The Hardness of Additively Manufactured Alloys
Abstract
1. Introduction
2. Influence of Process Variables
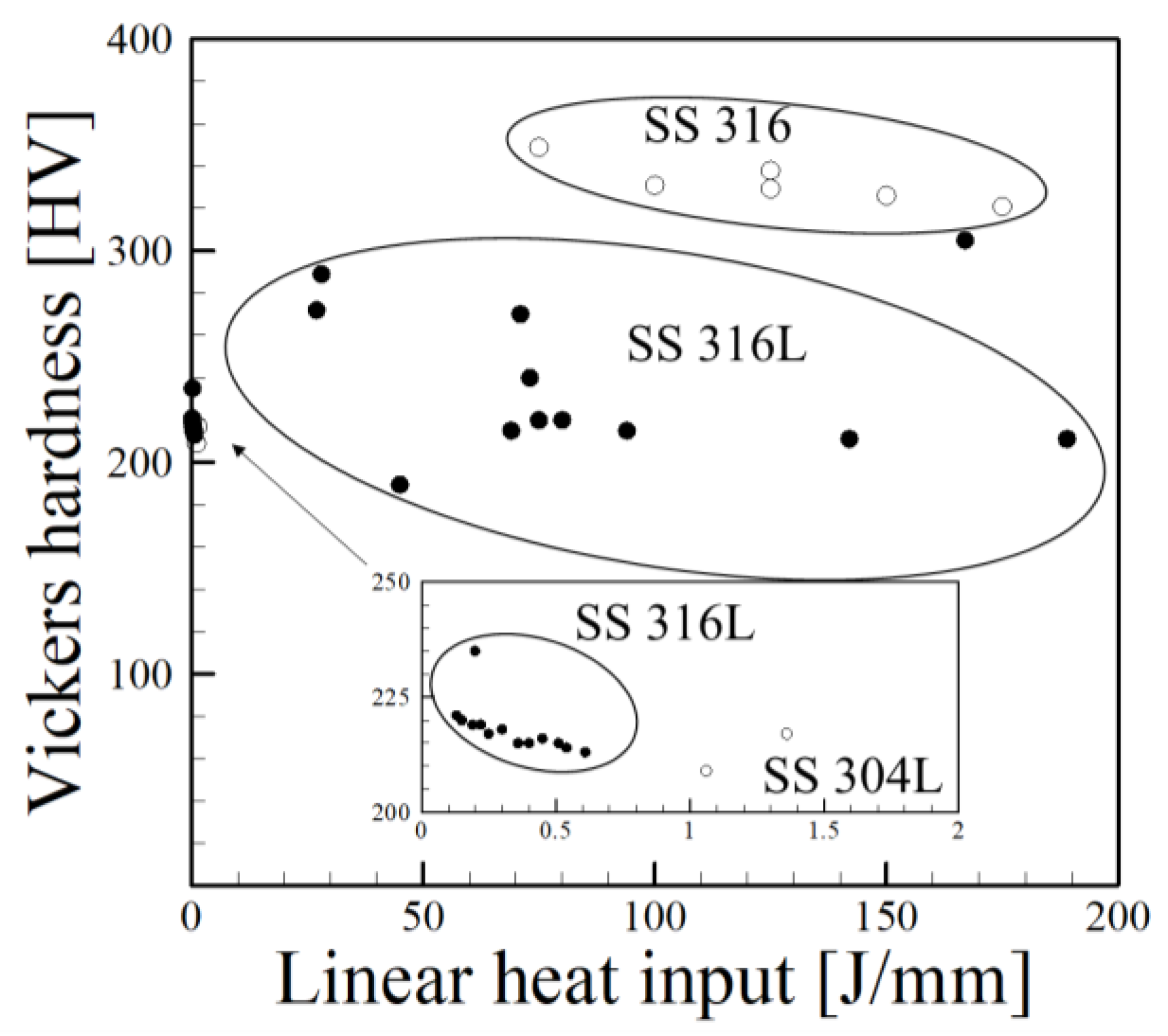
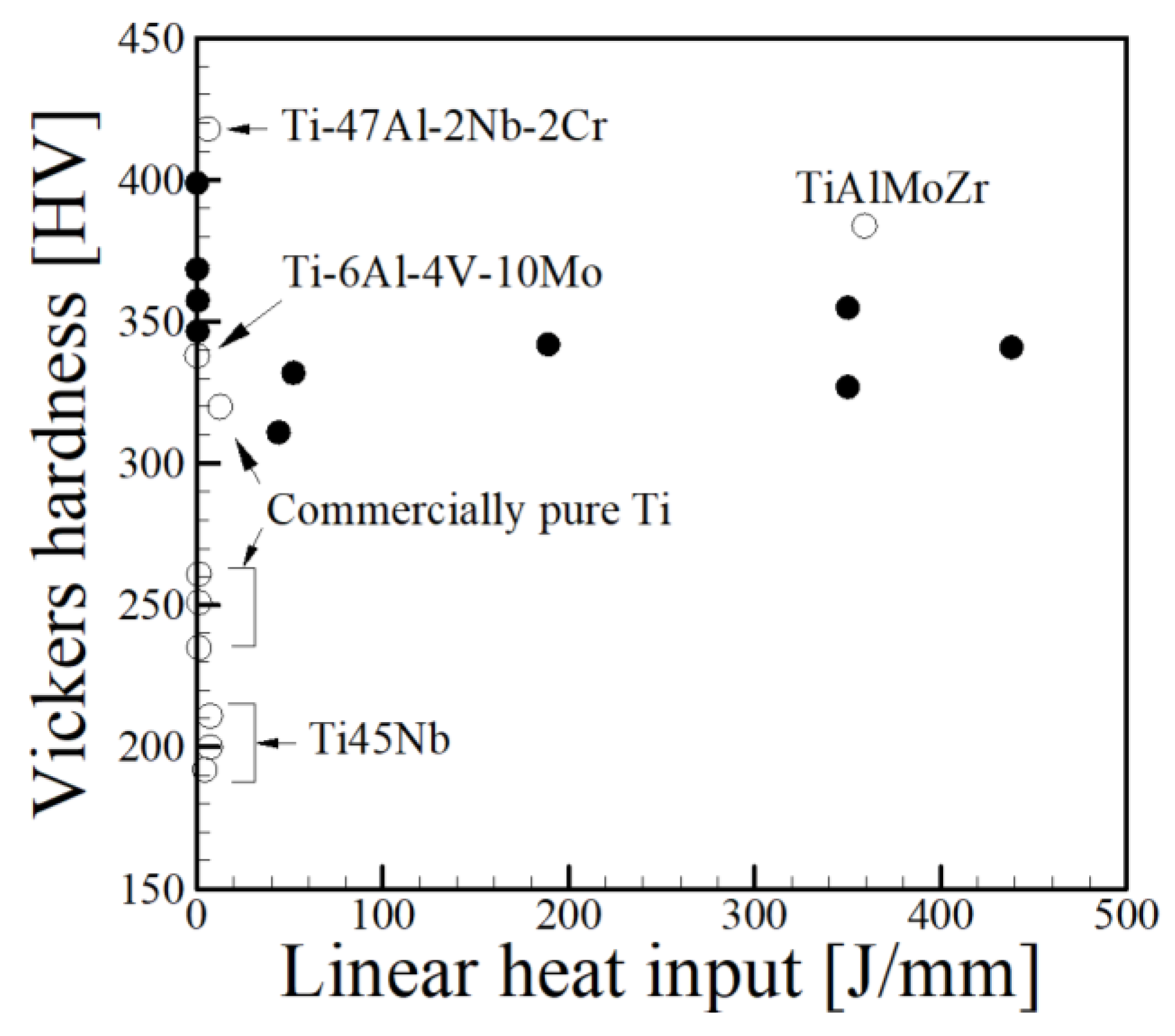
2.1. Energy Input
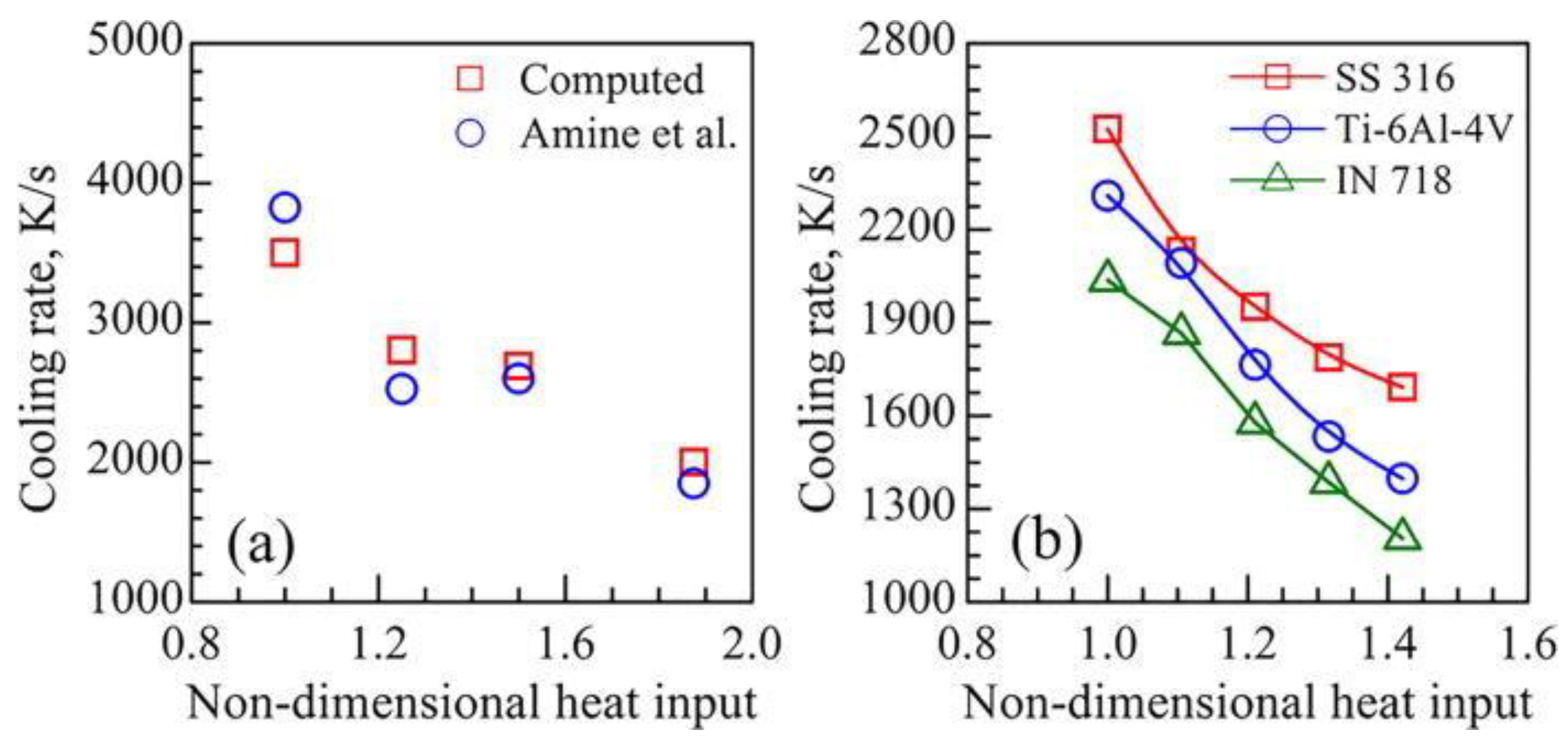
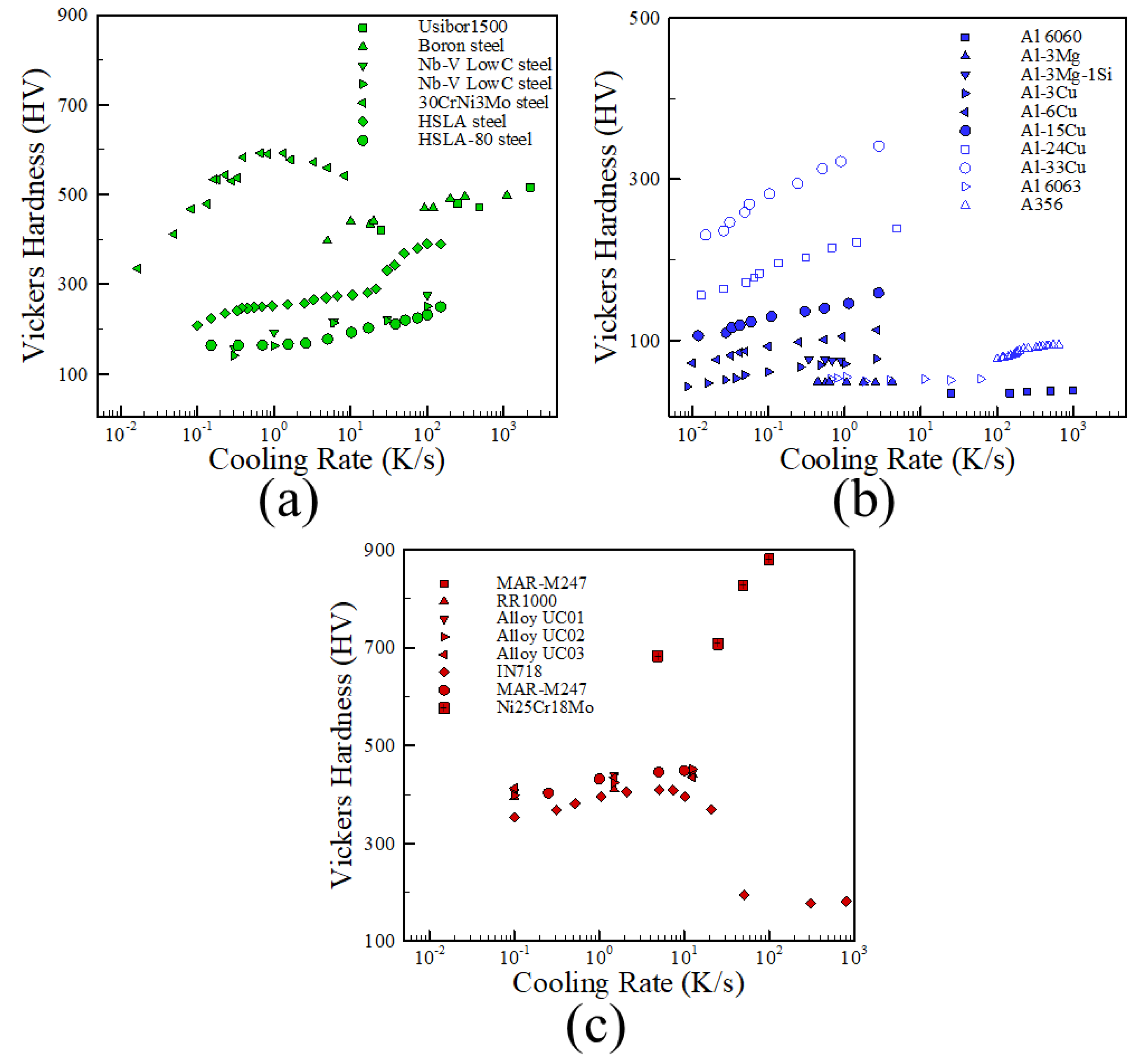
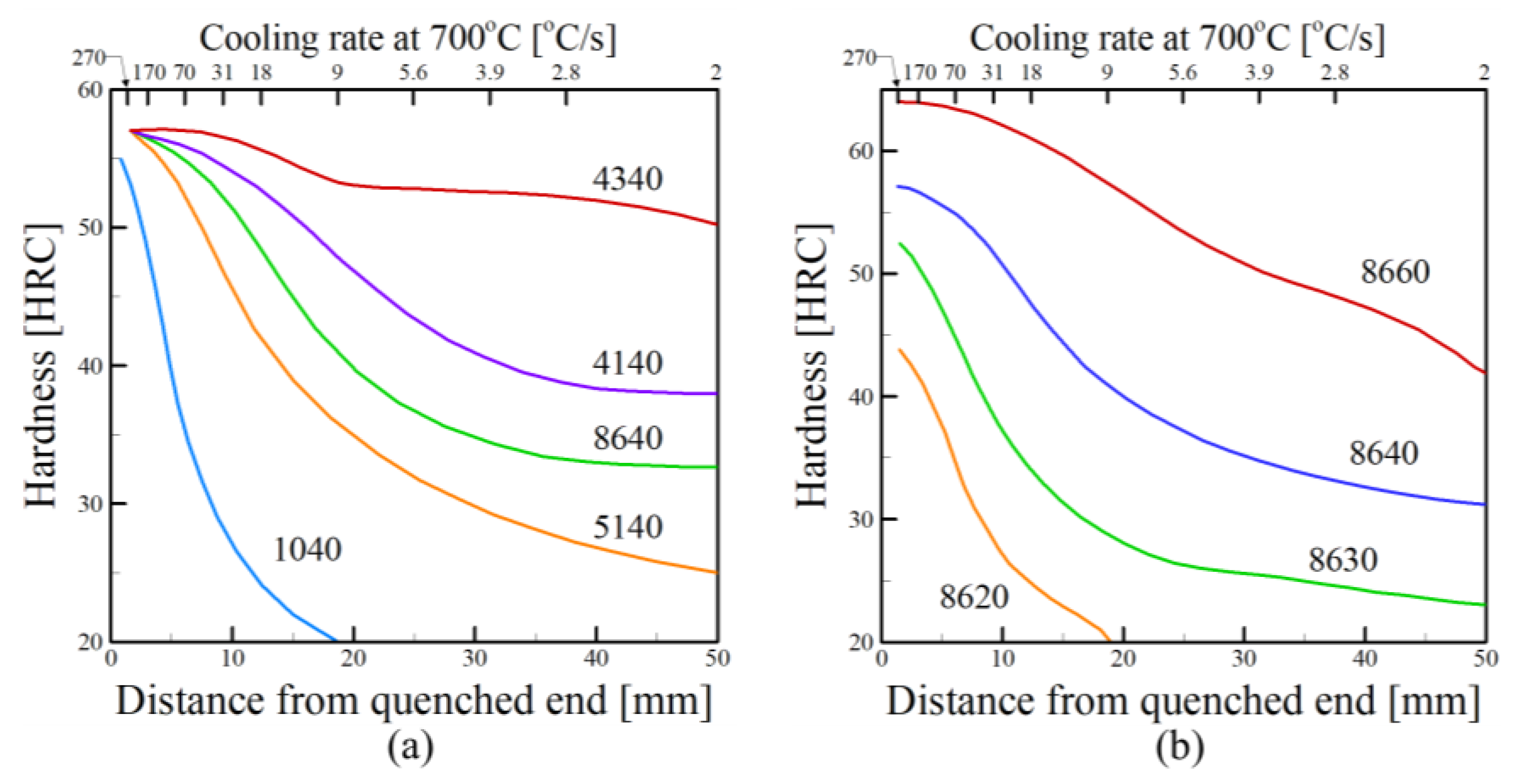
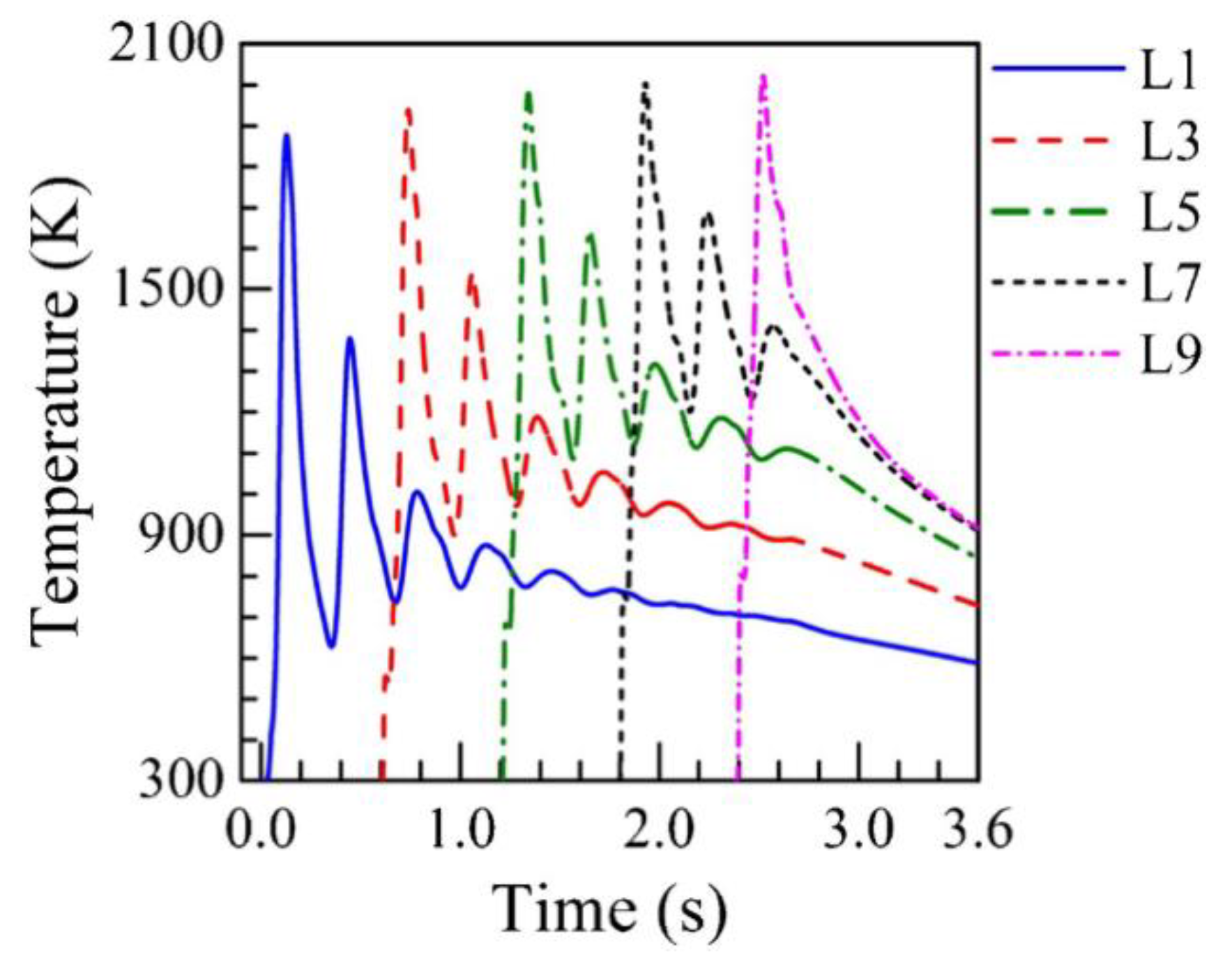
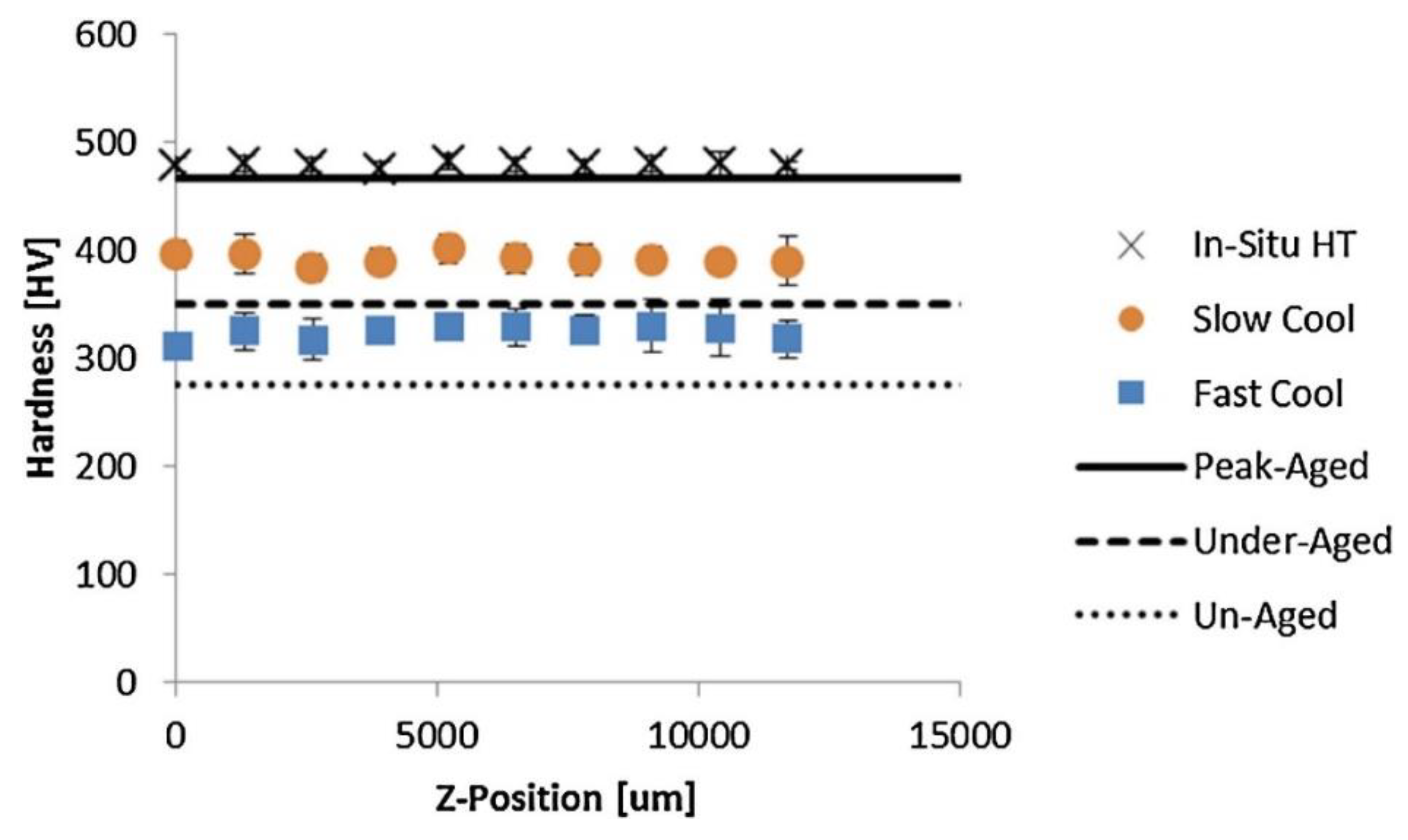
2.2. Cooling Rates
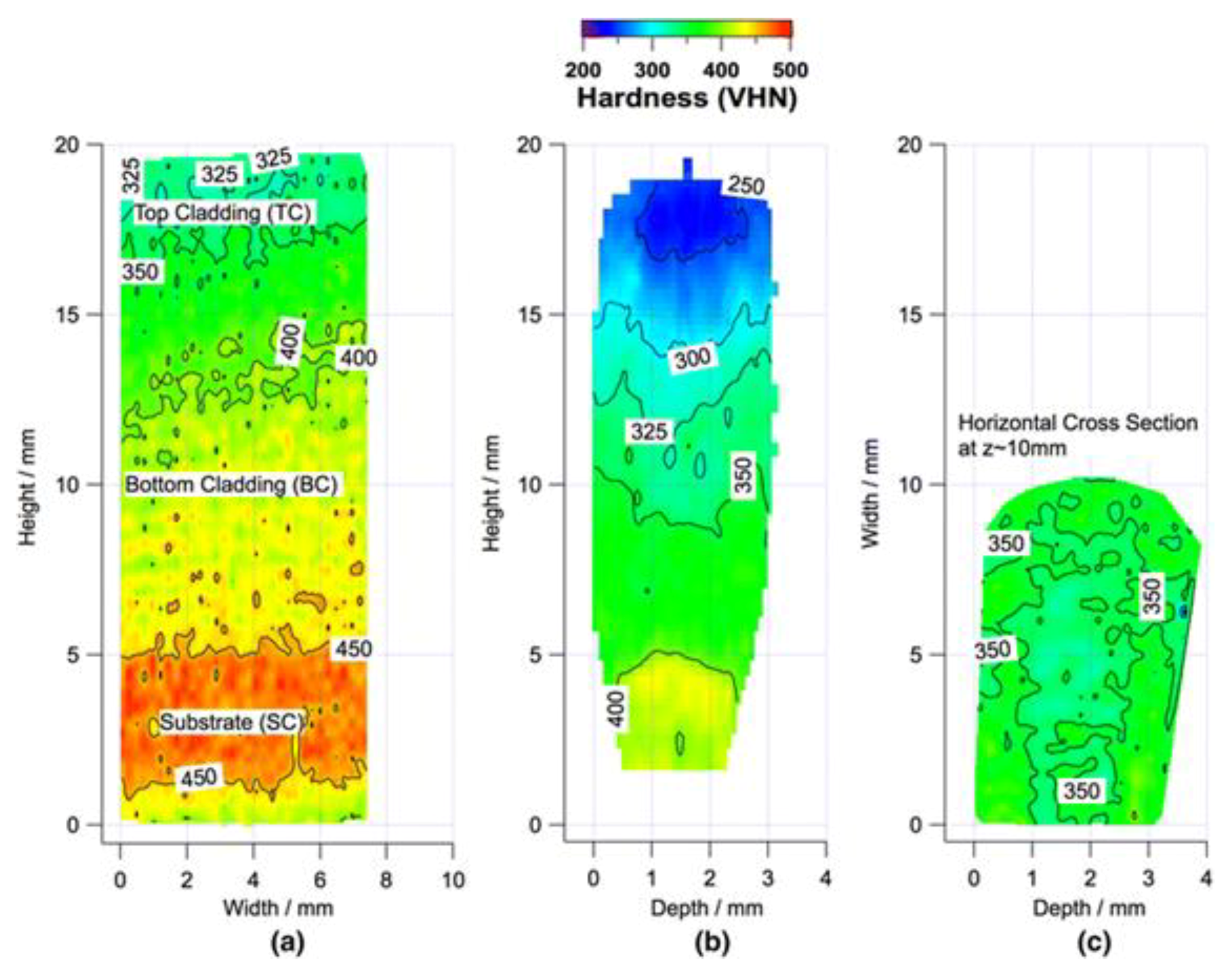
3. Effects of Microstructure
3.1. Iron Based Alloys
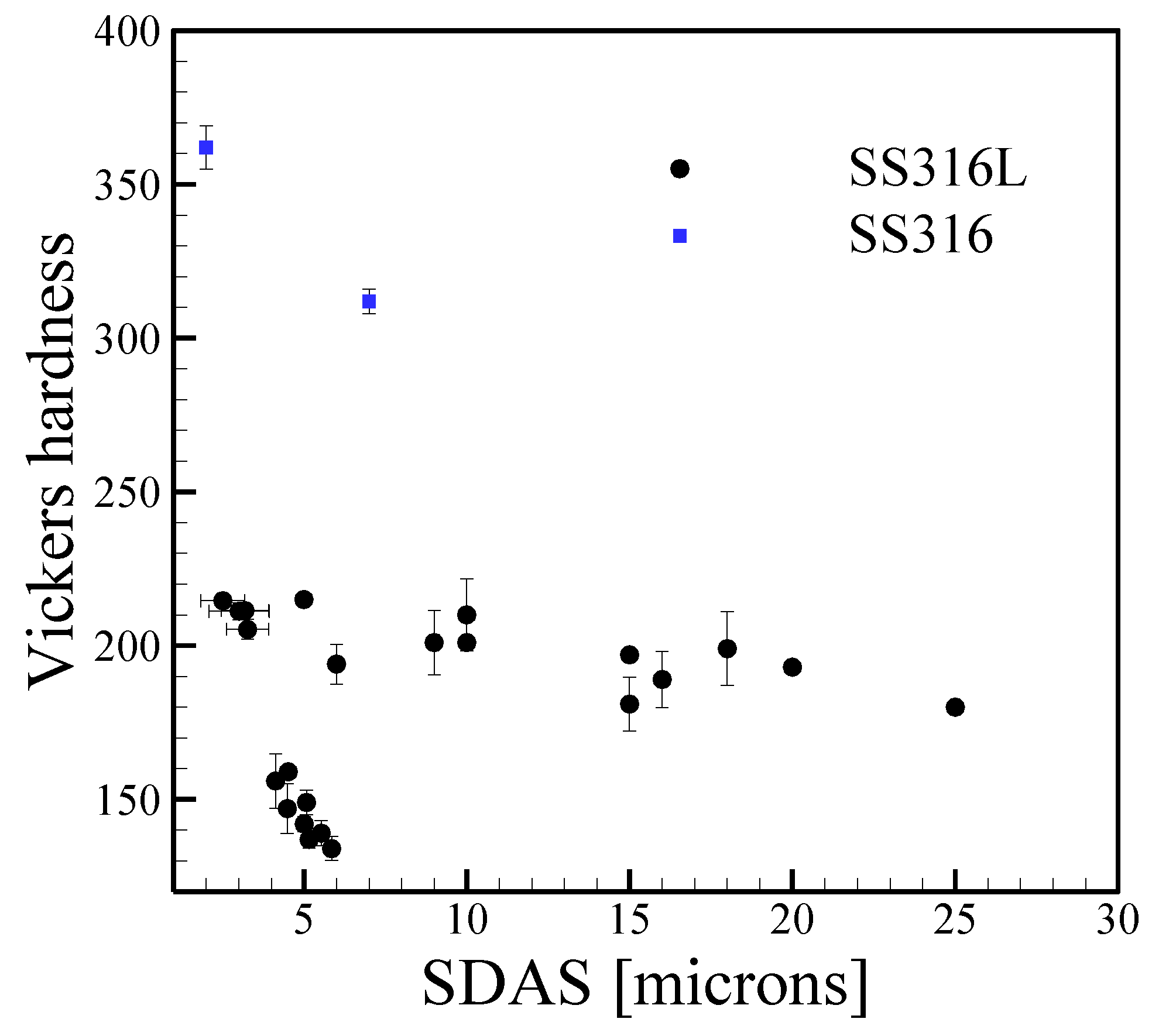
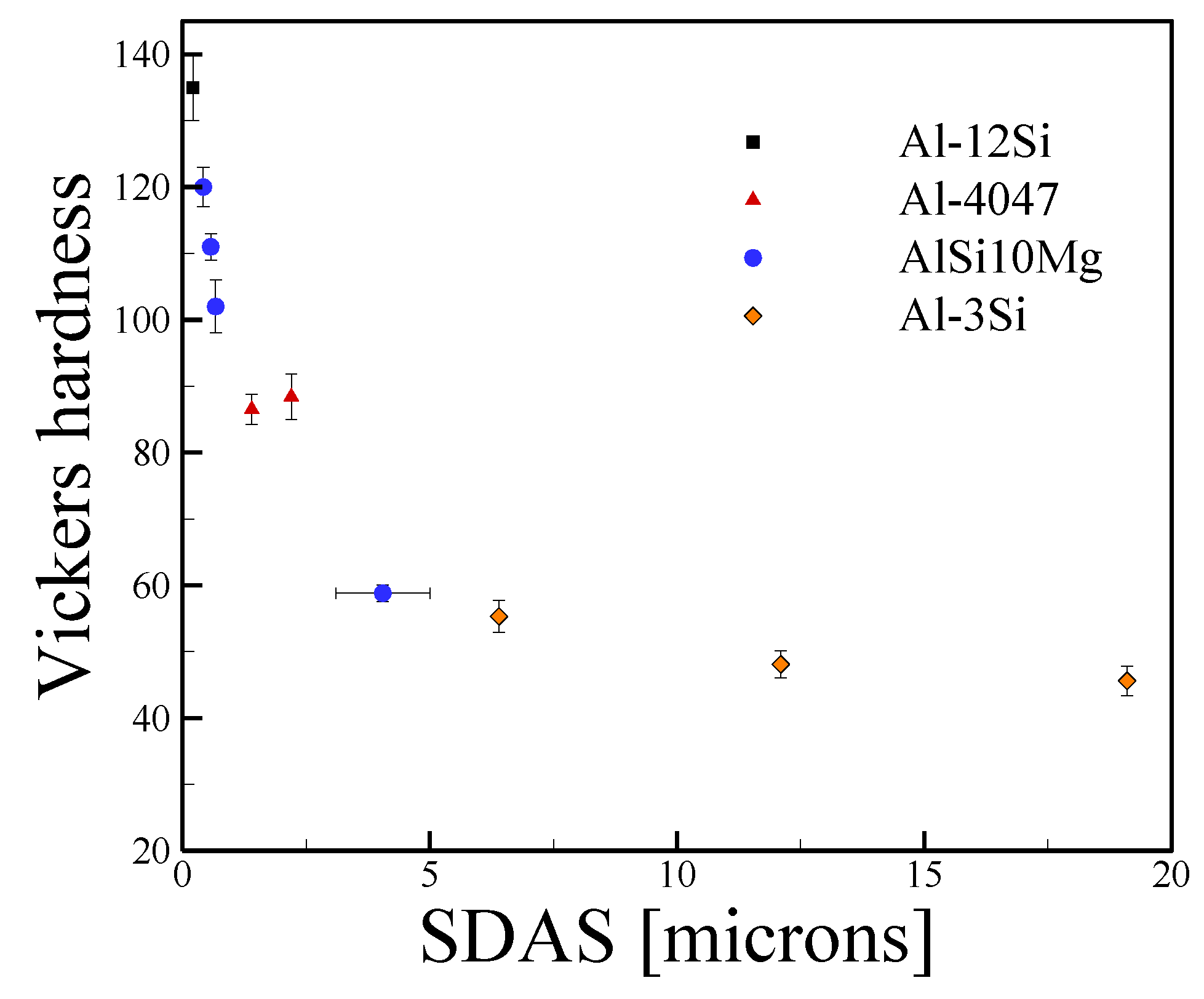
3.2. Aluminum Alloys
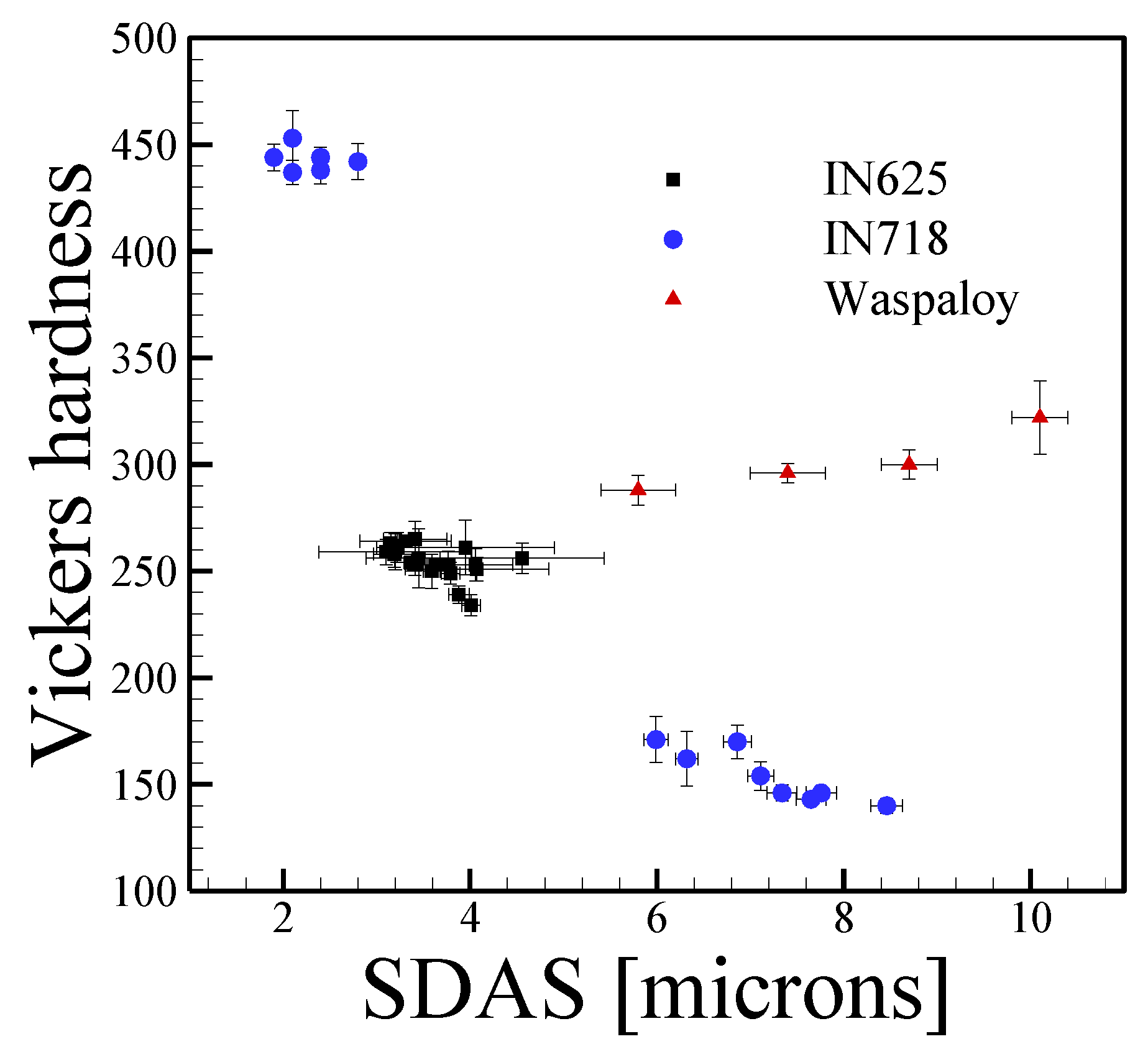
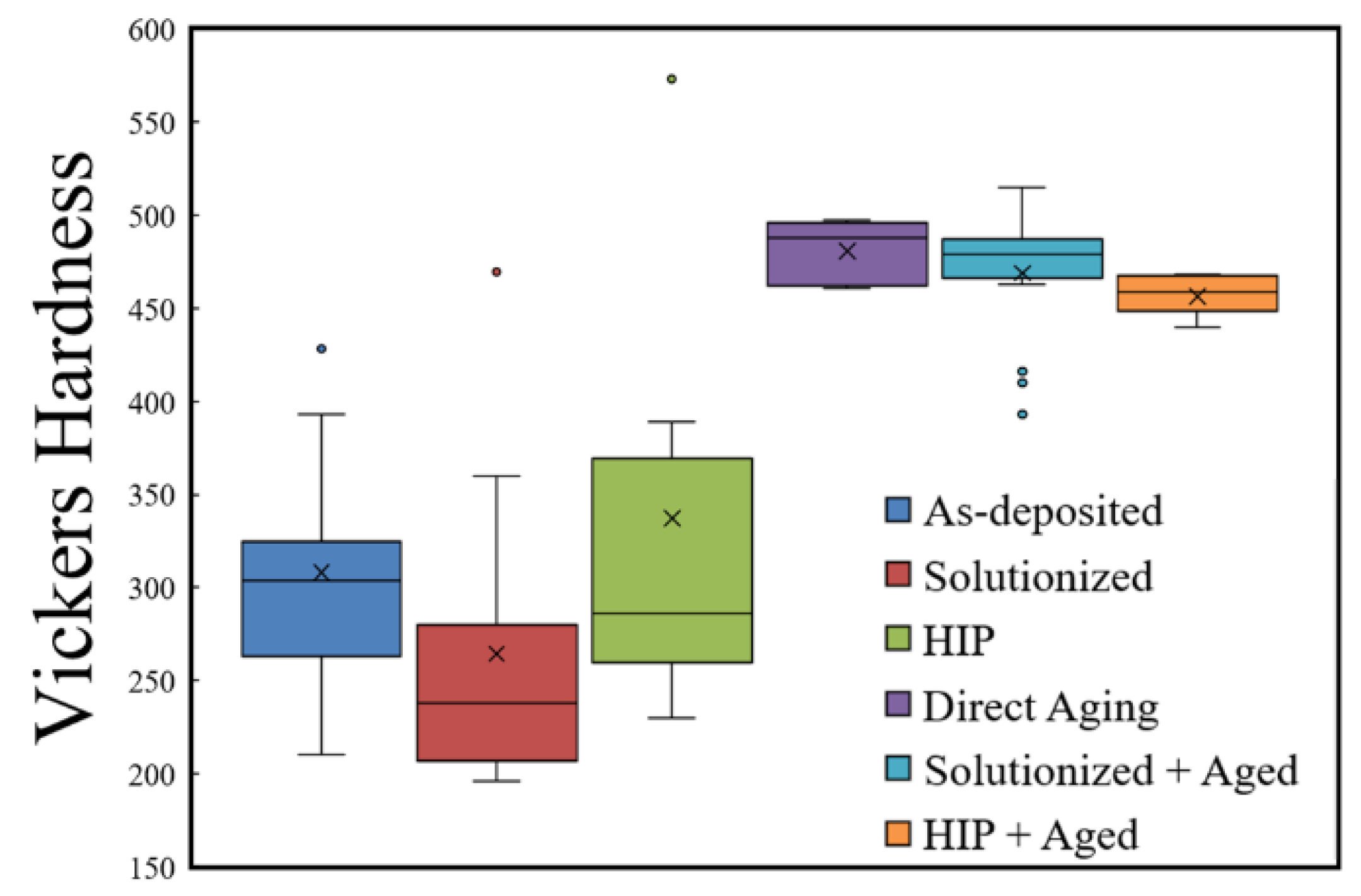
3.3. Nickel Alloys
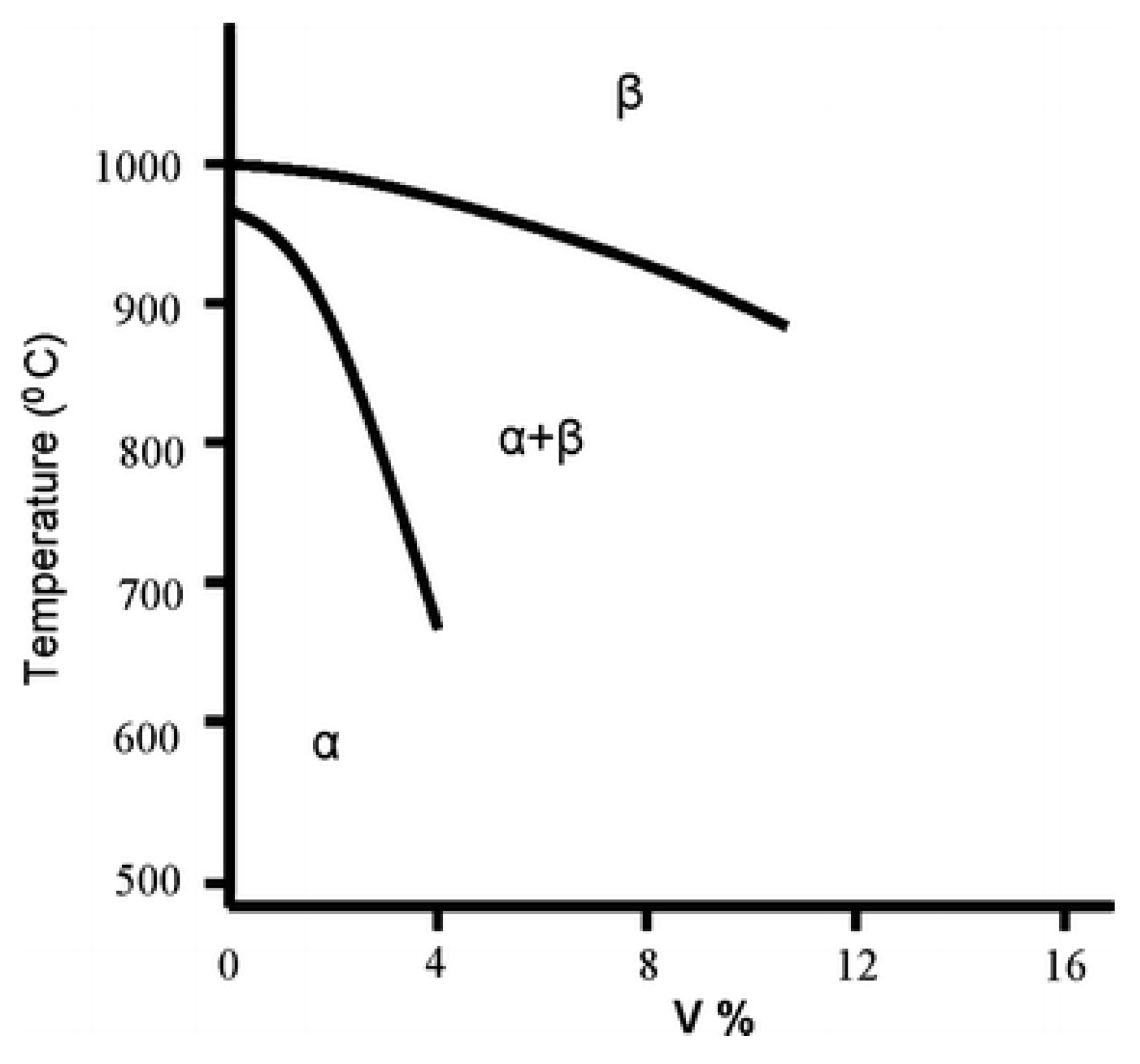
