Catalytic Intermediate Pyrolysis of Napier Grass in a Fixed Bed Reactor with ZSM-5, HZSM-5 and Zinc-Exchanged Zeolite-A as the Catalyst
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Characterization
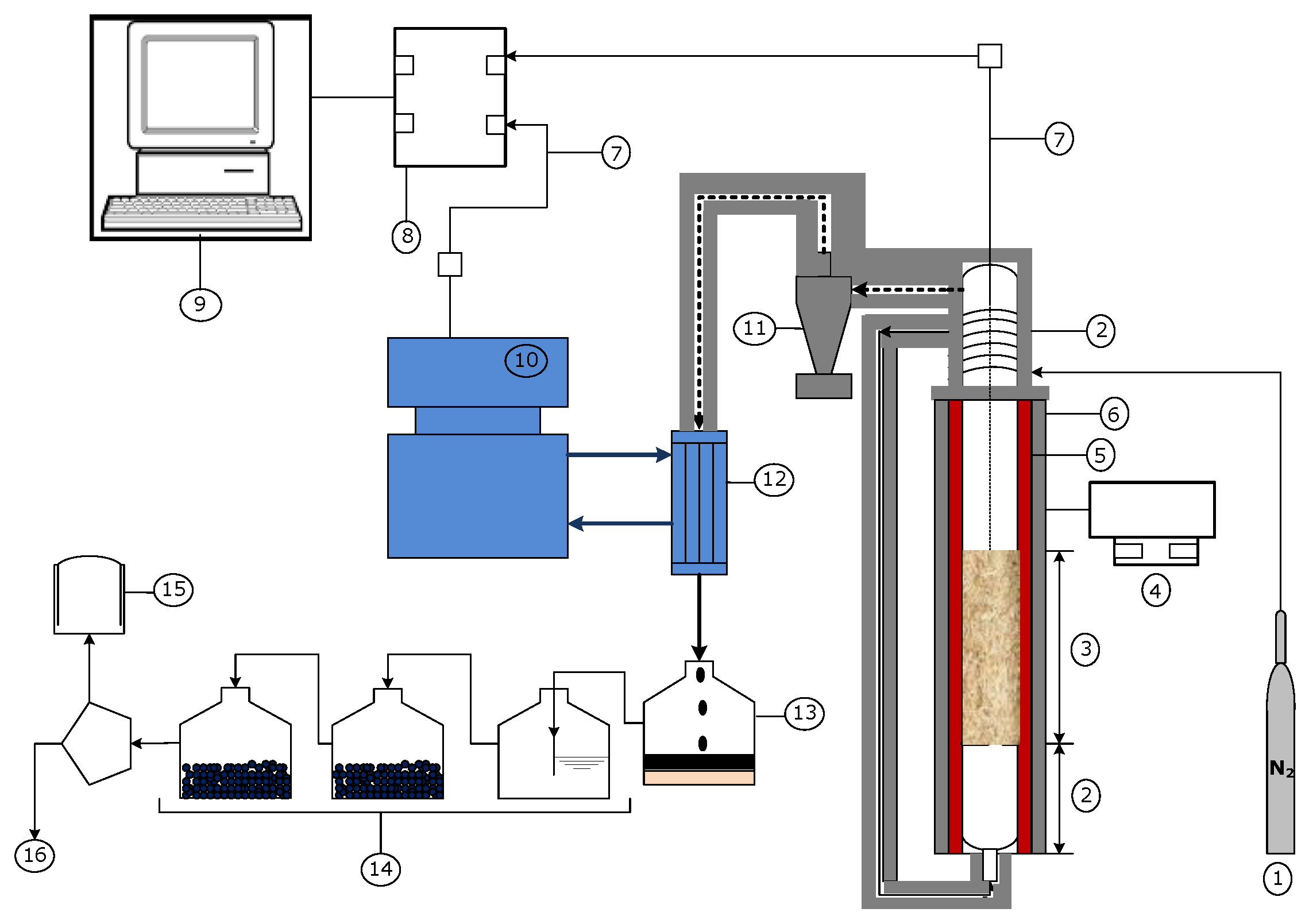
2.2. Catalytic Pyrolysis and Pyrolysis Oil Characterization
3. Results and Discussion
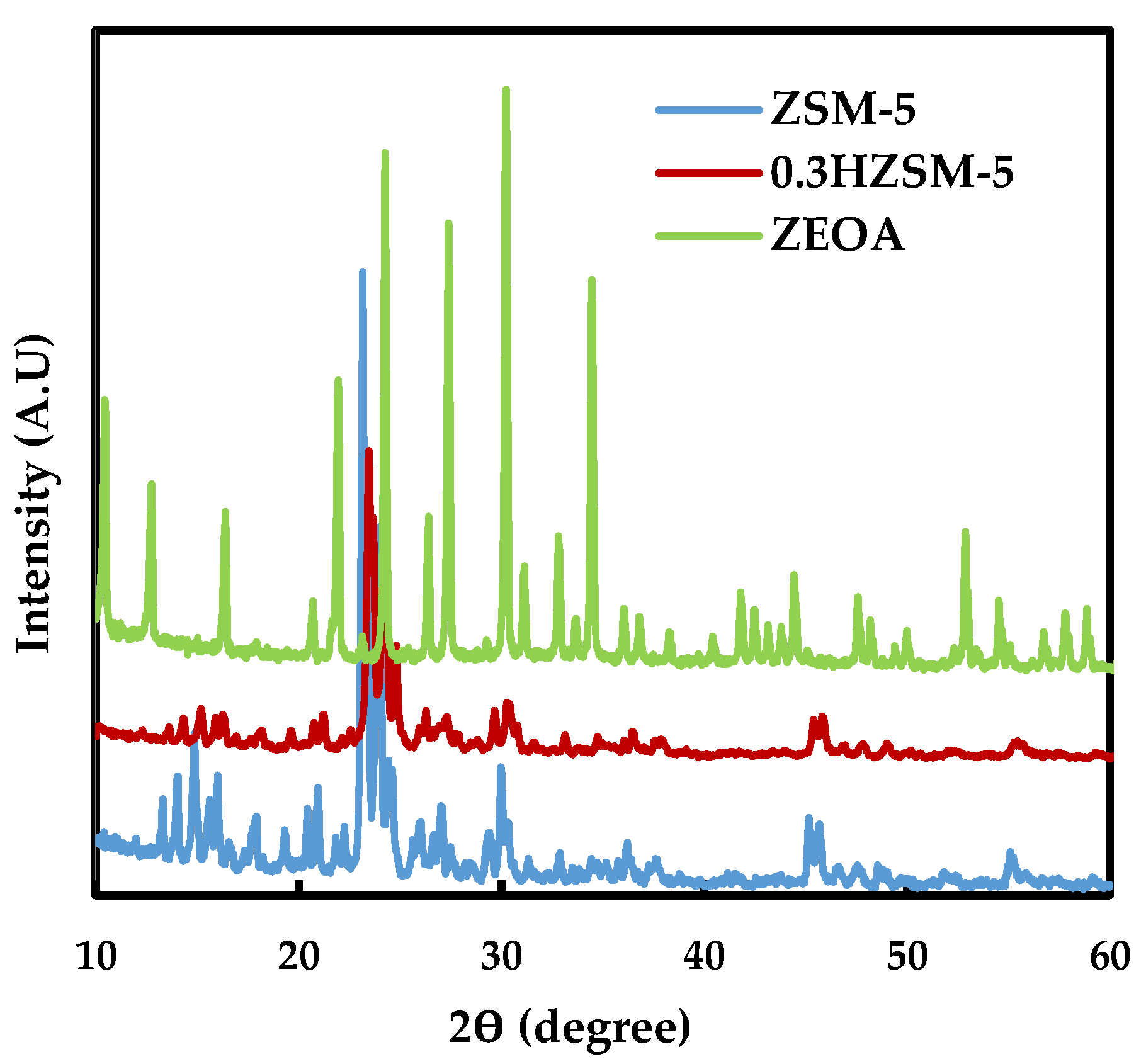
