Alkaline Materials and Regenerative Endodontics: A Review
Abstract
:1. Introduction
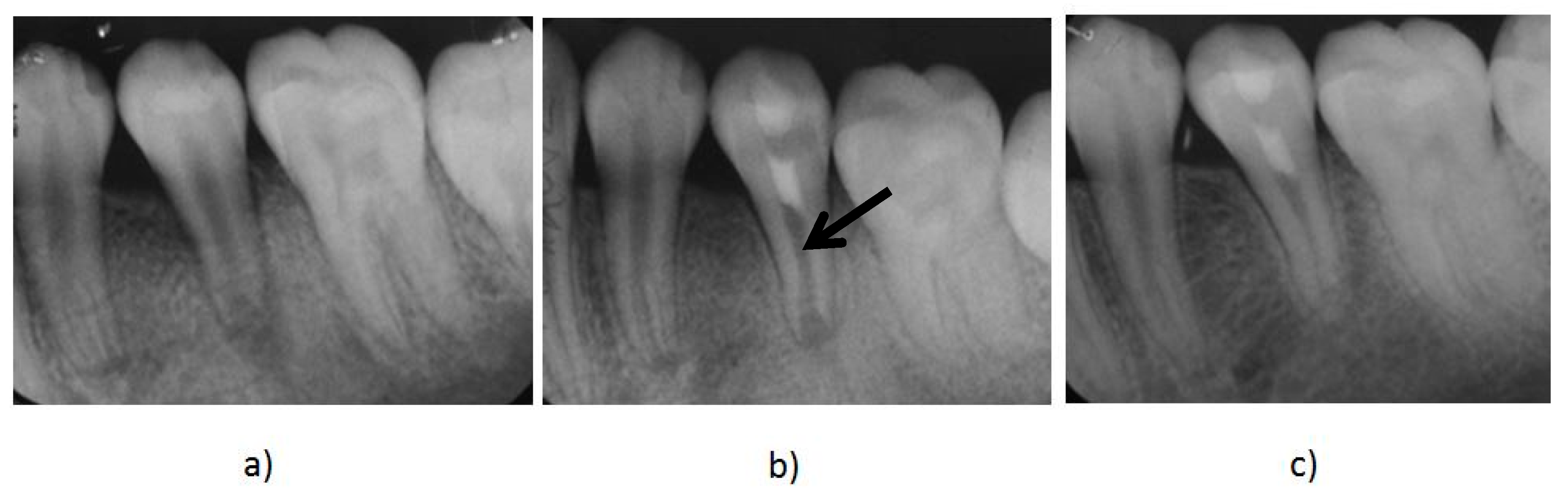
2. Regenerative Endodontics
3. Alkaline Materials Used in REPs
3.1. Sodium Hypochlorite (NaOCl)
3.2. Calcium Hydroxide
3.3. Mineral Trioxide Aggregate (MTA)
3.4. Biodentine
3.5. Calcium-Enriched Material (CEM)
3.6. EndoSequence Bioceramic Putty
4. Future Prospects
5. Conclusions
Conflicts of Interest
References
- Cvek, M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha. A retrospective clinical study. Dent. Traumatol. 1992, 8, 45–55. [Google Scholar] [CrossRef]
- Cvek, M. Treatment of non-vital permanent incisors with calcium hydroxide. I. Follow-up of periapical repair and apical closure of immature roots. Odontol. Revy 1972, 23, 27–44. [Google Scholar] [PubMed]
- Heithersay, G.S. Calcium hydroxide in the treatment of pulpless teeth with associated pathology. J. Br. Endod. Soc. 1975, 8, 74–93. [Google Scholar] [CrossRef] [PubMed]
- Andreasen, J.O.; Farik, B.; Munksgaard, E.C. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent. Traumatol. 2002, 18, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Witherspoon, D.E.; Small, J.C.; Regan, J.D.; Nunn, M. Retrospective analysis of open apex teeth obturated with mineral trioxide aggregate. J. Endod. 2008, 34, 1171–1176. [Google Scholar] [CrossRef] [PubMed]
- Iwaya, S.; Ikawa, M.; Kubota, M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent. Traumatol. 2001, 17, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Chueh, L.-H.; Huang, T.-J. Immature teeth with periradicular periodontitis or abscess undergoing apexogenesis: A paradigm shift. J. Endod. 2006, 32, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.T.-J. A paradigm shift in endodontic management of immature teeth: Conservation of stem cells for regeneration. J. Dent. 2008, 36, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.T.-J.; Sonoyama, W.; Liu, Y.; Liu, H.; Wang, G.S.; Shi, S. The hidden treasure in apical papilla: The potential role in pulp/dentine regeneration and bioroot engineering. J. Endod. 2008, 34, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.E.; Garcia-Godoy, F.; Hargreaves, K.M. Regenerative endodontics: A review of current status and a call for action. J. Endod. 2007, 33, 377–390. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Kahler, B.; Lin, L.M. Current developments in regenerative endodontics. Curr. Oral Health Rep. 2016, 3, 293–301. [Google Scholar] [CrossRef]
- Bezgin, T.; Sönmez, H. Review of current concepts of revascularization/revitalization. Dent. Traumatol. 2015, 31, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M. Clinical procedures for revitalization: Current knowledge and considerations. Int. Endod. J. 2016, 49, 926–936. [Google Scholar] [CrossRef] [PubMed]
- American Association of Endodontists. AAE Clinical Considerations for a Regenerative Procedure. Available online: https://www.aae.org/uploadedfiles/publications_and_research/research/currentregenerativeendodonticconsiderations.pdf (accessed on 2 December 2017).
- Fouad, A.F.; Nosrat, A. Pulp regeneration in previously infected root canal space. Endod. Top. 2013, 28, 24–37. [Google Scholar] [CrossRef]
