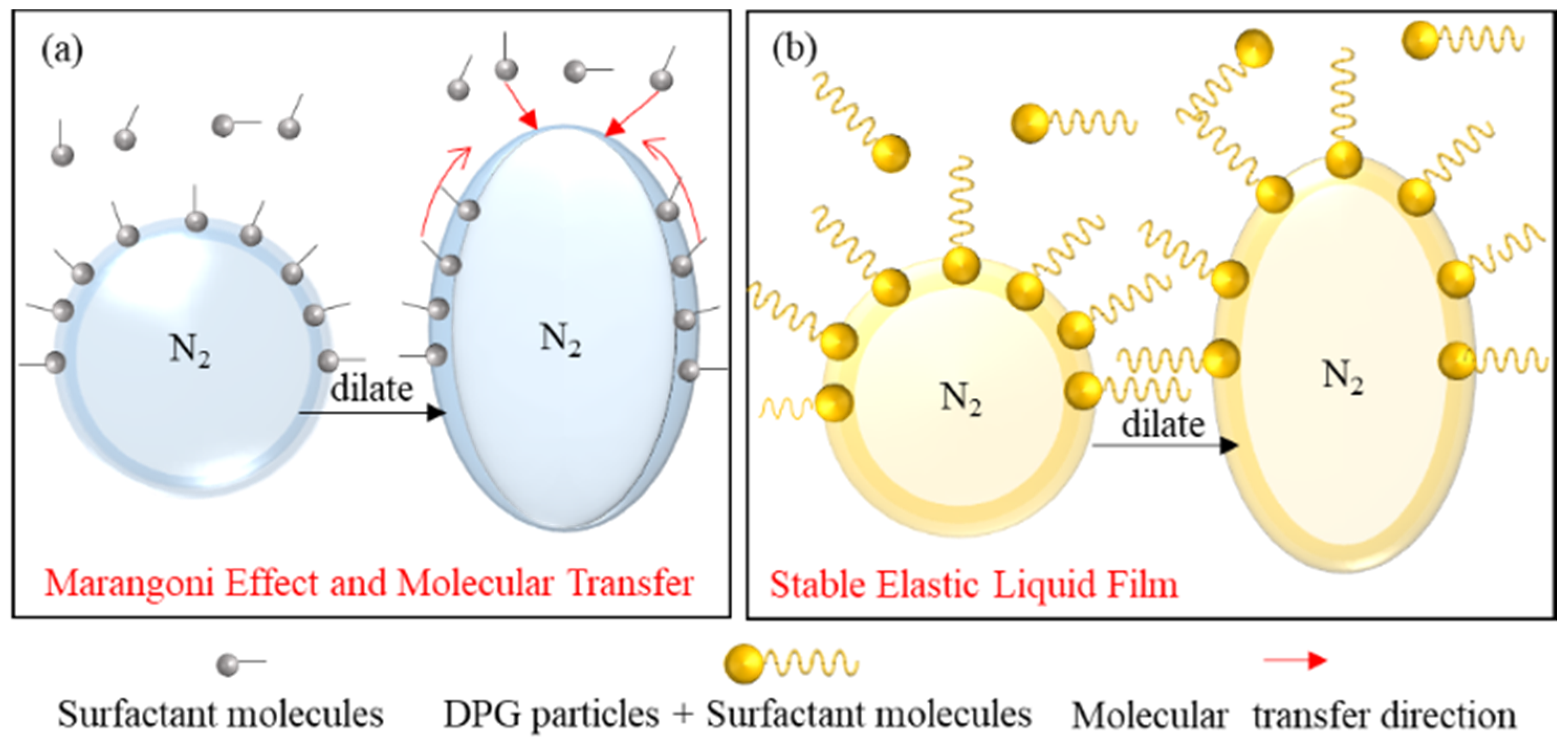
3.3. Analysis of the Foam Stabilization Mechanism
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Shchukin, E.D.; Pertsov, A.; Amelina, E.; Zelenev, A. Colloid and Surface Chemistry; Elsevier: New York, NY, USA, 2001; Volume 12. [Google Scholar]
- Murray, B.S. Stabilization of bubbles and foams. Curr. Opin. Colloids Interface Sci. 2007, 12, 232–241. [Google Scholar] [CrossRef]
- Wang, X.; Dong, M.; Zhou, W. Polymer/gel enhanced foam flood for improving post-waterflood heavy oil recovery. In Proceedings of the SPE Heavy Oil Conference-Canada, Calgary, AB, Canada, 11–13 June 2013; Society of Petroleum Engineers: Richardson, TX, USA, 2013. [Google Scholar]
- Chen, S.; Jian, G.; Hou, Q.; Gao, S.; Luo, Y.; Zhu, Y.; Li, W. Effect of oil on the stability of polymer enhanced foams. Int. J. Oil Gas Coal Technol. 2013, 6, 675–688. [Google Scholar] [CrossRef]
- Emrani, A.S.; Nasr-El-Din, H.A. An experimental study of nanoparticle-polymer-stabilized CO2 foam. Colloids Surf. A 2017, 524, 17–27. [Google Scholar] [CrossRef]
- Tang, L.; Fang, H.B.; Zong, H.; Zhang, L.; Jin, Z.Q.; Xu, Z.C.; Zhang, L.; Zhao, S. Dilational rheological properties of interfacial films containing Branch-Preformed particle gel and crude oil fractions. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Binks, B.P.; Kirkland, M.; Rodrigues, J.A. Origin of stabilisation of aqueous foams in nanoparticle–surfactant mixtures. Soft Matter 2008, 4, 2373–2382. [Google Scholar] [CrossRef]
- Fang, J.; Dai, C.; Yan, Y.; Yan, Z.; You, Q. Enhanced Foam Stability by Adding Dispersed Particle Gel: A New 3-Phase Foam Study. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 11–13 August 2015; Society of Petroleum Engineers: Richardson, TX, USA, 2015. [Google Scholar]
- Zhao, G.; Dai, C.; Wen, D.; Fang, J. Stability mechanism of a novel three—Phase foam by adding dispersed particle gel. Colloids Surf. A 2016, 497, 214–224. [Google Scholar] [CrossRef]
- Richardson, R.M.; Pelton, R.; Cosgrove, T.; Zhang, J. A Neutron Reflectivity Study of Poly (N-isopropylacrylamide) at the Air-Water Interface with and without Sodium Dodecyl Sulfate. Macromolecules 2000, 33, 6269–6274. [Google Scholar] [CrossRef]
- Noskov, B.; Alexandrov, D.; Loglio, G.; Miller, R. Characterisation of adsorbed polymer film structure by dynamic surface tension and dilational elasticity. Colloids Surf. A 1999, 156, 307–313. [Google Scholar] [CrossRef]
- Maldonado-Valderrama, J.; Patino, J.M.R. Interfacial rheology of protein–surfactant mixtures. Curr. Opin. Colloids Interface Sci. 2010, 15, 271–282. [Google Scholar] [CrossRef]
