Effects of Nitrogen and Tensile Direction on Stress Corrosion Cracking Susceptibility of Ni-Free FeCrMnC-Based Duplex Stainless Steels
Abstract
:1. Introduction
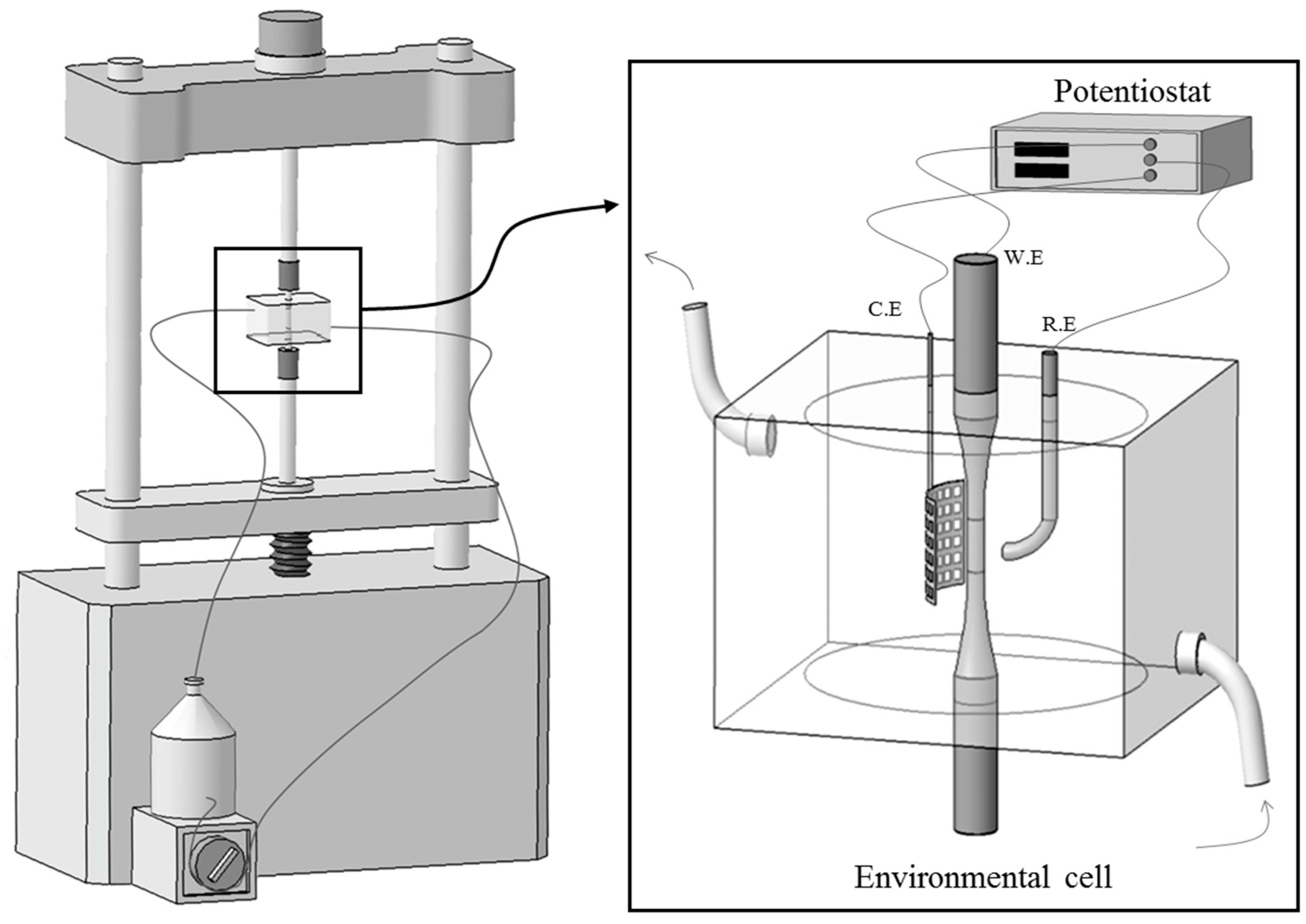
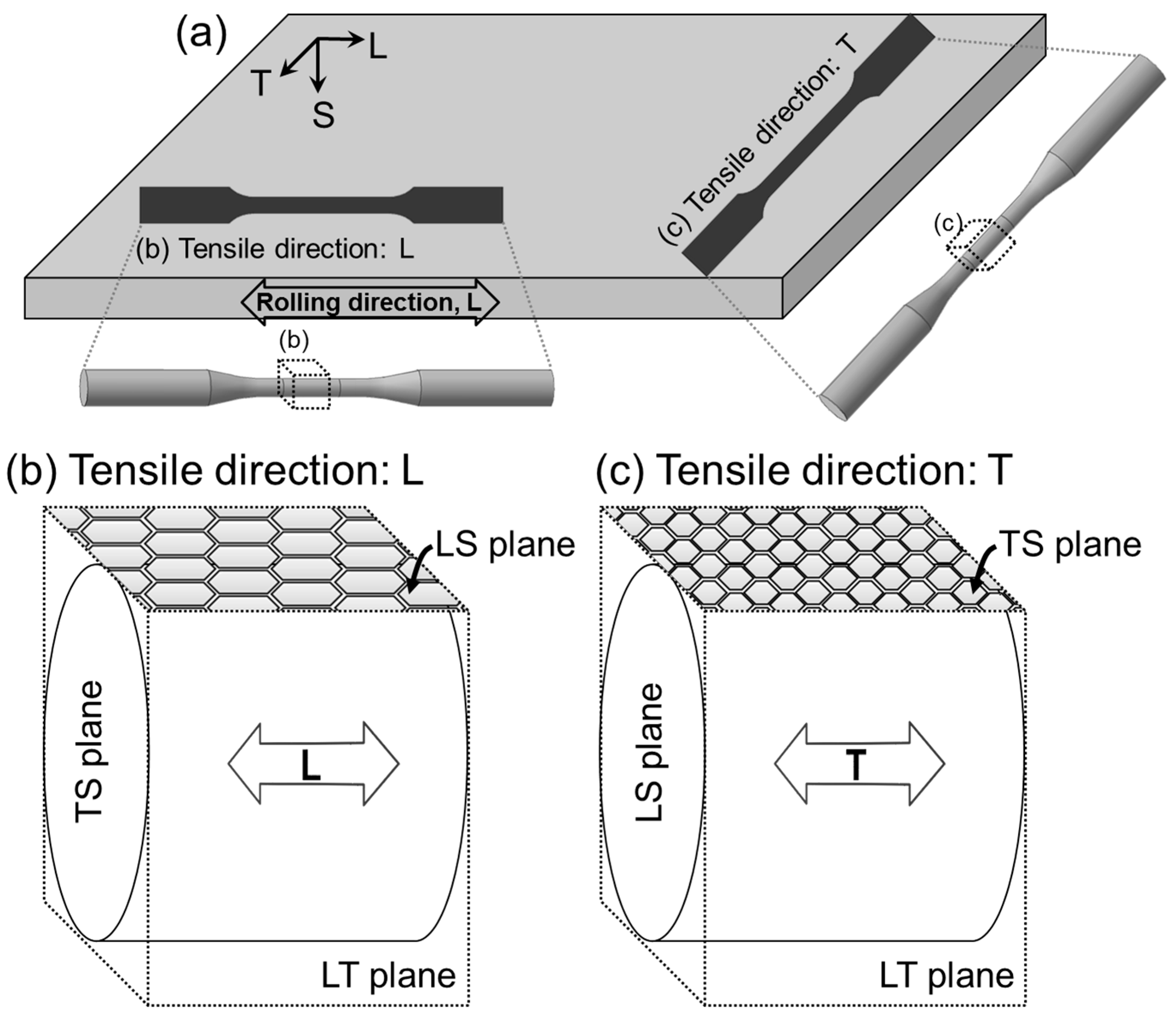
2. Experimental Procedure
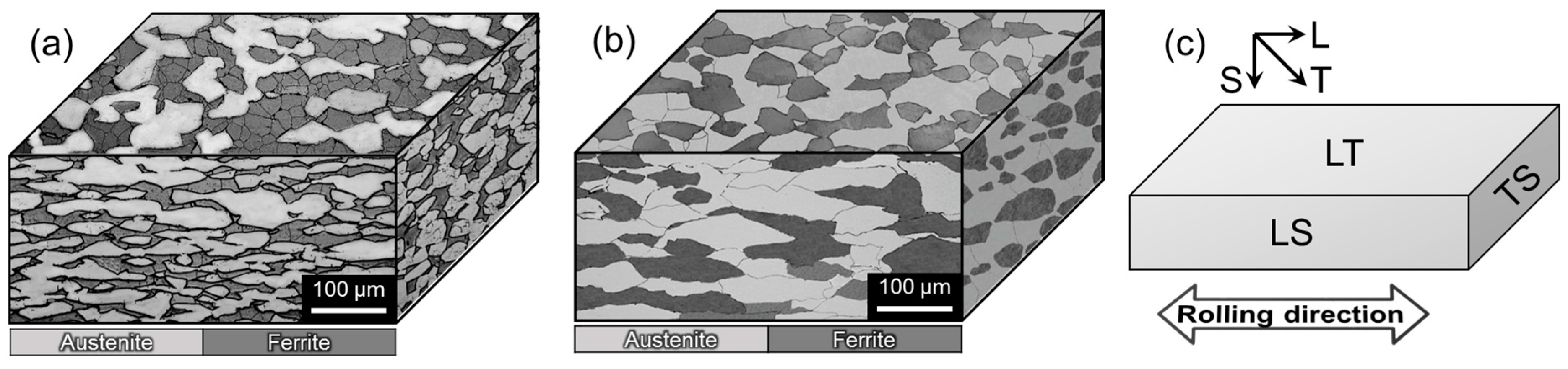
2.1. Materials
2.2. Corrosion Tests
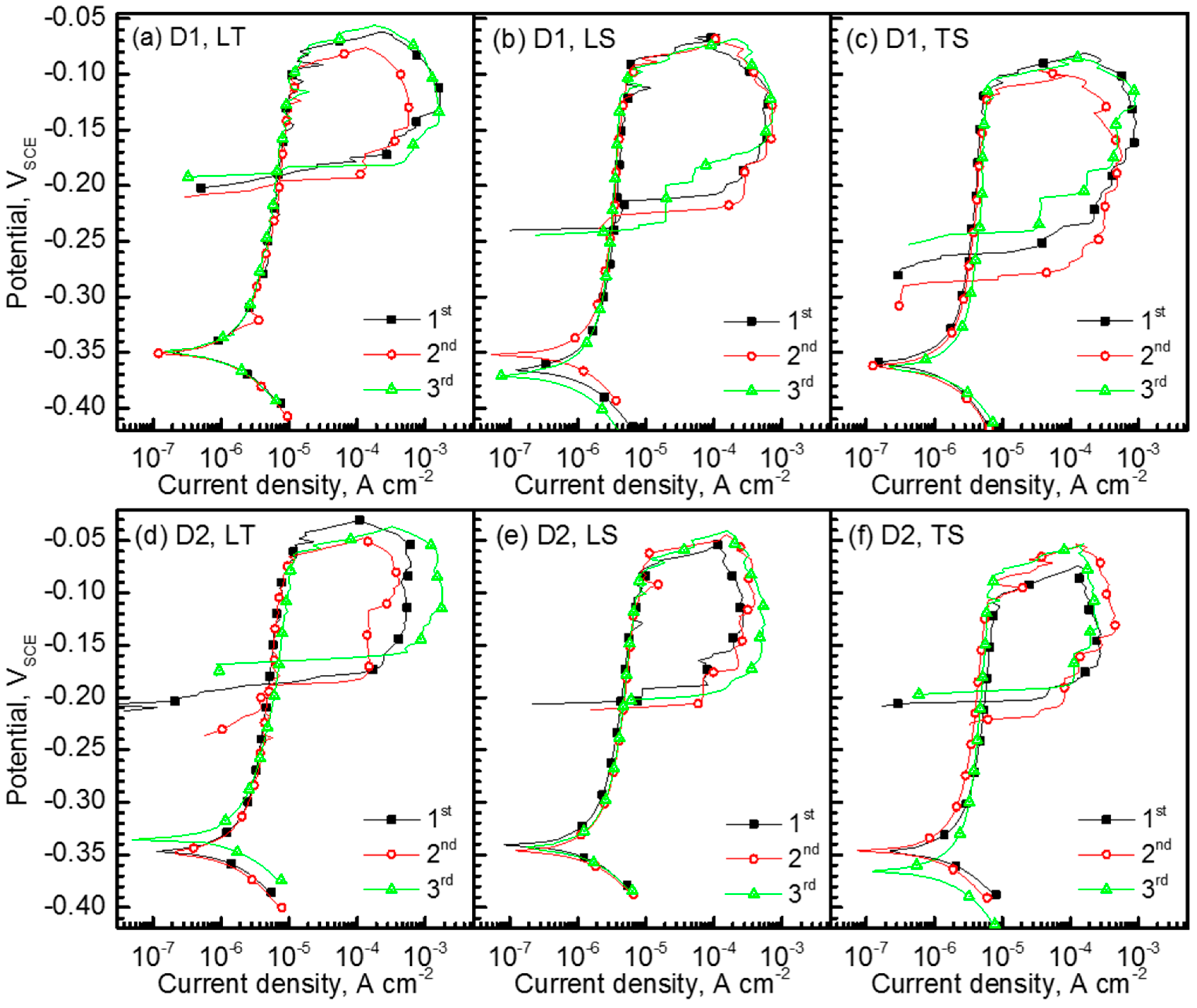
3. Results and Discussion
4. Conclusions
- (1)
- Alloying N was beneficial to resistance to pitting corrosion; it was also found that the TS plane with elongated small grains exhibited the lowest resistance to pitting corrosion, while the LT plane showed the highest resistance.
- (2)
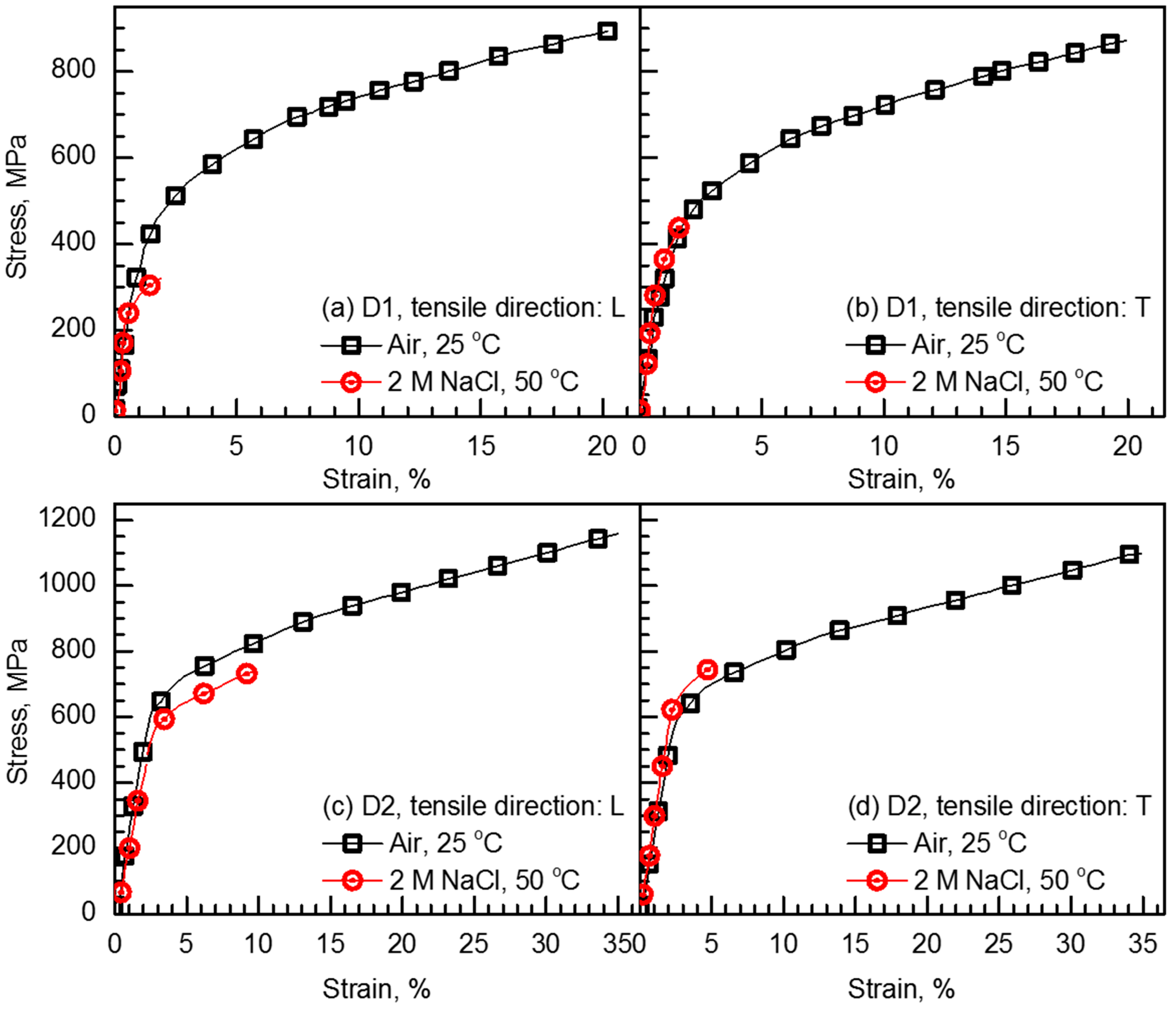
- The alloying N significantly improved both tensile strength and ductility of the FeCrMnC-based alloys. In addition, no notable anisotropy was observed in tensile properties in air; the strength and ductility for both L- and T-directions were similar to each other. The alloying N was effective in increasing the resistance to SCC, and higher SCC resistance was obtained when SSRTed along L-direction than T-direction.
- (3)
