Biological Degradation of Chinese Fir with Trametes Versicolor (L.) Lloyd
Abstract
1. Introduction
2. Results
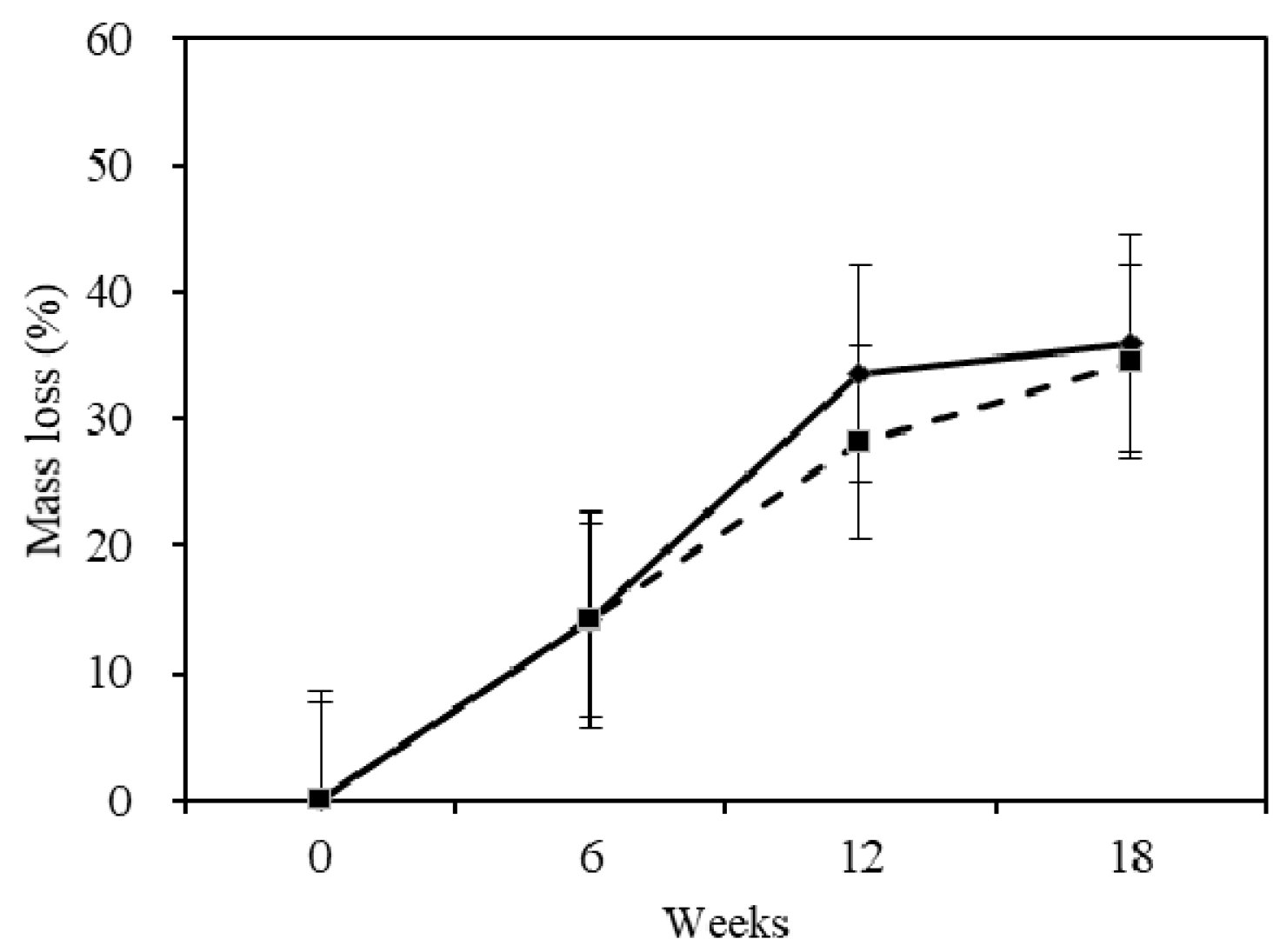
2.1. Mass Loss
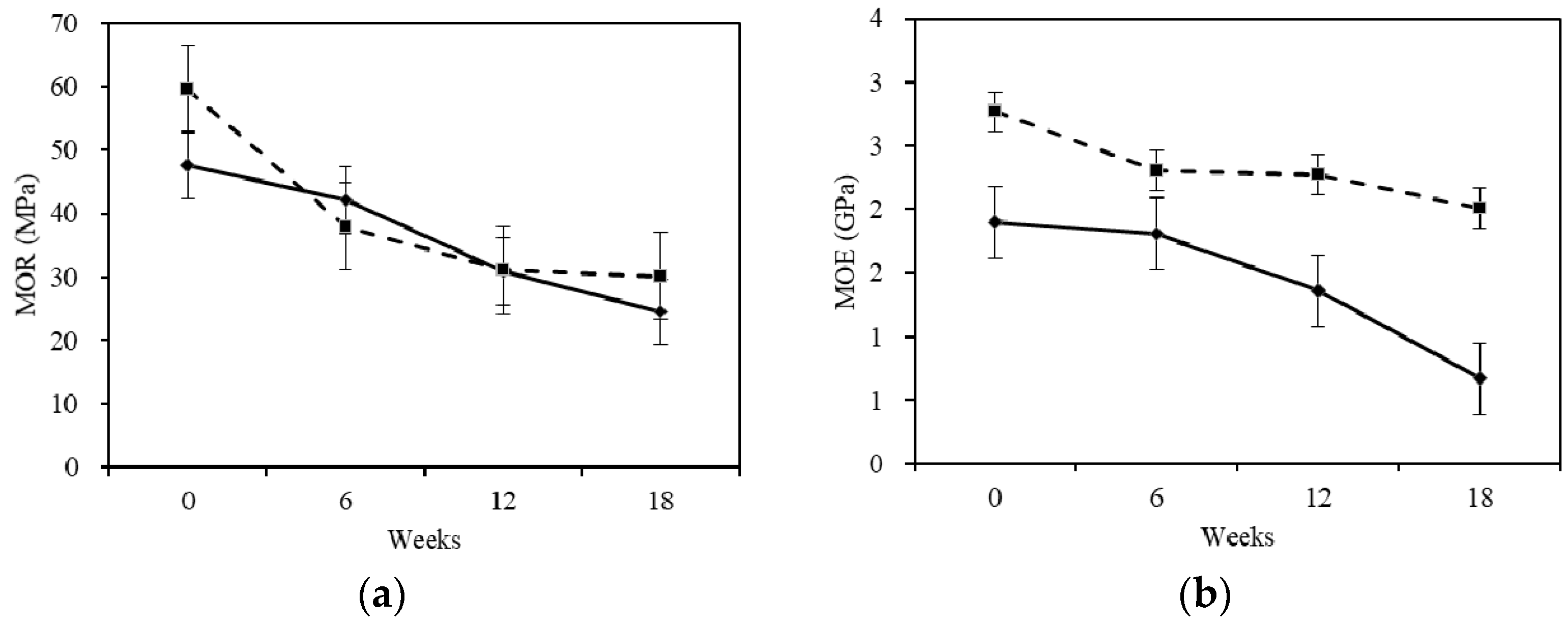
2.2. Mechanical Properties
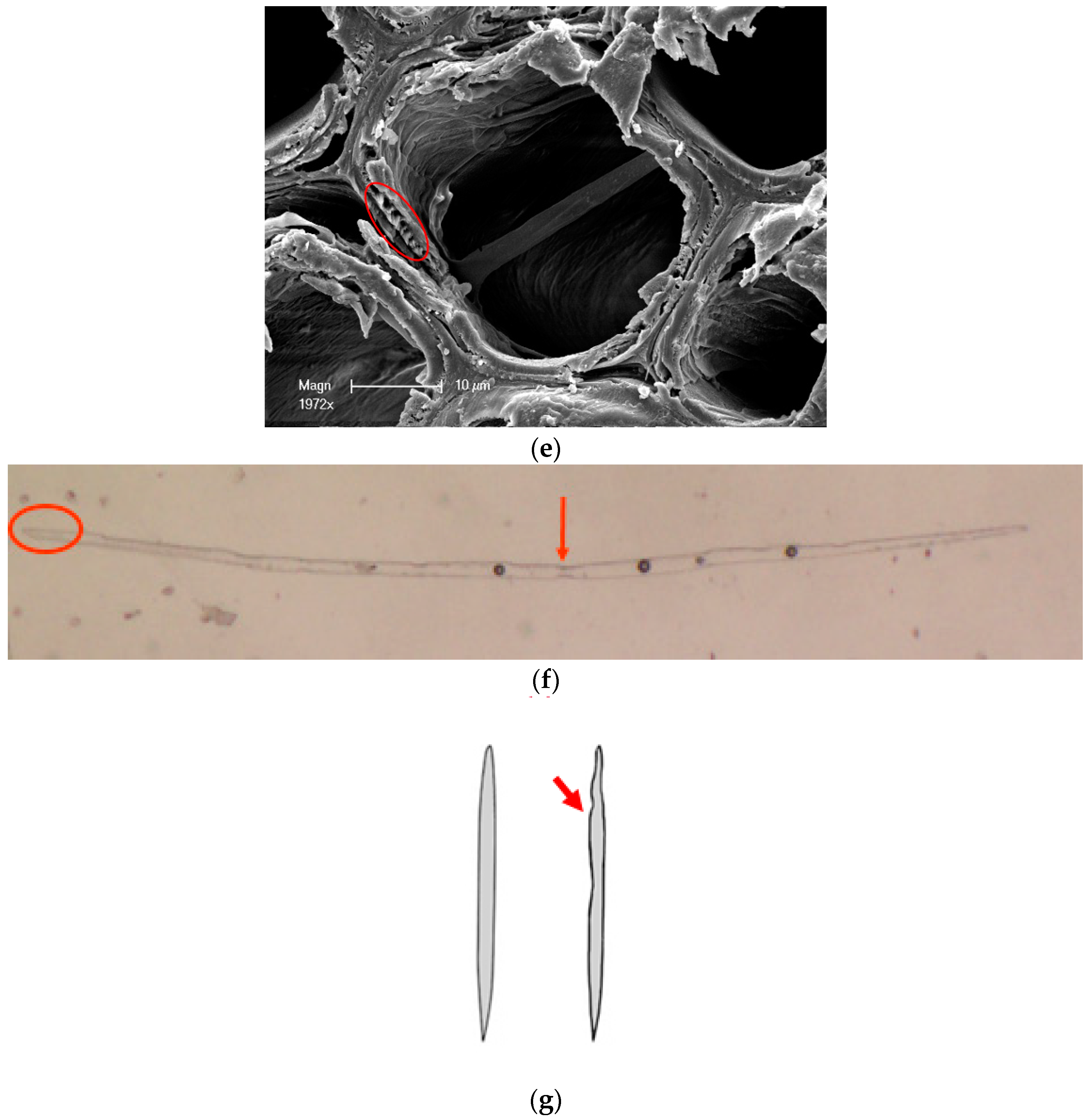
2.3. Characterization
