First-Row Transition Metal Doping in Calcium Phosphate Bioceramics: A Detailed Crystallographic Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sol-Gel Elaboration of M-Doped BCP Samples
2.2. X-ray Powder Diffraction (XRPD) and Rietveld Analyses
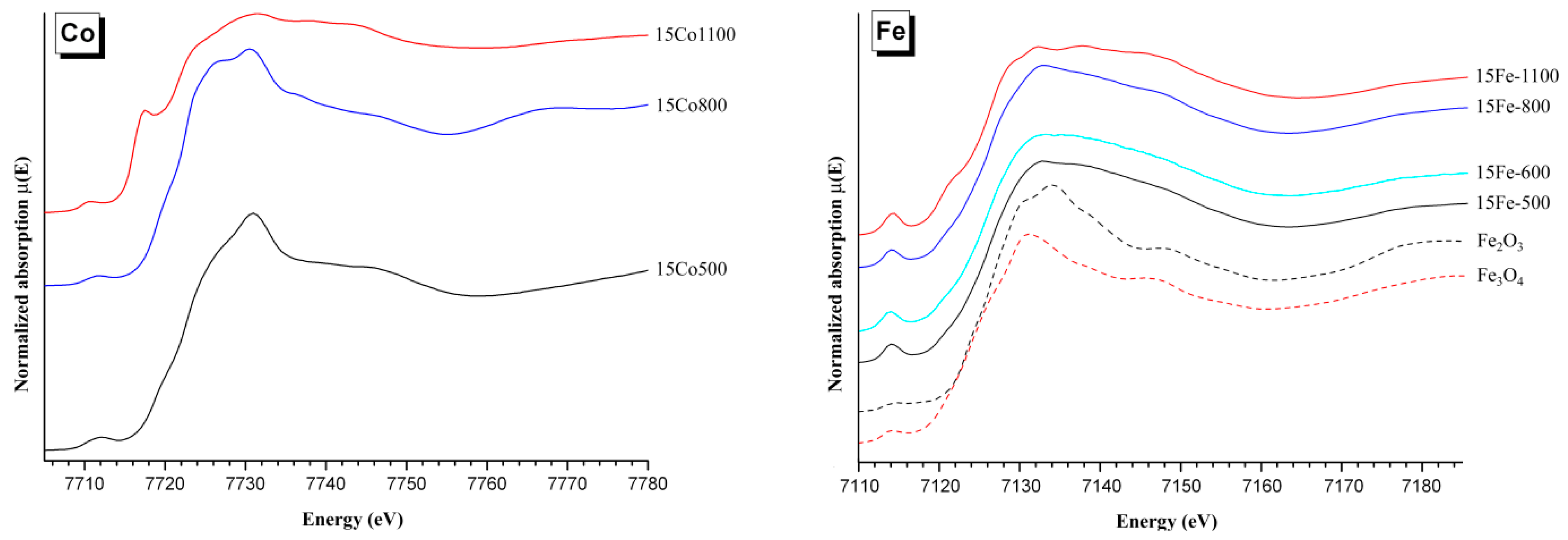
2.3. X-ray Absorption Spectroscopy (XAS)
3. Results
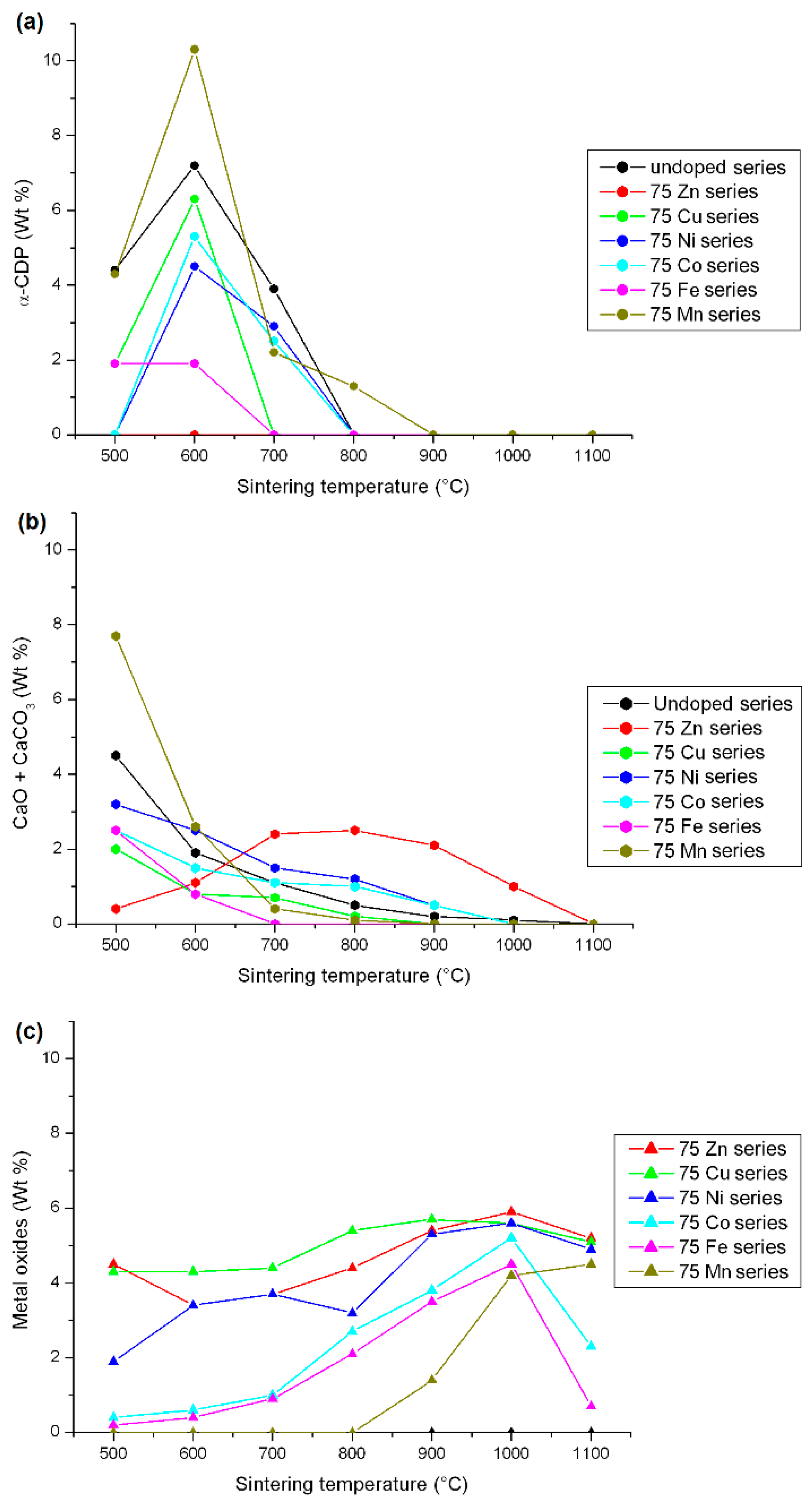
3.1. Mineral Composition of the M-Doped BCP Samples
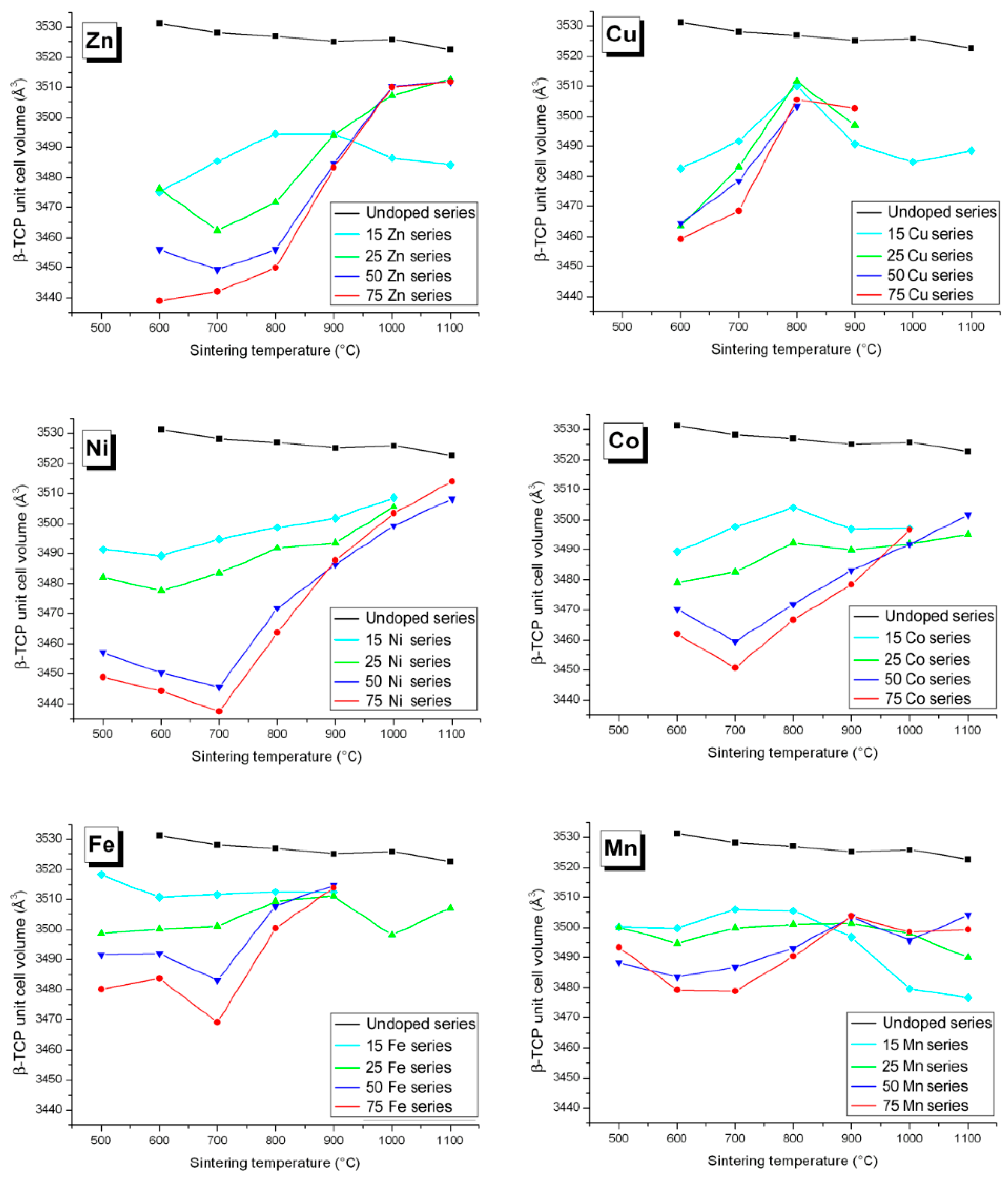
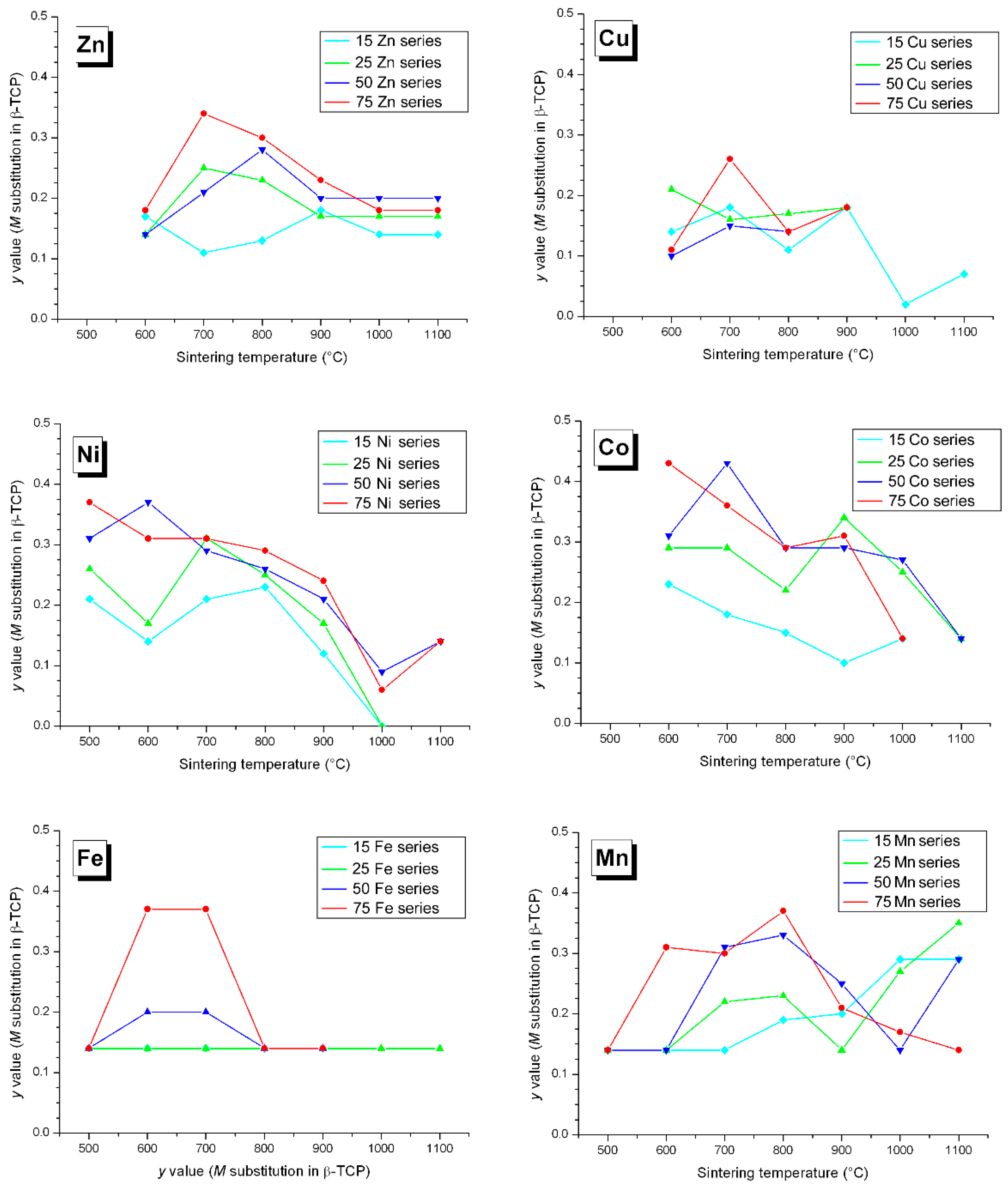
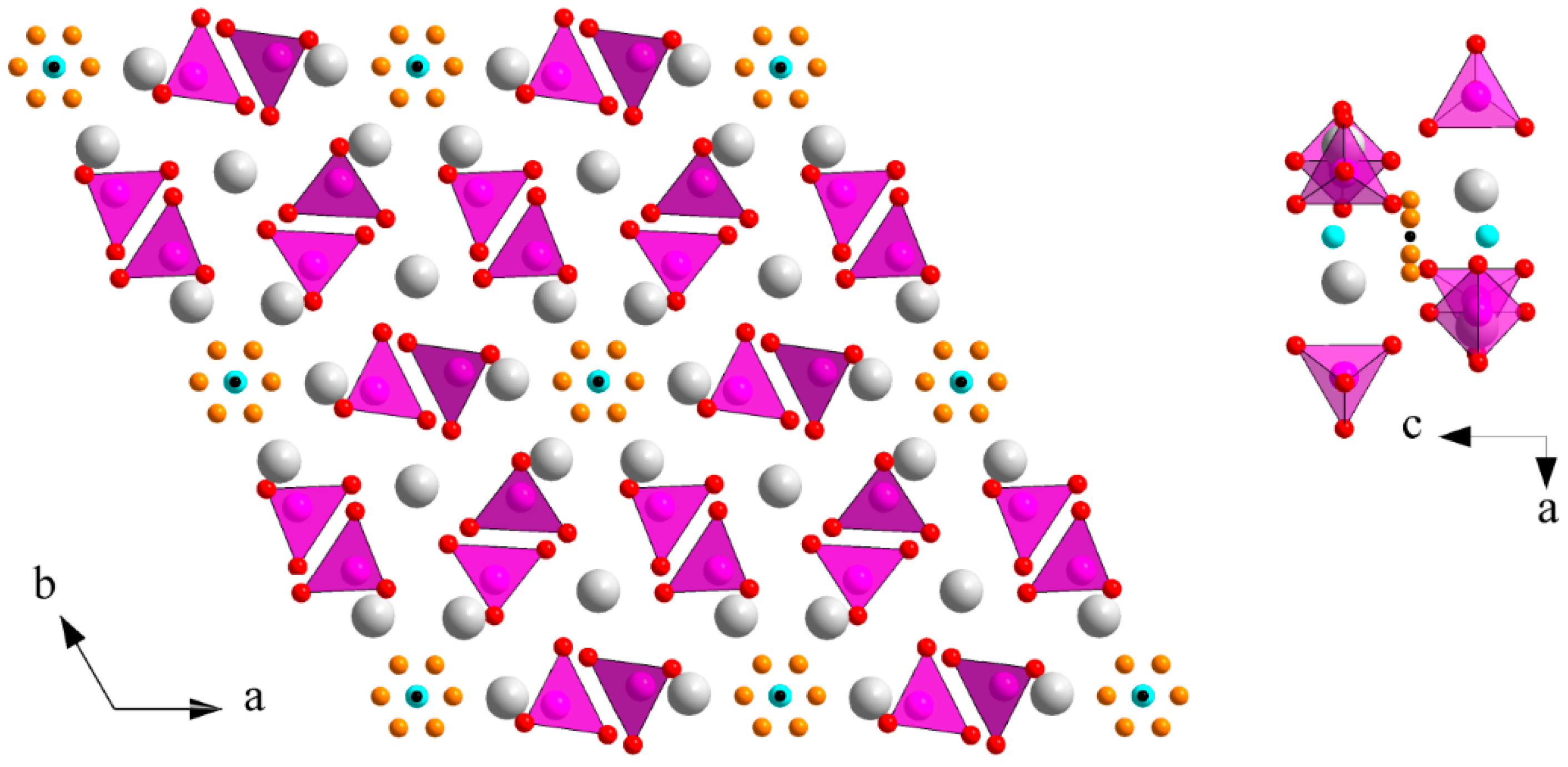
3.2. M Incorporation into the β-TCP Structure
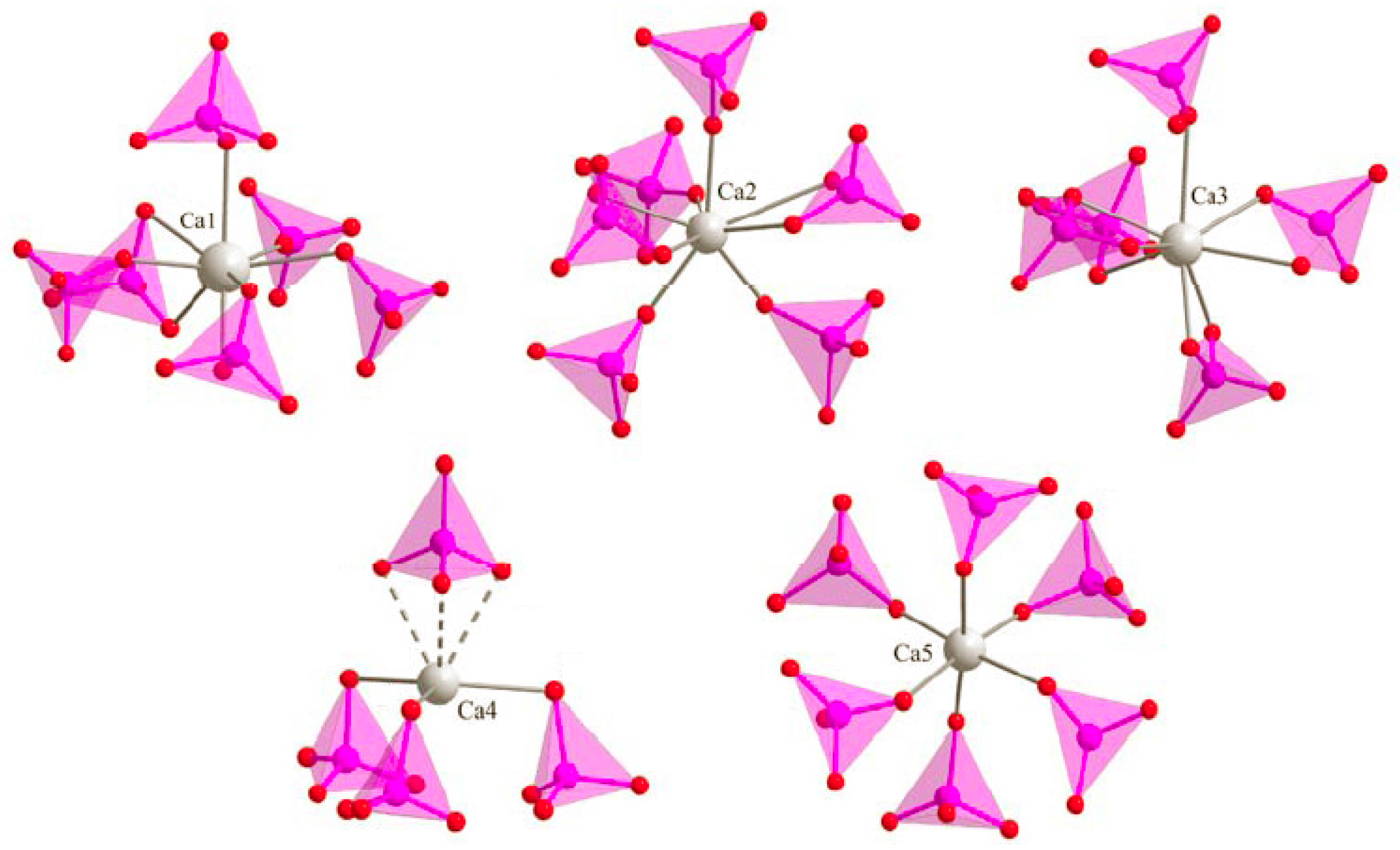
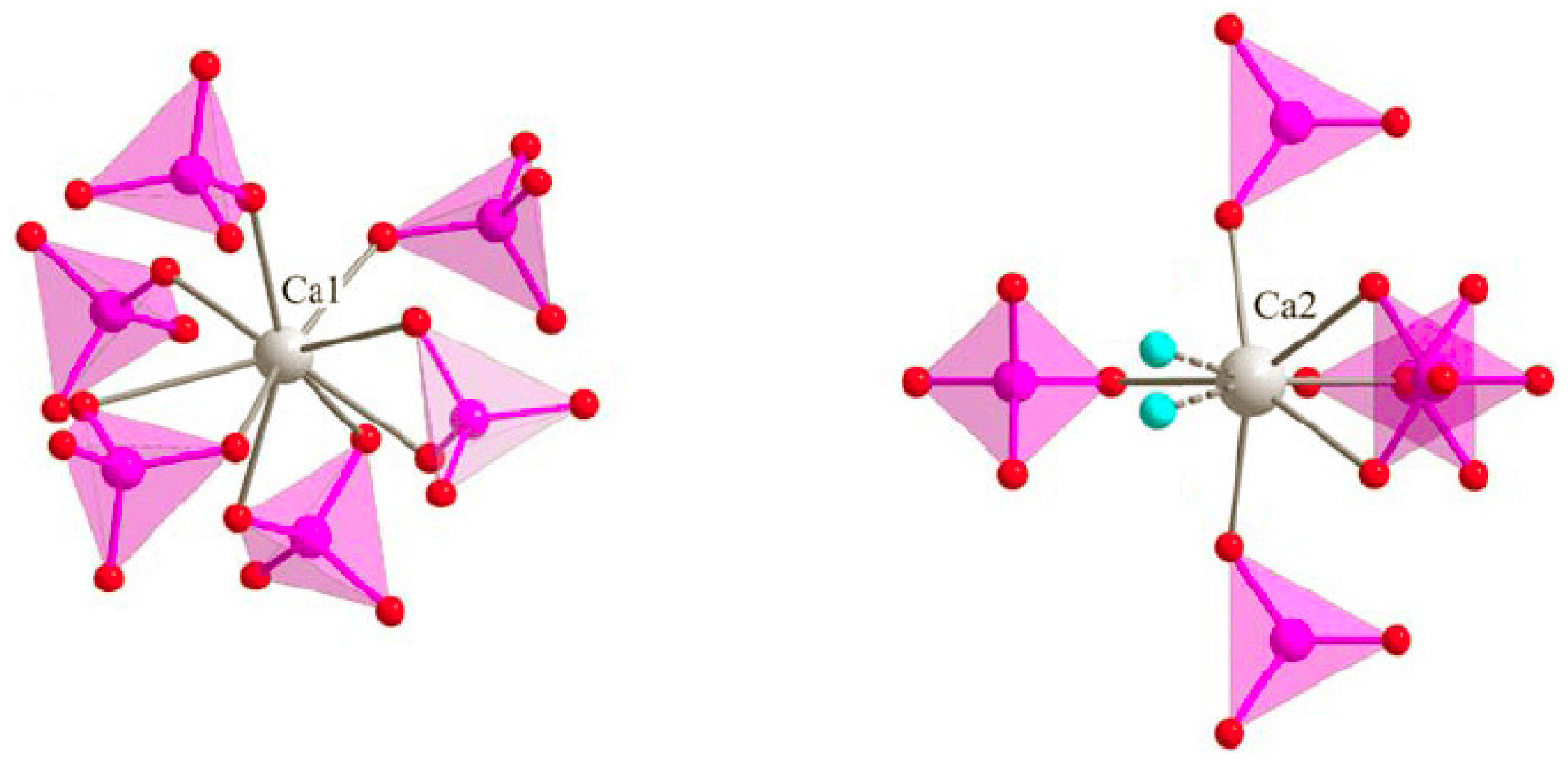
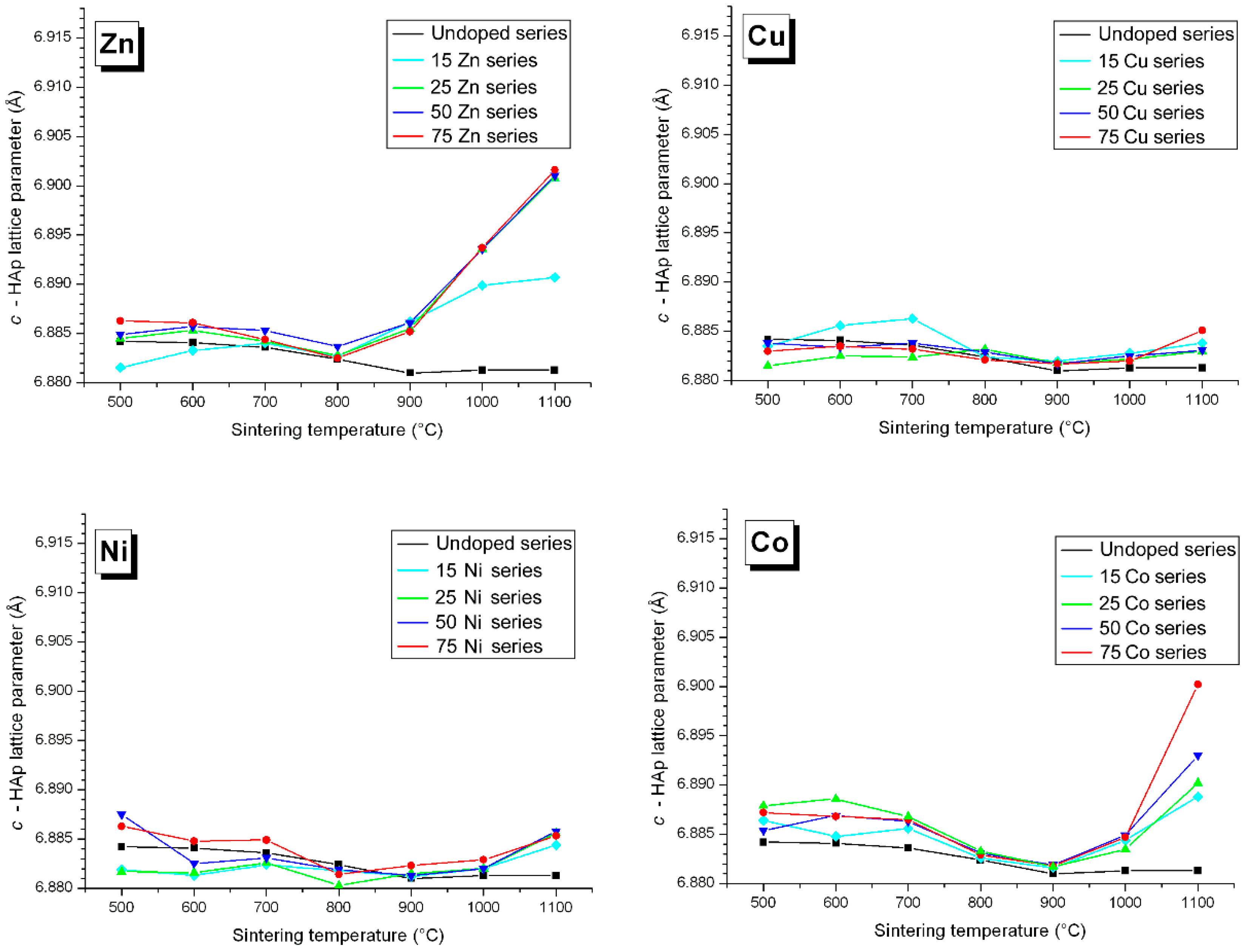
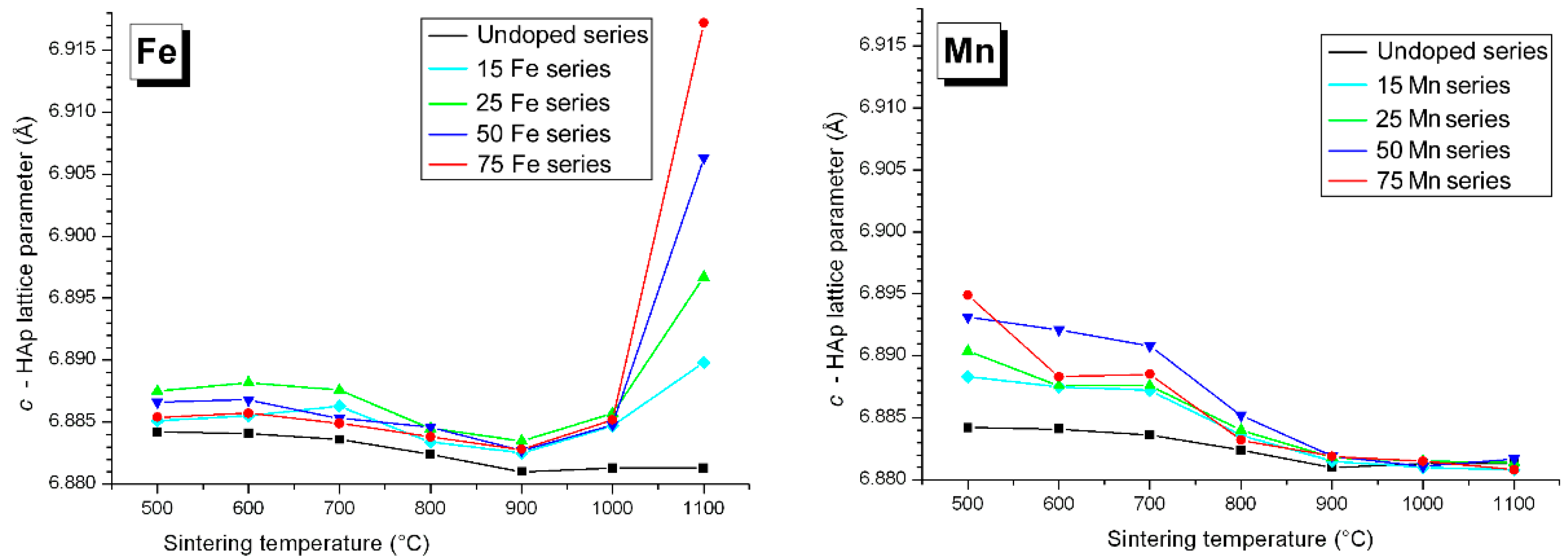
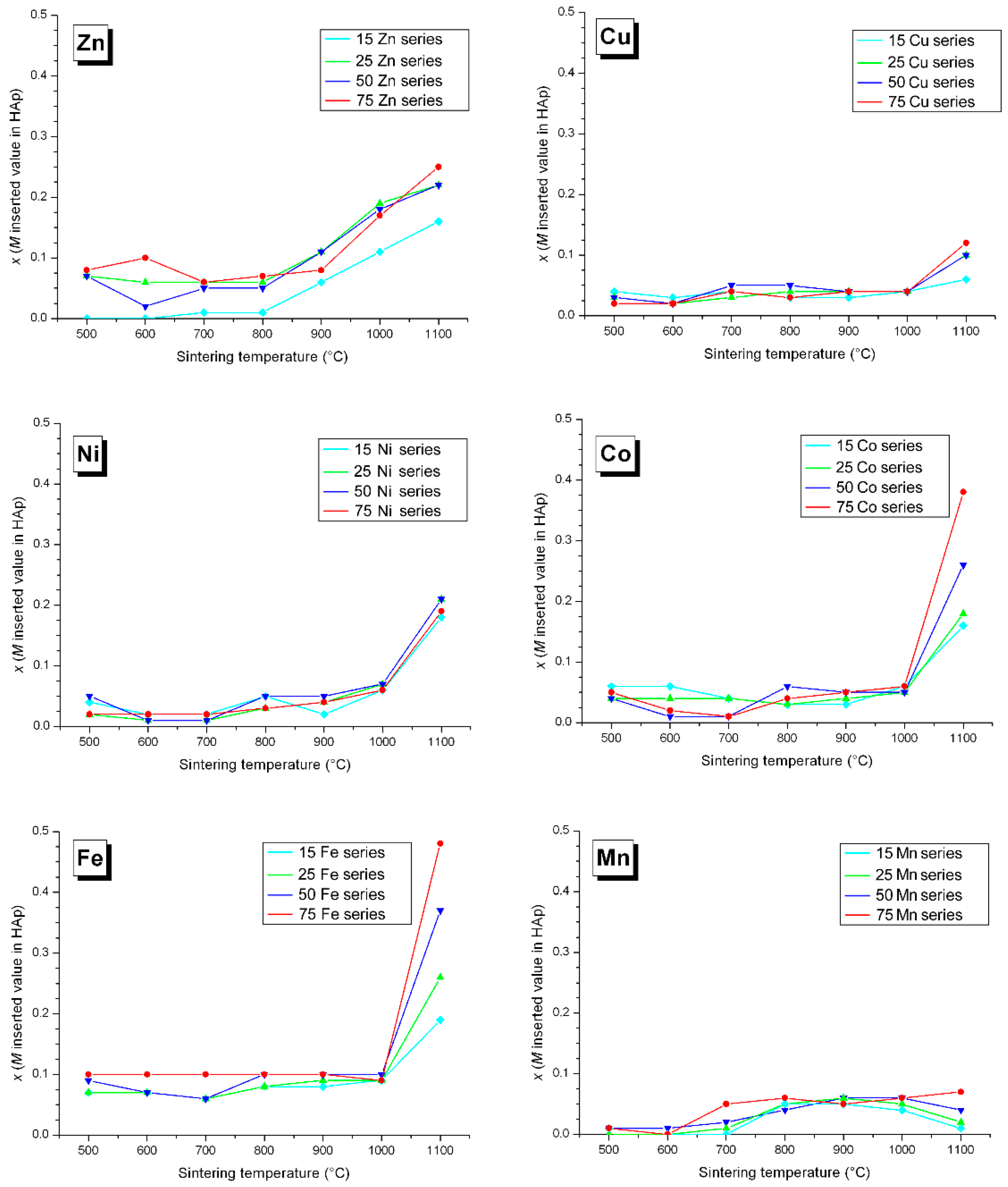
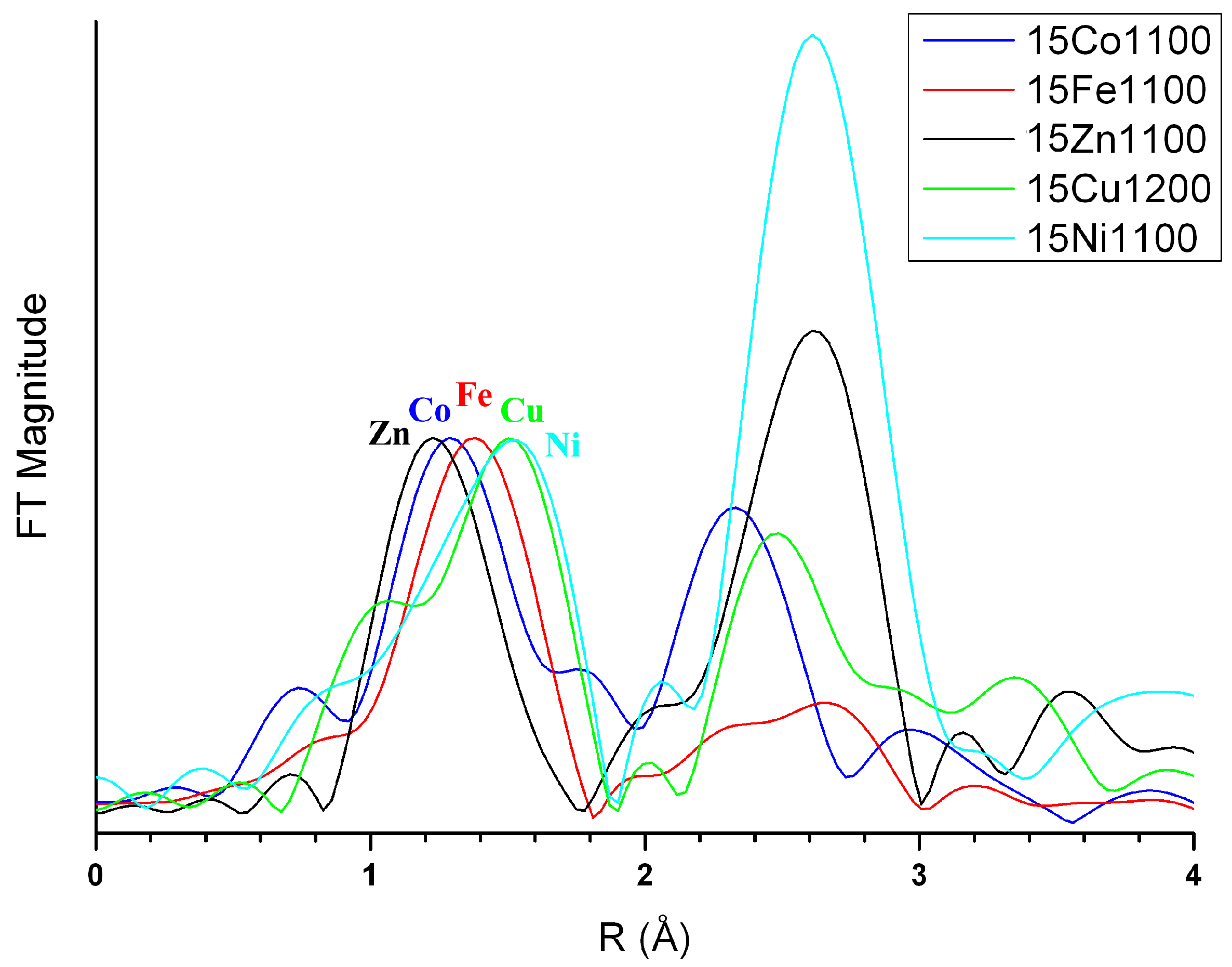
3.3. M Incorporation into the HAp Structure
4. Discussion
4.1. Iron and Cobalt Doping Mechanism
4.2. Nickel Doping Mechanism
4.3. Copper and Manganese Doping Mechanisms
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Supova, M. Substituted hydroxyapatites for biomedical applications: A review. Ceram. Int. 2015, 41, 9203–9231. [Google Scholar] [CrossRef]
- Kolmas, J.; Krukowski, S.; Laskus, A.; Jurkitewicz, M. Synthetic hydroxyapatite in pharmaceutical applications. Ceram. Int. 2016, 42, 2472–2487. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphates in nature. Biol. Med. Mater. 2009, 2, 399–498. [Google Scholar]
- Skinner, H.C.W. Biominerals. Miner. Mag. 2005, 69, 621–641. [Google Scholar] [CrossRef]
- Dorozhkin, S.V.; Epple, M. Biological and medical significance of calcium phosphates. Angew. Chem. Int. Ed. 2002, 41, 3130–3146. [Google Scholar] [CrossRef]
