Fabrication of Lead-Free Bi0.5Na0.5TiO3 Thin Films by Aqueous Chemical Solution Deposition
Abstract
:1. Introduction
2. Results
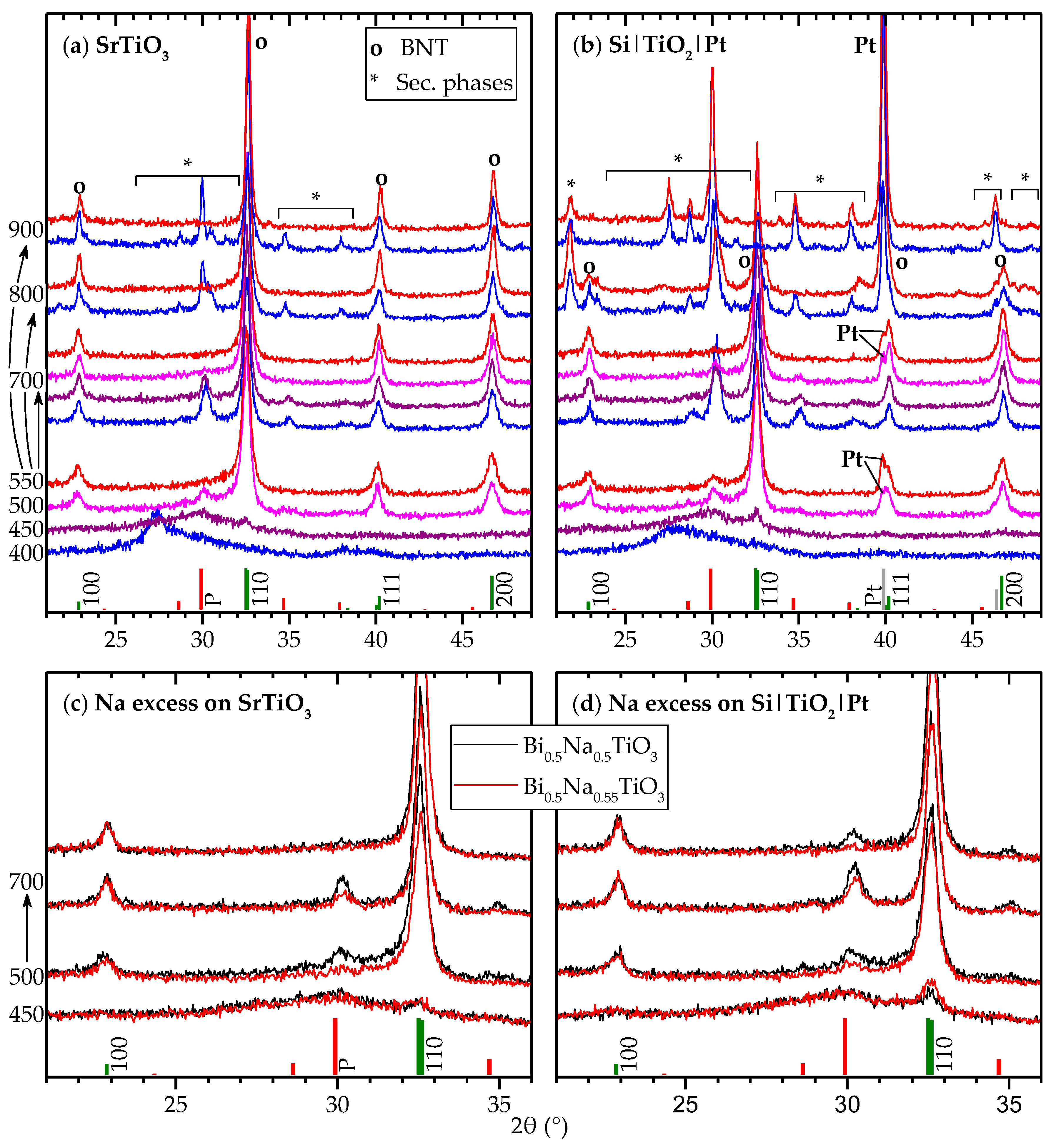
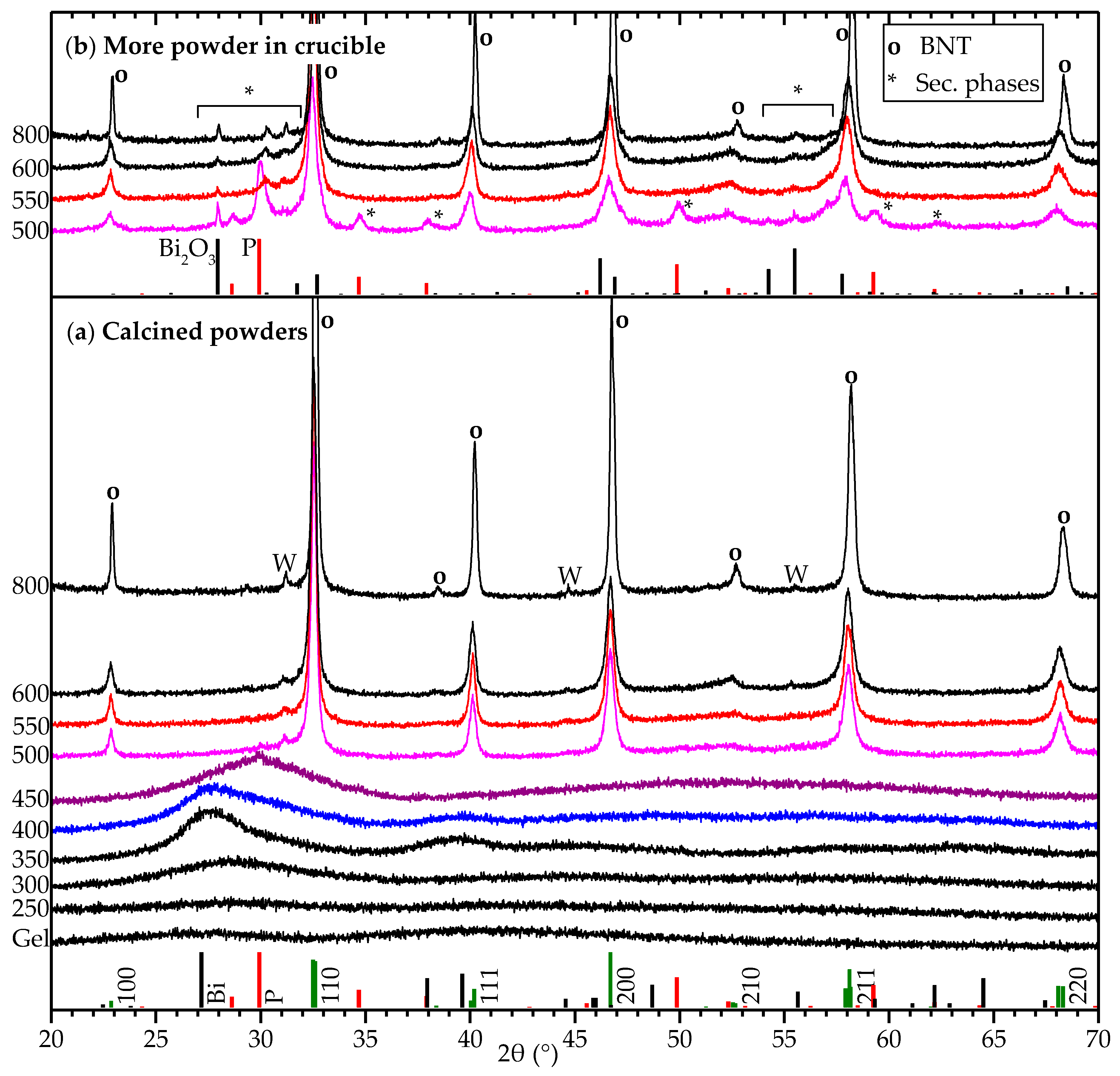
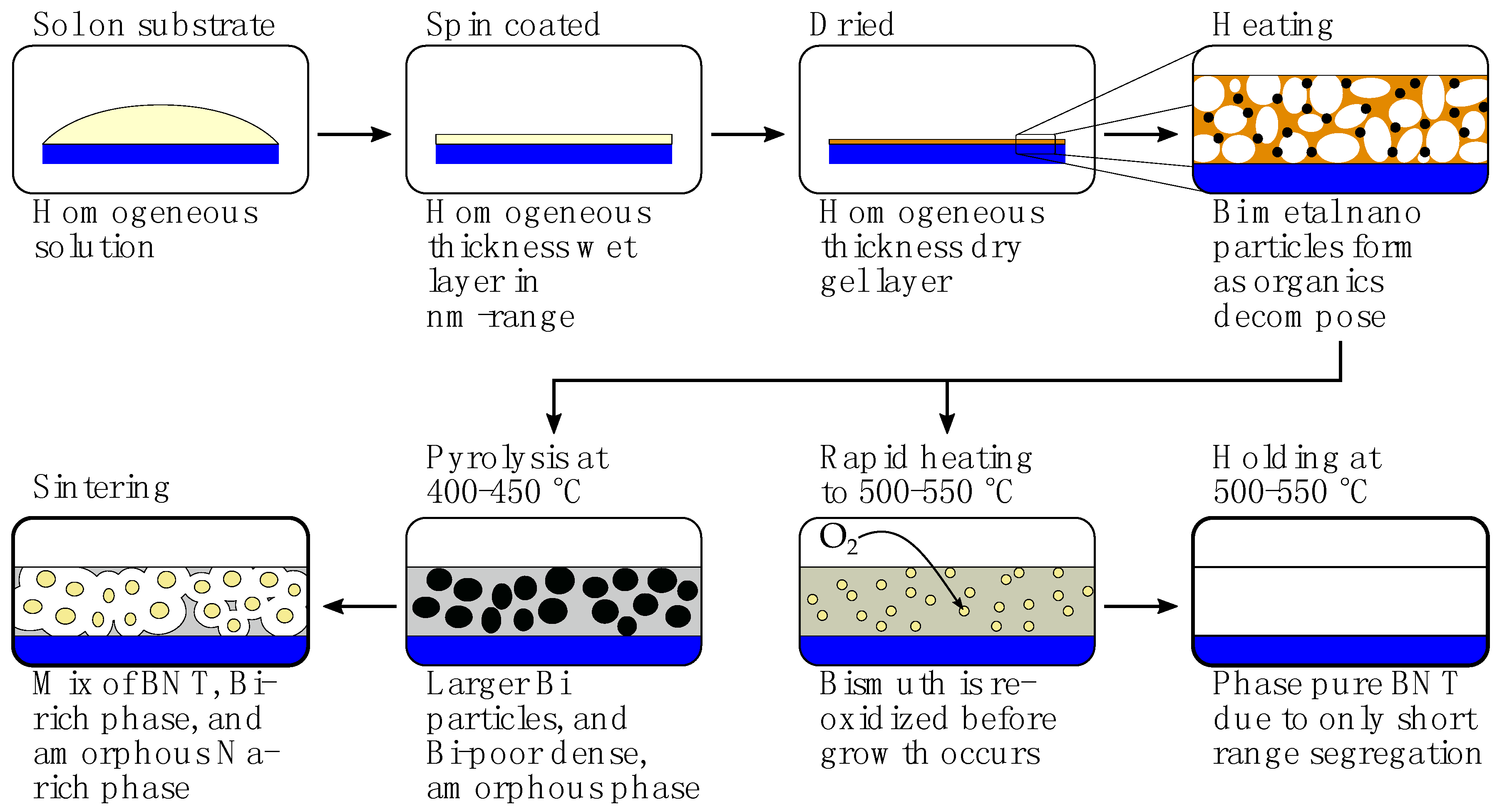
2.1. Deposition of Thin Film Using the Citrate Sol
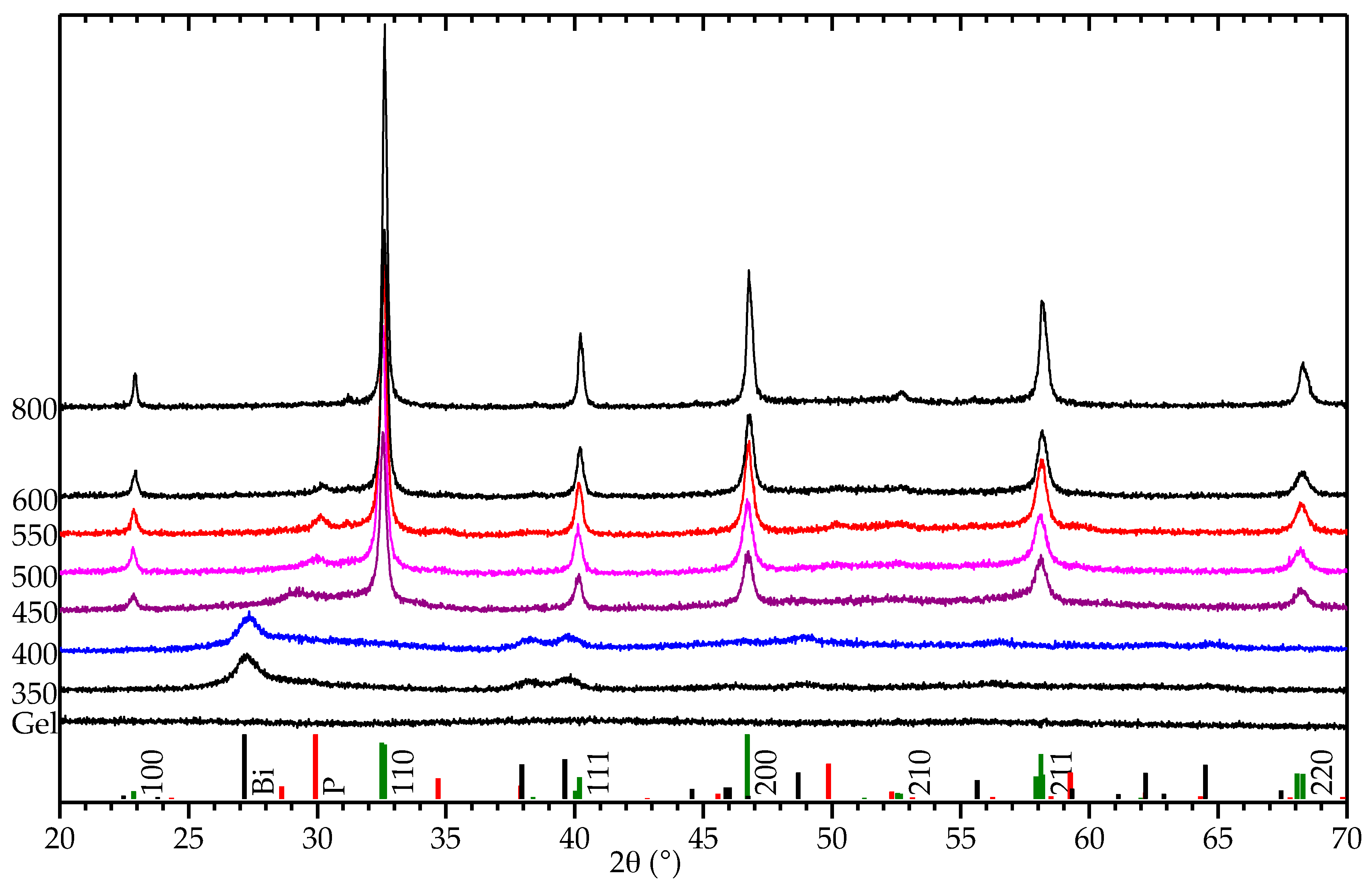
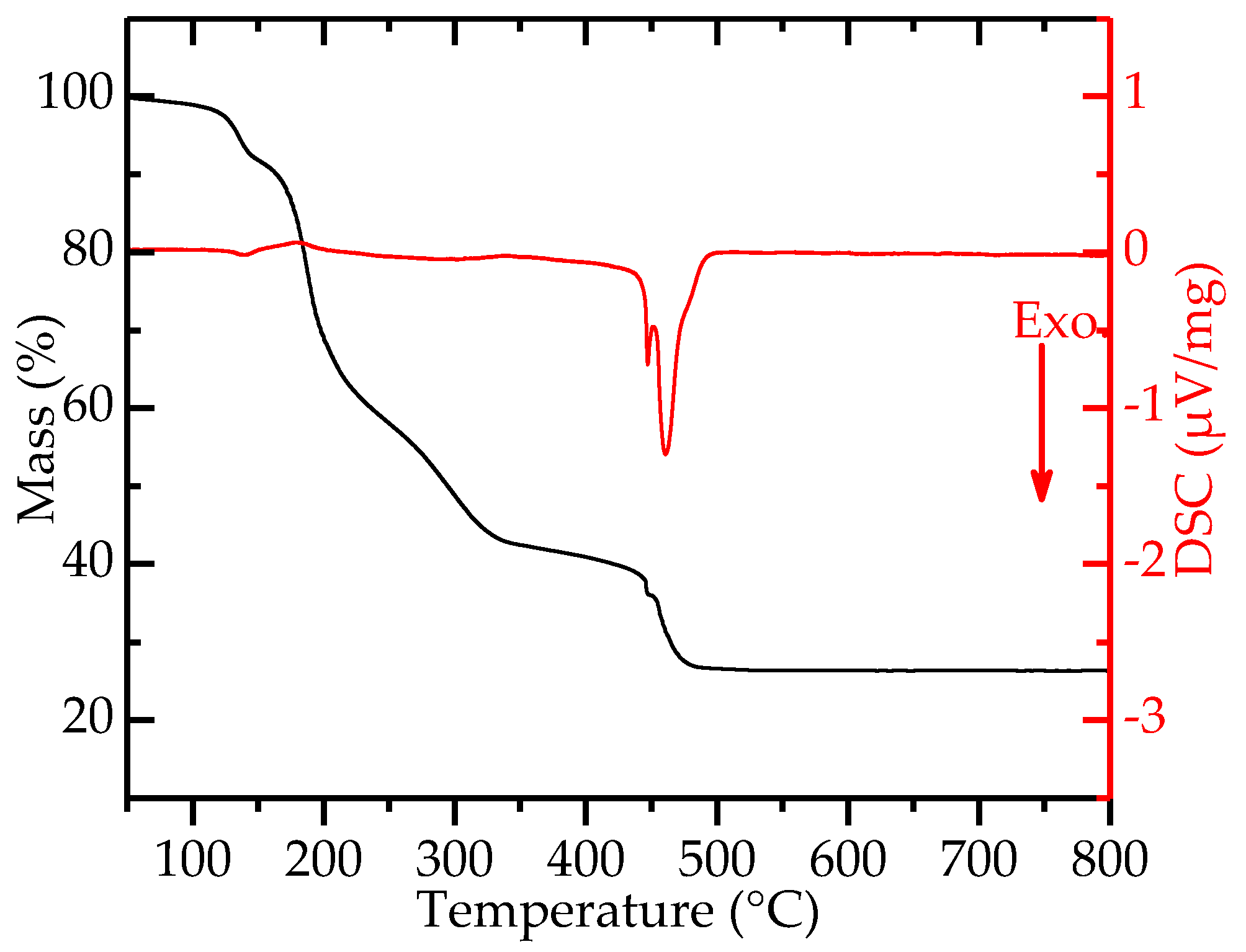
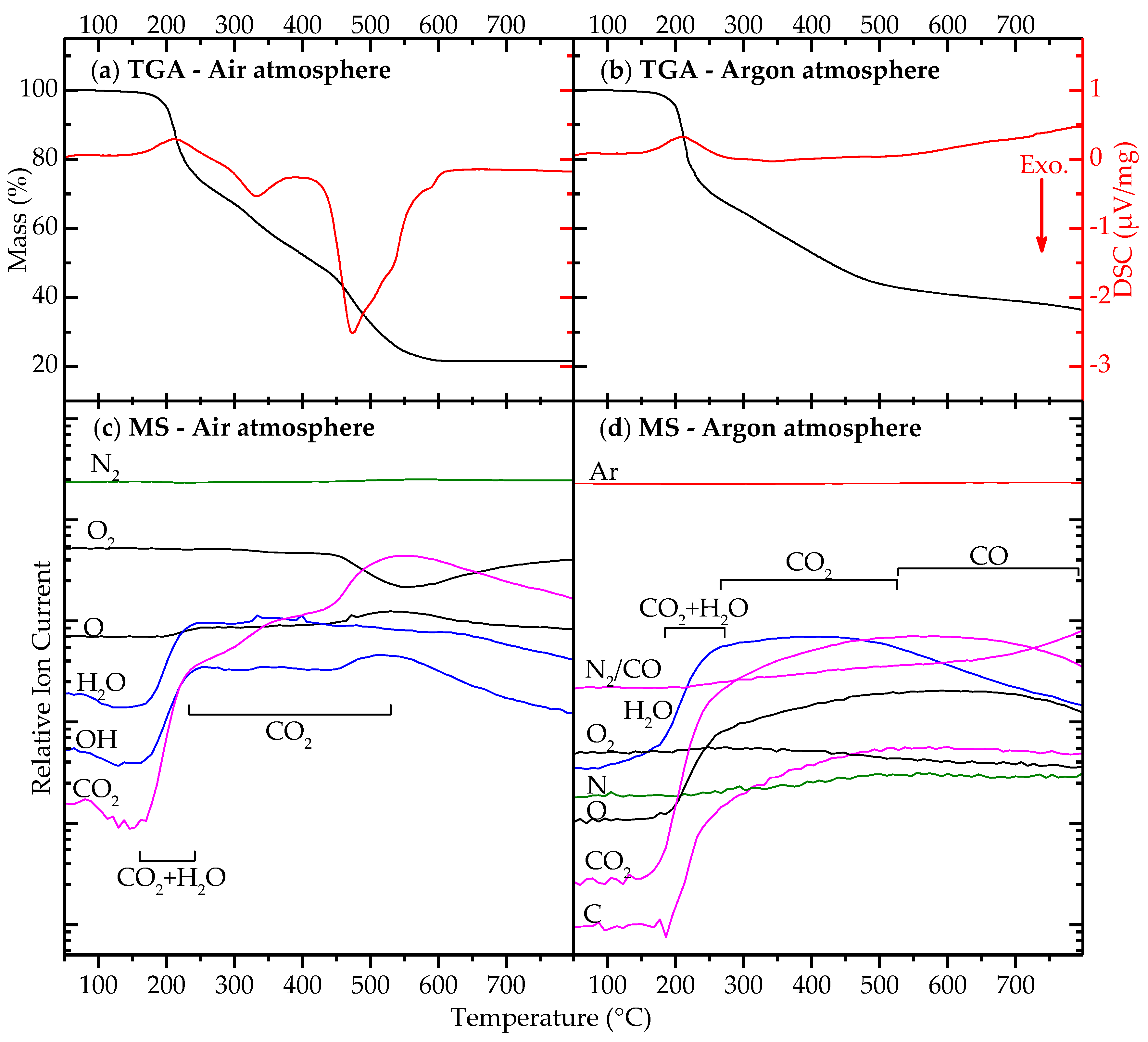
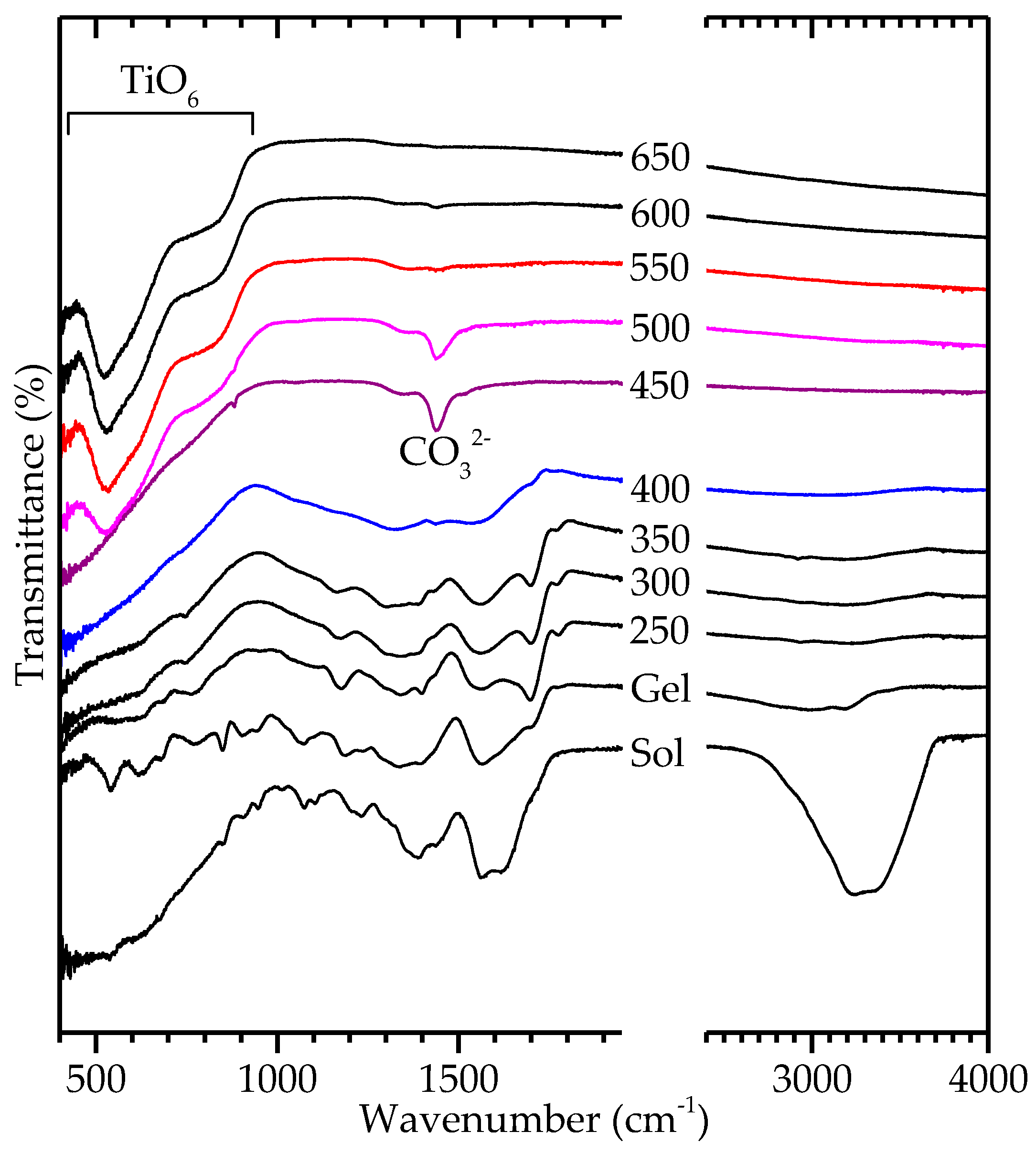
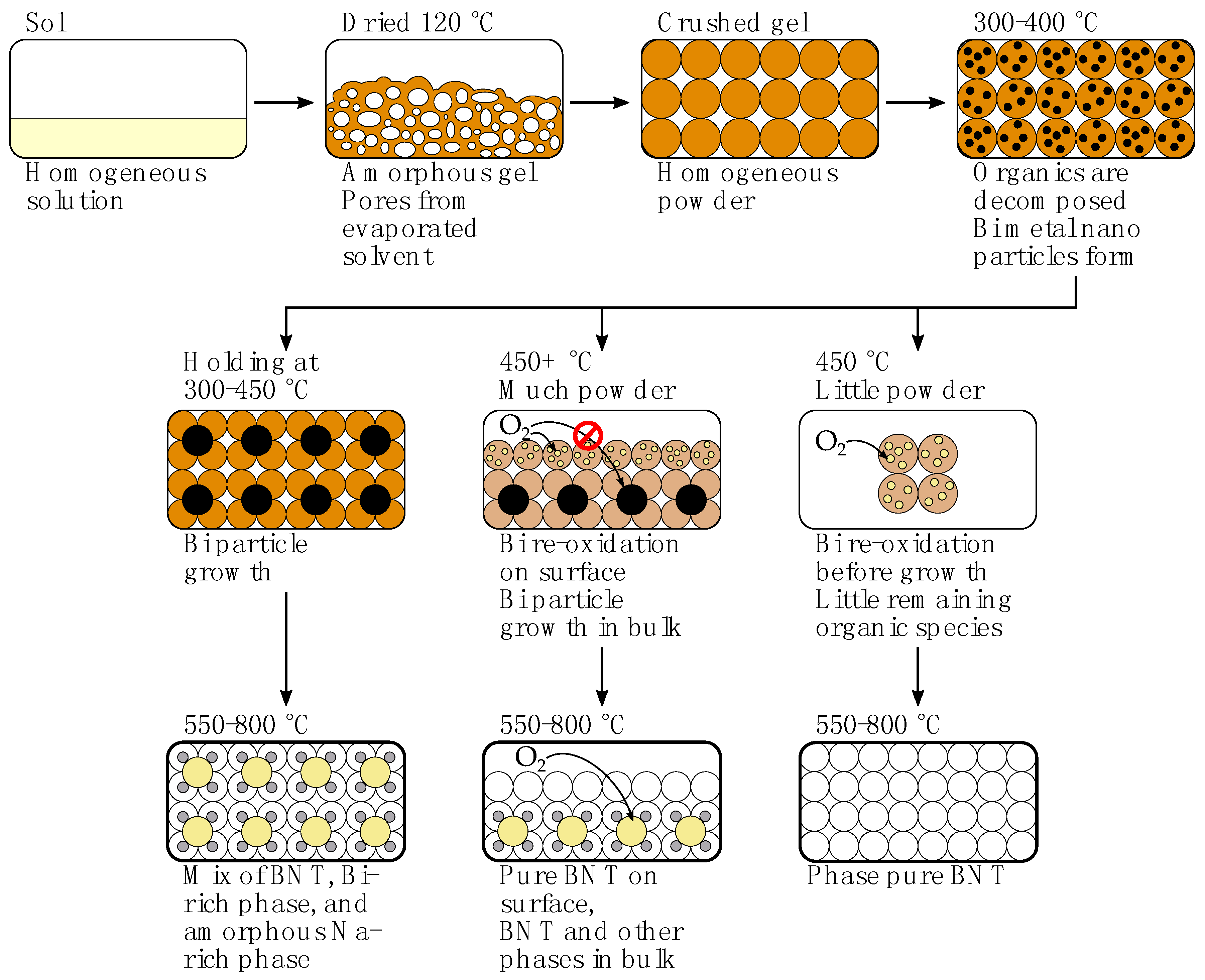
2.2. Decomposition of Gel from Citrate Sol
3. Discussion
4. Materials and Methods
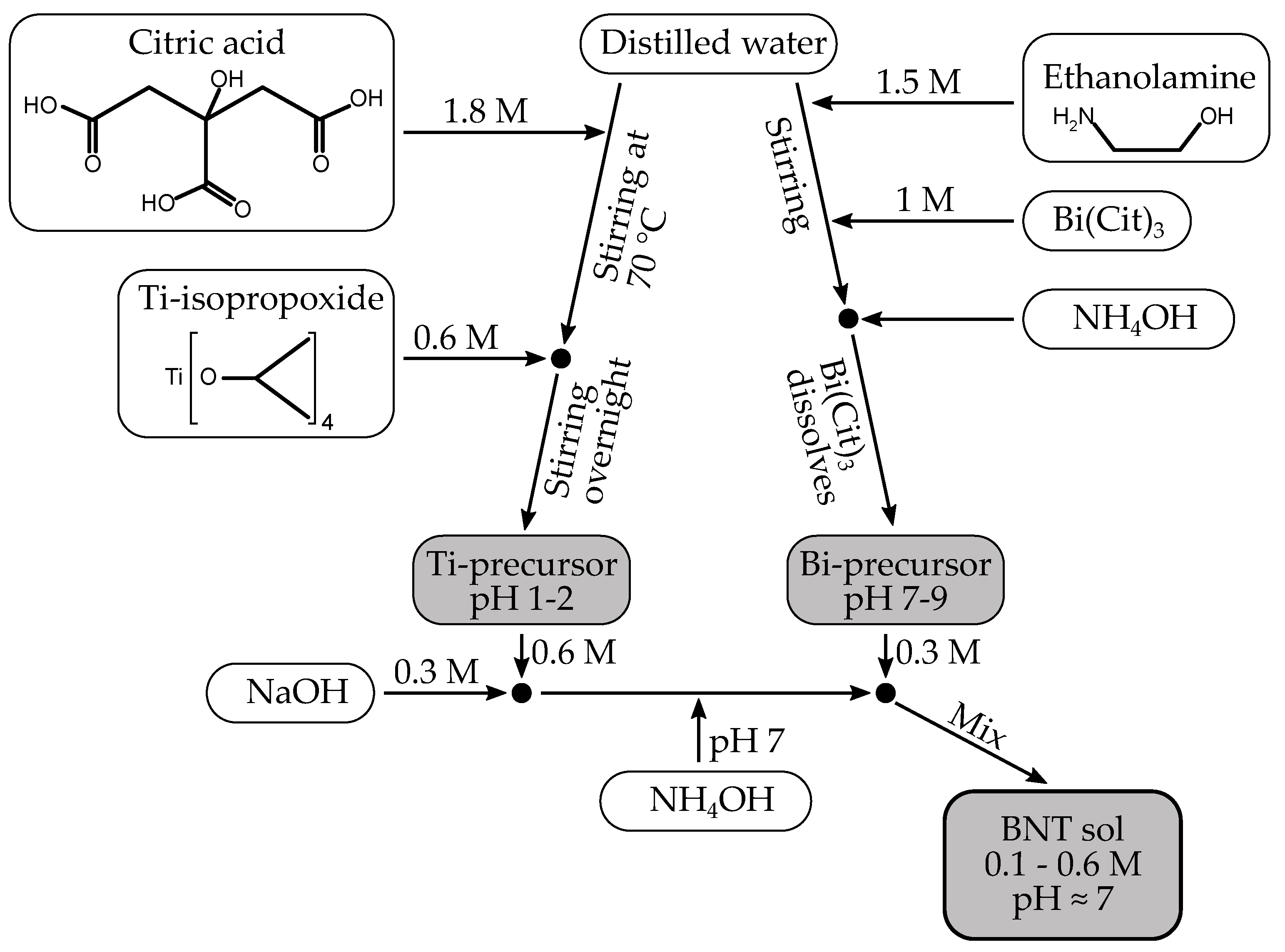
4.1. Sol Preparation
4.2. Powder and Film Preparation
4.3. Characterization
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A. The Nitrate Method
Appendix A.1. Synthesis by the Nitrate Method
Appendix A.2. Stability of the Sols
Appendix B. Rietveld Refinements
| Powders | Films on STO | Films on SiPt | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Heat Treatment (°C) | a (Å) | c (Å) | Cryst. Size (nm) | Heat Treatment (°C) | a (Å) | c (Å) | Cryst. Size (nm) | Heat Treatment (°C) | a (Å) | c (Å) | Cryst. Size (nm) |
| Reference: R3c [30] | 5.49 | 13.50 | Stoichiometric | Stoichiometric | |||||||
| 450 | 5.50 | 13.52 | 13 | 450 | 5.50 | 13.49 | 8 | ||||
| Citrate | 500 | 5.51 | 13.50 | 10 | 500 | 5.50 | 13.48 | 10 | |||
| 500 Little * | 5.50 | 13.51 | 20 | 550 * | 5.51 | 13.49 | 11 | 550 | 5.49 | 13.51 | 13 |
| 550 Little * | 5.50 | 13.51 | 23 | 400 + 700 | 5.50 | 13.49 | 16 | 400 + 700 | 5.49 | 13.44 | 19 |
| 600 Little * | 5.50 | 13.52 | 22 | 450 + 700 | 5.50 | 13.48 | 17 | 450 + 700 | 5.49 | 13.45 | 16 |
