Optical and Electrical Characterization of Biocompatible Polymeric Lines for Hemodialysis Applications
Abstract
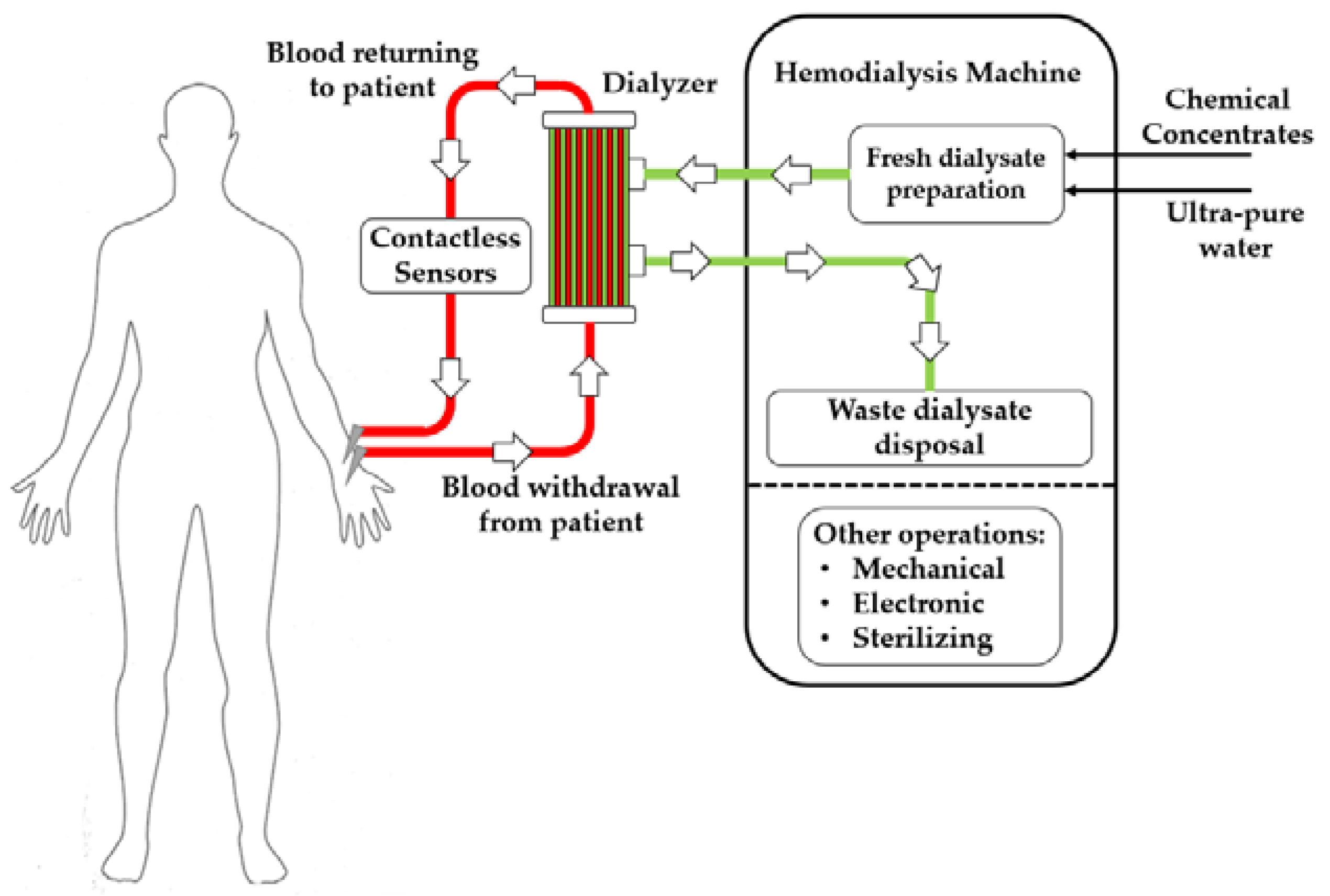
:1. Introduction
- Uremic toxins produced by the organism are transferred from blood to dialysate.
- Excess liquid accumulated by the patient due to lack of excretion through the kidneys is removed.
- Hematic concentrations of electrolytes are re-balanced.
