A Bio-Electro-Fenton System Employing the Composite FePc/CNT/SS316 Cathode
Abstract
:1. Introduction
Fe2+ + H2O2 → Fe3+ + ·HO + OH−,
2. Materials and Methods
2.1. Electrode Production
2.2. The Fabricaiton Process of Composite FePc/CNT/SS316 Cathode
2.3. Construction of Bio-Electro-Fenton Microbial Fuel Cell System
2.4. Measurement of Electrical Quantities
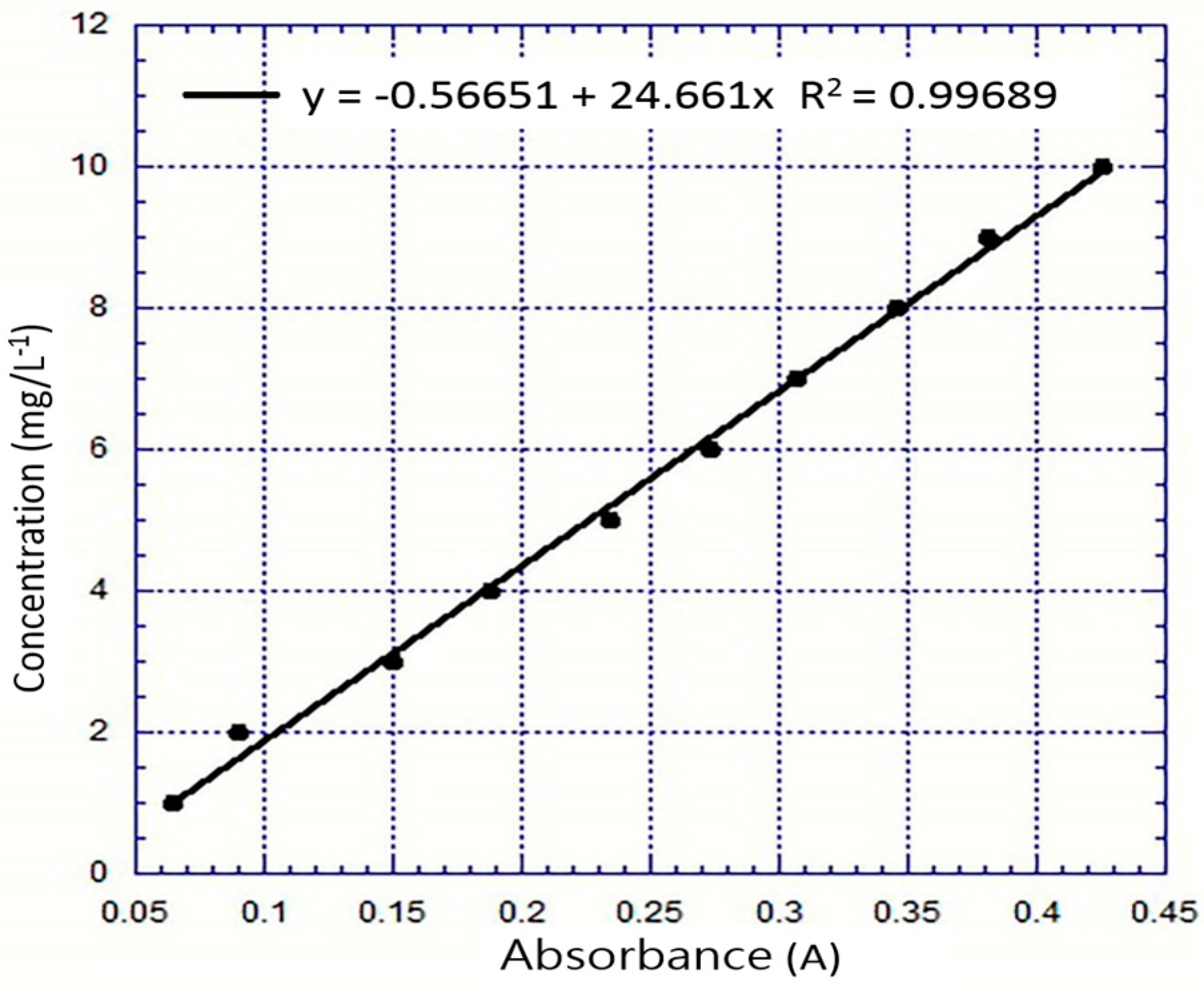
2.5. Preparation for Dye Decolorization
3. Results and Discussion
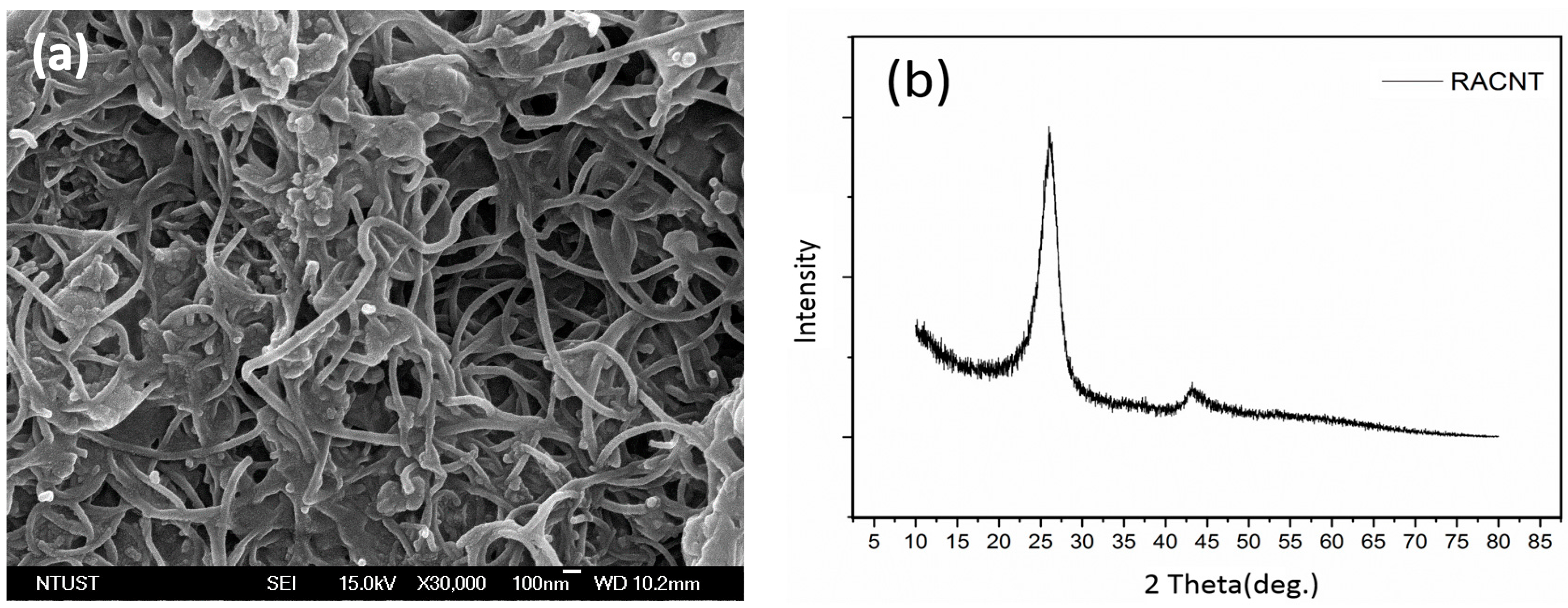
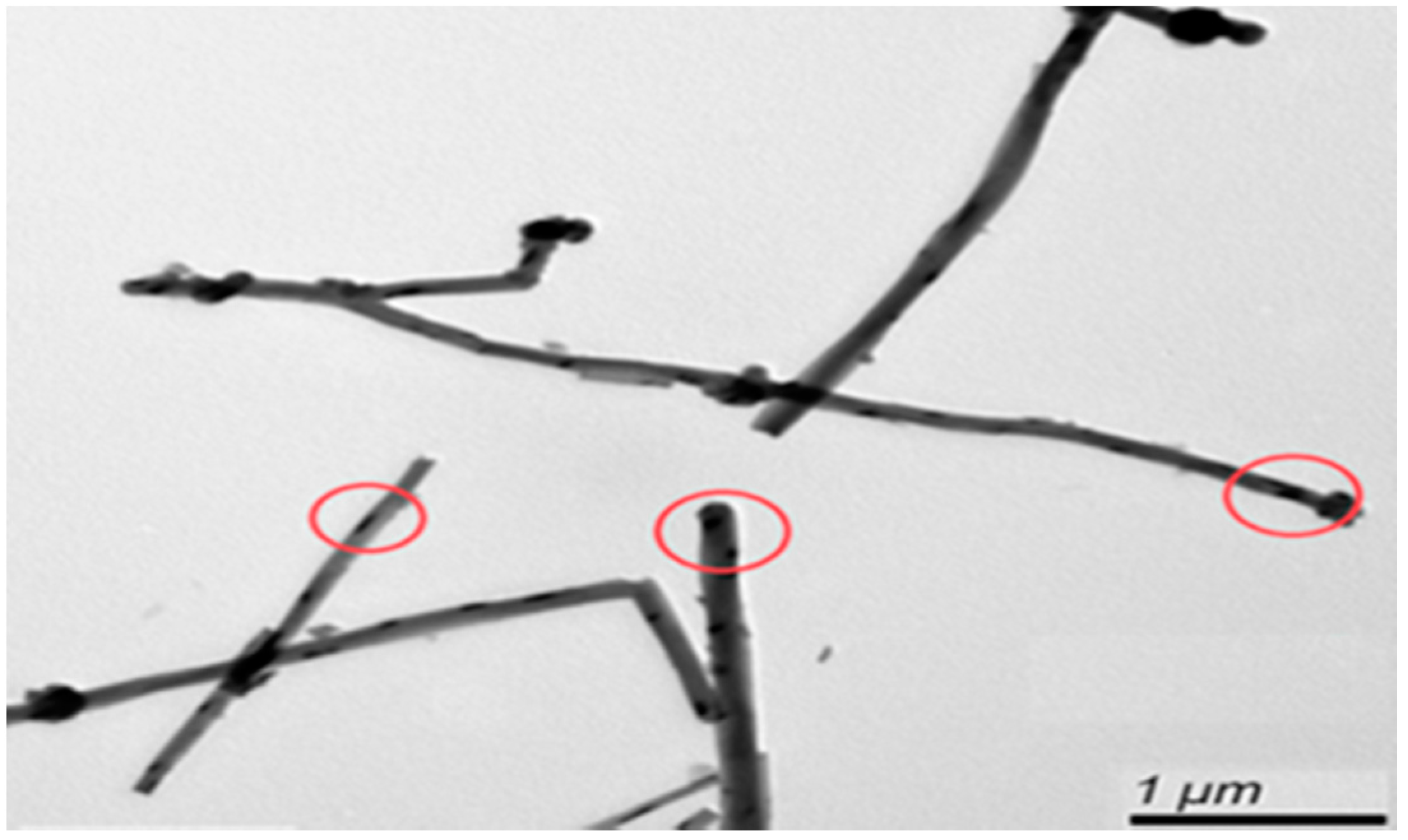
3.1. Morphology of CNT Formation
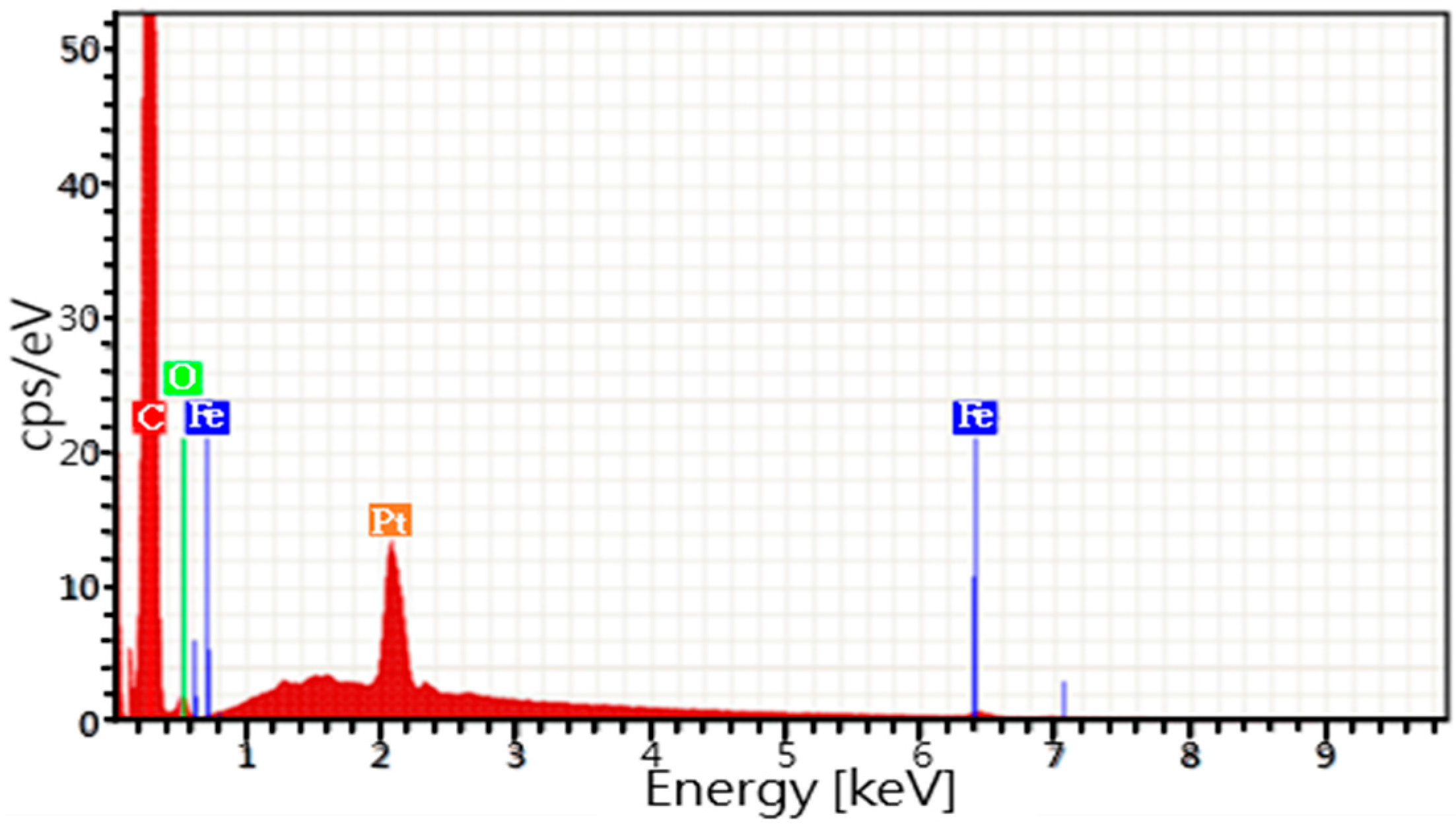
3.2. Analyses of Iron Phthalocyanine Component
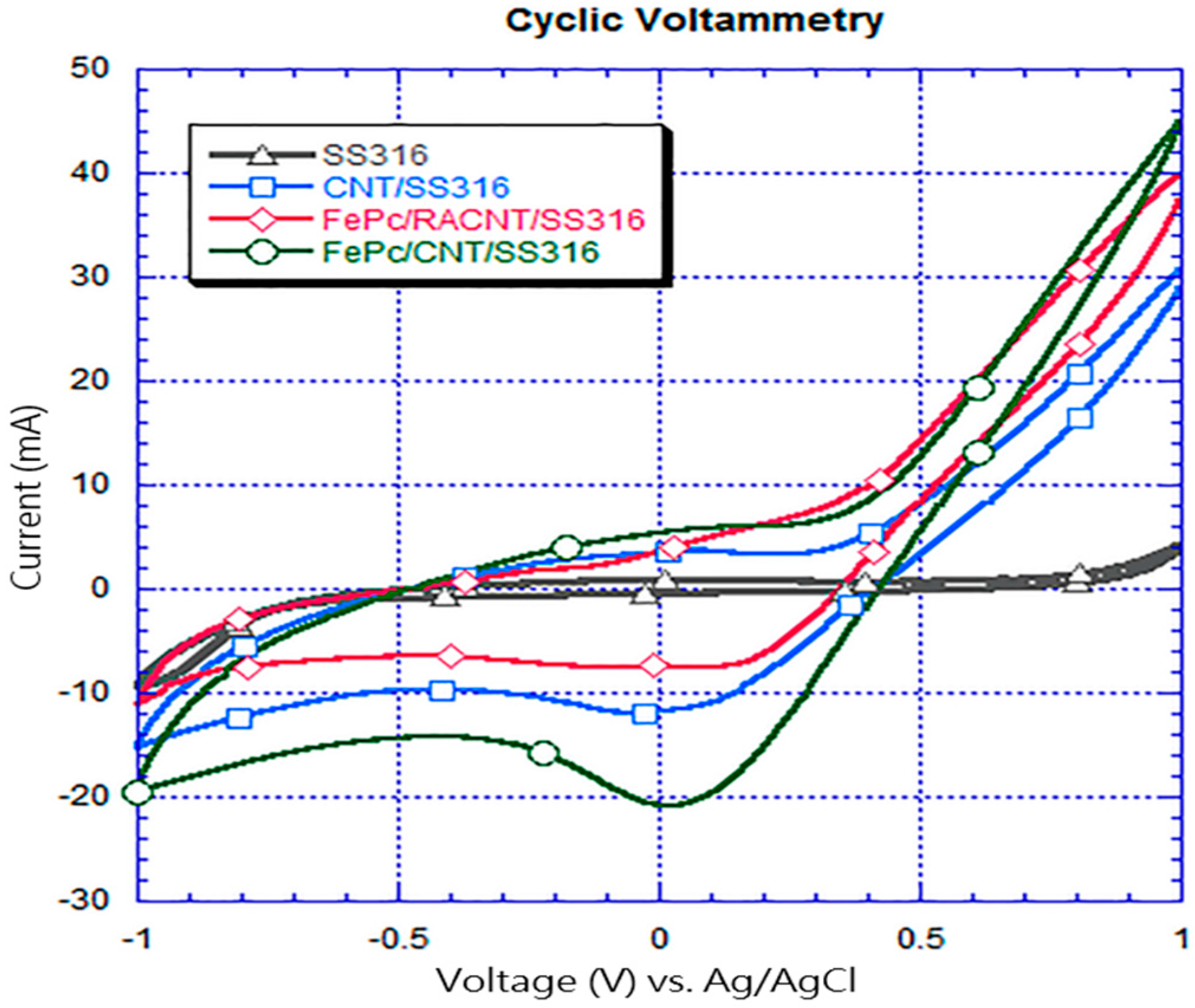
3.3. Cyclic Voltammetry Analyses
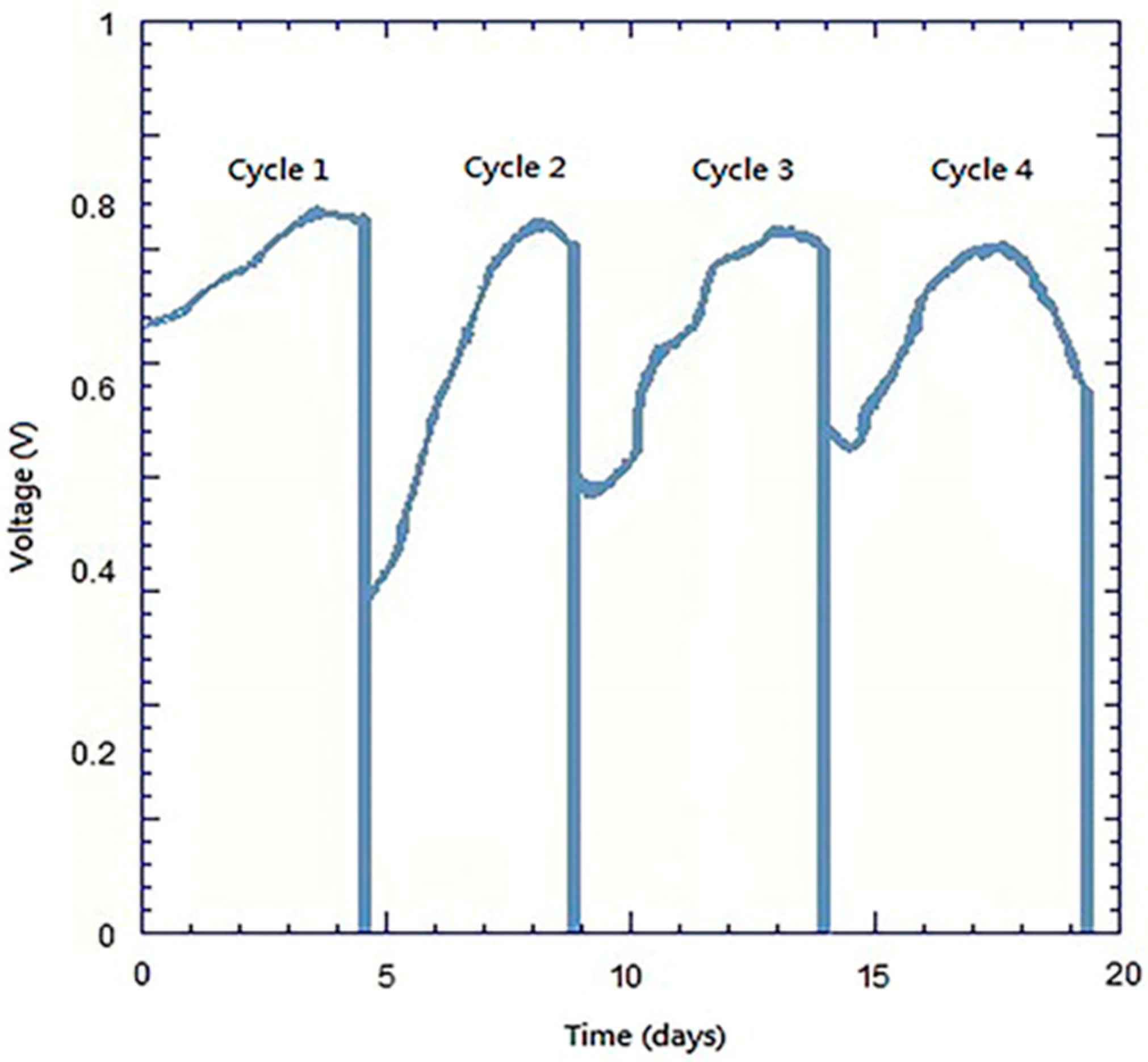
3.4. Analyses of Discharge with Constant Resistance
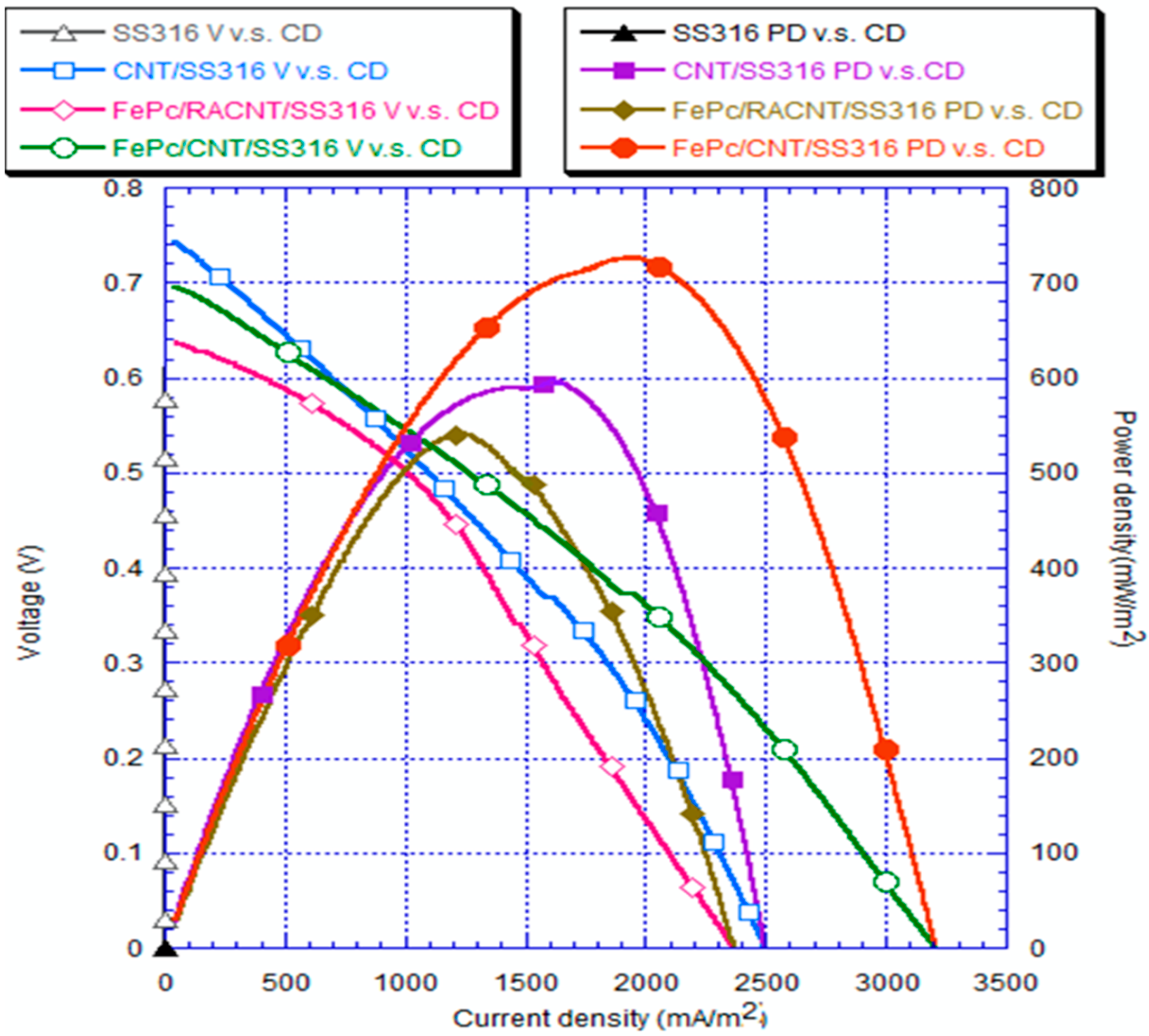
3.5. Analysis of Electrical Quantities
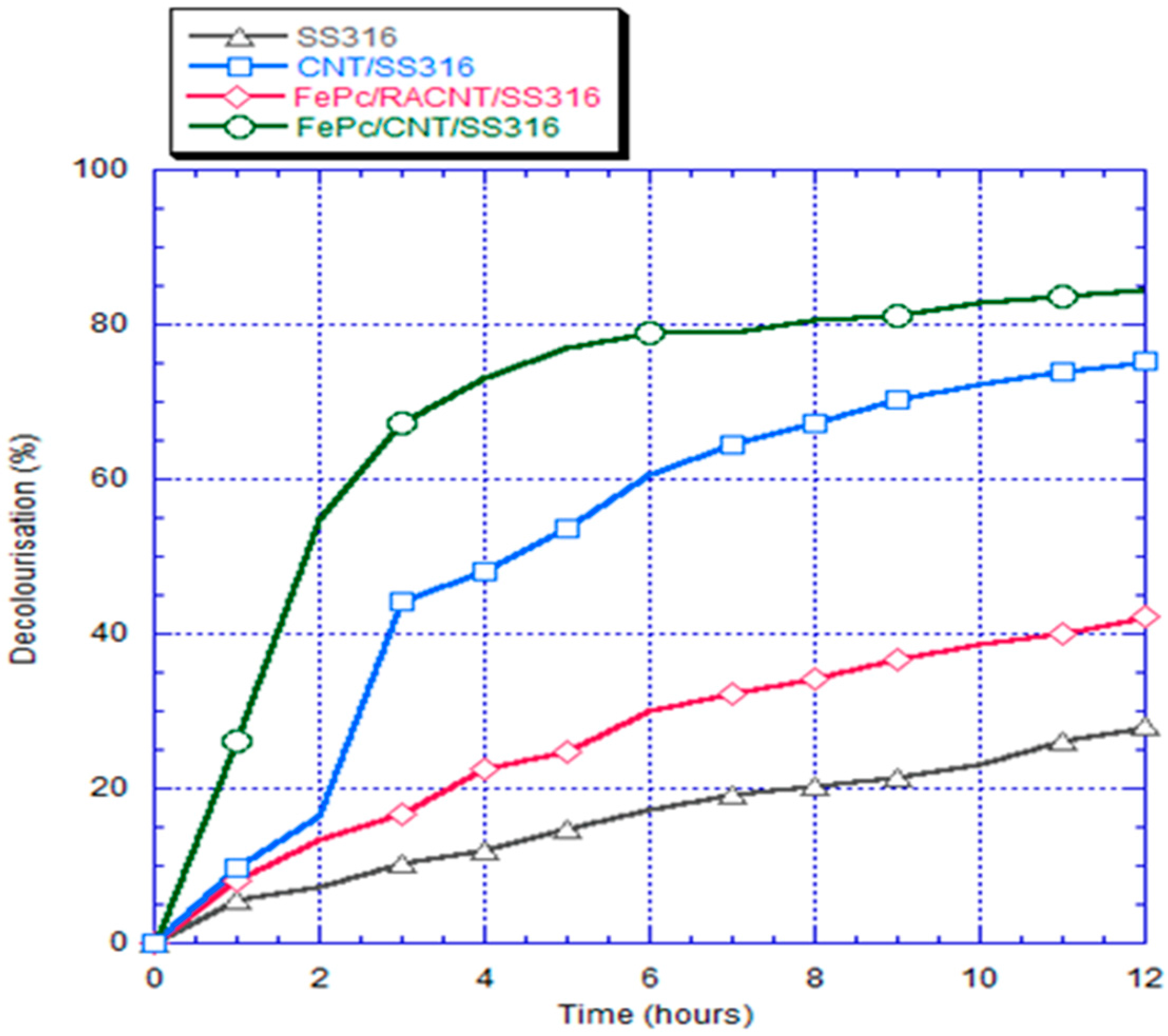
3.6. Decolorization Analyses
4. Conclusions
- CNTs were fabricated using TPCVD, and it was found that the argon gas flow velocity had an absolute impact upon the generation of CNTs. When the argon gas flow velocity was higher, the probability of curling in the generated CNT structure was lower, with aligned generation. The CNTs were generated with good alignment under an argon gas flow rate of 800 sccm and acetylene flow rate of 10 sccm.