- Daculsi, G.; LeGeros, R.Z.; Nery, E.; Lynch, K.; Kerebel, B. Transformation of biphasic calcium phosphate ceramics in vivo: Ultrastructural and physicochemical characterization. J. Biomed. Mater. Res. 1989, 23, 883–894. [Google Scholar] [CrossRef] [PubMed]
- Calvo, C.; Gopal, R. The crystal structure of whitlockite from the Palermo Quarry. Am. Mineral. 1975, 60, 120–133. [Google Scholar]
- LeGeros, R.Z. Calcium Phosphates in Oral Biology and Medicine; Karger Publisher: Basel, Switzerland, 1991. [Google Scholar]
- Hesse, A.; Heimbach, D. Causes of phosphate stone formation and the importance of metaphylaxis by urinary acidification: A review. World J. Urol. 1999, 17, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Lagier, R.; Baud, C.A. Magnesium whitlockite, a calcium phosphate crystal of special interest in pathology. Pathol. Res. Pract. 2003, 199, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Renaudin, G.; Laquerrière, P.; Filinchuk, Y.; Jallot, E.; Nedelec, J.-M. Structural characterization of sol-gel derived Sr-substituted calcium phosphates with anti-osteoporotic and anti-inflammatory properties. J. Mater. Chem. 2008, 18, 3593–3600. [Google Scholar] [CrossRef]
- Renaudin, G.; Jallot, E.; Nedelec, J.-M. Effect of strontium substitution on the composition and microstructure of sol-gel derived calcium phosphate. J. Sol Gel Sci. Technol. 2009, 51, 287–294. [Google Scholar] [CrossRef]
- Gomes, S.; Renaudin, G.; Jallot, E.; Nedelec, J.-M. Structural characterization and biological fluid interaction of sol-gel derived Mg-substituted biphasic calcium phosphate ceramics. Appl. Mater. Interfaces 2009, 1, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Gomes, S.; Nedelec, J.-M.; Jallot, E.; Sheptyakov, D.; Renaudin, G. Unexpected mechanism of Zn2+ insertion in calcium phosphate bioceramics. Chem. Mater. 2011, 23, 3072–3085. [Google Scholar] [CrossRef]
