Bilayer Poly(Lactic-co-glycolic acid)/Nano-Hydroxyapatite Membrane with Barrier Function and Osteogenesis Promotion for Guided Bone Regeneration
Abstract
:1. Introduction
2. Experimental Section
2.1. Fabrication and Characterization of Phase Inversion Membrane
- (1)
- PLGA phase inversion membrane without nHA (denoted PLGA PIM control);
- (2)
- PLGA phase inversion membrane + 5% nHA (denoted PLGA PIM + 5 nHA);
- (3)
- PLGA phase inversion membrane + 10% nHA (denoted PLGA PIM + 10 nHA);
- (4)
- PLGA phase inversion membrane + 20% nHA (denoted PLGA PIM + 20 nHA).
2.2. Barrier Function of Phase Inversion Membrane to Fibroblastic Cells
2.3. Fabrication of Functionally Graded Bilayer Membrane
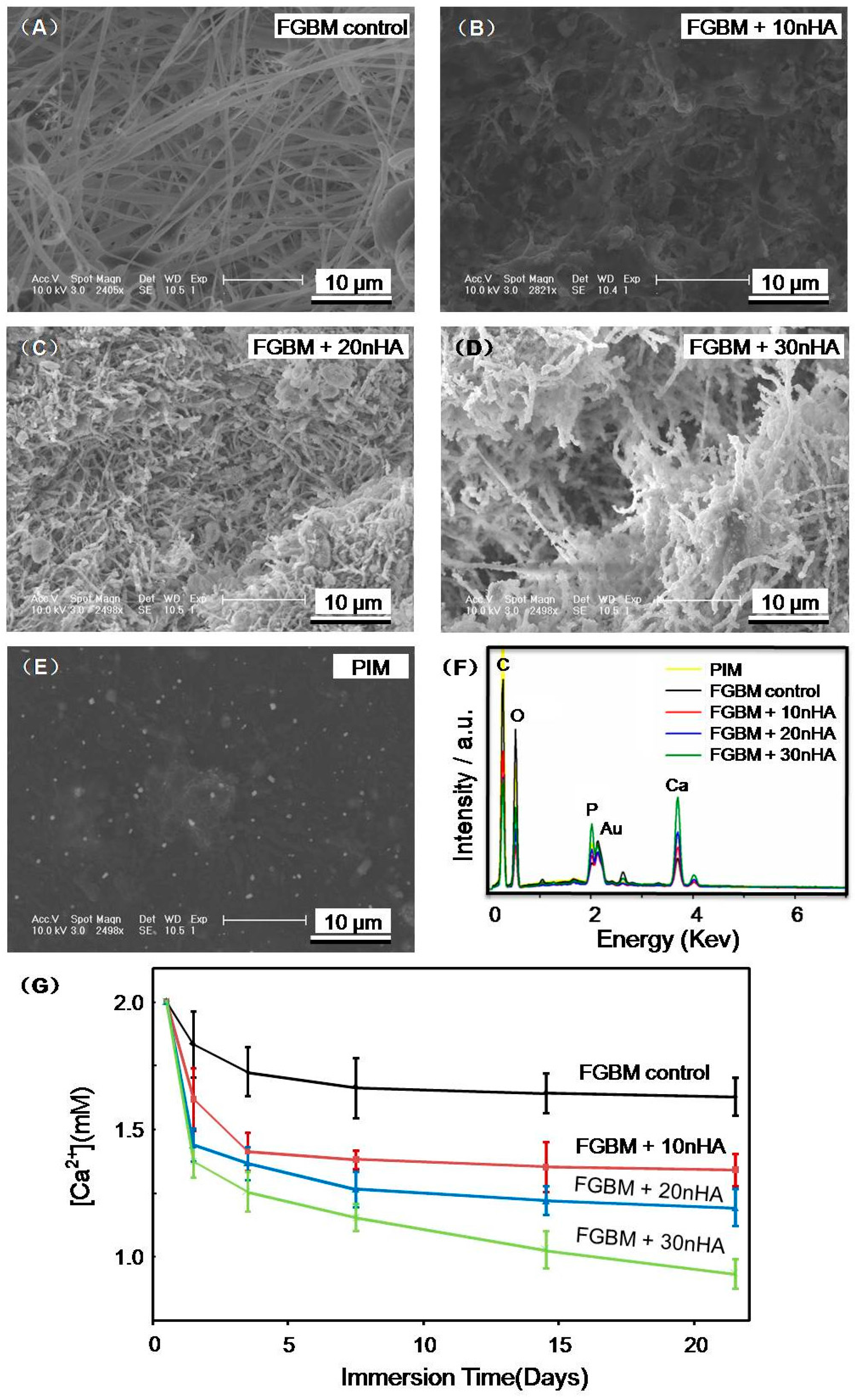
2.4. The Surface Morphology of FGBM
- (1)
- PLGA functionally graded bilayer membrane without nHA (denoted FGBM control);
- (2)
- PLGA functionally graded bilayer membrane + 10% nHA (denoted FGBM + 10 nHA);
- (3)
- PLGA functionally graded bilayer membrane + 20% nHA (denoted FGBM + 20 nHA);
- (4)
- PLGA functionally graded bilayer membrane + 30% nHA (denoted FGBM + 30 nHA);
- (5)
- PLGA functionally graded bilayer membrane + 40% nHA (denoted FGBM + 40 nHA).