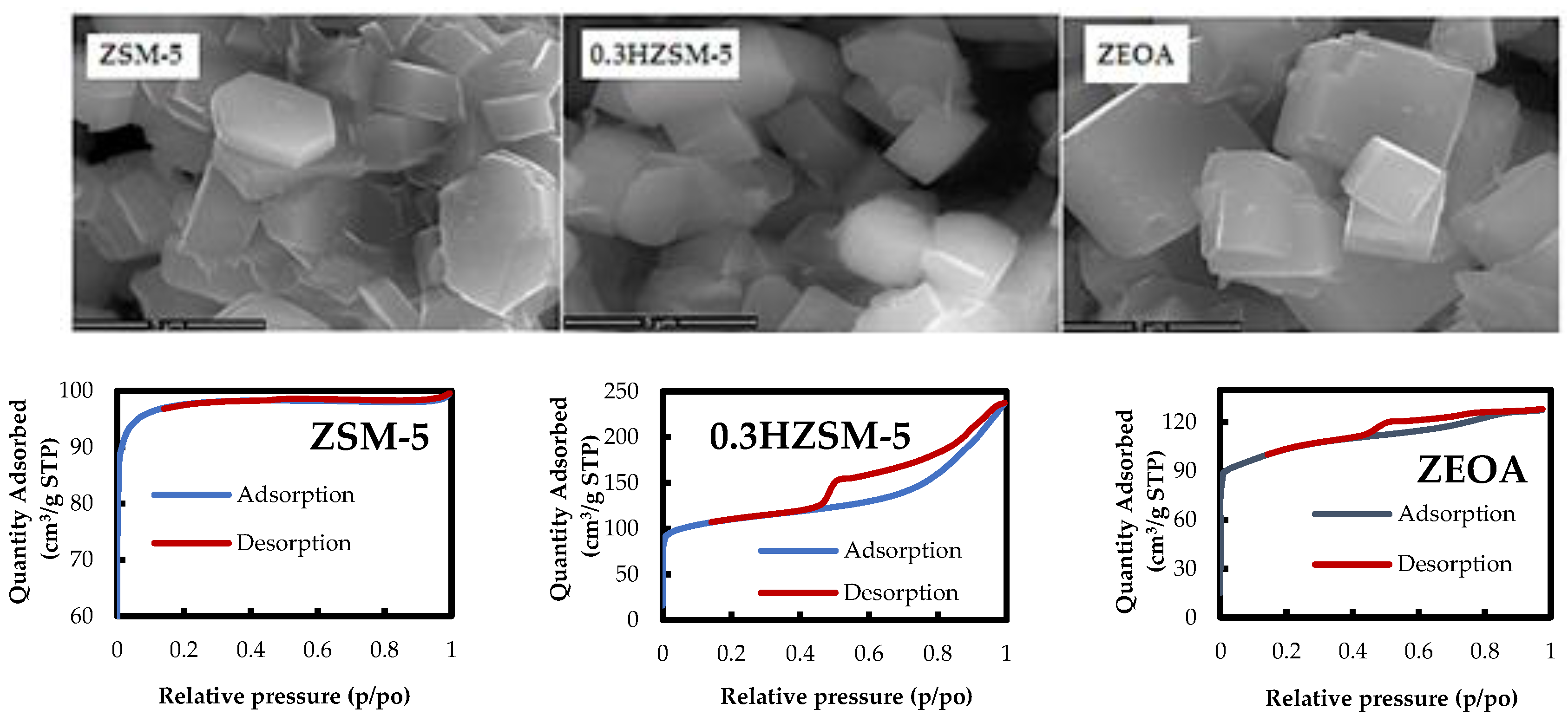
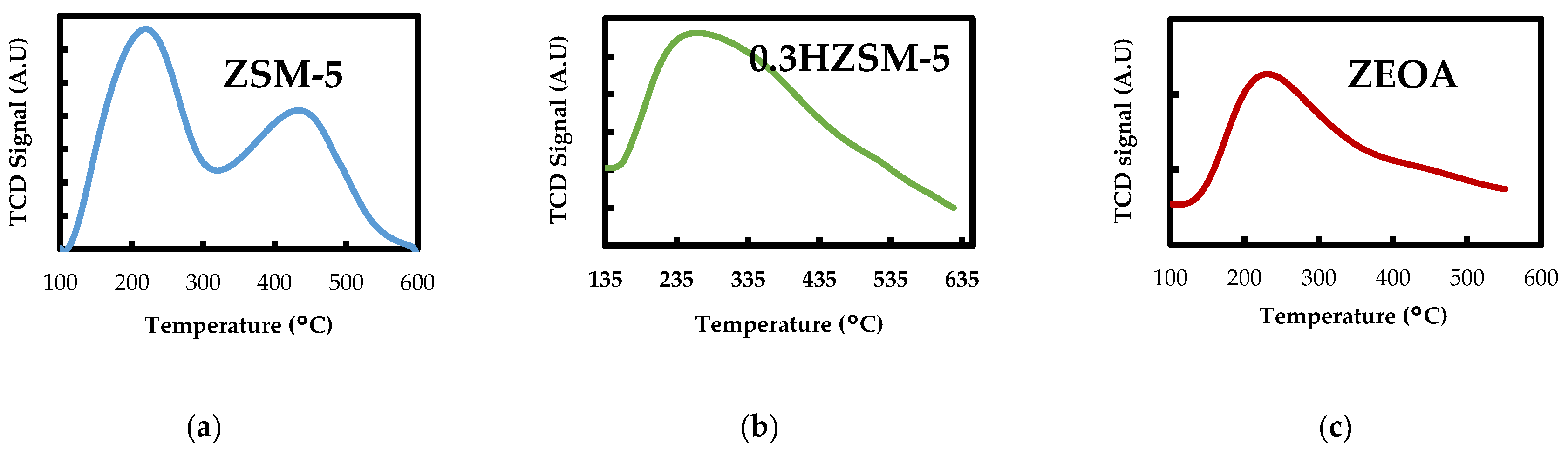
3.1. Catalyst Characteristics
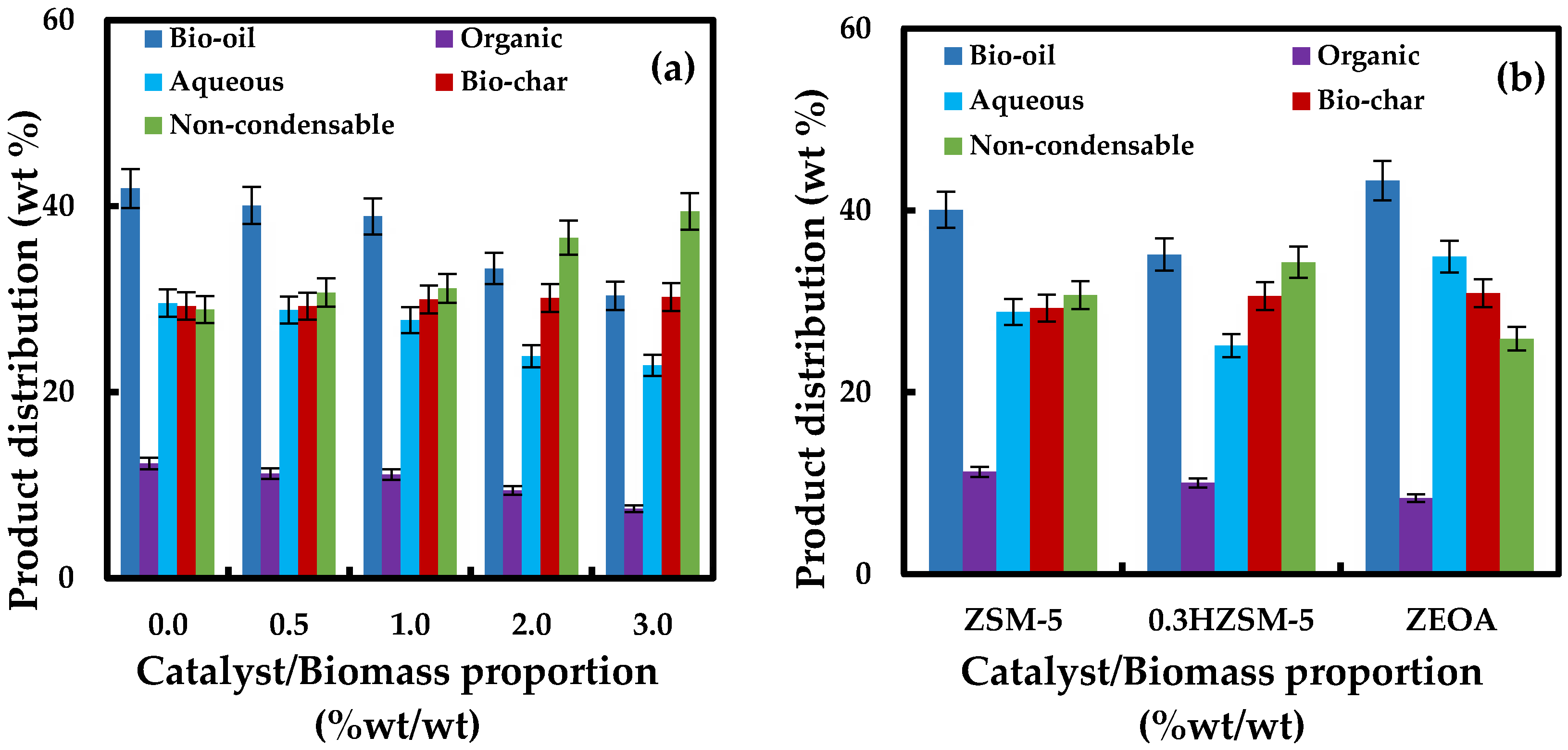
3.2. Pyrolysis Product Distribution
3.3. Physicochemical Properties of Organic Phase Product
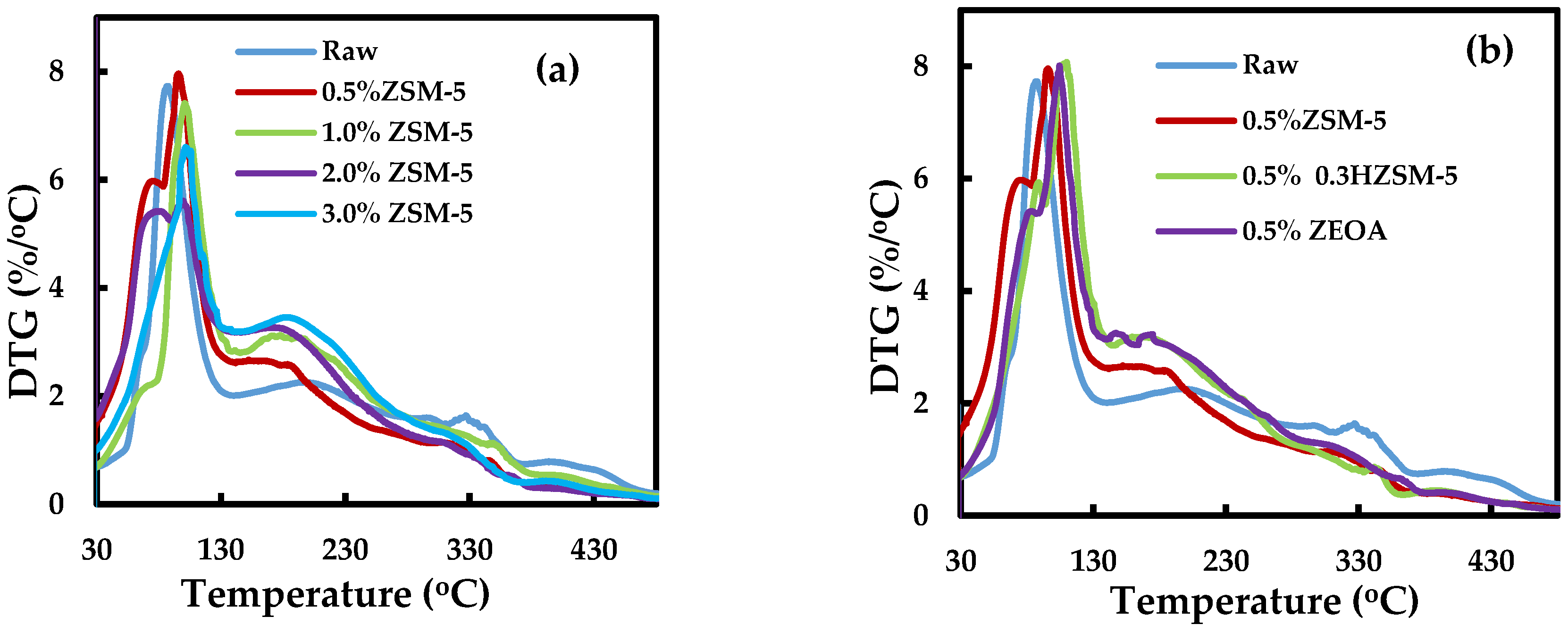
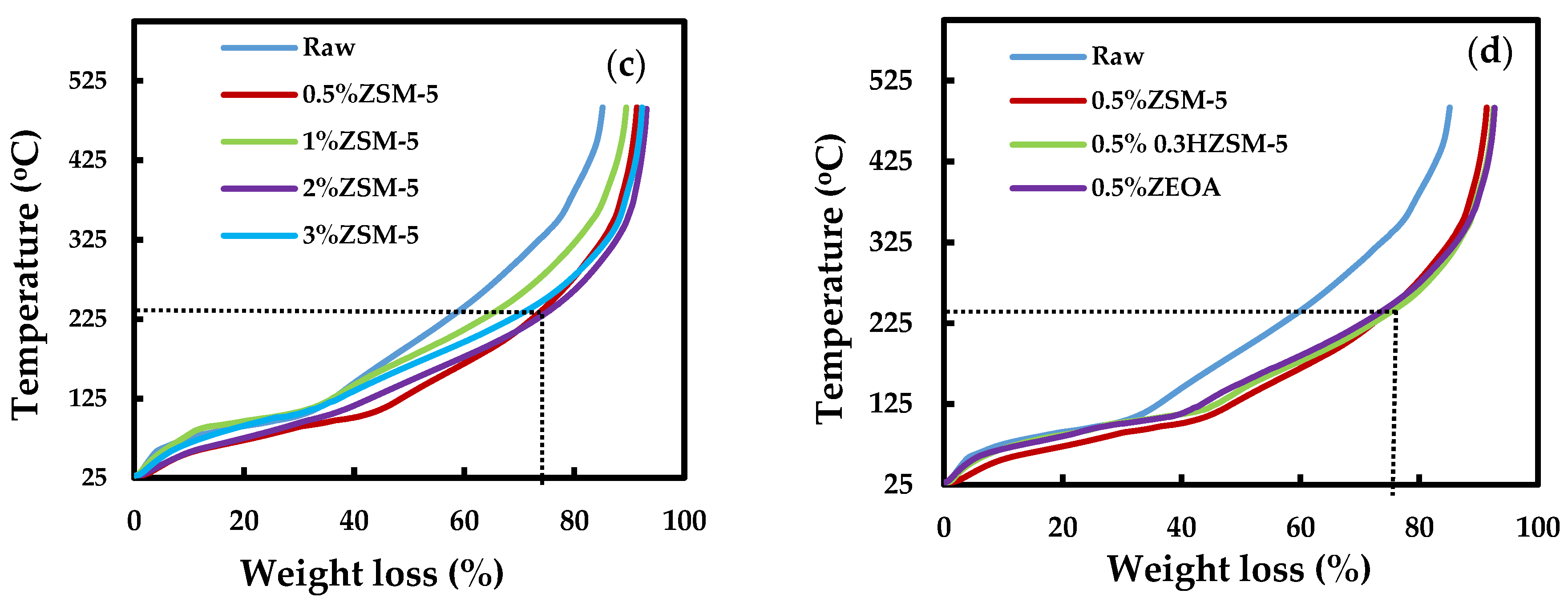
3.4. Thermogravimetric Analysis of the Organic Phase Bio-Oil