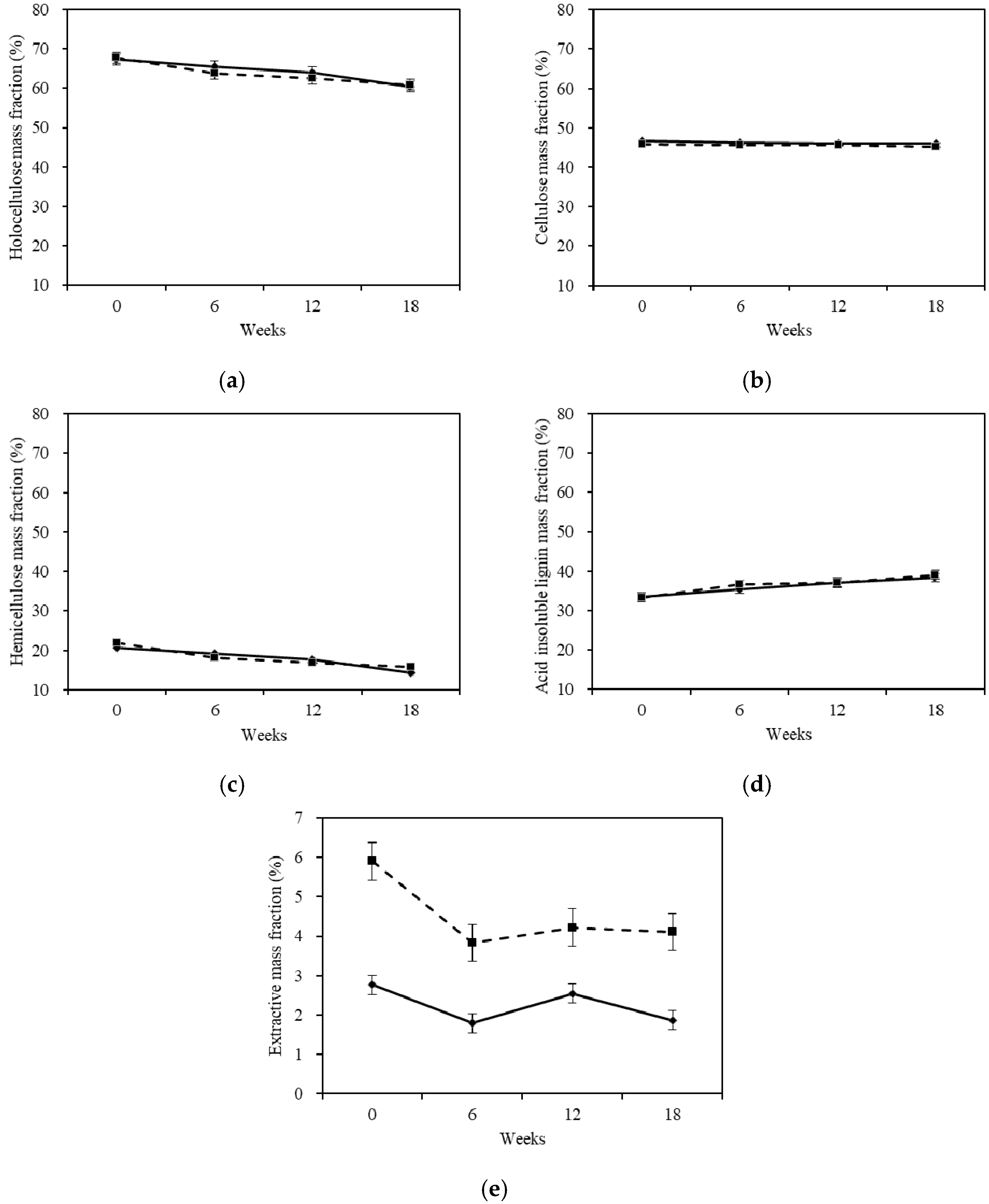
2.4. Chemical Properties Analysis
2.4.1. Chemical Compositions
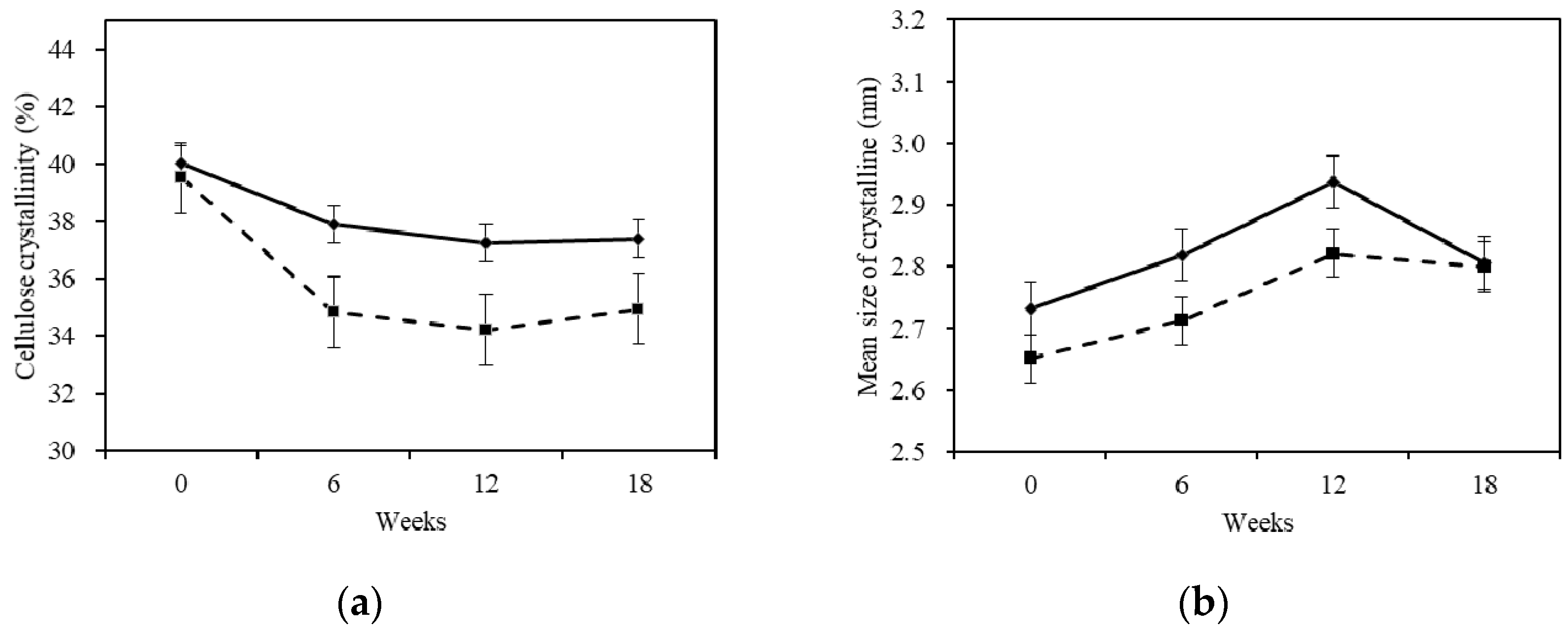
2.4.2. X-ray Diffraction
3. Materials and Methods
3.1. Materials
3.2. Biological Degradation
3.3. Mechanical Test
3.4. Characterization
3.4.1. Tracheid Morphology
3.4.2. Environment Scanning Electron Microscope Observation
3.5. Chemical Properties Analysis
3.5.1. Chemical Compositions
3.5.2. X-ray Diffraction Measurements
4. Conclusions
- Chinese fir was slightly durable, with a durability class of III. The heartwood was more durable than the sapwood.
- The MOR and MOE of fir wood decreased after the treatment with white-rot fungus and reached the minimum in the 18th week. The white-rot fungus had a greater influence on the mechanical properties (especially the MOE) for the sapwood.
- Trametes versicolor (L.) Lloyd degraded Chinese fir and colonized the lumen of various wood cell types in Chinese fir, penetrated cell walls via pits, caused erosion troughs and bore holes, and removed all cell layers.
- The ability of white-rot fungus to change the chemical composition mass fraction for Chinese fir was: hemicellulose > lignin > cellulose. The durability of the chemical compositions was: lignin > cellulose > hemicellulose.