| 800 Little * | 5.49 | 13.48 | 52 | 500 + 700 * | 5.50 | 13.47 | 16 | 500 + 700 | 5.49 | 13.45 | 16 |
| 500 much | 5.51 | 13.49 | 12 | 550 + 700 * | 5.49 | 13.51 | 19 | 550 + 700 * | 5.49 | 13.49 | 16 |
| 550 Much | 5.50 | 13.52 | 17 | 400 + 800 | 5.49 | 13.47 | 18 | 400 + 800 | 5.48 | 13.43 | 13 |
| 600 Much | 5.50 | 13.51 | 18 | 550 + 800 * | 5.49 | 13.49 | 22 | 550 + 800 | 5.48 | 13.46 | 15 |
| 800 Much | 5.48 | 13.46 | 54 | 400 + 900 | 5.49 | 13.44 | 22 | 400 + 900 | No BNT | ||
| Nitrate | 550 + 900 * | 5.49 | 13.41 | 30 | 550 + 900 | ||||||
| 450 | 5.49 | 13.50 | 22 | Na excess | Na excess | ||||||
| 500 | 5.49 | 13.51 | 21 | 450 | 5.52 | 13.51 | 6 | 450 | 5.50 | 13.48 | 8 |
| 550 | 5.49 | 13.50 | 26 | 500 * | 5.50 | 13.51 | 13 | 500 | 5.49 | 13.51 | 13 |
| 600 | 5.49 | 13.49 | 28 | 450 + 700 | 5.49 | 13.51 | 20 | 450 + 700 | 5.48 | 13.50 | 18 |
| 800 * | 5.49 | 13.47 | 44 | 500 + 700 * | 5.49 | 13.50 | 19 | 500 + 700 * | 5.48 | 13.49 | 20 |
References
- Jo, W.; Dittmer, R.; Acosta, M.; Zang, J.D.; Groh, C.; Sapper, E.; Wang, K.; Rödel, J. Giant electric-field-induced strains in lead-free ceramics for actuator applications—Status and perspective. J. Electroceram. 2012, 29, 71–93. [Google Scholar] [CrossRef]
- Rödel, J.; Jo, W.; Seifert, K.T.P.; Anton, E.-M.; Granzow, T.; Damjanovic, D. Perspective on the development of lead-free piezoceramics. J. Am. Ceram. Soc. 2009, 92, 1153–1177. [Google Scholar] [CrossRef]
- Aksel, E.; Jones, J.L. Advances in lead-free piezoelectric materials for sensors and actuators. Sensors 2010, 10, 1935–1954. [Google Scholar] [CrossRef] [PubMed]
