1. Introduction
There are a variety of techniques to produce energy and sustainable fuels from biomass, and interest in producing the bio-oil via flash pyrolysis of biomass has been increasing recently. However, the bio-oil obtained via conventional flash pyrolysis is very different from crude oil originating from petroleum sources. Compared to fossil oil, the bio-oil is rich in oxygen-containing molecules, it has a heating value of about half of that of the conventional fuel oil and it is only partly miscible with conventional fuels. Due to these properties many problems arise in the handling and utilization of the bio-oil [
1]. Therefore, the bio-oil obtained from conventional flash pyrolysis needs to be upgraded before it can be used as a transportation fuel. For catalytic upgrading of bio-oil, a number of process routes have been explored using various catalysts. Deoxygenation of bio-oil can be carried out by a hydrotreating process with a zeolite-based catalyst. This hydrodeoxygenation (HDO) process is similar to the hydrodesulfurization (HDS) process
i.e., it is an essential part of oil refining [
2]. Hydro-treating requires large volumes of hydrogen which has a negative impact on the process economics. However, research on catalytic aftertreatment of bio-oil has not shown any promising results. Catalytic aftertreatment processes suffer from fast deactivation of the catalysts caused by coking and poor yield of hydrocarbons due to bypassing of the larger molecules in bio-oils [
3].
The oxygen content of bio-oils is usually very high, up to 45 wt% [
3,
4]. The type of oxygen component depends on the raw material and on the pyrolysis process conditions (temperature, residence time and heating rate) [
5,
6,
7]. In bio-oil, carboxyl, carbonyl, hydroxyl and methoxy functionalities are typically present [
5,
7,
8,
9]. These oxygenated components are mainly responsible for most of the deleterious properties of the bio-oil such as high viscosity, non-volatility, high acidity and resulting corrosiveness and extreme instability upon storage, lower energy density than the conventional fuel by 50%, immiscibility with fossil fuels, thermal instability and tendency to polymerize under exposure to air [
5,
6,
7,
8,
10,
11]. Thus, upgrading of bio-oil means removal of oxygen. As bio-oil contains large molecules derived from lignin, the use of cracking catalysts could selectively break down the lignin derivatives in the bio-oil. Since the lignin derivatives also contain a high proportion of oxygen [
12], breaking down these molecules will decrease the oxygen content and increase the hydrocarbons in the bio-oil, with a resulting high heating value of the bio-oil.
In order to improve bio-oil quality, several catalysts have been proposed in the literature.
In-situ catalytic flash pyrolysis of biomass over zeolite-type catalysts has attracted interest in the past decade because it does not require reducing gases, and the process releases the oxygen as carbon dioxide and water. The catalytically produced bio-oil presents a series of improved properties, such as stability and lower acidity. Lappas
et al. [
13] have proposed that the bio-oil stability depends on the catalyst type and activity (in addition to pyrolysis process conditions). It is a challenge to develop a single catalyst that can selectively remove problematic oxygenates in bio-oil and till now most of studies have been based on off-theshelf catalysts with little effort dedicated to the design of dedicated catalysts that can tailor the targeted properties of bio-oil. In the present work, zeolite-based catalysts were developed and optimised based on lab scale studies of biomass model components. The criteria for catalyst development is: (i) deoxygenation of oxygenated components in the vapor phase
in-situ during the pyrolysis process; (ii) acceleration of primary cracking and (iii) inhibition of secondary cracking. Zeolites are typical fluid catalytic cracking (FCC) catalysts being used in oil refining industry for upgrading low-octane components in the gasoline boiling range, and to isomerize low-octane linear olefins to high-octane branched olefins [
14]. Zeolites are inexpensive and have long and rich history of use in FCC units. The key properties of zeolites that influence their acidity, thermal stability, and overall catalytic activity are their structure, Si/Al ratio, particle size, and nature of the exchanged cation.
It is well known that some of the inorganic constituents of biomass including alkali metals and alkaline earth metals act as a catalyst during the thermal degradation process that can determine the rate of degradation and yield of char in pyrolysis. To study the effect of the acidity of Y zeolites, sodium, calcium and magnesium cation forms were investigated as these alkaline earth metals are indigenous to a variety of biomasses. Broido
et al. proposed that the mechanism of thermal decomposition of the biomass during pyrolysis is affected by the alkali metal cations present that form natural polymer chains from the monomers. Raveendran
et al. [
15] claimed that it is the cations rather than the anions of alkali metals that are mainly affecting the catalytic biomass pyrolysis. In the present work, Y zeolites with different acidities were prepared by exchanging cations and used for
in-situ biomass catalytic pyrolysis to investigate the effect of the acidity and cation forms of the Y zeolite on biomass pyrolysis and bio-oil properties. H-ZSM5 is a well-known catalyst used for cracking, deoxygenation and synthesis of aromatic hydrocarbons, and it is investigated in current work to study the effect of different zeolite structures on biomass pyrolysis and bio-oil properties.
Fractionation of the bio-oil vapors by staged condensation in the downstream phase of the pyrolysis process is an option to tailor the bio-oil composition. Fractionation of the bio-oil can also be achieved for example by distillation, but re-heating of the bio-oil results in the formation of cross-linked solid carbonaceous residues because of the high reactivity of some fractions present in the bio-oil. There is very limited research available on fractionation during condensation of biomass pyrolysis vapors, Westerhof
et al. [
16] studied the effect of the condenser conditions on the water and organics content in the liquid obtained in the first condenser. Effendi
et al. [
17] studied the fractional condensation to concentrate phenolic compounds in a targeted fraction of the bio-oil. Oasmaa
et al. [
18] studied the use of fractional condensation to remove the light fractions from the bio-oil and found an increase the stability the bio-oil when the light fraction was replaced by an alcohol. In the present work, online fractionation of the catalytic pyrolysis vapors was used to improve the overall quality of the bio-oil, including its water content, elemental composition and calorific value. Online fractionation was employed using two condensers operated at different temperatures to produce two different liquids, an organic rich one and a water rich one.
A bench scale unit consisting of an entrained flow downer reactor system is designed for
in-situ catalytic flash pyrolysis of biomass. The entrained flow downer reactor type is selected because it features easy operation handling, and demonstrates high biomass heating rates while maintaining good control of the reaction conditions, for instance the residence time of reactants and products [
19,
20]. Experiments were conducted for
in-situ upgrading of pyrolysis vapor with premixed feedstocks of catalyst and biomass, using different cation forms of faujasite zeolites, different zeolite structures (MFI zeolites), different reaction temperatures and different catalyst to biomass ratios. Furthermore, fractional condensation results are presented where flash pyrolysis, catalytic upgrading and fractional condensation are integrated in a single pyrolysis system.
