Environment-Friendly Heterogeneous Alkaline-Based Mixed Metal Oxide Catalysts for Biodiesel Production
Abstract
:1. Introduction
2. Methods
2.1. Preparation of Mixed Metal Oxide (MMO) Catalysts
2.2. Characterization of MMO Catalysts
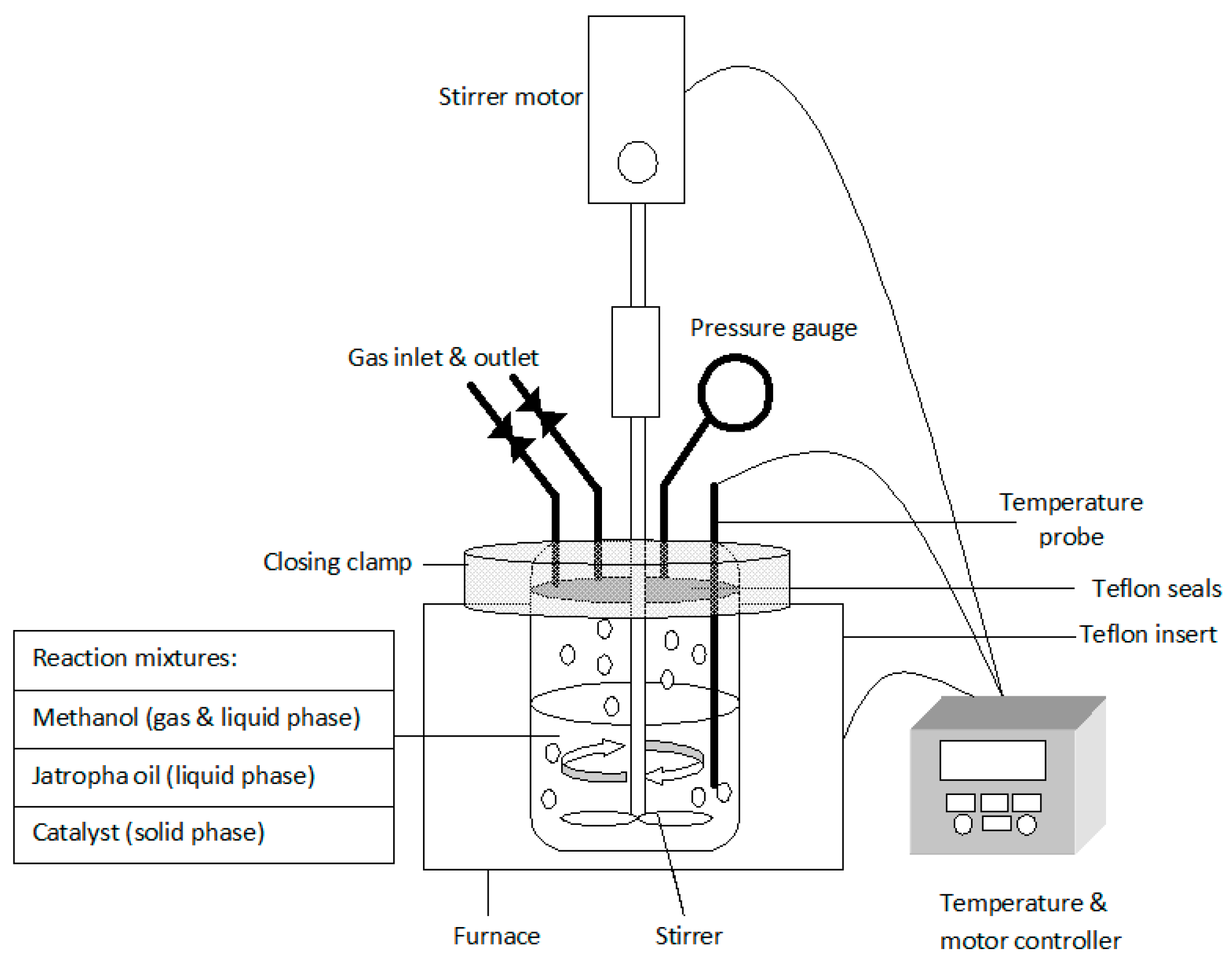
2.3. Transesterification Reaction of High Acid Jatropha Oil
3. Results and Discussion
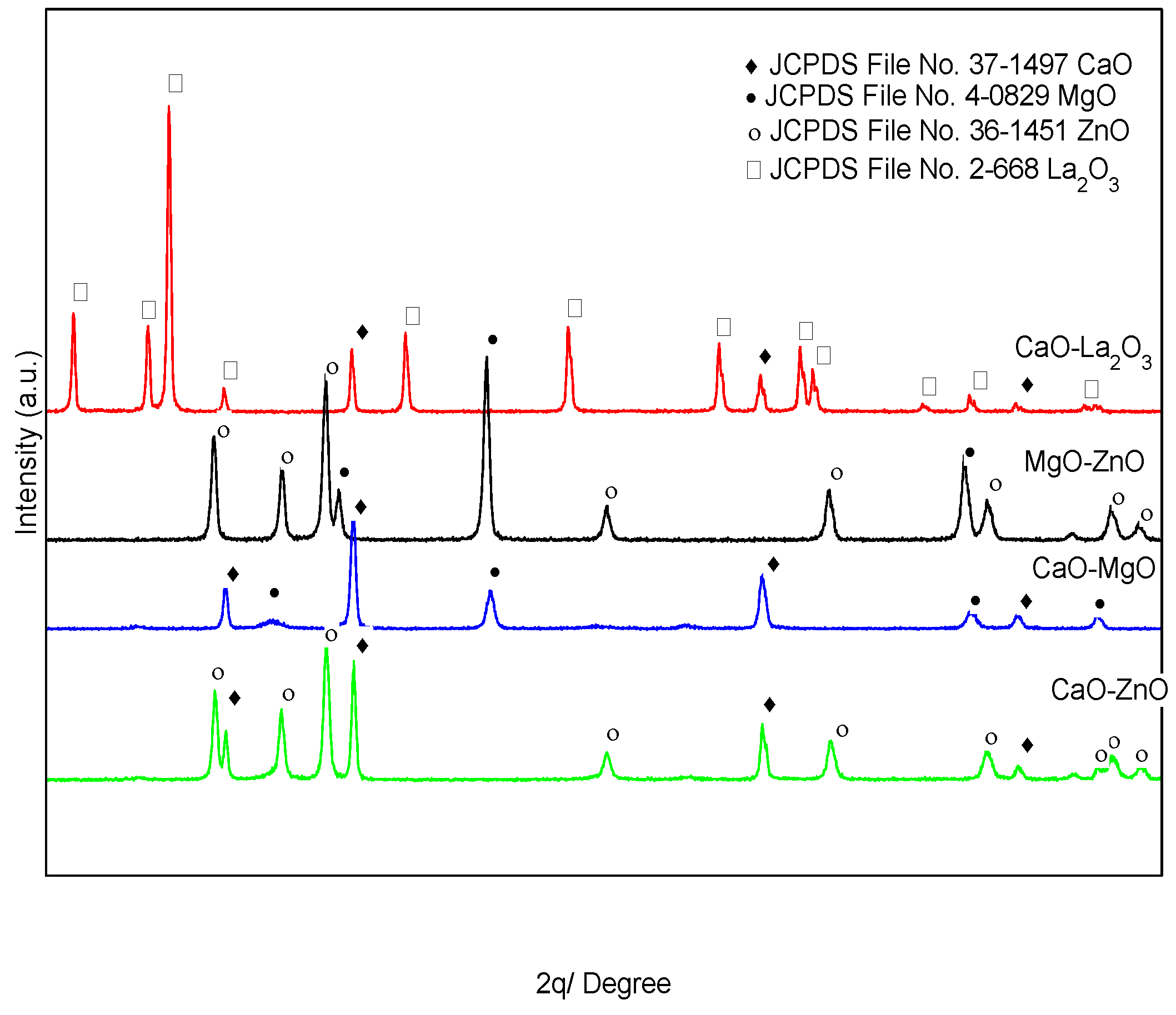
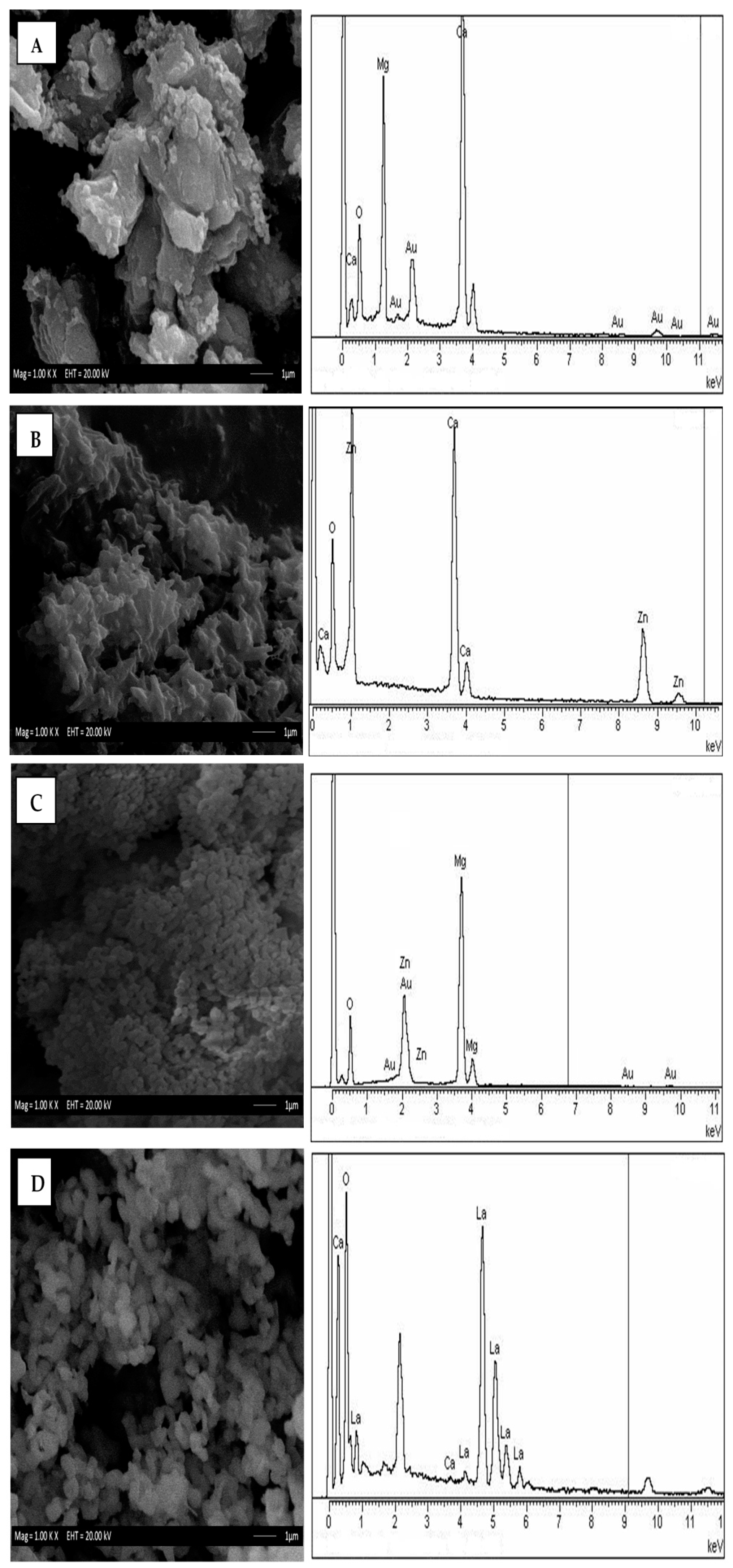
3.1. Physicochemical Properties of Mixed Metal Oxide (MMO) Catalysts
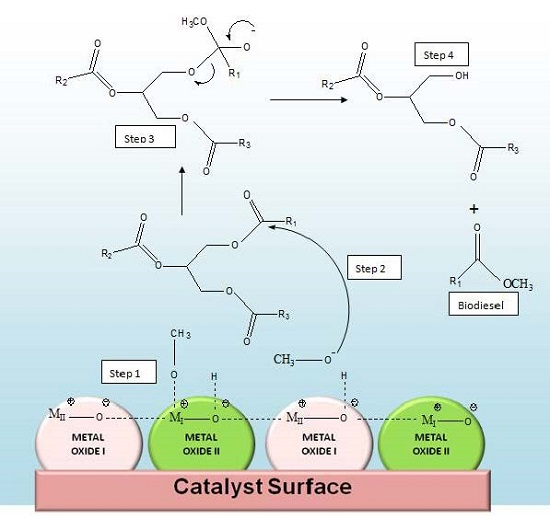
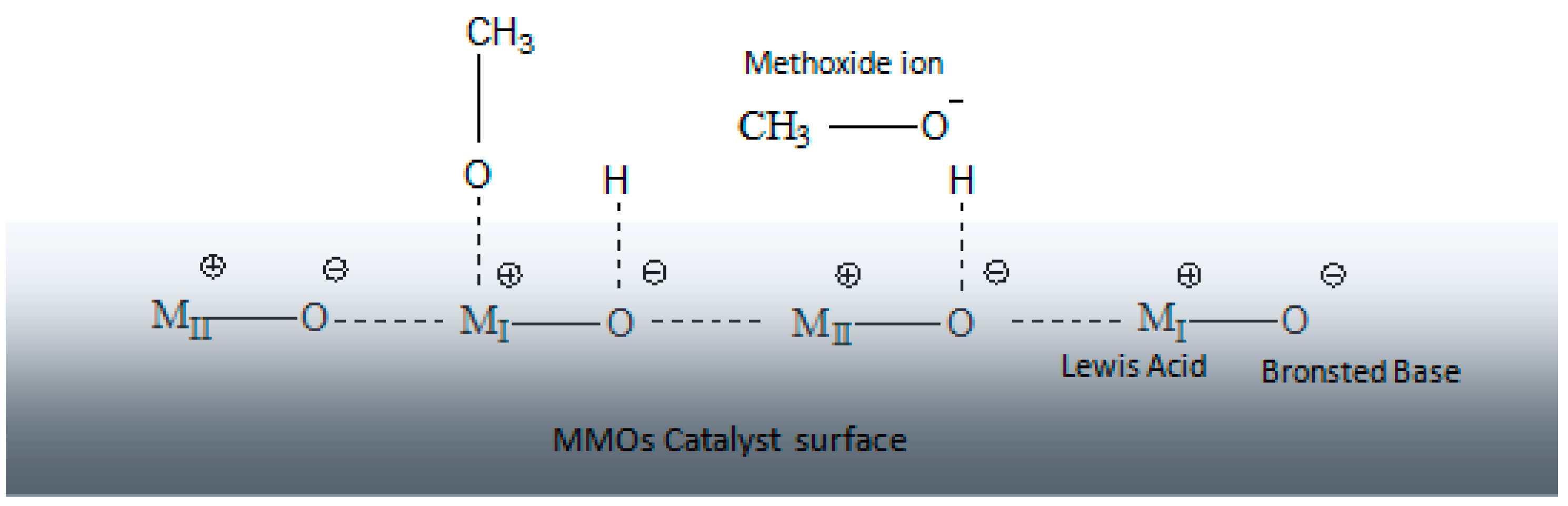
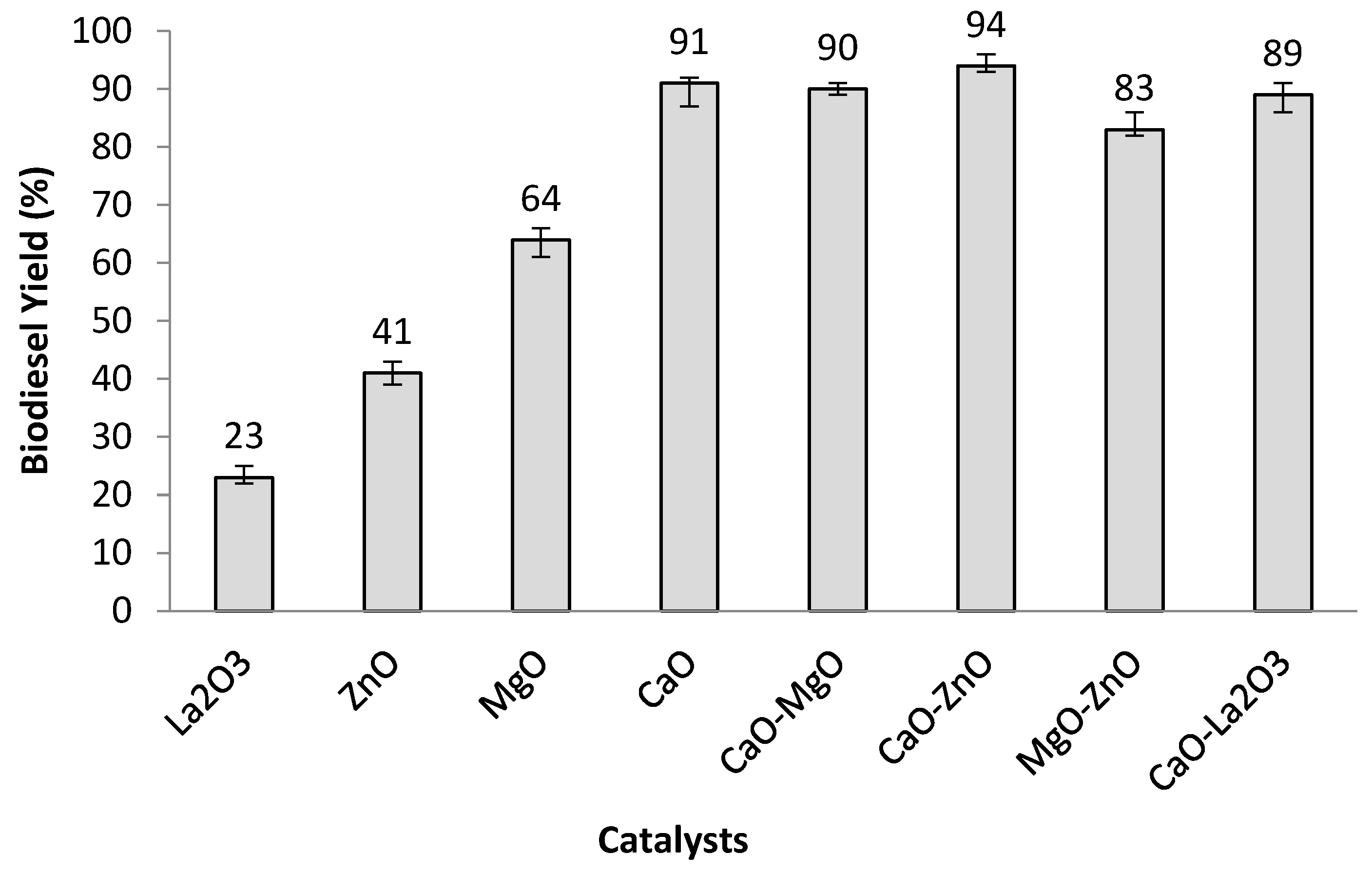
3.2. Transesterification Activity of the MMO Catalysed Reaction
3.3. Catalyst Reusability
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Luque, R.; Lovett, J.C.; Datta, B.; Clancy, J.; Campelo, J.M.; Romero, A.A. Biodiesel as feasible petrol fuel replacement: A multidisciplinary overview. Energy Environ. Sci. 2010, 3, 1706–1721. [Google Scholar] [CrossRef]
- Ganjehkaviri, A.; Mohd Jaafar, M.; Hosseini, S.; Musthafa, A. Performance Evaluation of Palm Oil-Based Biodiesel Combustion in an Oil Burner. Energies 2016, 9, 97. [Google Scholar] [CrossRef]
- Vyas, A.P.; Verma, J.L.; Subrahmanyam, N. A review on FAME production processes. Fuel 2010, 89, 1–9. [Google Scholar] [CrossRef]
- Leung, D.Y.; Wu, X.; Leung, M. A review on biodiesel production using catalyzed transesterification. Appl. Energy 2010, 87, 1083–1095. [Google Scholar] [CrossRef]