- The enhanced SCC resistance by N addition was due to an increase in the resistance to pitting corrosion and the mechanical properties. The difference in the SCC resistance with different tensile loading directions was primarily attributed to the resistance to pitting corrosion of the metallographic planes exposed to the environment during SSRT.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lo, K.H.; Shek, C.H.; Lai, J.K.L. Recent developments in stainless steels. Mater. Sci. Eng. R Rep. 2009, 65, 39–104. [Google Scholar] [CrossRef]
- Baddoo, N.R. Stainless steel in construction: A review of research, applications, challenges and opportunities. J. Constr. Steel. Res. 2008, 64, 1199–1206. [Google Scholar] [CrossRef]
- Davison, R.M.; Redmond, D. A guide to using duplex stainless steels. Mater. Des. 1991, 12, 187–192. [Google Scholar] [CrossRef]
- Maki, T.; Furuhara, T.; Tsuzaki, K. Microstructure development by thermomechanical processing in duplex stainless steel. ISIJ Int. 2001, 41, 571–579. [Google Scholar] [CrossRef]
- Olsson, J.; Snis, M. Duplex—A new generation of stainless steels for desalination plants. Desalination 2007, 205, 104–113. [Google Scholar] [CrossRef]
- Snis, M.; Olsson, J. Reduce costs for storage and distribution of desalted water—Use duplex stainless steel. Desalination 2008, 223, 476–486. [Google Scholar] [CrossRef]