3.5. Functional Group Analysis of Bio-Oils
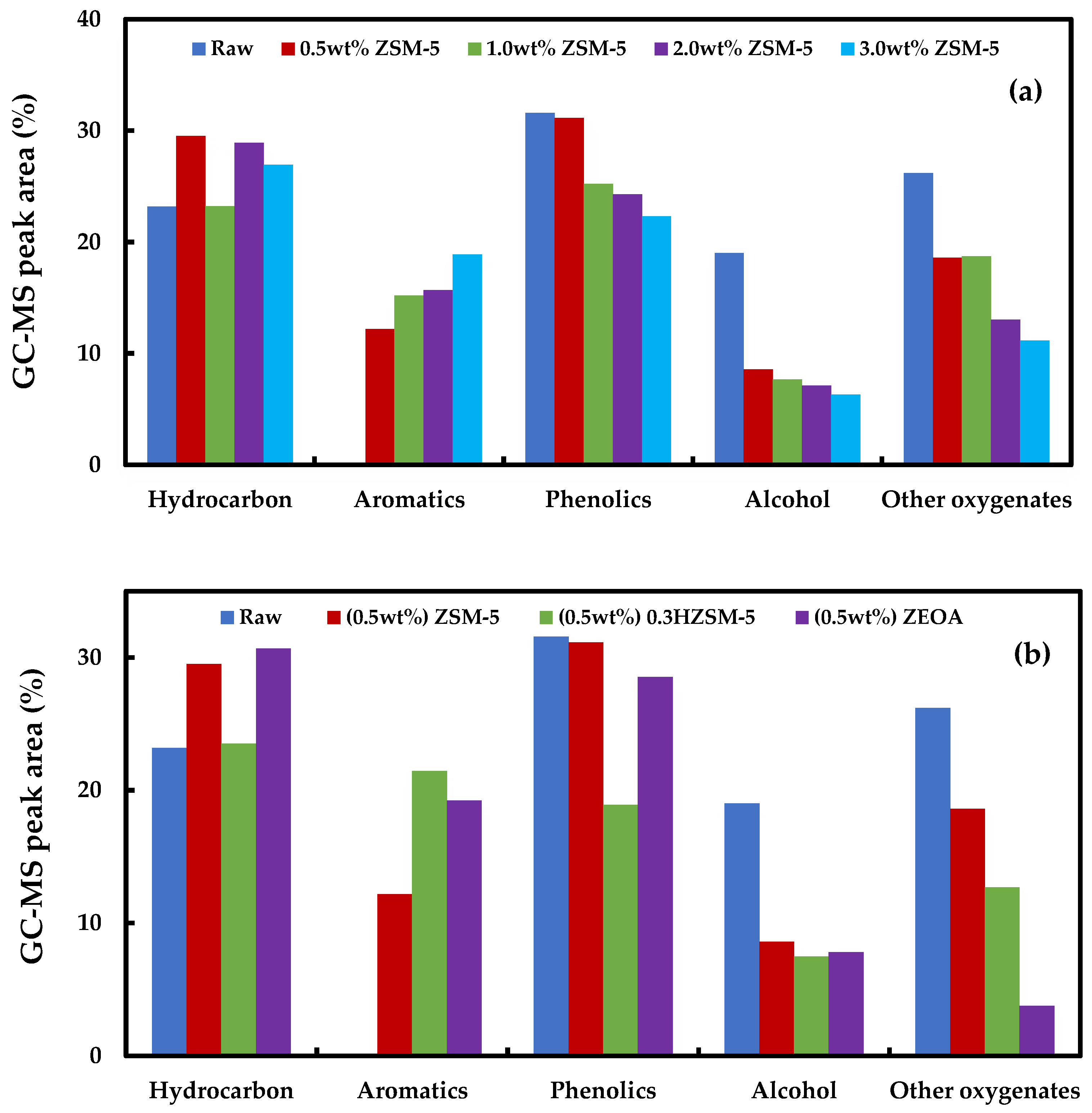
3.6. GC-MS Analysis of the Organic Phase Bio-Oil
3.7. GC Analysis of the Non-Condensable Gas
4. Conclusions
- ZSM-5 catalyst loading between 0.5 and 1.0 wt % had no significant impact on the oil yield compared to higher catalyst loadings at 2.0 and 3.0 wt %. The yield of non-condensable gas increased with catalyst loading. Impact of ZSM-5 on the yield of bio-char was minimal.
- Organic compounds in the bio-oil produced with ZSM-5 were made up of mainly hydrocarbons, aromatics and phenols. Catalyst loadings between 0.5 and 1.0 wt % promoted the yield of polyaromatic hydrocarbon (naphthalene) while benzene dominated the aromatics when 2.0 and 3.0 wt % catalyst loading were employed.