- Taufiq-Yap, Y.H.; Lee, H.V. Higher Grade Biodiesel Production by Using Solid Heterogeneous Catalysts. In Advances in Biofuels; Pogaku, R., Sarbatly, R.H., Eds.; Springer: New York, NY, USA, 2013; pp. 153–176. [Google Scholar]
- Meng, X.; Yang, J.; Xu, X.; Zhang, L.; Nie, Q.; Xian, M. Biodiesel production from oleaginous microorganisms. Renew. Energy 2009, 34, 1–5. [Google Scholar] [CrossRef]
- Bryan, R.M. Biodiesel production, properties and feedstocks. Vitro Cell. Dev. Biol. Plant 2009, 45, 229–266. [Google Scholar]
- Jain, S.; Sharma, M. Prospects of biodiesel from Jatropha in India: A review. Renew. Sustain. Energy Rev. 2010, 14, 763–771. [Google Scholar] [CrossRef]
- Chavan, S.B.; Kumbhar, R.R.; Madhu, D.; Singh, B.; Sharma, Y.C. Synthesis of biodiesel from Jatropha curcas oil using waste eggshell and study of its fuel properties. RSC Adv. 2015, 5, 63596–63604. [Google Scholar] [CrossRef]
- Deng, X.; Han, J.; Yin, F. Net Energy, CO2 Emission and Land-Based Cost-Benefit Analyses of Jatropha Biodiesel: A Case Study of the Panzhihua Region of Sichuan Province in China. Energies 2012, 5, 2150–2164. [Google Scholar] [CrossRef]
- Ruhul, A.M.; Kalam, M.A.; Masjuki, H.H.; Fattah, I.M.R.; Reham, S.S.; Rashed, M.M. State of the art of biodiesel production processes: A review of the heterogeneous catalyst. RSC Adv. 2015, 5, 101023–101044. [Google Scholar] [CrossRef]