2. Materials and Methods
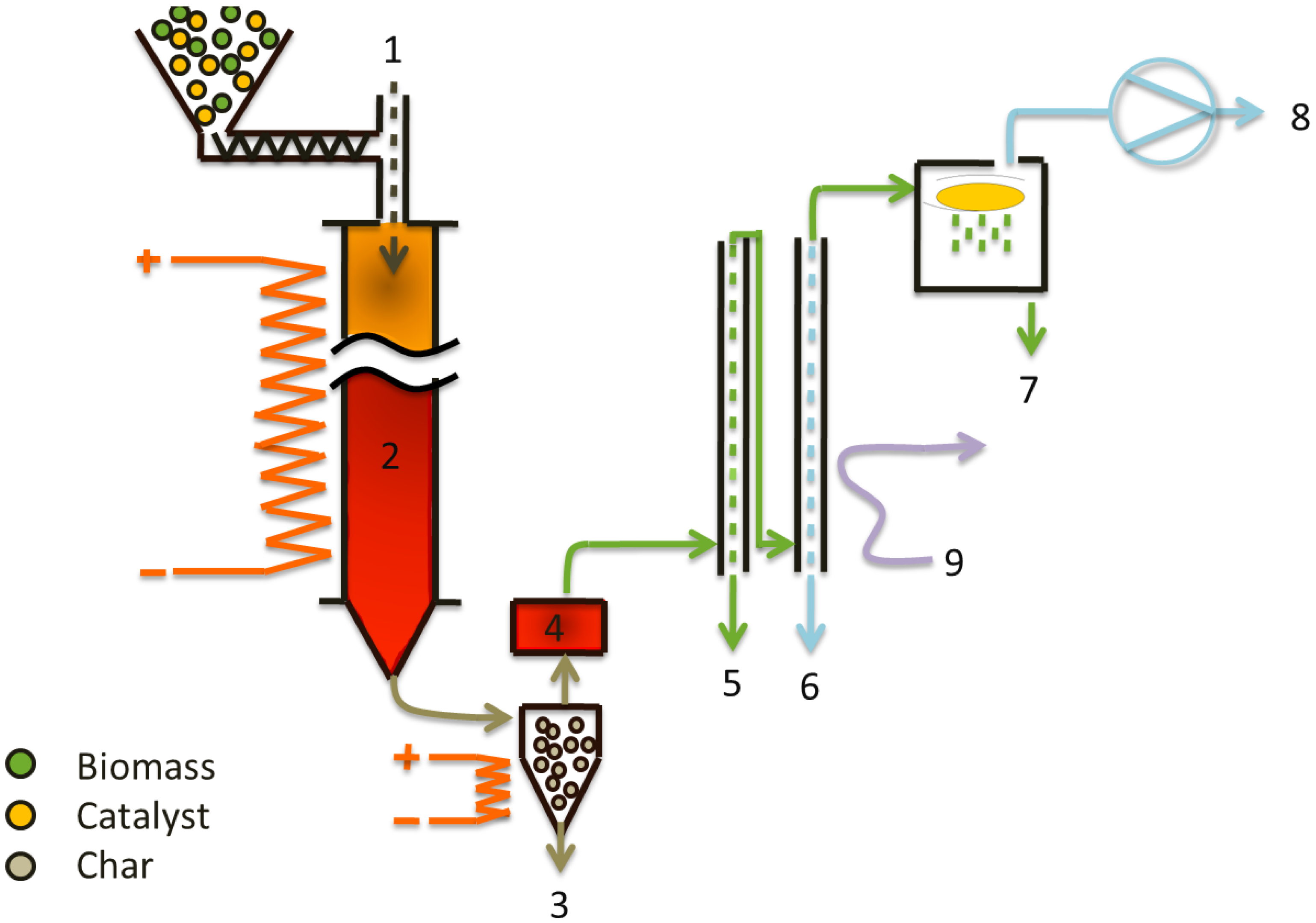
A continuous bench scale unit of 1 kg/h feedstock (biomass or/and catalyst) capacity has been used for the catalytic flash pyrolysis of woody biomass. The experimental set-up used was described in detail in an earlier publication [
21], and a schematic of the unit is presented in
Figure 1. An entrained flow reactor made of a cylindrical quartz tube of 4.2 m length with an internal diameter of 50 mm is used for
in-situ catalytic flash pyrolysis to produce bio-oil. The catalyst and biomass are mixed before feeding to the pyrolysis unit. The residence time of biomass and pyrolysis products in the reactor is controlled with nitrogen carrier gas. The vapors, gases and solids leaving the reactor enter tangentially into a cyclone that allows removal of solids particles up to 20 µm. The solids consist of char (ash and unconverted biomass) for non-catalytic experiments, and char plus spent catalyst for
in-situ catalytic experiments. Due to the wide particle size distribution of the biomass and the catalyst, fine particles are separated in a hot filter element downstream of the cyclone. The solids recovery system allows complete removal of char and catalyst. Knowing the concentration of catalyst in the biomass and the amount of feedstock consumed for each experiment, the amount of catalyst in the solids can be determined.
The condensation unit consists of two double tube heat exchangers that are operated with a coolant mixture of glycol and water at −5 °C, the bio-oil fractions are collected together from both condensers except for online fractionation experiments. When fractionation of pyrolysis vapors is required, tap water is used in first condenser instead of glycol/water mixture and water flow rate is controlled to set desired temperature of the outlet gas/vapor stream, the second condenser is operated with coolant at −5 °C to for maximum recovery of liquid. The light and heavy fractions of bio-oil are recovered in the first and second condenser respectively, and collected separately from both condensers. Separation of light fraction from bio oil can improve its quality because the light fraction carries mainly the water and oxygenate components. In this two-step condensation scheme, major portion of the condensable is recovered. Other operating conditions and parameters are listed in
Table 1.
2.1. Product Analysis
The non-condensable gases are mainly composed of carbon dioxide, carbon monoxide and C1–C3 hydrocarbons. The volumetric flow rate of these gases is measured with a gas flow meter and a sample of this stream is pumped to an on-line gas analysis unit. Infrared analyzers are used to measure CO, CO2 and an FID analyzer is used to measure hydrocarbons. The elemental composition of the liquid is analyzed with an elemental analyzer (Flash 2000 Thermo Scientific, Breda, The Netherlands). The water contents of the oil are quantified by Karl Fisher titration, and a bomb calorimeter is used to determine the heating value of the bio-oil. A GC-MS instrument (Agilent, Amstelveen, The Netherlands) is used to analyze the bio-oil and the NIST8 library is used to identify the components.
2.2. Biomass
The biomass used for the experiments consisted of wood fibres commercially available under the trade name LIGNOCEL from J. Rettenmaier & Söhne GmbH (Rosenberg, Germany). The biomass particle size varied from 0.1 to 1 mm. The ultimate and proximate analysis, and the bio-chemical composition [
16] of the biomass are presented in
Table 2.
2.3. Catalysts
Solid acid faujasite (Y and MFI ZSM5) zeolites were used to catalyze the deoxygenation reactions. The basic properties of these catalysts are presented in
Table 3. The faujasite zeolites were used in different cation forms, H-Y and Na-Y were obtained from Zeolyst International (Conshohocken, PA, USA), Ca-Y and Mg-Y were prepared by Petrobras (Rio de Janeiro, Brazil) by ion exchange of Na-Y zeolite. H-ZSM5 was obtained from Albemarle B.V. (Amsterdam, The Netherlands). All the catalysts were in powder form, with a particle size ranging between 5 µm to 50 µm.
3. Results and Discussion
Preliminary experiments were conducted to optimize the operating conditions, e.g., the solids residence time and reaction temperature (T = 450–550 °C, 2 s < residence time < 4 s) for a maximum solid conversion and liquid yield. In all these tests the stability of the unit was satisfactory and reproducible experiments could be performed for 1 to 2 h. A reasonably good mass balance of around 90% and liquid yields around 60% were achieved.
An optimum liquid yield was obtained at 500 °C reactor temperature and 4 s vapor residence time in the reactor. Maximum biomass conversion was achieved at 4 s vapor residence time, and vapor residence times longer than 4 s did not improve the biomass conversion and had a negative effect on pyrolysis product properties. Longer vapor residence times caused a significant increase in the production of gases and water contents of the liquid due to undesired secondary cracking of the bio-oil vapors. For catalytic experiments, these operating conditions were fixed in order to compare the effect of catalyst yield and characteristics of biomass pyrolysis products. Catalytic experiments were performed with a premixed feedstock of biomass and catalyst. The two step condensation scheme was employed for fractional condensation of bio-oil to separate the organic and aqueous rich phases of bio-oil.
3.1. Effect of the Catalyst to Biomass Ratio
For heterogeneous catalytic systems, the contact between catalyst and biomass/vapor is an important issue and poses practical challenges. The contact can be improved by increasing the catalyst to biomass ratio. ZSM5 and Y zeolites were used to study the effect of the catalyst to biomass ratio on the yield and properties of the pyrolysis products. The results are presented separately for both catalyst types in Subsections 3.1.1 and 3.1.2, respectively.
3.1.1. Effect of the H-ZSM5 to Biomass Ratio
To study the effect of the H-ZSM5/biomass ratio on the yields and characteristics of the pyrolysis products, experiments were performed with mixture of 0, 10, 30, and 50 wt% of H-ZSM5, and 100, 90, 70 and 50 wt% of biomass, respectively. H-ZSM5 and biomass were physically mixed and this premixed feedstock was used for in-situ catalytic pyrolysis.
