Synthesis and Characterization of WO3/Graphene Nanocomposites for Enhanced Photocatalytic Activities by One-Step In-Situ Hydrothermal Reaction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
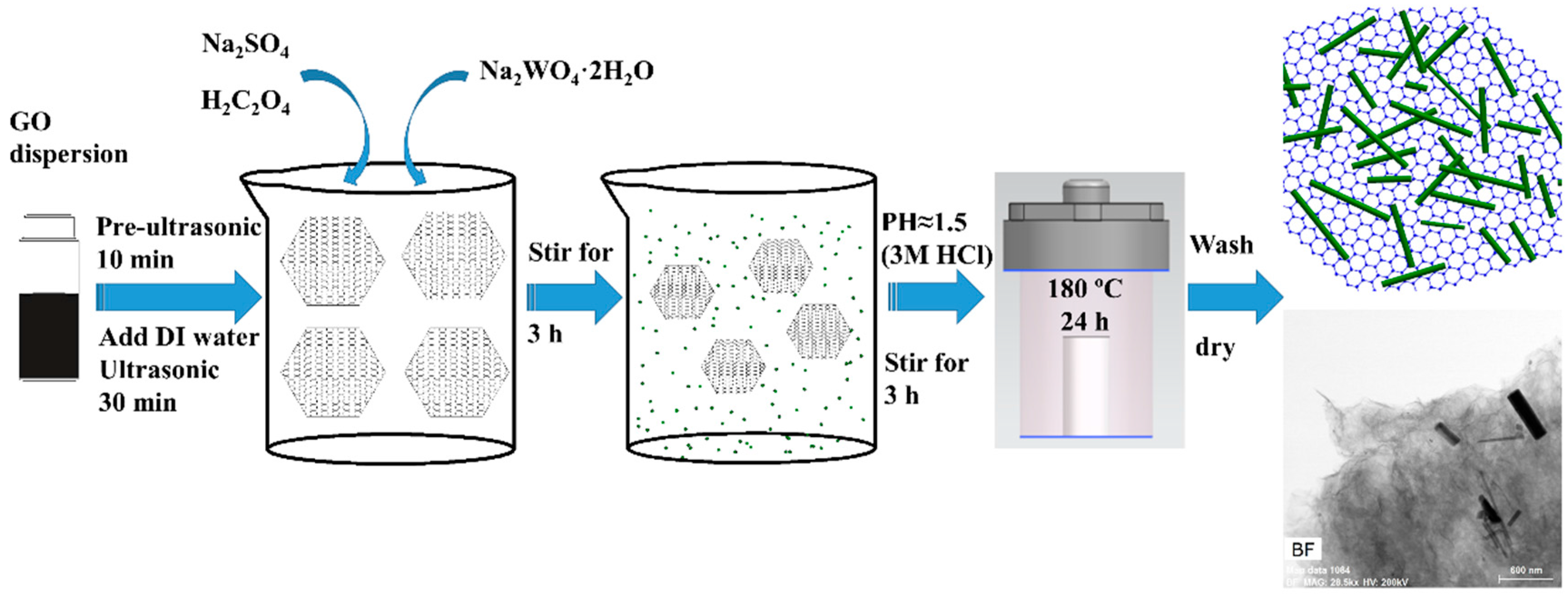
2.2. Synthesis of WO3/GR Nanocomposites
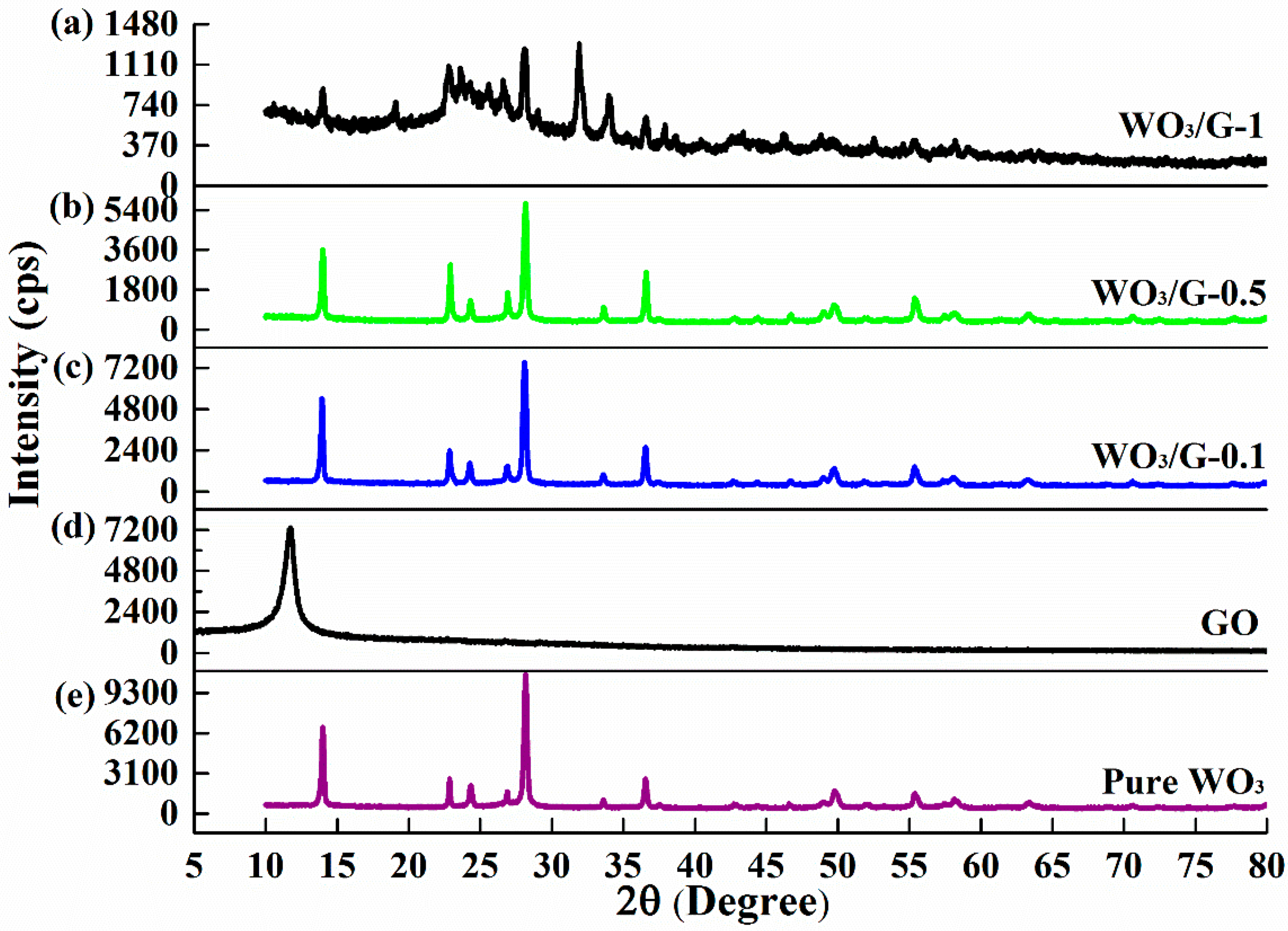
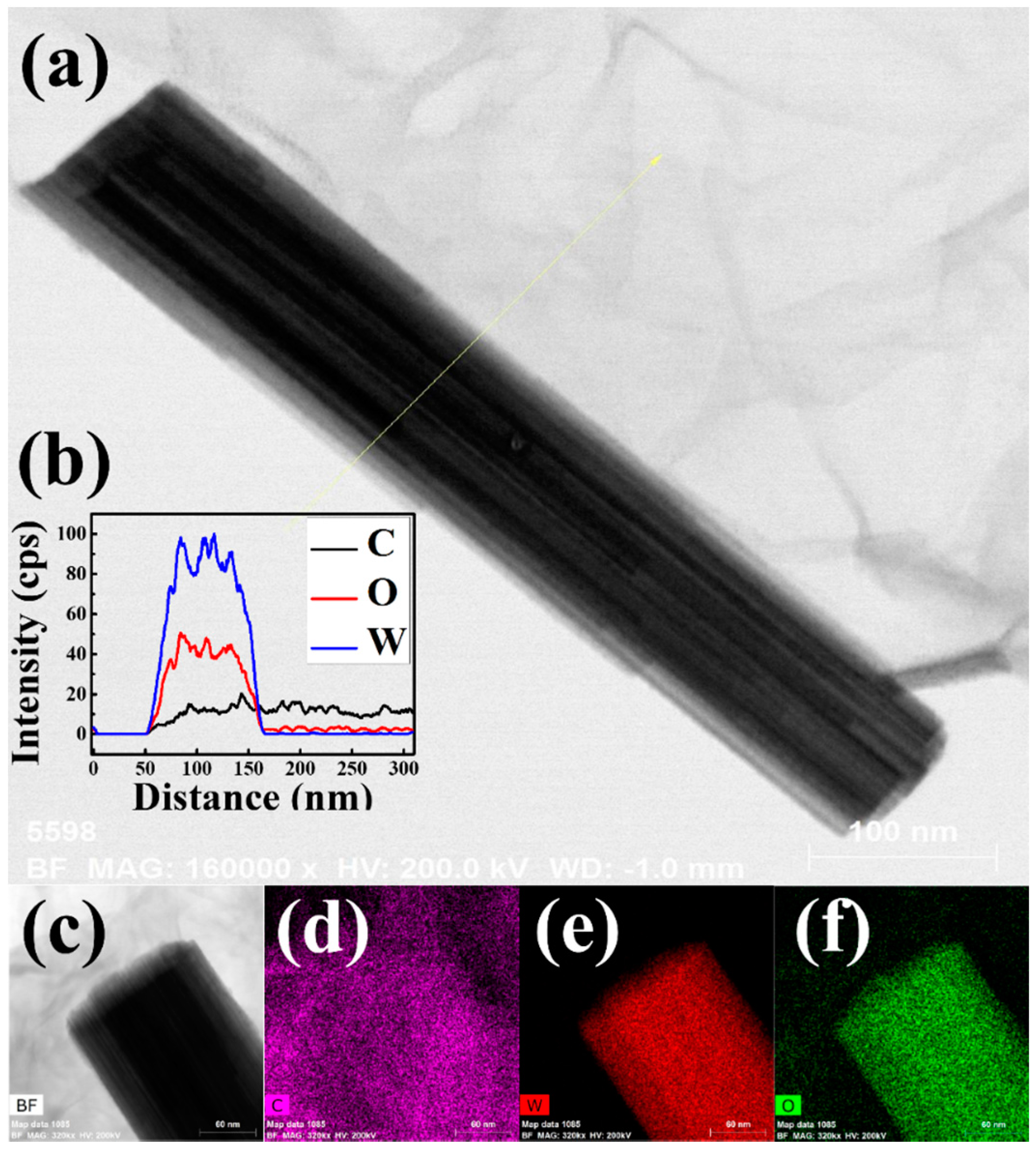
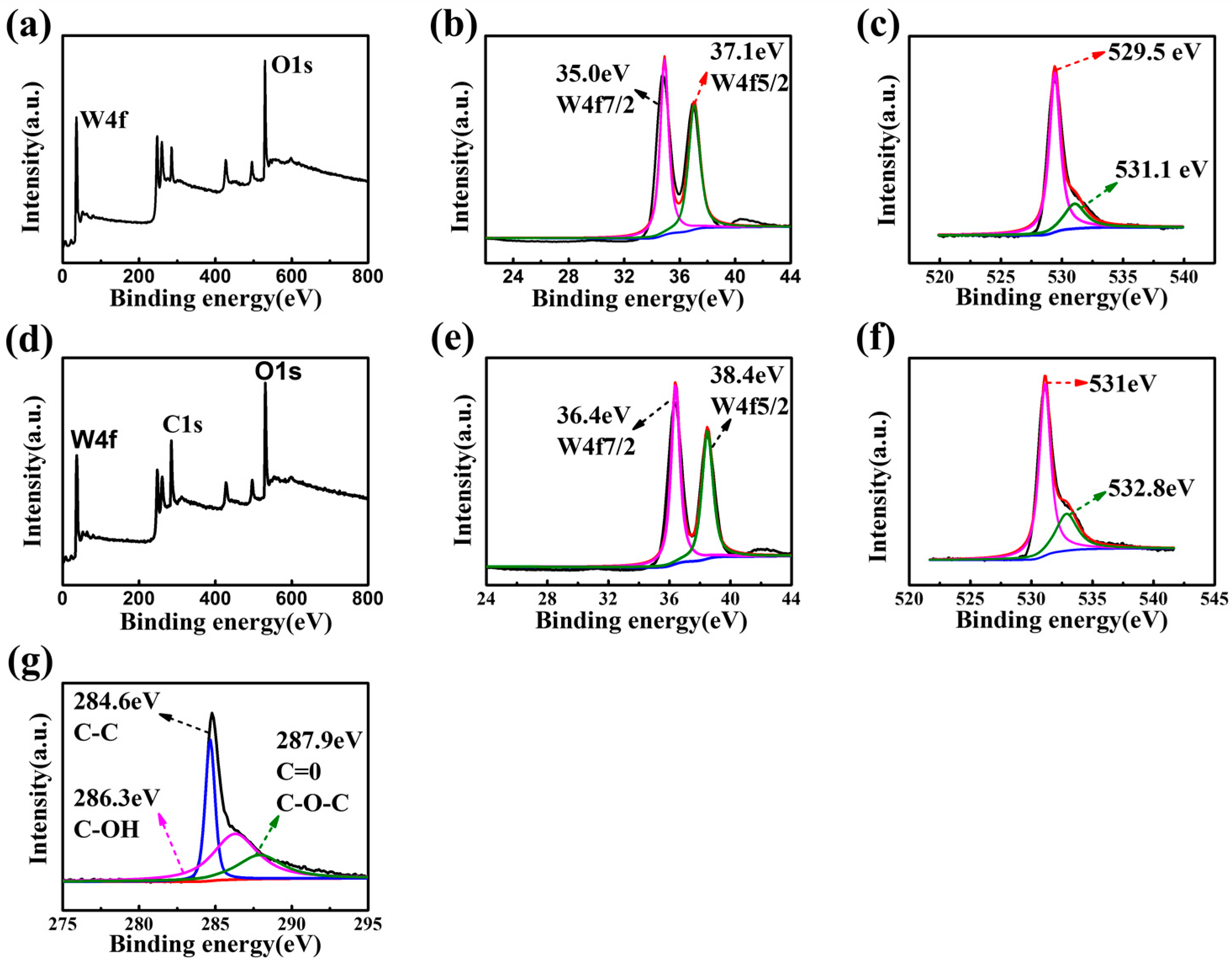
2.3. Characterizations of Materials
2.4. Evaluation of Photocatalytic Activity
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef] [PubMed]
- Lightcap, I.V.; Kosel, T.H.; Kamat, P.V. Anchoring semiconductor and metal nanoparticles on a two-dimensional catalyst mat. storing and shuttling electrons with reduced graphene oxide. Nano Lett. 2010, 10, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Wang, T.T.; Sun, H.G.; Shao, Q.; Zhao, J.; Song, K.; Hao, L.; Wang, L.; Guo, Z. Two-step hydrothermally synthesized carbon nanodots/WO3 photocatalysts with enhanced photocatalytic performance. Dalton Trans. 2017, 46, 15769–15777. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Fang, Y.; Wang, Y.; Sun, S.; He, J.; Yan, Z. Synthesis of Cu2O/graphene/rutile TiO2 nanorod ternary composites with enhanced photocatalytic activity. J. Alloys Compd. 2015, 650, 520–527. [Google Scholar] [CrossRef]
- Arutanti, O.; Nandiyanto, A.B.D.; Ogi, T.; Iskandar, F.; Kim, T.O.; Okuyama, K. Synthesis of composite WO3/TiO2 nanoparticles by flame-assisted spray pyrolysis and their photocatalytic activity. J. Alloys Compd. 2014, 591, 121–126. [Google Scholar] [CrossRef]
- Ramos-Delgado, N.A.; Hinojosa-Reyes, L.; Guzman-Mar, I.L.; Gracia-Pinilla, M.A.; Hernández-Ramírez, A. Synthesis by sol-gel of WO3/TiO2 for solar photocatalytic degradation of Malathion pesticide. Catal. Today 2013, 209, 35–40. [Google Scholar] [CrossRef]
- Huang, K.; Pan, Q.; Yang, F.; Ni, S.; Wei, X.; He, D. Controllable synthesis of hexagonal WO3 nanostructures and their application in lithium batteries. J. Phys. D Appl. Phys. 2008, 41, 155417. [Google Scholar] [CrossRef]