- Gomes, S.; Nedelec, J.-M.; Renaudin, G. On the effect of temperature on the insertion of zinc into hydroxyapatite. Acta Biomater. 2012, 8, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Gomes, S.; Kaur, A.; Nedelec, J.-M.; Renaudin, G. X-ray absorption spectroscopy shining (synchrotron) light onto the insertion of Zn2+ in calcium phosphate ceramics and its influence on their behaviour under biological conditions. J. Mater. Chem. B 2014, 2, 536–545. [Google Scholar] [CrossRef]
- Yamaguchi, M. Role of zinc in bone formation and bone resorption. J. Trace Elem. Exp. Med. 1998, 11, 119–135. [Google Scholar] [CrossRef]
- Moonga, B.S.; Dempster, D.W. Zinc is a potent inhibitor of osteoclastis bone resorption in vitro. J. Bone Miner. Res. 1995, 10, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Heidenau, F.; Mittelmeier, W.; Detsch, R.; Haenle, M.; Stenzel, F.; Ziegler, G.; Gollwitzer, H. A novel antibacterial titania coating: Metal ion toxicity and in vitro surface colonization. J. Mater. Sci. Mater. Med. 2005, 16, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Rameshbabu, N.; Sampath Kumar, T.S.; Prabhakar, T.G.; Sastry, V.S.; Murty, K.V.G.K.; Prasad Rao, K. Antibacterial nanosized silver substituted hydroxyapatite: Synthesis and characterization. J. Biomed. Mater. Res. A 2007, 80, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Sygnatowicz, M.; Keyshar, K.; Tiwari, A. Antimicrobial properties of silver-doped hydroxyapatite nano-powders and thin films. JOM J. Min. Met. Mater. Soc. 2010, 62, 65–70. [Google Scholar] [CrossRef]