- The crystallinity of the cellulose was decreased and the mean size of the ordered (crystalline) domains was increased after being treated with white-rot fungus.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yue, K.; Liu, W.Q.; Lu, X. Mechanical behavior and natural durability of fast-growing polar wood modified by chemical treatment. J. Anhui Agric. Univ. 2011, 38, 453–457. [Google Scholar]
- Yang, R.; Zhou, D.; Wang, S.; Zhang, Y.; Mao, H. Effect of hydrophobic modification on the wood nanomechanical properties of Chinese fir. China For. Sci. Technol. 2014, 28, 78–81. [Google Scholar]
- Wang, F.; Chai, Y.; Liu, J. Preparation and performance analysis of resin-based silicate modifier and modifed wood. Wood Proc. Mach. 2016, 4, 33–36. [Google Scholar]
- Crawford, R.L. Lignin Biodegradation and Transformation; Wiley: New York, NY, USA, 1981. [Google Scholar]
- Chen, M.; Wang, C.; Zhang, S.; Wu, H.; Pei, Y.; Liu, C. Selective decomposing of fir sapwood and heartwood with two wood-rotting fungi. J. Northeast For. Univ. 2015, 43, 82–85. [Google Scholar]
- Chen, M. Study on the Wood Properties after Biological Selectivity Degradation of Chinese Fir Wood; Anhui Agriculture University: Hefei, China, 2015. [Google Scholar]
- Cornelius, M.L.; Daigle, D.J.; Connick, W.J., Jr.; Parker, A.; Wunch, K. Responses of Coptotermes formosanus and Reticulitermes flavipes (Isoptera: Rhinotermitidae) to three types of wood rot fungi cultured on different substrates. J. Econ. Entomol. 2016, 95, 121. [Google Scholar] [CrossRef]
- Pérez, V.; Troya, M.T.D.; Martínez, A.T.; González-Vila, F.J.; Arias, E.; González, A.E. In vitro decay of Aextoxicon punctatum and Fagus sylvatica woods by white and brown-rot fungi. Wood Sci. Technol. 1993, 27, 295–307. [Google Scholar] [CrossRef]
- Davis, M.F.; Schroeder, H.A.; Maciel, G.E. Solid-state 13C nuclear magnetic resonance studies of wood decay. Pt. 2: White-rot decay of paper birch. Holzforsch. Int. J. Biol. Chem. Phys. Technol. Wood 1994, 48, 186–192. [Google Scholar]
- Shangguan, W.; Ren, H.; Lv, J.; Fei, B.; Chen, Z.; Zhao, R.; Zhao, Y. Cell wall property changes of white-rot larch during decay process. Bioresources 2014, 9. [Google Scholar] [CrossRef][Green Version]
- Bari, E.; Nazarnezhad, N.; Kazemi, S.M.; Ghanbary, M.A.T.; Mohebby, B.; Schmidt, O.; Clausen, C.A. Comparison between Degradation Capabilities of the White Rot Fungi Pleurotus ostreatus and Trametes versicolor in Beech wood. Int. Biodeterior. Biodegrad. 2015, 104, 231–237. [Google Scholar] [CrossRef]
- Bari, E.; Taghiyari, H.R.; Mohebby, B.; Clausen, C.A.; Schmidt, O.; Tajick Ghanbary, M.A.; Vaseghi, M.J. Mechanical properties and chemical composition of beech wood exposed for 30 and 120 days to white-rot fungi. Holzforschung 2014, 69, 587–593. [Google Scholar] [CrossRef]
- Jia, Z. Chinese Forest Resources Inventory Report-The Seventh National Forest Resources Inventory; China Forestry Publishing House: Beijing, China, 2009. [Google Scholar]
- Xiang, C.; Zhang, J.; Duan, A.; Zhang, X. Effects of planting density and site quality on stand volume of Chinese fir plantation. For. Res. 2014, 27, 801–808. [Google Scholar]
- GB/T 13942.1-2009 Durability of Wood-Part 1: Method for Laboratory Test of Natural Decay Resistance; General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China/Standardization Administration of the People’s Republic of China: Beijing, China, 2009.