3.4. Titanium Alloys
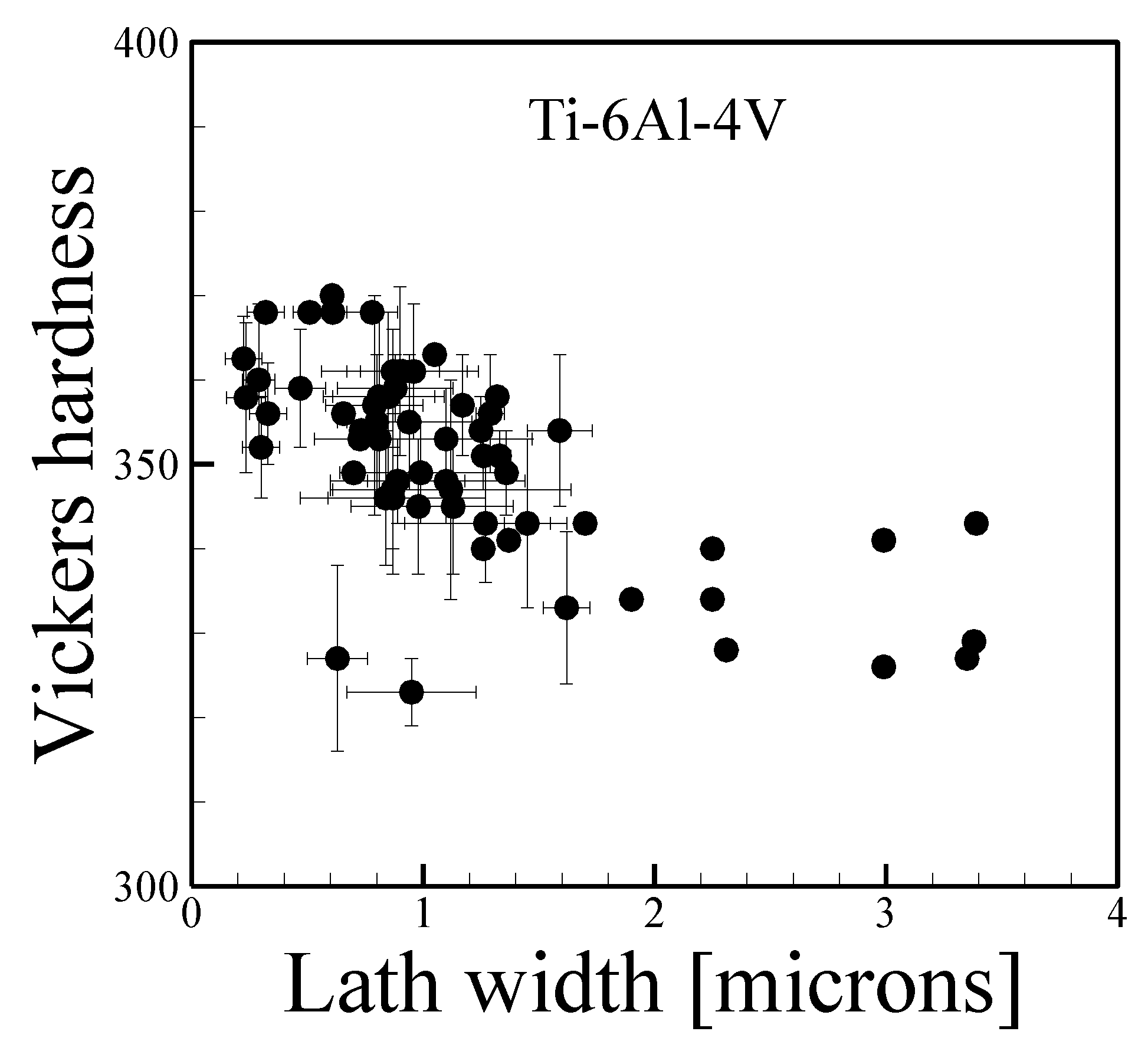
3.5. Grain Size
3.6. Dislocations in AM Materials
4. Compositional Variables
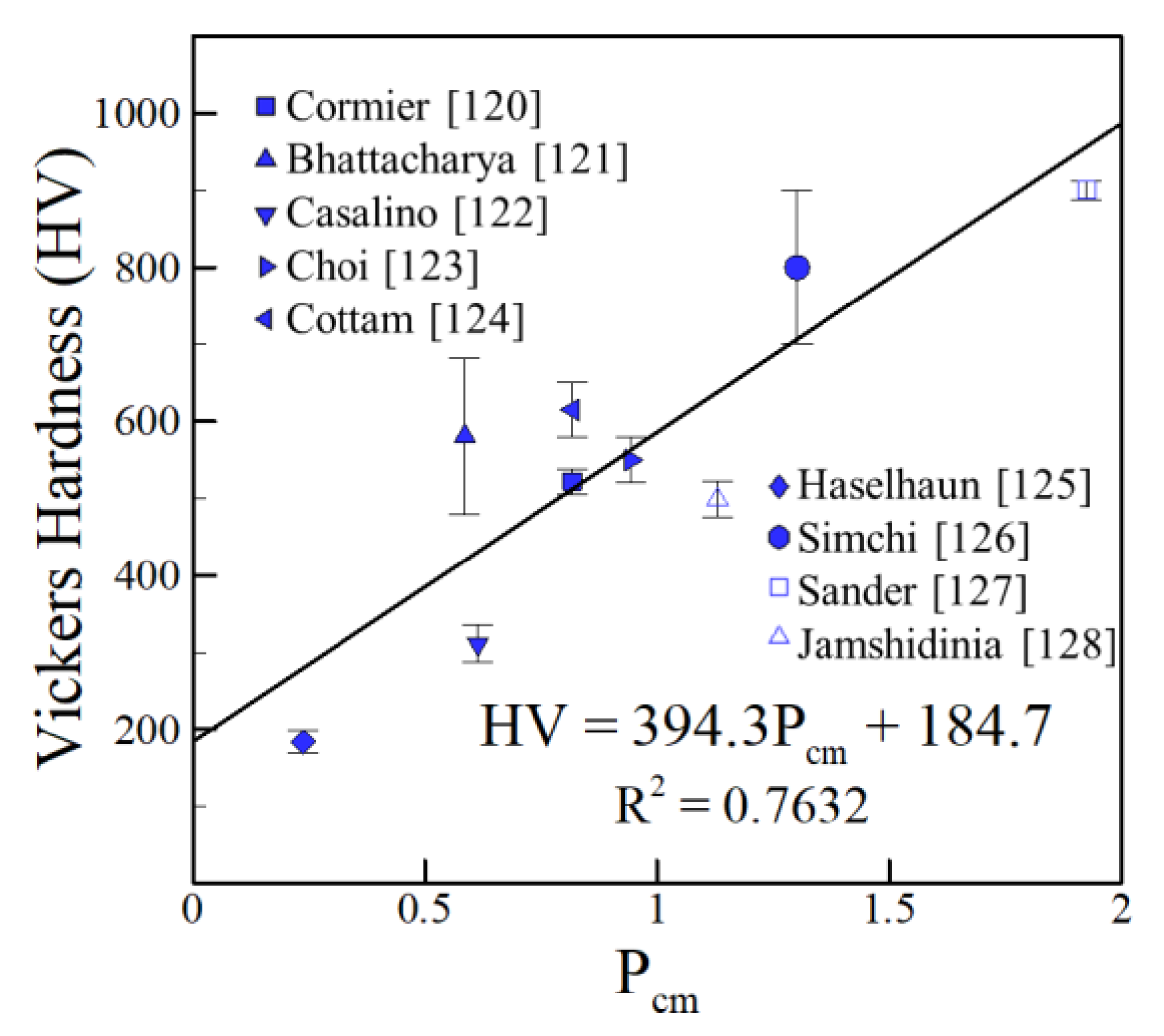
4.1. Iron Alloys
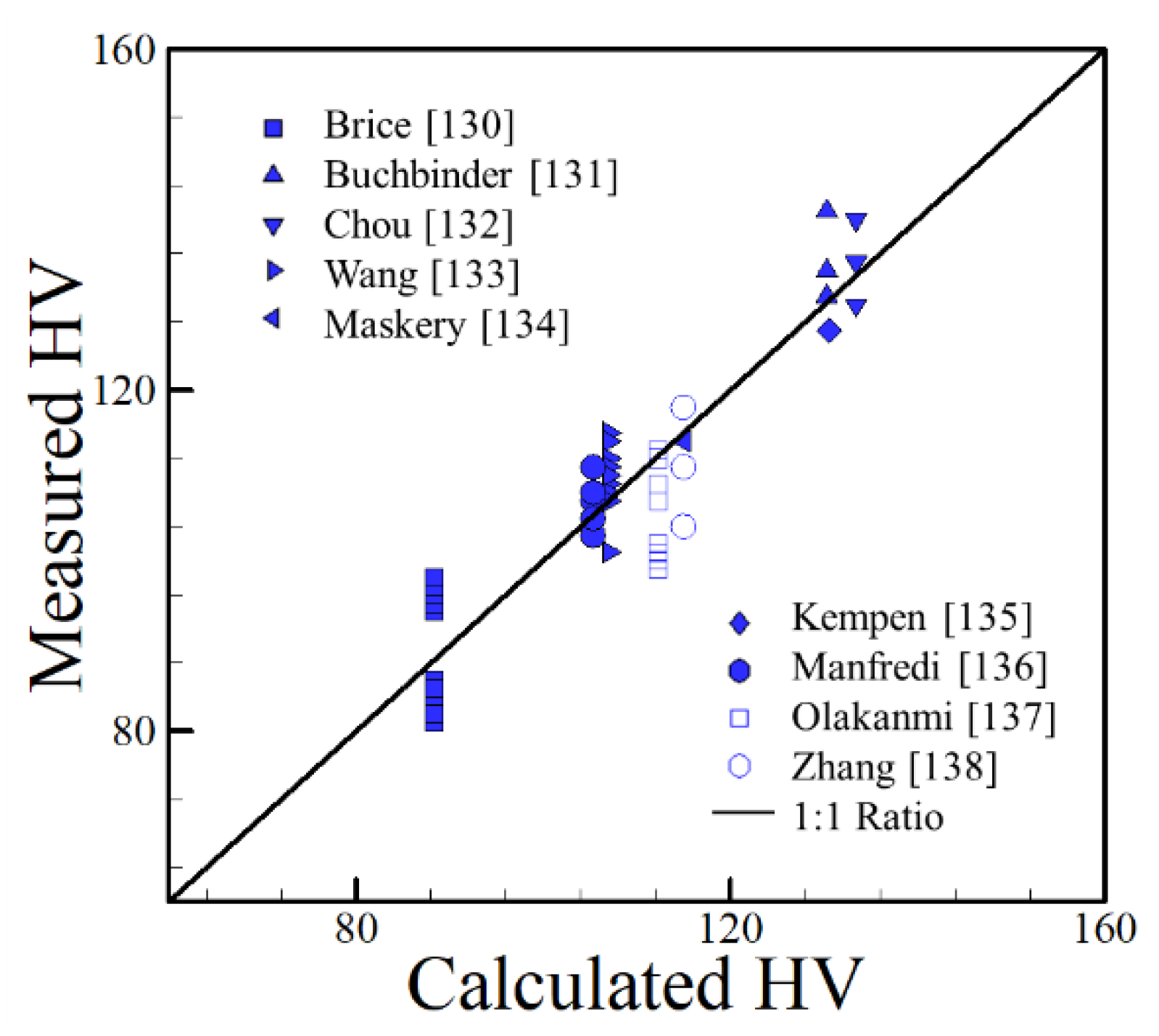
4.2. Aluminum Alloys
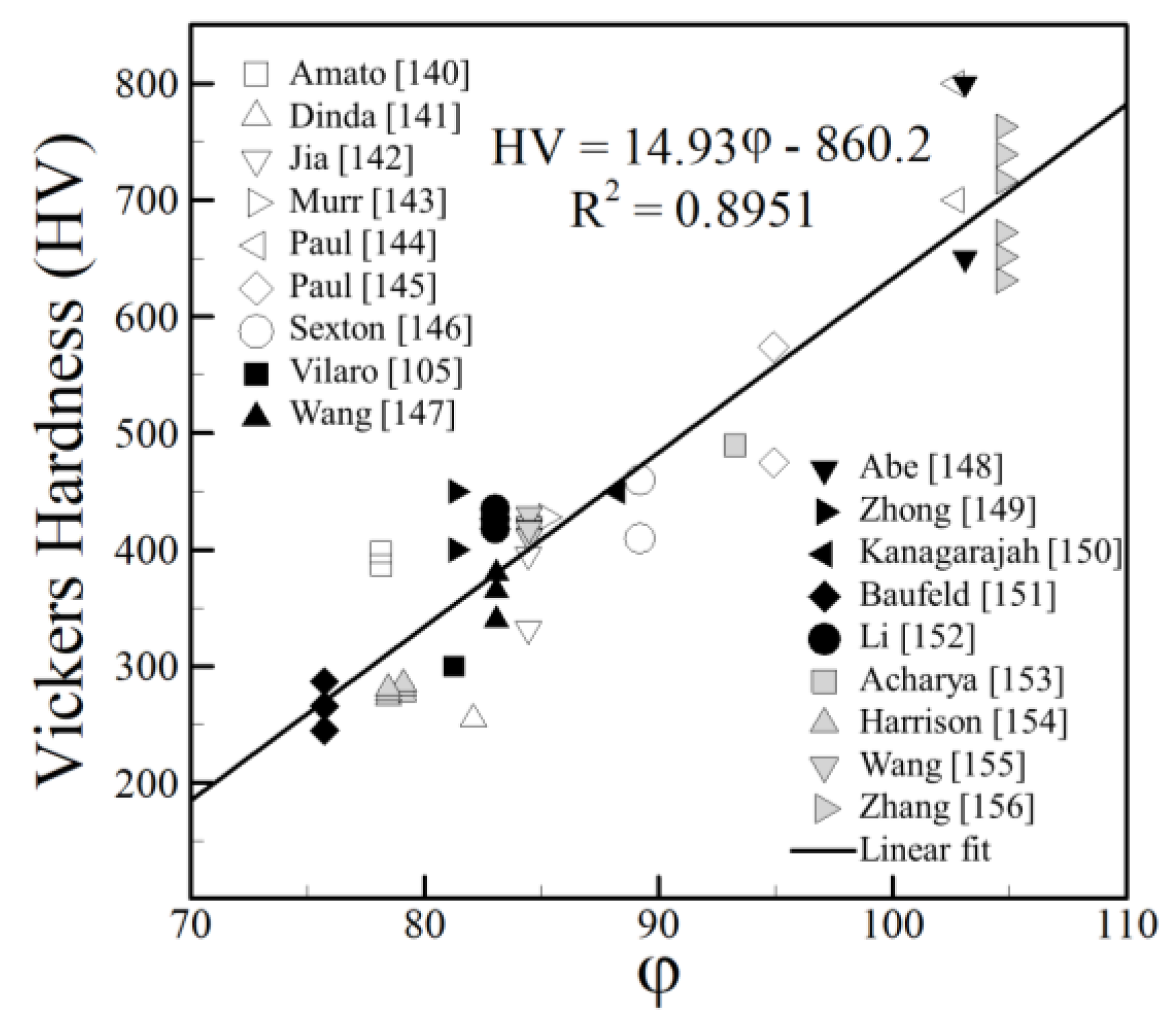
4.3. Nickel Alloys
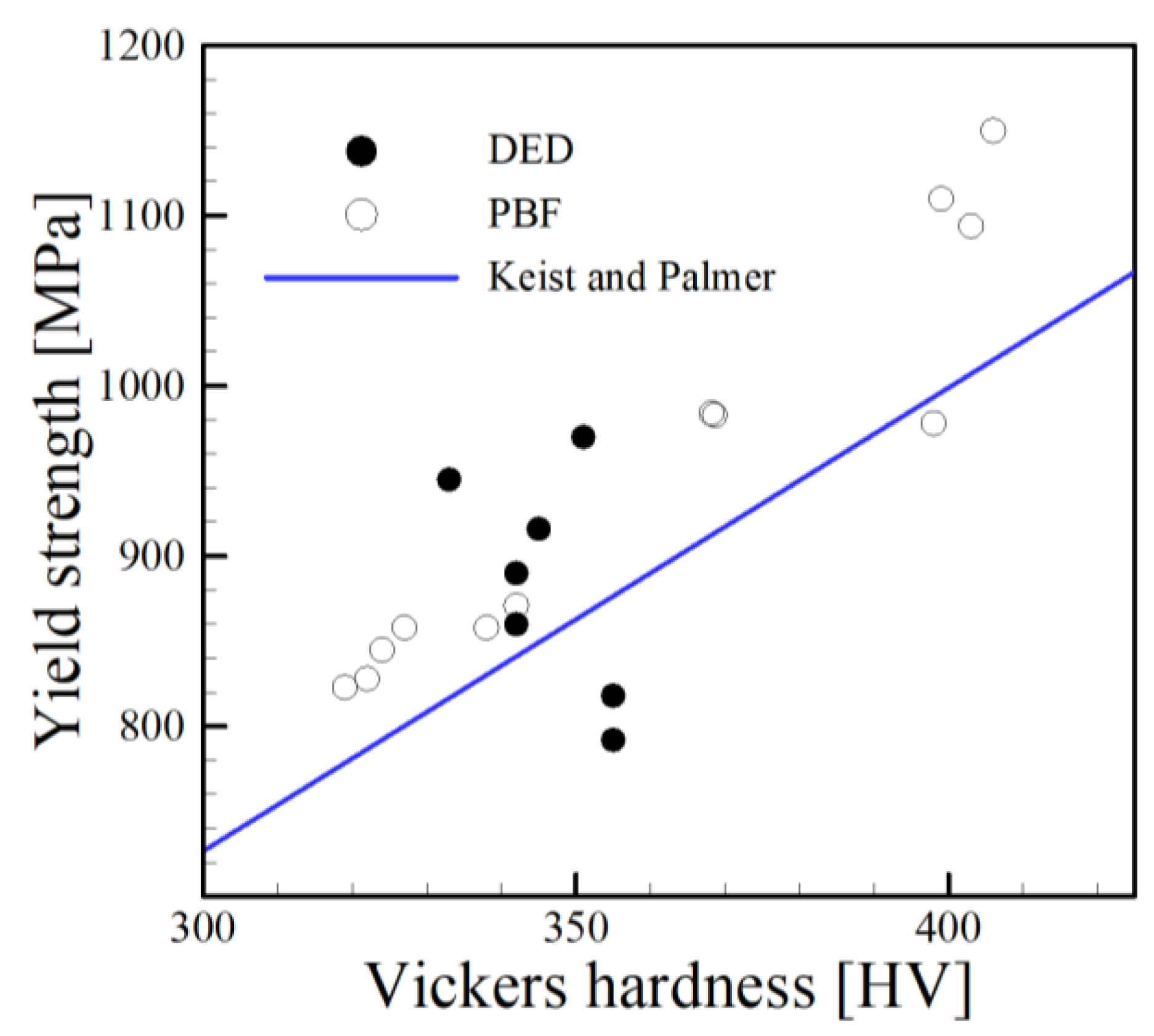
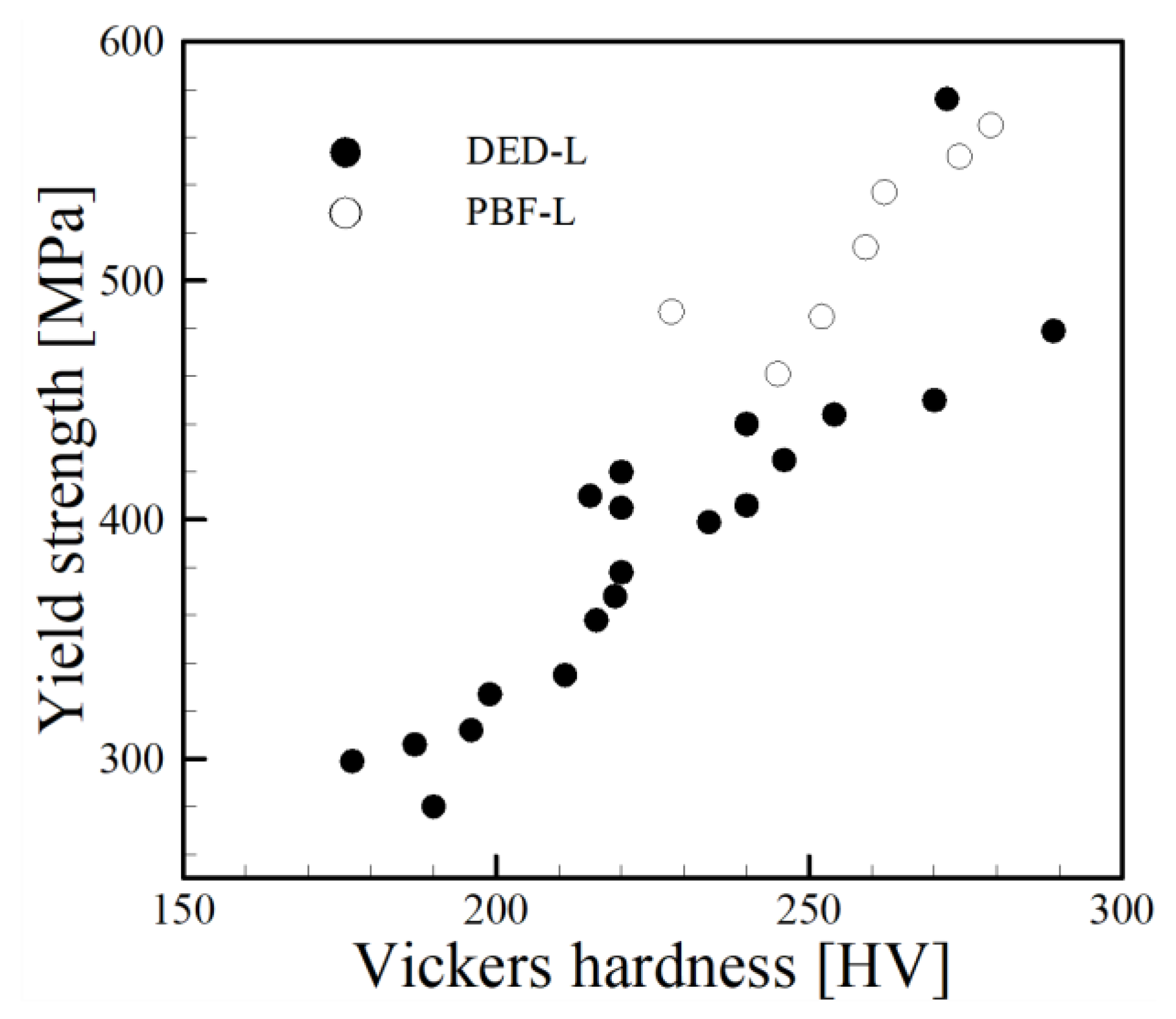
4.4. Comparison with Other Processes
5. Concluding Remarks
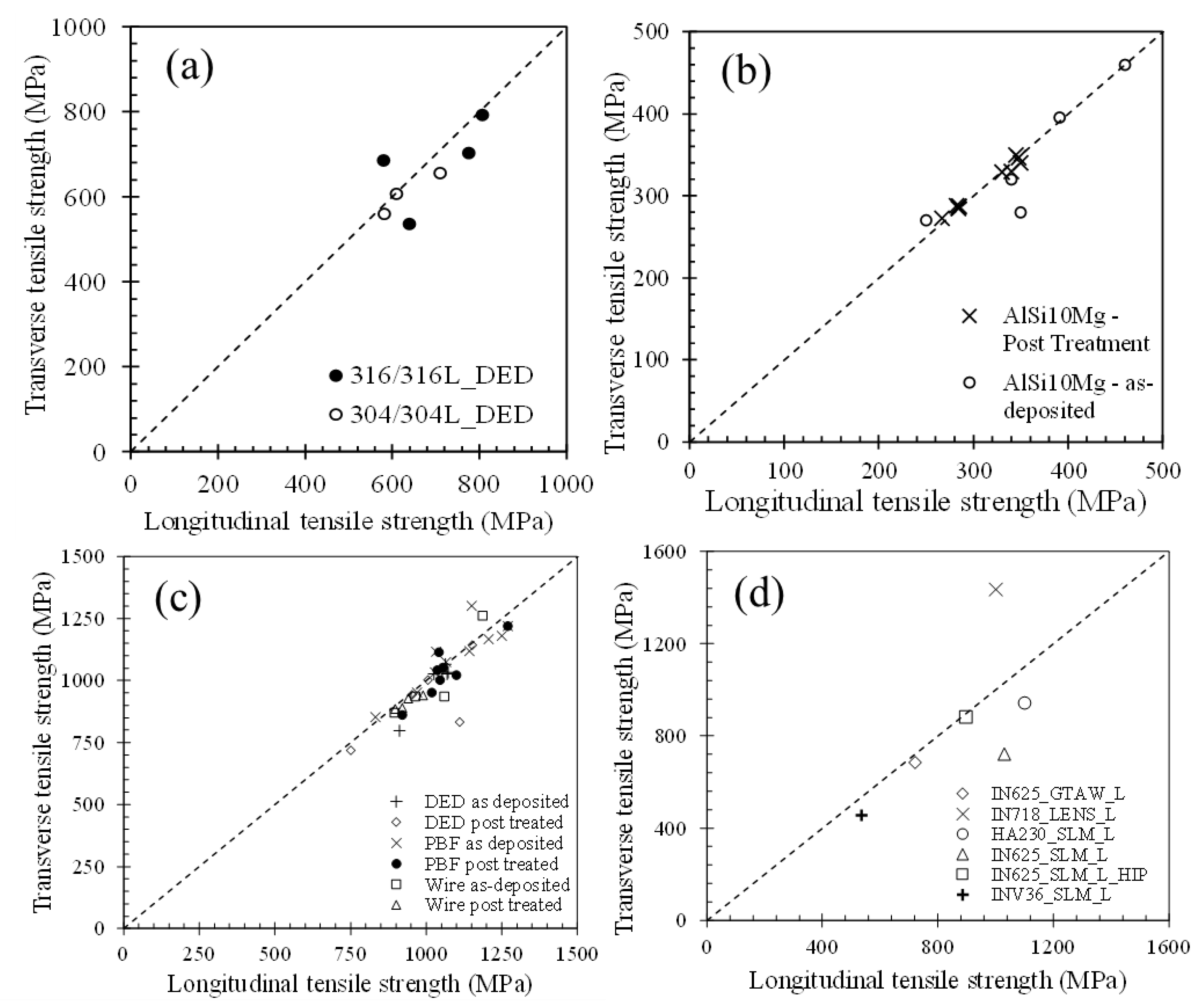
- (a)
- The variations of process parameters and cooling rates change microstructures of AM alloys. However, when the influence of AM process parameters on hardness is evaluated from the reported independent hardness values of fabricated