- Rodel, J.; Webber, K.G.; Dittmer, R.; Jo, W.; Kimura, M.; Damjanovic, D. Transferring lead-free piezoelectric ceramics into application. J. Eur. Ceram. Soc. 2015, 35, 1659–1681. [Google Scholar] [CrossRef]
- Shrout, T.R.; Zhang, S.J. Lead-free piezoelectric ceramics: Alternatives for PZT? J. Electroceram. 2007, 19, 113–126. [Google Scholar] [CrossRef]
- Takenaka, T.; Nagata, H. Current status and prospects of lead-free piezoelectric ceramics. J. Eur. Ceram. Soc. 2005, 25, 2693–2700. [Google Scholar] [CrossRef]
- Takenaka, T.; Nagata, H.; Hiruma, Y. Current developments and prospective of lead-free piezoelectric ceramics. Jpn. J. Appl. Phys. 2008, 47, 3787–3801. [Google Scholar] [CrossRef]
- Schneider, D.; Roedel, J.; Rytz, D.; Granzow, T. Orientation-dependence of thermal depolarization and phase development in Bi1/2Na1/2TiO3-BaTiO3 single crystals. J. Am. Ceram. Soc. 2015, 98, 3966–3974. [Google Scholar] [CrossRef]
- Reichmann, K.; Feteira, A.; Li, M. Bismuth sodium titanate based materials for piezoelectric actuators. Materials 2015, 8, 8467–8495. [Google Scholar] [CrossRef]
- Schneller, T.; Griesche, D.; Malic, B.; Glinsek, S.; Kosec, M.; Kessler, V.G.; van Bael, M.K.; Hardy, A.; Mullens, J.; Burrell, A.K. Chemical Solution Deposition of Functional Oxide Thin Films; Schneller, T., Waser, R., Kosec, M., Payne, D., Eds.; Springer: Wien, Austria, 2013. [Google Scholar]
- Bassiri-Gharb, N.; Bastani, Y.; Bernal, A. Chemical solution growth of ferroelectric oxide thin films and nanostructures. Chem. Soc. Rev. 2014, 43, 2125–2140. [Google Scholar] [CrossRef] [PubMed]