- Alvarez-Armas, I. Duplex stainless steels: Brief history and some recent alloys. Recent Pat. Mech. Eng. 2008, 1, 51–57. [Google Scholar] [CrossRef]
- Liljas, M.; Johansson, P.; Liu, H.-P.; Olsson, C.-O.A. Development of a lean duplex stainless steel. Steel Res. Int. 2008, 79, 466–473. [Google Scholar] [CrossRef]
- Sahu, J.K.; Krupp, U.; Ghosh, R.N.; Christ, H.-J. Effect of 475 °C embrittlement on the mechanical properties of duplex stainless steel. Mater. Sci. Eng. A 2009, 508, 1–14. [Google Scholar] [CrossRef]
- Jorge, A.M.; Reis, G.S.; Balancin, O. Influence of the microstructure on the plastic behaviour of duplex stainless steels. Mater. Sci. Eng. A 2011, 528, 2259–2264. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Kim, S. Effect of W on stress corrosion cracking susceptibility of newly developed Ni-saving duplex stainless steels. Met. Mater. Int. 2017, 23, 115–125. [Google Scholar] [CrossRef]
- Gavrilyuk, V.G.; Berns, H. Stainless and electrical steels. Met. Sci. Heat Treat. 2007, 49, 566–568. [Google Scholar] [CrossRef]
- Gavriljuk, V.G.; Shanina, B.D.; Berns, H. A physical concept for alloying steels with carbon + nitrogen. Mater. Sci. Eng. A 2008, 481–482, 707–712. [Google Scholar] [CrossRef]