- Taufiq-Yap, Y.H.; Lee, H.V.; Hussein, M.Z.; Yunus, R. Calcium-based mixed oxide catalysts for methanolysis of Jatropha curcas oil to biodiesel. Biomass Bioenergy 2011, 35, 827–834. [Google Scholar] [CrossRef]
- Taufiq-Yap, Y.H.; Lee, H.V.; Yunus, R.; Juan, J.C. Transesterification of non-edible Jatropha curcas oil to biodiesel using binary Ca–Mg mixed oxide catalyst: Effect of stoichiometric composition. Chem. Eng. J. 2011, 178, 342–347. [Google Scholar] [CrossRef]
- Lee, H.V.; Taufiq-Yap, Y.H.; Hussein, M.Z.; Yunus, R. Transesterification of jatropha oil with methanol over Mg-Zn mixed metal oxide catalysts. Energy 2013, 49, 12–18. [Google Scholar] [CrossRef]
- Wachs, I.E. Recent conceptual advances in the catalysis science of mixed metal oxide catalytic materials. Catal. Today 2005, 100, 79–94. [Google Scholar] [CrossRef]
- Gawande, M.B.; Pandey, R.K.; Jayaram, R.V. Role of mixed metal oxides in catalysis science—Versatile applications in organic synthesis. Catal. Sci. Technol. 2012, 2, 1113–1125. [Google Scholar] [CrossRef]
- Climent, M.J.; Corma, A.; de Frutos, P.; Iborra, S.; Noy, M.; Velty, A.; Concepción, P. Chemicals from biomass: Synthesis of glycerol carbonate by transesterification and carbonylation with urea with hydrotalcite catalysts: The role of acid–base pairs. J. Catal. 2010, 269, 140–149. [Google Scholar] [CrossRef]
- Xie, W.; Zhao, L. Heterogeneous CaO–MoO3–SBA-15 catalysts for biodiesel production from soybean oil. Energy Convers. Manag. 2014, 79, 34–42. [Google Scholar] [CrossRef]
