Preparation of Renewable Bio-Polyols from Two Species of Colliguaja for Rigid Polyurethane Foams
Abstract
1. Introduction
2. Experimental Procedures
2.1. Chemicals and Reagents
2.2. C. integerrima and C. salicifolia Seeds
2.3. Vegetable Oil Extraction
2.4. Synthesis of C. integerrima and C. salicifolia Polyol
2.5. Characterization Methods Used for Oils and Polyols
3. Results and Discussion
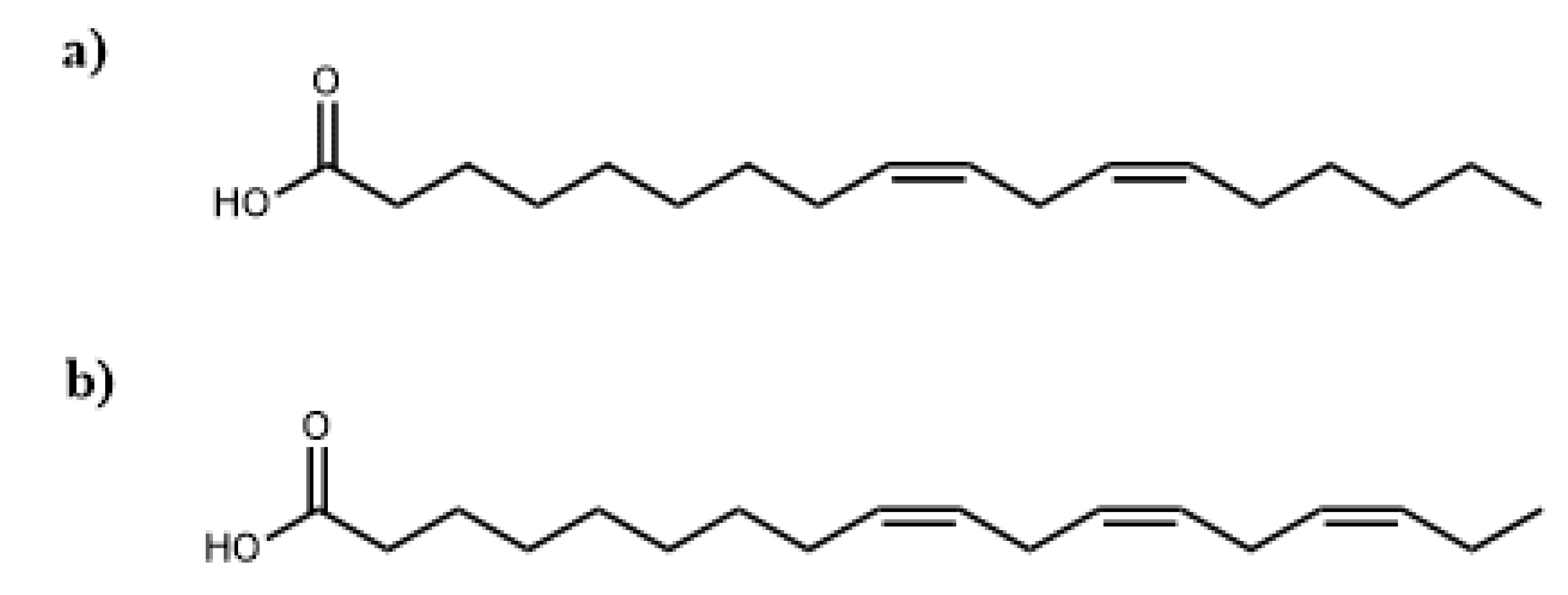
3.1. Physicochemical Characteristics of Vegetable Oils
3.2. Synthesis and Characterization of C. integerrima and C. salicifolia Polyols Prepared by a One-Step Synthesis
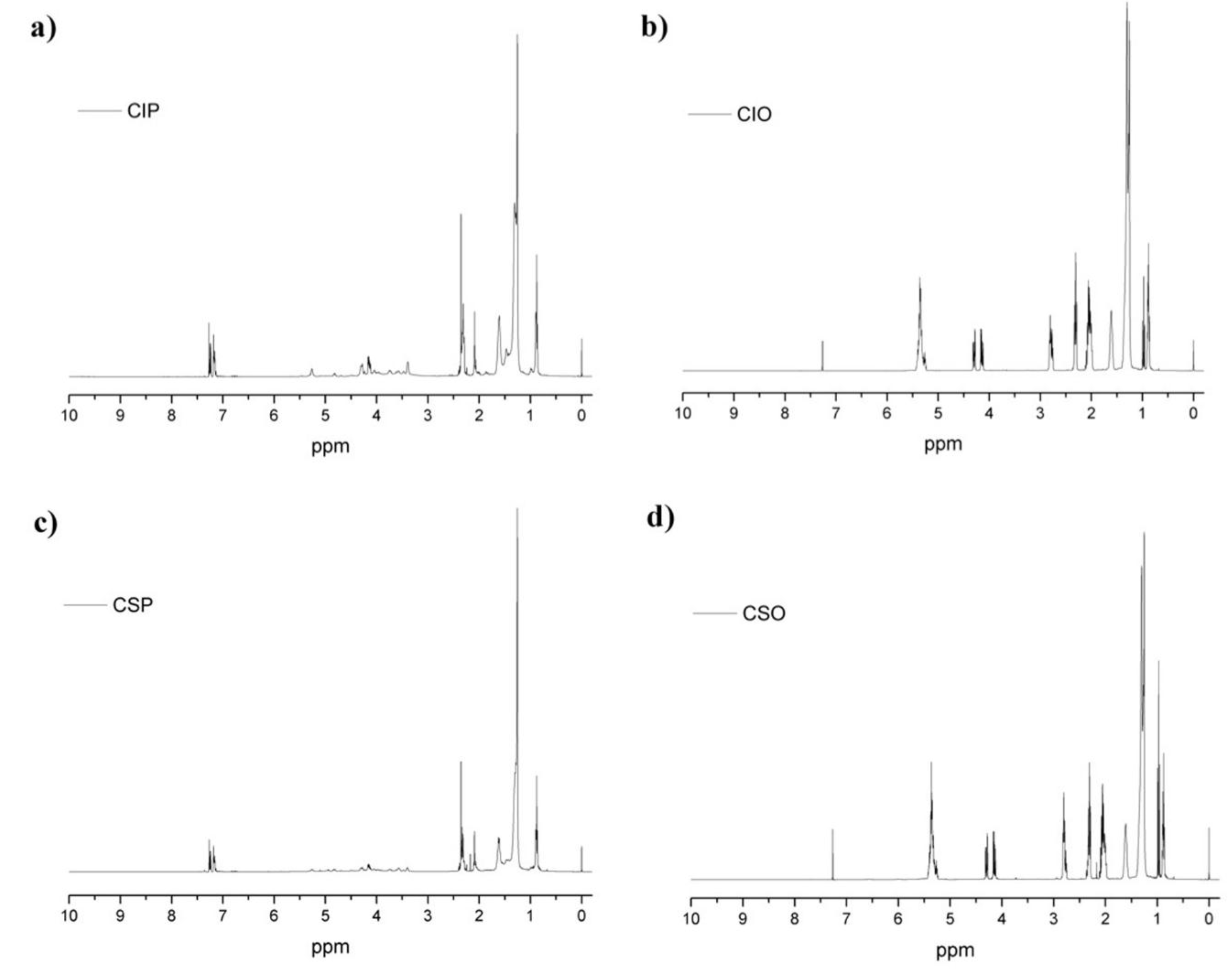
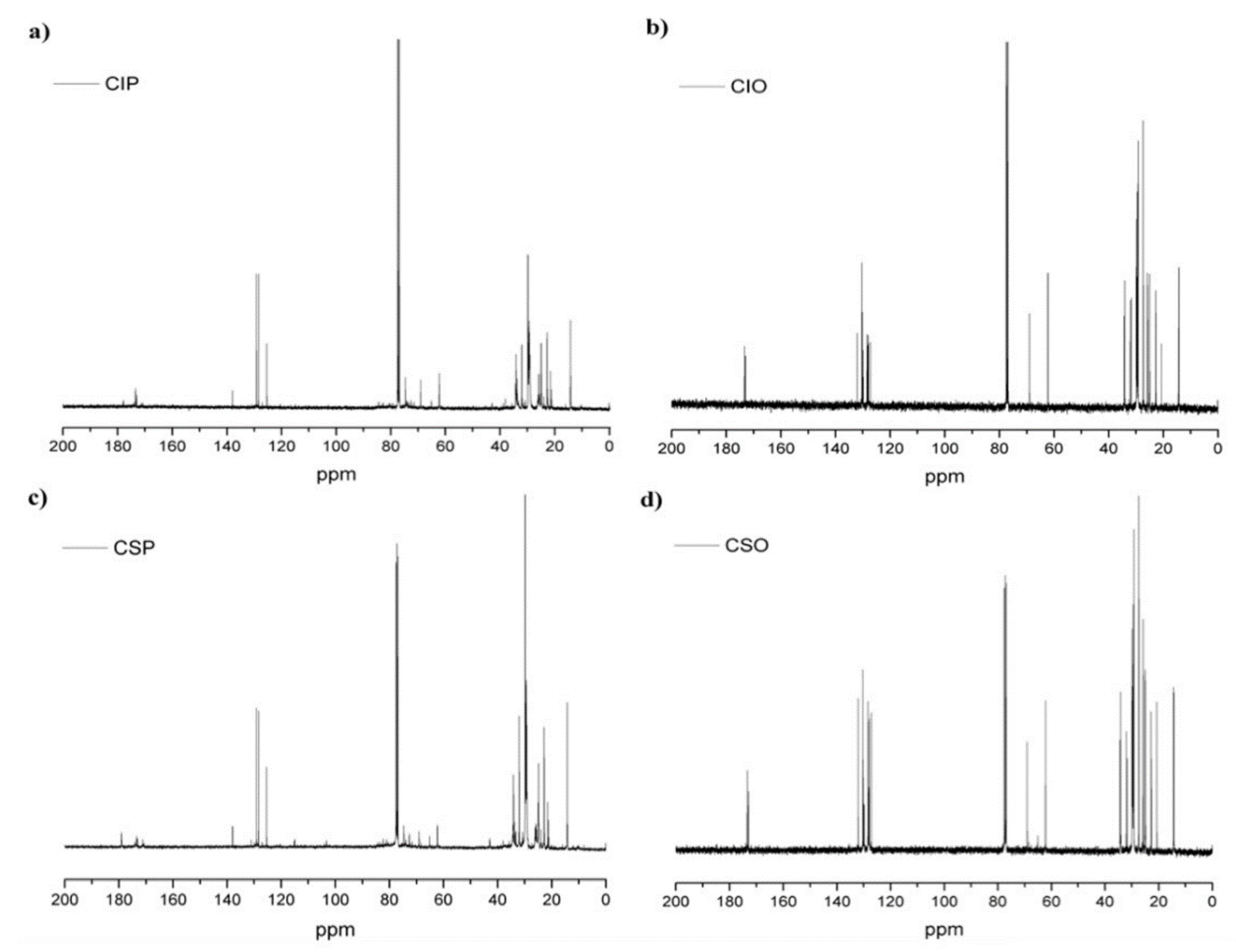
3.2.1. 1H and 13C NMR Analysis
3.2.2. FTIR Analysis
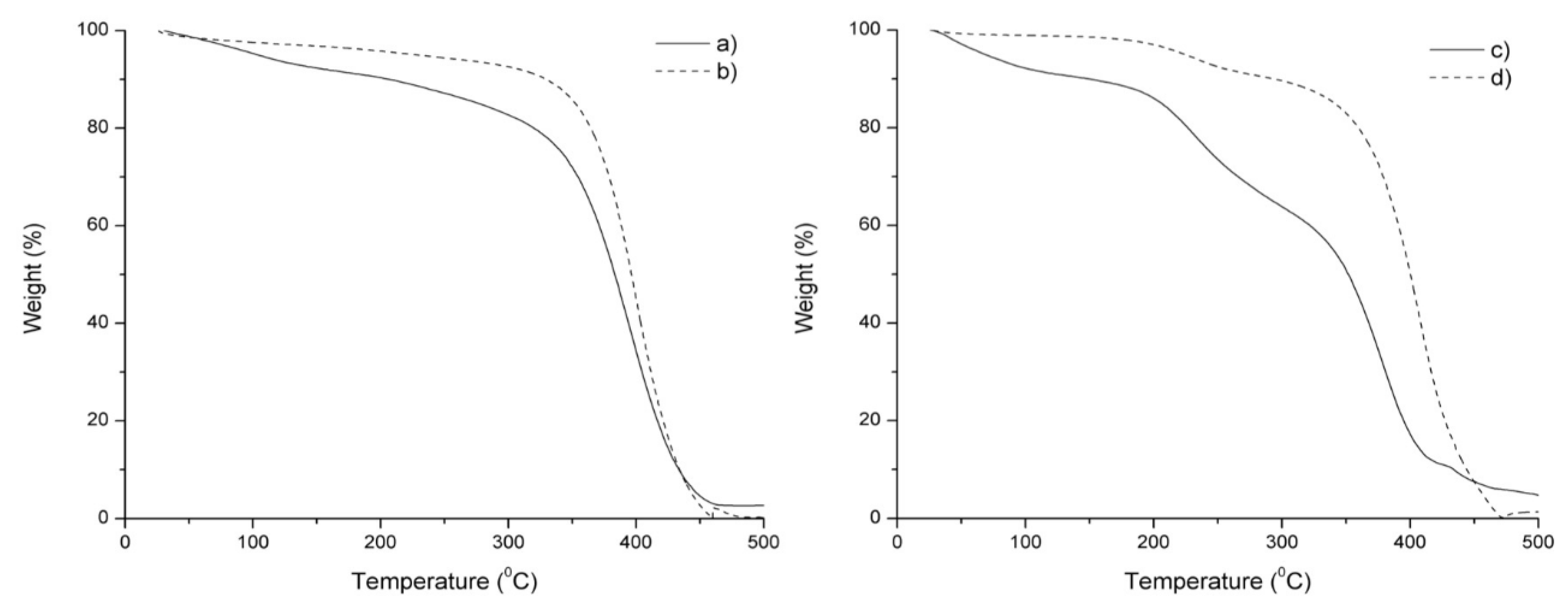
3.2.3. TGA Analysis
Physicochemical Characteristics of Vegetable Polyols
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eissen, M.; Metzger, J.O.; Schmidt, E.; Schneidewind, U. 10 Years after Rio-Concepts on the Contribution of Chemistry to a Sustainable Development. Angew. Chem. Int. Ed. 2002, 41, 414–436. [Google Scholar] [CrossRef]
- Tian, H.; Tang, Z.; Chen, X.; Jing, X. Biodegradable synthetic polymers: Preparation, functionalization and biomedical application. Prog. Polym. Sci. 2012, 37, 237–280. [Google Scholar] [CrossRef]
- Biermann, U.; Friedt, W.; Lang, S.; Luhs, W.; Machmuller, G.; Metzger, J.O.; Klaas, M.R.G.; Schafer, H.J.; Scheider, M.P. New syntheses whit oils and fast as renewable raw materials for the chemical industry. Angew. Chem. Int. Ed. 2000, 39, 2206–2224. [Google Scholar] [CrossRef]