- Shanina, B.D.; Tyshchenko, A.I.; Glavatskyy, I.N.; Runov, V.V.; Petrov, Y.N.; Berns, H.; Gavriljuk, V.G. Chemical nano-scale homogeneity of austenitic CrMnCN steels in relation to electronic and magnetic properties. J. Mater. Sci. 2011, 467, 7725–7736. [Google Scholar] [CrossRef]
- Thomann, U.I.; Uggowitzer, P.J. Wear-corrosion behavior of biocompatible austenitic stainless steels. Wear 2000, 239, 48–58. [Google Scholar] [CrossRef]
- Ha, H.Y.; Lee, T.H.; Oh, C.S.; Kim, S.J. Effects of carbon on the corrosion behaviour in Fe-18Cr-10Mn-N-C stainless steels. Steel Res. Int. 2009, 80, 488–492. [Google Scholar]
- Ha, H.Y.; Lee, T.H.; Oh, C.S.; Kim, S.J. Effects of combined addition of carbon and nitrogen on pitting corrosion behavior of Fe-18Cr-10Mn alloys. Scr. Mater. 2009, 61, 121–124. [Google Scholar] [CrossRef]
- Lee, T.H.; Shin, E.; Oh, C.S.; Ha, H.Y.; Kim, S.J. Correlation between stacking fault energy and deformation microstructure in high-interstitial-alloyed austenitic steels. Acta Mater. 2010, 58, 3173–3186. [Google Scholar] [CrossRef]
- Yoon, Y.S.; Ha, H.Y.; Lee, T.H.; Kim, S. Effect of N and C on stress corrosion cracking susceptibility of austenitic Fe18Cr10Mn-based stainless steels. Corros. Sci. 2014, 80, 28–36. [Google Scholar] [CrossRef]