components prior to post-deposition heat treatment, the variation of hardness falls within a narrow band of values for ferrous, aluminum and nickel alloys.
- (b)
- The range of hardness variation of AM alloys in the as-fabricated state attainable by varying AM process parameters is much lower than the hardness enhancement attainable by subsequent heat treatment or aging. The extent of hardness variation by varying AM process variables is roughly the same as the reported variations of hardness of iron and aluminum alloy weld metals when welding parameters are varied.
- (c)
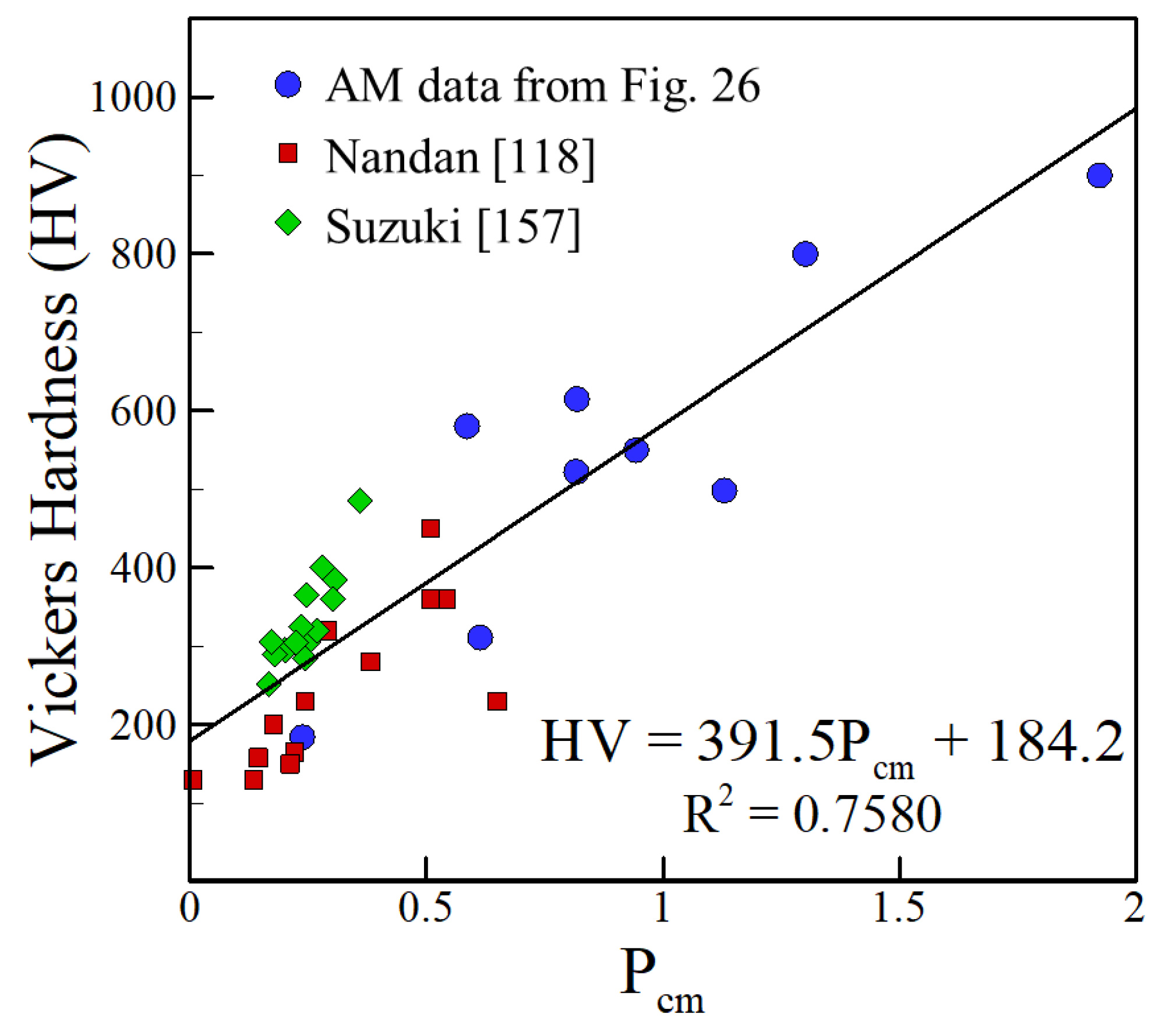
- The reported hardness data show approximate linear trends with appropriate compositional variables for iron, aluminum and nickel alloys over a wide range of AM variables and processes. The scatter in the hardness data for all alloy systems fall within a small band of values that correlates well with the concentration of alloying elements. The correlations developed are approximate and valid for the range of composition indicated but the findings are consistent over a wide range of processes and process parameters.
- (d)
- Although AM offers many advantages in fabricating metallic components, a target hardness of components in as fabricated condition is much more easily obtained by alloy selection rather than by changing AM processing variables. In this sense, the AM produced materials behave in a manner similar to other conventional metals processing technologies.