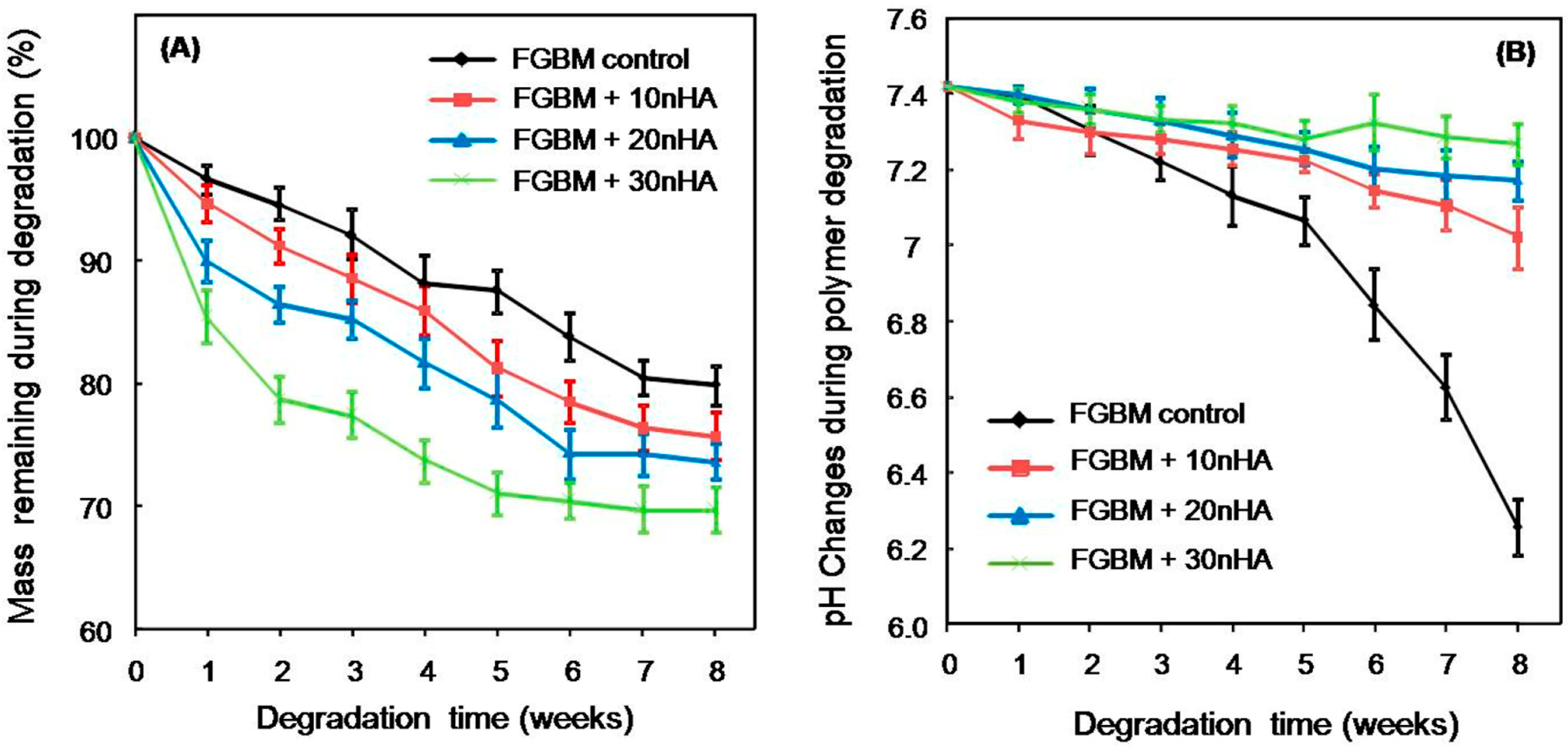
2.5. In Vitro Biodegradation of FGBM
2.6. SBF Immersion Test of FGBM
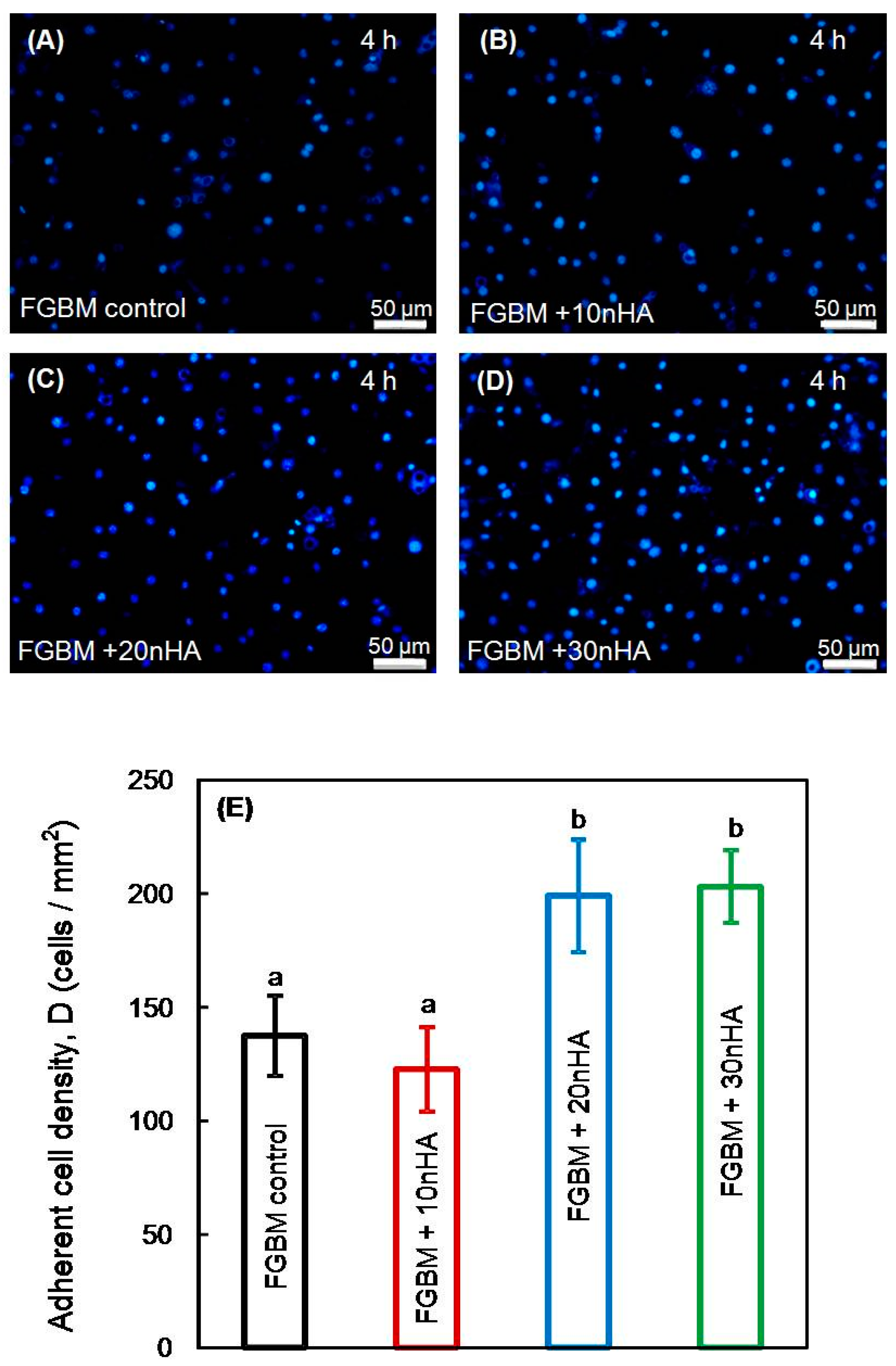
2.7. Cell Adhesion on the FGBM
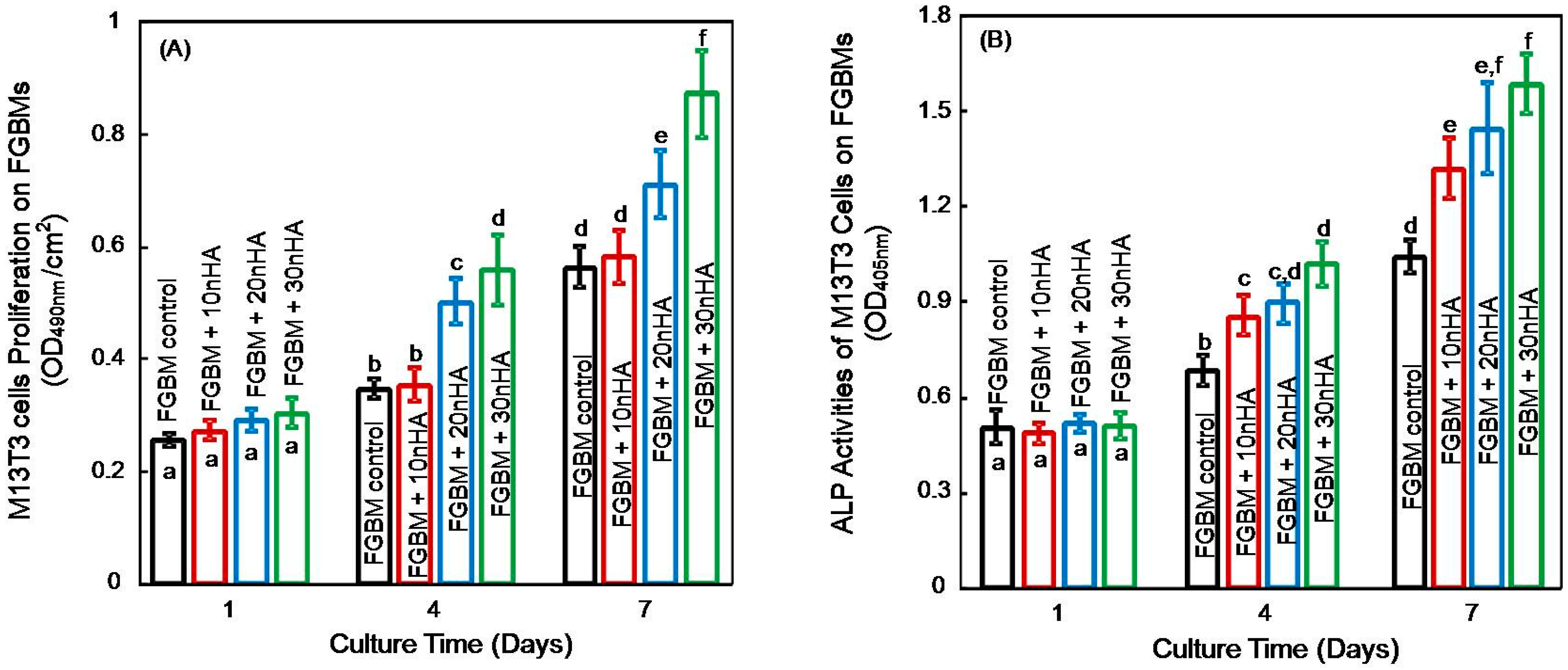
2.8. Cell Proliferation on the FGBM
2.9. Cells Differentiation on the FGBM
2.10. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chiapasco, M.; Casentini, P.; Zaniboni, M. Bone augmentation procedures in implant dentistry. Int. J. Oral Maxillofac. Implants 2009, 24, 237–259. [Google Scholar] [PubMed]
- Sezer, B.; Koyuncu, B.; Günbay, T.; Sezak, M. Alveolar distraction osteogenesis in the human mandible: A clinical and histomorphometric study. Implant Dent. 2012, 21, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Egusa, H.; Sonoyama, W.; Nishimura, M.; Atsuta, I.; Akiyama, K. Stem cells in dentistry-part II: Clinical applications. J. Prosthodont. Res. 2012, 56, 229–248. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.A.; Biagini, G.; Bellardini, M.; Simonelli, L.; Castaldini, C.; Fratto, G. Osteoconduction exerted by methylpyrrolidinone chitosan used in dental surgery. Biomaterials 1993, 14, 39–43. [Google Scholar] [CrossRef]
- Hoshikawa, A.; Fukui, N.; Fukuda, A.; Sawamura, T.; Hattori, M.; Nakamura, K.; Oda, H. Quantitative analysis of the resorption and osteoconduction process of a calcium phosphate cement and its mechanical effect for screw fixation. Biomaterials 2003, 24, 4967–4975. [Google Scholar] [CrossRef]
- Liao, S.; Watari, F.; Zhu, Y.; Akasaka, M.; Uo, T.; Wang, W.; Xu, G.; Cui, F. The degradation of the three layered nano-carbonated hydroxyapatite/collagen/PLGA composite membrane in vitro. Dent. Mater. 2007, 23, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- Isabella, R.; Massimo, S.; Maria, H.; Davide, T.; Marco, B.; Christer, D. Vertical Bone Augmentation with an Autogenous Block or Particles in Combination with Guided Bone Regeneration: A Clinical and Histological Preliminary Study in Humans. Clin. Implant Dent. Relat. Res. 2016, 18, 19–29. [Google Scholar]