- Gui, Y.; Zhao, J.; Wang, H.; Tian, J.; Zhang, H. Microwave-assisted gas–liquid interfacial synthesis of WO3 precursor and nano-WO3 for gas-sensing application. J. Mater. Sci. Mater. Electron. 2016, 155, 4–7. [Google Scholar] [CrossRef]
- Shi, J.; Cheng, Z.; Gao, L.; Zhang, Y.; Xu, J.; Zhao, H. Facile synthesis of reduced graphene oxide/hexagonal WO3 nanosheets composites with enhanced H2S sensing properties. Sens. Actuators B Chem. 2016, 230, 736–745. [Google Scholar] [CrossRef]
- An, X.; Yu, J.C.; Wang, Y.; Hu, Y.; Yu, X.; Zhang, G. WO3 nanorods/graphene nanocomposites for high-efficiency visible-light-driven photocatalysis and NO2 gas sensing. J. Mater. Chem. 2012, 22, 8525–8531. [Google Scholar] [CrossRef]
- Chu, X.; Hu, T.; Gao, F.; Dong, Y.; Sun, W.; Bai, L. Gas sensing properties of graphene-WO3 composites prepared by hydrothermal method. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2015, 193, 97–104. [Google Scholar] [CrossRef]
- Gu, X.; Wu, F.; Lei, B.; Wang, J.; Chen, Z.; Xie, K.; Song, Y.; Sun, D.; Sun, L.; Zhou, H.; et al. Three-dimensional nitrogen-doped graphene frameworks anchored with bamboo-like tungsten oxide nanorods as high performance anode materials for lithium ion batteries. J. Power Sources 2016, 320, 231–238. [Google Scholar] [CrossRef]
- Yu, M.; Sun, H.; Sun, X.; Lu, F.; Hu, T.; Wang, G.; Qiu, H.; Lian, J. 3D WO3 nanowires/graphene nanocomposite with improved reversible capacity and cyclic stability for lithium ion batteries. Mater. Lett. 2013, 108, 29–32. [Google Scholar] [CrossRef]
- Ma, Y.; Zhao, M.; Cai, B.; Wang, W.; Ye, Z.; Huang, J. 3D graphene network@WO3 nanowire composites: A multifunctional colorimetric and electrochemical biosensing platform. Chem. Commun. 2014, 50, 11135–11138. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Li, Y.; Zhu, S.; Chen, Z.; Liu, Q.; Zhang, D.; Moon, W.J.; Song, D.M. Synthesis of WO3@Graphene composite for enhanced photocatalytic oxygen evolution from water. RSC Adv. 2012, 2, 1356–1363. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, X.; Shen, Y.; Han, J.; Yuan, L.; Gong, L.; Xu, Z.; Bai, X.; Wei, M.; Tong, Y.; et al. Three-dimensional WO3 nanostructures on carbon paper: Photoelectrochemical property and visible light driven photocatalysis. Chem. Commun. 2011, 47, 5804–5806. [Google Scholar] [CrossRef] [PubMed]
- Vamvasakis, I.; Georgaki, I.; Vernardou, D.; Kenanakis, G.; Katsarakis, N. Synthesis of WO3 catalytic powders: Evaluation of photocatalytic activity under NUV/visible light irradiation and alkaline reaction pH. J. Sol-Gel Sci. Technol. 2015, 76, 120–128. [Google Scholar] [CrossRef]
- Dong, P.; Yang, B.; Liu, C.; Xu, F.; Xi, X.; Hou, G.; Shao, R. Highly enhanced photocatalytic activity of WO3 thin films loaded with Pt-Ag bimetallic alloy nanoparticles. RSC Adv. 2017, 7, 947–956. [Google Scholar] [CrossRef]
- Nayak, A.K.; Das, A.K.; Pradhan, D. High Performance Solid-State Asymmetric Supercapacitor using Green Synthesized Graphene-WO3 Nanowires Nanocomposite. ACS Sustain. Chem. Eng. 2017, 5, 10128–10138. [Google Scholar] [CrossRef]
- Ma, B.; Huang, E.; Wu, G.; Dai, W.; Guan, N.; Li, L. Fabrication of WO2.72/RGO nano-composites for enhanced photocatalysis. RSC Adv. 2017, 7, 2606–2614. [Google Scholar] [CrossRef]
- Geng, X.; You, J.; Wang, J.; Zhang, C. Visible light assisted nitrogen dioxide sensing using tungsten oxide-Graphene oxide nanocomposite sensors. Mater. Chem. Phys. 2017, 191, 114–120. [Google Scholar] [CrossRef]
- Thiyagarajan, K.; Muralidharan, M.; Sivakumar, K. Defects Induced Magnetism in WO3 and Reduced Graphene Oxide-WO3 Nanocomposites. J. Supercond. Novel Magn. 2018, 31, 117–125. [Google Scholar] [CrossRef]
- Wong, C.P.P.; Lee, K.M.; Lal, C.W. Hydrothermal preparation of reduced graphene oxide/tungsten trioxide nanocomposites with enhanced electrochemical performance. J. Mater. Sci. Mater. Electron. 2017, 28, 14554–14567. [Google Scholar] [CrossRef]