- SenihaGüner, F.; Yağcı, Y.; TuncerErciyes, A. Polymers from triglyceride oils. Prog. Polym. Sci. 2006, 31, 633–670. [Google Scholar] [CrossRef]
- de Espinosa, L.M.; Meier, M.A.R. Plant oils: The perfect renewable resource for polymer science? Eur. Polym. J. 2011, 47, 837–852. [Google Scholar] [CrossRef]
- Zhang, C.; Madbouly, S.A.; Kessler, M.R. Biobased polyurethanes prepared from different vegetable oils. ACS Appl. Mater. Interfaces 2015, 7, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.; Kulkarni, R.; Mahulikar, P.; Sohn, D.; Gite, V. Development of PU coatings from neem oil based alkyds prepared by the monoglyceride route. J. Am. Oil Chem. Soc. 2015, 92, 733–741. [Google Scholar] [CrossRef]
- Fernandes, F.C.; Kirwan, K.; Lehane, D.; Coles, S.R. Epoxy resin blends and composites from waste vegetable oil. Eur. Polym. J. 2017, 89, 449–460. [Google Scholar] [CrossRef]
- Sinadinović-Fišer, S.; Janković, M.; Petrović, Z.S. Kinetics of in situ epoxidation of soybean oil in bulk catalyzed by ion exchange resin. J. Am. Oil Chem. Soc. 2001, 78, 725–731. [Google Scholar] [CrossRef]
- Khoe, T.H.; Otey, F.H.; Frankel, E.N. Rigid urethane foams from hydroxymethylated linseed oil and polyol esters. J. Am. Oil Chem. Soc. 1972, 49, 615–618. [Google Scholar] [CrossRef]
- Guo, A.; Demydov, D.; Zhang, W.; Petrovic, Z.S. Polyols and polyurethanes from hydroformylation of soybean oil. J. Polym. Environ. 2002, 10, 49–52. [Google Scholar] [CrossRef]
- Petrović, Z.S.; Zhang, W.; Javni, I. Structure and properties of polyurethanes prepared from triglyceride polyols by ozonolysis. Biomacromolecules 2005, 6, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Caillol, S.; Desroches, M.; Carlotti, S.; Auvergne, R.; Boutevin, B. Synthesis of new polyurethanes from vegetable oil by thiol-ene coupling. Green Mater. 2012, 1, 16–26. [Google Scholar] [CrossRef]