- Rakhmatia, Y.D.; Ayukawa, Y.; Furuhashi, A.; Koyano, K. Current barrier membranes: Titanium mesh and other membranes for guided bone regeneration in dental applications. J. Prosthodont. Res. 2013, 57, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Behring, J.; Junker, R.; Walboomers, X.F.; Chessnut, B.; Jansen, J.A. Toward guided tissue and bone regeneration: Morphology, attachment, proliferation, and migration of cells cultured on collagen barrier membranes. A systematic review. Odontology 2008, 96, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Geurs, N.C.; Korostoff, J.M.; Vassilopoulos, P.J.; Kang, T.H.; Jeffcoat, M.; Kellar, R.; Reddy, M.S. Clinical and histologic assessment of lateral alveolar ridge augmentation using a synthetic long-term bioabsorbable membrane and an allograft. J. Periodontol. 2008, 79, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Leal, A.I.; Caridade, S.G.; Ma, J.; Yu, N.; Gomes, M.E.; Reis, R.L.; Jansen, J.A.; Walboomers, X.F.; Mano, J.F. Asymmetric PDLLA membranes containing Bioglass® for guided tissue regeneration: Characterization and in vitro biological behavior. Dent. Mater. 2013, 29, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Thomas, V.; Janowski, G.M. A novel spatially designed and functionally graded electrospun membrane for periodontal regeneration. Acta Biomater. 2011, 7, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Both, S.K.; Yang, X.; Walboomers, X.F.; Jansen, J.A. Development of an electrospun nano-apatite/PCL composite membrane for GTR/GBR application. Acta Biomater. 2009, 5, 3295–3304. [Google Scholar] [CrossRef] [PubMed]
- Watzinger, F.; Luksch, J.; Millesi, W. Guided bone regeneration with titanium membranes: A clinical study. Br. J. Oral Maxillofac. Surg. 2000, 38, 312–315. [Google Scholar] [CrossRef] [PubMed]
- Simion, M.; Jovanovic, S.A.; Trisi, P.; Scarano, A.; Piatelli, A. Vertical ridge augmentation around dental implants using a membrane technique and autogenous bone or allografts in humans. Int. J. Periodontics Restor. Dent. 1998, 18, 9–23. [Google Scholar]
- McGinnis, M.; Larsen, P.; Miloro, M.; Beck, M. Comparison of resorbable and nonresorbable guided bone regeneration materials: A preliminary study. Int. J. Oral Maxillofac. Implants 1998, 13, 30–35. [Google Scholar] [CrossRef]