- In the measurements using the cyclic voltammetry method, the measured position of the reduction peak of the SS316 electrode had values of 0.035 V and −0.37 mA; the position of the reduction peak of CNT/SS316 had values of −0.047 V and −12 mA; the position of the reduction peak of the FePc/RACNT/SS316 electrode had values of −0.022 V and −8.6 mA; and the position of the reduction peak of FePc/CNT/SS316 had values of 0.018 V and −21 mA. This showed that the iron phthalocyanine and serialized CNT catalysts were conducive to improving the redox activity of the electrode, while the FePc/CNT/SS316 electrode could improve the redox activity of the SS316 electrode.
- The current density and power density of the system with the FePc/CNT/SS316 electrode were 3206.30 mA/m2 and 726.55 mW/m2, respectively; while the current density and power density generated by the SS316 electrode were 3.42 mA/m2 and 0.28 mW/m2, respectively. These results showed that the current density and power density of the modified FePc/CNT/SS316 electrode were 937 and 2594 times higher than those of the system with only the SS316 electrode. In addition, in the experiment on the fixed resistance discharge of the bio-electro-Fenton microbial fuel cells, after exchanging the chamber solution over four cycles, the FePc/CNT/SS316 electrode could still reach a stable voltage output of 0.8 V.
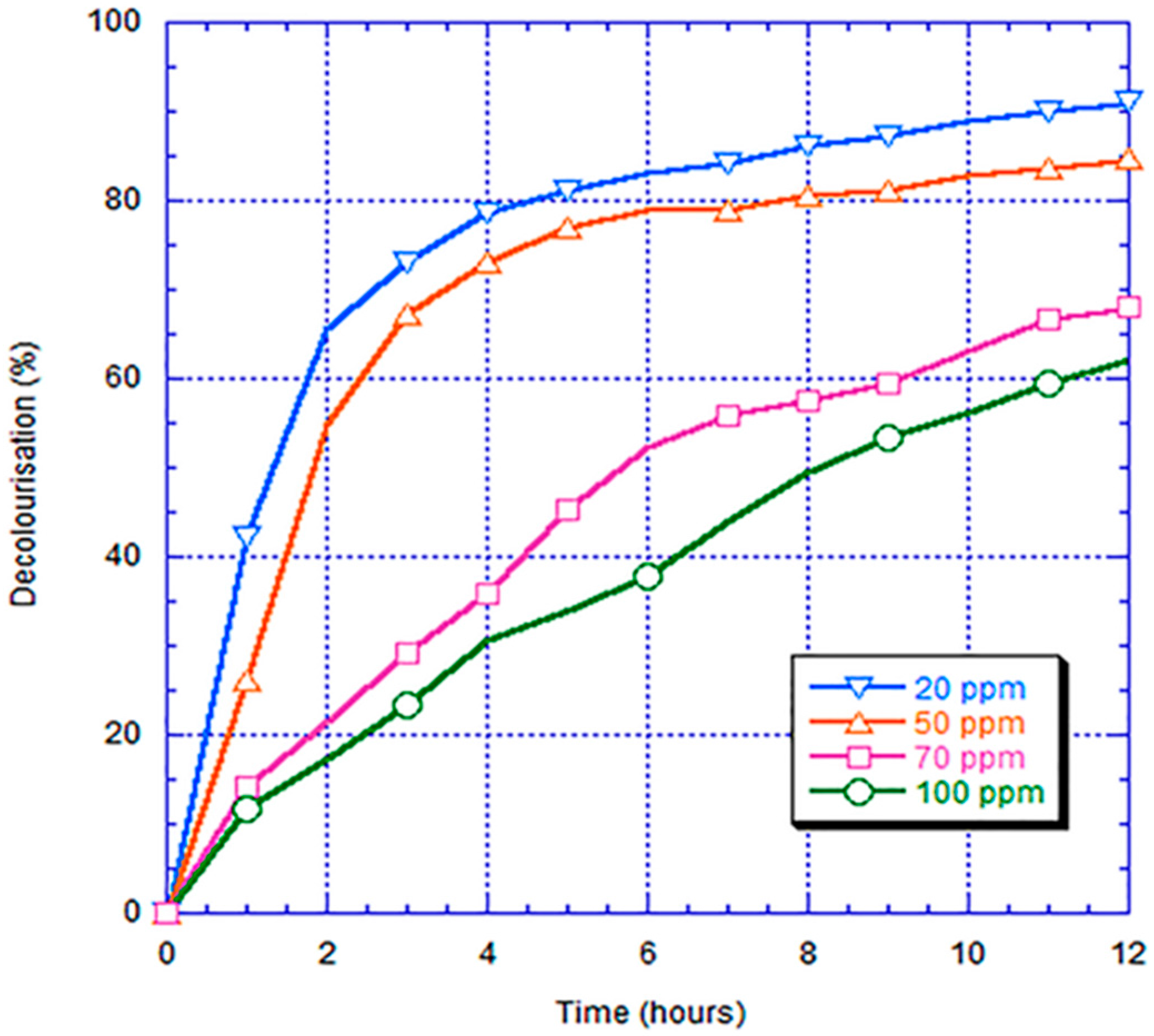
- In the decolorization experiment, the electricity generated by the anode chamber of the bio-electro-Fenton microbial fuel cell system could independently promote the initiation of the electron-Fenton system reaction of the cathode chamber. The decolorization rates of the SS316 electrode, FePc/RACNT/SS316 electrode, CNT/SS316 electrode, and FePc/CNT/SS316 electrode within 12 h were 28%, 42.1%, 74%, and 84.6%, respectively, among which the decolorization efficiency of the modified FePc/CNT/SS316 electrode was the best.
- In this research on a bio-electro-Fenton microbial fuel cell system, the anode was employed for electricity generation and the cathode was employed for the Fenton reaction, achieving the effects of energy generation and sewage disposal at the same time. In addition, an iron phthalocyanine and serialized CNT composite was adopted as the catalyst, which could improve the electrical properties and decolorization effect, and has great potential for improving bio-electro-Fenton microbial fuel cells in the future.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Allen, R.M.; Bennetto, H.P. Microbial fuel-cells electricity production from carbohydrates. Appl. Biochem. Biotechnol. 1993, 39, 27–40. [Google Scholar] [CrossRef]