- Khan, M.E.; Khan, M.M.; Cho, M.H. Fabrication of WO3 nanorods on graphene nanosheets for improved visible light-induced photocapacitive and photocatalytic performance. RSC Adv. 2016, 6, 20824–20833. [Google Scholar] [CrossRef]
- Won, J.M.; Son, M.Y.; Seo, J.H.; Kang, Y.C. Electrochemical properties of WO3-reduced graphene oxide composite powders prepared by one-pot spray pyrolysis process. J. Alloys Compd. 2016, 688, 647–653. [Google Scholar] [CrossRef]
- Liu, X.; Jin, A.; Jia, Y.; Xia, T.; Deng, C.; Zhu, M.; Chen, C.; Chen, X. Synergy of adsorption and visible-light photocatalytic degradation of methylene blue by a bifunctional Z-scheme heterojunction of WO3/g-C3N4. Appl. Surface Sci. 2017, 405, 359–371. [Google Scholar] [CrossRef]
- Dinari, M.; Momeni, M.M.; Ahangarpour, M. Efficient degradation of methylene blue dye over tungsten trioxide/multi-walled carbon nanotube system as a novel photocatalyst. Appl. Phys. A 2016, 122, 876. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.; Seger, B.; Kamat, P.V. TiO2-graphene nanocomposites. UV-assisted photocatalytic reduction of graphene oxide. ACS Nano 2008, 2, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Zhao, X.; Yang, J.; Shan, X.; Yang, L.; Zhang, Y.; Li, X.; Gao, M. ZnO-graphene composite for photocatalytic degradation of methylene blue dye. Catal. Commun. 2012, 29, 29–34. [Google Scholar] [CrossRef]
- Jie, X.; Zeng, D.; Zhang, J.; Xu, K.; Wu, J.; Zhu, B.; Xie, C. Graphene-wrapped WO3 nanospheres with room-temperature NO2 sensing induced by interface charge transfer. Sens. Actuators B Chem. 2015, 220, 201–209. [Google Scholar] [CrossRef]