- Pressure sensors on the arterial and venous sides of the bloodline
- Pressure sensors on both sides of the hemodialyzers to monitor transmembrane pressure
- Conductivity sensors on the inlet (fresh) and outlet (waste) sides of the dialysate circuit to monitor dialysate preparation
- Optical or ultrasonic sensors to monitor the potentially dangerous formation of bubbles in the bloodline
2. Materials and Methods
2.1. List of Bloodlines and Basic Properties
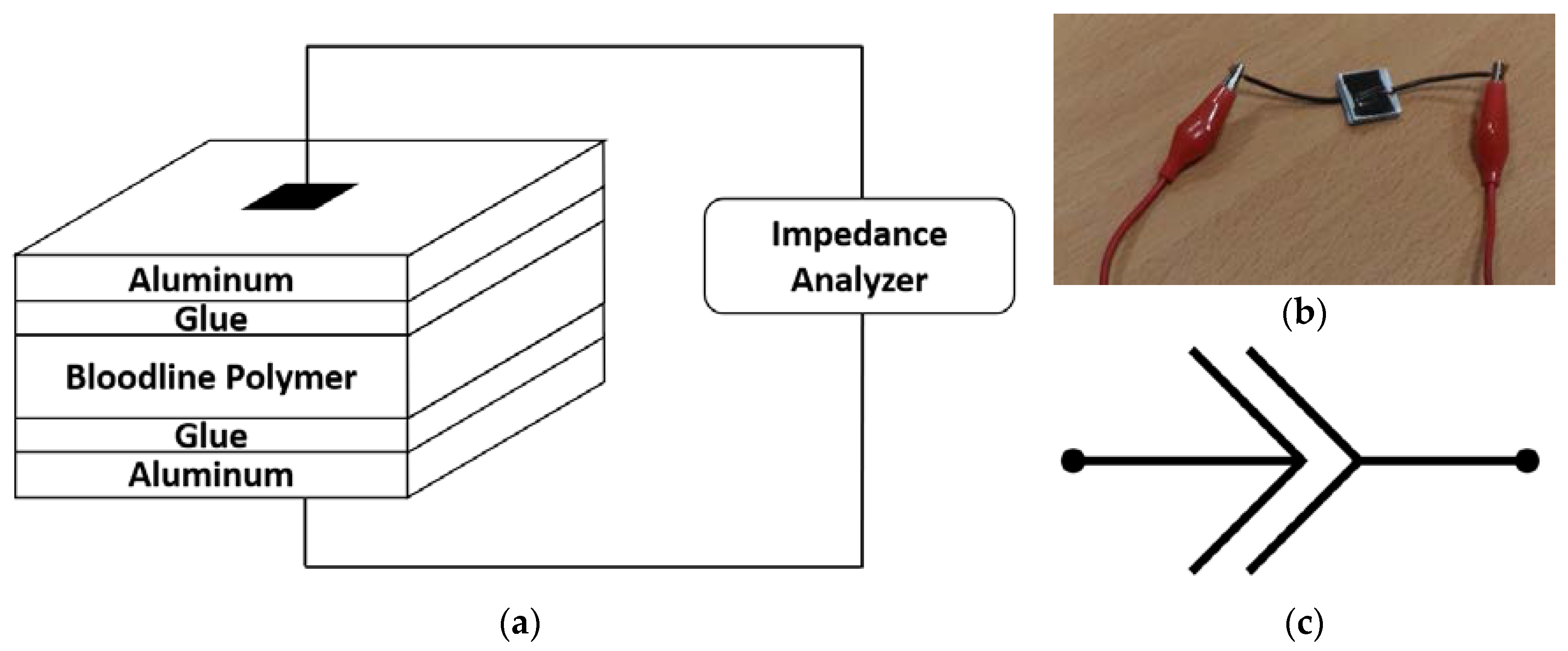
2.2. Optical Absorbance Spectroscopy
2.3. Electrical Impedance Spectroscopy
- The upper bound of the instrument range was 2 MHz (20 Hz–2 MHz).
- Polymers are usually dielectric in nature, so a reasonable lower bound is required to obtain proper readings (impedance → ∞ for frequency → 0).
- Previous work by our group [27] showed that: (a) at least one bloodline type showed nearly-capacitive behavior; and (b) 1–2 MHz is a valid range choice to decrease coupling impedance and thus increase sensitivity.