- Brennecka, G.L.; Ihlefeld, J.F.; Maria, J.-P.; Tuttle, B.A.; Clem, P.G. Processing technologies for high-permittivity thin films in capacitor applications. J. Am. Ceram. Soc. 2010, 93, 3935–3954. [Google Scholar] [CrossRef]
- Schwartz, R.W.; Schneller, T.; Waser, R. Chemical solution deposition of electronic oxide films. C. R. Chim. 2004, 7, 433–461. [Google Scholar] [CrossRef]
- Kupec, A.; Malic, B. Structural and dielectric properties of the lead-free (1 − x)K0.5Na0.5NbO3−xSrTiO3 thin films from solutions. J. Alloys Compd. 2014, 596, 32–38. [Google Scholar] [CrossRef]
- Lemoine, C.; Gilbert, B.; Michaux, B.; Pirard, J.-P.; Lecloux, A. Synthesis of barium titanate by the sol-gel process. J. Non-Cryst. Solids 1994, 175, 1–13. [Google Scholar] [CrossRef]
- Li, P.; Li, W.; Zeng, H.; Liu, S.; Wang, W.; Zhai, J. Ferroelectric and piezoelectric properties of La-modified lead-free (Bi0.5Na0.5)TiO3–(Bi0.5K0.5)TiO3–SrTiO3 thin films. Ceram. Int. 2015, 41, 4479–4486. [Google Scholar] [CrossRef]
- Zhou, Z.; Sun, W.; Li, J.-F. Sol-gel synthesis of 0.94(Bi0.5Na0.5)TiO3–0.06BaTiO3 lead-free piezoelectric films: Effect of pyrolysis temperature on phase evolution and electrical properties. J. Sol-Gel Sci. Technol. 2015, 77, 423–429. [Google Scholar] [CrossRef]
- Ojha, K.S. Structural optimization of bismuth sodium titanate thin films. Ferroelectrics 2015, 474, 163–168. [Google Scholar] [CrossRef]
- Peng, J.F.; Zheng, X.J.; Gong, Y.Q.; Zhan, K.; Dai, Z.H. Effects of annealing temperature on the electric properties of 0.94(Na0.5Bi0.5)TiO3–0.06BaTiO3 ferroelectric thin film. J. Electron. Mater. 2013, 43, 724–731. [Google Scholar] [CrossRef]