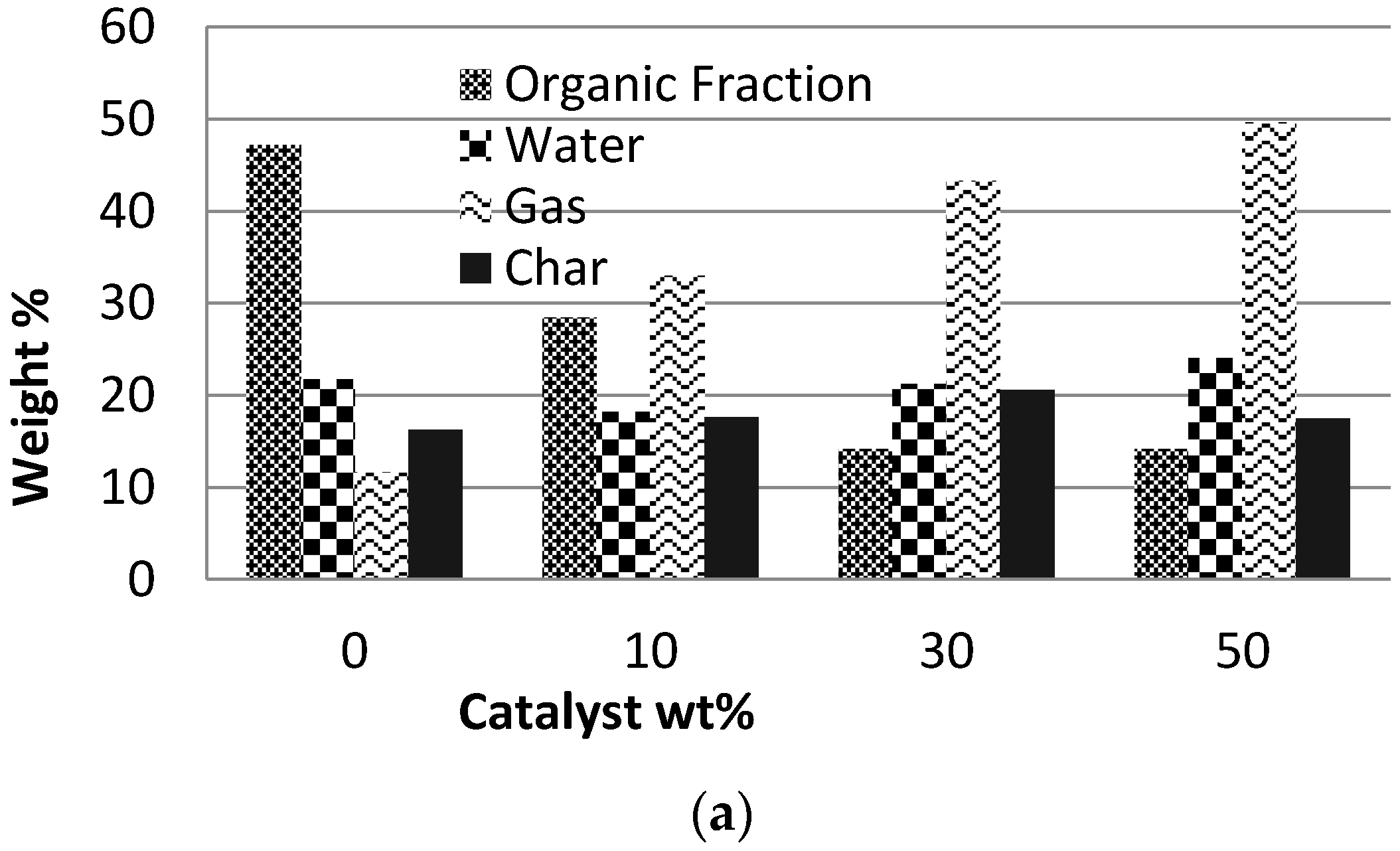
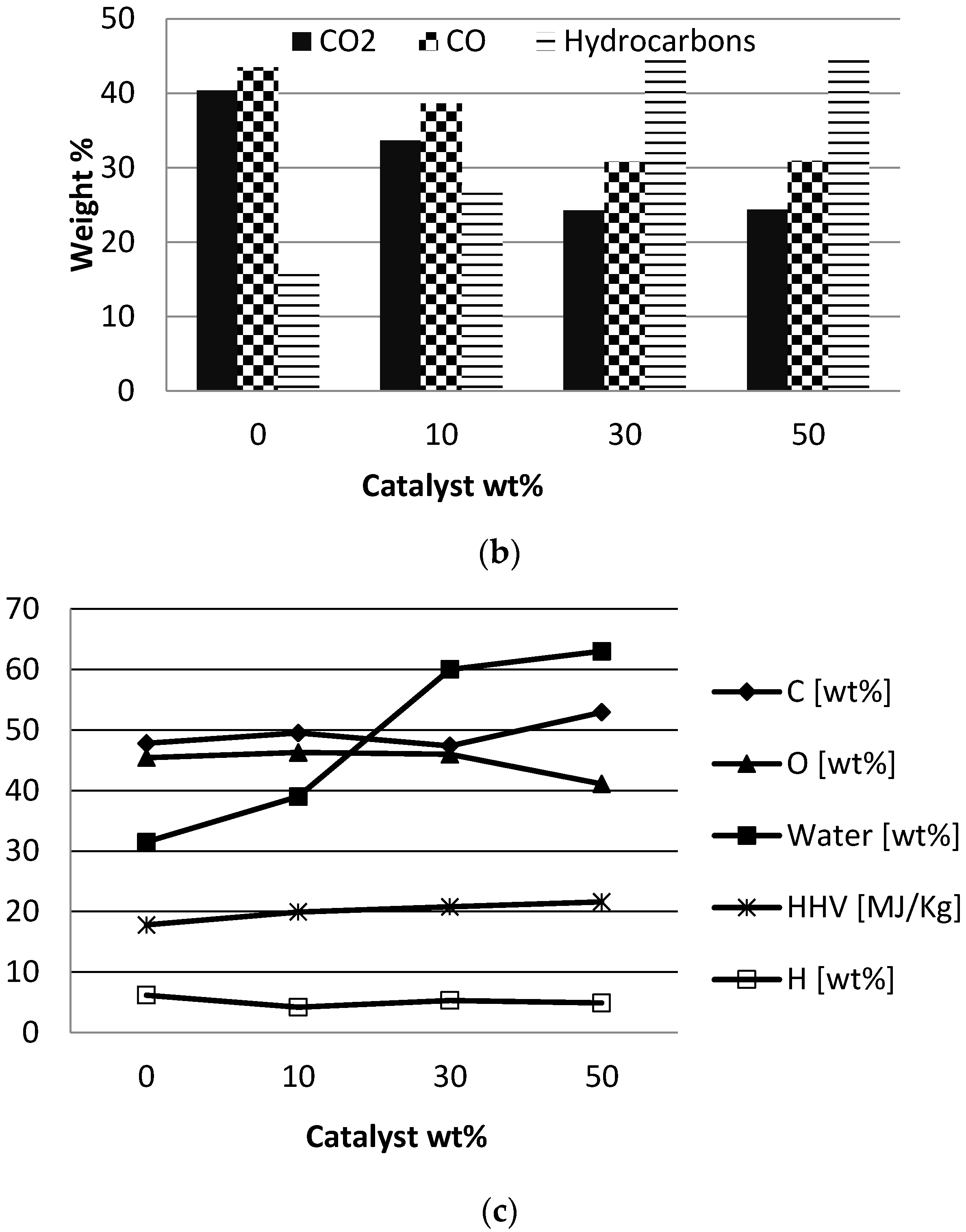
Figure 2 shows the product distribution (dry basis) and, the gas and boil-oil characteristics. Bio-oil was collected in one condenser (no fractionation) and its water content was determined by Karl Fisher titration, and the organic and water contents/fractions of the bio-oil are presented separately for a better comparison.
The catalyst/biomass ratio had a significant effect on the yields of the different pyrolysis products (see
Figure 2a). A higher catalyst/biomass ratio led to an increase in the gas production while the organic liquid fraction significantly reduced. Using 50% H-ZSM5, the organic liquid fraction reduced from 47.2% to 14.9% and the gas fraction was increased from 11.6% to 46.8% compared to the non-catalytic experiment.