- Chuayjuljit, S.; Maungchareon, A.; Saravari, O. Preparation and properties of palm oil-based rigid polyurethane nanocomposite foams. J. Reinf. Plast. Compos. 2010, 29, 218–225. [Google Scholar] [CrossRef]
- Hu, Y.H.; Gao, Y.; Wang, D.N.; Hu, C.P.; Zu, S.; Vanoverloop, L.; Randall, D. Rigid polyurethane foam prepared from a rape seed oil based polyol. J. Appl. Polym. Sci. 2002, 84, 591–597. [Google Scholar] [CrossRef]
- Desroches, M.; Escouvois, M.; Auvergne, R.; Caillol, S.; Boutevin, B. From vegetable oils to polyurethanes: Synthetic routes to polyols and main industrial products. Polym. Rev. 2012, 52, 38–79. [Google Scholar] [CrossRef]
- Kaushik, N.; Bhardwaj, D. Screening of Jatropha curcas germplasm for oil content and fatty acid composition. Biomass Bioenergy 2013, 58, 210–218. [Google Scholar] [CrossRef]
- John, J.; Bhattacharya, M.; Turner, R.B. Characterization of polyurethane foams from soybean oil. J. Appl. Polym. Sci. 2002, 86, 3097–3107. [Google Scholar] [CrossRef]
- Petrović, Z.S.; Javni, I.; Jing, X.; Hong, D.P.; Guo, A. Effect of hyperbranched vegetable oil polyols on properties of flexible polyurethane foams. Mater. Sci. Forum 2007, 555, 459–465. [Google Scholar]
- Prociak, A.; Rojek, P.; Pawlik, H. Flexible polyurethane foams modified with natural oil based polyols. J. Cell. Plast. 2012, 48, 489–499. [Google Scholar] [CrossRef]
- Singh, A.P.; Bhattacharya, M. Viscoelastic changes and cell opening of reacting polyurethane foams from soy oil. Polym. Eng. Sci. 2004, 44, 1977–1987. [Google Scholar] [CrossRef]