3. Results
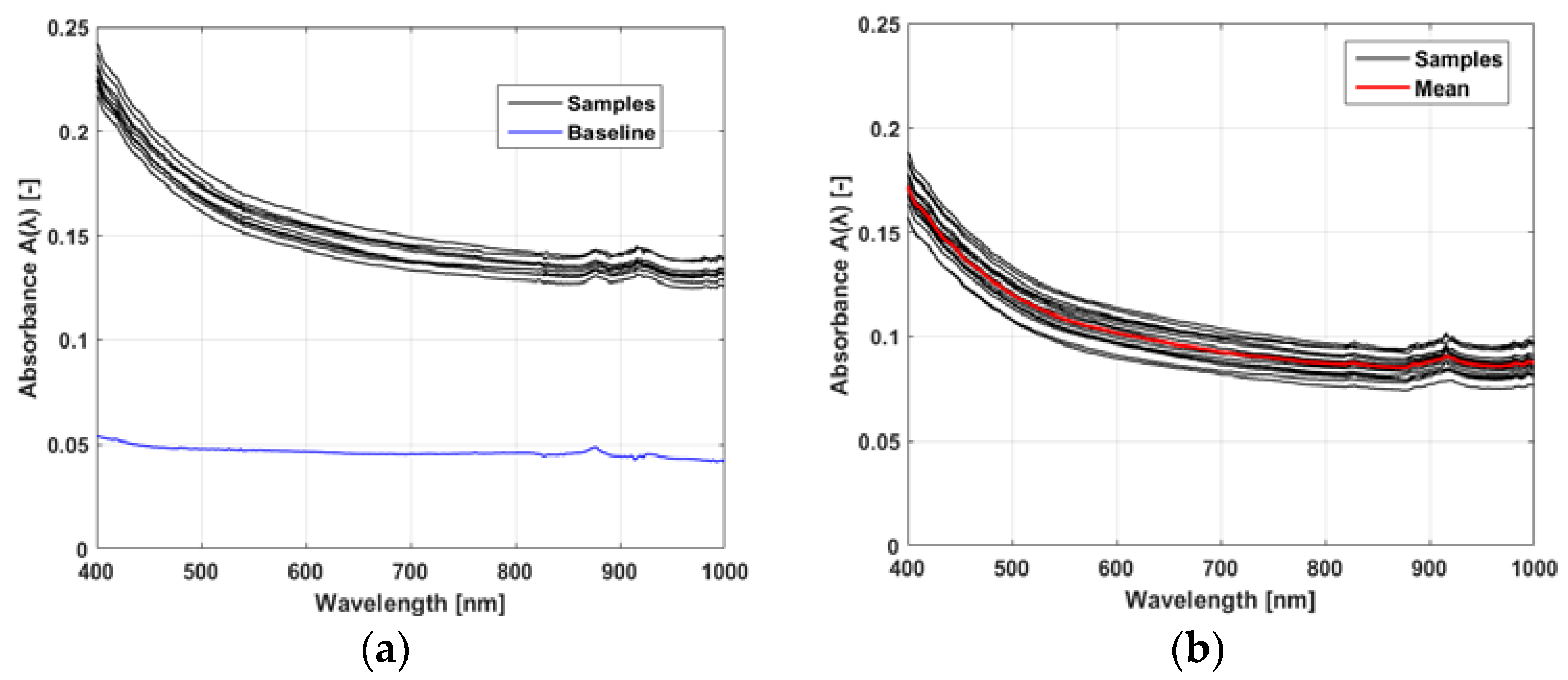
3.1. Optical Absorbance Spectroscopy
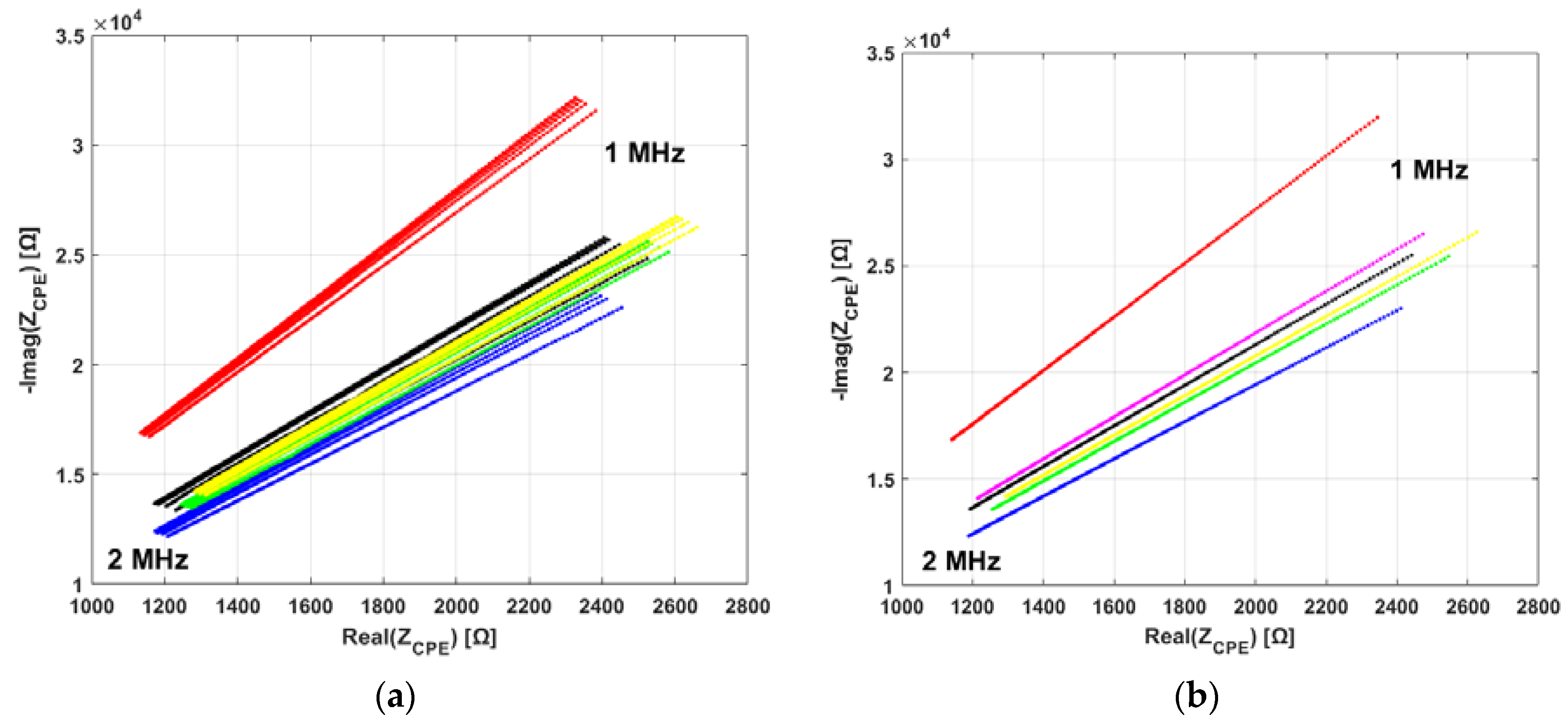
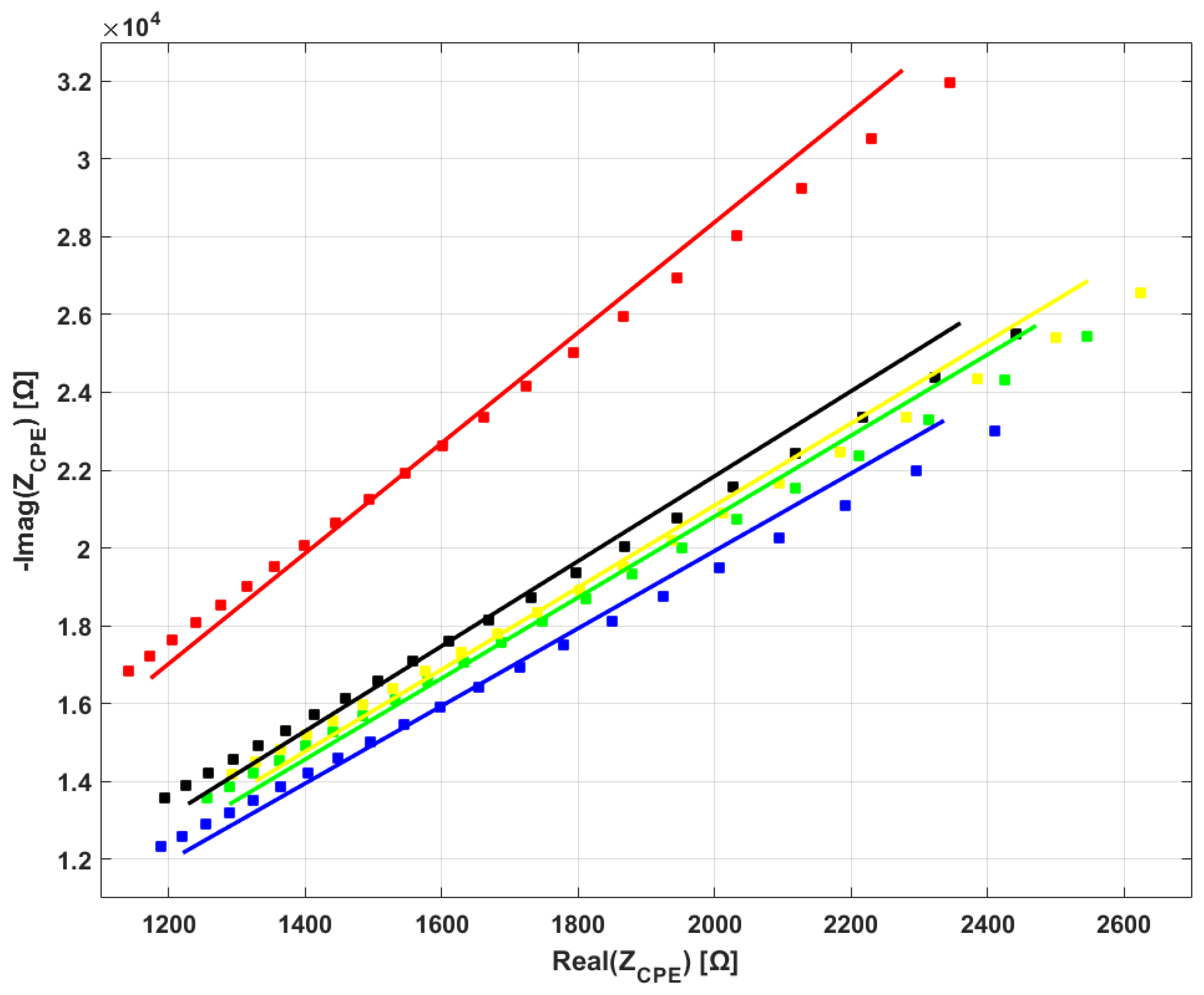
3.2. Electrical Impedance Spectroscopy
4. Discussion
4.1. Analysis of the Results
4.2. Limitations of the Study and Future Directions
- (1)
- UV light is damaging for biological tissues, so it cannot be realistically considered for continuous measurements on blood that will be reinjected into the organism.
- (2)
- Optical absorbance of blood at higher infrared wavelengths is dominated by water absorption.
- Repeating our investigation on a larger dataset (more bloodlines for each brand, more samples from each bloodline). In addition, performing sample manufacturing in an automated fashion where possible.
- Performing chemical structure analyses (IR, NMR, elemental analysis, etc.) and other material science analyses (e.g., FTIR) to compare the material composition of the bloodlines. Checking databases of physical properties for polymers where available.
- Investigating additional physical properties of the bloodlines, for example:
- ○
- Mechanical properties. These properties are useful to manage the flow of blood along the circuit and the interaction between the machine and the bloodline. They are studied independently by bloodline manufacturers but there are no comparisons in literature.
- ○
- Acoustic properties. This may be useful for ultrasound sensing, which is used in HD for bubble detection and to measure total protein concentration in blood.
- ○
- Optical index of refraction. This may be useful to estimate the amount of light emitted by optical sensors which is lost by refraction index mismatch across the air–polymer–blood optical path.
- ○
- Optical scattering. This property causes photons to deviate from their trajectory. Its estimation may help in determining an additional source of optical power loss in bloodlines.
4.3. Conclusions
- All bloodlines from the analyzed brands share similar optical spectra in the VIS-NIR range, with one exception of increased absorbance in the VIS region.
- All bloodlines from the analyzed brands share the same electrical behavior, that of the constant-phase element (CPE).
- Significant differences are present in CPE parameters among bloodlines.