- Santiago-Torres, N.; Romero-Ibarra, I.C.; Pfeiffer, H. Sodium zirconate (Na2ZrO3) as a catalyst in a soybean oil transesterification reaction for biodiesel production. Fuel Process. Technol. 2014, 120, 34–39. [Google Scholar] [CrossRef]
- Sawangkeaw, R.; Tejvirat, P.; Ngamcharassrivichai, C.; Ngamprasertsith, S. Supercritical Transesterification of Palm Oil and Hydrated Ethanol in a Fixed Bed Reactor with a CaO/Al2O3 Catalyst. Energies 2012, 5, 1062. [Google Scholar] [CrossRef]
- Dai, Y.-M.; Wu, J.-S.; Chen, C.-C.; Chen, K.-T. Evaluating the optimum operating parameters on transesterification reaction for biodiesel production over a LiAlO2 catalyst. Chem. Eng. J. 2015, 280, 370–376. [Google Scholar] [CrossRef]
- Dai, Y.-M.; Chen, K.-T.; Chen, C.-C. Study of the microwave lipid extraction from microalgae for biodiesel production. Chem. Eng. J. 2014, 250, 267–273. [Google Scholar] [CrossRef]
- Wang, J.-X.; Chen, K.-T.; Wu, J.-S.; Wang, P.-H.; Huang, S.-T.; Chen, C.-C. Production of biodiesel through transesterification of soybean oil using lithium orthosilicate solid catalyst. Fuel Process. Technol. 2012, 104, 167–173. [Google Scholar] [CrossRef]
- Endalew, A.K.; Kiros, Y.; Zanzi, R. Heterogeneous catalysis for biodiesel production from Jatropha curcas oil (JCO). Energy 2011, 36, 2693–2700. [Google Scholar] [CrossRef]
- Liu, S.; Ma, J.; Guan, L.; Li, J.; Wei, W.; Sun, Y. Mesoporous CaO–ZrO2 nano-oxides: A novel solid base with high activity and stability. Microporous Mesoporous Mater. 2009, 117, 466–471. [Google Scholar] [CrossRef]