The catalyst/biomass ratio had no significant effect on the char content. The gas composition is presented in
Figure 2b as relative selectivity to CO, CO
2 and hydrocarbons. H-ZSM5 had a significant effect on the gas composition compared to the non-catalytic situation, but changing the catalyst/biomass ratio did not alter the gas composition to a great extent. A biomass mixture with 50% H-ZSM5 produced a gas with 32.5% hydrocarbons and a relative concentration of CO
2 and CO of 25% and 42.5%. A high catalyst/biomass ratio enhanced the secondary cracking of the oil vapors and caused formation of hydrocarbon gases. Increasing the H-ZSM5 concentration did not result in deoxygenation of the bio-oil vapor via the preferred route of decarboxylation, but a significant amount of oxygen is released via decarbonization as CO gas. During catalytic de-oxygenation of bio-oil vapors, the oxygen may be given off by dehydration, decarbonization and decarboxylation leading to the formation of water, CO and CO
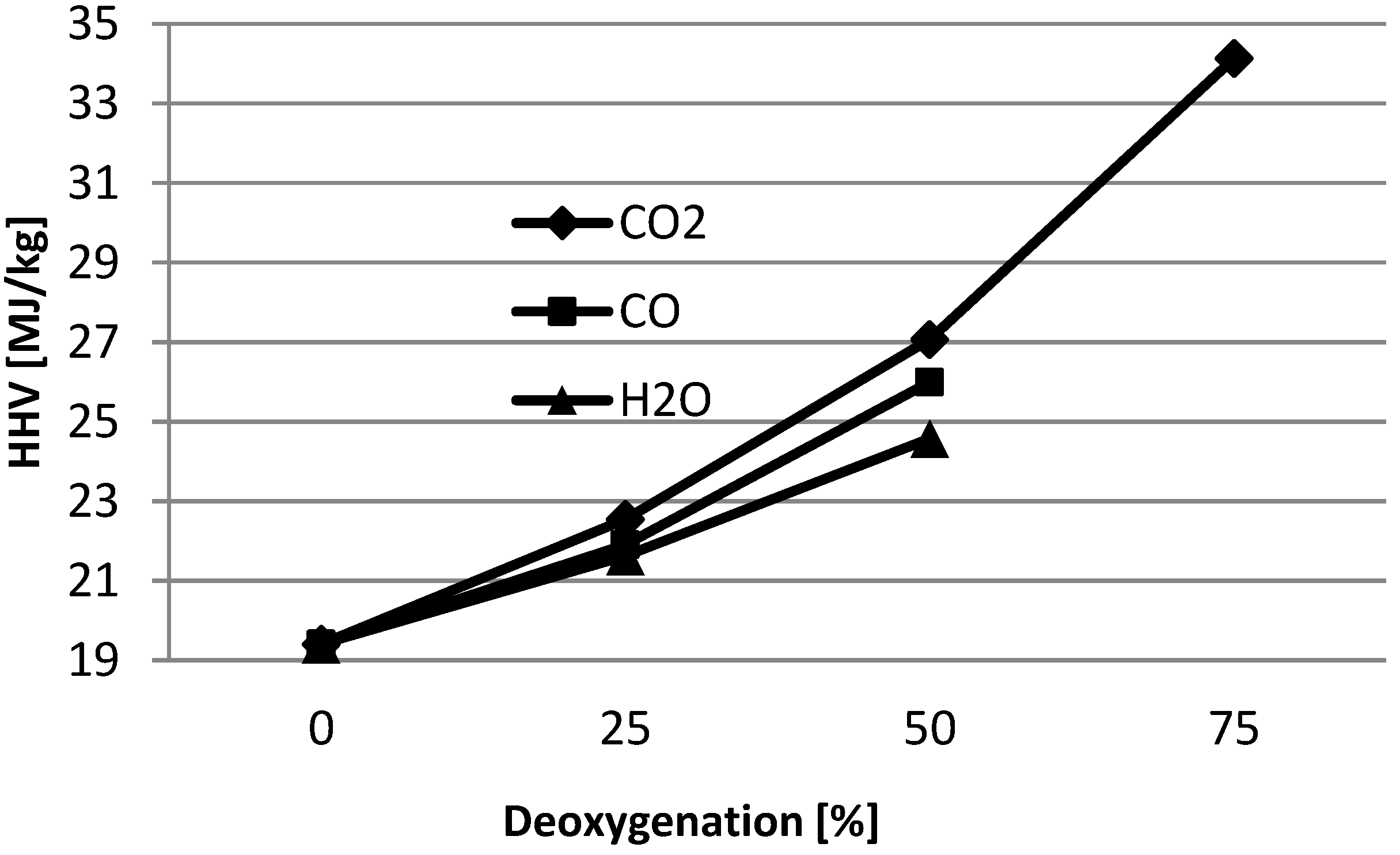
2, respectively. In order to assess the best mode of de-oxygenation of the vapors, basic calorific value calculations were done based on the elemental formula (C
6H
8O
4) for bio-oil (see
Figure 3). Calculation shows that 75% de-oxygenation via de-carboxylation will improve the heating value of the bio-oil from 19.4 MJ/Kg to 34.1 MJ/Kg. It is evident from these calculations that the decarboxylation is the preferred route for deoxygenation in comparison to dehydration. Decarboxylation removes oxygen with minimal carbon loss, retains the hydrogen in bio-oil and thereby increases heating value, decreases the aromatic compounds, minimizes the water content of the bio-oil. Hence, in order to the quality of the bio-oil, selective scission of bonds should follow the order of C-C > C-O > C-H.
A detailed analysis of bio-oil is presented in
Figure 2c. The elemental composition and higher heating value of bio-oil is given on a dry basis. H-ZSM5 caused an increase in the carbon and a decrease in the oxygen content, while the hydrogen content remained the same. At 50% H-ZSM5 concentration, the oxygen content of bio-oil is reduced from 45.4% to 41.6% and the higher heating value increased from 17.8 to 20.7 MJ/kg. Although this is a minor difference, a considerable difference in physical appearance of the bio-oil could be noted. The thermal pyrolysis oil was highly viscous and heavy tar was observed in the condenser, while the bio-oil obtained via catalytic pyrolysis was less viscous and no heavy fraction was observed in the condensers. Obviously the heavy fraction of the bio-oil was cracked to lighter fractions and gaseous components at high catalyst concentrations.
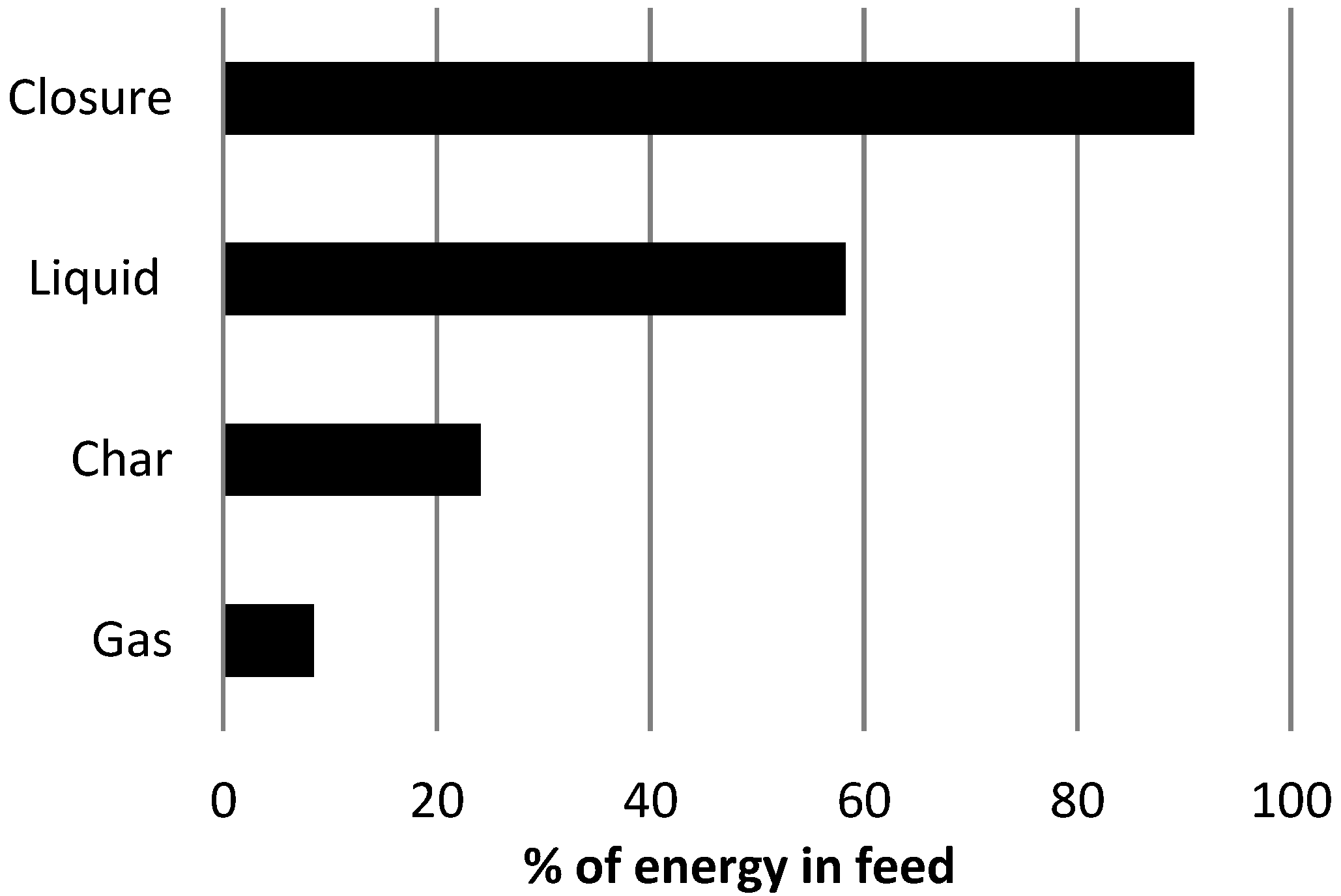
Energy balance for the non-catalytic pyrolysis of biomass is presented in
Figure 4. Energy yields for each product were calculated from the mass yields (see
Figure 2, non-catalytic pyrolysis experiment) and the gross energy contents, or higher heating values (HHV) of the corresponding products. HHV for liquid and char was measured, and calculated for the gas stream. A good energy balance closure was achieved. The optimum type of catalytic pyrolysis reactor should have an integrated heat recovery system from combustible gases, and char and can increase the overall process efficiency and economics.