- Ewald, A.; Hösel, D.; Patel, S.; Grover, L.M.; Barralet, J.E.; Gbureck, U. Silver-doped calcium phosphate cements with antimicrobial activity. Acta Biomater. 2011, 7, 4064–4070. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, C.S.; Massuyeau, F.; Constantin, L.V.; Predoi, D. Structural and physical properties of antibacterial Ag-doped nano-hydroxyapatite synthesized at 100 °C. Nanoscale Res. Lett. 2011, 6, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, C.S.; Andronescu, E.; Vasile, B.S.; Valsangiacom, C.M.; Ghita, R.V.; Predoi, D. Synthesis and antimicrobial activity of silver-doped hydroxyapatite nanoparticules. Optoelectron. Adv. Mater. 2010, 4, 1515–1519. [Google Scholar]
- Wang, X.; Cheng, F.; Liu, J.; Smått, J.-H.; Gepperth, D.; Lastusaari, M.; Xu, C.; Hupa, L. Biocomposites of copper-containing mesoporous bioactive glass and nanofibrillated cellulose: Biocompatibility and angiogenic promotion in chronic wound healing application. Acta Biomater. 2016, 46, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Popescu, R.A.; Magyari, K.; Vulpoi, A.; Trandafir, D.L.; Licarete, E.; Todea, M.; Ştefan, R.; Voica, C.; Vodnar, D.C.; Simon, S.; et al. Bioactive and biocompatible copper containing glass-ceramics with remarkable antibacterial properties and high cell viability designed for future in vivo trials. Biomater. Sci. 2016, 4, 1252–1265. [Google Scholar] [CrossRef] [PubMed]
- Bracci, B.; Torricelli, P.; Panzavolta, S.; Boanini, E.; Giordino, R.; Bigi, A. Effect Mg2+, Sr2+ and Mn2+ on the chemico-physical and in vitro biological properties of calcium phosphate biomimetic coatings. J. Inorg. Biochem. 2009, 103, 1666–1674. [Google Scholar] [CrossRef] [PubMed]