- Desilication of ZSM-5 with NaOH produced a mesoporous 0.3HZSM-5. Bio-oil yield decreased with HZSM-5 and increased with ZEOA compared to ZSM-5 at 0.5 wt % loading. The organic phase composition of the bio-oil from 0.3HZSM-5 and ZEOA were lower than that from of ZSM-5. Higher hydrocarbon yield was recorded, particularly with ZEOA, and the aromatics were mainly benzenes. Reduction in the phenolic content and other oxygenated compounds were also recorded. This observation was attributed to the improved pore structure and the acid sites of the catalysts. Higher composition of CO and CO2 was observed in the non-condensable gas from the catalytic pyrolysis compared to the non-catalytic pyrolysis and was attributed to decarbonylation and decarboxylation reactions.
- This study has demonstrated that bio-oil with high fuel quality and other value added chemicals can be produced from pyrolysis of Napier grass over acidic zeolite-based catalysts.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yakub, M.I.; Mohamed, S.; Danladi, S.U. Technical and Economic Considerations of Post-combustion Carbon Capture in a Coal Fired Power Plant. Int. J. Adv. Eng. Technol. 2014, 7, 1549–1581. [Google Scholar]
- Mohammed, I.Y. Optimization and Sensitivity Analysis of Post-combustion Carbon Capture Using DEA Solvent in a Coal Fired Power Plant. Int. J. Adv. Eng. Technol. 2015, 7, 1681–1690. [Google Scholar]
- Mohammed, I.Y.; Samah, M.; Mohamed, A.; Sabina, G. Comparison of SelexolTM and Rectisol® Technologies in an Integrated Gasification Combined Cycle (IGCC) Plant for Clean Energy Production. Int. J. Eng. Res. 2014, 3, 742–744. [Google Scholar] [CrossRef]
- Yakub, M.I.; Abdalla, A.Y.; Feroz, K.K.; Suzana, Y.; Ibraheem, A.; Chin, S.A. Pyrolysis of Oil Palm Residues in a Fixed Bed Tubular Reactor. J. Power Energy Eng. 2015, 3, 185–193. [Google Scholar] [CrossRef]
- Gebreslassie, B.H.; Slivinsky, M.; Wang, B.; You, F. Life cycle optimization for sustainable design and operations of hydrocarbon biorefinery via fast pyrolysis, hydrotreating and hydrocracking. Comput. Chem. Eng. 2013, 50, 71–91. [Google Scholar] [CrossRef]
- Liew, W.H.; Hassim, M.H.; Ng, D.S.K. Review of evolution, technology and sustainability assessments of biofuel production. J. Clean. Prod. 2014, 71, 11–29. [Google Scholar] [CrossRef]
- Park, S.R.; Pandey, A.K.; Tyagi, V.V.; Tyagi, S.K. Energy and exergy analysis of typical renewable energy systems. Renew. Sustain. Energy Rev. 2014, 30, 105–123. [Google Scholar] [CrossRef]
- Ming, Z.; Ximei, L.; Yulong, L.; Lilin, P. Review of renewable energy investment and financing in China: Status, mode, issues and countermeasures. Renew. Sustain. Energy Rev. 2014, 31, 23–37. [Google Scholar] [CrossRef]
- Nigam, P.S.; Singh, A. Production of liquid biofuels from renewable resources. Prog. Energy Combust. Sci. 2011, 37, 52–68. [Google Scholar] [CrossRef]
- Srirangan, K.; Akawi, L.; Moo-Young, M.; Chou, C.P. Towards sustainable production of clean energy carriers from biomass resources. Appl. Energy 2012, 100, 172–186. [Google Scholar] [CrossRef]
- Samson, R.; Mani, S.; Boddey, R.; Sokhansanj, S.; Quesada, D.; Urquiaga, S.; Reis, V.; Ho Lem, C. The Potential of C4 Perennial Grasses for Developing a Global BIOHEAT Industry. Crit. Rev. Plant Sci. 2005, 24, 461–495. [Google Scholar] [CrossRef]
- Mohammed, I.Y.; Abakr, Y.A.; Kazi, F.K.; Yusup, S.; Alshareef, I.; Chin, S.A. Comprehensive Characterization of Napier Grass as a Feedstock for Thermochemical Conversion. Energies 2015, 8, 3403–3417. [Google Scholar] [CrossRef]
- Mohammed, I.Y.; Abakr, Y.A.; Kazi, F.K.; Yusup, S.; Alshareef, I.; Chin, S.A. Pyrolysis of Napier Grass in a Fixed Bed Reactor: Effect of Operating Conditions on Product Yields and Characteristics. BioResources 2015, 10, 6457–6478. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of Fast Pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Kebelmann, K.; Hornung, A.; Karsten, U.; Griffiths, G. Intermediate pyrolysis and product identification by TGA and Py-GC/MS of green microalgae and their extracted protein and lipid components. Biomass Bioenergy 2013, 49, 38–48. [Google Scholar] [CrossRef]
- Mahmood, A.S.N.; Brammer, J.G.; Hornung, A.; Steele, A.; Poulston, S. The inter-mediate pyrolysis and catalytic steam reforming of Brewers spent grain. J. Anal. Appl. Pyrolysis 2013, 103, 328–342. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis: A review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Iliopoulou, E.F.; Stefanidis, S.D.; Kalogiannis, K.G.; Delimitis, A.; Lappas1, A.A.; Triantafyllidis, K.S. Catalytic upgrading of biomass pyrolysis vapors using transition metal-modified ZSM-5 zeolite. Appl. Catal. B Environ. 2010, 127, 281–290. [Google Scholar] [CrossRef]
- Carlson, T.R.; Cheng, Y.; Jae, J.; Huber, G.W. Production of green aromatics and olefins by catalytic fast pyrolysis of wood sawdust. Energy Environ. Sci. 2011, 4, 145–161. [Google Scholar] [CrossRef]
- Compton, D.L.; Jackson, M.A.; Mihalcik, D.J.; Mullen, C.A.; Boateng, A.A. Catalytic pyrolysis of oak via pyroprobe and bench scale, packed bed pyrolysis reactors. J. Anal. Appl. Pyrolysis 2011, 90, 174–181. [Google Scholar] [CrossRef]