3.1.2. Effect of Na-Y to Biomass Ratio
To study the effect of the faujasite zeolite/biomass ratio on product yields (liquid, gas and char) and on the liquid product characteristics, experiments were performed using 0, 10, 30, and 50 wt% of Na-Y zeolite. The product yields are presented in
Figure 5a and the results show that an increasing Na-Y/biomass ratio had a significant effect on the product yields. The trends are similar to those of H-ZSM5. A higher Na-Y/biomass ratio resulted in more formation of gas and a lower organic fraction yield. For 50% Na-Y, the organic liquid fraction reduced from 47.2% to 14.1% and the gas yield increased from 11.6% to 49.7% and there was no considerable change in the char yield compared to non-catalytic pyrolysis. The gas composition is presented in
Figure 5b. For 50% Na-Y, the hydrocarbons concentration in the gas increased from 16.1 % to 44.7% while the CO
2 and CO concentration decreased from 40.4% and 43.5% to 24.4% and 31% respectively compared to the non-catalytic experiment. A higher Na-Y/biomass ratio favored the secondary cracking of vapors and the effect is larger than it was in case of H-ZSM5. The bio-oil analysis is presented in
Figure 5c. For 50% Na-Y in the biomass mixture, the oxygen content of the bio-oil is reduced from 45.4% to 41.1% and the higher heating value of the bio-oil increased from 17.8 to 21.6 MJ/kg compared to the non-catalytic situation.
The experimental results show that an increasing catalyst/biomass ratio enhanced the formation of gas and lowered the organic fraction yield. The higher gas yields in the presence of the H-ZSM5 and Na-Y catalysts are due to the secondary cracking and deoxygenation reactions of the product vapors. Oxygen is removed as CO and CO2 and hydrocarbon gases are produced as a result of the cracking reactions. The lower organic liquid yield due to gas formation is more or less compensated by the better quality. The thus produced could be used for e.g., local electricity production in a gas engine.
3.2. Effect of the Catalytic Pyrolysis Reaction Temperature
To investigate the effect of the reaction temperature on the catalytic flash pyrolysis process, experiments were conducted with 50% H-ZSM5 at 550 °C and 500 °C. The product yields and composition of the gaseous products are compared in
Table 4 while the characteristics of the bio-oil are presented in
Table 5, respectively. Increasing the reaction temperature caused a decrease in the organic liquid yield and an increase in the gas yield. It is evident from the gas composition that a higher reaction temperature enhanced the secondary cracking of the bio-oil vapors resulting in a lower yield of organic liquid. A lower char yield at a higher temperature is expected because elevated reaction temperatures are favorable for a better conversion of lignin components in the biomass. A higher reaction temperature resulted in a significant increase in the higher heating value of the bio-oil (22.6 MJ/kg) because of a higher deoxygenation achieved, but this improvement is at the costs of a lower yield of organic liquid.
3.3. Effect of Zeolite Structures
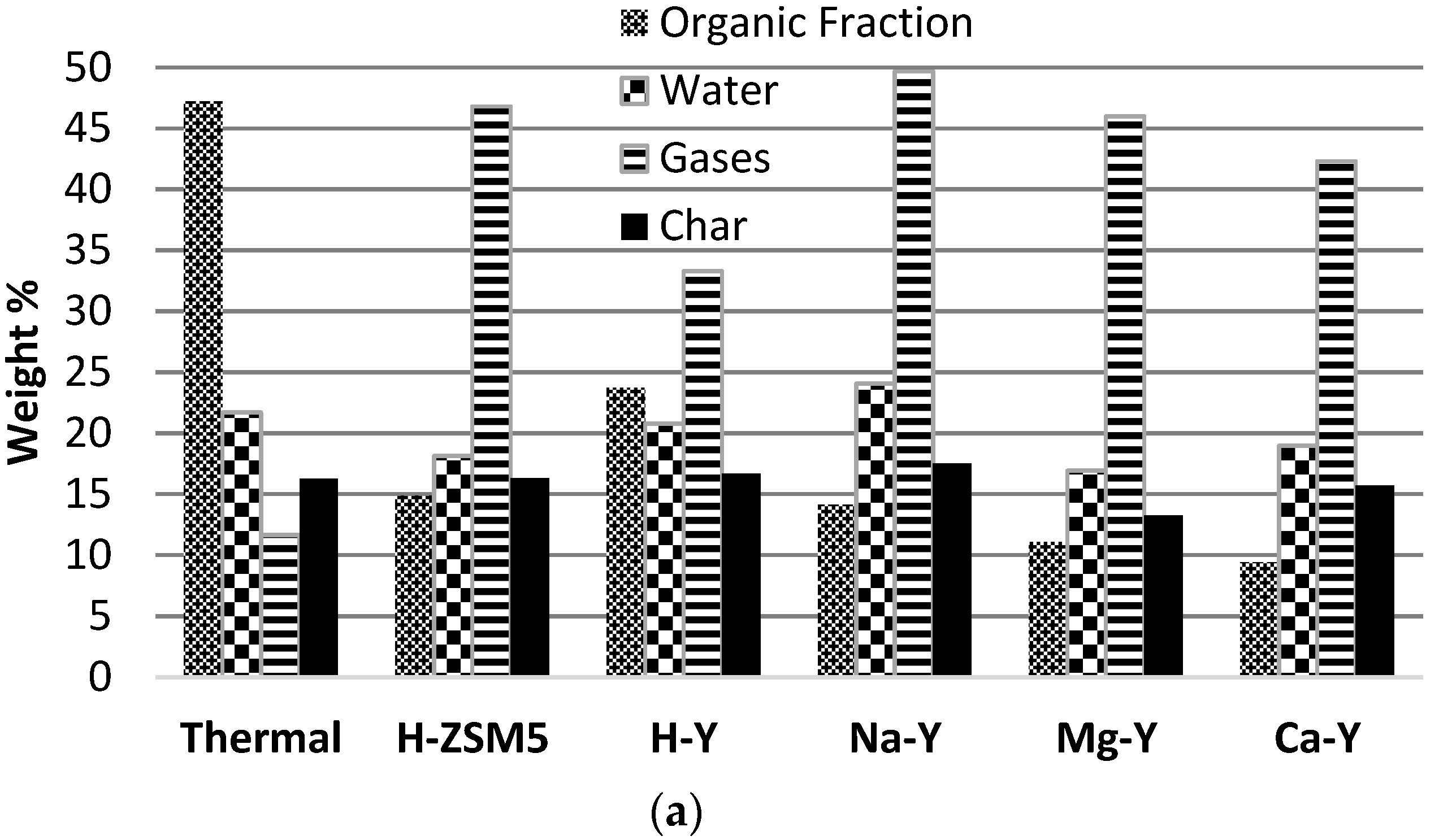
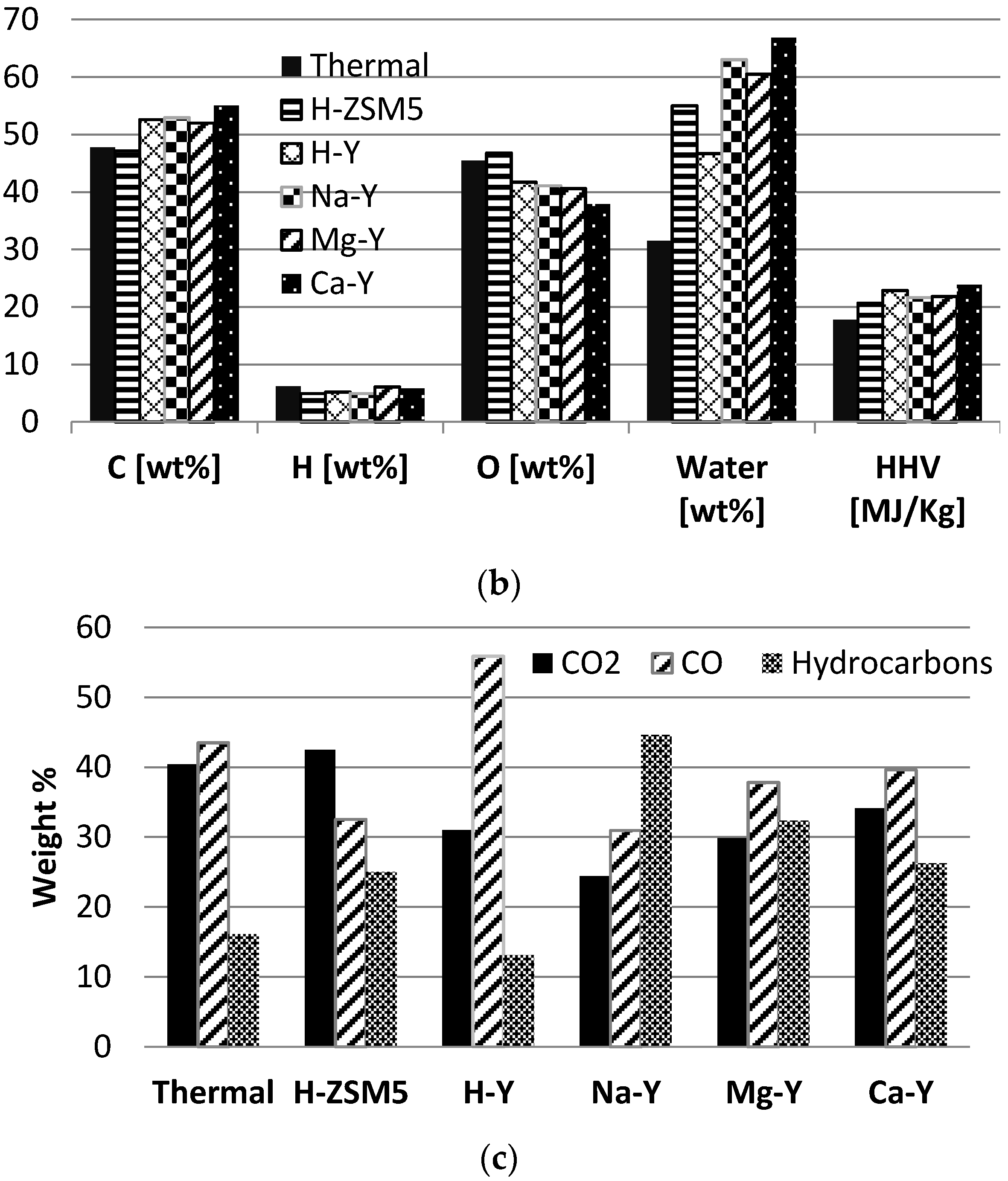