- Nair, P.N.R. Endodontic biofilm, technology and pulpal regenerative therapy: Where do we go from here? Int. Endod. J. 2014, 47, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.M.; Shimizu, E.; Gibbs, J.L.; Loghin, S.; Ricucci, D. Histologic and histobacteriologic observations of failed revascularization/revitalization therapy: A case report. J. Endod. 2014, 40, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Kontakiotis, E.G.; Filippatos, C.G.; Tzanetakis, G.N.; Agrafioti, A. Regenerative endodontic therapy: A data analysis of clinical protocols. J. Endod. 2015, 41, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Parirokh, M.; Dummer, P.M.H. Mineral trioxide aggregate and other bioactive endodontic cements: An updated overview—Part II: Other clinical applications and complications. Int. Endod. J. 2017. [Google Scholar] [CrossRef] [PubMed]
- Topçuoğlu, G.; Topçuoğlu, H.S. Regenerative endodontic therapy in a single visit using platelet-rich plasma and Biodentine in necrotic and asymptomatic immature molar teeth: A report of 3 cases. J. Endod. 2016, 42, 1344–1346. [Google Scholar] [CrossRef] [PubMed]
- Aldakak, M.M.; Capar, I.D.; Rekab, M.S.; Abboud, S. Single-visit pulp revascularization of a nonvital immature permanent tooth using Biodentine. Iran. Endod. J. 2016, 11, 246–249. [Google Scholar] [PubMed]
- Subash, D.; Shoba, K.; Aman, S.; Bharkavi, S.K. Revitalization of an immature permanent mandibular molar with a necrotic pulp using platelet-rich fibrin: A case report. J. Clin. Diagn. Res. 2016, 10, ZD21. [Google Scholar] [CrossRef] [PubMed]
- Nosrat, A.; Asgary, S.; Eghbal, M.J.; Ghoddusi, J.; Bayat-Movahed, S. Calcium-enriched mixture cement as artificial apical barrier: A case series. J. Conserv. Dent. 2011, 14, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, S.; Kohli, M.R.; Setzer, F.; Karabucak, B. Outcome of revascularization procedure: A retrospective case series. J. Endod. 2016, 42, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, R.M.; Moule, A.J.; Podlich, H.; Kellaway, R.; Macfarlane, R.; Lewis, D.; Rowell, J. Dissolution of porcine incisor pulps in sodium hypochlorite solutions of varying compositions and concentrations. Aust. Dent. J. 2006, 51, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M.; D’Souza, R.N.; Federlin, M.; Cavender, A.C.; Hartgerink, J.D.; Hecker, S.; Schmalz, G. Dentin conditioning codetermines cell fate in regenerative endodontics. J. Endod. 2011, 37, 1536–1541. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M.; Buchalla, W.; Hiller, K.A.; Federlin, M.; Eidt, A.; Schiefersteiner, M.; Schmalz, G. Influence of root canal disinfectants on growth factor release from dentin. J. Endod. 2015, 41, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M.; Widbiller, M.; Buchalla, W.; Eidt, A.; Hiller, K.A.; Hoffer, P.C.; Schmalz, G. EDTA conditioning of dentine promotes adhesion, migration and differentiation of dental pulp stem cells. Int. Endod. J. 2016, 49, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Diogenes, A.; Henry, M.A.; Teixeira, F.B.; Hargreaves, K.M. An update on clinical regenerative endodontics. Endod. Top. 2013, 28, 2–23. [Google Scholar] [CrossRef]
- Trevino, E.G.; Patwardhan, A.N.; Henry, M.A.; Perry, G.; Dybdal-Hargreaves, N.; Hargreaves, K.M.; Diogenes, A. Effect of irrigants on the survival of human stem cells of the apical papilla in a platelet-rich plasma scaffold in human root tips. J. Endod. 2011, 37, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.E.; De Almeida, J.F.; Henry, M.A.; Khaing, Z.Z.; Schmidt, C.E.; Teixeira, F.B.; Diogenes, A. Concentration-dependent effect of sodium hypochlorite on stem cells of apical papilla survival and differentiation. J. Endod. 2014, 40, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Banchs, F.; Trope, M. Revascularization of immature permanent teeth with apical periodontitis: New treatment protocol? J. Endod. 2004, 30, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Cotti, E.; Mereu, M.; Lusso, D. Regenerative treatment of an immature, traumatized tooth with apical periodontitis: Report of a case. J. Endod. 2008, 34, 611–616. [Google Scholar] [CrossRef] [PubMed]
