BPPO-Based Anion Exchange Membranes for Acid Recovery via Diffusion Dialysis
Abstract
:1. Introduction
2. Experimental
2.1. Materials
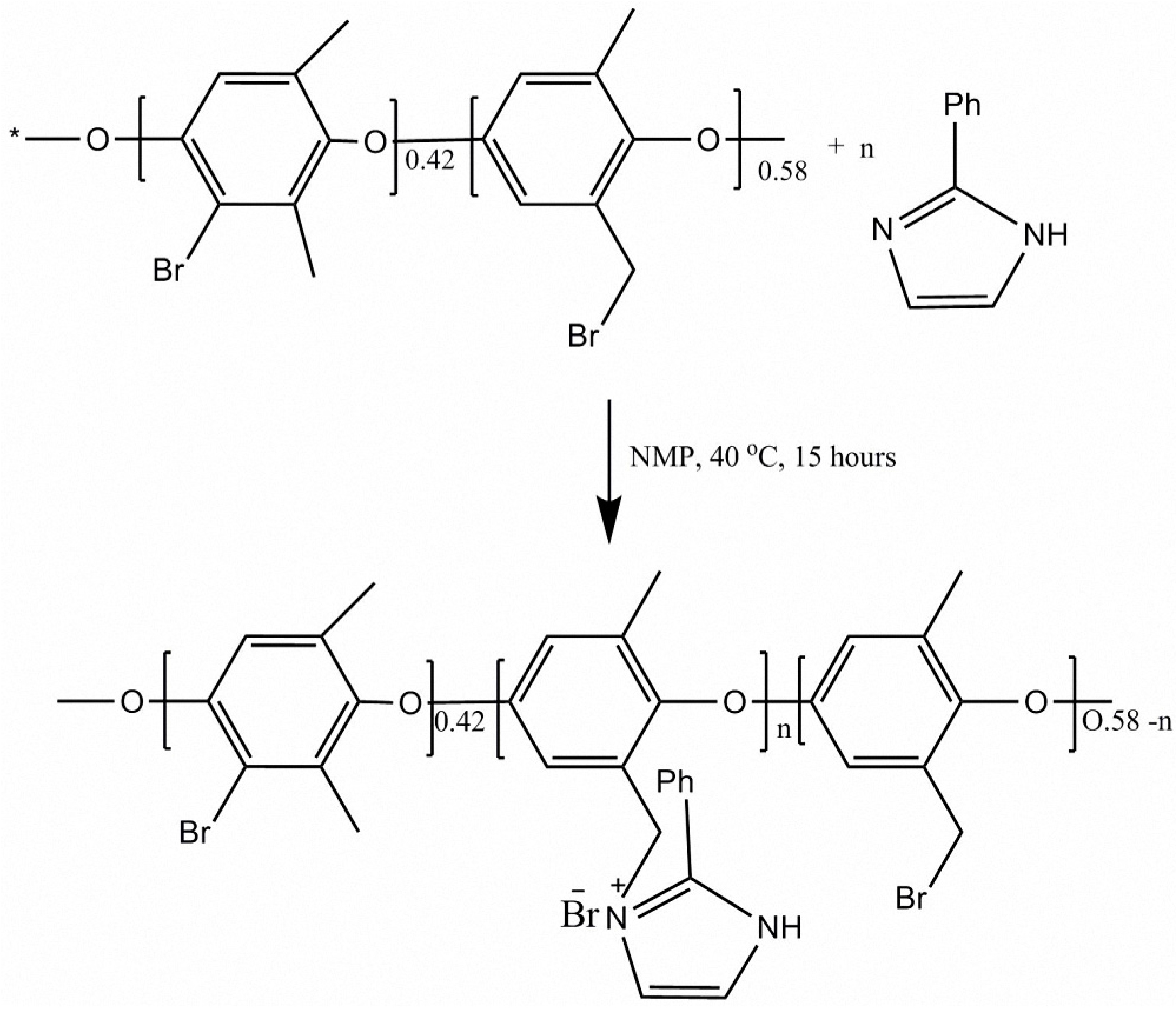
2.2. Membrane Preparation
2.3. Characterization
2.3.1. Functional Groups and Thermal Stability
2.3.2. Morphology
2.3.3. Ion Exchange Capacity (IEC)
2.3.4. Water Uptake, Linear Expansion Ratio and Thickness
2.3.5. Chemical and Mechanical Stability
2.3.6. Diffusion Dialysis of HCl/FeCl2 Mixtures
3. Results
3.1. Functional Groups
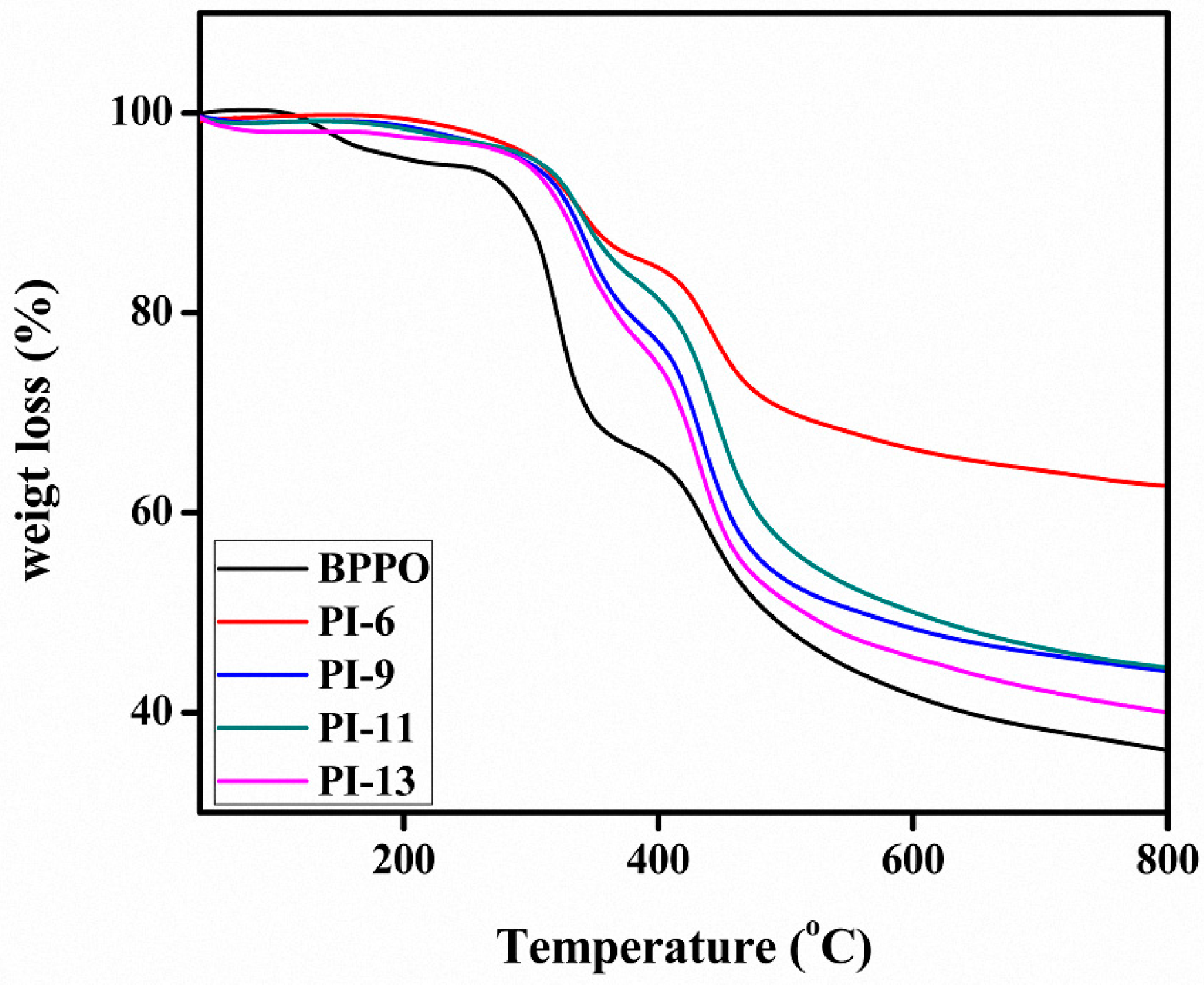
3.2. Thermal Stability
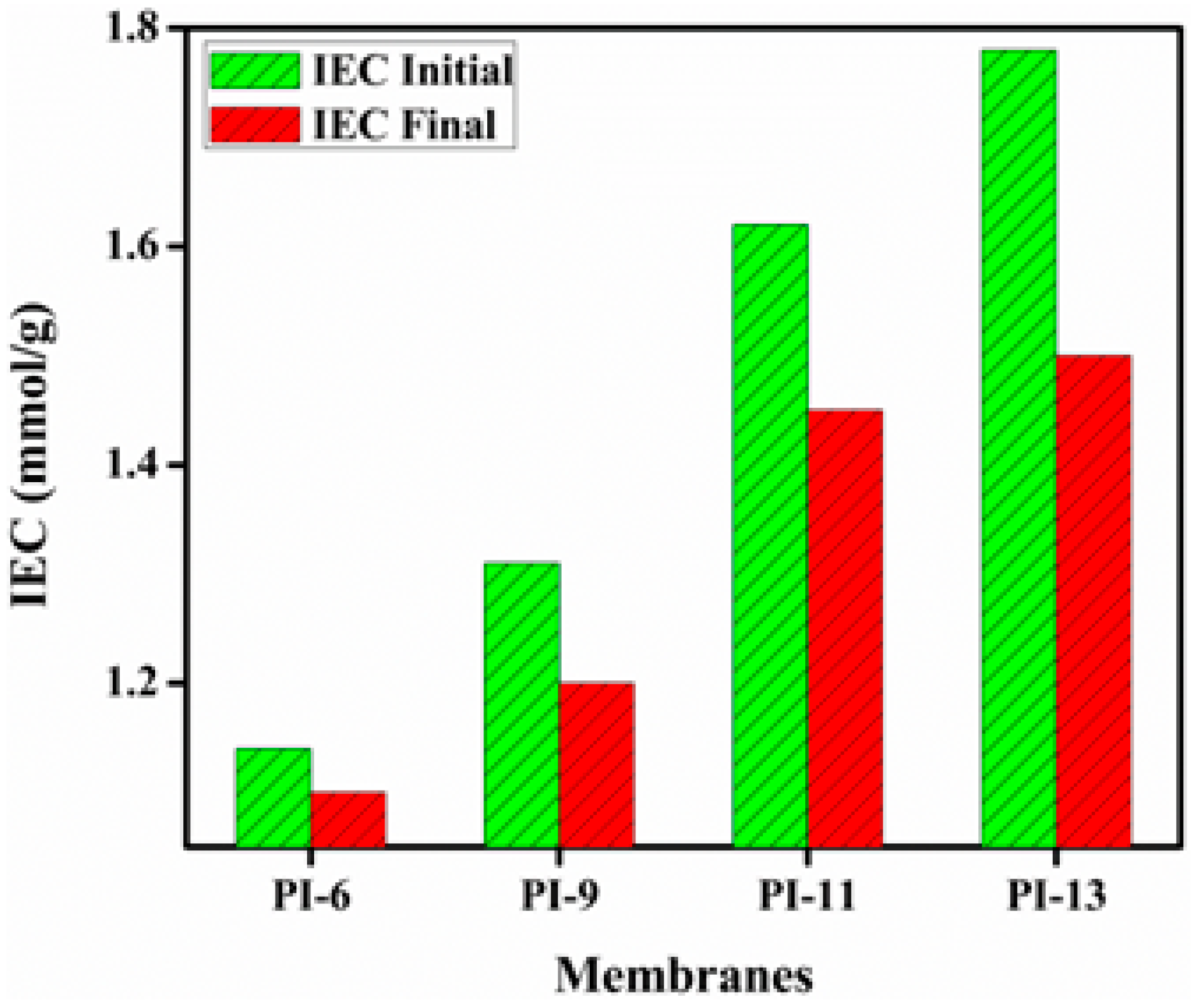
3.3. Ion Exchange Capacity (IEC), Water Uptake (WU) and Linear Expansion Ratio (LER)
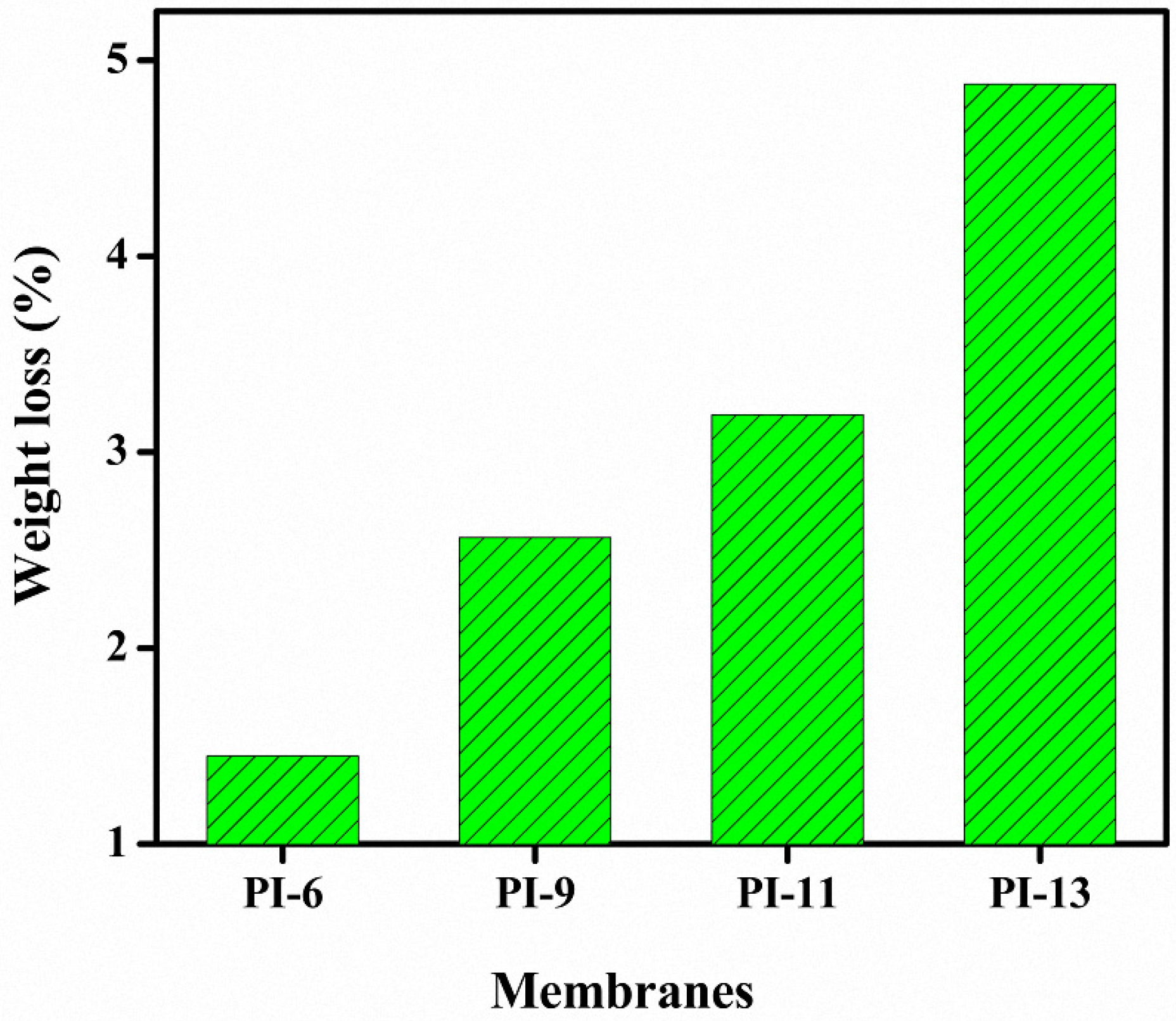
3.4. Chemical and Mechanical Stability
3.5. Morphology
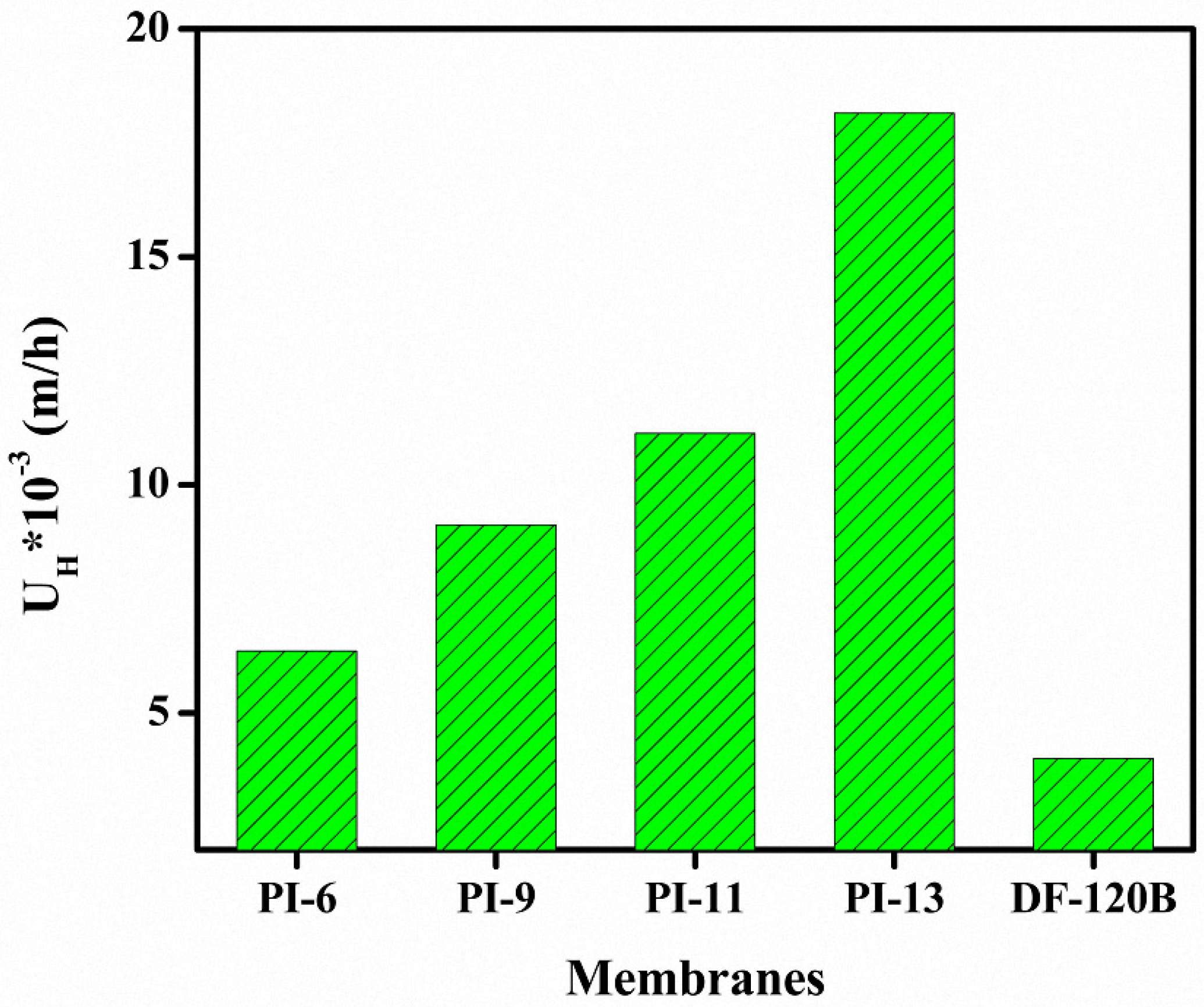
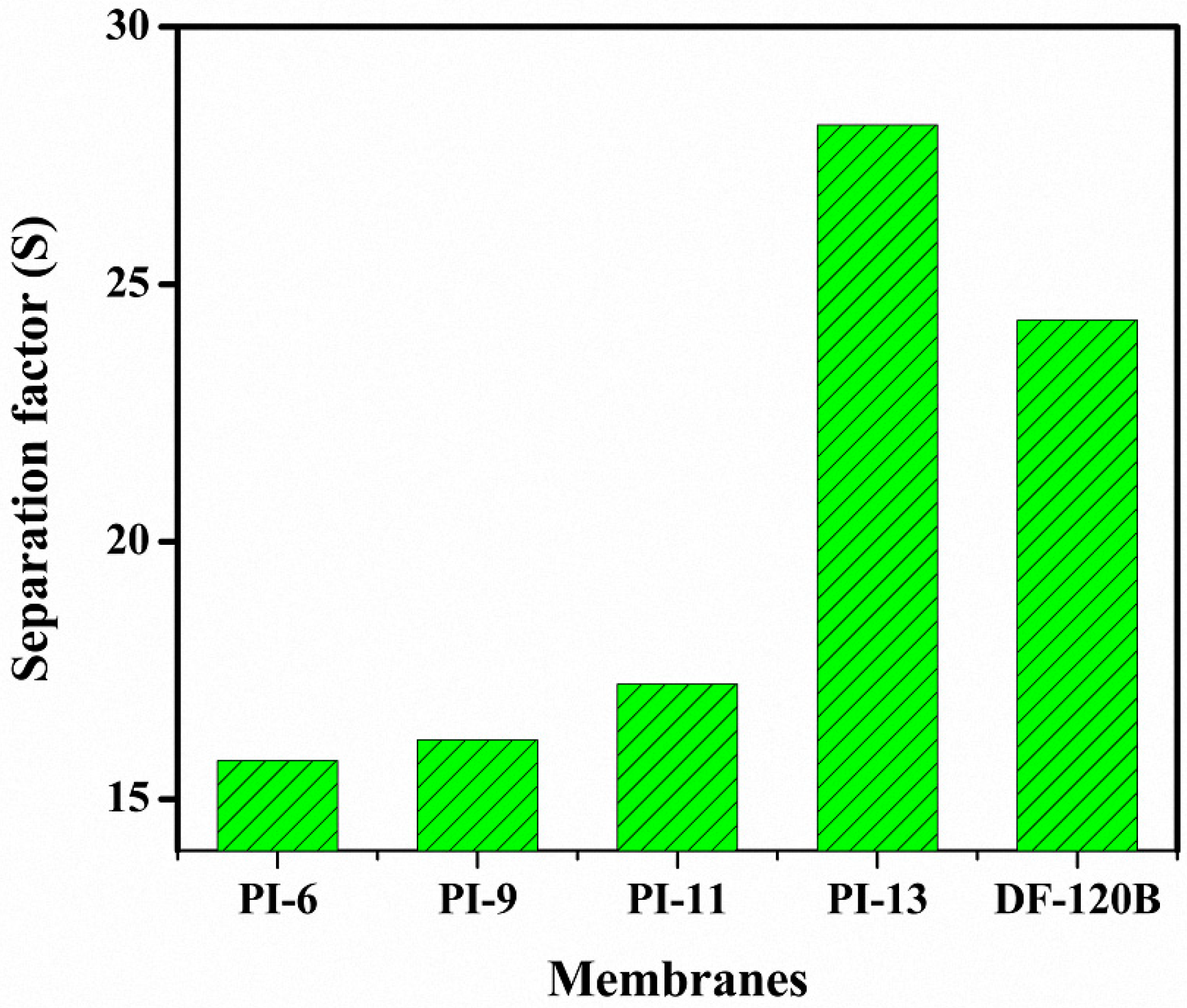
3.6. DD for HCl/FeCl2 Solution
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Strathmann, H. Ion-Exchange Membrane Separation Process; Elsevier: Amsterdam, The Netherlands, 2004. [Google Scholar]