To investigate the effect of different zeolite structures such as faujasite (H-Y) and MFI (Pentasil/H-ZSM5), experiments were performed with H-Y zeolite and the results were compared with those with H-ZSM5. For comparison 50 wt% of catalyst was used for this series of experiments to observe the clear effect of the zeolite structure. The product yields (dry basis), gas composition and liquid characteristics for 50% H-Y are compared with the results obtained for H-ZSM5 (
Figure 6a–c).
It can be seen that H-Y produced much less gas resulting in a higher yield of organic liquid. H-Y produced less hydrocarbon gas and more CO and CO2 gas; this is the preferred route for de-oxygenation of the bio-oil vapors to keep maximum hydrogen and carbon in the bio-oil. It is evident that H-Y is less prone to secondary cracking of the oil vapors.
A major difference between both catalysts structures becomes clear from the liquid characteristics. H-Y was more effective for the deoxygenation of the bio-oil vapors and lowered the oxygen content of the bio-oil to 41.7, and increased the carbon and hydrogen content of the bio-oil to 56.2 and 5.2 wt%, respectively. For 50% H-Y the heating value increased from 17.8 to 22.9 MJ/kg compared to non-catalytic experiment. This higher heating value can be attributed to the changed elemental composition of the bio-oil. H-Y has a superior performance compared to H-ZSM5 with respect to the energy recovery in the bio-oil.
H-Y produced an organic liquid with a higher yield and higher calorific value, and with lower water content. A detailed comparison of the two zeolite structures is carried out in
Section 3.4 based on the chemical composition of the produced bio-oils.
3.4. Effect of Varying Acidities of Faujasite Zeolite
The nature of the (exchanged) cation is a key property of faujasite zeolite catalysts and it influences the acidity of zeolite and its catalytic activity. To investigate the effect of varying acidities of faujasite zeolite on catalytic biomass pyrolysis, experiments were performed with Mg-Y and Ca-Y, and a comparison was made with H-Y, Na-Y and non-catalytic experiments. For all these experiments 50 wt% of catalyst was used in the biomass mixture. The acidity of Y zeolite is changed in the order of H-Y > Na-Y > Mg-Y > Ca-Y. The product yields (dry basis), gas composition and liquid characteristics are presented in
Figure 6c.
Compared to the non-catalytic experiment, all cation forms of faujasite zeolites produced less organic liquid and more gas. Among the all faujasite zeolites, H-Y produced the lowest amount of gas and highest amount of organic liquid. Reducing the acidity of Y zeolite resulted in more CO and CO2 formation and fewer hydrocarbons in the gas, except for H-Y. H-Y produced the highest amount of CO and lowest amount of hydrocarbon gas while the production of CO2 was quite similar to other zeolites.
The liquid product is not very different in terms of elemental composition. The oxygen content slightly decreased with a reducing acidity of Y zeolite while the carbon content increased in all cases. Acidic zeolite, H-Y, showed a superior performance compared to the other zeolites in terms of keeping the energy in the liquid product. It produced the highest amount of organic liquid with almost similar elemental composition, slightly lower calorific value and lower water formation compared to the other zeolites.