- Ruparel, N.B.; Teixeira, F.B.; Ferraz, C.C.; Diogenes, A. Direct effect of intracanal medicaments on survival of stem cells of the apical papilla. J. Endod. 2012, 38, 1372–1375. [Google Scholar] [CrossRef] [PubMed]
- Althumairy, R.I.; Teixeira, F.B.; Diogenes, A. Effect of dentin conditioning with intracanal medicaments on survival of stem cells of apical papilla. J. Endod. 2014, 40, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Bose, R.; Nummikoski, P.; Hargreaves, K.M. A retrospective evaluation of radiographic outcomes in immature teeth with necrotic root canal systems treated with regenerative endodontic procedures. J. Endod. 2009, 35, 1343–1349. [Google Scholar] [CrossRef] [PubMed]
- Nagata, J.Y.; de Almeida Gomes, B.P.F.; Lima, T.F.R.; Murakami, L.S.; de Faria, D.E.; Campos, G.R.; de Souza-Filho, F.J.; de Jesus Soares, A. Traumatized immature teeth treated with 2 protocols of pulp revascularization. J. Endod. 2014, 40, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Rajasekharan, S.; Martens, L.C.; Cauwels, R.G.; Verbeeck, R.M. Biodentine™ material characteristics and clinical applications: A review of the literature. Eur. Arch. Paediatr. Dent. 2014, 15, 147–1458. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J. Hydration mechanisms of mineral trioxide aggregate. Int. Endod. J. 2007, 40, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J. Characterization of hydration products of mineral trioxide aggregate. Int. Endod. J. 2008, 41, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, H.O.; Ozçelik, B.; Karabucak, B.; Cehreli, Z.C. Calcium ion diffusion from mineral trioxide aggregate through simulated root resorption defects. Dent. Traumatol. 2008, 24, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Antunes Bortoluzzi, E.; Juárez Broon, N.; Antonio Hungaro Duarte, M.; de Oliveira Demarchi, A.C.; Monteiro Bramante, C. The use of a setting accelerator and its effect on pH and calcium ion release of mineral trioxide aggregate and white Portland cement. J. Endod. 2006, 32, 1194–1197. [Google Scholar] [CrossRef] [PubMed]
- Eldeniz, A.U.; Hadimli, H.H.; Ataoglu, H.; Orstavik, D. Antibacterial effect of selected root-end filling materials. J. Endod. 2006, 32, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Tanomaru-Filho, M.; Tanomaru, J.M.; Barros, D.B.; Watanabe, E.; Ito, I.Y. In Vitro antimicrobial activity of endodontic sealers, MTA-based cements and Portland cement. J. Oral. Sci. 2007, 49, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Kuratate, M.; Yoshiba, K.; Shigetani, Y.; Yoshiba, N.; Ohshima, H.; Okiji, T. Immunohistochemical analysis of nestin, osteopontin, and proliferating cells in the reparative process of exposed dental pulp capped with mineral trioxide aggregate. J. Endod. 2008, 34, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Carmona, J.F.; Felippe, M.S.; Felippe, W.T. Biomineralization ability and interaction of mineral trioxide aggregate and white portland cement with dentin in a phosphate-containing fluid. J. Endod. 2009, 35, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part III: Clinical applications, drawbacks, and mechanism of action. J. Endod. 2010, 36, 400–413. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Hong, C.U.; McDonald, F.; Pitt Ford, T.R. Physical and chemical properties of a new root-end filling material. J. Endod. 1995, 21, 349–353. [Google Scholar] [CrossRef]
