Electrospun PVA/Bentonite Nanocomposites Mats for Drug Delivery
Abstract
:1. Introduction
2. Results
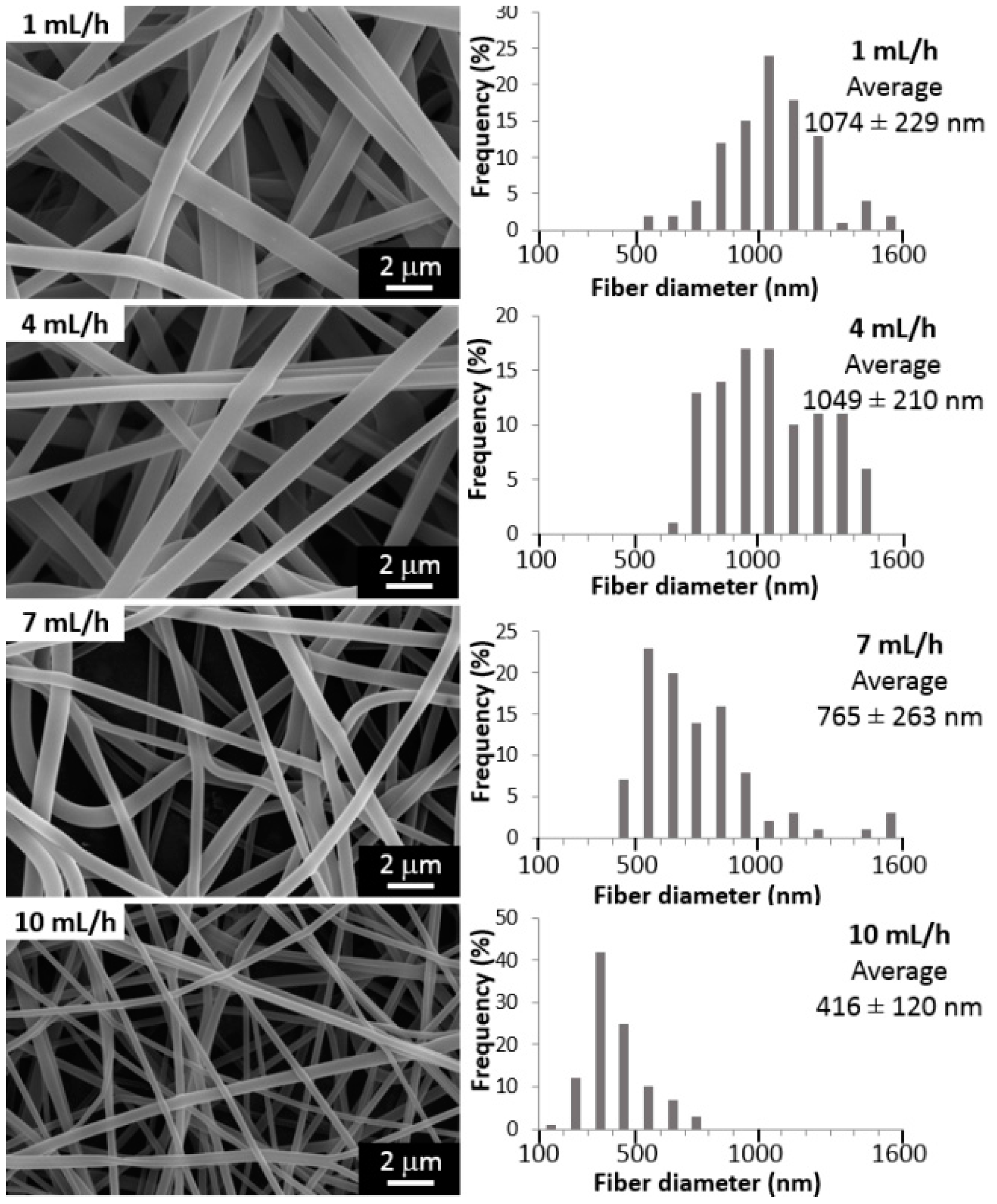
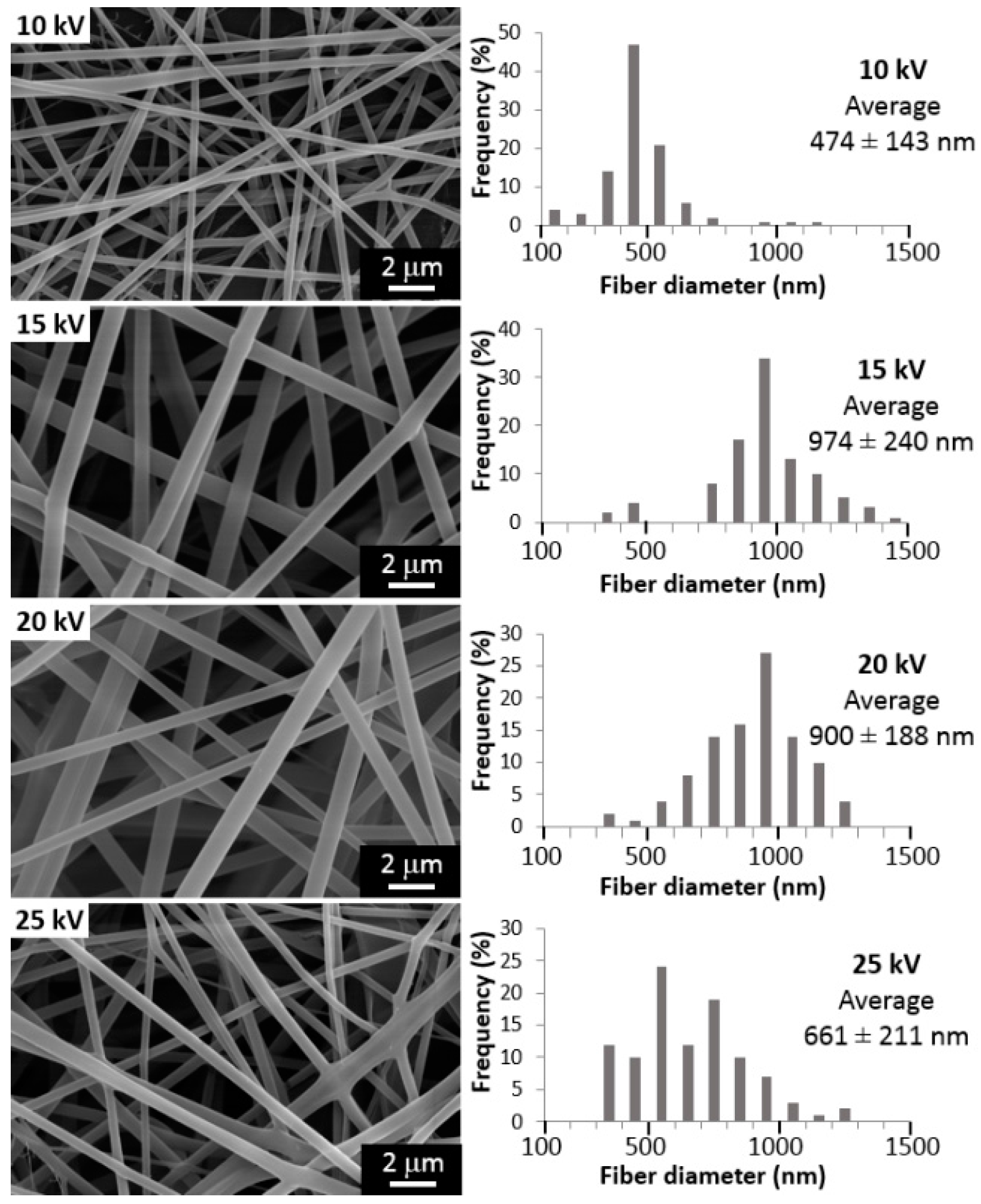
2.1. Influence of the Processing Parameters on PVA Electrospun Fibers Mean Diameter
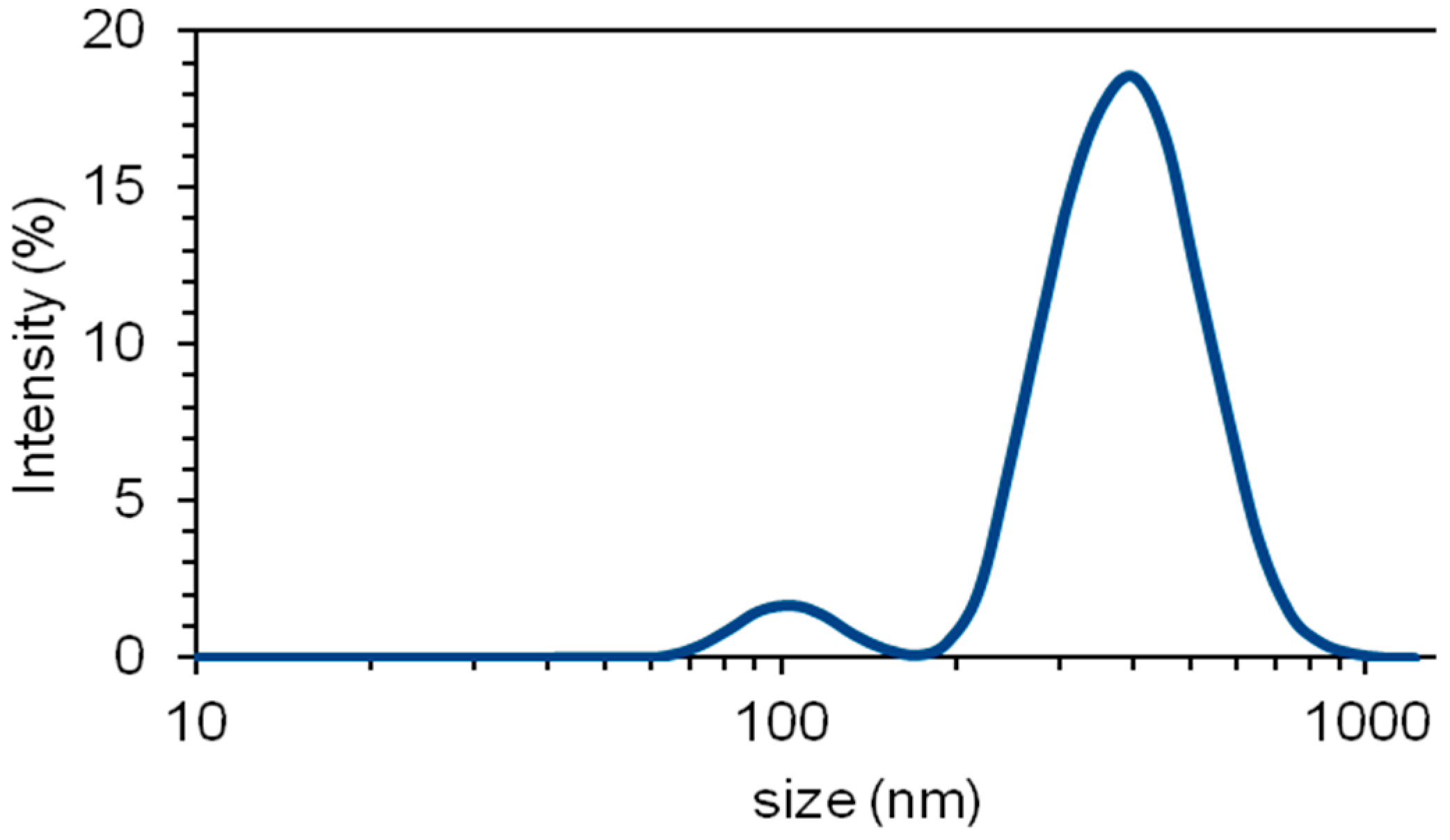
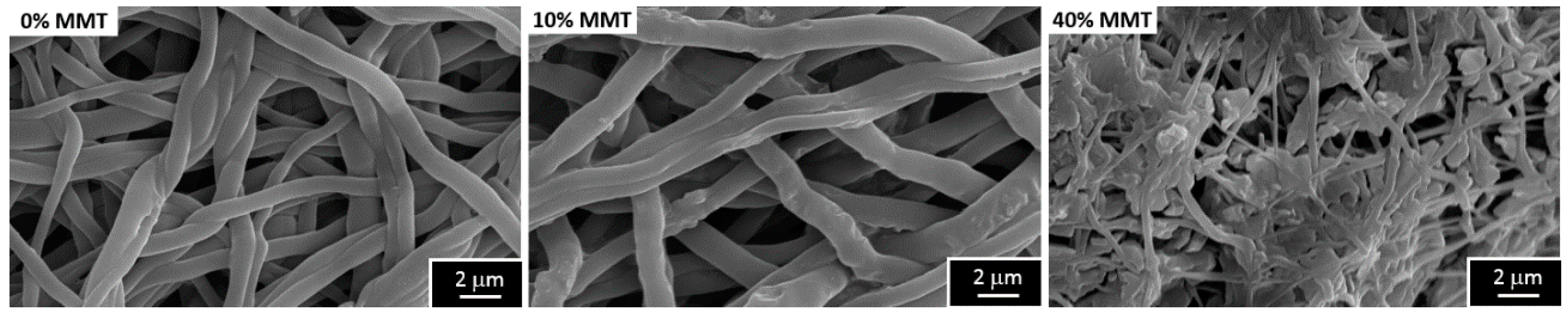
2.2. Influence of the Presence of Dispersed Nanoclay
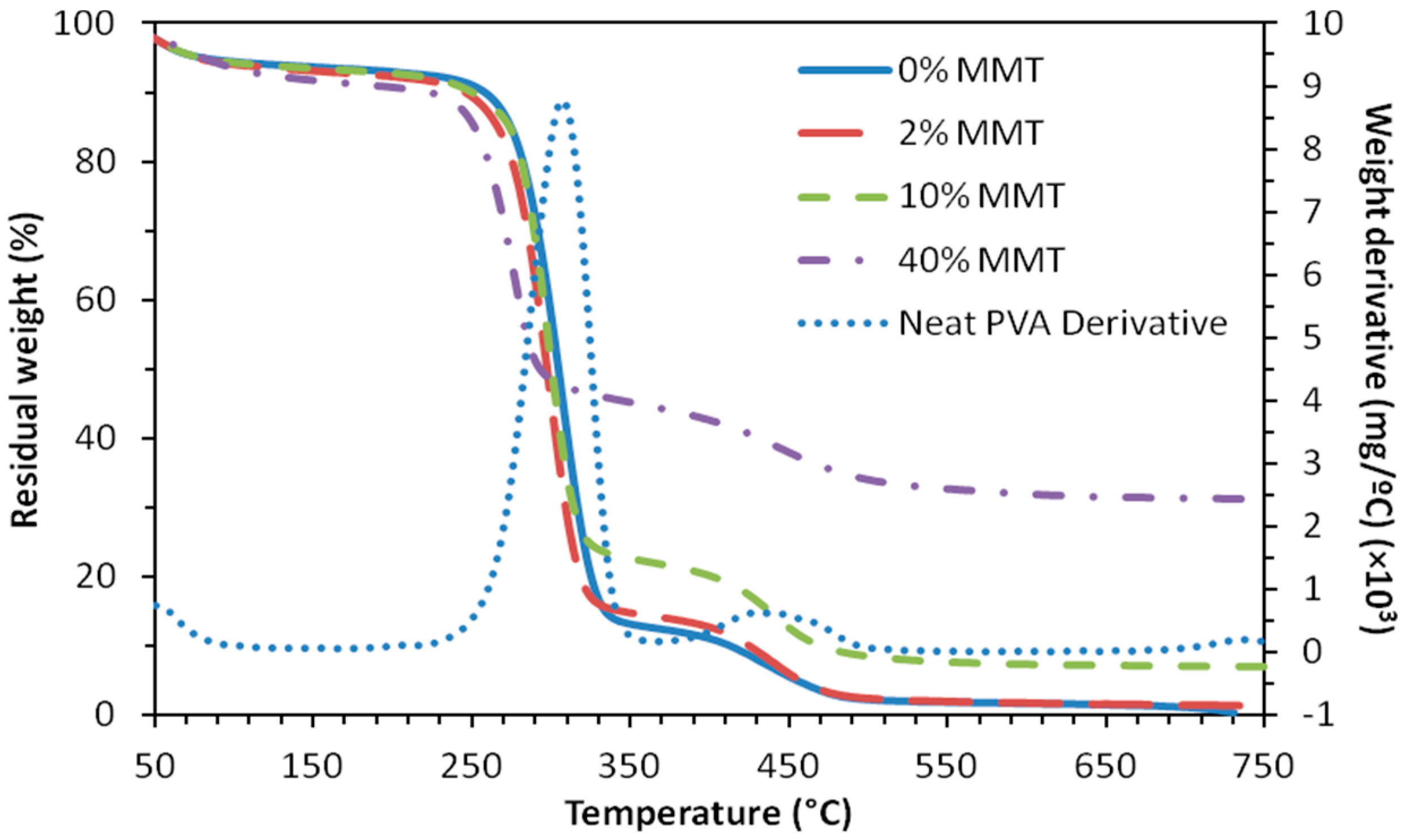
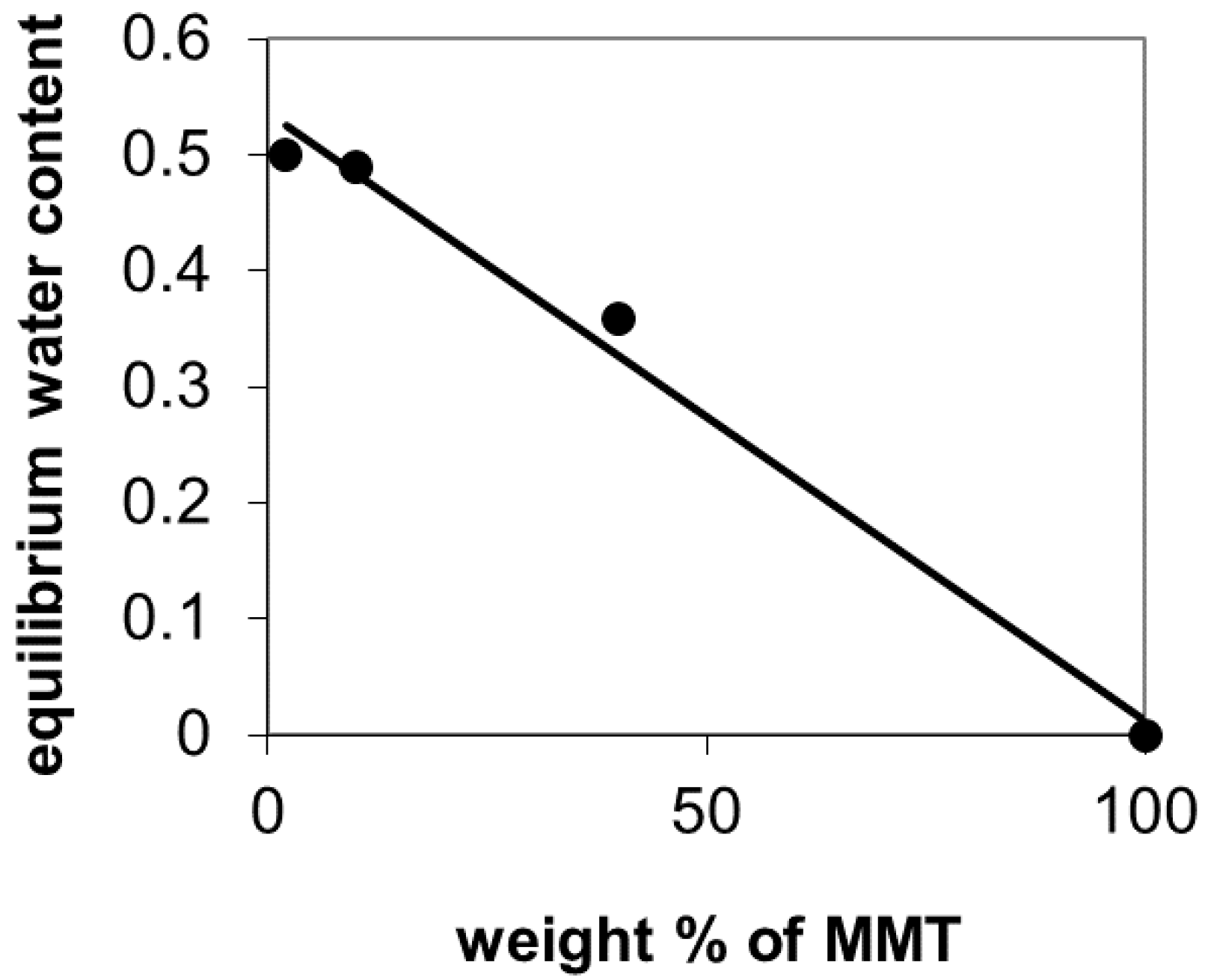
2.3. Thermal Stability and Swelling Behavior of PVA Films and Mats
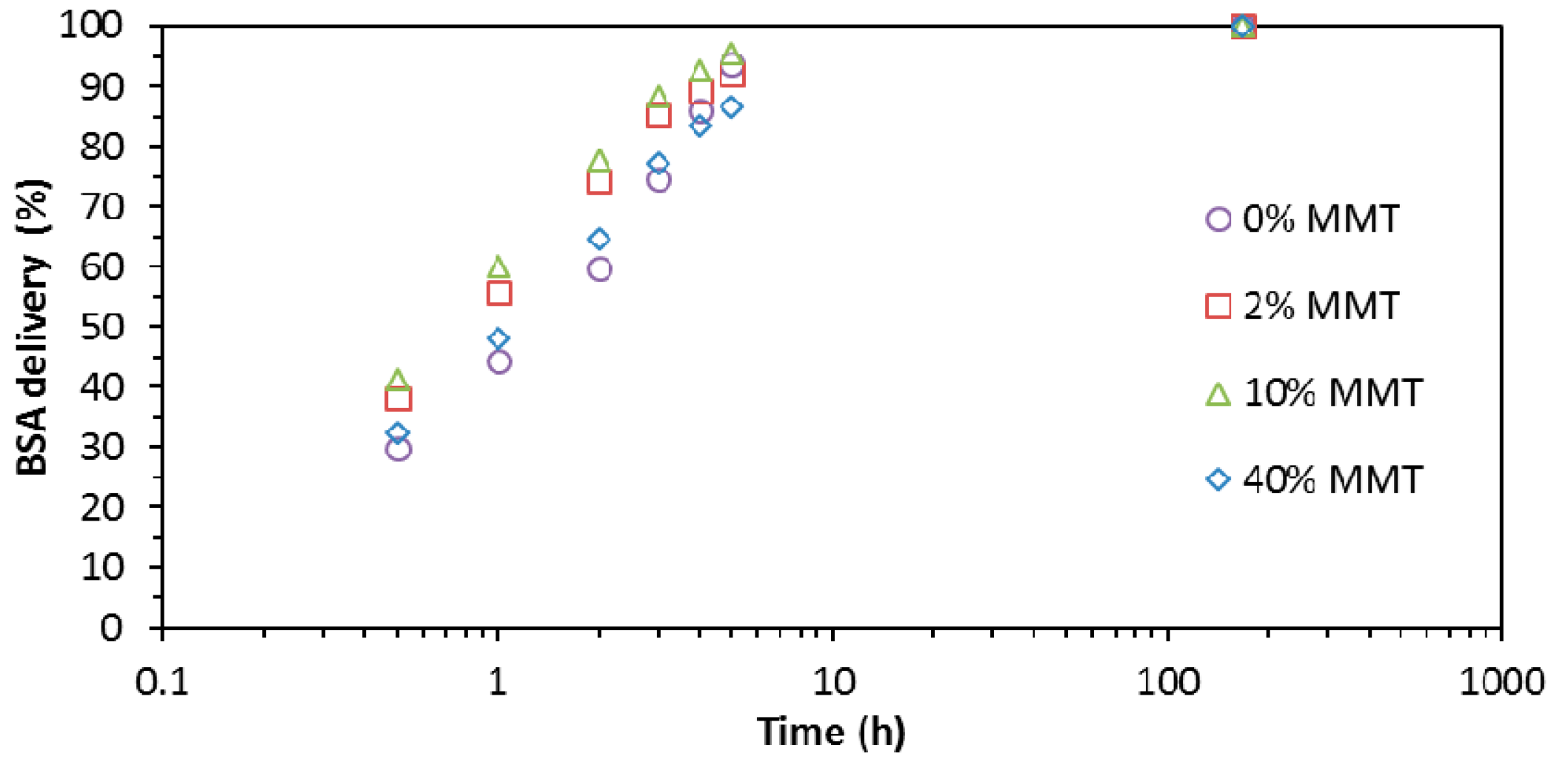
2.4. BSA Absorption and Delivery Assays
3. Discussion
3.1. Influence of the Processing Parameters on PVA Electrospun Fibers Mean Diameter
3.2. Influence of the Presence of Dispersed Nanoclay
3.3. Thermal Stability and Swelling Behavior of PVA Films and Mats
3.4. BSA Absorption and Delivery Assays
4. Materials and Methods
4.1. Materials
4.2. Preparation of PVA and PVA/MMT Solutions
4.3. Electrospinning Conditions
4.4. Films Preparation: PVA and PVA/MMT
4.5. Crosslinking of Films and Mats
4.6. Swelling Assay
4.7. Absorption, Release, and Quantification of Protein
4.8. Characterization
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paradossi, G.; Cavalieri, F.; Chiessi, E.; Spagnoli, C.; Cowman, M.K. Poly(vinyl alcohol) as versatile biomaterial for potential biomedical applications. J. Mater. Sci. Mater. Med. 2003, 14, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Sill, T.J.; von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Suntornnond, R.; An, J.; Yeong, W.Y.; Chua, C.K. Biodegradable Polymeric Films and Membranes Processing and Forming for Tissue Engineering. Macromol. Mater. Eng. 2015, 300, 858–877. [Google Scholar] [CrossRef]
- Nguyen, L.T.H.; Chen, S.; Elumalai, N.K.; Prabhakaran, M.P.; Zong, Y.; Vijila, C.; Allakhverdiev, S.I.; Ramakrishna, S. Biological, Chemical, and Electronic Applications of Nanofibers. Macromol. Mater. Eng. 2013, 298, 822–867. [Google Scholar] [CrossRef]