- Mayer, I.; Cuisinier, F.J.G.; Gdalya, S.; Popov, I. TEM study of the morphology of Mn2+-doped calcium hydroxyapatite and β-tricalcium phosphate. J. Inorg. Biochem. 2008, 102, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Shinkai, M.; Honda, H.; Kobayashi, T. Medical application of functionalized magnetic nanoparticles. J. Biosci. Bioeng. 2005, 100, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, S.; Veselinovic, L.; Markovic, S.; Ignjatovic, N.; Uskokovic, D. Hydrothermal synthesis of nanosized pure and cobalt-exchanged hydroxyapatite. Mater. Manuf. Process. 2009, 24, 1096–1103. [Google Scholar] [CrossRef]
- Pon-On, W.; Meejoo, S.; Tang, M. Incorporation of iron into nano hydroxyapatite particles synthesized by the microwave process. Int. J. Nanosci. 2007, 6, 9–16. [Google Scholar] [CrossRef]
- Chandra, V.S.; Baskar, G.; Suganthi, R.V.; Elayaraja, K.; Joshy, M.I.A.; Beaula, W.S.; Mythili, R.; Venkatraman, G.; Kalkura, S.N. Blood compatibility of iron-doped nanosize hydroxyapatite and its drug release. Appl. Mater. Interfaces 2012, 4, 1200–1210. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Widodo, J.; Lim, S.; Ooi, C.P. Synthesis and cytocompatibility of manganese (II) and iron (III) substituted hydroxyapatite nanoparticles. J. Mater. Sci. 2012, 47, 754–763. [Google Scholar] [CrossRef]
- Gomes, S.; Kaur, A.; Grenèche, J.-M.; Nedelec, J.-M.; Renaudin, G. Atomic scale modeling of iron-doped biphasic calcium phosphate bioceramics. Acta Biomater. 2016. [Google Scholar] [CrossRef] [PubMed]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Yashima, M.; Sakai, A.; Kamiyama, T.; Hoshikawa, A. Crystal structure analysis of beta-tricalcium phosphate Ca3(PO4)2 by neutron powder diffraction. J. Solid State Chem. 2003, 175, 272–277. [Google Scholar] [CrossRef]