- Kikuchi, M.; Koyama, Y.; Yamada, T.; Imamura, Y.; Okada, T.; Shirahama, N.; Akita, K.; Takakuda, K.; Tanaka, J. Development of guided bone regeneration membrane composed of beta tricalcium phosphate and poly(l-lactide-co-glycolide-epsilon-caprolactone) composites. Biomaterials 2004, 25, 5979–5986. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Chen, Z.; Qiao, F.; Sun, Y.; Yang, X.; Deng, X.; Cen, L.; Cai, Q.; Wu, M.; Zhang, X.; et al. Guided bone regeneration with tripolyphosphate cross-linked asymmetric chitosan membrane. J. Dent. 2014, 42, 1603–1612. [Google Scholar] [CrossRef] [PubMed]
- Jung, R.E.; Kokovic, V.; Jurisic, M.; Yaman, D.; Subramani, K.; Weber, F.E. Guided bone regeneration with a synthetic biodegradable membrane: A comparative study in dogs. Clin. Oral Implants Res. 2011, 22, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Mano, J.F.; Sousa, R.A.; Boesel, L.F.; Neves, N.M.; Reis, R.L. Bioinert, biodegradable and injectable polymeric matrix composites for hard tissue replacement: State of the art and recent developments. Compos. Sci. Technol. 2004, 64, 789–817. [Google Scholar] [CrossRef]
- Arinzeh, T.L.; Tran, T.; Mcalary, J.; Daculsi, G. A comparative study of biphasic calcium phosphate ceramics for human mesenchymal stem-cell-induced bone formation. Biomaterials 2005, 26, 3631–3638. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, S.; Ding, J.; Li, Z. Tailor-made poly(l-lactide)/poly(lactide-co-glycolide)/hydroxyl apatite composite scaffolds prepared via high-pressure compression molding/salt leaching. RSC Adv. 2016, 6, 47418–47426. [Google Scholar] [CrossRef]
- Deng, X.; Sui, G.; Zhao, M.; Chen, G.; Yang, X. Poly(l-lactic acid)/hydroxyapatite hybrid nanofibrous scaffolds prepared by electrospinning. J. Biomater. Sci. Polym. Ed. 2007, 18, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Thomas, V.; Jagani, S.; Johnson, K.; Jose, M.V.; Dean, D.R.; Vohra, Y.K.; Nyairo, E. Electrospun bioactive nanocomposite scaffolds of polycaprolactone and nano-hydroxyapatite for bone tissue engineering. J. Nanosci. Nanotechnol. 2006, 6, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Wang, W.; Uo, M.; Ohkawa, S.; Akasaka, T.; Tamura, K.; Cui, F.; Watari, F. A three-layered nano-carbonated hydroxyapatite-collagen-PLGA composite membrane for guided tissue regeneration. Biomaterials 2005, 26, 7564–7571. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zuo, Y.; Cheng, X.; Yang, W.; Wang, H.; Li, Y. Preparation and characterization of nano-hydroxyapatite/polyamide 66 composite GBR membrane with asymmetric porous structure. J. Mater. Sci. Mater. Med. 2009, 20, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Fujihara, K.; Kotaki, M.; Ramakrishna, S. Guided bone regeneration membrane made of polycaprolactone/calcium carbonate composite nano-fibers. Biomaterials 2005, 26, 4139–4147. [Google Scholar] [CrossRef] [PubMed]
- Amoroso, N.J.; D’Amore, A.; Hong, Y.; Wagner, W.R.; Sacks, M.S. Elastomeric electrospun polyurethane scaffolds: The interrelationship between fabrication conditions, fiber topology, and mechanical properties. Adv. Mater. 2011, 23, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Reis, R.L.; Neves, N.M. Electrospinning: Processing technique for tissue engineering scaffolding. Int. Mater. Rev. 2008, 53, 257–274. [Google Scholar] [CrossRef]
- Xu, L.; Jiang, L.; Xiong, C.; Jiang, L.; Li, Y. Study on a novel double-layered composite membrane of Mg-substituted nano-hydroxyapatite/poly(l-lactide-co-ε-caprolactone): Effect of different l-lactide/ε-caprolactone ratios. Mater. Sci. Eng. A 2014, 615, 361–366. [Google Scholar]
- Xue, J.; He, M.; Liu, H.; Niu, Y.; Crawford, A.; Coates, P.D.; Chen, D.; Shi, R.; Zhang, L. Drug loaded homogeneous electrospun PCL/gelatin hybrid nanofiber structures for anti-infective tissue regeneration membranes. Biomaterials 2014, 35, 9395–9405. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Ling, F.; Ma, L.; Yang, C.; Chen, X. Electrospun hydroxyapatite grafted poly(l-lactide)/poly(lactic-co-glycolic acid) nanofibers for guided bone regeneration membrane. Compos. Sci. Technol. 2013, 79, 8–14. [Google Scholar] [CrossRef]
- Kokubo, T.; Hanakawa, M.; Kawashita, M.; Minoda, M.; Beppu, T.; Miyamoto, T.; Nakamura, T. Apatite formation on non-woven fabric of carboxymethylated chitin in SBF. Biomaterials 2004, 25, 4485–4488. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, C.; Li, C.; Weir, M.D.; Wang, P.; Reynolds, M.A.; Zhao, L.; Xu, H.H. Injectable calcium phosphate with hydrogel fibers encapsulating induced pluripotent, dental pulp and bone marrow stem cells for bone repair. Mater. Sci. Eng. C 2016, 69, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Song, Y.; Weir, M.D.; Sun, J.; Zhao, L.; Simon, C.G.; Xu, H.H. A self-setting iPSMSC-alginate-calcium phosphate paste for bone tissue engineering. Dent. Mater. 2016, 32, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Mauney, J.R.; Blumberg, J.; Pirun, M.; Volloch, V.; Vunjak-Novakovic, G.; Kaplan, D.L. Osteogenic differentiation of human bone marrow stromal cells on partially demineralized bone scaffolds in vitro. Tissue Eng. 2004, 10, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Zhu, C.; Yang, J.; Sun, H.; Zhang, X.; Li, S.; Wang, Y.; Sun, L.; Yao, F. Electrospun PDLLA/PLGA composite membranes for potential application in guided tissue regeneration. Mater. Sci. Eng. C 2016, 58, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Liu, X.; Zhao, S.; Wu, H.; Xu, H.H. Porous chitosan bilayer membrane containing TGF-β1 loaded microspheres for pulp capping and reparative dentin formation in a dog model. Dent. Mater. 2014, 30, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Mi, F.L.; Wu, Y.B.; Shyu, S.S.; Chao, A.C.; Lai, J.Y.; Su, C.C. Asymmetric chitosan membranes prepared by dry/wet phase separation: A new type of wound dressing for controlled antibacterial release. J. Membr. Sci. 2003, 212, 237–254. [Google Scholar] [CrossRef]
- Jose, M.V.; Thomas, V.; Johnson, K.T.; Dean, D.R.; Nyairo, E. Aligned PLGA/HA nanofibrous nanocomposite scaffolds for bone tissue engineering. Acta Biomater. 2009, 5, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Kuo, S.M.; Chang, S.J.; Chen, T.W.; Kuan, T.C. Guided tissue regeneration for using a chitosan membrane: An experimental study in rats. J. Biomed. Mater. Res. A 2006, 76, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Iglhaut, J.; Aukhil, I.; Simpson, D.M.; Johnston, M.C.; Koch, G. Progenitor cell kinetics during guided tissue regeneration in experimental periodontal wounds. J. Periodontal Res. 1988, 23, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L. Principles in guided bone regeneration. Dent. Implantol. Update 1998, 9, 33–37. [Google Scholar] [PubMed]