- ontactless devices developed for use on one specific bloodline could be used with bloodlines of different manufacturers with proper recalibration.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nissenson, A.; Fine, R.N. Clinical Dialysis, 4th ed.; McGraw Hill Professional: New York, NY, USA, 2005; ISBN 978-0-07-141939-0. [Google Scholar]
- Ebnesajjad, S. Materials in Medicine: Hemodialysis and Prosthetic Vascular Grafts. Available online: https://chemical-materials.elsevier.com/new-materials-applications/materials-medicine-hemodialysis-prosthetic-vascular-grafts/?fsdsd=efwe (accessed on 9 January 2018).
- Sargent, J.A.; Gotch, F.A. Principles and biophysics of dialysis. In Replacement of Renal Function by Dialysis; Drukker, W., Parsons, F.M., Maher, J.F., Eds.; Springer: Dordrecht, The Netherlands, 1983. [Google Scholar]
- Ronco, C.; Ghezzi, P.M.; Brendolan, A.; Crepaldi, C.; La Greca, G. The haemodialysis system: Basic mechanisms of water and solute transport in extracorporeal renal replacement therapies. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc.-Eur. Ren. Assoc. 1998, 13 (Suppl. 6), 3–9. [Google Scholar] [CrossRef]
- Ahmad, S. Manual of Clinical Dialysis; Springer Science & Business Media: New York, NY, USA, 2009; ISBN 978-0-387-09651-3. [Google Scholar]
- Davison, A.M. Oxford Textbook of Clinical Nephrology; Oxford University Press: Oxford, UK, 2005; Volume 3, ISBN 978-0-19-856798-1. [Google Scholar]
- Ljunggren, L. Plasticizer migration from blood lines in hemodialysis. Artif. Organs 1984, 8, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Haishima, Y.; Matsuda, R.; Hayashi, Y.; Hasegawa, C.; Yagami, T.; Tsuchiya, T. Risk assessment of di(2-ethylhexyl)phthalate released from PVC blood circuits during hemodialysis and pump-oxygenation therapy. Int. J. Pharm. 2004, 274, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Tickner, J.A.; Schettler, T.; Guidotti, T.; McCally, M.; Rossi, M. Health risks posed by use of Di-2-ethylhexyl phthalate (DEHP) in PVC medical devices: a critical review. Am. J. Ind. Med. 2001, 39, 100–111. [Google Scholar] [CrossRef]
- Latini, G. Monitoring phthalate exposure in humans. Clin. Chim. Acta Int. J. Clin. Chem. 2005, 361, 20–29. [Google Scholar] [CrossRef] [PubMed]
- European Parliament; Council of the European Union. Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on Classification, Labelling and Packaging of Substances and Mixtures, Amending and Repealing Directives 67/548/EEC and 1999/45/EC, and Amending Regulation (EC) No 1907/2006 (Text with EEA Relevance) 2008; European Parliament and the Council of the European Union: Brussels, Belgium, 2008. [Google Scholar]
- NIKKISO: Blood Tubing Lines. Available online: http://www.nikkiso-europe.eu/blutschlauchsysteme.html?&L=1 (accessed on 9 January 2018).
- Nipro Set. Available online: http://www.nipro-europegroupcompanies.com/product/nipro-set/ (accessed on 9 January 2018).