- Liigadas, G.; Ronda, J.C.; Galià, M.; Cádiz, V. Plant as Platform chemical for polyurethane synthesis: Current-State-of-the-Art. Biomacromolecules 2010, 11, 2825–2835. [Google Scholar] [CrossRef] [PubMed]
- Piloto-Rodríguez, R.; Melo, E.A.; Goyos-Pérez, L.; Verhelst, S. Conversion of by-products from the vegetable oil industry into biodiesel and it is use in internal combustion engines: A review. Braz. J. Chem. Eng. 2014, 31, 287–301. [Google Scholar] [CrossRef]
- Bittner, M.; Alarcón, J.; Aqueveque, P.; Becerra, J.; Hernández, V.; Hoeneisen, M.; Silva, M. EstudioQuímico De Especies de la Familia EuphorbiaceaeEn Chile. Bol. Soc. Chil. Quím. 2001, 46, 419–431. [Google Scholar] [CrossRef]
- Galarce, G.; Trivelli, M. Reseñasobre la flora y vegetación de la serranía El Asiento, San Felipe, Región de Valparaíso. ServicioAgrícolaGanadero 2013, 1–118. Available online: https://www.sag.gob.cl/sites/default/files/flora_san_felipe_2013.pdf (accessed on 14 August 2018).
- Bull-Hereñu, K.; Martínez, E.A.; Squeo, F.A. Structure and genetic diversity in colliguajaodorifera Mol. (Euphorbiaceae), a shrub subjected to pleisto-holocenic natural perturbations in a Mediterranean South American Region. J. Biogeogr. 2005, 32, 1129–1138. [Google Scholar] [CrossRef]
- Pinto, G.; Toledo, I.; Córdoba, L.; Flores, M.; Cabrera, J. Análisisfitoquímico de C. integerrima (Hook.) Gill. Et Hook. (Euphorbiaceae), una planta de la Patagonia Argentina. Acta FarmacéuticaBonaerense 2004, 23, 459–465. [Google Scholar]
- Alvarez, M.E.; Gil, R.; Acosta, M.G.; Saad, R.J.; Borkowski, E.; María, A.O.M. Diuretic activity of aqueous extract and betulin from C. integerrima in rats. Pharm. Biol. 2009, 47, 274–278. [Google Scholar] [CrossRef]