- Luo, J.; Wu, C.; Xu, T.; Wu, Y. Diffusion dialysis-concept, principle and applications. J. Membr. Sci. 2011, 366, 1–16. [Google Scholar] [CrossRef]
- Kang, M.-S.; Yoo, K.-S.; Oh, S.-J.; Moon, S.-H. A lumped parameter model to predict hydrochloric acid recovery in diffusion dialysis. J. Membr. Sci. 2001, 188, 61–70. [Google Scholar] [CrossRef]
- Khan, M.I.; Mondal, A.N.; Emmanuel, K.; Hossain, M.M.; Afsar, N.U.; Wu, L.; Xu, T. Preparation of pyrrolidinium-based anion-exchange membranes for acid recovery via diffusion dialysis. Sep. Sci. Technol. 2016, 51, 1881–1890. [Google Scholar] [CrossRef]
- Khan, M.I.; Mondal, A.N.; Tong, B.; Jiang, C.; Emmanuel, K.; Yang, Z.; Wu, L.; Xu, T. Development of bppo-based anion exchange membranes for electrodialysis desalination applications. Desalination 2016, 391, 61–68. [Google Scholar] [CrossRef]
- Xu, J.; Lu, S.; Fu, D. Recovery of hydrochloric acid from the waste acid solution by diffusion dialysis. J. Hazard. Mater. 2009, 165, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Palatý, Z.; Žáková, A. Competitive transport of hydrochloric acid and zinc chloride through polymeric anion-exchange membrane. J. Appl. Polym. Sci. 2006, 101, 1391–1397. [Google Scholar] [CrossRef]