- Rodriguez-Carvajal, J. PROGRAM FullProf.2k—Version 3.20; Laboratoire Léon Brillouin (CEA-CNRS): Saclay, France, 2005. [Google Scholar]
- McCusker, L.B.; Von Dreele, R.B.; Cox, D.E.; Louër, D.; Scardi, P. Rietveld refinement guidelines. J. Appl. Crystallogr. 1999, 32, 36–50. [Google Scholar] [CrossRef]
- Hill, R.J. Rietveld refinement round-robin. I. Analysis of standard X-ray and neutron data for PbSO4. J. Appl. Crystallogr. 1992, 25, 589–610. [Google Scholar] [CrossRef]
- Scarlett, N.V.Y.; Madsen, I.C.; Cranswick, L.M.D.; Lwin, T.; Groleau, E.; Stephenson, G.; Aylmore, M.; Agron-Olshina, N. Outcomes of the international union of crystallography commission on powder diffraction round robin on quantitative phase analysis: Samples 2, 3, 4, synthetic bauxite, natural granodiorite and pharmaceuticals. J. Appl. Crystallogr. 2002, 35, 383–400. [Google Scholar] [CrossRef]
- Newville, N. IFFEFIT: Interactive EXAFS analysis and FEFF fitting. J. Synchrotron Radiat. 2001, 8, 322–324. [Google Scholar] [CrossRef] [PubMed]
- Ravel, B. ATOMS: Crystallography for X-ray absorption spectroscopist. J. Synchrotron Radiat. 2001, 8, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Rehr, J.J.; Mustre de Leon, J.; Zabinsky, S.I.; Albers, R.C. Theoretical X-ray absorption fine structure standards. J. Am. Chem. Soc. 1991, 113, 5135–5145. [Google Scholar] [CrossRef]
- Rodriguez-Lorenzo, L.M.; Hart, J.N.; Gross, K.A. Structural and chemical analysis of well-crystallized hydroxyfluorapatites. J. Phys. Chem. B 2003, 107, 8316–8320. [Google Scholar] [CrossRef]