- Wu, H.; Xu, M.; Da, P.; Li, W.; Jia, D.; Zheng, G. WO3-reduced graphene oxide composites with enhanced charge transfer for photoelectrochemical conversion. Phys. Chem. Chem. Phys. 2013, 15, 16138–16142. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Demadrille, F.R.; Grévin, B.; Jang, B.-H.; Lee, S.J.; Lee, J.H.; Tuller, H.L.; Kim, I. Fast Responding Exhaled-Breath Sensors Using WO3 Hemitubes Functionalized by Graphene-Based Electronic Sensitizers for Diagnosis of Diseases. ACS Appl. Mater. Interfaces 2014, 6, 9061–9070. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Da, P.; Zhang, Y.; Wang, Y.; Lin, X.; Gong, X.; Zheng, G. WO3 Nanoflakes for Enhanced Photoelectrochemical Conversion. ACS Nano 2014, 8, 11770–11777. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.; Foo, C.; Lee, P.S. One-step facile electrochemical preparation of WO3/graphene nanocomposites with improved electrochromic properties. Electrochim. Acta 2014, 117, 139–144. [Google Scholar] [CrossRef]
- Kovtyukhova, N.I.; Ollivier, P.J.; Martin, B.R.; Mallouk, T.E.; Buzaneva, S.A.C.E.V.; Gorchinskiy, A.D. Layer-by-layer assembly of ultrathin composite films from micron-sized graphite oxide sheets and polycations. Chem. Mater. 1999, 11, 771–778. [Google Scholar] [CrossRef]
- Weng, B.; Wu, J.; Zhang, N.; Xu, Y. Observing the role of graphene in boosting the two-electron reduction of oxygen in graphene-WO3 nanorod photocatalysts. Langmuir 2014, 30, 5574–5584. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wei, X.; Wang, Y.; Luo, F. One-pot twelve tungsten phosphate acid assisted electrochemical synthesis of WO3-decorated graphene sheets for high-efficiency UV-light-driven photocatalysis. Chem. Phys. Lett. 2014, 607, 34–38. [Google Scholar] [CrossRef]
- Zhou, M.; Yan, J.; Cui, P. Synthesis and enhanced photocatalytic performance of WO3 nanorods@graphene nanocomposites. Mater. Lett. 2012, 89, 258–261. [Google Scholar] [CrossRef]
- Gui, Y.; Yuan, J.; Wang, W.; Zhao, J.; Tian, J.; Xie, B. Facile solvothermal synthesis and gas sensitivity of graphene/WO3 nanocomposites. Materials 2014, 7, 4587–4600. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Bai, Y.; Luo, P.; Liu, J. Graphene-WO3 nanobelt composite: Elevated conduction band toward photocatalytic reduction of CO2 into hydrocarbon fuels. Catal. Commun. 2013, 38, 82–85. [Google Scholar] [CrossRef]