- Torabinejad, M.; Rastegar, A.F.; Kettering, J.D.; Pitt Ford, T.R. Bacterial leakage of mineral trioxide aggregate as a root-end filling material. J. Endod. 1995, 21, 109–112. [Google Scholar] [CrossRef]
- Adamo, H.L.; Buruiana, R.; Schertzer, L.; Boylan, R.J. A comparison of MTA, Super-EBA, composite and amalgam as root-end filling materials using a bacterial microleakage model. Int. Endod. J. 1999, 32, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Hong, C.U.; Pitt Ford, T.R.; Kettering, J.D. Cytotoxicity of four root end filling materials. J. Endod. 1995, 21, 489–492. [Google Scholar] [CrossRef]
- Tselnik, M.; Baumgartner, J.C.; Marshall, J.G. Bacterial leakage with mineral trioxide aggregate or a resin-modified glass ionomer used as a coronal barrier. J. Endod. 2004, 30, 782–784. [Google Scholar] [CrossRef] [PubMed]
- John, A.D.; Webb, T.D.; Imamura, G.; Goodell, G.G. Fluid flow evaluation of Fuji Triage and gray and white ProRoot mineral trioxide aggregate intraorifice barriers. J. Endod. 2008, 34, 830–832. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J.; Pitt Ford, T.R. Evaluation of the effect of tracer pH on the sealing ability of glass ionomer cement and mineral trioxide aggregate. J. Mater. Sci. Mater. Med. 2008, 19, 2941–2948. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, Y.; Ogawa, M.; Arakawa, T.; Kadowaki, T.; Saito, T. The effect of mineral trioxide aggregate on the mineralization ability of rat dental pulp cells: An in vitro study. J. Endod. 2008, 34, 1057–1060. [Google Scholar] [CrossRef] [PubMed]
- Moghaddame-Jafari, S.; Mantellini, M.G.; Botero, T.M.; McDonald, N.J.; Nör, J.E. Effect of ProRoot MTA on pulp cell apoptosis and proliferation in vitro. J. Endod. 2005, 31, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Lourenço Neto, N.; Marques, N.C.; Fernandes, A.P.; Rodini, C.O.; Sakai, V.T.; Abdo, R.C.; Machado, M.A.; Santos, C.F.; Oliveira, T.M. Immunolocalization of dentin matrix protein-1 in human primary teeth treated with different pulp capping materials. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, K.; Mistakidis, I.; Beltes, P.; Karagiannis, V. Spectrophotometric analysis of coronal discolouration induced by grey and white MTA. Int. Endod. J. 2013, 46, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Kahler, B.; Rossi-Fedele, G. A review of tooth discoloration following regenerative endodontic therapy. J. Endod. 2016, 42, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Grech, L.; Mallia, B.; Camilleri, J. Characterization of set intermediate restorative material, Biodentine, Bioaggregate and a prototypecalcium silicate cement for use as root-end filling materials. Int. Endod. J. 2013, 46, 632–641. [Google Scholar] [CrossRef] [PubMed]
- Laurent, P.; Camps, J.; De Méo, M.; Déjou, J.; About, I. Induction of specific cell responses to a Ca3SiO5-based posterior restorative material. Dent. Mater. 2008, 24, 1486–1494. [Google Scholar] [CrossRef] [PubMed]