- Yan, S.; Kim, M.; Mohan, S.; Salley, S.O.; Ng, K.Y.S. Effects of preparative parameters on the structure and performance of Ca-La metal oxide catalysts for oil transesterification. Appl. Catal. A Gen. 2010, 373, 104–111. [Google Scholar] [CrossRef]
- Boynton, R.S. Chemistry and Technology of Lime and Limestone; Interscience: New York, NY, USA, 1966. [Google Scholar]
- Fraile, J.M.; García, N.; Mayoral, J.A.; Pires, E.; Roldán, L. The basicity of mixed oxides and the influence of alkaline metals: The case of transesterification reactions. Appl. Catal. A Gen. 2010, 387, 67–74. [Google Scholar] [CrossRef]
- Di Serio, M.; Ledda, M.; Cozzolino, M.; Minutillo, G.; Tesser, R.; Santacesaria, E. Transesterification of soybean oil to biodiesel by using heterogeneous basic catalysts. Ind. Eng. Chem. Res. 2006, 45, 3009–3014. [Google Scholar] [CrossRef]
- Zhang, P.; Han, Q.; Fan, M.; Jiang, P. Magnetic solid base catalyst CaO/CoFe2O4 for biodiesel production: Influence of basicity and wettability of the catalyst in catalytic performance. Appl. Surf. Sci. 2014, 317, 1125–1130. [Google Scholar] [CrossRef]
- Hattori, H. Solid Base Catalysts: Generation, Characterization, and Catalytic Behavior of Basic Sites. J. Jpn. Pet. Inst. 2004, 47, 67–81. [Google Scholar] [CrossRef]
- Corma, A.; Hamid, S.B.A.; Iborra, S.; Velty, A. Lewis and Brönsted basic active sites on solid catalysts and their role in the synthesis of monoglycerides. J. Catal. 2005, 234, 340–347. [Google Scholar] [CrossRef]