- Negro, C.; Blanco, M.; Lopez-Mateos, F.; DeJong, A.; LaCalle, G.; van Erkel, J.; Schmal, D. Free acids and chemicals recovery from stainless steel pickling baths. Sep. Sci. Technol. 2001, 36, 1543–1556. [Google Scholar] [CrossRef]
- Jung Oh, S.; Moon, S.-H.; Davis, T. Effects of metal ions on diffusion dialysis of inorganic acids. J. Membr. Sci. 2000, 169, 95–105. [Google Scholar] [CrossRef]
- Luo, J.; Wu, C.; Wu, Y.; Xu, T. Diffusion dialysis of hydrochloride acid at different temperatures using ppo–sio 2 hybrid anion exchange membranes. J. Membr. Sci. 2010, 347, 240–249. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, X.; Li, C.; Feng, H.; Wang, Y.; Luo, J.; Xu, T. A quantification of diffusion dialysis process: Single electrolyte system (sodium chloride solution). Sep. Purif. Technol. 2013, 105, 48–54. [Google Scholar] [CrossRef]
- Zhou, J.; Unlu, M.; Vega, J.A.; Kohl, P.A. Anionic polysulfone ionomers and membranes containing fluorenyl groups for anionic fuel cells. J. Power Sources 2009, 190, 285–292. [Google Scholar] [CrossRef]
- Khan, M.I.; Mondal, A.N.; Cheng, C.; Pan, J.; Emmanuel, K.; Wu, L.; Xu, T. Porous bppo-based membranes modified by aromatic amine for acid recovery. Sep. Purif. Technol. 2016, 157, 27–34. [Google Scholar] [CrossRef]
- Khan, M.; Luque, R.; Akhtar, S.; Shaheen, A.; Mehmood, A.; Idress, S.; Buzdar, A.S. Design of anion exchange membranes and electrodialysis studies for water desalination. Materials 2016, 9, 365. [Google Scholar] [CrossRef]
- Khan, M.I.; Wu, L.; Hossain, M.M.; Pan, J.; Ran, J.; Mondal, A.N.; Xu, T. Preparation of diffusion dialysis membrane for acid recovery via a phase-inversion method. Membr. Water Treat. 2015, 6, 365–378. [Google Scholar] [CrossRef]
- Wu, C.; Wu, Y.; Luo, J.; Xu, T.; Fu, Y. Anion exchange hybrid membranes from pva and multi-alkoxy silicon copolymer tailored for diffusion dialysis process. J. Membr. Sci. 2010, 356, 96–104. [Google Scholar] [CrossRef]
- Li, N.; Guiver, M.D. Ion transport by nanochannels in ion-containing aromatic copolymers. Macromolecules 2014, 47, 2175–2198. [Google Scholar] [CrossRef]