- Remondiere, F.; Malic, B.; Kosec, M.; Mercurio, J.P. Study of the crystallization pathway of Na0.5Bi0.5TiO3 thin films obtained by chemical solution deposition. J. Sol-Gel Sci. Technol. 2008, 46, 117–125. [Google Scholar] [CrossRef]
- Sakamoto, W.; Makino, N.; Katayama, T.; Moriya, M.; Yogo, T. Improvement of the ferroelectric properties of chemically synthesized Bi1/2Na1/2TiO3 thin films via Mn doping. Ferroelectrics 2015, 479, 56–63. [Google Scholar] [CrossRef]
- Sui, H.; Yang, C.; Geng, F.; Feng, C. Effects of Zr doping content on microstructure, ferroelectric and dielectric properties of Na0.5Bi0.5TiO3 thin film. Mater. Lett. 2015, 139, 284–287. [Google Scholar] [CrossRef]
- West, D.L.; Payne, D.A. Preparation of 0.95Bi1/2Na1/2TiO3·0.05BaTiO3 ceramics by an aqueous citrate-gel route. J. Am. Ceram. Soc. 2003, 86, 192–194. [Google Scholar] [CrossRef]
- Chen, M.; Xu, Q.; Kim, B.H.; Ahn, B.K.; Ko, J.H.; Kang, W.J.; Nam, O.J. Structure and electrical properties of (Na0.5Bi0.5)1−xBaxTiO3 piezoelectric ceramics. J. Eur. Ceram. Soc. 2008, 28, 843–849. [Google Scholar] [CrossRef]
- Xu, Q.; Chen, S.; Chen, W.; Wu, S.; Zhou, J.; Sun, H.; Li, Y. Synthesis and piezoelectric and ferroelectric properties of (Na0.5Bi0.5)1−xBaxTiO3 ceramics. Mater. Chem. Phys. 2005, 90, 111–115. [Google Scholar] [CrossRef]
- Xu, Q.; Chen, X.-L.; Chen, W.; Chen, M.; Xu, S.-L.; Kim, B.-H.; Lee, J.-H. Effect of MnO addition on structure and electrical properties of (Na0.5Bi0.5)0.94Ba0.06TiO3 ceramics prepared by citrate method. Mater. Sci. Eng. B 2006, 130, 94–100. [Google Scholar] [CrossRef]
- Xu, Q.; Chen, X.-L.; Chen, W.; Kim, B.-H.; Xu, S.-L.; Chen, M. Structure and electrical properties of (Na0.5Bi0.5)1−xBaxTiO3 ceramics made by a citrate method. J. Electroceram. 2007, 21, 617–620. [Google Scholar] [CrossRef]