- Huang, Z. Studies on Some Lesser-Utilised Tropical Hardwoods from Ghana; Brunel University: London, UK, 2005. [Google Scholar]
- Zabel, R.A.; Morrell, J.J. Wood Microbiology: Decay and Its Prevention; Academic Press, Inc.: London, UK, 1992. [Google Scholar]
- Wilcox, W.W. Anatomical changes in wood cell walls attacked by fungi and bacteria. Bot. Rev. 1970, 36, 1–28. [Google Scholar] [CrossRef]
- Guo, M.L.; Lan, H.F.; Qiu, J. Wood Deterioration and Preservation; China Metrology Publishing House: Beijing, China, 2010. [Google Scholar]
- Highley, T.L.; Murmanis, L.L. Micromorphology of degradation in western hemlock and sweetgum by the white-rot fungus Coriolus Versicolor. Holzforschung 1987, 41, 67–72. [Google Scholar] [CrossRef]
- Li, M.Y. Microstructure and Chemical Comoisition Changes of Betula Luminifera, Moso Bamboo and Phyllostachys Praecox in Decay; Zhejiang University: Hangzhou, China, 2012. [Google Scholar]
- Bari, E.; Taghiyari, H.R.; Naji, H.R.; Schmidt, O.; Ohno, K.M.; Clausen, C.A.; Bakar, E.S. Assessing the destructive behaviors of two white-rot fungi on beech wood. Int. Biodeterior. Biodegrad. 2016, 114, 129–140. [Google Scholar] [CrossRef]
- Kirk, T.K.; Cowling, E.B. Biological decomposition of solid wood. Biol. Decompos. Solid Wood 1984, 455–487. [Google Scholar]
- Kirk, T.K.; Chang, H.; Lorenz, L.F. Topochemistry of the fungal degradation of lignin in birch wood as related to the distribution of guaiacyl and syringyl lignins. Wood Sci. Technol. 1975, 9, 81–86. [Google Scholar] [CrossRef]
- Faix, O.; Mozuch, M.D.; Kirk, T.K. Degradation of gymnosperm (guaiacyl) vs. angiosperm (syringyl/guaiacyl) lignins by Phanerochaete chrysosporium. Holzforschung 1985, 39, 203–208. [Google Scholar] [CrossRef]
- Yatagai, M.; Takahashi, T. Tropical wood extractives’ effects on durability, paint curing time, and pulp sheet resin spotting. Wood Sci. 1980, 12, 176–181. [Google Scholar]
- Li, M.Y.; Qian, J.; Li, G.Y. Review of Research Progress on Wood-Decay Fungi and the Decay Mechanism. Anhui For. Sci. Technol. 2012, 38, 33–35. [Google Scholar]
- Liu, X.; Zhao, M.; Wang, Q. Biological characters of five species of wood rot fungi and decay capacity to Betula Platyphylla. J. Northeast For. Univ. 2008, 36, 41–44. [Google Scholar]
- Liu, X.; Xu, Y.; Wang, Q. Comparison of main components among those white birch varieties more susseptible to decaying. Trans. Beijing Inst. Technol. 2010, 30, 348–352. [Google Scholar]
- Nie, C.R. SPSS Tutorial; Yunnan University publishing house: Kunming, China, 2011. [Google Scholar]
- ASTM-D143-94 Standard Test Methods for Small Clear Specimens of Timber; American Society for Testing and Materials: West Conshohocken, PA, USA, 2000.
- Dixon, P.G.; Ahvenainen, P.; Aijazi, A.N.; Chen, S.H.; Lin, S.; Augusciak, P.K. Comparison of the structure and flexural properties of Moso, Guadua and Tre Gai bamboo. Constr. Build. Mater. 2015, 90, 11–17. [Google Scholar] [CrossRef]
- GB/T 747-2003 Determination of Insoluble Lignin in Pulp; General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China/Standardization Administration of the People’s Republic of China: Beijing, China, 2003.
- GB/T 744-1989 Determination of Cellulose in Pulp; General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China/Standardization Administration of the People’s Republic of China: Beijing, China, 1989.
- GB/T 2677-1994 Determination of Extraction in Pulp; General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China/Standardization Administration of the People’s Republic of China: Beijing, China, 1994.