- Azimirad, R.; Safa, S. Preparation of three dimensional graphene foam WO3 nanocomposite with enhanced visible light photocatalytic activity. Mater. Chem. Phys. 2015, 162, 686–691. [Google Scholar] [CrossRef]
- Prabhu, S.; Manikumar, S.; Cindrella, L.; Kwon, O.J. Charge transfer and intrinsic electronic properties of rGO-WO3 nanostructures for efficient photoelectrochemical and photocatalytic applications. Mater. Sci. Semicond. Process. 2018, 74, 136–146. [Google Scholar] [CrossRef]
- Ahmed, B.; Ojha, A.K.; Hirsch, F.; Fischer, I.; Patrice, D.; Materny, A. Tailoring of enhanced interfacial polarization in WO3 nanorods grown over reduced graphene oxide synthesized by a one-step hydrothermal method. RSC Adv. 2017, 7, 985–996. [Google Scholar] [CrossRef]
- Bissessur, R.; Liu, P.K.Y.; Scully, S.F. Intercalation of polypyrrole into graphite oxide. Synth. Met. 2006, 156, 1023–1027. [Google Scholar] [CrossRef]
- Shang, S.; Gan, L.; Yuen, C.W.M.; Jiang, S.; Luo, N.M. The synthesis of graphene nanoribbon and its reinforcing effect on poly (vinyl alcohol). Compos. Part A 2015, 68, 149–154. [Google Scholar] [CrossRef]
- Cao, N.; Zhang, Y. Study of reduced graphene oxide preparation by hummers method and related characterization. J. Nanomater. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Zeng, F.; Ren, Y.; Chen, L.; Yang, Y.; Li, Q.; Gu, G. Hierarchical sandwich-type tungsten trioxide nanoplatelets/graphene anode for high-performance lithium-ion batteries with long cycle life. Electrochim. Acta 2016, 190, 964–971. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [PubMed]
- Park, S.K.; Lee, H.J.; Lee, M.H.; Park, H.S. Hierarchically structured reduced graphene oxide/WO3 frameworks for an application into lithium ion battery anodes. Chem. Eng. J. 2015, 281, 724–729. [Google Scholar] [CrossRef]