- Pham, Q.P.; Sharma, U.; Mikos, A.G. Electrospunpoly(epsilon-caprolactone) microfiber and multilayer nanofiber/microfiber scaffolds: Characterization of scaffolds and measurement of cellular infiltration. Biomacromolecules 2006, 7, 2796–2805. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.A.; Mikos, A.G.; Kasper, F.K. Scaffold/extracellular matrix hybrid constructs for bone-tissue engineering. Adv. Healthc. Mater. 2013, 2, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Kasuga, T.; Hosoi, Y.; Nogami, M.; Niinomi, M. Apatite formation on calcium phosphate invert glasses in simulated body fluid. J. Am. Ceram. Soc. 2001, 84, 450–452. [Google Scholar] [CrossRef]
- Wong, K.L.; Wong, C.T.; Liu, W.C.; Pan, H.B.; Fong, M.K.; Lam, W.M.; Cheung, W.L.; Tang, W.M.; Chiu, K.Y.; Luk, K.D.K. Mechanical properties and in vitro response of strontium-containing hydroxyapatite/polyetheretherketone composites. Biomaterials 2009, 30, 3810–3817. [Google Scholar] [CrossRef] [PubMed]
- Wenstrup, R.J.; Fowlkes, J.L.; Witte, D.P.; Florer, J.B. Discordant expression of osteoblast markers in MC3T3-E1 cells that synthesize a high tunoverr matrix. J. Biol. Chem. 1996, 271, 10271–10276. [Google Scholar] [PubMed]
- Thomas, V.; Dean, D.R.; Jose, M.V.; Mathew, B.; Chowdhury, S.; Vohra, Y.K. Nanostructured biocomposite scaffolds based on collagen coelectrospun with nanohydroxyapatite. Biomacromolecules 2007, 8, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.I.; Lee, A.Y.; Lee, Y.M.; Shin, H. Electrospun gelatin/poly(l-lactide-co-epsiloncaprolactone) nanofibers for mechanically functional tissue-engineering scaffolds. J. Biomater. Sci. Polym. Ed. 2008, 19, 339–357. [Google Scholar] [CrossRef] [PubMed]
- Kilpadi, K.L.; Chang, P.L.; Bellis, S.L. Hydroxyapatite binds more serum proteins, purified integrins, and osteoblast precursor cells than titanium or steel. J. Biomed. Mater. Res. 2001, 57, 258–267. [Google Scholar] [CrossRef]