- Du, S.; Sun, Y.; Gamliel, D.P.; Valla, J.A.; Bollas, G.M. Catalytic pyrolysis of miscanthus × giganteus in a spouted bed reactor. Bioresour. Technol. 2014, 169, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Lee, K.-H.; Zhang, J.; Brown, R.C.; Shanks, B.H. Manipulation of chemical species in bio-oil using in situ catalytic fast pyrolysis in both a bench-scale fluidized bed pyrolyzer and micropyrolyzer. Biomass Bioenergy 2015, 81, 256–264. [Google Scholar] [CrossRef]
- Park, Y.-K.; Yoo, M.L.; Jin, S.H.; Park, S.H. Catalytic fast pyrolysis of waste pepper stems over HZSM-5. Renew. Energy 2015, 79, 20–27. [Google Scholar] [CrossRef]
- Jae, J.; Tompsett, G.A.; Foster, A.J.; Hammond, K.D.; Auerbach, S.M.; Lobo, R.F.; Huber, G.W. Investigation into the shape selectivity of zeolite catalysts for biomass conversion. J. Catal. 2011, 279, 257–268. [Google Scholar] [CrossRef]
- Zhang, B.; Zhong, Z.-P.; Wang, X.-B.; Ding, K.; Song, Z.-W. Catalytic upgrading of fast pyrolysis biomass vapors over fresh, spent and regenerated ZSM-5 zeolites. Fuel Process. Technol. 2015, 138, 430–434. [Google Scholar] [CrossRef]
- Carlson, T.R.; Vispute, T.P.; Huber, G.W. Green gasoline by catalytic fast pyrolysis of solid biomass derived compounds. ChemSusChem 2008, 1, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Gamliel, D.P.; Du, S.; Bollas, G.M.; Valla, J.A. Investigation of in situ and ex situ catalytic pyrolysis of miscanthus-giganteus using a PyGC–MS microsystem and comparison with a bench-scale spouted-bed reactor. Bioresour. Technol. 2015, 191, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, G.; Ronsse, F.; Venderbosch, R.; Van Duren, R.; Kersten, S.R.A.; Prins, W. Effect of biomass ash in catalytic fast pyrolysis of pine wood. Appl. Catal. B Environ. 2015, 168–169, 203–211. [Google Scholar] [CrossRef]
- Liu, G.; Wright, M.M.; Zhao, Q.; Brown, R.C. Catalytic fast pyrolysis of duckweed: Effects of pyrolysis parameters and optimization of aromatic production. J. Anal. Appl. Pyrolysis 2015, 112, 29–36. [Google Scholar] [CrossRef]
- Kim, B.-S.; Kim, Y.-M.; Jae, J.; Watanabe, C.; Kim, S.; Jung, S.-C.; Kim, S.C.; Park, Y.-K. Pyrolysis and catalytic upgrading of Citrus unshiu peel. Bioresour. Technol. 2015, 194, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Ojha, D.K.; Vinu, R. Resource recovery via catalytic fast pyrolysis of polystyrene using zeolites. J. Anal. Appl. Pyrolysis 2015, 113, 349–359. [Google Scholar] [CrossRef]
- Standard Test Method for Water Using Volumetric Karl Fischer Titration; ASTM International: West Conshohocken, PA, USA, 2001.
- Mohammed, I.Y.; Kazi, F.K.; Abakr, Y.A.; Yusuf, S.; Razzaque, M.A. Novel Method for the Determination of Water Content and Higher Heating Value of Pyrolysis Oil. BioResources 2015, 10, 2681–2690. [Google Scholar] [CrossRef]
- Standard Test Method for Heat of Combustion of Liquid Hydrocarbon Fuels by Bomb Calorimeter; ASTM International: West Conshohocken, PA, USA, 2009.
- Alaba, P.A.; Sani, Y.M.; Mohammed, I.Y.; Abakr, Y.A.; Daud, W.M.A.W. Synthesis and application of hierarchical mesoporous HZSM-5 for biodiesel production from shea butter. J. Taiwan Inst. Chem. Eng. 2016, 59, 405–412. [Google Scholar] [CrossRef]
- Shirazi, L.; Jamshidi, E.; Ghasemi, M.R. Effect of Si/Al ratio of ZSM-5 zeolite on its morphology, acidity and crystal size. Cryst. Res. Technol. 2008, 43, 1300–1306. [Google Scholar] [CrossRef]
- Gong, F.; Yang, Z.; Hong, C.; Huang, W.; Ning, S.; Zhang, Z.; Xu, Y.; Li, Q. Selective conversion of bio-oil to light olefins: Controlling catalytic cracking for maximum olefins. Bioresour. Technol. 2011, 102, 9247–9254. [Google Scholar] [CrossRef] [PubMed]
- Suyitno; Purbaningrum, P.S.; Danardono, D.; Salem, A.E.; Mansur, F.A. Synthesis of zeolite Socony mobil from blue silica gel and rice husk ash as catalysts for hydrothermal liquefaction. J. Eng. Sci. Technol. 2015, 10, 982–993. [Google Scholar]
- Shaikh, I.R.; Shaikh, R.A.; Shaikh, A.A.; War, J.A.; Hangirgekar, S.P.; Shaikh, A.L.; Shaikh, P.R.; Shaikh, R.R. H-ZSM-5 Zeolite Synthesis by Sourcing Silica from the Wheat Husk Ash: Characterization and Application as a Versatile Heterogeneous Catalyst in Organic Transformations including Some Multicomponent Reactions. J. Catal. 2015, 2015, 805714. [Google Scholar] [CrossRef]
- Huang, Y.; Wei, L.; Julson, J.; Gao, Y.; Zhao, X. Converting pine sawdust to advanced biofuel over HZSM-5 using a two-stage catalytic pyrolysis reactor. J. Anal. Appl. Pyrolysis 2015, 111, 148–155. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, S.; Zhang, J.; Zhao, T.-S. Effect of Synthesis Conditions on the yields and properties of HZSM-5. Cryst. Res. Technol. 2015, 50, 522–527. [Google Scholar] [CrossRef]
- Ayele, L.; Perez-Pariente, J.; Chebude, Y.; Díaz, I. Synthesis of zeolite A from Ethiopian kaolin. Microporous Mesoporous Mater. 2015, 215, 29–36. [Google Scholar] [CrossRef]
- Bieseki, L.; Penha, F.G.; Perghera, S.B.C. Zeolite A Synthesis Employing a Brazilian Coal Ash as the Silicon and Aluminum Sourceand its Applications in Adsorption and Pigment Formulation. Mater. Res. 2013, 16, 38–43. [Google Scholar] [CrossRef]
- Dyballa, M.; Obenaus, U.; Lang, S.; Gehring, B.; Traa, Y.; Koller, H.; Hunger, M. Brønsted sites and structural stabilization effect of acidic low-silica zeolite A prepared by partial ammonium exchange. Microporous Mesoporous Mater. 2015, 212, 110–116. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Poerotto, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid system with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Zhou, G.; Wang, W.; Wang, C.; Komarneni, S.; Wang, Y. Catalytic fast pyrolysis of biomass with mesoporous ZSM-5 zeolites prepared by desilication with NaOH solutions. Appl. Catal. A Gen. 2014, 470, 115–122. [Google Scholar] [CrossRef]