- AOAC. Official Method Cd 3d-63, Am2-93, Official Methods and Recommended Practices of the American Oil Chemists’ Society; AOCS Press: Champaign, IL, USA, 1995. [Google Scholar]
- Monteavaro, L.; da Silva, E.; Costa, A.; Samios, D.; Gervase, A.; Petzhold, C. Polyurethane networks from formiated soy polyols: Synthesis and mechanical characterization. J. Am. Oil Chem. Soc. 2005, 82, 365–371. [Google Scholar] [CrossRef]
- Morrison, W.R.; Smith, L.M. Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol. J. Lipid. Res. 1964, 5, 600–608. [Google Scholar] [PubMed]
- Fatemi, S.H.; Hammond, E.G. Analysis of oleate, linoleate and linolenate hydroperoxides in oxidized ester mixtures. Lipids 1980, 15, 379–385. [Google Scholar] [CrossRef]
- AOAC. Official Method Cd 3d-63, Official Methods and Recommended Practices of the American Oil Chemists’ Society; AOCS Press: Champaign, IL, USA, 1993. [Google Scholar]
- AOAC-Association of Official Analytical Chemists. Official Methods of Analysis, 18th ed.; Association of Official Analytical: Washington, DC, USA, 2005. [Google Scholar]
- Zlatanić, A.; Lava, C.; Zhnag, W.; Petrović, Z.S. Effect of structure on properties of polyols and polyurethanes based on different vegetable oils. J. Polym. Sci. B 2004, 42, 809–819. [Google Scholar] [CrossRef]
- American Society for Testing Materials. Polyurethane Raw Materials: Determination of Hydroxyl Numbers of Polyols; ASTM (D 4274-05); ASTM: West Conshohocken, PA, USA, 2005; pp. 1–9. [Google Scholar]
- Gunstone, F.D.; John, L.H.; Fred, B.P. The Lipid Handbook; Chapman & Hall Chemical Database: Washington, DC, USA, 1994. [Google Scholar]