Author Contributions
Funding
Conflicts of Interest
References
- Keist, J.S.; Palmer, T.A. evelopment of strength-hardness relationships in additively manufactured titanium alloys. Mater. Sci. Eng. A 2017, 693, 214–224. [Google Scholar] [CrossRef]
- Attar, H.; Ehtemam-Haghighi, S.; Kent, D.; Okulov, I.V.; Wendrock, H.; Bӧnisch, M.; Volegov, A.S.; Calin, M.; Eckert, J.; Dargusch, M.S. Nanoindentation and wear properties of Ti and Ti-TiB composite materials produced by selective laser melting. Mater. Sci. Eng. A 2017, 688, 20–26. [Google Scholar] [CrossRef]
- Baufeld, B. Effect of deposition parameters on mechanical properties of shaped metal deposition parts. Proc. Inst. Mech. Eng. B J. Eng. 2012, 226, 126–136. [Google Scholar] [CrossRef]
- Wang, Z.; Palmer, T.A.; Beese, A.M. Effect of processing parameters on microstructure and tensile properties of austenitic stainless steel 304L made by directed energy deposition additive manufacturing. Acta Mater. 2016, 110, 226–235. [Google Scholar] [CrossRef]
- Mertens, R.; Clijsters, S.; Kempen, K.; Kruth, J.P. Optimization of scan strategies in Selective laser melting of aluminum parts with downfacing areas. J. Manuf. Sci. Eng. 2014, 136, 061012. [Google Scholar] [CrossRef]
- Kruth, J.P.; Froyen, L.; Van Vaerenbergh, J.; Mercelis, P.; Rombouts, M.; Lauwers, B. Selective laser melting of iron-based powder. J. Mater. Process. Technol. 2004, 149, 616–622. [Google Scholar] [CrossRef]
- Mukherjee, T.; Manvatkar, V.; De, A.; DebRoy, T. Mitigation of thermal distortion during additive manufacturing. Scr. Mater. 2017, 127, 79–83. [Google Scholar] [CrossRef]
- Manvatkar, V.; De, A.; DebRoy, T. Spatial variation of melt pool geometry, peak temperature and solidification parameters during laser assisted additive manufacturing process. Mater. Sci. Technol. 2015, 31, 924–930. [Google Scholar] [CrossRef]
- Manvatkar, V.; De, A.; DebRoy, T. Heat transfer and material flow during laser assisted multi-layer additive manufacturing. J. Appl. Phys. 2014, 116, 124905. [Google Scholar] [CrossRef]
- Mukherjee, T.; Zuback, J.; De, A.; DebRoy, T. Printability of alloys for additive manufacturing. Sci. Rep. 2016, 6, 19717. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, A.; Wei, H.L.; Palmer, T.A.; DebRoy, T. Heat transfer and fluid flow in additive manufacturing. J. Laser Appl. 2013, 25, 052006. [Google Scholar] [CrossRef]
- Wei, H.; Mazumder, J.; DebRoy, T. Evolution of solidification texture during additive manufacturing. Sci. Rep. 2015, 5, 16446. [Google Scholar] [CrossRef] [PubMed]
- Leuders, S.; Thöne, M.; Riemer, A.; Niendorf, T.; Tröster, T.; Richard, H.; Maier, H. On the mechanical behaviour of titanium alloy TiAl6V4 manufactured by selective laser melting: Fatigue resistance and crack growth performance. Int. J. Fatigue 2013, 48, 300–307. [Google Scholar] [CrossRef]
- Zhao, X.; Lin, X.; Chen, J.; Xue, L.; Huang, W. The effect of hot isostatic pressing on crack healing, microstructure, mechanical properties of Rene88DT superalloy prepared by laser solid forming. Mater. Sci. Eng. A 2009, 504, 129–134. [Google Scholar] [CrossRef]
- Jägle, E.A.; Sheng, Z.; Wu, L.; Lu, L.; Risse, J.; Weisheit, A.; Raabe, D. Precipitation reactions in age-hardenable alloys during laser additive manufacturing. JOM 2016, 68, 943–949. [Google Scholar] [CrossRef]
- Beese, A.M.; Carroll, B.E. Review of mechanical properties of Ti-6Al-4V made by laser-based additive manufacturing using powder feedstock. JOM 2016, 68, 724–734. [Google Scholar] [CrossRef]
- DebRoy, T.; Wei, H.; Zuback, J.; Mukherjee, T.; Elmer, J.; Milewski, J.; Beese, A.; Wilson-Heid, A.; De, A.; Zhang, W. Additive manufacturing of metallic components–process, structure and properties. Prog. Mater. Sci. 2018, 92, 112–224. [Google Scholar] [CrossRef]
- Lewandowski, J.J.; Seifi, M. Metal additive manufacturing: A review of mechanical properties. Ann. Rev. Mater. Res. 2016, 46, 151–186. [Google Scholar] [CrossRef]
- Gorsse, S.; Hutchinson, C.; Gouné, M.; Banerjee, R. Additive manufacturing of metals: A brief review of the characteristic microstructures and properties of steels, Ti-6Al-4V and high-entropy alloys. Sci. Technol. Adv. Mater. 2017, 18, 584–610. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.D.; Meiners, W.; Wissenbach, K.; Poprawe, R. Laser additive manufacturing of metallic components: Materials, processes and mechanisms. Int. Mater. Rev. 2012, 57, 133–164. [Google Scholar] [CrossRef]
- Sames, W.J.; List, F.; Pannala, S.; Dehoff, R.R.; Babu, S.S. The metallurgy and processing science of metal additive manufacturing. Int. Mater. Rev. 2016, 61, 315–360. [Google Scholar] [CrossRef]
- Frazier, W.E. Metal additive manufacturing: A review. J. Mater. Eng. Perform. 2014, 23, 1917–1928. [Google Scholar] [CrossRef]
- Murr, L.E.; Gaytan, S.M.; Ramirez, D.A.; Martinez, E.; Hernandez, J.; Amato, K.N.; Shindo, P.W.; Medina, F.R.; Wicker, R.B. Metal fabrication by additive manufacturing using laser and electron beam melting technologies. J. Mater. Sci. Technol. 2012, 28, 1–14. [Google Scholar] [CrossRef]
- Körner, C. Additive manufacturing of metallic components by selective electron beam melting—A review. Int. Mater. Rev. 2016, 61, 361–377. [Google Scholar] [CrossRef]
- Yap, C.; Chua, C.; Dong, Z.; Liu, Z.; Zhang, D.; Loh, L.; Sing, S. Review of selective laser melting: Materials and applications. Appl. Phys. Rev. 2015, 2, 041101. [Google Scholar] [CrossRef]
- Herzog, D.; Seyda, V.; Wycisk, E.; Emmelmann, C. Additive manufacturing of metals. Acta Mater. 2016, 117, 371–392. [Google Scholar] [CrossRef]
- Zhang, D.; Sun, S.; Qiu, D.; Gibson, M.A.; Dargusch, M.S.; Brandt, M.; Qian, M.; Easton, M. Metal alloys for fusion-based additive manufacturing. Adv. Eng. Mater. 2018. [Google Scholar] [CrossRef]
- Yan, W.; Lin, S.; Kafka, O.L.; Yu, C.; Liu, Z.; Lian, Y.; Wolff, S.; Cao, J.; Wagner, G.J.; Liu, W.K. Modeling process-structure-property relationships for additive manufacturing. Front. Mech. Eng. 2018, 13, 482–492. [Google Scholar] [CrossRef]
- Cao, F.; Zhang, T.; Ryder, M.A.; Lados, D.A. A review of the fatigue properties of additively manufactured Ti-6Al-4V. JOM 2018, 70, 349–357. [Google Scholar] [CrossRef]
- Kok, Y.; Tan, X.P.; Wang, P.; Nai, M.; Loh, N.H.; Liu, E.; Tor, S.B. Anisotropy and heterogeneity of microstructure and mechanical properties in metal additive manufacturing: A critical review. Mater. Des. 2018, 139, 565–586. [Google Scholar] [CrossRef]
- Agius, D.; Kourousis, K.I.; Wallbrink, C. A review of the as-built SLM Ti-6Al-4V mechanical properties towards achieving fatigue resistant designs. Metals 2018, 8, 75. [Google Scholar] [CrossRef]
- Yusuf, S.M.; Gao, N. Influence of energy density on metallurgy and properties in metal additive manufacturing. Mater. Sci. Technol. 2017, 33, 1269–1289. [Google Scholar] [CrossRef]
- Easton, M.; Qian, M.; Prasad, A.; StJohn, D. Recent advances in grain refinement of light metals and alloys. Curr. Opin. Solid State Mater. Sci. 2016, 20, 13–24. [Google Scholar] [CrossRef]
- Collins, P.; Brice, D.; Samimi, P.; Ghamarian, I.; Fraser, H. Microstructural control of additively manufactured metallic materials. Ann. Rev. Mater. Res. 2016, 46, 63–91. [Google Scholar] [CrossRef]
- Derekar, K. A review of wire arc additive manufacturing and advances in wire arc additive manufacturing of aluminium. Mater. Sci. Technol. 2018, 34, 1–22. [Google Scholar] [CrossRef]
- Shipley, H.; McDonnell, D.; Culleton, M.; Lupoi, R.; O’Donnell, G.; Trimble, D. Optimisation of process parameters to address fundamental challenges during selective laser melting of Ti-6Al-4V: A review. Int. J. Mach. Tools Manuf. 2018, 128, 1–20. [Google Scholar] [CrossRef]
- Hitzler, L.; Merkel, M.; Hall, W.; Öchsner, A. A review of metal fabricated with laser-and powder-bed based additive manufacturing techniques: Process, nomenclature, materials, achievable properties and its utilization in the medical sector. Adv. Eng. Mater. 2018, 20, 1700658. [Google Scholar] [CrossRef]
- Zhang, L.C.; Liu, Y.; Li, S.; Hao, Y. Additive manufacturing of titanium alloys by electron beam melting: A review. Adv. Eng. Mater. 2018, 20, 1700842. [Google Scholar] [CrossRef]
- Song, B.; Zhao, X.; Li, S.; Han, C.; Wei, Q.; Wen, S.; Liu, J.; Shi, Y. Differences in microstructure and properties between selective laser melting and traditional manufacturing for fabrication of metal parts: A review. Front. Mech. Eng. 2015, 10, 111–125. [Google Scholar] [CrossRef]
- Shamsaei, N.; Yadollahi, A.; Bian, L.; Thompson, S.M. An overview of Direct Laser Deposition for additive manufacturing; Part II: Mechanical behavior, process parameter optimization and control. Addit. Manuf. 2015, 8, 12–35. [Google Scholar] [CrossRef]
- Trevisan, F.; Calignano, F.; Lorusso, M.; Pakkanen, J.; Aversa, A.; Ambrosio, E.P.; Lombardi, M.; Fino, P.; Manfredi, D. On the selective laser melting (SLM) of the AlSi10Mg alloy: Process, microstructure and mechanical properties. Materials 2017, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gong, X.; Chou, K. Review on powder-bed laser additive manufacturing of Inconel 718 parts. Proc. Inst. Mech. Eng. B J. Eng. 2017, 231, 1890–1903. [Google Scholar] [CrossRef]
- Yan, M.; Xu, W.; Dargusch, M.; Tang, H.; Brandt, M.; Qian, M. Review of effect of oxygen on room temperature ductility of titanium and titanium alloys. Powder Metall. 2014, 57, 251–257. [Google Scholar] [CrossRef]
- Saboori, A.; Gallo, D.; Biamino, S.; Fino, P.; Lombardi, M. An overview of additive manufacturing of titanium components by directed energy deposition: Microstructure and mechanical properties. Appl. Sci. 2017, 7, 883. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, S.; Liu, W.; Shang, X. Characterization of stainless steel parts by laser metal deposition shaping. Mater. Des. 2014, 55, 104–119. [Google Scholar] [CrossRef]
- Yu, J.; Rombouts, M.; Maes, G. Cracking behavior and mechanical properties of austenitic stainless steel parts produced by laser metal deposition. Mater. Des. 2013, 45, 228–235. [Google Scholar] [CrossRef]
- Li, J.; Deng, D.; Hou, X.; Wang, X.; Ma, G.; Wu, D.; Zhang, G. Microstructure and performance optimisation of stainless steel formed by laser additive manufacturing. Mater. Sci. Technol. 2016, 32, 1223–1230. [Google Scholar] [CrossRef]
- Ziętala, M.; Durejko, T.; Polański, M.; Kunce, I.; Płociński, T.; Zieliński, W.; Łazińska, M.; Stępniowski, W.; Czujko, T.; Kurzydłowski, K.J.; et al. The microstructure, mechanical properties and corrosion resistance of 316L stainless steel fabricated using laser engineered net shaping. Mater. Sci. Eng. A 2016, 677, 1–10. [Google Scholar] [CrossRef]
- Ma, M.; Wang, Z.; Wang, D.; Zeng, X. Control of shape and performance for direct laser fabrication of precision large-scale metal parts with 316L Stainless Steel. Opt. Laser Technol. 2013, 45, 209–216. [Google Scholar] [CrossRef]
- Abd-Elghany, K.; Bourell, D.L. Property evaluation of 304L stainless steel fabricated by selective laser melting. Rapid Prototyp. J. 2012, 18, 420–428. [Google Scholar] [CrossRef]
- Tolosa, I.; Garciandia, F.; Zubiri, F.; Zapirain, F.; Esnaola, A. Study of mechanical properties of AISI 316 stainless steel processed by “selective laser melting”, following different manufacturing strategies. Int. J. Adv. Manuf. Technol. 2010, 51, 639–647. [Google Scholar] [CrossRef]
- Sun, Z.; Tan, X.; Tor, S.B.; Yeong, W.Y. Selective laser melting of stainless steel 316L with low porosity and high build rates. Mater. Des. 2016, 104, 197–204. [Google Scholar] [CrossRef]
- Knapp, G.L.; Mukherjee, T.; Zuback, J.S.; Wei, H.L.; Palmer, T.A.; De, A.; DebRoy, T. Building blocks for a digital twin of additive manufacturing. Acta Mater. 2017, 135, 390–399. [Google Scholar] [CrossRef]
- Galarraga, H.; Lados, D.A.; Dehoff, R.R.; Kirka, M.M.; Nandwana, P. Effects of the microstructure and porosity on properties of Ti-6Al-4V ELI alloy fabricated by electron beam melting (EBM). Addit. Manuf. 2016, 10, 47–57. [Google Scholar] [CrossRef]
- Brandl, E.; Baufeld, B.; Leyens, C.; Gault, R. Additive manufactured Ti-6Al-4V using welding wire: Comparison of laser and arc beam deposition and evaluation with respect to aerospace material specifications. Phys. Procedia 2010, 5, 595–606. [Google Scholar] [CrossRef]
- Brandl, E.; Schoberth, A.; Leyens, C. Morphology, microstructure and hardness of titanium (Ti-6Al-4V) blocks deposited by wire-feed additive layer manufacturing (ALM). Mater. Sci. Eng. A 2012, 532, 295–307. [Google Scholar] [CrossRef]
- Carroll, B.E.; Palmer, T.A.; Beese, A.M. Anisotropic tensile behavior of Ti-6Al-4V components fabricated with directed energy deposition additive manufacturing. Acta Mater. 2015, 87, 309–320. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, J.; Tian, X.; Wang, H.; Liu, D. Microstructure and mechanical properties of hybrid fabricated Ti–6.5 Al–3.5 Mo–1.5 Zr–0.3 Si titanium alloy by laser additive manufacturing. Mater. Sci. Eng. A 2014, 607, 427–434. [Google Scholar] [CrossRef]
- Vrancken, B.; Thijs, L.; Kruth, J.P.; Van Humbeeck, J. Microstructure and mechanical properties of a novel β titanium metallic composite by selective laser melting. Acta Mater. 2014, 68, 150–158. [Google Scholar] [CrossRef]
- Baufeld, B.; Van der Biest, O. Mechanical properties of Ti-6Al-4V specimens produced by shaped metal deposition. Sci. Technol. Adv. Mater. 2009, 10, 015008. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.; Osakada, K.; Kitamura, Y.; Matsumoto, M.; Shiomi, M. Manufacturing of titanium parts for medical purposes by selective laser melting. In Proceedings of the 8th International Conference Rapid Prototyping, Tokyo, Japan, 12–13 June 2000; pp. 288–293. [Google Scholar]
- Hrabe, N.; Quinn, T. Effects of processing on microstructure and mechanical properties of a titanium alloy (Ti-6Al-4V) fabricated using electron beam melting (EBM), Part 2: Energy input, orientation and location. Mater. Sci. Eng. A 2013, 573, 271–277. [Google Scholar] [CrossRef]
- Attar, H.; Calin, M.; Zhang, L.C.; Scudino, S.; Eckert, J. Manufacture by selective laser melting and mechanical behavior of commercially pure titanium. Mater. Sci. Eng. A 2014, 593, 170–177. [Google Scholar] [CrossRef]
- Murr, L.E.; Gaytan, S.M.; Ceylan, A.; Martinez, E.; Martinez, J.L.; Hernandez, D.H.; Machado, B.I.; Ramirez, D.A.; Medina, F.; Collins, S.; et al. Characterization of titanium aluminide alloy components fabricated by additive manufacturing using electron beam melting. Acta Mater. 2010, 58, 1887–1894. [Google Scholar] [CrossRef]
- Schwab, H.; Prashanth, K.; Löber, L.; Kühn, U.; Eckert, J. Selective Laser Melting of Ti-45Nb Alloy. Metals 2015, 5, 686. [Google Scholar] [CrossRef]
- Cherry, J.A.; Davies, H.M.; Mehmood, S.; Lavery, N.P.; Brown, S.G.R.; Sienz, J. Investigation into the effect of process parameters on microstructural and physical properties of 316L stainless steel parts by selective laser melting. Int. J. Adv. Manuf. Technol. 2015, 76, 869–879. [Google Scholar] [CrossRef]
- Jia, Q.B.; Gu, D.D. Selective laser melting additive manufacturing of Inconel 718 superalloy parts: Densification, microstructure and properties. J. Alloys Compd. 2014, 585, 713–721. [Google Scholar] [CrossRef]
- Thijs, L.; Verhaeghe, F.; Craeghs, T.; Van Humbeeck, J.; Kruth, J.-P. A study of the microstructural evolution during selective laser melting of Ti–6Al–4V. Acta Mater. 2010, 58, 3303–3312. [Google Scholar] [CrossRef]
- Mukherjee, T.; Manvatkar, V.; De, A.; DebRoy, T. Dimensionless numbers in additive manufacturing. J. Appl. Phys. 2017, 121, 064904. [Google Scholar] [CrossRef]
- Amine, T.; Newkirk, J.W.; Liou, F. Investigation of effect of process parameters on multilayer builds by direct metal deposition. Appl. Therm. Eng. 2014, 73, 500–511. [Google Scholar] [CrossRef]
- Bardelcik, A.; Salisbury, C.P.; Winkler, S.; Wells, M.A.; Worswick, M.J. Effect of cooling rate on the high strain rate properties of boron steel. Int. J. Impact Eng. 2010, 37, 694–702. [Google Scholar] [CrossRef]
- Nishibata, T.; Kojima, N. Effect of quenching rate on hardness and microstructure of hot-stamped steel. J. Alloys Compd. 2013, 577, S549–S554. [Google Scholar] [CrossRef]