- Li, C.; Sun, G.; Ren, S.; Liu, J.; Wang, Q.; Wu, Z.; Sun, H.; Jin, W. Casting nafion–sulfonated organosilica nano-composite membranes used in direct methanol fuel cells. J. Membr. Sci. 2006, 272, 50–57. [Google Scholar] [CrossRef]
- Spry, D.B.; Goun, A.; Glusac, K.; Moilanen, D.E.; Fayer, M.D. Proton transport and the water environment in nafion fuel cell membranes and aot reverse micelles. J. Am. Chem. Soc. 2007, 129, 8122–8130. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, C.; Wu, Y.; Gu, J.; Xu, T. Polyelectrolyte complex/PVA membranes for diffusion dialysis. J. Hazard. Mater. 2013, 261, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, T.; Singh, A.K.; Shahi, V.K. Zwitterionic silica copolymer based crosslinked organic-inorganic hybrid polymer electrolyte membranes for fuel cell applications. RSC Adv. 2012, 2, 1949–1961. [Google Scholar] [CrossRef]
- Khan, M.I.; Zheng, C.; Mondal, A.N.; Hossain, M.M.; Wu, B.; Emmanuel, K.; Wu, L.; Xu, T. Preparation of anion exchange membranes from bppo and dimethylethanolamine for electrodialysis. Desalination 2017, 402, 10–18. [Google Scholar] [CrossRef]
