Microfluidic Fabrication of Morphology-Controlled Polymeric Microspheres of Blends of Poly(4-butyltriphenylamine) and Poly(methyl methacrylate)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
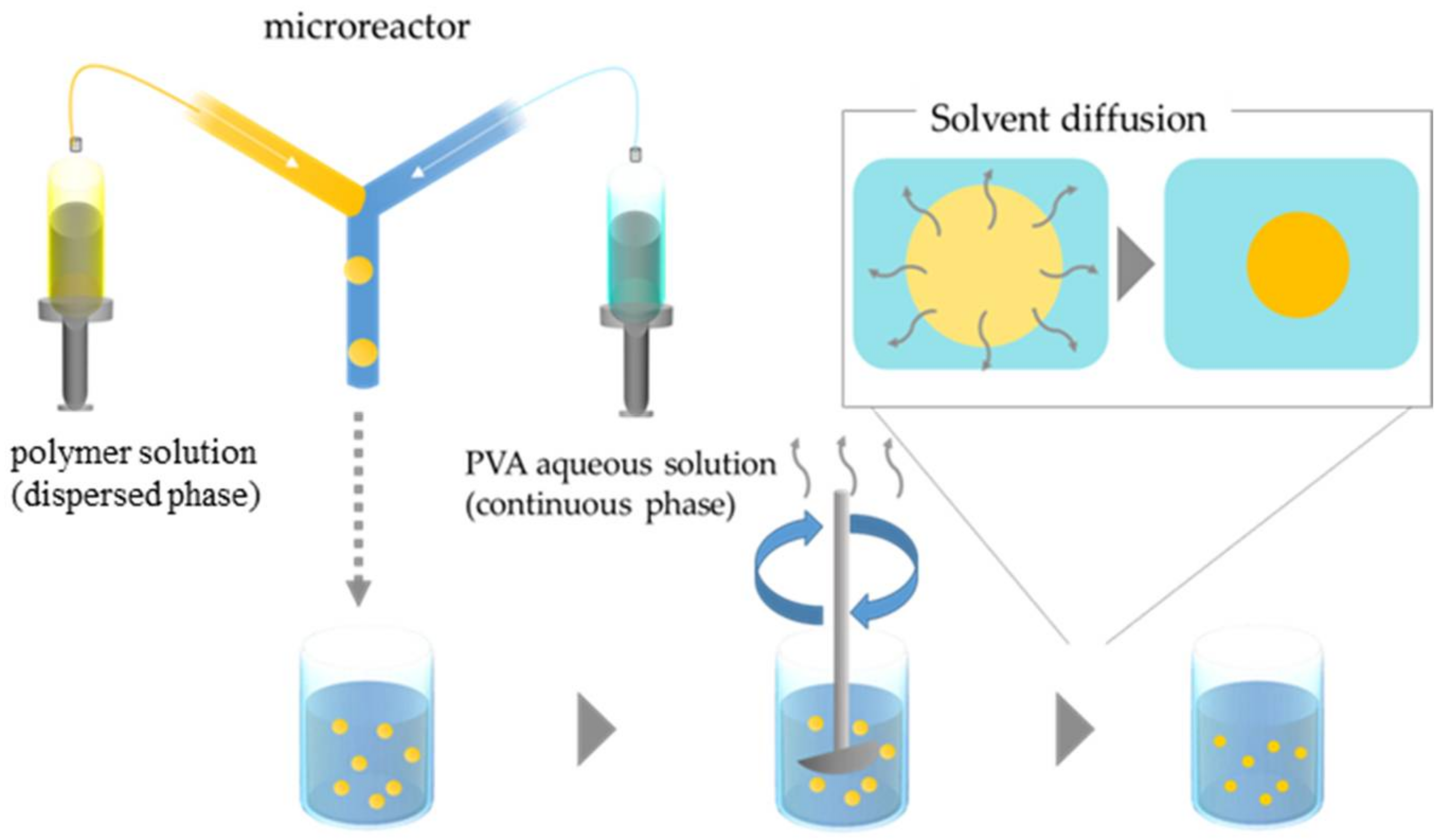
2.2. Microfluidic Device and Particle Fabrication
3. Results
3.1. Effect of Flow Rate of Continuous Phase and Concentration of Dispersed Phase
3.2. Effect of Molecular Weight of PMMA
3.3. Effect of SDS
4. Discussion
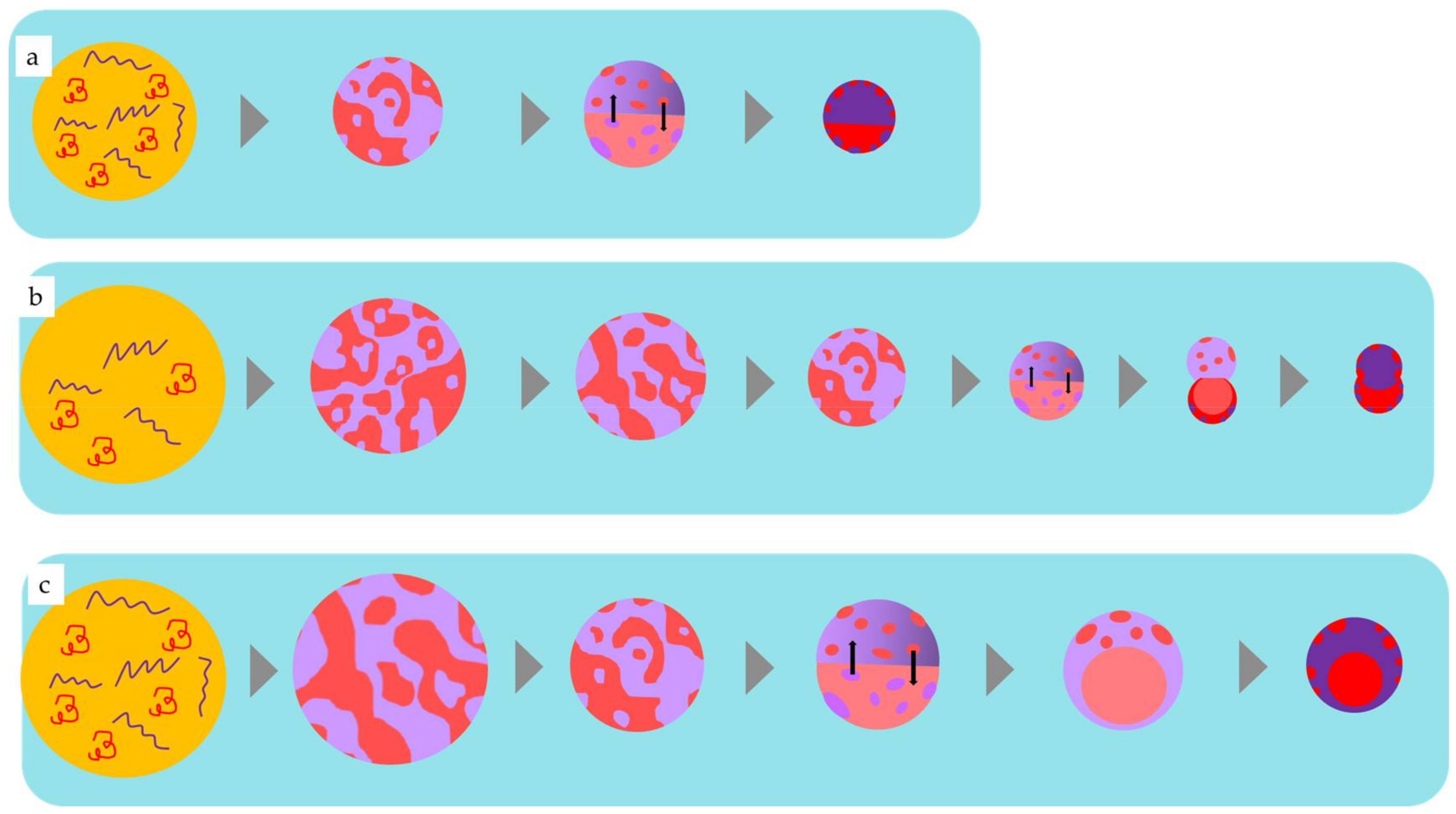
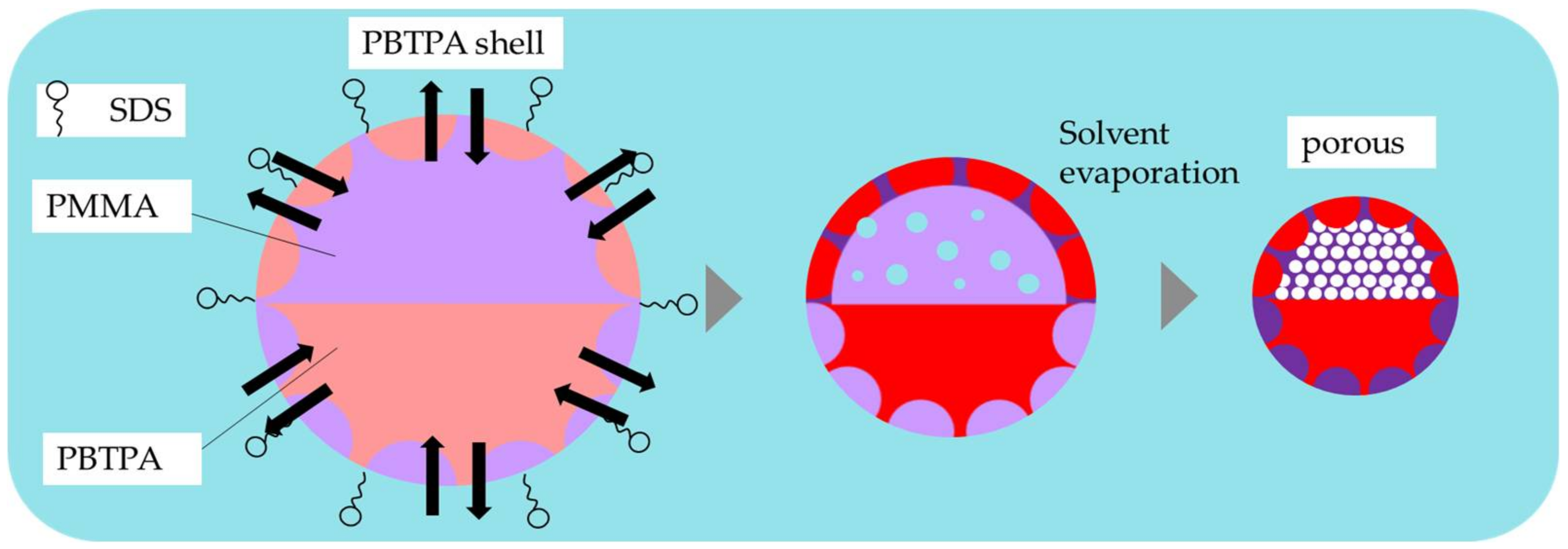
4.1. Phase Separation of Polymers in Emulsion Drops
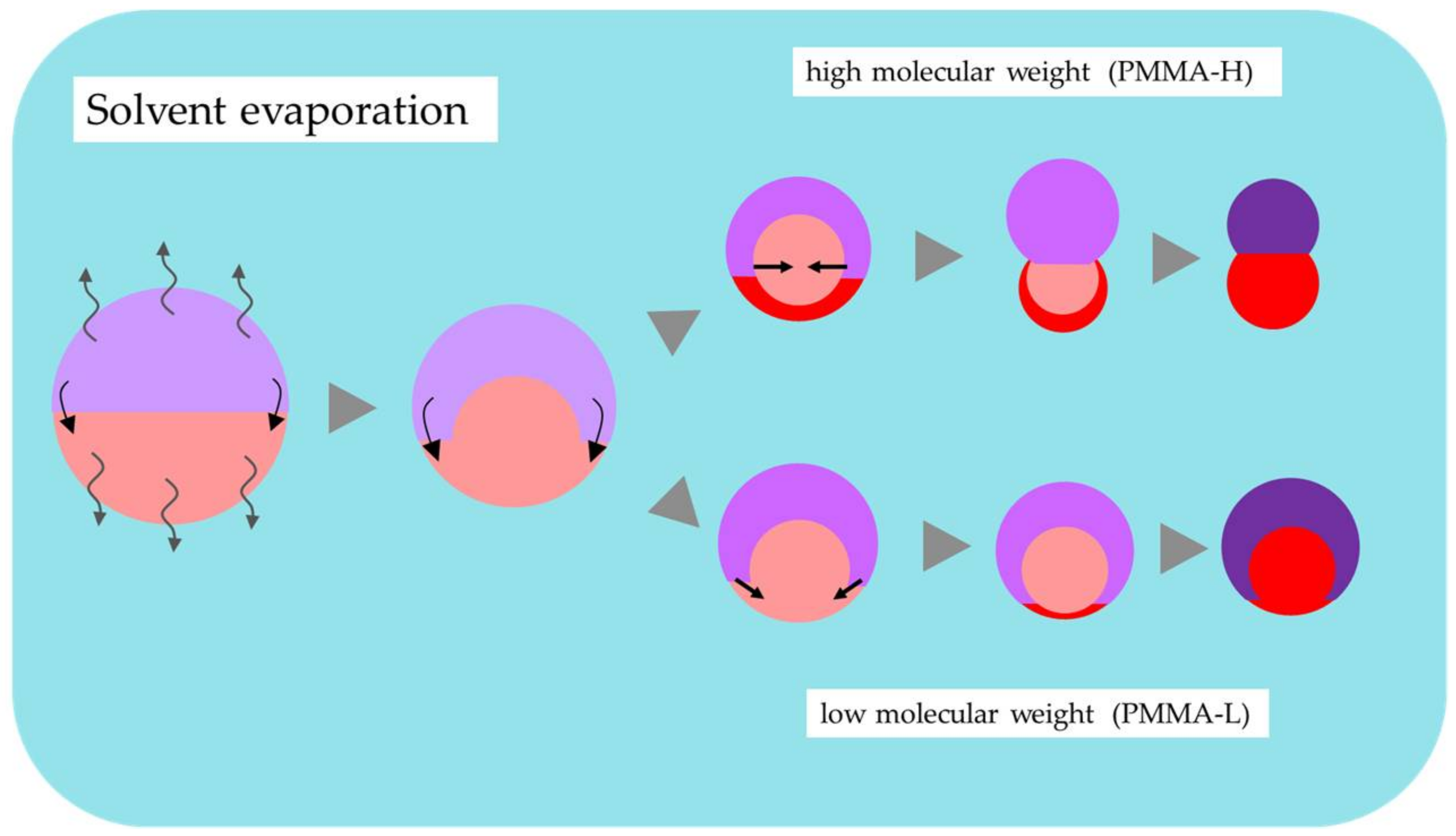
4.2. Effect of Molecular Weight of PMMA
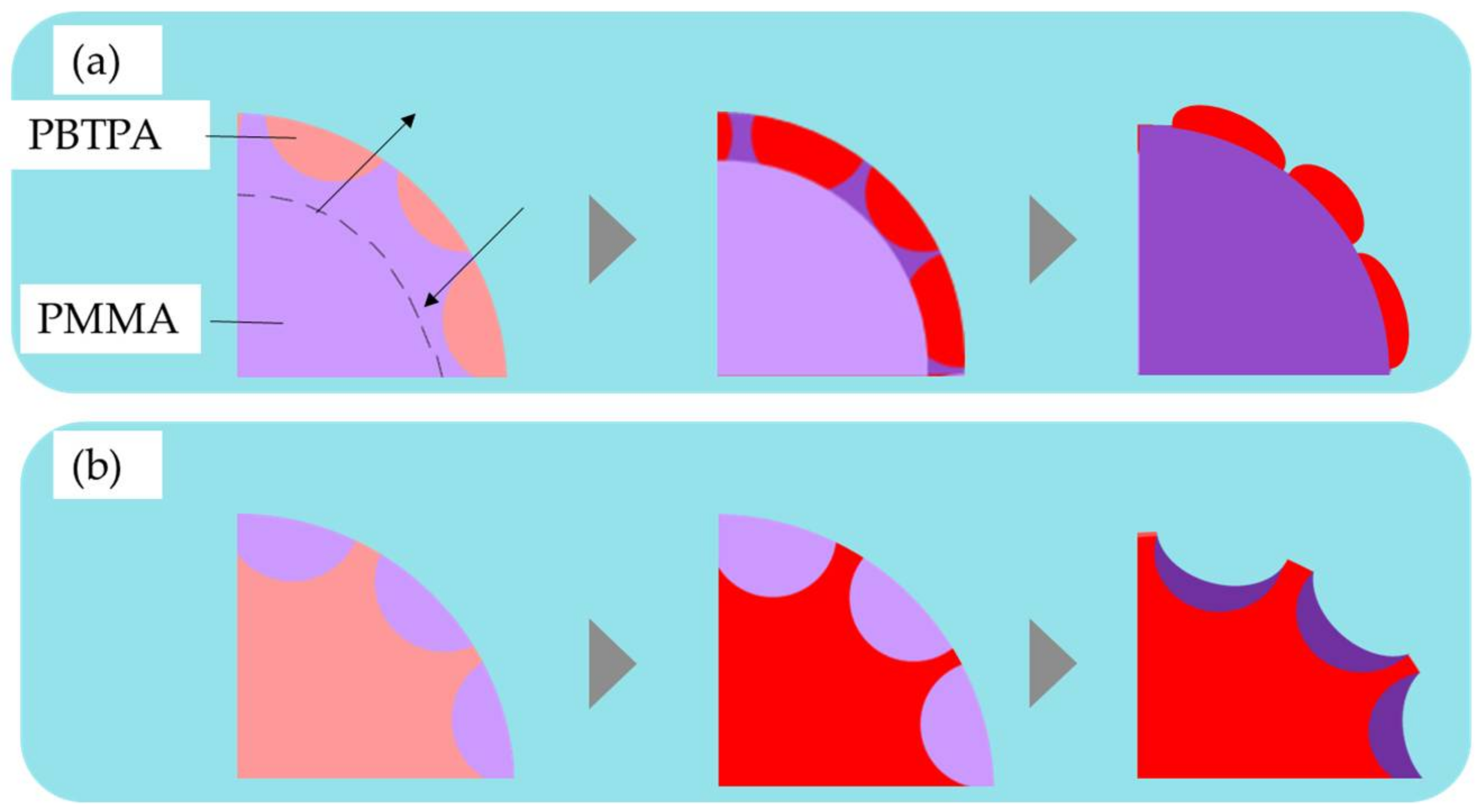
4.3. Effect of Surfactant
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mora-Huertas, C.E.; Fessi, H.; Elaissari, A. Polymer-based nanocapsules for drug delivery. Int. J. Pharm. 2010, 385, 113–142. [Google Scholar] [CrossRef] [PubMed]