- Seifert, M.; Siebert, S.; Huth, S.; Theisen, W.; Berns, H. New developments in martensitic stainless steels containing C + N. Steel Res. Int. 2015, 86, 1508–1516. [Google Scholar] [CrossRef]
- Frankel, G.S. Pitting corrosion of metals—A review of the critical factors. J. Electrochem. Soc. 1998, 145, 2186–2198. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Lee, T.-H.; Kim, S.-J. Role of nitrogen in the active-passive transition behavior of binary Fe-Cr alloy system. Electrochim. Acta 2012, 80, 432–439. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Jang, H.J.; Kwon, H.S.; Kim, S.J. Effects of nitrogen on the passivity of Fe-20Cr alloy. Corros. Sci. 2009, 51, 48–53. [Google Scholar] [CrossRef]
- Levey, P.R.; Bennekom, A. A mechanistic study of the effects of nitrogen on the corrosion properties of stainless steels. Corrosion 1995, 51, 911–921. [Google Scholar] [CrossRef]
- Jargelius-Pettersson, R.F.A. Electrochemical investigation of the influence of nitrogen alloying on pitting corrosion of austenitic stainless steels. Corros. Sci. 1999, 41, 1639–1664. [Google Scholar] [CrossRef]
- Mozhi, T.A.; Clark, W.A.T.; Wilde, B.E. The effect of nitrogen and carbon on the stress corrosion cracking performance of sensitized AISI 304 stainless steel in chloride and sulfate solutions at 250 °C. Corros. Sci. 1987, 27, 257–273. [Google Scholar] [CrossRef]
- Ha, H.-Y.; Seo, W.-G.; Park, J.Y.; Lee, T.-H.; Kim, S. Influences of Mo on stress corrosion cracking susceptibility of newly developed FeCrMnNiNC-based lean austenitic stainless steels. Mater. Charact. 2016, 119, 200–208. [Google Scholar] [CrossRef]
- Congleton, J.; Yang, W. The effect of applied potential on the stress corrosion cracking of sensitized type 316 stainless steel in high temperature water. Corros. Sci. 1995, 37, 429–444. [Google Scholar] [CrossRef]
- Kwon, H.S. Prediction of stress corrosion cracking susceptibility of stainless steels based on repassivation kinetics. Corrosion 2000, 56, 32–40. [Google Scholar] [CrossRef]
- Bernard, F.; Shankar Rao, V.; Kwon, H.-S. A study on the repassivation kinetics and SCC behavior of duplex stainless steel in chloride solution. J. Electrochem. Soc. 2005, 152, B415–B420. [Google Scholar] [CrossRef]
- Yoon, Y.S.; Ha, H.Y.; Lee, T.H.; Kim, S. Comparative study of stress corrosion cracking susceptibility of Fe18Cr10Mn- and Fe18Cr10Mn1Ni-based high nitrogen stainless steels. Corros. Sci. 2014, 88, 337–348. [Google Scholar] [CrossRef]