- Chaudhuri, S.; Lovely, D.R. Electricity generation by direct oxidation of glucose in mediatorless microbial fuel cells. Nat. Biotechnol. 2003, 21, 1229–1232. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.H.; Li, F.B.; Mai, H.J.; Li, X.Z. Bio-electro-Fenton process driven by microbial fuel cell for wastewater treatment. Environ. Sci. Technol. 2010, 44, 1875–1880. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Dios, M.A.; Iglesias, O.; Bocos, E.; Pazos, M.; Sanromán, M.A. Application of benthonic microbial fuel cells and electro-Fenton process to dye decolourisation. J. Ind. Eng. Chem. 2014, 20, 3754–3760. [Google Scholar] [CrossRef]
- Wang, X.Q.; Liu, C.P.; Yuan, Y.; Li, F.B. Arsenite oxidation and removal driven by a bio-electro-Fenton process under neutral pH conditions. J. Hazard. Mater. 2014, 275, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yin, X.; Li, S.F.Y. Bio-electrochemical degradation of paracetamol in a microbial fuel cell-Fenton system. Chem. Eng. J. 2015, 276, 185–192. [Google Scholar] [CrossRef]
- Logan, B.E.; Hamelers, B.; Rozendal, R.; Schröder, U.; Keller, J.; Freguia, S.; Rabaey, K. Microbial fuel cells: Methodology and technology. Environ. Sci. Technol. 2006, 40, 5181–5192. [Google Scholar] [CrossRef]
- Ren, H.; Lee, H.S.; Chae, J. Miniaturizing microbial fuel cells for potential portable power sources: Promises and challenges. Microfluid. Nanofluid. 2012, 13, 353–381. [Google Scholar] [CrossRef]