- Refaat, A. Biodiesel production using solid metal oxide catalysts. Int. J. Environ. Sci. Technol. 2011, 8, 203–221. [Google Scholar] [CrossRef]
- Om Tapanes, N.C.; Gomes Aranda, D.A.; de Mesquita Carneiro, J.W.; Ceva Antunes, O.A. Transesterification of Jatropha curcas oil glycerides: Theoretical and experimental studies of biodiesel reaction. Fuel 2008, 87, 2286–2295. [Google Scholar] [CrossRef]
- Boey, P.-L.; Maniam, G.P.; Hamid, S.A. Performance of calcium oxide as a heterogeneous catalyst in biodiesel production: A review. Chem. Eng. J. 2011, 168, 15–22. [Google Scholar] [CrossRef]
- Atadashi, I.M.; Aroua, M.K.; Abdul Aziz, A.R.; Sulaiman, N.M.N. Production of biodiesel using high free fatty acid feedstocks. Renew. Sustain. Energy Rev. 2012, 16, 3275–3285. [Google Scholar] [CrossRef]
- Kesica, Z.; Lukic, I.; Zdujic, M.; Liu, H.; Skala, D. Mechanochemically Synthesized CaO·ZnO Catalyst For Biodiesel Production. Procedia Eng. 2012, 42, 1169–1178. [Google Scholar] [CrossRef]
- Ngamcharussrivichai, C.; Totarat, P.; Bunyakiat, K. Ca and Zn mixed oxide as a heterogeneous base catalyst for transesterification of palm kernel oil. Appl. Catal. A Gen. 2008, 341, 77–85. [Google Scholar] [CrossRef]
- Istadi, I.; Prasetyo, S.A.; Nugroho, T.S. Characterization of K2O/CaO-ZnO Catalyst for Transesterification of Soybean Oil to Biodiesel. Procedia Environ. Sci. 2015, 23, 394–399. [Google Scholar] [CrossRef]