- Nissenson, A.R.; Fine, R.E. Handbook of Dialysis Therapy E-Book, 5th ed.; Elsevier Health Sciences: Philadelphia, PA, USA, 2016; ISBN 978-0-323-44550-4. [Google Scholar]
- Paolini, F.; Mancini, E.; Bosetto, A.; Santoro, A. Hemoscan: A dialysis machine-integrated blood volume monitor. Int. J. Artif. Organs 1995, 18, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, I.; Ando, K.; Ando, Y.; Ookawara, S.; Suzuki, M.; Furuya, H.; Iimura, O.; Takada, D.; Kajiya, M.; Komada, T.; et al. BVM Study Group A new device to monitor blood volume in hemodialysis patients. Ther. Apher. Dial. 2010, 14, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Stiller, S.; Bonnie-Schorn, E.; Grassmann, A.; Uhlenbusch-Körwer, I.; Mann, H. A critical review of sodium profiling for hemodialysis. Semin. Dial. 2001, 14, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Mancini, E.; Basile, C.; Amoroso, L.; Di Giulio, S.; Usberti, M.; Colasanti, G.; Verzetti, G.; Rocco, A.; Imbasciati, E.; et al. Blood volume controlled hemodialysis in hypotension-prone patients: A randomized, multicenter controlled trial. Kidney Int. 2002, 62, 1034–1045. [Google Scholar] [CrossRef] [PubMed]
- Locatelli, F.; Buoncristiani, U.; Canaud, B.; Köhler, H.; Petitclerc, T.; Zucchelli, P. Haemodialysis with on-line monitoring equipment: Tools or toys? Nephrol. Dial. Transplant. 2005, 20, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Dasselaar, J.J. Relative blood volume based biofeedback during haemodialysis. J. Ren. Care 2007, 33, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Javed, F.; Savkin, A.V.; Chan, G.S.H.; Mackie, J.D.; Lovell, N.H. Identification and Control for Automated Regulation of Hemodynamic Variables During Hemodialysis. IEEE Trans. Biomed. Eng. 2011, 58, 1686–1697. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Mancini, E.; Azar, A.T. Biofeedback Systems and Their Application in the Hemodialysis Therapy. In Modeling and Control of Dialysis Systems; Studies in Computational Intelligence; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1081–1107. ISBN 978-3-642-27557-9. [Google Scholar]
- Holmer, M.; Sandberg, F.; Solem, K.; Grigonyte, E.; Olde, B.; Sörnmo, L. Extracting a cardiac signal from the extracorporeal pressure sensors of a hemodialysis machine. IEEE Trans. Biomed. Eng. 2015, 62, 1305–1315. [Google Scholar] [CrossRef] [PubMed]
- Holmer, M.; Sandberg, F.; Solem, K.; Olde, B.; Sörnmo, L. Cardiac signal estimation based on the arterial and venous pressure signals of a hemodialysis machine. Physiol. Meas. 2016, 37, 1499–1515. [Google Scholar] [CrossRef] [PubMed]
- Holmer, M.; Martínez, J.P.; Gil, E.; Sandberg, F.; Olde, B.; Sörnmo, L. Detection of ventricular premature beats based on the pressure signals of a hemodialysis machine. Med. Eng. Phys. 2018, 51, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Cattini, S.; Rovati, L. An Optical Technique for Real-Time Monitoring of Hemolysis During Hemodialysis. IEEE Trans. Instrum. Meas. 2016, 65, 1060–1069. [Google Scholar] [CrossRef]
- Ravagli, E.; Crescentini, M.; Tartagni, M.; Severi, S. Non-invasive measurement of electrical conductivity of liquids in biocompatible polymeric lines for hemodialysis applications. Sens. Actuators Phys. 2017, 261, 252–260. [Google Scholar] [CrossRef]
- Solem, K.; Olde, B.; Sörnmo, L. Prediction of intradialytic hypotension using photoplethysmography. IEEE Trans. Biomed. Eng. 2010, 57, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, F.; Bailón, R.; Hernando, D.; Laguna, P.; Martínez, J.P.; Solem, K.; Sörnmo, L. Prediction of hypotension in hemodialysis patients. Physiol. Meas. 2014, 35, 1885–1898. [Google Scholar] [CrossRef] [PubMed]