- Kazin, P.E.; Gazizova, O.R.; Karpov, A.S.; Jansen, M.; Tretyakov, Y.D. Incorporation of 3d-metal ions in the hexagonal channels of the Sr5(PO4)3OH apatite. Solid State Sci. 2007, 9, 82–87. [Google Scholar] [CrossRef]
- Bernhardt, F.; Hoppe, R. Das erste Oxocobaltat des Typs A2CoIIO2: K2CoO2 = K4[OCoO2CoO]. Z. Anorg. Allg. Chem. 1994, 620, 586–591. [Google Scholar] [CrossRef]
- Vialat, P.; Mousty, C.; Taviot-Gueho, C.; Renaudin, G.; Martinez, H.; Dupin, J.-C.; Elkaim, E.; Leroux, F. High-performing monometallic cobalt layered double hydroxide supercapacitor with defined local structure. Adv. Funct. Mater. 2014, 24, 4831–4842. [Google Scholar] [CrossRef]
- Yoon, W.-S.; Kim, K.-B.; Kim, M.-G.; Lee, M.-K.; Shin, H.-J.; Lee, J.-M.; Lee, J.-S.; Yo, C.-H. Oxygen Contribution on Li-Ion Intercalation–Deintercalation in LiCoO2 Investigated by O K-Edge and Co L-Edge X-ray Absorption Spectroscopy. J. Phys. Chem. B 2002, 106, 2526–2532. [Google Scholar] [CrossRef]
- Rieck, H.; Hoppe, R. Über Oxoniccolate (II) der Alkalimetalle: K2NiO2, Rb2NiO2 und Cs2NiO2. Z. Anorg. Allg. Chem. 1973, 400, 311–320. [Google Scholar] [CrossRef]
- Hoppe, R.; Baier, R.; Carl, W.; Glaum, H.; Untenecker, H. Zum Aufbau von A2[HgO2] (A = K, Rb), Rb2[NiO2] und Rb1,4K0,6[NiO2] [1–4]. Z. Anorg. Allg. Chem. 1988, 567, 69–76. [Google Scholar] [CrossRef]
- Mayer, I.; Jacobsohn, O.; Niazov, T.; Werckmann, J.; Iliescu, M.; Richard-Plouet, M.; Burghaus, O.; Reinen, D. Manganese in precipitated hydroxyapatites. Eur. J. Inorg. Chem. 2003, 7, 1445–1451. [Google Scholar] [CrossRef]
- Suitch, P.R.; LaCout, J.L.; Hewat, A.; Young, R.A. The structural location and role of Mn2+ partially substituted for Ca2+ in fluorapatite. Acta Crystallogr. B 1985, 41, 173–179. [Google Scholar] [CrossRef]





