A Critical Review of Mineral Matter Related Issues during Gasification of Coal in Fixed, Fluidized, and Entrained Flow Gasifiers
Abstract
:1. Introduction
| Classification | Fixed /Moving bed | Fluidized bed | Entrained flow | ||
|---|---|---|---|---|---|
| Ash conditions | Dry | Slagging | Dry | Agglomerating | Slagging |
| Typical Processes | Lurgi | BGL | HTW, CFB, HRL | KRW, U-Gas | Shell, GEE, Siemens, MHI, PWR |
| Coal Rank | Any | High | Low | Any | Any (dry feed) High (Slurry feed) |
| Particle Size | 5–80 mm | <80 mm | <6 mm | <6 mm | <100 µm (dry) <1000 µm (slurry) |
| Coal ash yield | No limit | <25% preferred | No limit | No limit | <25% preferred |
| Acceptability of fines | Limited | Injection of tuyeres | Good | Better | Unlimited |
| Operating Temperature | 973–1473 K | 1773–2073 K | 1173–1323 K | 1423–1533 K | >1573 K |
| Heating rate | Very Low (<50 K/s) | Low (<200 K/s) * | Extremely high (>10,000 K/s) | ||
| Average residence time | ~3600 s | >100 s | 0.5–10 s | ||
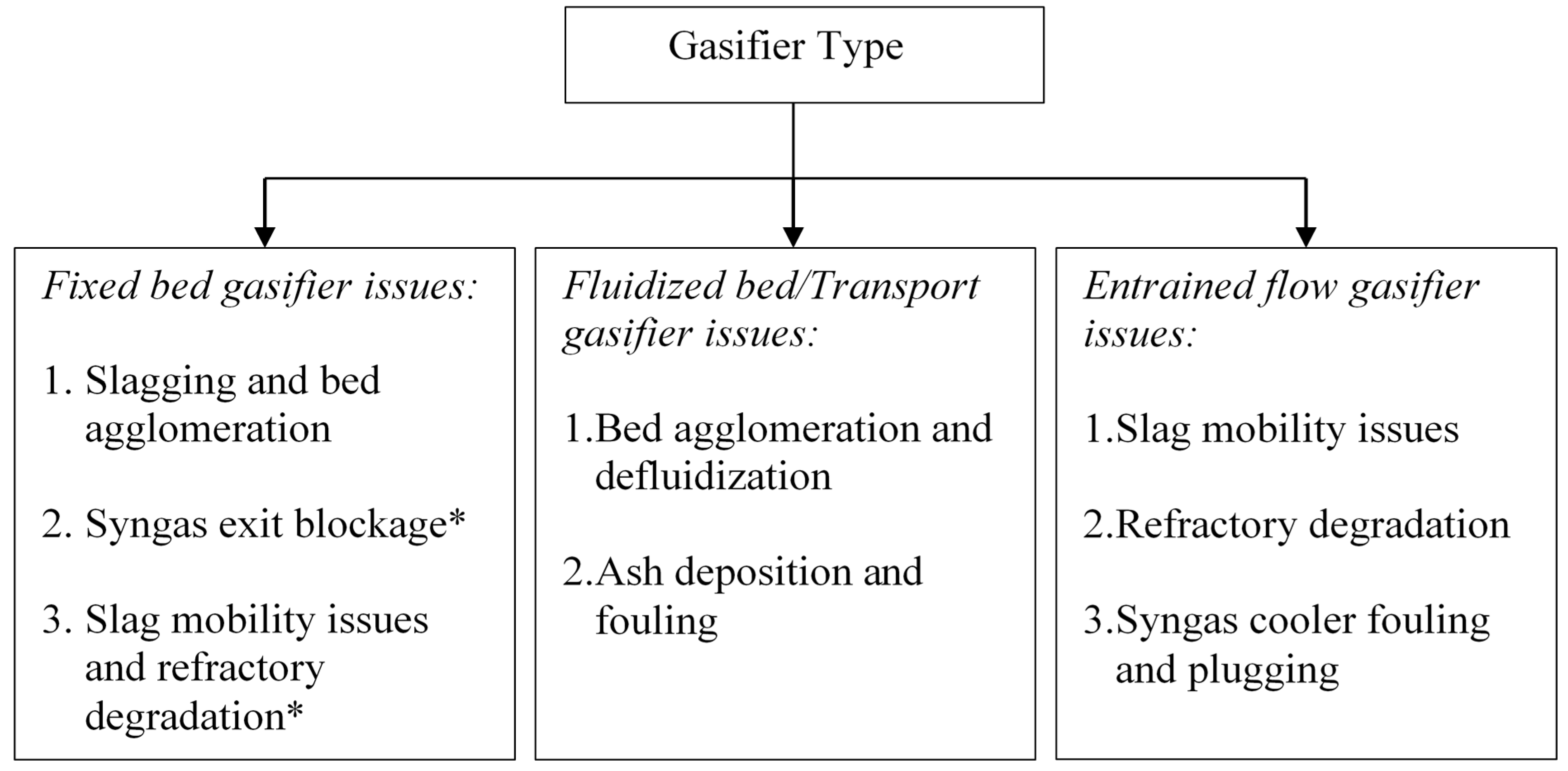
2. Fixed Bed/Moving Bed Gasifiers
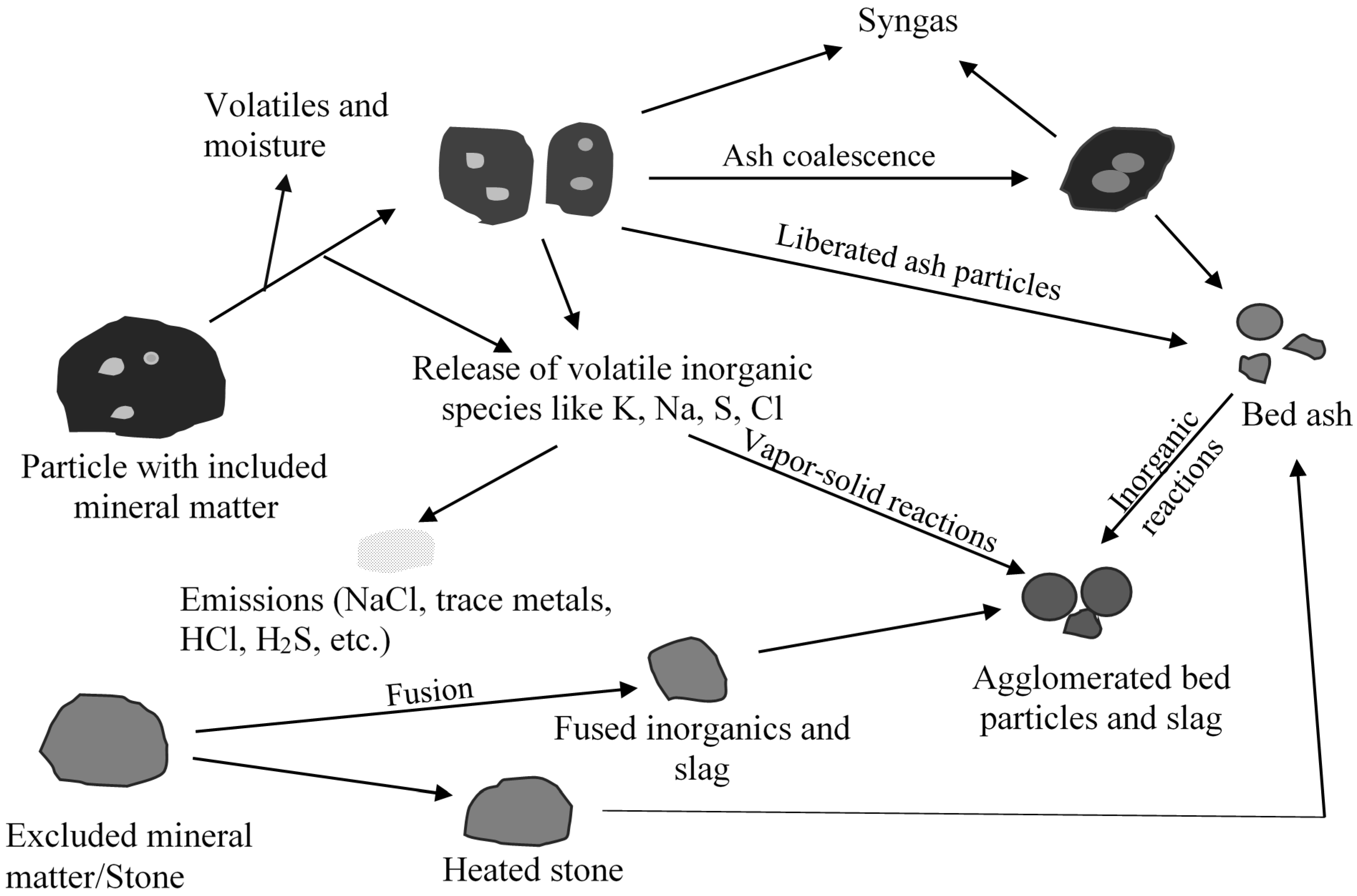
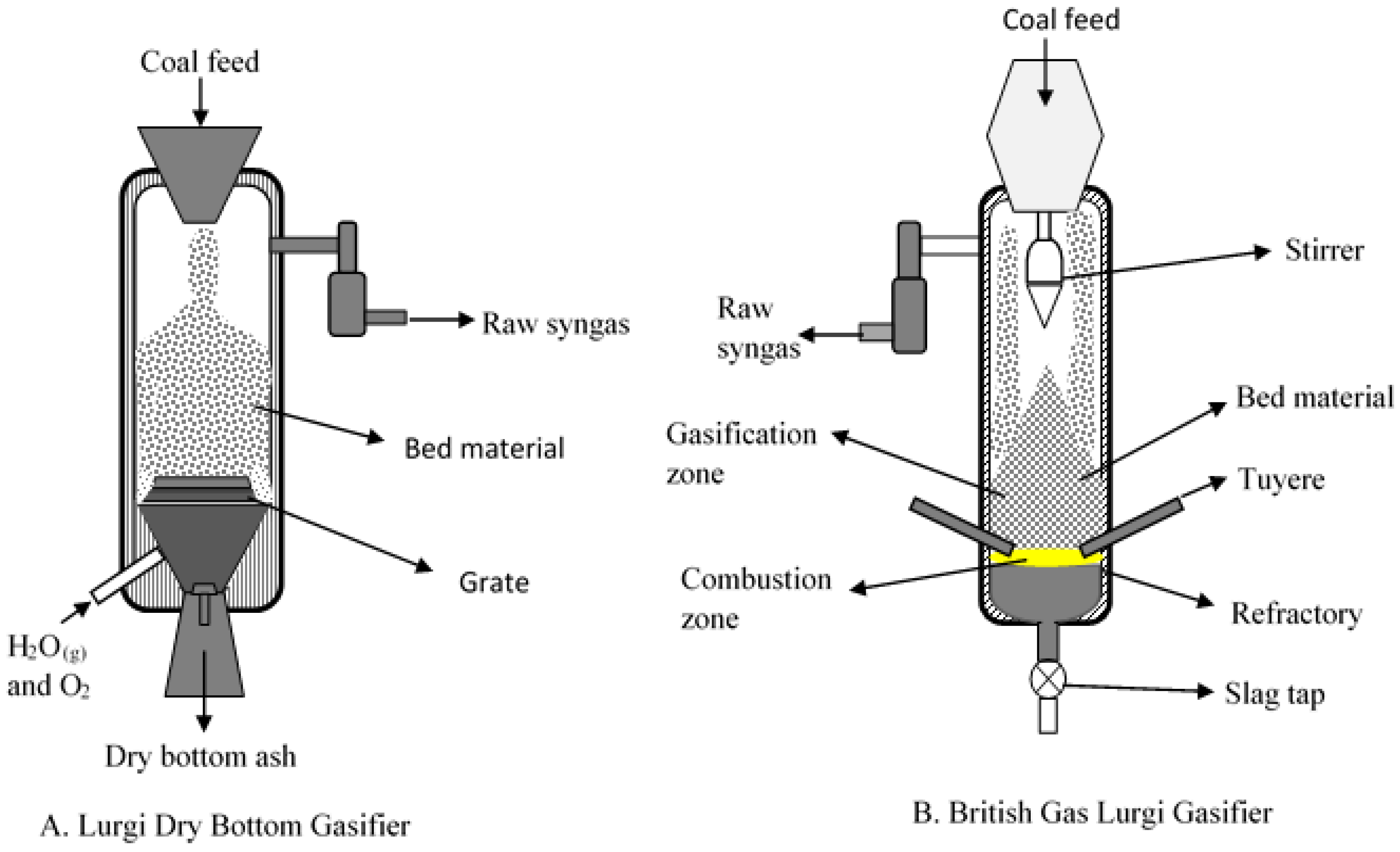
2.1. Role of Inorganic Interactions in Agglomerate and Deposit Formation
2.2. Prediction of Slag Formation
2.3. Slag Mobility in Slagging Gasifier
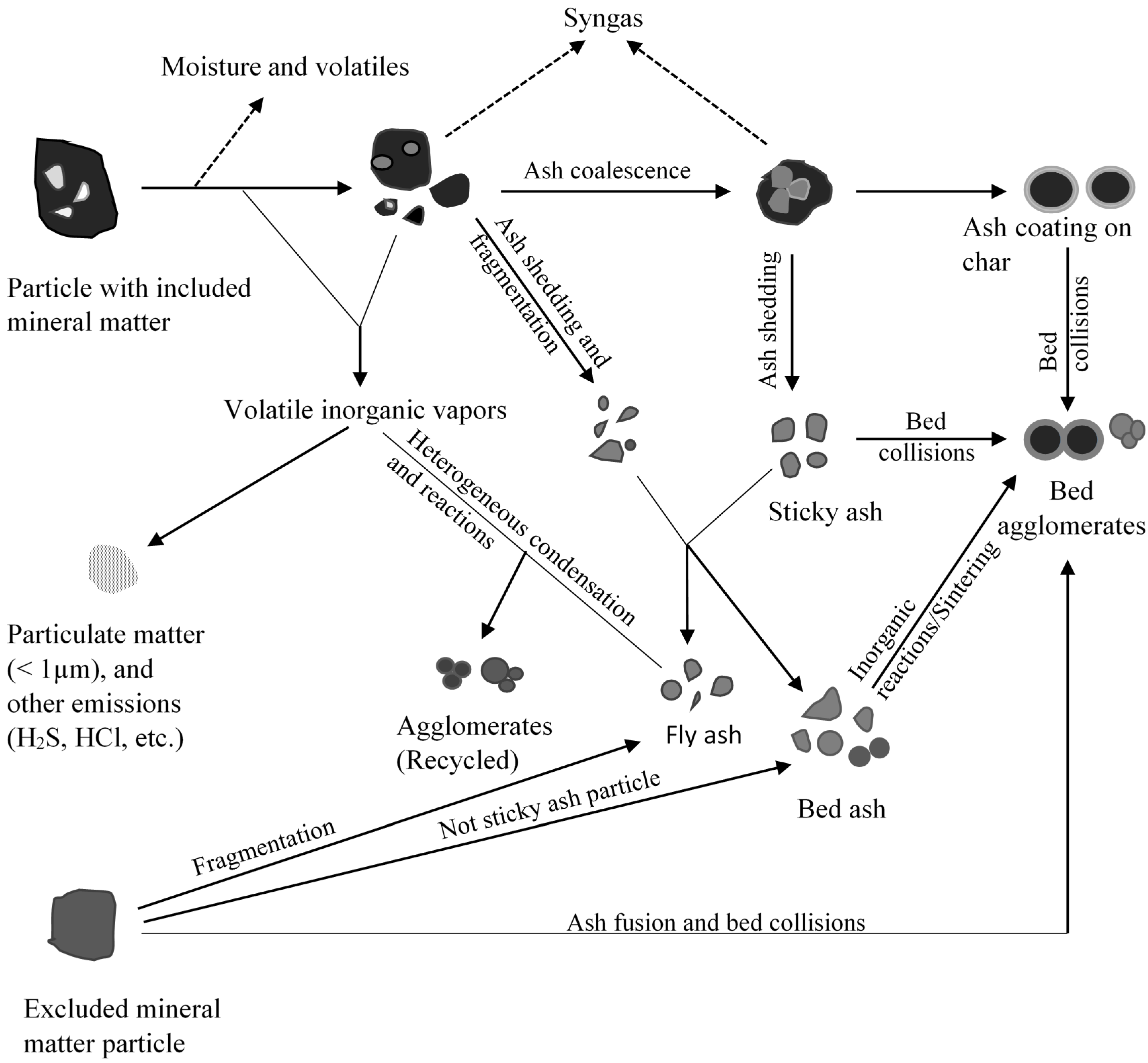
3. Fluidized Bed Gasifiers