- GB/T 2677.10-1995 Determination of Holocellulose in Pulp; General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China/Standardization Administration of the People’s Republic of China: Beijing, China, 1995.
- Toba, K.; Nakai, T.; Shirai, T.; Yamamoto, H. Changes in the cellulose crystallinity of Moso bamboo cell walls during the growth process by X-ray diffraction techniques. J. Wood Sci. 2015, 61, 1–8. [Google Scholar] [CrossRef]
- Wang, Y.; Leppänen, K.; Andersson, S.; Serimaa, R.; Ren, H.; Fei, B. Studies on the nanostructure of the cell wall of bamboo using X-ray scattering. Wood Sci. Technol. 2012, 46, 317–332. [Google Scholar] [CrossRef]
- Patterson, A. The Scherrer Formula for X-ray Particle Size Determination. Phys. Rev. 1939, 56, 978–982. [Google Scholar] [CrossRef]


| Durability Class | Description | Mass Loss |
|---|---|---|
| I | Very durable | ML ≤ 0 |
| II | Durable | 0.11 < ML ≤ 0.24 |
| III | Slightly durable | 0.25 < ML ≤ 0.45 |
| IV | Not durable | ML > 0.45 |
| Samples | Weeks | Holocellulose (%) | Cellulose (%) | Hemicellulose (%) | Acid-Insoluble Lignin (%) |
|---|---|---|---|---|---|
| Sapwood | 0 | 67.3 | 46.7 | 20.6 | 33.6 |
| 6 | 56.2 | 39.6 | 16.5 | 30.3 | |
| 12 | 42.4 | 30.6 | 11.8 | 24.5 | |
| 18 | 38.7 | 29.5 | 9.1 | 24.6 | |
| Heartwood | 0 | 67.7 | 45.8 | 22.0 | 33.3 |
| 6 | 54.7 | 39.2 | 15.6 | 31.4 | |
| 12 | 44.9 | 32.7 | 12.2 | 26.6 | |
| 18 | 39.9 | 29.5 | 10.4 | 25.6 |
| Samples | Weeks | Holocellulose (%) | Cellulose (%) | Hemicellulose (%) | Acid-Insoluble Lignin (%) |
|---|---|---|---|---|---|
| Sapwood | 0 | 0 | 0 | 0 | 0 |
| 6 | 16.5 | 15.0 | 19.9 | 9.6 | |
| 12 | 36.9 | 34.4 | 42.7 | 26.9 | |
| 18 | 42.5 | 36.7 | 55.7 | 26.7 | |
| Heartwood | 0 | 0 | 0 | 0 | 0 |
| 6 | 19.2 | 14.3 | 29.2 | 5.8 | |
| 12 | 33.8 | 28.5 | 44.6 | 20.1 | |
| 18 | 41.1 | 35.4 | 52.9 | 23.4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, M.; Wang, C.; Fei, B.; Ma, X.; Zhang, B.; Zhang, S.; Huang, A. Biological Degradation of Chinese Fir with Trametes Versicolor (L.) Lloyd. Materials 2017, 10, 834. https://doi.org/10.3390/ma10070834
Chen M, Wang C, Fei B, Ma X, Zhang B, Zhang S, Huang A. Biological Degradation of Chinese Fir with Trametes Versicolor (L.) Lloyd. Materials. 2017; 10(7):834. https://doi.org/10.3390/ma10070834
Chicago/Turabian StyleChen, Meiling, Chuangui Wang, Benhua Fei, Xinxin Ma, Bo Zhang, Shuangyan Zhang, and Anmin Huang. 2017. "Biological Degradation of Chinese Fir with Trametes Versicolor (L.) Lloyd" Materials 10, no. 7: 834. https://doi.org/10.3390/ma10070834
APA StyleChen, M., Wang, C., Fei, B., Ma, X., Zhang, B., Zhang, S., & Huang, A. (2017). Biological Degradation of Chinese Fir with Trametes Versicolor (L.) Lloyd. Materials, 10(7), 834. https://doi.org/10.3390/ma10070834