- Hardy, A.; Mondelaers, D.; van Bael, M.K.; Mullens, J.; van Poucke, L.C.; Vanhoyland, G.; D’Haen, J. Synthesis of (Bi,La)4Ti3O12 by a new aqueous solution-gel route. J. Eur. Ceram. Soc. 2004, 24, 905–909. [Google Scholar] [CrossRef]
- Haugen, A.B.; Kumakiri, I.; Simon, C.; Einarsrud, M.-A. TiO2, TiO2/Ag and TiO2/Au photocatalysts prepared by spray pyrolysis. J. Eur. Ceram. Soc. 2011, 31, 291–298. [Google Scholar] [CrossRef]
- Jones, G.O.; Thomas, P.A. Investigation of the structure and phase transitions in the novel A-site substituted distorted perovskite compound Na0.5Bi0.5TiO3. Acta Crystallogr. Sect. B Struct. Sci. 2002, 58, 168–178. [Google Scholar] [CrossRef]
- Dong, Q.; Jiang, H.; Kumada, N.; Yonesaki, Y.; Takei, T.; Kinomura, N. Preparation of a new pyrochlore-type compound Na0.32Bi1.68Ti2O6.46(OH)0.44 by hydrothermal reaction. J. Solid State Chem. 2011, 184, 1899–1902. [Google Scholar] [CrossRef]
- Geguzina, G.A.; Shuvayev, A.T.; Vlasenko, V.G.; Shuvayeva, E.T.; Shilkina, L.A. Synthesis and structure of new Am−1Bi2BmO3m+3 (m = 3) phases. Crystallogr. Rep. 2003, 48, 406–412. [Google Scholar] [CrossRef]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies; John Wiley & Sons Ltd.: Chichester, UK, 2001. [Google Scholar]
- Samajdar, S.; Becker, F.F.; Banik, B.K. Surface-mediated highly efficient oxidation of alcohols by bismuth itrate. Synth. Commun. 2001, 31, 2691–2695. [Google Scholar] [CrossRef]