- Nisisako, T.; Torii, T.; Takahashi, T.; Takizawa, Y. Synthesis of monodisperse bicolored Janus particles with electrical anisotropy using a microfluidic co-flow system. Adv. Mater. 2006, 18, 1152–1156. [Google Scholar] [CrossRef]
- Tu, F.; Lee, D. Shape-changing and amphiphilicity-reversing Janus particles with pH-responsive surfactant properties. J. Am. Chem. Soc. 2014, 136, 9999–10006. [Google Scholar] [CrossRef] [PubMed]
- Charreyre, M.T.; Boullanger, P.; Delair, T.; Pichot, C. Preparation and characterization of polystyrene latexes bearing disaccharide surface groups. Colloid Polym. Sci. 1993, 271, 668–679. [Google Scholar] [CrossRef]
- Urakami, Y.; Kasuya, Y.; Fujimoto, K.; Miyamoto, M.; Kawaguchi, H. Phagocytosis of microspheres with modified surfaces. Colloids Surf. B 1994, 3, 183–190. [Google Scholar] [CrossRef]
- Kawamura, A.; Kohri, M.; Morimoto, G.; Nannichi, Y.; Taniguchi, T.; Kishikawa, K. Full-color biomimetic photonic materials with iridescent and non-iridescent structural colors. Sci. Rep. 2016, 6, 33984. [Google Scholar] [CrossRef] [PubMed]