- Ren, H.; Pyo, S.; Lee, J.I.; Park, T.J.; Gittleson, F.S.; Leung, F.C.C.; Kim, J.; Taylor, A.D.; Lee, H.-S.; Chae, J. A high power density miniaturized microbial fuel cell having carbon nanotube anodes. J. Power Sources 2015, 273, 823–830. [Google Scholar] [CrossRef]
- Ren, H.; Tina, H.; Gardner, C.L.; Ren, T.L.; Chae, J. A miniaturized microbial fuel cell with three-dimensional graphene macroporous scaffold anode demonstrating a record power density of over 10000 W/m3. Nanoscale 2016, 8, 3539–3547. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K. Recent developments in microbial fuel cell technologies for sustainable bioenergy. J. Biosci. Bioeng. 2008, 106, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Chang, I.S.; Kim, B.H. Continuous electricity production from artificial wastewater using a mediator-less microbial fuel cell. Bioresour. Technol. 2006, 97, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Liu, H.; Logam, B.E. Power densities using different cathode catalysts (Pt and CoTMPP) and polymer binders (Nafion and PTFE) in single chamber microbial fuel cells. Environ. Sci. Technol. 2006, 40, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.H.; Cheng, S.; Scott, K.; Logan, B.E. Microbial fuel cell performance with non-Pt cathode catalysts. J. Power Sources 2007, 171, 275–281. [Google Scholar]