- Tuninstra, F.; Koenic, J.L. Raman Spectrum of Graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef]
- Akhavan, O.; Choobtashani, M.; Ghaderi, E. Protein Degradation and RNA Efflux of Viruses Photocatalyzed by Graphene-Tungsten Oxide Composite under Visible Light Irradiation. J. Phys. Chem. C 2012, 116, 9653–9659. [Google Scholar] [CrossRef]
- Zhang, S.; Li, H.; Yang, Z. Controllable synthesis of WO3 with different crystalline phases and its applications on methylene blue removal from aqueous solution. J. Alloys Compd. 2017, 722, 555–563. [Google Scholar] [CrossRef]
- Umukoro, E.H.; Peleyeju, M.G.; Ngila, J.C.; Arotiba, O.A. Towards wastewater treatment: Photo-assisted electrochemical degradation of 2-nitrophenol and orangeII dye at a tungsten trioxide-exfoliated graphite composite electrode. Chem. Eng. J. 2017, 317, 290–301. [Google Scholar] [CrossRef]
- Sun, M.; Li, W.; Sun, S.; He, J.; Zhang, Q.; Shi, Y. One-step in situ synthesis of grapheme-TiO2 nanorod hybrid composites with enhanced photocatalytic activity. Mater. Res. Bull. 2015, 61, 280–286. [Google Scholar] [CrossRef]




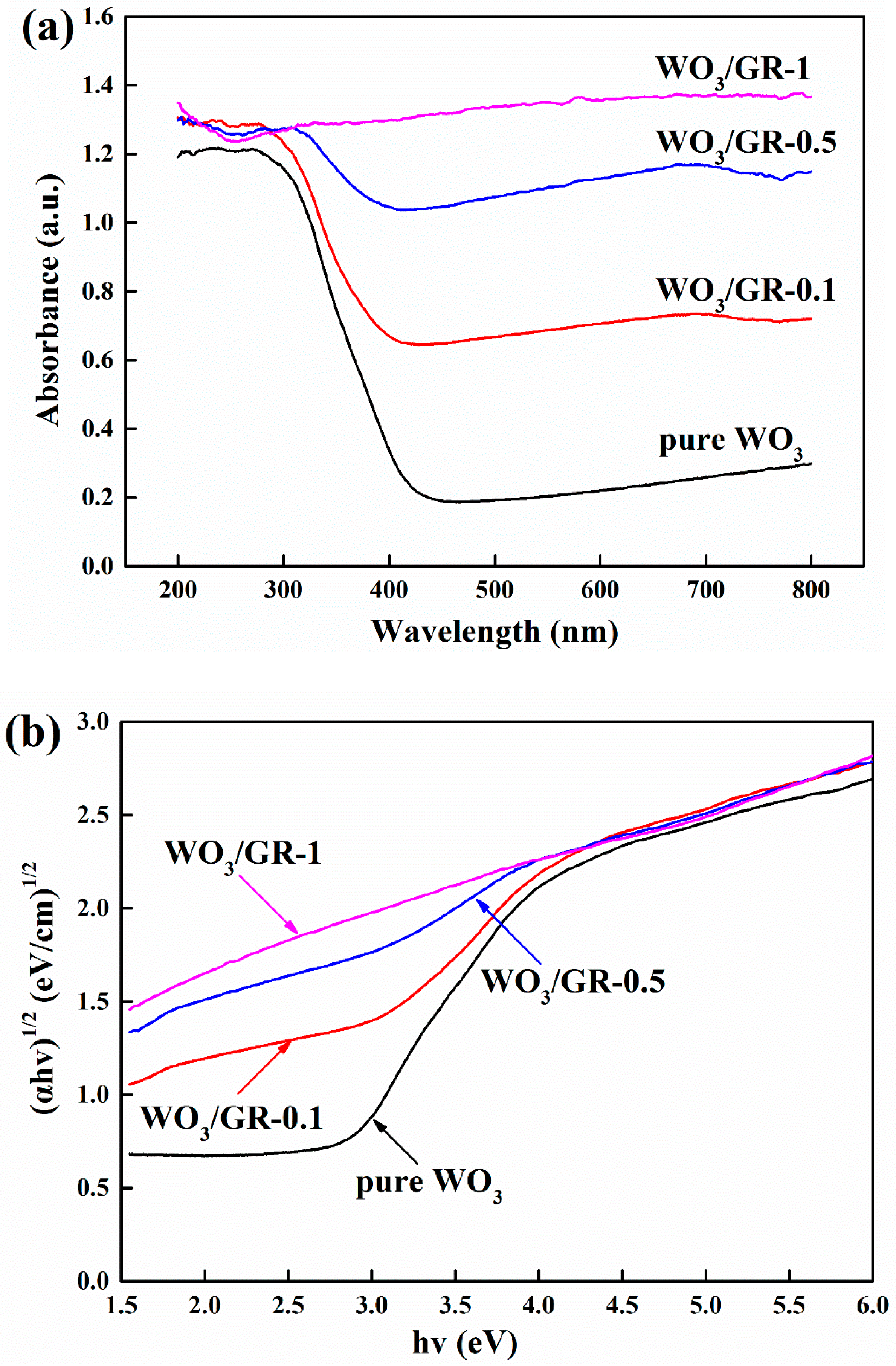
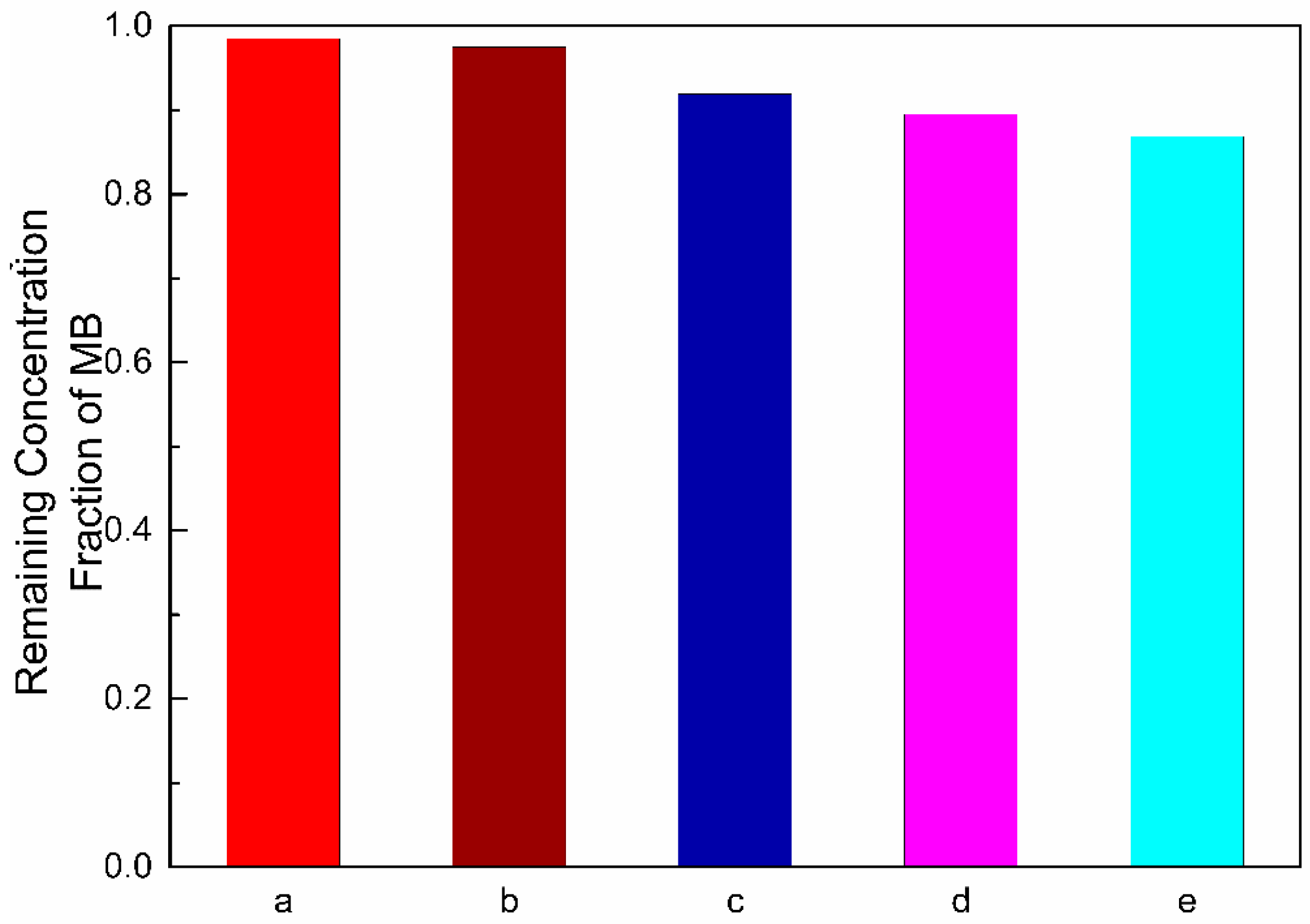
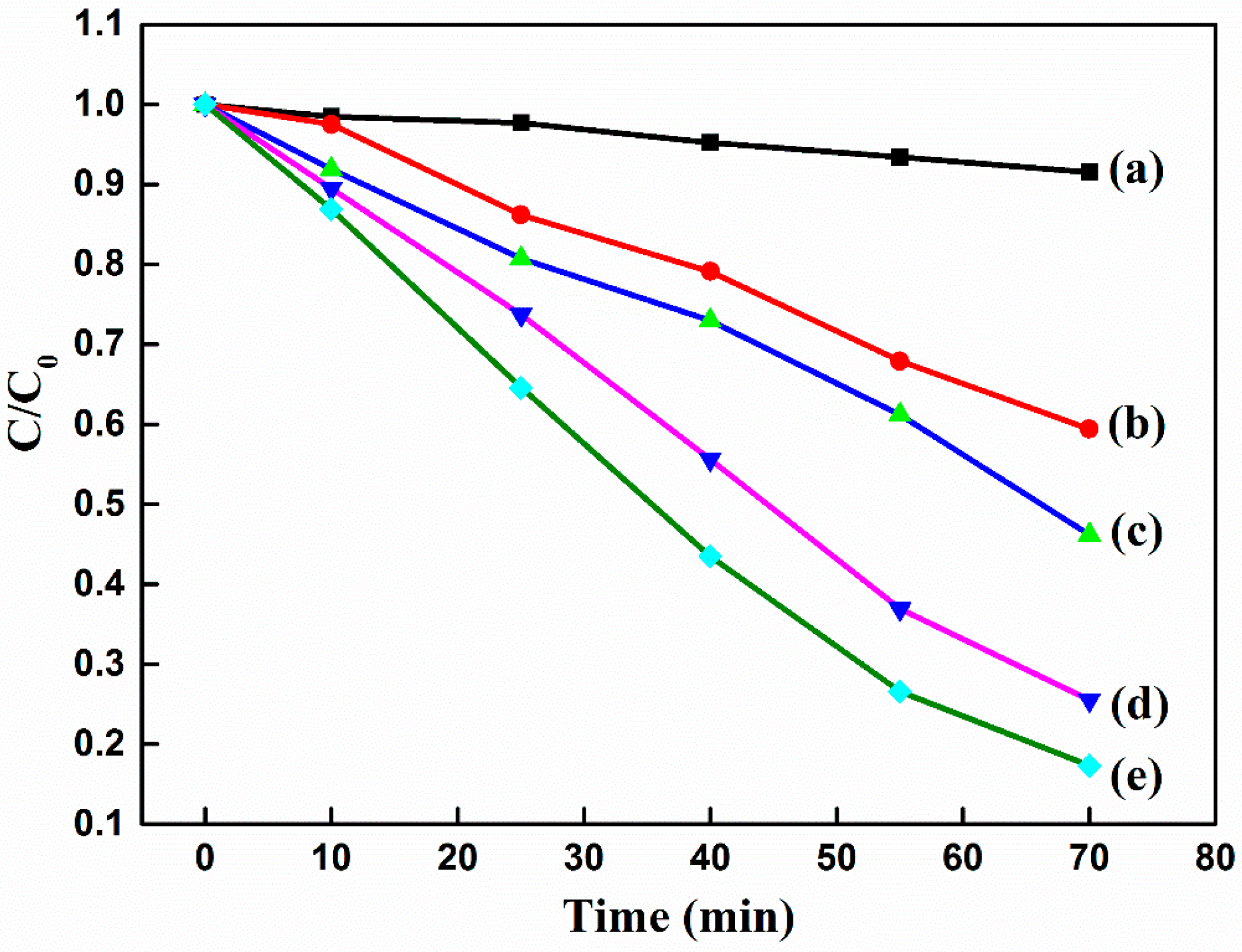
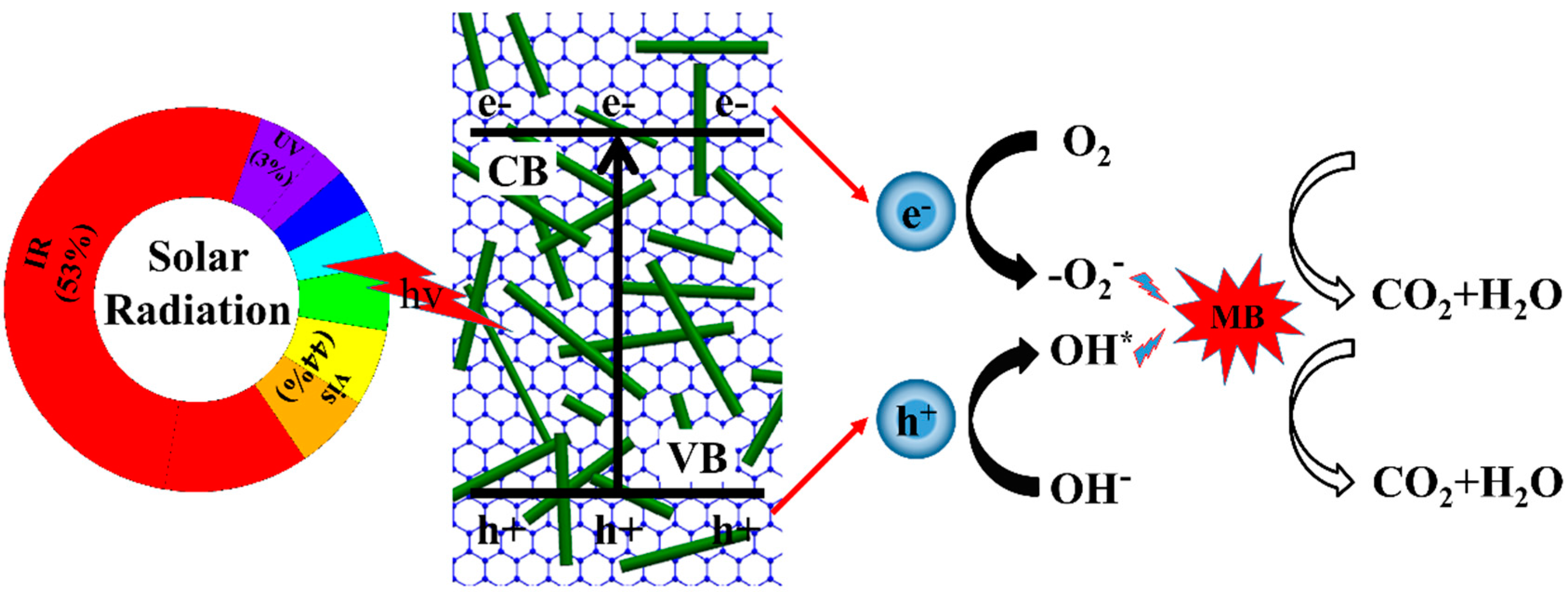
| Photocatalyst | Methods of Synthesis | Experimental Conditions | Photodecomposition | Photocatalytic Efficiency | References |
|---|---|---|---|---|---|
| WO3/GR | one-step in-situ hydrothermal reaction | Precursor: Na2WO4·2H2O and GO the structure-directing agent: H2C2O4 and Na2SO4 Time: 24 h Temp:180 °C | Degradation of methylene blue | Degradation percentage of 83% within 70 min | This work |
| 3D graphene foam-WO3 | chemical vapor deposition(CVD) (two steps) | Precursor: the nickel foam, CH4, tungsten wire Time: >54 h Temp: - | degradation of Rhodamine B dye | Degradation percentage of 85% within 140 min | [42] |
| rGO-WO3 | wet chemistry and thermal decomposition method | Precursor: GO and H2WO4 | Degradation of methylene blue | Degradation percentage of 65% within 60 min | [43] |