- Toor, I.H.; Park, K.J.; Kwon, H.S. Manganese effects on repassivation kinetics and SCCsusceptibility of high Mn-N austenitic stainless steel alloys. J. Electrochem. Soc. 2007, 154, C494–C499. [Google Scholar] [CrossRef]
- Tseng, C.M.; Liou, H.Y.; Tsai, W.T. Effect of nitrogen content on the environmentally assisted cracking susceptibility of duplex stainless steels. Metall. Mater. Trans. A 2003, 34, 95–103. [Google Scholar] [CrossRef]
- Zhu, L.K.; Yan, Y.; Qiao, L.J.; Volinsky, A.A. Stainless steel pitting and early-stage stress corrosion cracking under ultra-low elastic load. Corros. Sci. 2013, 77, 360–368. [Google Scholar] [CrossRef]
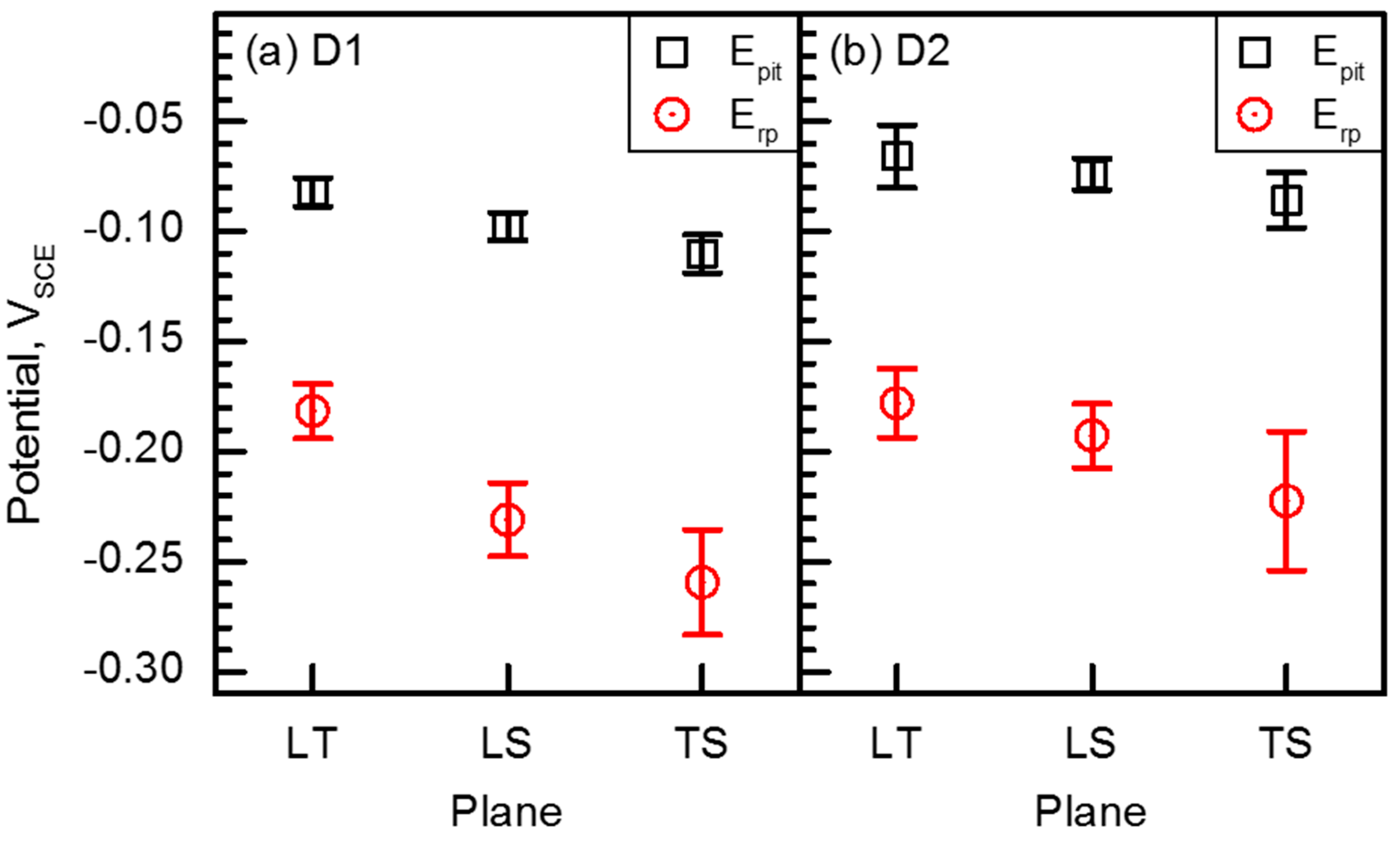






| Alloy | Fe | Cr | Mn | N | C |
|---|---|---|---|---|---|
| D1 | Balance | 19.151 | 7.889 | 0.034 | 0.278 |
| D2 | Balance | 19.203 | 7.827 | 0.210 | 0.234 |
| Si ≤ 0.15, Ni ≤ 0.01, S < 0.005, P < 0.003 | |||||
| Alloy | Tensile Direction | Environment | Yield Strength MPa | Tensile Strength MPa | Elongation % | Reduction of Tensile Elongation % |
|---|---|---|---|---|---|---|
| D1 | L | Air, 25 °C | 420 | 858 | 15.8 | |
| 2 M NaCl, 50 °C | 287 | 308 | 2.0 | 87.3 | ||
| T | Air, 25 °C | 413 | 888 | 16.3 | ||
| 2 M NaCl, 50 °C | 366 | 449 | 1.8 | 89.0 | ||