- Zhao, F.; Harnisch, F.; Schröder, U.; Scholz, F.; Bogdanoff, P.; Herrmann, I. Application of pyrolysed iron (II) phthalocyanine and CoTMPP based oxygen reduction catalysts as cathode materials in microbial fuel cells. Electrochem. Commun. 2005, 7, 1405–1410. [Google Scholar] [CrossRef]
- Zagal, J.H.; Bedioui, F.; Dodelet, J.-P. N4-Macrocyclic Metal Complexes; Springer: New York, NY, USA, 2006. [Google Scholar]
- Zhao, F.; Harnisch, F.; Schröder, U.; Scholz, F.; Bogdanoff, P.; Herrmann, I. Challenges and constraints of using oxygen cathodes in microbial fuel cells. Environ. Sci. Technol. 2006, 40, 5193–5199. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Zhou, M.; Liu, C.; Liu, L.; Liu, C.; Dong, S. Development of high performance of Co/Fe/N/CNT nanocatalyst for oxygen reduction in microbial fuel cells. Talanta 2010, 81, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, B.; Cui, D.; Xiang, X.; Li, W. Nano-molybdenum carbide/carbon nanotubes composite as bifunctional anode catalyst for high-performance Escherichia coli-based microbial fuel cell. Biosens. Bioelectron. 2014, 51, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, M.; Daud, W.R.W.; Hassan, S.H.A.; Jafary, T.; Rahimnejad, M.; Ahmad, A.; Yazdi, M.H. Carbon nanotube/polypyrrole nanocomposite as a novel cathode catalyst and proper alternative for Pt in microbial fuel cell. Int. J. Hydrog. Energy 2016, 41, 4872–4878. [Google Scholar] [CrossRef]