- Gómez-Tejedor, J.A.; Van Overberghe, N.; Rico, P.; Ribelles, J.L.G. Assessment of the parameters influencing the fiber characteristics of electrospun poly(ethyl methacrylate) membranes. Eur. Polym. J. 2011, 47, 119–129. [Google Scholar] [CrossRef]
- Silva, C.S.R.; Luz, G.M.; Gamboa-martÍnez, T.C.; Mano, J.F.; GÓmez ribelles, J.L.; GÓmez-tejedor, J.A. Poly(ɛ-caprolactone) Electrospun Scaffolds Filled with Nanoparticles. Production and Optimization According to Taguchi’s Methodology. J. Macromol. Sci. Part B 2014, 53, 781–799. [Google Scholar] [CrossRef]
- Nugroho, R.W.N.; Roy, P.K.; Odelius, K.; Albertsson, A.-C. Crosslinked PVAL nanofibers with enhanced long-term stability prepared by single-step electrospinning. Polym. Adv. Technol. 2013, 24, 421–429. [Google Scholar] [CrossRef]
- Destaye, A.G.; Lin, C.-K.; Lee, C.-K. Glutaraldehyde Vapor Cross-linked Nanofibrous PVA Mat with in Situ Formed Silver Nanoparticles. ACS Appl. Mater. Interfaces 2013, 5, 4745–4752. [Google Scholar] [CrossRef] [PubMed]
- Pathan, S.G.; Fitzgerald, L.M.; Ali, S.M.; Damrauer, S.M.; Bide, M.J.; Nelson, D.W.; Ferran, C.; Phaneuf, T.M.; Phaneuf, M.D. Cytotoxicity associated with electrospun polyvinyl alcohol. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 1652–1662. [Google Scholar] [CrossRef] [PubMed]
- Arayanarakul, K.; Choktaweesap, N.; Aht-ong, D.; Meechaisue, C.; Supaphol, P. Effects of Poly(ethylene glycol), Inorganic Salt, Sodium Dodecyl Sulfate, and Solvent System on Electrospinning of Poly(ethylene oxide). Macromol. Mater. Eng. 2006, 291, 581–591. [Google Scholar] [CrossRef]
- Koosha, M.; Mirzadeh, H.; Shokrgozar, M.A.; Farokhi, M. Nanoclay-reinforced electrospun chitosan/PVA nanocomposite nanofibers for biomedical applications. RSC Adv. 2015, 5, 10479–10487. [Google Scholar] [CrossRef]
- Hassan, C.M.; Peppas, N.A. Structure and Applications of Poly(vinyl alcohol) Hydrogels Produced by Conventional Crosslinking or by Freezing/Thawing Methods. In Biopolymers PVA Hydrogels, Anionic Polymerisation Nanocomposites; Springer: Berlin/Heidelberg, Germany, 2000; pp. 37–65. [Google Scholar]