- Tran, X.V.; Gorin, C.; Willig, C.; Baroukh, B.; Pellat, B.; Decup, F.; Opsahl Vital, S.; Chaussain, C.; Boukpessi, T. Effect of a calcium-silicate-based restorative cement on pulp repair. J. Dent. Res. 2012, 91, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Bakhtiar, H.; Esmaeili, S.; Fakhr Tabatabayi, S.; Ellini, M.R.; Nekoofar, M.H.; Dummer, P.M. Second-generation platelet concentrate (Platelet-rich fibrin) as a scaffold in regenerative endodontics: A case series. J. Endod. 2017, 43, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Yoldaş, S.E.; Bani, M.; Atabek, D.; Bodur, H. Comparison of the potential discoloration effect of Bioaggregate, Biodentine, and white Mineral Trioxide Aggregate on bovine teeth: In vitro research. J. Endod. 2016, 42, 1815–1818. [Google Scholar] [CrossRef] [PubMed]
- Shokouhinejad, N.; Nekoofar, M.H.; Pirmoazen, S.; Shamshiri, A.R.; Dummer, P.M. Evaluation and comparison of occurrence of tooth discoloration after the application of various calcium silicate-based cements: An ex vivo study. J. Endod. 2016, 42, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Marconyak, L.J., Jr.; Kirkpatrick, T.C.; Roberts, H.W.; Roberts, M.D.; Aparicio, A.; Himel, V.T.; Sabey, K.A. A comparison of coronal tooth discoloration elicited by various endodontic reparative materials. J. Endod. 2016, 42, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Dawood, A.E.; Parashos, P.; Wong, R.H.K.; Reynolds, E.C.; Manton, D.J. Calcium silicate-based cements: Composition, properties, and clinical applications. J. Investig. Clin. Dent. 2017, 8, e12195. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Shahabi, S.; Jafarzadeh, T.; Amini, S.; Kheirieh, S. The properties of a new endodontic material. J. Endod. 2008, 34, 990–993. [Google Scholar] [CrossRef] [PubMed]
- EndoSequence BC Obturation Kit BC RRM-Fast Set Putty™. Available online: http://brasselerusadental.com/wp-content/uploads/sites/9/2015/03/B_4164_Endosquence-Fast-Set-Putty-Syringe.pdf (accessed on 2 December 2017).
- Paryani, K.; Kim, S.G. Regenerative endodontic treatment of permanent teeth after completion of root development: A report of 2 cases. J. Endod. 2013, 39, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Chaniotis, A. The use of MTA/blood mixture to induce hard tissue healing in a root fractured maxillary central incisor. Case report and treatment considerations. Int. Endod. J. 2014, 47, 989–999. [Google Scholar] [CrossRef] [PubMed]
- Saoud, T.M.; Martin, G.; Chen, Y.H.; Chen, K.L.; Chen, C.A.; Songtrakul, K.; Malek, M.; Sigurdsson, A.; Lin, L.M. Treatment of mature permanent teeth with necrotic pulps and apical periodontitis using regenerative endodontic procedures: A case series. J. Endod. 2016, 42, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Saoud, T.M.; Mistry, S.; Kahler, B.; Sigurdsson, A.; Lin, L.M. Regenerative endodontic procedures for traumatized teeth after horizontal root fracture, avulsion, and perforating root resorption. J. Endod. 2016, 42, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahler, B.; Chugal, N.; Lin, L.M. Alkaline Materials and Regenerative Endodontics: A Review. Materials 2017, 10, 1389. https://doi.org/10.3390/ma10121389
Kahler B, Chugal N, Lin LM. Alkaline Materials and Regenerative Endodontics: A Review. Materials. 2017; 10(12):1389. https://doi.org/10.3390/ma10121389
Chicago/Turabian StyleKahler, Bill, Nadia Chugal, and Louis M. Lin. 2017. "Alkaline Materials and Regenerative Endodontics: A Review" Materials 10, no. 12: 1389. https://doi.org/10.3390/ma10121389
APA StyleKahler, B., Chugal, N., & Lin, L. M. (2017). Alkaline Materials and Regenerative Endodontics: A Review. Materials, 10(12), 1389. https://doi.org/10.3390/ma10121389