- Olasolo, M.; Uranga, P.; Rodriguez-Ibabe, J.; López, B. Effect of austenite microstructure and cooling rate on transformation characteristics in a low carbon Nb–V microalloyed steel. Mater. Sci. Eng. A 2011, 528, 2559–2569. [Google Scholar] [CrossRef]
- Thompson, S.; Col, D.V.; Krauss, G. Continuous cooling transformations and microstructures in a low-carbon, high-strength low-alloy plate steel. Metall. Trans. A 1990, 21, 1493–1507. [Google Scholar] [CrossRef]
- Thompson, S.; Colvin, D.; Krauss, G. Austenite decomposition during continuous cooling of an HSLA-80 plate steel. Metall. Mater. Trans. A 1996, 27, 1557–1571. [Google Scholar] [CrossRef]
- Qiao, Z.; Liu, Y.; Yu, L.; Gao, Z. Effect of cooling rate on microstructural formation and hardness of 30CrNi3Mo steel. Appl. Phys. A Mater. 2009, 95, 917–922. [Google Scholar] [CrossRef]
- Birol, Y. Optimization of homogenization for a low alloyed AlMgSi alloy. Mater. Charact. 2013, 80, 69–75. [Google Scholar] [CrossRef]
- Brito, C.; Costa, T.A.; Vida, T.A.; Bertelli, F.; Cheung, N.; Spinelli, J.E.; Garcia, A. Characterization of dendritic microstructure, intermetallic phases and hardness of directionally solidified Al-Mg and Al-Mg-Si alloys. Metall. Mater. Trans. A 2015, 46, 3342–3355. [Google Scholar] [CrossRef]
- Çadırlı, E. Effect of solidification parameters on mechanical properties of directionally solidified Al-rich Al-Cu alloys. Met. Mater.-Int. 2013, 19, 411–422. [Google Scholar] [CrossRef]
- Cavazos, J.L.; Colás, R. Precipitation in a heat-treatable aluminum alloy cooled at different rates. Mater. Charact. 2001, 47, 175–179. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, Y.; Ma, Z.; Shan, S.; Jia, Y.; Fan, C.; Wang, W. Effect of cooling rate on solidified microstructure and mechanical properties of aluminium-A356 alloy. J. Mater. Process. Technol. 2008, 207, 107–111. [Google Scholar] [CrossRef]
- Milenkovic, S.; Sabirov, I.; Llorca, J. Effect of the cooling rate on microstructure and hardness of MAR-M247 Ni-based superalloy. Mater. Lett. 2012, 73, 216–219. [Google Scholar] [CrossRef]
- Mitchell, R.; Preuss, M.; Tin, S.; Hardy, M. The influence of cooling rate from temperatures above the γ’ solvus on morphology, mismatch and hardness in advanced polycrystalline nickel-base superalloys. Mater. Sci. Eng. A 2008, 473, 158–165. [Google Scholar] [CrossRef]
- Geng, L.; Na, Y.-S.; Park, N.-K. Continuous cooling transformation behavior of alloy 718. Mater. Lett. 1997, 30, 401–405. [Google Scholar] [CrossRef]
- Rahimian, M.; Milenkovic, S.; Sabirov, I. Microstructure and hardness evolution in MAR-M247 Ni-based superalloy processed by controlled cooling and double heat treatment. J. Alloys Compd. 2013, 550, 339–344. [Google Scholar] [CrossRef]
- Karaköse, E.; Keskin, M. Effect of microstructural evolution and elevated temperature on the mechanical properties of Ni–Cr–Mo alloys. J. Alloys Compd. 2015, 619, 82–90. [Google Scholar] [CrossRef]
- Callister, W.D.; Rethwisch, D.G. Materials Science and Engineering; John Wiley & Sons: Hoboken, NJ, USA, 2011; Volume 5. [Google Scholar]
- Farshidianfar, M.H.; Khajepour, A.; Gerlich, A.P. Effect of real-time cooling rate on microstructure in laser additive manufacturing. J. Mater. Process. Technol. 2016, 231, 468–478. [Google Scholar] [CrossRef]
- Keist, J.S.; Palmer, T.A. Role of geometry on properties of additively manufactured Ti-6Al-4V structures fabricated using laser based directed energy deposition. Mater. Des. 2016, 106, 482–494. [Google Scholar] [CrossRef]
- Tian, Y.; McAllister, D.; Colijn, H.; Mills, M.; Farson, D.; Nordin, M.; Babu, S. Rationalization of microstructure heterogeneity in Inconel 718 builds made by the direct laser additive manufacturing process. Metall. Mater. Trans. A 2014, 45, 4470–4483. [Google Scholar] [CrossRef]
- Cormier, D.; Harrysson, A.; West, H. Characterization of H13 steel produced via electron beam melting. Rapid Prototyp. J. 2004, 10, 35–41. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Dinda, G.P.; Dasgupta, A.K.; Mazumder, J. Microstructural evolution of AISI 4340 steel during direct metal deposition process. Mater. Sci. Eng. A 2011, 528, 2309–2318. [Google Scholar] [CrossRef]
- Casalino, G.; Campanelli, S.L.; Contuzzi, N.; Ludovico, A.D. Experimental investigation and statistical optimisation of the selective laser melting process of a maraging steel. Opt. Laser Technol. 2015, 65, 151–158. [Google Scholar] [CrossRef]
- Choi, J.; Chang, Y. Characteristics of laser aided direct metal/material deposition process for tool steel. Int. J. Mach. Tools Manuf. 2005, 45, 597–607. [Google Scholar] [CrossRef]
- Cottam, R.; Wang, J.; Luzin, V. Characterization of microstructure and residual stress in a 3D H13 tool steel component produced by additive manufacturing. J. Mater. Res. 2014, 29, 1978–1986. [Google Scholar] [CrossRef]
- Haselhuhn, A.S.; Wijnen, B.; Anzalone, G.C.; Sanders, P.G.; Pearce, J.M. In situ formation of substrate release mechanisms for gas metal arc weld metal 3-D printing. J. Mater. Process. Technol. 2015, 226, 50–59. [Google Scholar] [CrossRef]
- Simchi, A.; Asgharzadeh, H. Densification and microstructural evaluation during laser sintering of M2 high speed steel powder. Mater. Sci. Technol. 2004, 20, 1462–1468. [Google Scholar] [CrossRef]
- Sander, J.; Hufenbach, J.; Giebeler, L.; Wendrock, H.; Kuhn, U.; Eckert, J. Microstructure and properties of FeCrMoVC tool steel produced by selective laser melting. Mater. Des. 2016, 89, 335–341. [Google Scholar] [CrossRef]
- Jamshidinia, M.; Sadek, A.; Wang, W.; Kelly, S. Additive manufacturing of steel alloys using laser powder-bed fusion. Adv. Mater. Process. 2015, 173, 20–24. [Google Scholar]
- Brice, C.; Shenoy, R.; Kral, M.; Buchannan, K. Precipitation behavior of aluminum alloy 2139 fabricated using additive manufacturing. Mater. Sci. Eng. A 2015, 648, 9–14. [Google Scholar] [CrossRef]
- Buchbinder, D.; Meiners, W.; Pirch, N.; Wissenbach, K.; Schrage, J. Investigation on reducing distortion by preheating during manufacture of aluminum components using selective laser melting. J. Laser Appl. 2014, 26, 012004. [Google Scholar] [CrossRef]
- Chou, R.; Milligan, J.; Paliwal, M.; Brochu, M. Additive manufacturing of Al-12Si alloy via pulsed selective laser melting. JOM 2015, 67, 590–596. [Google Scholar] [CrossRef]
- Wang, X.J.; Zhang, L.C.; Fang, M.H.; Sercombe, T.B. The effect of atmosphere on the structure and properties of a selective laser melted Al-12Si alloy. Mater. Sci. Eng. A 2014, 597, 370–375. [Google Scholar] [CrossRef]
- Maskery, I.; Aboulkhair, N.T.; Corfield, M.R.; Tuck, C.; Clare, A.T.; Leach, R.K.; Wildman, R.D.; Ashcroft, I.A.; Hague, R.J.M. Quantification and characterisation of porosity in selectively laser melted Al-Si10-Mg using X-ray computed tomography. Mater. Charact. 2016, 111, 193–204. [Google Scholar] [CrossRef]
- Kempen, K.; Thijs, L.; Van Humbeeck, J.; Kruth, J.-P. Mechanical properties of AlSi10Mg produced by selective laser melting. Phys. Procedia 2012, 39, 439–446. [Google Scholar] [CrossRef]
- Manfredi, D.; Calignano, F.; Krishnan, M.; Canali, R.; Ambrosio, E.P.; Atzeni, E. From powders to dense metal parts: Characterization of a commercial AlSiMg alloy processed through direct metal laser sintering. Materials 2013, 6, 856–869. [Google Scholar] [CrossRef] [PubMed]
- Olakanmi, E.O.; Cochrane, R.F.; Dalgarno, K.W. Densification mechanism and microstructural evolution in selective laser sintering of Al-12Si powders. J. Mater. Process. Technol. 2011, 211, 113–121. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, H.H.; Qi, T.; Hu, Z.H.; Zeng, X.Y. Selective laser melting of high strength Al-Cu-Mg alloys: Processing, microstructure and mechanical properties. Mater. Sci. Eng. A 2016, 656, 47–54. [Google Scholar] [CrossRef]
- Amato, K.N.; Gaytan, S.M.; Murr, L.E.; Martinez, E.; Shindo, P.W.; Hernandez, J.; Collins, S.; Medina, F. Microstructures and mechanical behavior of Inconel 718 fabricated by selective laser melting. Acta Mater. 2012, 60, 2229–2239. [Google Scholar] [CrossRef]
- Dinda, G.P.; Dasgupta, A.K.; Mazumder, J. Texture control during laser deposition of nickel-based superalloy. Scr. Mater. 2012, 67, 503–506. [Google Scholar] [CrossRef]
- Murr, L.E.; Martinez, E.; Pan, X.M.; Gaytan, S.M.; Castro, J.A.; Terrazas, C.A.; Medina, F.; Wicker, R.B.; Abbott, D.H. Microstructures of Rene 142 nickel-based superalloy fabricated by electron beam melting. Acta Mater. 2013, 61, 4289–4296. [Google Scholar] [CrossRef]
- Paul, C.P.; Jain, A.; Ganesh, P.; Negi, J.; Nath, A.K. Laser rapid manufacturing of Colmonoy-6 components. Opt. Laser Eng. 2006, 44, 1096–1109. [Google Scholar] [CrossRef]
- Paul, C.P.; Ganesh, P.; Mishra, S.K.; Bhargava, P.; Negi, J.; Nath, A.K. Investigating laser rapid manufacturing for Inconel-625 components. Opt. Laser Technol. 2007, 39, 800–805. [Google Scholar] [CrossRef]
- Sexton, L.; Lavin, S.; Byrne, G.; Kennedy, A. Laser cladding of aerospace materials. J. Mater. Process. Technol. 2002, 122, 63–68. [Google Scholar] [CrossRef]
- Vilaro, T.; Colin, C.; Bartout, J.D.; Nazé, L.; Sennour, M. Microstructural and mechanical approaches of the selective laser melting process applied to a nickel-base superalloy. Mater. Sci. Eng. A 2012, 534, 446–451. [Google Scholar] [CrossRef]
- Wang, Z.; Guan, K.; Gao, M.; Li, X.; Chen, X.; Zeng, X. The microstructure and mechanical properties of deposited-IN718 by selective laser melting. J. Alloys Compd. 2012, 513, 518–523. [Google Scholar] [CrossRef]
- Abe, F.; Osakada, K.; Shiomi, M.; Uematsu, K.; Matsumoto, M. The manufacturing of hard tools from metallic powders by selective laser melting. J. Mater. Process. Technol. 2001, 111, 210–213. [Google Scholar] [CrossRef]
- Zhong, M.; Yang, L.; Liu, W.; Huang, T.; He, J. Laser rapid manufacturing of special pattern Inco 718 nickel-based alloy component. In Proceedings of the SPIE 5629, Photonics Asia, Beijing, China, 13 January 2005; pp. 59–66. [Google Scholar]
- Kanagarajah, P.; Brenne, F.; Niendorf, T.; Maier, H.J. Inconel 939 processed by selective laser melting: Effect of microstructure and temperature on the mechanical properties under static and cyclic loading. Mater. Sci. Eng. A 2013, 588, 188–195. [Google Scholar] [CrossRef]
- Baufeld, B. Mechanical properties of Inconel 718 parts manufactured by shaped metal deposition (SMD). J. Mater. Eng. Perform 2012, 21, 1416–1421. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.M.; Tang, H.B. Effect of heat treatment on microstructure and mechanical properties of laser melting deposited Ni-base superalloy Rene’41. Mater. Sci. Eng. A 2012, 550, 97–102. [Google Scholar] [CrossRef]
- Acharya, R.; Bansal, R.; Gambone, J.J.; Kaplan, M.A.; Fuchs, G.E.; Rudawski, N.G.; Das, S. Additive manufacturing and characterization of René 80 superalloy processed through scanning laser epitaxy for turbine engine hot-section component repair. Adv. Eng. Mater. 2015, 17, 942–950. [Google Scholar] [CrossRef]
- Harrison, N.J.; Todd, I.; Mumtaz, K. Reduction of micro-cracking in nickel superalloys processed by selective laser melting: A fundamental alloy design approach. Acta Mater. 2015, 94, 59–68. [Google Scholar] [CrossRef]
- Wang, X.; Chou, Y.K. A method to estimate residual stress in metal parts made by selective laser melting. In Proceedings of the ASME 2015 International Mechanical Engineering Congress and Exposition, Houston, TX, USA, 13–19 November 2015. [Google Scholar]
- Zhang, K.; Liu, W.J.; Shang, X.F. Characteristics of laser aided direct metal powder deposition process for nickel-based superalloy. Mater. Sci. Forum 2007, 534–536, 457–460. [Google Scholar] [CrossRef]
- Sridharan, N.; Noakes, M.W.; Nycz, A.; Love, L.J.; Dehoff, R.R.; Babu, S.S. On the toughness scatter in low alloy C-Mn steel samples fabricated using wire arc additive manufacturing. Mater. Sci. Eng. A 2018, 713, 18–27. [Google Scholar] [CrossRef]
- Zuback, J.S.; Palmer, T.A.; DebRoy, T. Additive manufacturing of functionally graded transition joints between ferritic and austenitic alloys. J. Alloys Compd. 2019, 770, 995–1003. [Google Scholar] [CrossRef]
- Pinkerton, A.J.; Li, L. Direct additive laser manufacturing using gas-and water-atomised H13 tool steel powders. Int. J. Adv. Manuf. Technol. 2005, 25, 471–479. [Google Scholar] [CrossRef]
- Hentschel, O.; Scheitler, C.; Fedorov, A.; Junker, D.; Gorunov, A.; Haimerl, A.; Merklein, M.; Schmidt, M. Experimental investigations of processing the high carbon cold-work tool steel 1.2358 by laser metal deposition for the additive manufacturing of cold forging tools. J. Laser Appl. 2017, 29, 022307. [Google Scholar] [CrossRef]
- Mazur, M.; Brincat, P.; Leary, M.; Brandt, M. Numerical and experimental evaluation of a conformally cooled H13 steel injection mould manufactured with selective laser melting. Int. J. Adv. Manuf. Technol. 2017, 93, 881–900. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, C.; Qin, L.; Yan, K.; Cheng, G.; Jing, H.; Zou, T. Laser additive manufacturing of M2 high-speed steel. Mater. Sci. Technol. 2018, 34, 69–78. [Google Scholar] [CrossRef]
- Chen, H.; Gu, D.; Dai, D.; Ma, C.; Xia, M. Microstructure and composition homogeneity, tensile property and underlying thermal physical mechanism of selective laser melting tool steel parts. Mater. Sci. Eng. A 2017, 682, 279–289. [Google Scholar] [CrossRef]
- Sander, J.; Hufenbach, J.; Giebeler, L.; Bleckmann, M.; Eckert, J.; Kühn, U. Microstructure, mechanical behavior and wear properties of FeCrMoVC steel prepared by selective laser melting and casting. Scr. Mater. 2017, 126, 41–44. [Google Scholar] [CrossRef]
- Fayazfar, H.; Salarian, M.; Rogalsky, A.; Sarker, D.; Russo, P.; Paserin, V.; Toyserkani, E. A critical review of powder-based additive manufacturing of ferrous alloys: Process parameters, microstructure and mechanical properties. Mater. Des. 2018, 144, 98–128. [Google Scholar] [CrossRef]
- David, S.; Vitek, J.; Reed, R.; Hebble, T. Effect of Rapid Solidification on Stainless Steel Weld Metal Microstructures and Its Implications on the Schaeffler Diagram; Oak Ridge National Lab: Oak Ridge, TN, USA, 1987. [Google Scholar]
- Amine, T.; Newkirk, J.W.; Liou, F. An investigation of the effect of direct metal deposition parameters on the characteristics of the deposited layers. Case Stud. Therm. Eng. 2014, 3, 21–34. [Google Scholar] [CrossRef]
- Shah, K.; ul Haq, I.; Khan, A.; Shah, S.A.; Khan, M.; Pinkerton, A.J. Parametric study of development of Inconel-steel functionally graded materials by laser direct metal deposition. Mater. Des. 2014, 54, 531–538. [Google Scholar] [CrossRef]
- Rafi, H.K.; Pal, D.; Patil, N.; Starr, T.L.; Stucker, B.E. Microstructure and mechanical behavior of 17-4 precipitation hardenable steel processed by selective laser melting. J. Mater. Eng. Perform. 2014, 23, 4421–4428. [Google Scholar] [CrossRef]
- Murr, L.E.; Martinez, E.; Hernandez, J.; Collins, S.; Amato, K.N.; Gaytan, S.M.; Shindo, P.W. Microstructures and properties of 17-4 PH stainless steel fabricated by selective laser melting. J. Mater. Res. Technol. 2012, 1, 167–177. [Google Scholar] [CrossRef]
- Cheruvathur, S.; Lass, E.A.; Campbell, C.E. Additive manufacturing of 17-4 PH stainless steel: Post-processing heat treatment to achieve uniform reproducible microstructure. JOM 2016, 68, 930–942. [Google Scholar] [CrossRef]
- Thijs, L.; Kempen, K.; Kruth, J.P.; Van Humbeeck, J. Fine-structured aluminium products with controllable texture by selective laser melting of pre-alloyed AlSi10Mg powder. Acta Mater. 2013, 61, 1809–1819. [Google Scholar] [CrossRef]
- Dinda, G.P.; Dasgupta, A.K.; Bhattacharya, S.; Natu, H.; Dutta, B.; Mazumder, J. Microstructural characterization of laser-deposited Al 4047 alloy. Metall. Mater. Trans. A 2012, 44, 2233–2242. [Google Scholar] [CrossRef]
- Kaya, H.; Çadırlı, E.; Böyük, U.; Maraşlı, N. Variation of microindentation hardness with solidification and microstructure parameters in the Al based alloys. Appl. Surf. Sci. 2008, 255, 3071–3078. [Google Scholar] [CrossRef]