- Nelis, D.; van Werde, K.; Mondelaers, D.; Vanhoyland, G.; van den Rul, H.; van Bael, M.K.; Mullens, J.; van Poucke, L.C. Aqueous solution-gel synthesis of strontium bismuth niobate (SrBi2Nb2O9). J. Sol-Gel Sci. Technol. 2003, 26, 1125–1129. [Google Scholar] [CrossRef]
- Li, W.; Zeng, H.; Zhao, K.; Hao, J.; Zhai, J. Structural, dielectric and piezoelectric properties of (Bi0.5Na0.5)TiO3-(Bi0.5K0.5)TiO3-Bi(Zn0.5Ti0.5)O3 thin films prepared by sol-gel method. Ceram. Int. 2014, 40, 7947–7951. [Google Scholar] [CrossRef]
- Alonso-Sanjose, D.; Jimenez, R.; Bretos, I.; Calzada, M.L. Lead-free ferroelectric (Na1/2Bi1/2)TiO3-BaTiO3 thin films in the morphotropic phase boundary composition: Solution processing and properties. J. Am. Ceram. Soc. 2009, 92, 2218–2225. [Google Scholar] [CrossRef]
- Li, M.; Zhang, H.; Cook, S.N.; Li, L.; Kilner, J.A.; Reaney, I.M.; Sinclair, D.C. Dramatic influence of A-site nonstoichiometry on the electrical conductivity and conduction mechanisms in the perovskite oxide Na0.5Bi0.5TiO3. Chem. Mater. 2015, 27, 629–634. [Google Scholar] [CrossRef]
- Li, M.; Pietrowski, M.J.; de Souza, R.A.; Zhang, H.; Reaney, I.M.; Cook, S.N.; Kilner, J.A.; Sinclair, D.C. A family of oxide ion conductors based on the ferroelectric perovskite Na0.5Bi0.5TiO3. Nat. Mater. 2014, 13, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Prasertpalichat, S.; Cann, D.P. Hardening in non-stoichiometric (1 − x)Bi0.5Na0.5TiO3−xBaTiO3 lead-free piezoelectric ceramics. J. Mater. Sci. 2016, 51, 476–486. [Google Scholar] [CrossRef]
- Okamoto, H. The Bi-Pt (bismuth-platinum) system. J. Phase Equilib. 1991, 12, 207–210. [Google Scholar] [CrossRef]
- Kim, K.; Jang, G.; Kim, C.; Yoon, D. Ferroelectric properties of Bi3.25Nd0.75Ti3O12 thin films prepared by MOD process. J. Electroceram. 2004, 13, 77–81. [Google Scholar] [CrossRef]