- Kim, K.-J.; Lee, S.-B.; Han, N.W. Effects of the Degree of Crosslinking on Properties of Poly(vinyl alcohol) Membranes. Polym. J. 1993, 25, 1295–1302. [Google Scholar] [CrossRef]
- Zhang, X.; Tang, K.; Zheng, X. Electrospinning and Crosslinking of COL/PVA Nanofiber-microsphere Containing Salicylic Acid for Drug Delivery. J. Bionic Eng. 2016, 13, 143–149. [Google Scholar] [CrossRef]
- Lee, I.W.; Li, J.; Chen, X.; Park, H.J. Electrospun poly(vinyl alcohol) composite nanofibers with halloysite nanotubes for the sustained release of sodium d-pantothenate. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Tsai, R.-Y.; Hung, S.-C.; Lai, J.-Y.; Wang, D.-M.; Hsieh, H.-J. Electrospun chitosan-gelatin-polyvinyl alcohol hybrid nanofibrous mats: Production and characterization. J. Taiwan Inst. Chem. Eng. 2014, 45, 1975–1981. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Regí, M.; Colilla, M.; González, B. Medical applications of organic-inorganic hybrid materials within the field of silica-based bioceramics. Chem. Soc. Rev. 2011, 40, 596–607. [Google Scholar] [CrossRef] [PubMed]
- Armentano, I.; Dottori, M.; Fortunati, E.; Mattioli, S.; Kenny, J.M. Biodegradable polymer matrix nanocomposites for tissue engineering: A review. Polym. Degrad. Stab. 2010, 95, 2126–2146. [Google Scholar] [CrossRef]
- Lee, W.-F.; Chen, Y.-C. Effect of bentonite on the physical properties and drug-release behavior of poly(AA-co-PEGMEA)/bentonite nanocomposite hydrogels for mucoadhesive. J. Appl. Polym. Sci. 2004, 91, 2934–2941. [Google Scholar] [CrossRef]
- Bhattacharya, S.S.; Sen, K.K.; Sen, S.O.; Banerjee, S.; Kaity, S.; Ghosh, A.K.; Ghosh, A. Synthesis and Characterization of Poly(acrylic acid)/modified Bentonite Superabsorbent Polymer. Int. J. Polym. Mater. 2011, 60, 1015–1025. [Google Scholar] [CrossRef]
- Zerda, A.S.; Lesser, A.J. Intercalated clay nanocomposites: Morphology, mechanics, and fracture behavior. J. Polym. Sci. Part B Polym. Phys. 2001, 39, 1137–1146. [Google Scholar] [CrossRef]