- Javidani, M.; Arreguin-Zavala, J.; Danovitch, J.; Tian, Y.; Brochu, M. Additive manufacturing of AlSi10Mg alloy using direct energy deposition: Microstructure and hardness characterization. J. Therm. Spray Technol. 2017, 26, 587–597. [Google Scholar] [CrossRef]
- Yan, C.; Hao, L.; Hussein, A.; Young, P.; Huang, J.; Zhu, W. Microstructure and mechanical properties of aluminium alloy cellular lattice structures manufactured by direct metal laser sintering. Mater. Sci. Eng. A 2015, 628, 238–246. [Google Scholar] [CrossRef]
- Aboulkhair, N.T.; Tuck, C.; Ashcroft, I.; Maskery, I.; Everitt, N.M. On the precipitation hardening of selective laser melted AlSi10Mg. Metall. Mater. Trans. A 2015, 46, 3337–3341. [Google Scholar] [CrossRef]
- Li, W.; Li, S.; Liu, J.; Zhang, A.; Zhou, Y.; Wei, Q.; Yan, C.; Shi, Y. Effect of heat treatment on AlSi10Mg alloy fabricated by selective laser melting: Microstructure evolution, mechanical properties and fracture mechanism. Mater. Sci. Eng. A 2016, 663, 116–125. [Google Scholar] [CrossRef]
- Kempen, K.; Thijs, L.; Van Humbeeck, J.; Kruth, J.-P. Processing AlSi10Mg by selective laser melting: Parameter optimisation and material characterisation. Mater. Sci. Technol. 2015, 31, 917–923. [Google Scholar] [CrossRef]
- Rosenthal, I.; Shneck, R.; Stern, A. Heat treatment effect on the mechanical properties and fracture mechanism in AlSi10Mg fabricated by additive manufacturing selective laser melting process. Mater. Sci. Eng. A 2018, 729, 310–322. [Google Scholar] [CrossRef]
- Tradowsky, U.; White, J.; Ward, R.; Read, N.; Reimers, W.; Attallah, M. Selective laser melting of AlSi10Mg: Influence of post-processing on the microstructural and tensile properties development. Mater. Des. 2016, 105, 212–222. [Google Scholar] [CrossRef]
- Feng, K.; Chen, Y.; Deng, P.; Li, Y.; Zhao, H.; Lu, F.; Li, R.; Huang, J.; Li, Z. Improved high-temperature hardness and wear resistance of Inconel 625 coatings fabricated by laser cladding. J. Mater. Process. Technol. 2017, 243, 82–91. [Google Scholar] [CrossRef]
- Foster, B.; Beese, A.; Keist, J.; McHale, E.; Palmer, T. Impact of interlayer dwell time on microstructure and mechanical properties of nickel and titanium alloys. Metall. Mater. Trans. A 2017, 48, 4411–4422. [Google Scholar] [CrossRef]
- Hussein, N.; Segal, J.; McCartney, D.; Pashby, I. Microstructure formation in Waspaloy multilayer builds following direct metal deposition with laser and wire. Mater. Sci. Eng. A 2008, 497, 260–269. [Google Scholar] [CrossRef]
- Jones, J.; Whittaker, M.; Buckingham, R.; Johnston, R.; Bache, M.; Clark, D. Microstructural characterisation of a nickel alloy processed via blown powder direct laser deposition (DLD). Mater. Des. 2017, 117, 47–57. [Google Scholar] [CrossRef]
- Khayat, Z.; Palmer, T. Impact of iron composition on the properties of an additively manufactured solid solution strengthened nickel base alloy. Mater. Sci. Eng. A 2018, 718, 123–134. [Google Scholar] [CrossRef]
- Zhang, F.; Levine, L.E.; Allen, A.J.; Stoudt, M.R.; Lindwall, G.; Lass, E.A.; Williams, M.E.; Idell, Y.; Campbell, C.E. Effect of heat treatment on the microstructural evolution of a nickel-based superalloy additive-manufactured by laser powder bed fusion. Acta Mater. 2018, 152, 200–214. [Google Scholar] [CrossRef]
- Floreen, S.; Fuchs, G.E.; Yang, W.J. The metallurgy of alloy 625. Superalloys 1994, 718, 13–37. [Google Scholar]
- Zhao, X.; Chen, J.; Lin, X.; Huang, W. Study on microstructure and mechanical properties of laser rapid forming Inconel 718. Mater. Sci. Eng. A 2008, 478, 119–124. [Google Scholar] [CrossRef]
- Zhong, C.; Gasser, A.; Kittel, J.; Wissenbach, K.; Poprawe, R. Improvement of material performance of Inconel 718 formed by high deposition-rate laser metal deposition. Mater. Des. 2016, 98, 128–134. [Google Scholar] [CrossRef]
- Stevens, E.L.; Toman, J.; To, A.C.; Chmielus, M. Variation of hardness, microstructure and Laves phase distribution in direct laser deposited alloy 718 cuboids. Mater. Des. 2017, 119, 188–198. [Google Scholar] [CrossRef]
- Tabernero, I.; Lamikiz, A.; Martínez, S.; Ukar, E.; Figueras, J. Evaluation of the mechanical properties of Inconel 718 components built by laser cladding. Int. J. Mach. Tools Manuf. 2011, 51, 465–470. [Google Scholar] [CrossRef]
- Tucho, W.M.; Cuvillier, P.; Sjolyst-Kverneland, A.; Hansen, V. Microstructure and hardness studies of Inconel 718 manufactured by selective laser melting before and after solution heat treatment. Mater. Sci. Eng. A 2017, 689, 220–232. [Google Scholar] [CrossRef]
- Zhang, D.; Niu, W.; Cao, X.; Liu, Z. Effect of standard heat treatment on the microstructure and mechanical properties of selective laser melting manufactured Inconel 718 superalloy. Mater. Sci. Eng. A 2015, 644, 32–40. [Google Scholar] [CrossRef]
- Tillmann, W.; Schaak, C.; Nellesen, J.; Schaper, M.; Aydinöz, M.; Hoyer, K.-P. Hot isostatic pressing of IN718 components manufactured by selective laser melting. Addit. Manuf. 2017, 13, 93–102. [Google Scholar] [CrossRef]
- Aydinöz, M.; Brenne, F.; Schaper, M.; Schaak, C.; Tillmann, W.; Nellesen, J.; Niendorf, T. On the microstructural and mechanical properties of post-treated additively manufactured Inconel 718 superalloy under quasi-static and cyclic loading. Mater. Sci. Eng. A 2016, 669, 246–258. [Google Scholar] [CrossRef]
- Popovich, V.; Borisov, E.; Popovich, A.; Sufiiarov, V.S.; Masaylo, D.; Alzina, L. Impact of heat treatment on mechanical behaviour of Inconel 718 processed with tailored microstructure by selective laser melting. Mater. Des. 2017, 131, 12–22. [Google Scholar] [CrossRef]
- Chlebus, E.; Gruber, K.; Kuźnicka, B.; Kurzac, J.; Kurzynowski, T. Effect of heat treatment on the microstructure and mechanical properties of Inconel 718 processed by selective laser melting. Mater. Sci. Eng. A 2015, 639, 647–655. [Google Scholar] [CrossRef]
- Deng, D.; Peng, R.L.; Brodin, H.; Moverare, J. Microstructure and mechanical properties of Inconel 718 produced by selective laser melting: Sample orientation dependence and effects of post heat treatments. Mater. Sci. Eng. A 2017, 713, 294–306. [Google Scholar] [CrossRef]
- Deng, D.; Moverare, J.; Peng, R.L.; Söderberg, H. Microstructure and anisotropic mechanical properties of EBM manufactured Inconel 718 and effects of post heat treatments. Mater. Sci. Eng. A 2017, 693, 151–163. [Google Scholar] [CrossRef]
- Deng, D.; Saarimäki, J.; Söderberg, H.; Peng, R.; Brodin, H.; Moverare, J. Microstructural characterization of as-manufactured and heat treated electron beam melted Inconel 718. In Proceedings of the Materials Science and Technology, Salt Lake City, UT, USA, 23–27 October 2016; pp. 105–112. [Google Scholar]
- Sames, W.J.; Unocic, K.A.; Helmreich, G.W.; Kirka, M.M.; Medina, F.; Dehoff, R.R.; Babu, S.S. Feasibility of in situ controlled heat treatment (ISHT) of Inconel 718 during electron beam melting additive manufacturing. Addit. Manuf. 2017, 13, 156–165. [Google Scholar] [CrossRef]
- Schwab, H.; Bönisch, M.; Giebeler, L.; Gustmann, T.; Eckert, J.; Kuehn, U. Processing of Ti-5553 with improved mechanical properties via an in-situ heat treatment combining selective laser melting and substrate plate heating. Mater. Des. 2017, 130, 83–89. [Google Scholar] [CrossRef]
- Dinda, G.; Song, L.; Mazumder, J. Fabrication of Ti-6Al-4V scaffolds by direct metal deposition. Metall. Mater. Trans. A 2008, 39, 2914–2922. [Google Scholar] [CrossRef]
- Kelly, S.; Kampe, S. Microstructural evolution in laser-deposited multilayer Ti-6Al-4V builds: Part I. Microstructural characterization. Metall. Mater. Trans. A 2004, 35, 1861–1867. [Google Scholar] [CrossRef]
- Hrabe, N.; Quinn, T. Effects of processing on microstructure and mechanical properties of a titanium alloy (Ti–6Al–4V) fabricated using electron beam melting (EBM), part 1: Distance from build plate and part size. Mater. Sci. Eng. A 2013, 573, 264–270. [Google Scholar] [CrossRef]
- Galarraga, H.; Warren, R.J.; Lados, D.A.; Dehoff, R.R.; Kirka, M.M.; Nandwana, P. Effects of heat treatments on microstructure and properties of Ti-6Al-4V ELI alloy fabricated by electron beam melting (EBM). Mater. Sci. Eng. A 2017, 685, 417–428. [Google Scholar] [CrossRef]
- Nassar, A.R.; Keist, J.S.; Reutzel, E.W.; Spurgeon, T.J. Intra-layer closed-loop control of build plan during directed energy additive manufacturing of Ti–6Al–4V. Addit. Manuf. 2015, 6, 39–52. [Google Scholar] [CrossRef]
- Wang, P.; Tan, X.; Nai, M.L.S.; Tor, S.B.; Wei, J. Spatial and geometrical-based characterization of microstructure and microhardness for an electron beam melted Ti–6Al–4V component. Mater. Des. 2016, 95, 287–295. [Google Scholar] [CrossRef]
- Jamshidinia, M.; Atabaki, M.M.; Zahiri, M.; Kelly, S.; Sadek, A.; Kovacevic, R. Microstructural modification of Ti–6Al–4V by using an in-situ printed heat sink in electron beam melting® (EBM). J. Mater. Process. Technol. 2015, 226, 264–271. [Google Scholar] [CrossRef]
- Hall, E. The deformation and ageing of mild steel: III discussion of results. Proc. Phys. Soc. Lond. B 1951, 64, 747–753. [Google Scholar] [CrossRef]
- Petch, N.J. The cleavage strengh of polycrystals. J. Iron Steel Inst. 1953, 174, 25–28. [Google Scholar]
- Petch, N. The fracture of metals. Prog. Metal Phys. 1954, 5, 1–52. [Google Scholar] [CrossRef]
- Tabor, D. The Hardness of Metals; Oxford University Press Inc.: New York, NY, USA, 2000. [Google Scholar]
- Cahoon, J.; Broughton, W.; Kutzak, A. The determination of yield strength from hardness measurements. Metall. Trans. 1971, 2, 1979–1983. [Google Scholar]
- Tan, X.; Kok, Y.; Tan, Y.J.; Descoins, M.; Mangelinck, D.; Tor, S.B.; Leong, K.F.; Chua, C.K. Graded microstructure and mechanical properties of additive manufactured Ti–6Al–4V via electron beam melting. Acta Mater. 2015, 97, 1–16. [Google Scholar] [CrossRef]
- Åkerfeldt, P.; Antti, M.-L.; Pederson, R. Influence of microstructure on mechanical properties of laser metal wire-deposited Ti-6Al-4V. Mater. Sci. Eng. A 2016, 674, 428–437. [Google Scholar] [CrossRef]
- Palanivel, S.; Dutt, A.; Faierson, E.; Mishra, R. Spatially dependent properties in a laser additive manufactured Ti–6Al–4V component. Mater. Sci. Eng. A 2016, 654, 39–52. [Google Scholar] [CrossRef]
- Ma, M.; Wang, Z.; Zeng, X. A comparison on metallurgical behaviors of 316L stainless steel by selective laser melting and laser cladding deposition. Mater. Sci. Eng. A 2017, 685, 265–273. [Google Scholar] [CrossRef]
- Zhong, Y.; Liu, L.; Wikman, S.; Cui, D.; Shen, Z. Intragranular cellular segregation network structure strengthening 316L stainless steel prepared by selective laser melting. J. Nucl. Mater. 2016, 470, 170–178. [Google Scholar] [CrossRef]
- Li, C.; Guo, Y.; Zhao, J. Interfacial phenomena and characteristics between the deposited material and substrate in selective laser melting Inconel 625. J. Mater. Process. Technol. 2017, 243, 269–281. [Google Scholar] [CrossRef]
- Griffith, M.L.; Ensz, M.T.; Puskar, J.D.; Robino, C.V.; Brooks, J.A.; Philliber, J.A.; Smugeresky, J.E.; Hofmeister, W. Understanding the microstructure and properties of components fabricated by laser engineered net shaping (LENS). MRS Proc. 2000, 625, 9. [Google Scholar] [CrossRef]
- Manfredi, D.; Ambrosio, E.; Calignano, F.; Krishnan, M.; Canali, R.; Biamino, S.; Pavese, M.; Atzeni, E.; Iuliano, L.; Fino, P. Direct metal laser sintering: An additive manufacturing technology ready to produce lightweight structural parts for robotic applications. Metall. Ital. 2013, 10, 15–24. [Google Scholar]
- Read, N.; Wang, W.; Essa, K.; Attallah, M.M. Selective laser melting of AlSi10Mg alloy: Process optimisation and mechanical properties development. Mater. Des. 2015, 65, 417–424. [Google Scholar] [CrossRef]
- Rosenthal, I.; Stern, A.; Frage, N. Strain rate sensitivity and fracture mechanism of AlSi10Mg parts produced by Selective Laser Melting. Mater. Sci. Eng. A 2017, 682, 509–517. [Google Scholar] [CrossRef]
- Rosenthal, I.; Stern, A.; Frage, N. Microstructure and mechanical properties of AlSi10Mg parts produced by the laser beam additive manufacturing (AM) technology. Metall. Microstruct. Anal. 2014, 3, 448–453. [Google Scholar] [CrossRef]
- Tang, M.; Pistorius, P.C. Oxides, porosity and fatigue performance of AlSi10Mg parts produced by selective laser melting. Int. J. Fatigue 2017, 94, 192–201. [Google Scholar] [CrossRef]
- Vilaro, T.; Colin, C.; Bartout, J.D. As-fabricated and heat-treated microstructures of the Ti-6Al-4V alloy processed by selective laser melting. Metall. Mater. Trans. A 2011, 42, 3190–3199. [Google Scholar] [CrossRef]
- Simonelli, M.; Tse, Y.Y.; Tuck, C. Effect of the build orientation on the mechanical properties and fracture modes of SLM Ti–6Al–4V. Mater. Sci. Eng. A 2014, 616, 1–11. [Google Scholar] [CrossRef]
- Zhao, X.; Li, S.; Zhang, M.; Liu, Y.; Sercombe, T.B.; Wang, S.; Hao, Y.; Yang, R.; Murr, L.E. Comparison of the microstructures and mechanical properties of Ti–6Al–4V fabricated by selective laser melting and electron beam melting. Mater. Des. 2016, 95, 21–31. [Google Scholar] [CrossRef]
- Edwards, P.; O’conner, A.; Ramulu, M. Electron beam additive manufacturing of titanium components: Properties and performance. J. Manuf. Sci. Eng. 2013, 135, 061016. [Google Scholar] [CrossRef]
- Zhai, Y.; Galarraga, H.; Lados, D.A. Microstructure evolution, tensile properties and fatigue damage mechanisms in Ti-6Al-4V alloys fabricated by two additive manufacturing techniques. Procedia Eng. 2015, 114, 658–666. [Google Scholar] [CrossRef]
- Qiu, C.; Ravi, G.; Dance, C.; Ranson, A.; Dilworth, S.; Attallah, M.M. Fabrication of large Ti–6Al–4V structures by direct laser deposition. J. Alloys Compd. 2015, 629, 351–361. [Google Scholar] [CrossRef]
- Alcisto, J.; Enriquez, A.; Garcia, H.; Hinkson, S.; Steelman, T.; Silverman, E.; Valdovino, P.; Gigerenzer, H.; Foyos, J.; Ogren, J. Tensile properties and microstructures of laser-formed Ti-6Al-4V. J. Mater. Eng. Perform. 2011, 20, 203–212. [Google Scholar] [CrossRef]
- Qiu, C.; Adkins, N.J.; Attallah, M.M. Microstructure and tensile properties of selectively laser-melted and of HIPed laser-melted Ti–6Al–4V. Mater. Sci. Eng. A 2013, 578, 230–239. [Google Scholar] [CrossRef]
- Rafi, H.; Karthik, N.; Gong, H.; Starr, T.L.; Stucker, B.E. Microstructures and mechanical properties of Ti6Al4V parts fabricated by selective laser melting and electron beam melting. J. Mater. Eng. Perform. 2013, 22, 3872–3883. [Google Scholar] [CrossRef]
- Kobryn, P.; Semiatin, S. Mechanical properties of laser-deposited Ti-6Al-4V. In Solid Freeform Fabrication Proceedings; The University of Texas: Austin, TX, USA, 2001; pp. 6–8. [Google Scholar]
- Amsterdam, E.; Kool, G. High cycle fatigue of laser beam deposited Ti-6Al-4V and Inconel 718. In ICAF 2009, Bridging the Gap between Theory and Operational Practice; Springer: Berlin, Germany, 2009; pp. 1261–1274. [Google Scholar]
- Brandl, E.; Leyens, C.; Palm, F. Mechanical properties of additive manufactured Ti-6Al-4V using wire and powder based processes. In Proceedings of the IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2011. [Google Scholar]
- Yao, B.; Ma, X.-L.; Lin, F.; Ge, W.-J. Microstructure and mechanical properties of Ti-6Al-4V components fabricated by laser micro cladding deposition. Rare Met. 2015, 34, 445–451. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Q.; Wang, H.; Liu, J.; Feng, J. Effect of location on microstructure and mechanical properties of additive layer manufactured Inconel 625 using gas tungsten arc welding. Mater. Sci. Eng. A 2016, 676, 395–405. [Google Scholar] [CrossRef]
- Amato, K.; Hernandez, J.; Murr, L.; Martinez, E.; Gaytan, S.; Shindo, P.; Collins, S. Comparison of microstructures and properties for a Ni-base superalloy (alloy 625) fabricated by electron beam melting. J Mater. Sci. Res. 2012, 1, 3. [Google Scholar] [CrossRef]
- Qiu, C.; Adkins, N.J.; Attallah, M.M. Selective laser melting of Invar 36: Microstructure and properties. Acta Mater. 2016, 103, 382–395. [Google Scholar] [CrossRef]
- Bauer, T.; Dawson, K.; Spierings, A.; Wegener, K. Microstructure and mechanical characterisation of SLM processed Haynes® 230®. In Proceedings of the 26th Annual International Solid Freeform Fabrication Symposium, Austin, TX, USA, 10–12 August 2015; pp. 813–822. [Google Scholar]