- Monteux, C.; Fuller, G.G.; Bergeron, V. Shear and dilational surface rheology of oppositely charged polyelectrolyte/surfactant microgels adsorbed at the air-water interface. Influence on foam stability. J. Phys. Chem. B 2004, 108, 16473–16482. [Google Scholar] [CrossRef]
- Bhamla, M.S.; Giacomin, C.E.; Balemans, C.; Fuller, G.G. Influence of interfacial rheology on drainage from curved surfaces. Soft Matter 2014, 10, 6917–6925. [Google Scholar] [CrossRef] [PubMed]
- Narsimhan, G.; Wang, Z. Stability of thin stagnant film on a solid surface with a viscoelastic air–liquid interface. J. Colloid Interface Sci. 2005, 291, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Nguyen, A.V.; Farrokhpay, S. Effects of surface rheology and surface potential on foam stability. Colloids Surf. A 2016, 488, 70–81. [Google Scholar] [CrossRef]
- Li, Z.-Q.; Zhang, L.; Yan, F.; Zhang, L.; Zhao, S.; Yu, J.-Y. Surface dilational properties of N-acyltaurate amphiphiles with aromatic side chains. Acta Phys.-Chim. Sin. 2009, 25, 1939–1944. [Google Scholar]
- Cui, X.-H.; Zhang, L.; Zhao, R.-H.; Luo, L.; Zhang, L.; Zhao, S.; Yu, J.-Y. Interfacial Interactions Among Aromatic Side Chained N-Acyltaurates Studied by Interfacial Tension Relaxation Measurements. Chem. J. Chin. Univ. 2011, 32, 1556–1562. [Google Scholar]
- Wang, H.; Gong, Y.; Lu, W.; Chen, B. Influence of nano-SiO2 on dilational viscoelasticity of liquid/air interface of cetyltrimethyl ammonium bromide. Appl. Surf. Sci. 2008, 254, 3380–3384. [Google Scholar] [CrossRef]
- Dong, X.; Xu, J.; Cao, C.; Sun, D.; Jiang, X. Aqueous foam stabilized by hydrophobically modified silica particles and liquid paraffin droplets. Colloids Surf. A 2010, 353, 181–188. [Google Scholar] [CrossRef]
- Noskov, B.; Mikhailovskaya, A.; Lin, S.-Y.; Loglio, G.; Miller, R. Bovine serum albumin unfolding at the air/water interface as studied by dilational surface rheology. Langmuir 2010, 26, 17225–17231. [Google Scholar] [CrossRef] [PubMed]
- Noskov, B.A. Dilational surface rheology of polymer and polymer/surfactant solutions. Curr. Opin. Colloids Interface Sci. 2010, 15, 229–236. [Google Scholar] [CrossRef]
- Yarranton, H.; Sztukowski, D.; Urrutia, P. Effect of interfacial rheology on model emulsion coalescence: I. Interfacial rheology. J. Colloids Interface Sci. 2007, 310, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Bhamla, M.S.; Chai, C.; Àlvarez-Valenzuela, M.A.; Tajuelo, J.; Fuller, G.G. Interfacial mechanisms for stability of surfactant-laden films. PLoS ONE 2017, 12, e0175753. [Google Scholar] [CrossRef] [PubMed]