3.5. Chemical Characterization of Liquid
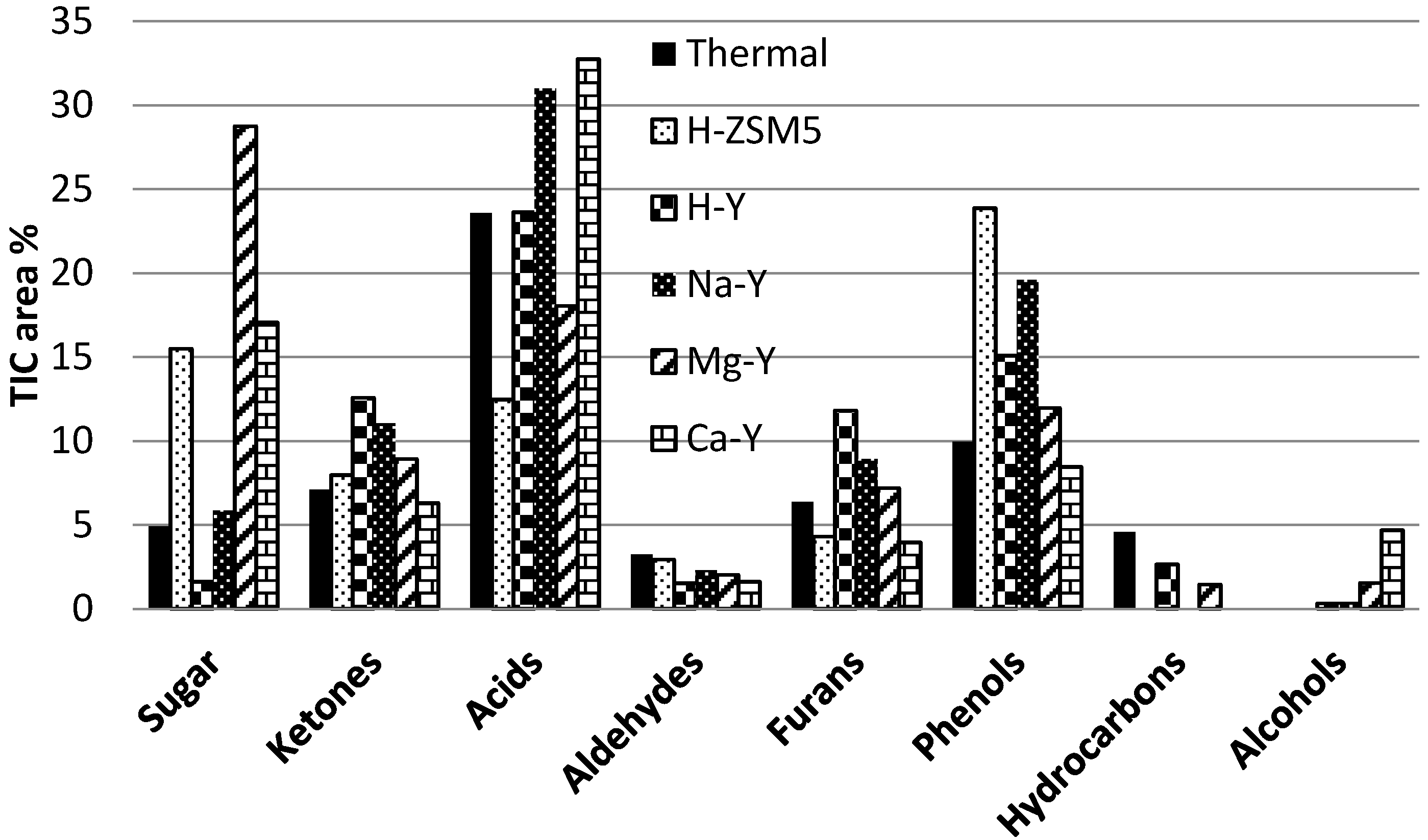
The bio-oil obtained from the catalytic and non-catalytic pyrolysis experiments was diluted in acetone and analyzed in a GC–MS. A wide range of organic compounds are found in the bio-oil. In
Figure 7, the chemical composition of the bio-oil has been described and the compounds have been classified according to their chemical nature as hydrocarbons, phenols, furans, acids, alcohols, aldehydes, ketones, sugars and aromatics.
Hydrocarbons are assumed to be the desirable fractions since they are chemicals of high commercial value. In addition to the hydrocarbons, compounds like phenol and its alkylated derivatives also have a high value, especially for the resin or adhesive industry and they could make the pyrolysis process economically more feasible. Furans are stable compounds with a high energy value and fuel compatible. While the oxygen-containing compounds, such as acids and carbonyls, are considered as undesirable fractions. The reduction of the acids is important factor to improve the corrosive nature of the bio-oil and the reduction of carbonyls is important for its stability. These two factors are critical with respect to handling, storage and fuel applications of the bio-oil. A catalyst is expected to produce more desirable and less undesirable compounds in the bio-oil.
Acids, phenols, ketones and furans were the dominant groups of bio-oil and acetic acid was the single most dominant and undesired compound in the bio-oil. In general it can be said that increasing the acidity of the zeolite catalyst lowered the acid yields except for Mg-Y. H-ZSM5 has the highest acidity among the investigated zeolites and produced the lowest amount of acids. The acid yield was reduced to half of what was obtained during non-catalytic pyrolysis. Increasing the acidity of zeolites enhanced the formation of ketones except for H-ZSM5. H-Y produced the highest amount of ketones and Ca-Y formed the lowest amount of ketones. All zeolites reduced the amount of aldehydes in the bio-oil, and the effect was more pronounced for lower acidities of the zeolites, except for H-Y that produced the least amount of aldehydes. Increasing the acidity of the zeolites favored the production of furans except for H-ZSM5. Both H-ZSM5 and Ca-Y produced the lowest amount of furans. Reducing the acidity of zeolites favored the formation of valuable alcohols, and Ca-Y produced the highest amount of alcohols. Compared to the non-catalytic runs, the application of zeolite catalysts increased the production of phenols, a valuable commodity for resins and adhesive industry. In general, increasing the acidity of zeolites favored the phenols production except for H-Y. Zeolites did not favor the production of hydrocarbons i.e., the important compound for fuel application. Only H-Y zeolite was able to almost completely convert the sugar components.
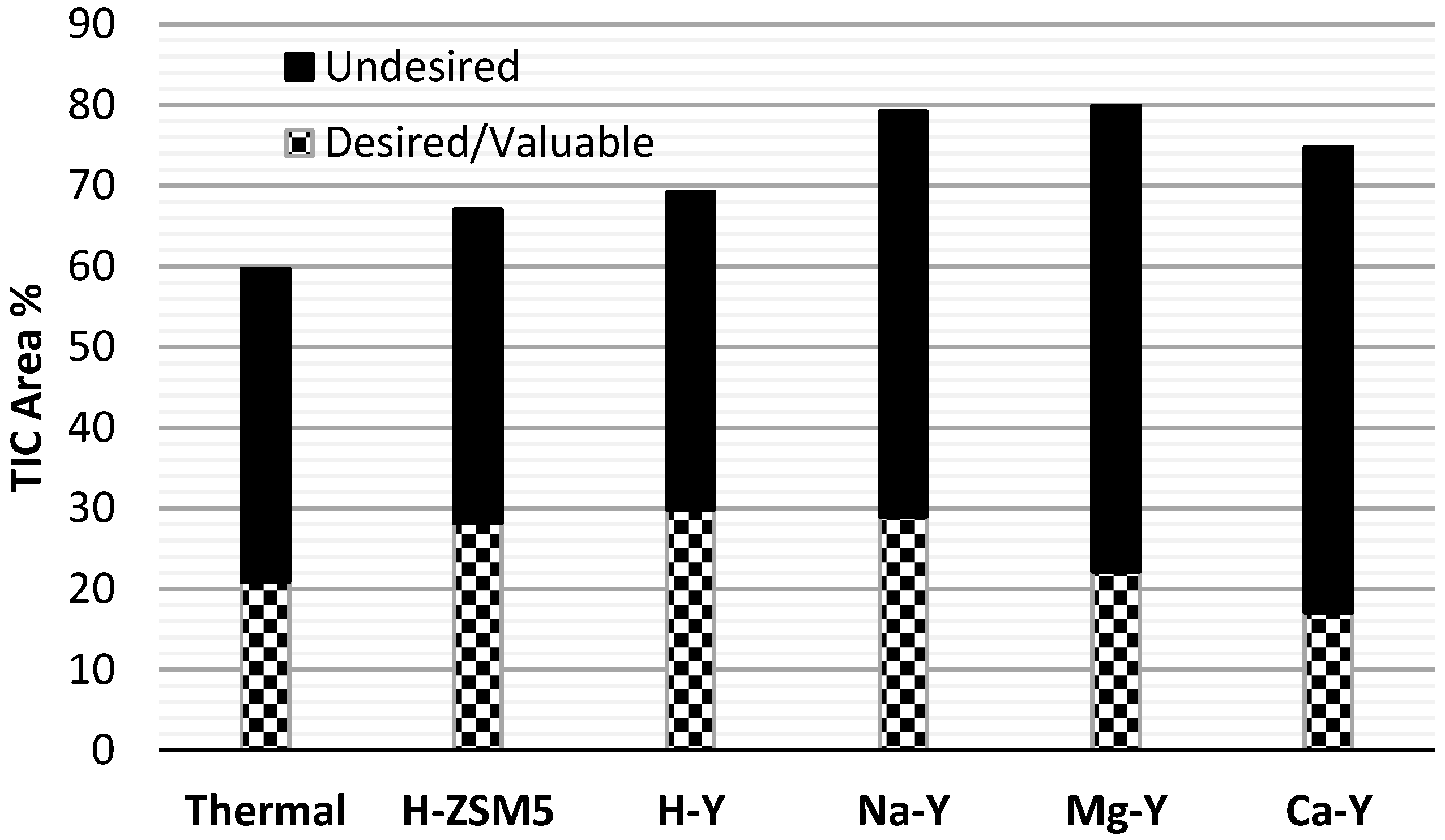
To evaluate the overall performance of the zeolite catalysts and the effect of their acid nature on the chemical composition of the bio-oil, the chemical compounds are grouped in so called undesired and desired/valuable compounds (see
Figure 8). Acids, ketones, aldehydes and sugars are grouped as undesired compounds, while hydrocarbons, phenols, furans and alcohols are grouped as desired and valuable compounds. Compared to the non-catalytic (thermal) experiments, both H-ZSM5 and H-Y were successful in enhancing the yields of the desired components and reducing the undesired components in the bio-oil. Na-Y also produced a high amount of desired compound but also enhanced the formation of undesired compounds. Reducing the acidity of Y-zeolite had a negative effect on the bio-oil composition and produced less desired compounds and more undesired compounds. Summarizing it can be stated that H-Y produced a bio-oil with both a high chemical and calorific value in combination with a high organic liquid yield. H-Y has a high potential for the production of a high quality fuel via catalytic flash pyrolysis of lignocellulosic biomass. H-ZSM5 is a very close competitor of H-Y but it results in a lower yield of organic liquid.