- Yadroitsev, I.; Thivillon, L.; Bertrand, P.; Smurov, I. Strategy of manufacturing components with designed internal structure by selective laser melting of metallic powder. Appl. Surf. Sci. 2007, 254, 980–983. [Google Scholar] [CrossRef]
- Blackwell, P. The mechanical and microstructural characteristics of laser-deposited IN718. J. Mater. Process. Technol. 2005, 170, 240–246. [Google Scholar] [CrossRef]
- Wang, Y.M.; Voisin, T.; McKeown, J.T.; Ye, J.; Calta, N.P.; Li, Z.; Zeng, Z.; Zhang, Y.; Chen, W.; Roehling, T.T. Additively manufactured hierarchical stainless steels with high strength and ductility. Nat. Mater. 2018, 17, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Parsa, A.B.; Ramsperger, M.; Kostka, A.; Somsen, C.; Körner, C.; Eggeler, G. Transmission electron microscopy of a CMSX-4 Ni-base superalloy produced by selective electron beam melting. Metals 2016, 6, 258. [Google Scholar] [CrossRef]
- Divya, V.; Muñoz-Moreno, R.; Messé, O.; Barnard, J.; Baker, S.; Illston, T.; Stone, H.J. Microstructure of selective laser melted CM247LC nickel-based superalloy and its evolution through heat treatment. Mater. Charact. 2016, 114, 62–74. [Google Scholar] [CrossRef]
- Akino, K.; Kakehi, K. Strengths and microstructure of SUS316L fabricated by selective laser melting. Mater. Trans. 2018, 59, 482–487. [Google Scholar] [CrossRef]
- Morrow, B.M.; Lienert, T.J.; Knapp, C.M.; Sutton, J.O.; Brand, M.J.; Pacheco, R.M.; Livescu, V.; Carpenter, J.S.; Gray, G.T. Impact of defects in powder feedstock materials on microstructure of 304L and 316L stainless steel produced by additive manufacturing. Metall. Mater. Trans. A 2018, 49, 3637–3650. [Google Scholar] [CrossRef]
- Qiu, Z.; Yao, C.; Feng, K.; Li, Z.; Chu, P.K. Cryogenic deformation mechanism of CrMnFeCoNi high-entropy alloy fabricated by laser additive manufacturing process. Int. J. Light. Mater. Manuf. 2018, 1, 33–39. [Google Scholar] [CrossRef]
- Yoo, Y.S.J.; Book, T.A.; Sangid, M.D.; Kacher, J. Identifying strain localization and dislocation processes in fatigued Inconel 718 manufactured from selective laser melting. Mater. Sci. Eng. A 2018, 724, 444–451. [Google Scholar] [CrossRef]
- Martinez, E.; Murr, L.E.; Hernandez, J.; Pan, X.; Amato, K.; Frigola, P.; Terrazas, C.; Gaytan, S.; Rodriguez, E.; Medina, F. Microstructures of niobium components fabricated by electron beam melting. Metall. Microstruct. Anal. 2013, 2, 183–189. [Google Scholar] [CrossRef]
- Hayes, B.J.; Martin, B.W.; Welk, B.; Kuhr, S.J.; Ales, T.K.; Brice, D.A.; Ghamarian, I.; Baker, A.H.; Haden, C.V.; Harlow, D.G. Predicting tensile properties of Ti-6Al-4V produced via directed energy deposition. Acta Mater. 2017, 133, 120–133. [Google Scholar] [CrossRef]
- Pokharel, R.; Balogh, L.; Brown, D.; Clausen, B.; Gray, G.; Livescu, V.; Vogel, S.; Takajo, S. Signatures of the unique microstructure of additively manufactured steel observed via diffraction. Scr. Mater. 2018, 155, 16–20. [Google Scholar] [CrossRef]
- Nandan, R.; DebRoy, T.; Bhadeshia, H.K.D.H. Recent advances in friction-stir welding—Process, weldment structure and properties. Prog. Mater. Sci. 2008, 53, 980–1023. [Google Scholar] [CrossRef]
- Lancaster, J.F. Metallurgy of Welding, 6th ed.; Elsevier: Cambridge, MA, USA; London, UK, 1999. [Google Scholar]
- Lienert, T.; Siewert, T.; Babu, S.; Acoff, V. ASM Handbook, Welding Fundamentals and Processes; ASM International: Materials Park, OH, USA, 2011; Volume 6A. [Google Scholar]
- Zhang, L.; Wen, M.; Imade, M.; Fukuyama, S.; Yokogawa, K. Effect of nickel equivalent on hydrogen gas embrittlement of austenitic stainless steels based on type 316 at low temperatures. Acta Mater. 2008, 56, 3414–3421. [Google Scholar] [CrossRef]
- Suzuki, H. Carbon equivalent and maximum hardness. Trans. Jpn Weld. Soc. 1984, 15, 25–33. [Google Scholar]
- Ito, Y.; Bessyo, K. Weldability Formula of High Strength Steels: Related to Heat-Affected Zone Cracking; International Institute of Welding: Villepinte, France, 1968; pp. 416–428. [Google Scholar]
- Lienert, T.; Stellwag, W., Jr.; Grimmett, B.; Warke, R. Friction stir welding studies on mild steel. Weld. J. 2003, 82, 1–9. [Google Scholar] [CrossRef]
- Nandan, R.; Roy, G.; Lienert, T.; Debroy, T. Three-dimensional heat and material flow during friction stir welding of mild steel. Acta Mater. 2007, 55, 883–895. [Google Scholar] [CrossRef]
- Cui, L.; Fujii, H.; Tsuji, N.; Nogi, K. Friction stir welding of a high carbon steel. Scr. Mater. 2007, 56, 637–640. [Google Scholar] [CrossRef]
- Khandkar, M.; Khan, J.A.; Reynolds, A.P. Prediction of temperature distribution and thermal history during friction stir welding: Input torque based model. Sci. Technol. Weld. Join. 2003, 8, 165–174. [Google Scholar] [CrossRef]
- Hofmann, D.C.; Vecchio, K.S. Thermal history analysis of friction stir processed and submerged friction stir processed aluminum. Mater. Sci. Eng. A 2007, 465, 165–175. [Google Scholar] [CrossRef]
- Kasuya, T.; Yurioka, N. Prediction of welding thermal history by a comprehensive solution. Weld J. 1993, 72, 107–115. [Google Scholar]
- DebRoy, T.; David, S.A. Physical processes in fusion welding. Rev. Mod. Phys. 1995, 67, 85–112. [Google Scholar] [CrossRef]
- Kenel, C.; Grolimund, D.; Li, X.; Panepucci, E.; Samson, V.A.; Sanchez, D.F.; Marone, F.; Leinenbach, C. In situ investigation of phase transformations in Ti-6Al-4V under additive manufacturing conditions combining laser melting and high-speed micro-X-ray diffraction. Sci. Rep. 2017, 7, 16358. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Zhou, Y.; Smugeresky, J.E.; Schoenung, J.M.; Lavernia, E.J. Thermal Behavior and Microstructural Evolution during Laser Deposition with Laser-Engineered Net Shaping: Part I. Numerical Calculations. Metall. Mater. Trans. A 2008, 39, 2228–2236. [Google Scholar] [CrossRef]
- Hooper, P.A. Melt pool temperature and cooling rates in laser powder bed fusion. Addit. Manuf. 2018, 22, 548–559. [Google Scholar] [CrossRef]
- Arora, A.; DebRoy, T.; Bhadeshia, H. Back-of-the-envelope calculations in friction stir welding–velocities, peak temperature, torque and hardness. Acta Mater. 2011, 59, 2020–2028. [Google Scholar] [CrossRef]









| Alloy | * Process | † Power (W) | Scanning Speed (mm/s) | Ref. | |
|---|---|---|---|---|---|
| Iron Alloys | H13 | EBM | (20 mA) | 500 | [91] |
| 4340 | DMD | 500 | 7.5 | [92] | |
| 18Ni300 | SLM | 86–100 | 180–220 | [93] | |
| H13 | DMD | 1000–1400 | 10.5–19.0 | [94] | |
| H13 | DMD | 2500 | 5 | [95] | |
| ER70S-6 | 3D GMAW | (14–16 V, 65–76A) | 5.23 | [96] | |
| M2 steel | DMLS | 200 | 50–175 | [97] | |
| Tool steel | SLM | 75–175 | 300-450 | [98] | |
| 420 SS | DMLS | 283–317 | 600–1000 | [99] | |
| Aluminum Alloys | Al 2139 | EBF3 | 1350 | 8.47 | [100] |
| AlSi10Mg | SLM | 195 | 800 | [101] | |
| Al-12Si | Pulsed SLM | 500–4500 | 1.5–3.0 | [102] | |
| Al-12Si | SLM | 200 | 37–2000 | [103] | |
| AlSi10Mg | SLM | 200 | 318 | [104] | |
| AlSi10Mg | SLM | 200 | Not reported | [105] | |
| AlSi10Mg | DMLS | 120 | 900 | [106] | |
| Al-12Si | SLS | 100–200 | 80–200 | [107] | |
| Al 2024 | SLM | 200 | 83–333 | [108] | |
| Nickel Alloys | IN718 | SLM | 200 | 800–1200 | [109] |
| IN718 | DMD | 750 | 6.25 | [110] | |
| IN718 | SLM | 110–130 | 400–600 | [67] | |
| Rene 142 | EBM | Not reported | Not reported | [111] | |
| Colmonoy 6 | LRM | 2500 | 4.2 | [112] | |
| IN625 | LRM | 1000–1500 | 5.0–13 | [113] | |
| Rene 142 | LC | 550 | Not reported | [114] | |
| Nimonic 263 | SLM | 200 | 100 | [115] | |
| IN718 | SLM | 170 | 417 | [116] | |
| Experimental | SLM | 1000 | 2 | [117] | |
| IN718 | SLM | 3000 | Not reported | [118] | |
| IN939 | SLM | 400 | 540–620 | [119] | |
| IN718 | SMD | (220A) | 5 | [120] | |
| Rene 41 | LMD | 4500–5000 | 5.00–5.83 | [121] | |
| Rene 80 | SLE | 1000 | ~100 | [122] | |
| Hastelloy X | SLM | 165–195 | Not reported | [123] | |
| Hastelloy X | SLM | Not reported | Not reported | [123] | |
| IN718 | SLM | Not reported | Not reported | [124] | |
| Ni60A | LMDS | Not reported | Not reported | [125] | |
| Alloy | Process | Phases | HV | Ref. | |
|---|---|---|---|---|---|
| Iron alloys | H13 | EBM | Martensite | ‡ 498.5 ± 14.5 | [91] |
| 4340 | DMD | Ferrite, Martensite, Cementite | 580.5 ± 100.5 | [92] | |
| 18Ni300 Maraging steel | SLM | Not reported | ‡ 323.5 ± 21.5 | [93] | |
| H13 | DMD | Not reported | 550 ± 30 | [94] | |
| H13 | DMD | Fine martensite, retained austenite, fine carbides | 615 ± 35 | [95] | |
| ER70S-6 | 3D-GMAW | Polygonal ferrite, acicular ferrite | 184 ± 15 | [96] | |
| M2 steel | DMLS | Martensite, austenite, fine carbides | 800 ± 100 | [97] | |
| FeCrMoVCtool steel | SLM | Fine martensite, austenite, carbides | 900 ± 12 | [98] | |
| 420 SS | DMLS | Martensite, little retained austenite | ‡ 478 ± 20 | [99] | |
| Aluminum alloys | Al 2139 | EBF3 | Not reported | 81–103 | [100] |
| AlSi10Mg | SLM | FCC dendrites, interdendritic eutectic | 131–141 | [101] | |
| Al-12Si | Pulsed SLM | FCC dendrites, small eutectic phases | 130–140 | [102] | |
| Al-12Si | SLM | FCC Al matrix, nanosized Si precipitates | 107–115 | [103] | |
| AlSi10Mg | SLM | FCC | 114 | [104] | |
| AlSi10Mg | SLM | FCC, fine Si precipitates | 127 | [105] | |
| AlSi10Mg | DMLS | FCC | 103–111 | [106] | |
| Al-12Si | SLS | FCC Al-Si matrix | 99–113 | [107] | |
| Al 2024 | SLM | FCC | 104–118 | [108] | |
| Nickel alloys | IN718 | SLM | FCC-γ, ellipsoidal Ni3Nb precipitates | 387–398 | [109] |
| IN718 | DMD | FCC-γ | 255 | [110] | |
| IN718 | SLM | FCC-γ, fine γ’ precipitates | 331.9–395.8 | [67] | |
| Rene 142 | EBM | FCC-γ, cuboidal γ’ precipitates | 428.1 | [111] | |
| Colmonoy 6 | LRM | FCC-γ, interdendritic eutectic | 700–800 | [112] | |
| IN625 | LRM | FCC-γ | ‡ 474.9–574.1 | [113] | |
| Rene 142 | LC | Not reported | 410–460 | [114] | |
| Nimonic 263 | SLM | γ, carbides in interdendritic regions | 300 | [115] | |
| IN718 | SLM | FCC-γ | 340–380 | [116] | |
| Experimental | SLM | Not reported | 650–800 | [117] | |
| IN718 | SLM | FCC-γ | 400–450 | [118] | |
| IN939 | SLM | FCC-γ | 450 | [119] | |
| IN718 | SMD | FCC-γ, interdendritic carbides/Laves | 245–287 | [120] | |
| Rene 41 | LMD | FCC-γ, MC carbides | 418.1–435.1 | [121] | |
| Rene 80 | SLE | FCC-γ, fine carbides/γ’ particles | 489.8 | [122] | |
| Hastelloy X | SLM | FCC-γ | 276.9–284.9 | [123] | |
| Hastelloy X | SLM | FCC-γ | 273.2–281.0 | [123] | |
| IN718 | SLM | FCC-γ | 410.8–430.2 | [124] | |
| Ni60A | LMDS | FCC-γ | ‡ 631.1–762.9 | [125] | |
| Alloy | Process | Dislocation Density (m−2) | Reference |
|---|---|---|---|
| SS316L | PBF-L | 1.5 × 1014 | [219] |
| SS316L | PBF-L + Solutionized | 9.7 × 1013 | [219] |
| SS316L | Hot worked + Solutionized | 3.5 × 1013 | [219] |
| SS316L | DED-L | 2.77 × 1014 | [220] |
| SS304L | DED-L | 4.31 to 7.45 × 1012 | [220] |
| SS304L | PBF-EB | 2.72 × 1014 | [220] |
| SS304L | Wrought | 1.84 × 1014 | [220] |
| CrMnFeCoNi | DED-L | 0.89 to 1.19 × 1014 | [221] |
| IN718 | PBF-L | 2.00 × 1013 to 5.62 × 1015 | [222] |
| Nb | PBF-EB | 1013 to 1014 | [223] |
| Ti-6Al-4V | DED-EB + HIP | 1011 | [224] |
| Ti-6Al-4V | DED-EB + Stress relieved | 1015 | [224] |
| SS304L | PBF-L | (3.8 ± 1) × 1014 | [225] |
| SS304L | DED-L | (2.5 ± 1) × 1014 | [225] |
| SS304L | Wrought (deformed) | (6.8 ± 1) × 1014 | [225] |
| Alloy | C | Co | Cr | Cu | Mn | Mo | Ni | P | Si | Ti | V | W | Pcm | Average HV | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H13 | 0.37 | - | 4.99 | - | 0.2 | 1.1 | - | 0.011 | 1.02 | - | 0.8 | - | 0.817 | ‡ 498.5 ± 14.5 | [91] |
| 4340 | 0.42 | - | 0.9 | - | 0.74 | 0.45 | 2.63 | - | 0.29 | - | - | - | 0.586 | 580.5 ± 100.5 | [92] |
| 18Ni300 Maraging steel | 0.02 | 10.2 | - | - | - | 4.2 | 18.8 | - | - | 0.88 | - | - | 0.613 | ‡ 323.5 ± 21.5 | [93] |
| H13 | 0.47 | - | 5.01 | - | 0.2 | 1.2 | - | - | 0.63 | - | 1.12 | - | 0.944 | 550 ± 30 | [94] |
| H13 | 0.35 | - | 5 | - | 0.35 | 1.5 | - | - | - | - | 1 | - | 0.818 | 615 ± 35 | [95] |
| ER70S-6 | 0.1 | - | 0.15 | - | 1.62 | 0.15 | 0.15 | 0.025 | 1 | - | 0.03 | - | 0.237 | 184 ± 15 | [96] |
| M2 steel | 0.86 | - | 1.25 | - | 0.37 | 5.23 | - | - | 0.33 | - | - | 6.32 | 1.301 | 800 ± 100 | [97] |
| FeCrMoVCtool steel | 0.99 | - | 4.02 | - | - | 7.97 | - | - | - | - | 2.01 | - | 1.923 | 900 ± 12 | [98] |
| 420 SS | 0.42 | - | 13.3 | - | 0.33 | 0.06 | 0.37 | - | 0.54 | - | - | - | 1.130 | ‡ 478 ± 20 | [99] |
| Alloy | Ag | Cu | Fe | Mg | Mn | Si | Ti | Zn | HV | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Al 2139 | 0.5 | 5.3 | 0.08 | 0.52 | 0.31 | 0.051 | 0.064 | - | 81–103 | [100] |
| AlSi10Mg | - | - | 0.55 | 0.4 | 0.45 | 10 | - | 0.1 | 131–141 | [101] |
| Al-12Si | - | 0.3 | 0.8 | 0.1 | 0.15 | 12 | - | 0.2 | 130–140 | [102] |
| Al-12Si | - | 0.003 | 0.12 | - | - | 12.2 | - | - | 107–115 | [103] |
| AlSi10Mg | - | 0.05 | 0.25 | 0.4 | 0.1 | 10 | 0.1 | 0.1 | 114 | [104] |
| AlSi10Mg | - | 0.1 | 0.55 | 0.4 | 0.45 | 10 | - | 0.1 | 127 | [105] |
| AlSi10Mg | - | 0.001 | 0.16 | 0.35 | 0.002 | 10.08 | 0.01 | 0.002 | 103–111 | [106] |
| Al-12Si | - | 0.08 | 0.36 | - | - | 12.1 | - | - | 99–113 | [107] |
| Al 2024 | - | 4.47 | - | 1.95 | 0.55 | - | - | - | 104–118 | [108] |
| Alloy | Al | Co | Cr | Fe | Mo | Nb | Si | Ti | C | Other | HV | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IN718 | 0.5 | 1.0 | 19.0 | 22.0 | 3.0 | 5.0 | - | 1.0 | - | - | 387–398 | [109] |
| IN718 | 0.5 | - | 19.0 | 22.0 | 3.0 | 5.0 | - | 1.0 | - | - | 255 | [110] |
| IN718 | 0.3 | - | 18.4 | 17.7 | 4.2 | 5.1 | - | 0.9 | 0.08 | - | 331.9–395.8 | [67] |
| Rene 142 | 6.15 | 12.0 | 6.8 | - | 1.5 | 5.1 | - | 0.9 | 0.12 | 0.02B-1.5Hf-6.35Ta-4.9W | 428.1 | [111] |
| Colmonoy 6 | - | 0.24 | 13.6 | 4.75 | - | - | 4.25 | - | 0.6 | 2.5B | 700–800 | [112] |
| IN625 | 0.4 | 1.0 | 21.3 | 5.0 | 9.2 | 1.8 | 0.5 | 0.4 | 0.1 | 1.8Ta | ‡ 474.9–574.1 | [113] |
| Rene 142 | 3.0 | 9.5 | 14.0 | 0.1 | 3.8 | 0.03 | 0.01 | 5.0 | 0.14 | 0.02B-0.01Hf-0.01Mn-0.01Ta | 410–460 | [114] |
| Nimonic 263 | 0.5 | 19.2 | 19.5 | 0.5 | 6.0 | - | 0.2 | 2.4 | - | - | 300 | [115] |
| IN718 | 0.29 | - | 18.2 | 18.9 | 3.1 | 5.1 | - | 0.9 | 0.03 | - | 340–380 | [116] |
| Experimental | - | - | 9.4 | 2.0 | - | - | 2.8 | - | 0.4 | 1.8B | 650–800 | [117] |
| IN718 | 0.41 | - | 15.9 | 17.1 | 1.9 | 2.23 | - | 1.27 | - | 0.31W | 400–450 | [118] |
| IN939 | 1.9 | 19.0 | 22.4 | - | - | 1.0 | - | 3.7 | 0.15 | 0.01B-1.4Ta-2.0W | 450 | [119] |
| IN718 | - | - | 19.0 | 24.7 | 3.0 | - | 0.35 | - | 0.08 | 0.35Mn | 245–287 | [120] |
| Rene 41 | 1.6 | 11.0 | 19.0 | 5.0 | 9.75 | - | 0.5 | 3.25 | 0.09 | 0.01B-0.5Mn | 418.1–435.1 | [121] |
| Rene 80 | 3.0 | 9.0 | 14.0 | - | 4.0 | - | - | 4.7 | 0.16 | 0.02B-0.8Hf | 489.8 | [122] |
| Hastelloy X | - | 1.77 | 21.8 | 18.6 | 9.4 | - | 0.31 | - | 0.05 | 0.22Mn-1.05W | 276.9–284.9 | [123] |
| Hastelloy X | - | 1.04 | 21.3 | 19.5 | 9.0 | - | 0.32 | - | 0.06 | 0.48Mn-0.56W | 273.2–281.0 | [123] |
| IN718 | - | - | 18.3 | 18.9 | 2.0 | 4.6 | - | 0.83 | - | - | 410.8–430.2 | [124] |
| Ni60A | - | - | 16.5 | 8.0 | - | - | 4.25 | - | 0.75 | 3.75B | ‡ 631.1–762.9 | [125] |
| Process | Values/Ranges (K/s) | Reference |
|---|---|---|
| FSW | ~5 | [232] |
| FSW | ~3 to 5 | [233] |
| FSW | ~90 to 120 | [234] |
| FSW | ~10 | [235] |
| Submerged FSW | ~20 | [236] |
| Fusion welding | ~5 | [237] |
| Laser welding | 100 to 105 | [238] |
| Fusion welding | ~103 | [227] |
| DED-L | 4500 | [239] |
| PBF-L | 103 to 104 | [197] |
| DED-L | 104 | [240] |
| DED-L | ~103–104 | [9] |
| PBF-L | 1.0 × 106 to 4.0 × 107 | [241] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuback, J.S.; DebRoy, T. The Hardness of Additively Manufactured Alloys. Materials 2018, 11, 2070. https://doi.org/10.3390/ma11112070
Zuback JS, DebRoy T. The Hardness of Additively Manufactured Alloys. Materials. 2018; 11(11):2070. https://doi.org/10.3390/ma11112070
Chicago/Turabian StyleZuback, J.S., and T. DebRoy. 2018. "The Hardness of Additively Manufactured Alloys" Materials 11, no. 11: 2070. https://doi.org/10.3390/ma11112070
APA StyleZuback, J. S., & DebRoy, T. (2018). The Hardness of Additively Manufactured Alloys. Materials, 11(11), 2070. https://doi.org/10.3390/ma11112070