- Gryglewicz, S. Rapeseed oil methyl esters preparation using heterogeneous catalysts. Bioresour. Technol. 1999, 70, 249–253. [Google Scholar] [CrossRef]
- Gryglewicz, S. Alkaline-earth metal compounds as alcoholysis catalysts for ester oils synthesis. Appl. Catal. A Gen. 2000, 192, 23–28. [Google Scholar] [CrossRef]
- Sharma, Y.C.; Singh, B.; Korstad, J. Latest developments on application of heterogenous basic catalysts for an efficient and eco friendly synthesis of biodiesel: A review. Fuel 2011, 90, 1309–1324. [Google Scholar] [CrossRef]
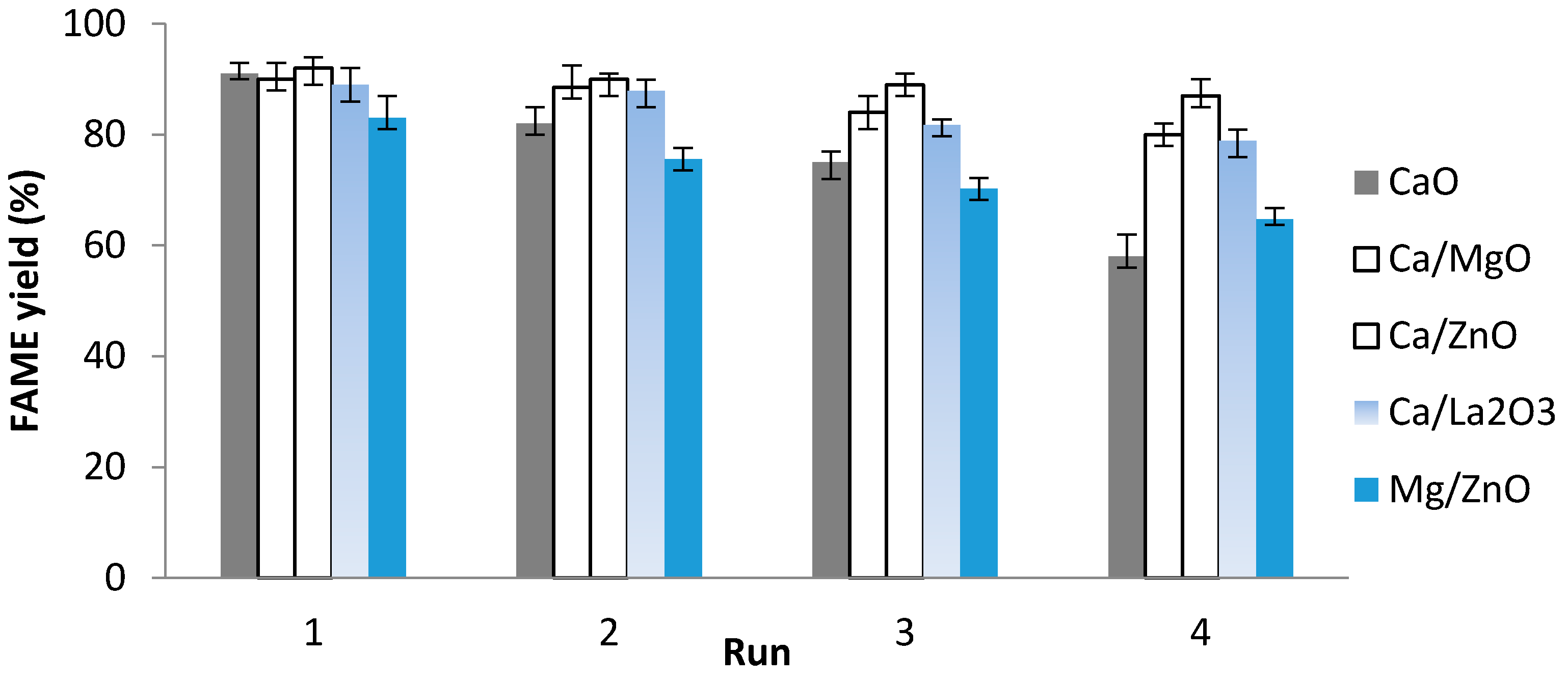

| Catalysts | Amount of Basic Sites | |
|---|---|---|
| Tmax (300–500 °C) Medium Basic Strength | Tmax (>500 °C) Strong Basic Strength | |
| ZnO | - | - |
| MgO | 50.3 | - |
| CaO | - | 290.4 |
| La2O3 | - | 179.9 |
| MgO-ZnO | 73.2 | 257.9 |
| CaO-MgO | - | 1210.5 |
| CaO-ZnO | - | 1383.0 |
| CaO-La2O3 | - | 737.8 |
| Catalyst | Catalyst Synthesis Method | Feedstock and Transesterification Condition | Biodiesel Yield (%) | Reference |
|---|---|---|---|---|
| CaO·ZnO | Ball milling of CaO and ZnO powder mixture (CaO:ZnO = 1:2) | Methanolysis of sunflower oil, at 60 °C and 1 bar, using molar ratio of sunflower oil to methanol of 1:10 and with 2 wt % of catalyst based on oil weight, 3 h | 99% | [37] |
| CaO·ZnO | Conventional co-precipitation (Ca:Zn = 0.25:1) | Transesterification of palm kernel oil under conditions at 60 °C, catalyst amount = 10 wt %, methanol/oil molar ratio = 30, reaction time = 1 h | >94% | [38] |