- Villafuerte-Castrejón, M.; Morán, E.; Reyes-Montero, A.; Vivar-Ocampo, R.; Peña-Jiménez, J.-A.; Rea-López, S.-O.; Pardo, L. Towards lead-free piezoceramics: Facing a synthesis challenge. Materials 2016, 9, 21. [Google Scholar] [CrossRef]
- Jo, W.; Granzow, T.; Aulbach, E.; Rodel, J.; Damjanovic, D. Origin of the large strain response in (K0.5Na0.5)NbO3-modified (Bi0.5Na0.5)TiO3-BaTiO3 lead-free piezoceramics. J. Appl. Phys. 2009, 105. [Google Scholar] [CrossRef]
- Deng, Y.-F.; Jiang, Y.-Q.; Hong, Q.-M.; Zhou, Z.-H. Speciation of water-soluble titanium citrate: Synthesis, structural, spectroscopic properties and biological relevance. Polyhedron 2007, 26, 1561–1569. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christensen, M.; Einarsrud, M.-A.; Grande, T. Fabrication of Lead-Free Bi0.5Na0.5TiO3 Thin Films by Aqueous Chemical Solution Deposition. Materials 2017, 10, 213. https://doi.org/10.3390/ma10020213
Christensen M, Einarsrud M-A, Grande T. Fabrication of Lead-Free Bi0.5Na0.5TiO3 Thin Films by Aqueous Chemical Solution Deposition. Materials. 2017; 10(2):213. https://doi.org/10.3390/ma10020213
Chicago/Turabian StyleChristensen, Mads, Mari-Ann Einarsrud, and Tor Grande. 2017. "Fabrication of Lead-Free Bi0.5Na0.5TiO3 Thin Films by Aqueous Chemical Solution Deposition" Materials 10, no. 2: 213. https://doi.org/10.3390/ma10020213
APA StyleChristensen, M., Einarsrud, M.-A., & Grande, T. (2017). Fabrication of Lead-Free Bi0.5Na0.5TiO3 Thin Films by Aqueous Chemical Solution Deposition. Materials, 10(2), 213. https://doi.org/10.3390/ma10020213