- Bakangura, E.; Cheng, C.; Wu, L.; He, Y.; Ge, X.; Ran, J.; Emmanuel, K.; Xu, T. Highly charged hierarchically structured porous anion exchange membranes with excellent performance. J. Membr. Sci. 2016, 515, 154–162. [Google Scholar] [CrossRef]
- He, Y.; Pan, J.; Wu, L.; Ge, L.; Xu, T. Facile preparation of 1, 8-diazabicyclo [5.4. 0] undec-7-ene based high performance anion exchange membranes for diffusion dialysis applications. J. Membr. Sci. 2015, 491, 45–52. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, J.; Wu, C.; Xu, T.; Fu, Y. Bionic multisilicon copolymers used as novel cross-linking agents for preparing anion exchange hybrid membranes. J. Phys. Chem. B 2011, 115, 6474–6483. [Google Scholar] [CrossRef] [PubMed]
- Palatý, Z.; Bendová, H. Separation of HCl + FeCl2 mixture by anion-exchange membrane. Sep. Purif. Technol. 2009, 66, 45–50. [Google Scholar] [CrossRef]
- Mondal, A.N.; Cheng, C.; Yao, Z.; Pan, J.; Hossain, M.M.; Khan, M.I.; Yang, Z.; Wu, L.; Xu, T. Novel quaternized aromatic amine based hybrid PVA membranes for acid recovery. J. Membr. Sci. 2015, 490, 29–37. [Google Scholar] [CrossRef]
- Sun, F.; Wu, C.; Wu, Y.; Xu, T. Porous bppo-based membranes modified by multisilicon copolymer for application in diffusion dialysis. J. Membr. Sci. 2014, 450, 103–110. [Google Scholar] [CrossRef]


| Membranes | PI-6 | PI-9 | PI-11 | PI-13 |
|---|---|---|---|---|
| BPPO (g) | 0.4 | 0.4 | 0.4 | 0.4 |
| PI (g) | 0.026 | 0.035 | 0.043 | 0.052 |
| IDT (°C) | 289 | 290 | 294 | 285 |
| Td (°C) | 307 | 308 | 311 | 303 |
| Membranes | PI-6 | PI-9 | PI-11 | PI-13 |
|---|---|---|---|---|
| IEC (mmol/g) | 1.14 | 1.31 | 1.62 | 1.78 |
| WU (%) | 22 | 27 | 36 | 47 |
| LER (%) | 1.2 | 2.0 | 2.3 | 6.1 |
| Thickness (μm) | 17 | 19 | 31 | 20 |
| TS (MPa) | 10.2 | 8.1 | 2.4 | 0.8 |
| PI Content (wt %) | Residual Br− Content (wt %) | |||
|---|---|---|---|---|
| 100 | 60 | 30 | 5 | |
| 6 | 1.3 | 0.8 | 0.4 | 0.1 |
| 9 | 1.9 | 1.1 | 0.6 | 0.1 |
| 11 | 2.3 | 1.4 | 0.7 | 0.1 |