- Rao, Y.; Pochan, J.M. Mechanics of Polymer-Clay Nanocomposites. Macromolecules 2007, 40, 290–296. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, T.G.; Park, H.S.; Lee, D.S.; Lee, Y.K.; Yoon, S.C.; Nam, J.-D. Thermal and mechanical characteristics of poly(l-lactic acid) nanocomposite scaffold. Biomaterials 2003, 24, 2773–2778. [Google Scholar] [CrossRef]
- Marras, S.I.; Kladi, K.P.; Tsivintzelis, I.; Zuburtikudis, I.; Panayiotou, C. Biodegradable polymer nanocomposites: The role of nanoclays on the thermomechanical characteristics and the electrospun fibrous structure. Acta Biomater. 2008, 4, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Yeum, J.H.; Das, A.K. Effect of pullulan/poly(vinyl alcohol) blend system on the montmorillonite structure with property characterization of electrospun pullulan/poly(vinyl alcohol)/montmorillonite nanofibers. J. Colloid Interface Sci. 2012, 368, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.M.; Lee, H.W.; Karim, M.R.; Cheong, I.W.; Bae, E.A.; Kim, T.H.; Islam, M.S.; Ji, B.C.; Yeum, J.H. Electrospinning and characterization of medium-molecular-weight poly(vinyl alcohol)/high-molecular-weight poly(vinyl alcohol)/montmorillonite nanofibers. Colloid Polym. Sci. 2009, 287, 751–758. [Google Scholar] [CrossRef]
- Kahraman, H.T.; Gevgilili, H.; Kalyon, D.M.; Pehlivan, E. Nanoclay dispersion into a thermosetting binder using sonication and intensive mixing methods. J. Appl. Polym. Sci. 2013, 129, 1773–1783. [Google Scholar] [CrossRef]
- Kaboorani, A.; Riedl, B.; Blanchet, P. Ultrasonication Technique: A Method for Dispersing Nanoclay in Wood Adhesives. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Koski, A.; Yim, K.; Shivkumar, S. Effect of molecular weight on fibrous PVA produced by electrospinning. Mater. Lett. 2004, 58, 493–497. [Google Scholar] [CrossRef]