- Shaijumon, M.; Sundara, R.; Rajalakshmi, N. Platinum/multiwalled carbon nanotubes-platinum/carbon composites as electrocatalysts for oxygen reduction reaction in proton exchange membrane fuel cell. Appl. Phys. Lett. 2006, 88, 1–3. [Google Scholar] [CrossRef]
- Lee, S.W.; Sigmund, W.M. Formation of anatase TiO2 nanoparticles on carbon nanotubes. Chem. Commun. 2003, 21, 780–781. [Google Scholar] [CrossRef]
- Hang, S.T.; Yeh, C.Y.; Yao, P.C. Synthesis of carbon nanotubes over sol-gel deposited substrates from acetylene by thermal chemical vapor deposition. J. Sci. Eng. Technol. 2007, 3, 47–54. [Google Scholar]
- Pacios, M.; Martín-Fernández, I.; Villa, R.; Godignon, P.; Valle, M.D.; Bartrolí, J.; Esplandiu, M.J. Carbon Nanotubes as Suitable Electrochemical Platforms for Metalloprotein Sensors and Genosensors; InTech: Rijeka, Croatia, 2011. [Google Scholar]
- Li, Y.-H.; Wang, S.; Luan, Z.; Ding, J.; Xu, C.; Wu, D. Adsorption of cadmium(II) from aqueous solution by surface oxidized carbon nanotubes. Carbon 2012, 41, 1057–1062. [Google Scholar] [CrossRef]
- Lu, C.; Chiu, H. Adsorption of zinc (II) from water with purified carbon nanotubes. Chem. Eng. Sci. 2006, 61, 1138–1145. [Google Scholar] [CrossRef]
- Pocaznoi, D.; Calmet, A.; Etcheverry, L.; Bergel, A. Stainless steel is a promising electrode material for anodes of microbial fuel cells. Energy Environ. Sci. 2012, 5, 9645–9652. [Google Scholar] [CrossRef]