| 13 | 2.6 | 1.6 | 0.8 | 0.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.I.; Luque, R.; Prinsen, P.; Ur Rehman, A.; Anjum, S.; Nawaz, M.; Shaheen, A.; Zafar, S.; Mustaqeem, M. BPPO-Based Anion Exchange Membranes for Acid Recovery via Diffusion Dialysis. Materials 2017, 10, 266. https://doi.org/10.3390/ma10030266
Khan MI, Luque R, Prinsen P, Ur Rehman A, Anjum S, Nawaz M, Shaheen A, Zafar S, Mustaqeem M. BPPO-Based Anion Exchange Membranes for Acid Recovery via Diffusion Dialysis. Materials. 2017; 10(3):266. https://doi.org/10.3390/ma10030266
Chicago/Turabian StyleKhan, Muhammad Imran, Rafael Luque, Pepijn Prinsen, Aziz Ur Rehman, Saima Anjum, Muhammad Nawaz, Aqeela Shaheen, Shagufta Zafar, and Mujahid Mustaqeem. 2017. "BPPO-Based Anion Exchange Membranes for Acid Recovery via Diffusion Dialysis" Materials 10, no. 3: 266. https://doi.org/10.3390/ma10030266
APA StyleKhan, M. I., Luque, R., Prinsen, P., Ur Rehman, A., Anjum, S., Nawaz, M., Shaheen, A., Zafar, S., & Mustaqeem, M. (2017). BPPO-Based Anion Exchange Membranes for Acid Recovery via Diffusion Dialysis. Materials, 10(3), 266. https://doi.org/10.3390/ma10030266