| K2O/CaO-ZnO | CaO-ZnO prepared by co-precipitation was promoted by K2O via impregnation method (Ca:Zn = 1:3) | Tranesterification of soybean oil under reaction temperature of 60 °C, catalyst loading of 2 wt %, methanol to oil molar ratio of 15:1, and reaction time of 4 h | 81% | [39] |
| CaO-ZnO | Co-precipitation method (Ca:Zn ratio = 1:1) | Transesterification of high-acid crude jatrophas oil (12 wt % of FFA) under reaction conditions: catalyst amount,3 wt %; methanol/oil ratio = 25:1; 120 °C; 3 h | 94% | Present study |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.V.; Juan, J.C.; Yun Hin, T.-Y.; Ong, H.C. Environment-Friendly Heterogeneous Alkaline-Based Mixed Metal Oxide Catalysts for Biodiesel Production. Energies 2016, 9, 611. https://doi.org/10.3390/en9080611
Lee HV, Juan JC, Yun Hin T-Y, Ong HC. Environment-Friendly Heterogeneous Alkaline-Based Mixed Metal Oxide Catalysts for Biodiesel Production. Energies. 2016; 9(8):611. https://doi.org/10.3390/en9080611
Chicago/Turabian StyleLee, Hwei Voon, Joon Ching Juan, Taufiq-Yap Yun Hin, and Hwai Chyuan Ong. 2016. "Environment-Friendly Heterogeneous Alkaline-Based Mixed Metal Oxide Catalysts for Biodiesel Production" Energies 9, no. 8: 611. https://doi.org/10.3390/en9080611
APA StyleLee, H. V., Juan, J. C., Yun Hin, T.-Y., & Ong, H. C. (2016). Environment-Friendly Heterogeneous Alkaline-Based Mixed Metal Oxide Catalysts for Biodiesel Production. Energies, 9(8), 611. https://doi.org/10.3390/en9080611