- Okubo, M.; Saito, N.; Fujibayashi, T. Preparation of polystyrene/poly(methyl methacrylate) composite particles having a dent. Colloid Polym. Sci. 2005, 283, 691–698. [Google Scholar] [CrossRef]
- Ge, X.; Wang, M.; Ji, X.; Ge, X.; Liu, H. Effects of concentration of nonionic surfactant and molecular weight of polymers on the morphology of anisotropic polystyrene/poly(methyl methacrylate) composite particles prepared by solvent evaporation method. Colloid Polym. Sci. 2009, 287, 819–827. [Google Scholar] [CrossRef]
- Tanaka, T.; Nakatsuru, R.; Kagari, Y.; Saito, N.; Okubo, M. Effect of molecular weight on the morphology of polystyrene/poly(methyl methacrylate) composite particles prepared by the solvent evaporation method. Langmuir 2008, 24, 12267–12271. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, T.; Tajima, A.; Yabu, H.; Shimomura, M. Suprapolymer structures from nanostructured polymer particles. Angew. Chem. Int. Ed. 2009, 48, 5125–5128. [Google Scholar] [CrossRef] [PubMed]
- Motoyoshi, K.; Tajima, A.; Higuchi, T.; Yabu, H.; Shimomura, M. Static and dynamic control of phase separation structures in nanoparticles of polymer blends. Soft Matter 2010, 6, 1253–1257. [Google Scholar] [CrossRef]
- Tanaka, T.; Saito, N.; Okubo, M. Control of Layer Thickness of Onionlike Multilayered Composite Polymer Particles Prepared by the Solvent Evaporation Method. Macromolecules 2009, 42, 7423–7429. [Google Scholar] [CrossRef]
- Taherzadeh, H.; Ogino, K. Fabrication of microspheres based on poly(4-butyltriphenylamine) blends with poly(methyl methacrylate) and block copolymer by solvent evaporation method. Open J. Org. Polym. Mater. 2015, 5, 37–42. [Google Scholar] [CrossRef][Green Version]
- Kikuchi, S.; Kanehashi, S.; Ogino, K. Effect of solvent and surfactant on morphology of poly(4-butyltriphenylamine) and poly(methyl methacrylate) blend particles prepared by solvent evaporation method. 2018. manuscript in preparation. [Google Scholar]
- Kandori, K.; Kishi, K.; Ishikawa, T. Preparation of monodispersed W/O emulsions by Shirasu-porous-glass filter emulsification technique. Colloids Surf. 1991, 55, 73–78. [Google Scholar] [CrossRef]