- Ahlmén, J.; Gydell, K.-H.; Hadimeri, H.; Hernandez, I.; Rogland, B.; Strömbom, U. A new safety device for hemodialysis. Hemodial. Int. 2008, 12, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.-C.; Lim, B.-Y.; Ciou, W.-S.; Wu, M.-J. Novel Wearable Device for Blood Leakage Detection during Hemodialysis Using an Array Sensing Patch. Sensors 2016, 16, 849. [Google Scholar] [CrossRef] [PubMed]
- Barsoukov, E.; Macdonald, J.R. Impedance Spectroscopy: Theory, Experiment, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2005; ISBN 978-0-471-71622-8. [Google Scholar]
- Morgan, H.; Green, N.G. AC Electrokinetics: Colloids and Nanoparticles; Research Studies Press: Philadelphia, PA, USA, 2003; ISBN 978-0-86380-255-3. [Google Scholar]
- Brug, G.J.; van den Eeden, A.L.G.; Sluyters-Rehbach, M.; Sluyters, J.H. The analysis of electrode impedances complicated by the presence of a constant phase element. J. Electroanal. Chem. Interfacial Electrochem. 1984, 176, 275–295. [Google Scholar] [CrossRef]
- Zoltowski, P. On the electrical capacitance of interfaces exhibiting constant phase element behaviour. J. Electroanal. Chem. 1998, 443, 149–154. [Google Scholar] [CrossRef]
- Shoar Abouzari, M.R.; Berkemeier, F.; Schmitz, G.; Wilmer, D. On the physical interpretation of constant phase elements. Solid State Ion. 2009, 180, 922–927. [Google Scholar] [CrossRef]
- Mancini, E.; Perazzini, C.; Gesualdo, L.; Aucella, F.; Limido, A.; Scolari, F.; Savoldi, S.; Tramonti, M.; Corazza, L.; Atti, M.; et al. Italian Oxygen Saturation Study Group (SOGLIA) Intra-dialytic blood oxygen saturation (SO2): Association with dialysis hypotension (the SOGLIA Study). J. Nephrol. 2017, 30, 811–819. [Google Scholar] [CrossRef] [PubMed]



| Manufacturer | Model Name | Composition Information (from Package) | Abbreviation | Internal Diameter (mm) | Thickness (mm) |
|---|---|---|---|---|---|
| B.Braun Avitum AG (Germany) | A/V Set | DEHP-FREE PVC | BB | 4.83 | 1.00 |
| Bellco (Italy), now part of Medtronic (USA) | Extracorporeal Bloodlines | PVC V 326-1/F | BE | 4.37 | 1.20 |
| Gambro Dasco S.p.a (Italy), now part of Baxter (USA) | ArtiSet | DEHP-FREE | BG | 4.27 | 1.17 |
| Fresenius Medical Care (Germany) | LifeLine Beta AV-Set ONLINEplus BVM 5008-R | - | FC | 4.1 | 1.17 |
| EffeEmme Fabbricazioni Medicali (Italy) | DiaLine | DEHP-FREE | FM | 4.9 | 1 |
| GAMA Group (Czech Republic) | Standardline DIS 06-16 UNIV | DEHP-FREE (pump segment) | GA | 4.5 | 1.17 |
| NIPRO Corporation (Japan) | NIPRO Set | DEHP-FREE | NI | 4.33 | 1.13 |
| Brand | N (-) (Mean ± std) | Q0 (pS) (Mean ± std) | Q0’ (pS∙mm) (Mean ± std) |
|---|---|---|---|
| BB | 0.942 ± 0.004 (b,d,e,f) | 16.64 ± 1.65 | 16.64 ± 1.65 |
| BE | 0.957 ± 0.003 (a,c) | 10.35 ± 1.12 | 12.42 ± 1.34 (c) |
| BG | 0.951 ± 0.004 | 12.58 ± 1.04 | 14.67 ± 1.16 |
| FC | 0.942 ± 0.007 (b,d,e) | 14.95 ± 3.11 | 17.50 ± 3.64 (b,f) |
| FM | 0.954 ± 0.002 (a,c) | 13.94 ± 0.83 | 13.94 ± 0.83 |
| GA | 0.955 ± 0.007 (a,c) | 12.03 ± 2.86 | 14.08 ± 3.35 |
| NI | 0.952 ± 0.007 (a) | 10.98 ± 2.72 | 12.41 ± 3.07 (c) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravagli, E.; Severi, S. Optical and Electrical Characterization of Biocompatible Polymeric Lines for Hemodialysis Applications. Materials 2018, 11, 438. https://doi.org/10.3390/ma11030438
Ravagli E, Severi S. Optical and Electrical Characterization of Biocompatible Polymeric Lines for Hemodialysis Applications. Materials. 2018; 11(3):438. https://doi.org/10.3390/ma11030438
Chicago/Turabian StyleRavagli, Enrico, and Stefano Severi. 2018. "Optical and Electrical Characterization of Biocompatible Polymeric Lines for Hemodialysis Applications" Materials 11, no. 3: 438. https://doi.org/10.3390/ma11030438
APA StyleRavagli, E., & Severi, S. (2018). Optical and Electrical Characterization of Biocompatible Polymeric Lines for Hemodialysis Applications. Materials, 11(3), 438. https://doi.org/10.3390/ma11030438