| Coordination | Mn2+ | Mn3+ | Fe3+ | Co2+ | Co3+ | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| LS | HS | LS | HS | LS | HS | LS | HS | LS | HS | |
| II | - | - | - | - | - | - | - | - | - | - |
| IV | - | 0.66 | - | - | - | 0.49 | - | 0.58 | - | - |
| VI | 0.67 | 0.83 | 0.58 | 0.64 | 0.55 | 0.64 | 0.65 | 0.74 | 0.54 | 0.61 |
| VII | - | 0.90 | - | - | - | - | - | |||
| VIII | 0.96 | - | - | 0.78 | 0.90 | - | ||||
| IX | - | - | - | - | - | |||||
| Coordination | Ni2+ | Cu+ | Cu2+ | Zn2+ | Ca2+ | |||||
| II | - | 0.46 | - | - | - | |||||
| IV | 0.55 (0.49 *) | 0.60 | 0.57 (0.57 *) | 0.60 | - | |||||
| VI | 0.69 | 0.77 | 0.73 | 0.74 | 1.00 | |||||
| VII | - | - | - | - | 1.06 | |||||
| VIII | - | - | - | 0.90 | 1.12 | |||||
| IX | - | - | - | - | 1.18 | |||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renaudin, G.; Gomes, S.; Nedelec, J.-M. First-Row Transition Metal Doping in Calcium Phosphate Bioceramics: A Detailed Crystallographic Study. Materials 2017, 10, 92. https://doi.org/10.3390/ma10010092
Renaudin G, Gomes S, Nedelec J-M. First-Row Transition Metal Doping in Calcium Phosphate Bioceramics: A Detailed Crystallographic Study. Materials. 2017; 10(1):92. https://doi.org/10.3390/ma10010092
Chicago/Turabian StyleRenaudin, Guillaume, Sandrine Gomes, and Jean-Marie Nedelec. 2017. "First-Row Transition Metal Doping in Calcium Phosphate Bioceramics: A Detailed Crystallographic Study" Materials 10, no. 1: 92. https://doi.org/10.3390/ma10010092
APA StyleRenaudin, G., Gomes, S., & Nedelec, J.-M. (2017). First-Row Transition Metal Doping in Calcium Phosphate Bioceramics: A Detailed Crystallographic Study. Materials, 10(1), 92. https://doi.org/10.3390/ma10010092