- Lee, J.S.; Choi, K.H.; Ghim, H.D.; Kim, S.S.; Chun, D.H.; Kim, H.Y.; Lyoo, W.S. Role of molecular weight of atactic poly(vinyl alcohol) (PVA) in the structure and properties of PVA nanofabric prepared by electrospinning. J. Appl. Polym. Sci. 2004, 93, 1638–1646. [Google Scholar] [CrossRef]
- Ding, B.; Kim, H.-Y.; Lee, S.-C.; Shao, C.-L.; Lee, D.-R.; Park, S.-J.; Kwag, G.-B.; Choi, K.-J. Preparation and characterization of a nanoscale poly(vinyl alcohol) fiber aggregate produced by an electrospinning method. J. Polym. Sci. Part B Polym. Phys. 2002, 40, 1261–1268. [Google Scholar] [CrossRef]
- Adomavičiūtė, E.; Milašius, R.; Levinskas, R. The Influence of Main Technological Parameters on the Diameter of Poly(vinyl alcohol) (PVA) Nanofibre and Morphology of Manufactured Mat. Mater. Sci. 2007, 13, 3–6. [Google Scholar]
- Zhang, C.; Yuan, X.; Wu, L.; Han, Y.; Sheng, J. Study on morphology of electrospun poly(vinyl alcohol) mats. Eur. Polym. J. 2005, 41, 423–432. [Google Scholar] [CrossRef]
- Lee, H.W.; Karim, M.R.; Ji, H.M.; Choi, J.H.; Ghim, H.D.; Park, S.M.; Oh, W.; Yeum, J.H. Electrospinning fabrication and characterization of poly(vinyl alcohol)/montmorillonite nanofiber mats. J. Appl. Polym. Sci. 2009, 113, 1860–1867. [Google Scholar] [CrossRef]
- Belozerov, A.G.; Karasev, N.S.; Ovchinnikov, N.L.; Butman, M.F. The effect of preliminary delamination of silicate filler on its exfoliation degree in polyvinyl alcohol/montmorillonite nanocomposites. Nanotechnol. Russ. 2014, 9, 410–415. [Google Scholar] [CrossRef]
- Wickham, A.M.; Islam, M.M.; Mondal, D.; Phopase, J.; Sadhu, V.; Tamás, É.; Polisetti, N.; Richter-Dahlfors, A.; Liedberg, B.; Griffith, M. Polycaprolactone-thiophene-conjugated carbon nanotube meshes as scaffolds for cardiac progenitor cells. J. Biomed. Mater. Res. B Appl. Biomater. 2014, 102, 1553–1561. [Google Scholar] [CrossRef] [PubMed]
- Purwar, R.; Sai Goutham, K.; Srivastava, C.M. Electrospun Sericin/PVA/Clay nanofibrous mats for antimicrobial air filtration mask. Fibers Polym. 2016, 17, 1206–1216. [Google Scholar] [CrossRef]