| WO3/g-C3N4 | in-situ liquid phase process and heat treatment | Precursor: ammonium metatungstate hydrate and g-C3N4 the structure-directing agent: oxalic acid-ethanol solution | Degradation of methylene blue | Degradation percentage of 95% within 90 min | [26] |
| WO3/NWCNT | Solvothermal process | Precursor: acid-functionalized MWCNTs and Na2WO4·2H2O the structure-directing agent: HNO3 | Degradation of methylene blue | Degradation percentage of 75% within 150 min | [27] |
| Graphene/TiO2 | hydrothermal method | Precursor: GO and TiO2 (P25) Time: 24 h Temp:200 °C | Degradation of methylene blue | Degradation percentage of 85% within 100 min | [55] |
| ZnO-graphene | hydrothermal method | Precursor: GO and the commercial ZnO nanoparticles Time: 12 h Temp:200 °C | Degradation of methylene blue | Degradation percentage of 72.1% within 56 min | [30] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.; Xu, P.; Gong, H.; Yin, G. Synthesis and Characterization of WO3/Graphene Nanocomposites for Enhanced Photocatalytic Activities by One-Step In-Situ Hydrothermal Reaction. Materials 2018, 11, 147. https://doi.org/10.3390/ma11010147
Hu X, Xu P, Gong H, Yin G. Synthesis and Characterization of WO3/Graphene Nanocomposites for Enhanced Photocatalytic Activities by One-Step In-Situ Hydrothermal Reaction. Materials. 2018; 11(1):147. https://doi.org/10.3390/ma11010147
Chicago/Turabian StyleHu, Xiaoxiao, Peiquan Xu, Hongying Gong, and Guotao Yin. 2018. "Synthesis and Characterization of WO3/Graphene Nanocomposites for Enhanced Photocatalytic Activities by One-Step In-Situ Hydrothermal Reaction" Materials 11, no. 1: 147. https://doi.org/10.3390/ma11010147
APA StyleHu, X., Xu, P., Gong, H., & Yin, G. (2018). Synthesis and Characterization of WO3/Graphene Nanocomposites for Enhanced Photocatalytic Activities by One-Step In-Situ Hydrothermal Reaction. Materials, 11(1), 147. https://doi.org/10.3390/ma11010147