- Krisnangkura, K.; Yimsuwan, T.; Pairintra, R. An empirical approach in predicting biodiesel viscosity at various temperatures. Fuel 2006, 85, 107–113. [Google Scholar] [CrossRef]
- Rodrigues, J.A., Jr.; Cardoso, F.P.; Lachter, E.R.; Estevao, L.R.M.; Lima, E.; Nascimento, R.S.V. Correlating chemical structure and physical properties of vegetable oil esters. J. Am. Oil Chem. Soc. 2006, 30, 353–357. [Google Scholar] [CrossRef]
- Demirbas, A. Relationships derived from physical properties of vegetable oil and biodiesel fuels. Fuel 2008, 87, 1743–1748. [Google Scholar] [CrossRef]
- Vollhardt, K.P.C.; Schore, N.E. QuímicaOrgánica: Estructura y Función, 5th ed.; OMEGA D.L.: Barcelona, Spain, 2007. [Google Scholar]
- Javni, I.; Petrovic, Z.S.; Guo, A.; Fuller, R. Thermal stability of polyurethanes based on vegetable oils. J. Appl. Polym. Sci. 2000, 77, 1723–1734. [Google Scholar] [CrossRef]
- Dileesh, S.; Adithya, M.; Sankar, A.; Peter, C.V. Determination of saponification, acid and ester values; percentage of free fatty acids and glycerol in some selected edible oils: Calculation of concentration of lye needed to prepare soap from these oils. Res. Sch. ISSN 2013, 2249–6696. [Google Scholar]
- Knothe, G.; Steidly, K.R. Kinematic viscosity of biodiesel fuel components and related compounds. Influence of compound structure and comparison to petrodiesel fuel components. Fuel 2005, 84, 1059–1065. [Google Scholar] [CrossRef]
- Tersac, G. Chemistry and Technology of Polyols for Polyurethanes; Ionescu, M., Ed.; Rapra Technology: Shrewsbury, UK, 2007; Chapter 12; pp. 317–319. [Google Scholar]