- Tsigko, O.; Jones, J.R.; Polak, J.M.; Stevens, M.M. Differentiation of fetal osteoblasts and formation of mineralized nodules by 45S5 bioglass conditioned medium in the absence of osteogenic supplements. Biomaterials 2009, 30, 3542–3550. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.; Andersson, A.S.; Sutherland, D.; Petronis, S.; Kasemo, B.; Thomsen, P. Response of rat osteoblast-like cells to microstructured model surfaces in vitro. Biomaterials 2003, 24, 649–654. [Google Scholar] [CrossRef]
- Jaiswal, A.K.; Kadam, S.S.; Soni, V.P.; Bellare, J.R. Improved functionalization of electrospun PLLA/gelatin scaffold by alternate soaking method for bone tissue engineering. Appl. Surf. Sci. 2013, 268, 477–488. [Google Scholar] [CrossRef]





| Types of PIM | Tensile Strength (MPa) |
|---|---|
| PLGA PIM control | 2.90 (0.31) a |
| PLGA PIM + 5 nHA | 3.55 (0.45) b |
| PLGA PIM + 10 nHA | 2.82 (0.32) a |
| PLGA PIM + 20 nHA | 2.03 (0.11) a |
| Types of FGBM | Tensile Strength (MPa) |
|---|---|
| FGBM control | 3.80 (0.59) a |
| FGBM + 10 nHA | 3.59 (0.44) a |
| FGBM + 20 nHA | 3.35 (0.30) a,b |
| FGBM + 30 nHA | 3.18 (0.28) a,b |
| FGBM + 40 nHA | 3.09 (0.32) b |
| Commercial control Bio-Gide | 3.06 (0.24) b |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, L.; Wang, Z.; Dong, S.; Cai, Y.; Ni, Y.; Zhang, T.; Wang, L.; Zhou, Y. Bilayer Poly(Lactic-co-glycolic acid)/Nano-Hydroxyapatite Membrane with Barrier Function and Osteogenesis Promotion for Guided Bone Regeneration. Materials 2017, 10, 257. https://doi.org/10.3390/ma10030257
Fu L, Wang Z, Dong S, Cai Y, Ni Y, Zhang T, Wang L, Zhou Y. Bilayer Poly(Lactic-co-glycolic acid)/Nano-Hydroxyapatite Membrane with Barrier Function and Osteogenesis Promotion for Guided Bone Regeneration. Materials. 2017; 10(3):257. https://doi.org/10.3390/ma10030257
Chicago/Turabian StyleFu, Li, Zhanfeng Wang, Shujun Dong, Yan Cai, Yuxin Ni, Tianshou Zhang, Lin Wang, and Yanmin Zhou. 2017. "Bilayer Poly(Lactic-co-glycolic acid)/Nano-Hydroxyapatite Membrane with Barrier Function and Osteogenesis Promotion for Guided Bone Regeneration" Materials 10, no. 3: 257. https://doi.org/10.3390/ma10030257
APA StyleFu, L., Wang, Z., Dong, S., Cai, Y., Ni, Y., Zhang, T., Wang, L., & Zhou, Y. (2017). Bilayer Poly(Lactic-co-glycolic acid)/Nano-Hydroxyapatite Membrane with Barrier Function and Osteogenesis Promotion for Guided Bone Regeneration. Materials, 10(3), 257. https://doi.org/10.3390/ma10030257