- Yue, L.; He, Z.-M.; Wang, Y.-Q.; Shang, Y.-Z.; Liu, H.-L. Effect of the surfactant C12mimBr on the aggregation behavior of gemini12-2-12 at an air/water interface, investigated using an interfacial dilational rheology method. Acta Phys.-Chim. Sin. 2014, 30, 2291–2299. [Google Scholar]
- Rane, J.P.; Pauchard, V.; Couzis, A.; Banerjee, S. Interfacial rheology of asphaltenes at oil–water interfaces and interpretation of the equation of state. Langmuir 2013, 29, 4750–4759. [Google Scholar] [CrossRef] [PubMed]
- Samaniuk, J.R.; Hermans, E.; Verwijlen, T.; Pauchard, V.; Vermant, J. Soft-glassy rheology of asphaltenes at liquid interfaces. J. Dispers. Sci. Technol. 2015, 36, 1444–1451. [Google Scholar] [CrossRef]
- Simjoo, M.; Rezaei, T.; Andrianov, A.; Zitha, P. Foam stability in the presence of oil: Effect of surfactant concentration and oil type. Colloids Surf. A 2013, 438, 148–158. [Google Scholar] [CrossRef]
- Subramaniam, A.B.; Abkarian, M.; Mahadevan, L.; Stone, H.A. Mechanics of interfacial composite materials. Langmuir 2006, 22, 10204–10208. [Google Scholar] [CrossRef] [PubMed]
- Zang, D.; Rio, E.; Langevin, D.; Wei, B.; Binks, B. Viscoelastic properties of silica nanoparticle monolayers at the air-water interface. Eur. Phys. J. E Soft Matter Biol. Phys. 2010, 31, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Deo, P.; Somasundaran, P. Interactions of hydrophobically modified polyelectrolytes with nonionic surfactants. Langmuir 2005, 21, 3950–3956. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Khin, C.C.; Chen, S.B.; Chen, B.-H. Nonionic surfactant and temperature effects on the viscosity of hydrophobically modified hydroxyethyl cellulose solutions. J. Phys. Chem. B 2005, 109, 14198–14204. [Google Scholar] [CrossRef] [PubMed]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, X.; Yi, P.; Zhao, G.; Sun, X.; Dai, C. A Study of the Stability Mechanism of the Dispersed Particle Gel Three-Phase Foam Using the Interfacial Dilational Rheology Method. Materials 2018, 11, 699. https://doi.org/10.3390/ma11050699
Yao X, Yi P, Zhao G, Sun X, Dai C. A Study of the Stability Mechanism of the Dispersed Particle Gel Three-Phase Foam Using the Interfacial Dilational Rheology Method. Materials. 2018; 11(5):699. https://doi.org/10.3390/ma11050699
Chicago/Turabian StyleYao, Xue, Ping Yi, Guang Zhao, Xin Sun, and Caili Dai. 2018. "A Study of the Stability Mechanism of the Dispersed Particle Gel Three-Phase Foam Using the Interfacial Dilational Rheology Method" Materials 11, no. 5: 699. https://doi.org/10.3390/ma11050699
APA StyleYao, X., Yi, P., Zhao, G., Sun, X., & Dai, C. (2018). A Study of the Stability Mechanism of the Dispersed Particle Gel Three-Phase Foam Using the Interfacial Dilational Rheology Method. Materials, 11(5), 699. https://doi.org/10.3390/ma11050699