- Liu, Y.; Yue, X.; Li, K.; Qiao, J.; Wilkinson, D.P.; Zhang, J. PEM fuel cell electrocatalysts based on transition metal macrocyclic compounds. Coord. Chem. Rev. 2016, 315, 153–177. [Google Scholar] [CrossRef]
- Gong, K.; Du, F.; Xia, Z.; Durstock, M.; Dai, L. Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction. Science 2009, 323, 760–764. [Google Scholar] [CrossRef] [PubMed]




| Component | Si | Mo | Cr | Mn | Fe | Ni |
|---|---|---|---|---|---|---|
| Content (%) | 0.86 | 2.23 | 17.78 | 1.09 | 67.41 | 10.63 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.-T.; Wang, R.-S. A Bio-Electro-Fenton System Employing the Composite FePc/CNT/SS316 Cathode. Materials 2017, 10, 169. https://doi.org/10.3390/ma10020169
Wang Y-T, Wang R-S. A Bio-Electro-Fenton System Employing the Composite FePc/CNT/SS316 Cathode. Materials. 2017; 10(2):169. https://doi.org/10.3390/ma10020169
Chicago/Turabian StyleWang, Yi-Ta, and Ruei-Shiang Wang. 2017. "A Bio-Electro-Fenton System Employing the Composite FePc/CNT/SS316 Cathode" Materials 10, no. 2: 169. https://doi.org/10.3390/ma10020169
APA StyleWang, Y.-T., & Wang, R.-S. (2017). A Bio-Electro-Fenton System Employing the Composite FePc/CNT/SS316 Cathode. Materials, 10(2), 169. https://doi.org/10.3390/ma10020169