- You, S.J.; Park, E.D. Effects of dealumination and desilication of H-ZSM-5 on xylose dehydration. Microporous Mesoporous Mater. 2014, 186, 121–129. [Google Scholar] [CrossRef]
- Wang, L.; Lei, H.; Ren, S.; Bu, Q.; Liang, J.; Wei, Y.; Liu, Y.; Lee, G.-S.J.; Chen, S.; Tang, J.; et al. Aromatics and phenols from catalytic pyrolysis of Douglas fir pellets in microwave with ZSM-5 as a catalyst. J. Anal. Appl. Pyrolysis 2012, 98, 194–200. [Google Scholar] [CrossRef]
- Jae, J.; Coolman, R.; Mountziaris, T.J.; Huber, G.W. Catalytic fast pyrolysis of lignocellulosic biomass in a process development unit with continual catalyst addition and removal. Chem. Eng. Sci. 2014, 108, 33–46. [Google Scholar] [CrossRef]
- Naqvi, S.R.; Uemura, Y.; Yusup, S.B. Catalytic pyrolysis of paddy husk in a drop type pyrolyzer for bio-oil production: The role of temperature and catalyst. J. Anal. Appl. Pyrolysis 2014, 106, 57–62. [Google Scholar] [CrossRef]
- Park, H.J.; Park, K.-H.; Jeon, J.-K.; Kim, J.; Ryoo, R.; Jeong, K.-E.; Park, S.H.; Park, Y.-K. Production of phenolics and aromatics by pyrolysis of Miscanthus. Fuel 2012, 97, 379–384. [Google Scholar] [CrossRef]
- Elordi, G.; Olazar, M.; Lopez, G.; Castaño, P.; Bilbao, J. Role of pore structure in the deactivation of zeolites (HZSM-5, Hb and HY) by coke in the pyrolysis of polyethylene in a conical spouted bed reactor. Appl. Catal. B Environ. 2011, 102, 224–231. [Google Scholar] [CrossRef]
- Lappas, A.A.; Samolada, M.C.; Iatridis, D.K.; Voutetakis, S.S.; Vasalos, I.A. Biomass pyrolysis in a circulating fluid bed reactor for the production of fuels and chemicals. Fuel 2002, 81, 2087–2095. [Google Scholar] [CrossRef]
- Veses, A.; Puértolas, B.; López, M.J.; Callén, M.S.; Solsona, B.; García, T. Promoting Deoxygenation of Bio-Oil by Metal-Loaded Hierarchical ZSM-5 Zeolites. ACS Sustain. Chem. Eng. 2016, 4, 1653–1660. [Google Scholar] [CrossRef]
- Nilsen, M.H.; Antonakou, E.; Bouzga, A.; Lappas, A.; Mathisen, K.; Stöcker, M. Investigation of the effect of metal sites in Me-Al-MCM-41 (Me = Fe, Cu or Zn) on the catalytic behavior during the pyrolysis of wooden based biomass. Microporous Mesoporous Mater. 2007, 105, 189–203. [Google Scholar] [CrossRef]
- Kelkar, S.; Saffron, C.M.; Andreassi, K.; Li, Z.; Murkute, A.; Miller, D.J.; Pinnavaia, T.J.; Kriegel, R.M. A survey of catalysts for aromatics from fast pyrolysis of biomass. Appl. Catal. B Environ. 2015, 174–175, 85–95. [Google Scholar] [CrossRef]
- Imam, T.; Capareda, S. Characterization of bio-oil, syn-gas and bio-char from switchgrass pyrolysis at various temperatures. J. Anal. Appl. Pyrolysis 2012, 93, 170–177. [Google Scholar] [CrossRef]
- Abu Bakar, M.S.; Titiloye, J.O. Catalytic pyrolysis of rice husk for bio-oil production. J. Anal. Appl. Pyrolysis 2013, 103, 362–368. [Google Scholar] [CrossRef]
- Fan, Y.; Cai, Y.; Li, X.; Yin, H.; Yu, N.; Zhang, R.; Zhao, W. Rape straw as a source of bio-oil via vacuum pyrolysis: Optimization of bio-oil yield using orthogonal design method and characterization of bio-oil. J. Anal. Appl. Pyrolysis 2014, 106, 63–70. [Google Scholar] [CrossRef]
- De Miguel Mercader, F.; Groeneveld, M.J.; Kersten, S.R.A.; Venderbosch, R.H.; Hogendoorn, J.A. Pyrolysis oil upgrading by high pressure thermal treatment. Fuel 2010, 89, 2829–2837. [Google Scholar] [CrossRef]
- Li, X.; Gunawan, R.; Wang, Y.; Chaiwat, W.; Hu, X.; Gholizadeh, M.; Mourant, D.; Bromly, J.; Li, C.-Z. Upgrading of bio-oil into advanced biofuels and chemicals. Part III. Changes in aromatic structure and coke forming propensity during the catalytic hydrotreatment of a fast pyrolysis bio-oil with Pd/C catalyst. Fuel 2014, 116, 642–649. [Google Scholar] [CrossRef]
- Garcia-Perez, M.; Chaala, A.; Pakdel, H.; Kretschmer, D.; Roy, C. Characterization of bio-oils in chemical families. Biomass Bioenergy 2007, 31, 222–242. [Google Scholar] [CrossRef]
- Nguyen, M.T.; Sengupta, D.; Raspoet, G.; Vanquickenborne, L.G. Theoretical Study of the Thermal Decomposition of Acetic Acid: Decarboxylation Versus Dehydration. J. Phys. Chem. 1995, 99, 11883–11888. [Google Scholar] [CrossRef]
- Guo, Y.; Song, W.; Lu, J.; Ma, Q.; Xu, D.; Wang, S. Hydrothermal liquefaction of Cyanophyta: Evaluation of potential bio-crude oil production and component analysis. Algal Res. 2015, 11, 242–247. [Google Scholar] [CrossRef]
- Bordoloi, N.; Narzari, R.; Chutia, R.S.; Bhaskar, T.; Kataki, R. Pyrolysis of Mesua ferrea and Pongamia glabra seed cover: Characterization of bio-oil and its sub-fractions. Bioresour. Technol. 2015, 178, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Lazdovica, K.; Liepina, L.; Kampars, V. Comparative wheat straw catalytic pyrolysis in the presence of zeolites, Pt/C, and Pd/C by using TGA-FTIR method. Fuel Process. Technol. 2015, 138, 645–653. [Google Scholar] [CrossRef]
- Yorgun, S.; Yildiz, D. Slow pyrolysis of paulownia wood: Effects of pyrolysis parameters on product yields and bio-oil characterization. J. Anal. Appl. Pyrolysis 2015, 114, 68–78. [Google Scholar] [CrossRef]