The result obtained in this study with zeolite catalysts in an entrained flow reactor are comparable to other studies conducted at bench scale units. Aho
et al. [
22] performed catalytic pyrolysis of pine wood in a fluidized bed reactor to investigate the effect of different structures of acidic zeolite catalysts. H-Y and H-ZSM5 were tested as catalysts and the chemical composition of the bio-oil was found to be dependent on the structure of acidic zeolite catalysts. H-Y zeolite and H-ZSM5 catalyst significantly reduced the organic liquid yield. The formation of acids and alcohols over H-ZSM5 was lower while the formation of ketones was higher than over the H-Y zeolite. Lappas
et al. [
23] used FCC and H-ZSM5 for catalytic flash pyrolysis of biomass in a circulating fluid bed reactor with continuous solids regeneration. The effect of specific operating variables including the type of catalyst and the catalyst/biomass ratio on the final liquid product quality and yield was studied. FCC and H-ZSM5 catalysts produce high gas yields, with dominating oxygenated gaseos compounds as CO
2 and CO. A high catalyst to biomass ratio enhanced the production of additional water and resulted in a lower yield of organic liquid obtained with less oxygenated compounds but no improvement in the calorific value. H-ZSM5 produced more carbonyls while the production of acids, HCs, phenols and heavy oxygenates was not significantly changed. Atutxa
et al. [
24] also reported that bio-oil from catalytic pyrolysis with H-ZSM5 was less oxygenated than oil from thermal pyrolysis. Atutxa
et al. used a conical spouted bed reactor to investigate catalytic pyrolysis of pine wood at 400 °C and found a substantial increase in gaseous products (mainly CO
2 and CO) and water, and a corresponding decrease in the organic liquid and char yield compared to thermal pyrolysis. The obtained liquid product was less corrosive and more stable than thermal bio-oil.
It can be concluded that in general zeolites reduce oxygenates compounds in the bio-oil via various routes of deoxygenation e.g., dehydration, decarbonylation and decarboxylation. However, main drawback of the zeolites for catalytic pyrolysis is a significant decrease in bio-oil organics yield due to the increase in the production of gases, water and coke. Organics yield in the bio-oil can be increased if deep cracking and formation of hydrocarbon gas can be avoided, it may be controlled by manipulating the strength and concentration of acid sites in zeolites.
3.6. Online Fractionation via Staged Condensation of Pyrolysis Vapors
Online fractionation via two staged condensation of pyrolysis vapors was employed to investigate the possibilities to improve the composition and thus the quality of the bio-oil. The pyrolysis vapors leaving the pyrolysis reactor were condensed via two condensers at different temperatures. The first condenser was used to condense the heavy and middle fractions of the bio-oil and it was operated at an outlet temperature of 55 °C. The second condenser was used to condense the water and light oxygenate fractions of the bio-oil and operated with a coolant (glycol/water) temperature of −5 °C resulting in an outlet gas temperature of 6–8 °C. The fractionation experiments were performed with 50% Na-Y and 50% H-ZSM5 and the liquid characteristics from the first condenser are compared with the results of the experiments without fractionation in
Table 6.
Experiments 1 and 3 were performed with both condensers at the same low temperature and the liquid was collected together (no fractionation), while experiments 2 and 4 were performed with two staged condensation. For the fractionation experiments, the liquid obtained in the second condenser was mainly water (the water content was more than 90 wt%) and it was not possible to perform a reliable and accurate elemental analysis of the liquid because of this high water content. The liquid obtained from the first condenser was rich in organic compounds and its characteristics are presented here in
Table 6 being the fraction of interest. A significant difference in the composition of the bio-oil is seen in case of fractionation. In case of Na-Y and fractionation, the water content goes down to 31 wt%, the hydrogen content increased and the higher heating value of the bio-oil increased to 23.1 MJ/kg.
In case of H-ZSM5, the fractionation experiment was more effective and resulted in a bio-oil with a water content of 11 wt%. The oxygen content of the bio-oil decreased to 37.9 wt% while both the hydrogen and carbon content increased. The higher heating value of the bio-oil increased to 23 MJ/kg, this effect can be attributed to the concentration of lignin derivatives in the bio-oil that are mainly condensed in the second condenser, the lignin derivatives contains a relative low oxygen compared to the light organic compounds. The GC/MS analysis of the water-rich liquid collected from the second condenser (see
Table 7) revealed that acetic acid and furfural were the dominant components in the liquid, contributing up to 60% of the total organic fraction of the liquid collected in the second condenser. The remaining 40% of the liquid mainly consisted of ketone and phenol groups. In general, online fractionation was effective to concentrate the high calorific value compounds of the bio-oil in the first condenser and the water and oxygenated compounds in the second condenser. The online fractionation could be cheap downstream approach to improve the quality of the bio-oil. Operating the first condenser at higher temperatures generates a bio-oil with up to 90 wt% organics, which could be promising fraction for gasoline production via e.g., FCC, hydrotreatment. The decrease in the oxygen content and the increase in the carbon and hydrogen content of the bio-oil could help to reduce the amount of hydrogen needed in case of e.g., hydrotreatment of this bio-oil. The water-rich fraction of the pyrolysis liquid, obtained in the second condenser, contains up to 10 wt% light organics and could be an interesting raw stock for further extraction applications or for supercritical water gasification to produce hydrogen for hydro-treatment of the bio-oil [
22,
23]
4. Conclusions
The bench scale unit can be operated successfully for in situ catalytic flash pyrolysis of biomass. The unit demonstrated good operating stability and flexibility for varying operating conditions. The bio-oil recovery system provides the possibility for fractionation of different bio-oil fractions and adds to the degrees of freedom available for tailoring bio-oil properties.
Zeolite-based acidic catalysts can perform deoxygenation, at the cost of reduced organic liquid yield because of secondary cracking reaction of vapors leading to formation of gases. Nevertheless, the gas or char generated during the catalytic pyrolysis can supply a part of the heat required for the pyrolysis step in an integrated process or it can be used for biomass drying. In general, increasing the zeolite acidity favored the formation of desirable compounds such as phenols and furans and reduced the most deleterious components in the bio-oil such as acids. However some of the undesired components in the bio-oil such as ketones, and aldehydes increased with increasing zeolite acidity.
Online fractionation via staged condensation was a successful technique to produce high calorific value bio-oil with very low water content of 10 wt%. It can be concluded that fractionation is a promising cheap downstream approach to control the bio-oil properties. The integrated process of catalytic pyrolysis and in-situ fractionation can be used for the production of tailor made biofuels and/or biochemicals. The good quality bio-oil produced by integrated catalytic pyrolysis of biomass may be used as a co-feedstock for conventional refineries and existing infrastructure can be used for the production of sustainable transportation fuels.