| D2 | L | Air, 25 °C | 631 | 1156 | 29.7 | |
| 2 M NaCl, 50 °C | 554 | 770 | 10.6 | 62.1 | ||
| T | Air, 25 °C | 590 | 1094 | 30.8 | ||
| 2 M NaCl, 50 °C | 595 | 767 | 5.5 | 82.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, H.-Y.; Lee, C.-H.; Lee, T.-H.; Kim, S. Effects of Nitrogen and Tensile Direction on Stress Corrosion Cracking Susceptibility of Ni-Free FeCrMnC-Based Duplex Stainless Steels. Materials 2017, 10, 294. https://doi.org/10.3390/ma10030294
Ha H-Y, Lee C-H, Lee T-H, Kim S. Effects of Nitrogen and Tensile Direction on Stress Corrosion Cracking Susceptibility of Ni-Free FeCrMnC-Based Duplex Stainless Steels. Materials. 2017; 10(3):294. https://doi.org/10.3390/ma10030294
Chicago/Turabian StyleHa, Heon-Young, Chang-Hoon Lee, Tae-Ho Lee, and Sangshik Kim. 2017. "Effects of Nitrogen and Tensile Direction on Stress Corrosion Cracking Susceptibility of Ni-Free FeCrMnC-Based Duplex Stainless Steels" Materials 10, no. 3: 294. https://doi.org/10.3390/ma10030294
APA StyleHa, H.-Y., Lee, C.-H., Lee, T.-H., & Kim, S. (2017). Effects of Nitrogen and Tensile Direction on Stress Corrosion Cracking Susceptibility of Ni-Free FeCrMnC-Based Duplex Stainless Steels. Materials, 10(3), 294. https://doi.org/10.3390/ma10030294