- Ben, H.; Ragauskas, A.J. Torrefaction of Loblolly Pine. Green Chem. 2012, 14, 72–76. [Google Scholar] [CrossRef]
- Gudka, B.; Jones, J.M.; Lea-Langton, A.R.; Williams, A.; Saddawi, A. A review of the mitigation of deposition and emission problems during biomass combustion through washing pre-treatment. J. Energy Inst. 2015, in press. [Google Scholar] [CrossRef]
- Zhang, H.; Xiao, R.; Jin, B.; Xiao, G.; Chen, R. Biomass catalytic pyrolysis to produce olefins and aromatics with a physically mixed catalyst. Bioresour. Technol. 2013, 140, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, P.S.; Shafaghat, H.; Daud, W.M.A.W. Production of green aromatics and olefins by catalytic cracking of oxygenate compounds derived from biomass pyrolysis: A review. Appl. Catal. A Gen. 2014, 469, 490–511. [Google Scholar] [CrossRef]
- Alaba, P.A.; Sani, Y.M.; Mohammed, I.Y.; Daud, W.M.A.W. Insight into catalyst deactivation mechanism and suppression techniques in thermocatalytic deoxygenation of bio-oil over zeolites. Rev. Chem. Eng. 2015, 32, 71–91. [Google Scholar] [CrossRef]
- Puertolas, B.; Veses, A.; Callen, M.S.; Mitchell, S.; Garcia, T.; Perez-Ramirez, J. Porosity-Acidity Interplay in Hierarchical ZSM-5 Zeolites for Pyrolysis Oil Valorization to Aromatics. Chem. Sus. Chem. 2015, 8, 3283–3293. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Du, S.; Valla, J.A.; Bollas, G.M. The eff ect of temperature, heating rate, and ZSM-5 catalyst on the product selectivity of the fast pyrolysis of spent coffee grounds. RSC Adv. 2015, 5, 29252–29261. [Google Scholar] [CrossRef]

| Property | ZSM-5 | 0.3HZSM-5 | ZEOA |
|---|---|---|---|
| Si/Al ratio | 20.7600 | 12.5100 | 1.0000 |
| SBET (m2/g) | 385.2000 | 374.8800 | 367.0000 |
| Smicro (m2/g) | 356.5400 | 240.2300 | 315.7200 |
| Smeso (m2/g) | 28.6600 | 134.6500 | 93.3100 |
| Vmicro (m3/g) | 0.1383 | 0.1114 | 0.1240 |
| Total acidity (mmol/g) | 3.8085 | 2.9635 | 1.2100 |
| Bio-Oil | ZSM-5 (wt %) | 0.3HZSM-5 | ZEOA | ||||
|---|---|---|---|---|---|---|---|
| Property | Raw | 0.5 | 1.0 | 2.0 | 3.0 | 0.5 | 0.5 |
| Proximate analysis | |||||||
| pH | 2.71 ± 0.01 | 2.83 ± 0.01 | 2.93 ± 0.01 | 2.98 ± 0.01 | 2.79 ± 0.01 | 2.91 ± 0.01 | 3.57 ± 0.01 |
| H2O (wt %) | 8.64 ± 0.23 | 9.50 ± 0.25 | 9.60 ± 0.23 | 10.00 ± 0.26 | 10.20 ± 0.23 | 9.60 ± 0.24 | 10.80 ± 0.26 |
| Viscosity (cP) * | 2.82 ± 0.14 | 2.80 ± 0.13 | 2.80 ± 0.13 | 2.78 ± 0.13 | 2.74 ± 0.14 | 2.81 ± 0.15 | 2.70 ± 0.13 |
| Density (g/cm3) * | 1.082 ± 0.0 | 1.059 ± 0.0 | 1.056 ± 0.0 | 1.040 ± 0.0 | 1.002 ± 0.0 | 1.051 ± 0.0 | 0.998 ± 0.0 |
| Ultimate analysis (wt % w.b) | |||||||
| Carbon (C) | 49.97 ± 1.50 | 59.92 ± 1.79 | 61.65 ± 1.66 | 63.84 ± 1.84 | 64.69 ± 1.82 | 65.61 ± 1.80 | 63.98 ± 1.79 |
| Hydrogen (H) | 6.79 ± 0.07 | 6.82 ± 0.07 | 6.63 ± 0.06 | 7.47 ± 0.07 | 6.6 ± 0.06 | 6.46 ± 0.07 | 6.34 ± 0.07 |
| Nitrogen (N) | 1.35 ± 0.03 | 0.95 ± 0.02 | 0.85 ± 0.02 | 0.53 ± 0.02 | 0.97 ± 0.02 | 0.89 ± 0.02 | 0.57 ± 0.02 |
| Sulfur (S) | 0.6 ± 0.01 | 0.51 ± 0.01 | 0.46 ± 0.01 | 0.43 ± 0.01 | 0.4 ± 0.01 | 0.44 ± 0.01 | 0.41 ± 0.01 |
| Oxygen (O) ** | 41.29 ± 1.07 | 31.8 ± 0.86 | 30.41 ± 0.88 | 27.73 ± 0.83 | 27.34 ± 0.79 | 26.6 ± 0.88 | 27.34 ± 0.88 |
| HHV (MJ/kg) | 26.23 ± 0.10 | 28.29 ± 0.10 | 27.87 ± 0.10 | 27.57 ± 0.10 | 28.23 ± 0.10 | 28.24 ± 0.10 | 27.29 ± 0.10 |
| Ultimate analysis (wt % d.b) | |||||||
| C | 54.7 | 66.21 | 68.2 | 70.93 | 71.17 | 72.58 | 71.73 |
| H | 6.38 | 6.37 | 6.15 | 7.07 | 6.15 | 5.97 | 5.76 |
| O | 36.79 | 25.81 | 24.2 | 20.93 | 21.18 | 19.99 | 21.41 |
| HHV (MJ/kg) | 28.92 | 31.5 | 31.07 | 30.88 | 31.29 | 31.48 | 30.87 |
| DOD (%) | 0.00 | 29.85 | 34.22 | 43.11 | 42.43 | 45.66 | 41.80 |
| Catalyst Type | Catalyst Loading (wt %) | Gas Composition (vol %) N2-Free Basis | |||
|---|---|---|---|---|---|
| CH4 | H2 | CO | CO2 | ||
| Raw | 0.00 | 2.72 | 0.56 | 14.04 | 25.32 |
| ZSM-5 | 0.50 | 2.40 | 0.32 | 20.87 | 29.94 |
| ZSM-5 | 1.00 | 2.26 | 0.31 | 22.97 | 33.32 |
| ZSM-5 | 2.00 | 2.01 | 0.34 | 24.67 | 36.07 |
| ZSM-5 | 3.00 | 1.94 | 0.36 | 26.14 | 37.95 |
| ZEOA | 0.50 | 2.19 | 0.33 | 26.16 | 31.65 |
| 0.3HZSM-5 | 0.50 | 2. 32 | 0.29 | 29.23 | 31.80 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammed, I.Y.; Kazi, F.K.; Yusup, S.; Alaba, P.A.; Sani, Y.M.; Abakr, Y.A. Catalytic Intermediate Pyrolysis of Napier Grass in a Fixed Bed Reactor with ZSM-5, HZSM-5 and Zinc-Exchanged Zeolite-A as the Catalyst. Energies 2016, 9, 246. https://doi.org/10.3390/en9040246
Mohammed IY, Kazi FK, Yusup S, Alaba PA, Sani YM, Abakr YA. Catalytic Intermediate Pyrolysis of Napier Grass in a Fixed Bed Reactor with ZSM-5, HZSM-5 and Zinc-Exchanged Zeolite-A as the Catalyst. Energies. 2016; 9(4):246. https://doi.org/10.3390/en9040246
Chicago/Turabian StyleMohammed, Isah Yakub, Feroz Kabir Kazi, Suzana Yusup, Peter Adeniyi Alaba, Yahaya Muhammad Sani, and Yousif Abdalla Abakr. 2016. "Catalytic Intermediate Pyrolysis of Napier Grass in a Fixed Bed Reactor with ZSM-5, HZSM-5 and Zinc-Exchanged Zeolite-A as the Catalyst" Energies 9, no. 4: 246. https://doi.org/10.3390/en9040246
APA StyleMohammed, I. Y., Kazi, F. K., Yusup, S., Alaba, P. A., Sani, Y. M., & Abakr, Y. A. (2016). Catalytic Intermediate Pyrolysis of Napier Grass in a Fixed Bed Reactor with ZSM-5, HZSM-5 and Zinc-Exchanged Zeolite-A as the Catalyst. Energies, 9(4), 246. https://doi.org/10.3390/en9040246