- Yuan, H.; Li, B.; Liang, K.; Lou, X.; Zhang, Y. Regulating drug release from pH- and temperature-responsive electrospun CTS-g-PNIPAAm/poly(ethylene oxide) hydrogel nanofibers. Biomed. Mater. 2014, 9, 55001. [Google Scholar] [CrossRef] [PubMed]
- Vakilian, S.; Mashayekhan, S.; Shabani, I.; Khorashadizadeh, M.; Fallah, A.; Soleimani, M. Structural stability and sustained release of protein from a multilayer nanofiber/nanoparticle composite. Int. J. Biol. Macromol. 2015, 75, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Raheja, A.; Agarwal, A.; Muthuvijayan, V.; Chandra, T.S.; Natarajan, T.S. Studies on Encapsulation of Bovine Serum Albumin, Lysozyme and Insulin through Coaxial Electrospinning. J. Biomater. Tissue Eng. 2013, 3, 669–672. [Google Scholar] [CrossRef]
- Duque Sánchez, L.M.; Rodríguez, L.; López, M. Electrospinning: La era de las nanofibras. Rev. Iberoam. Polímeros 2013, 14, 10–27. [Google Scholar]
- Pandey, J.K.; Reddy, K.R.; Mohanty, A.K.; Misra, M. Handbook of Polymernanocomposites. Processing, Performance and Application; Pandey, J.K., Reddy, K.R., Mohanty, A.K., Misra, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; Volume C, ISBN 978-3-642-38648-0. [Google Scholar]
- Riba, J.-R.; Esteban, B. A simple laboratory experiment to measure the surface tension of a liquid in contact with air. Eur. J. Phys. 2014, 35, 55003. [Google Scholar] [CrossRef]
- Lee, B.-B.; Ravindra, P.; Chan, E.-S. New drop weight analysis for surface tension determination of liquids. Colloids Surf. A Physicochem. Eng. Asp. 2009, 332, 112–120. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Barzegar, F.; Bello, A.; Fabiane, M.; Khamlich, S.; Momodu, D.; Taghizadeh, F.; Dangbegnon, J.; Manyala, N. Preparation and characterization of poly(vinyl alcohol)/graphene nanofibers synthesized by electrospinning. J. Phys. Chem. Solids 2015, 77, 139–145. [Google Scholar] [CrossRef]