- Kawakatsu, T.; Kikuchi, Y.; Nakajima, M. Regular-sized cell creation in microchannel emulsification by visual micro-processing method. J. Am. Oil Chem. Soc. 1997, 74, 317–321. [Google Scholar] [CrossRef]
- Nisisako, T.; Torii, T.; Higuchi, T. Novel microreactors for functional polymer beads. Chem. Eng. J. 2004, 101, 23–29. [Google Scholar] [CrossRef]
- Nisisako, T.; Okushima, S.; Torii, T. Controlled formulation of monodisperse double emulsions in a multiple-phase microfluidic system. Soft Matter 2005, 1, 23–27. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Shimomura, T.; Ogino, K. Preparation of diblock copolymer based on poly(4-n-butyltriphenylamine). Polymer 2009, 50, 95–101. [Google Scholar] [CrossRef]
- Broseta, D.; Leibler, L.; Kaddour, L.O.; Strazielle, C. A theoretical and experimental study of interfacial tension of immiscible polymer blends in solution. J. Chem. Phys. 1987, 87, 7248–7256. [Google Scholar] [CrossRef]
- Gao, X.; Gao, F.; Chen, L.; Yao, Y.; Chen, T.; Lin, S. Tuning the morphology of amphiphilic copolymer aggregates by compound emulsifier via emulsion–solvent evaporation. J. Saudi Chem. Soc. 2018, 22, 297–305. [Google Scholar] [CrossRef]


| Condition | Continuous Phase 1 | Dispersed Phase 2 | Average Diameter (μm) | CV 4 (%) | Type | ||
|---|---|---|---|---|---|---|---|
| Flow Rate (μL/min) | SDS (0.06 wt %) | Conc. of Polymers (wt %) 3 | Mw of PMMA (/104) | ||||
| A1 | 140 | - | 1.0 | 12 | 72.4 | 13.1 | core-shell |
| A2 5 | 140 | + | 1.0 | 12 | 96.7 | 3.1 | Janus |
| A3 6 | 140 | + | 1.0 | 12 | 52.0 | 13.7 | core-shell |
| B1 | 700 | - | 1.0 | 12 | 44.9 | 4.5 | Janus |
| B2 | 700 | - | 1.0 | 1.5 | 44.6 | 8.6 | incomplete core-shell |
| C1 | 140 | - | 0.1 | 12 | 51.9 | 8.5 | dumbbell |
| C2 | 140 | - | 0.1 | 1.5 | 39.7 | 11.3 | incomplete core-shell |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshida, S.; Kikuchi, S.; Kanehashi, S.; Okamoto, K.; Ogino, K. Microfluidic Fabrication of Morphology-Controlled