- Narine, S.S.; Kong, X.; Bouzidi, L.; Sporns, P. Physical properties of polyurethanes produced from polyols from seed oils: II. Foams. J. Am. Oil Chem. Soc. 2007, 84, 65–72. [Google Scholar] [CrossRef]
- Guo, A.; Cho, Y.; Petrovic, Z.S. Structure and properties of halogenated and nonhalogenated soy-based polyols. J. Polym. Sci. Part A Polym. Chem. 2000, 38, 3900–3910. [Google Scholar] [CrossRef]
- Webster, D.C.; Pan, X. New biobased high functionality polyols and their use in polyurethane coatings. ChemSusChem 2012, 5, 419–429. [Google Scholar]
- Nadji, H.; Bruzzèse, C.; Belgacem, M.N.; Benaboura, A.; Gandini, A. Oxypropylation of lignins and preparation of rigid polyurethane foams from the ensuing polyols. Macromol. Mater. Eng. 2005, 290, 1009–1016. [Google Scholar] [CrossRef]
- Gandini, A.; Belgacem, M.N. Partial or total oxypropylation of natural polymers and the use of the ensuing materials as composites or polyol macromonomers. In Monomers, Polymers and Composites from Renewable Resources, 1st ed.; Belgacem, M.N., Gandini, A., Eds.; Elsevier: Oxford, UK, 2008. [Google Scholar]

| Parameters | Oils | |
|---|---|---|
| CIO | CSO | |
| Fatty acid (%) | ||
| Myristic, C14:0 | 0.06 | 0.04 |
| Palmitic, C16:0 | 10.54 | 8.46 |
| Palmitoleic, C16:1 | 0.05 | 0.08 |
| Margaric, C17:0 | 0.00 | 0.00 |
| Stearic, C18:0 | 2.03 | 2.06 |
| Oleic, C18:1 | 23.50 | 15.08 |
| Linoleic, C18:2 | 31.11 | 20.40 |
| Gamma- linolenic, C18:3 | 0.48 | 0.00 |
| Alpha-linolenic, C18:3 | 26.39 | 46.60 |
| Gondoic, C20:1 | 5.35 | 6.34 |
| Eicosadienoic C20:2 | 0.48 | 0.73 |
| Cox value | 9.35 | 12.24 |
| AV (mg KOH/g oil) | 0.17 | 0.25 |
| IV (g I2/100 g oil) | 143.8 | 179.3 |