- Islam, M.S.; Rahaman, M.S.; Yeum, J.H. Electrospun novel super-absorbent based on polysaccharide-polyvinyl alcohol-montmorillonite clay nanocomposites. Carbohydr. Polym. 2015, 115, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hsieh, Y.-L. Immobilization of lipase enzyme in polyvinyl alcohol (PVA) nanofibrous membranes. J. Membr. Sci. 2008, 309, 73–81. [Google Scholar] [CrossRef]
- Buchko, C.J.; Chen, L.C.; Shen, Y.; Martin, D.C. Processing and microstructural characterization of porous biocompatible protein polymer thin films. Polymer 1999, 40, 7397–7407. [Google Scholar] [CrossRef]
- Pham, Q.P.; Sharma, U.; Mikos, A.G. Electrospinning of Polymeric Nanofibers for Tissue Engineering Applications: A Review. Tissue Eng. 2006, 12, 1197–1211. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, C. One-Dimensional Nanostructures; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Haghi, A.K.; Akbari, M. Trends in electrospinning of natural nanofibers. Phys. Status Solidi 2007, 204, 1830–1834. [Google Scholar] [CrossRef]
- Reneker, D.H.; Chun, I. Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 1996, 7, 216–223. [Google Scholar] [CrossRef]
- Zuo, W.; Zhu, M.; Yang, W.; Yu, H.; Chen, Y.; Zhang, Y. Experimental study on relationship between jet instability and formation of beaded fibers during electrospinning. Polym. Eng. Sci. 2005, 45, 704–709. [Google Scholar] [CrossRef]
- Zong, X.; Kim, K.; Fang, D.; Ran, S.; Hsiao, B.S.; Chu, B. Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 2002, 43, 4403–4412. [Google Scholar] [CrossRef]
- Qin, Q.; Liu, Y.; Chen, S.-C.; Zhai, F.-Y.; Jing, X.-K.; Wang, Y.Z. Electrospinning fabrication and characterization of poly(vinyl alcohol)/layered double hydroxides composite fibers. J. Appl. Polym. Sci. 2012, 126, 1556–1563. [Google Scholar] [CrossRef]
- Ristolainen, N.; Heikkilä, P.; Harlin, A.; Seppälä, J. Poly(vinyl alcohol) and Polyamide-66 Nanocomposites Prepared by Electrospinning. Macromol. Mater. Eng. 2006, 291, 114–122. [Google Scholar] [CrossRef]
- Daga, V.K.; Helgeson, M.E.; Wagner, N.J. Electrospinning of neat and laponite-filled aqueous poly(ethylene oxide) solutions. J. Polym. Sci. Part B Polym. Phys. 2006, 44, 1608–1617. [Google Scholar] [CrossRef]