Polymeric Microspheres of Blends of Poly(4-butyltriphenylamine) and Poly(methyl methacrylate). Materials 2018, 11, 582. https://doi.org/10.3390/ma11040582
Yoshida S, Kikuchi S, Kanehashi S, Okamoto K, Ogino K. Microfluidic Fabrication of Morphology-Controlled Polymeric Microspheres of Blends of Poly(4-butyltriphenylamine) and Poly(methyl methacrylate). Materials. 2018; 11(4):582. https://doi.org/10.3390/ma11040582
Chicago/Turabian StyleYoshida, Saki, Shu Kikuchi, Shinji Kanehashi, Kazuo Okamoto, and Kenji Ogino. 2018. "Microfluidic Fabrication of Morphology-Controlled Polymeric Microspheres of Blends of Poly(4-butyltriphenylamine) and Poly(methyl methacrylate)" Materials 11, no. 4: 582. https://doi.org/10.3390/ma11040582
APA StyleYoshida, S., Kikuchi, S., Kanehashi, S., Okamoto, K., & Ogino, K. (2018). Microfluidic Fabrication of Morphology-Controlled Polymeric Microspheres of Blends of Poly(4-butyltriphenylamine) and Poly(methyl methacrylate). Materials, 11(4), 582. https://doi.org/10.3390/ma11040582