| PV (meq O2/kg oil) | 19 | 18 |
| SN (mg KOH/g oil) | 196 | 194 |
| Kinematic Viscosity (mm2/s) | 58.6 | 48.8 |
| Density (g/cm3) | 0.884 | 0.896 |
| Parameters | Polyols | |
|---|---|---|
| CIP | CSP | |
| AV (mg KOH/g) | 4.50 | 12.95 |
| IV (g I2/100 g) | 0.2 | 0.5 |
| SN (mg KOH/g) | 227 | 235 |
| Kinematic Viscosity (mm2/s) | 3637 | 5746 |
| Density (g/cm3) | 1.012 | 0.921 |
| Molar Mass (g/mol) | 1122.4 | 1166.1 |
| OH numbers (mg KOH/g) | 225.0 | 240.8 |
| OH functionality | 4.50 | 5.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abril-Milán, D.; Valdés, O.; Mirabal-Gallardo, Y.; F. de la Torre, A.; Bustamante, C.; Contreras, J. Preparation of Renewable Bio-Polyols from Two Species of Colliguaja for Rigid Polyurethane Foams. Materials 2018, 11, 2244. https://doi.org/10.3390/ma11112244
Abril-Milán D, Valdés O, Mirabal-Gallardo Y, F. de la Torre A, Bustamante C, Contreras J. Preparation of Renewable Bio-Polyols from Two Species of Colliguaja for Rigid Polyurethane Foams. Materials. 2018; 11(11):2244. https://doi.org/10.3390/ma11112244
Chicago/Turabian StyleAbril-Milán, Diana, Oscar Valdés, Yaneris Mirabal-Gallardo, Alexander F. de la Torre, Carlos Bustamante, and Jorge Contreras. 2018. "Preparation of Renewable Bio-Polyols from Two Species of Colliguaja for Rigid Polyurethane Foams" Materials 11, no. 11: 2244. https://doi.org/10.3390/ma11112244
APA StyleAbril-Milán, D., Valdés, O., Mirabal-Gallardo, Y., F. de la Torre, A., Bustamante, C., & Contreras, J. (2018). Preparation of Renewable Bio-Polyols from Two Species of Colliguaja for Rigid Polyurethane Foams. Materials, 11(11), 2244. https://doi.org/10.3390/ma11112244