- Zhao, C.; Qin, H.; Gong, F.; Feng, M.; Zhang, S.; Yang, M. Mechanical, thermal and flammability properties of polyethylene/clay nanocomposites. Polym. Degrad. Stab. 2005, 87, 183–189. [Google Scholar] [CrossRef]
- Lee, B.-B.; Ravindra, P.; Chan, E.-S. A critical review: Surface and interfacial tension measurement by the drop weight method. Chem. Eng. Commun. 2008, 195, 889–924. [Google Scholar] [CrossRef]


| % | Density | Surface Tension (mN/m) | |
|---|---|---|---|
| MMT | (kg/m3) | Method 1 | Method 2 |
| 0 | 1076 | 31.7 | 20.7 |
| 2 | 1082 | 32.4 | 26.3 |
| 10 | 1089 | 29.6 | 21.8 |
| 40 | 1097 | 31.1 | 20.6 |
| Product | Theoretical Content of MMT (%) | TGA Measured Nanoclay Content (%) | Swelling Ratio at 5 h (g/g) |
|---|---|---|---|
| Films | 0 | 0 | --- |
| 2 | 1.9 | 1.00 | |
| 10 | 8.4 | 0.96 | |
| 40 | 35.1 | 0.56 | |
| Electrospun mats | 0 | 0 | 2.46 |
| 2 | 1.6 | 2.52 | |
| 10 | 7.5 | 2.66 | |
| 40 | 34.5 | 2.18 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrández-Rives, M.; Beltrán-Osuna, Á.A.; Gómez-Tejedor, J.A.; Gómez Ribelles, J.L. Electrospun PVA/Bentonite Nanocomposites Mats for Drug Delivery. Materials 2017, 10, 1448. https://doi.org/10.3390/ma10121448
Ferrández-Rives M, Beltrán-Osuna ÁA, Gómez-Tejedor JA, Gómez Ribelles JL. Electrospun PVA/Bentonite Nanocomposites Mats for Drug Delivery. Materials. 2017; 10(12):1448. https://doi.org/10.3390/ma10121448
Chicago/Turabian StyleFerrández-Rives, Mariola, Ángela Aurora Beltrán-Osuna, José Antonio Gómez-Tejedor, and José Luis Gómez Ribelles. 2017. "Electrospun PVA/Bentonite Nanocomposites Mats for Drug Delivery" Materials 10, no. 12: 1448. https://doi.org/10.3390/ma10121448
APA StyleFerrández-Rives, M., Beltrán-Osuna, Á. A., Gómez-Tejedor, J. A., & Gómez Ribelles, J. L. (2017). Electrospun PVA/Bentonite Nanocomposites Mats for Drug Delivery. Materials, 10(12), 1448. https://doi.org/10.3390/ma10121448