| Factors | Effect on agglomeration and defluidization |
|---|---|
| Temperature | Increase in temperature increase the possibility of agglomeration and defluidization |
| Particle size distribution | Presence of a bimodal or multimodal particle size distribution increase the possibility of agglomeration and defluidization |
| Fluidization velocity | Increase in fluidization velocity increases the segregation force and reduce the agglomeration tendencies below ash sintering temperature |
| Alkalis, iron sulfides, and siderite | Increase the possibility of agglomeration and defluidization through formation of melt phase |
| Steam | Increase in steam can increase the agglomeration and defluidization |
3.1. Mineral Matter Transformations Leading to Agglomeration and Deposition
3.2. Strategies to Minimize Agglomeration and Ash Deposition
4. Entrained Flow Gasifiers
| Name | Wabash River | Tampa electric | Nuon | ELCOGAS |
|---|---|---|---|---|
| Power output | 262 MWe | 250 MWe | 253 MWe | 300 MWe |
| Gasifier Technology | Lummus E-Gas Technology | GE/Texaco | Shell | Shell (formerly Prenflo) |
| Gasifier type | Two stage upflow | Single stage downflow | Single stage upflow | Single stage upflow |
| Feed system | Coal-water slurry | Coal-water slurry | Dry lock hopper | Dry lock hopper |
| Operating temperature/Pressure | >1600 K (firststage)/~2.7 MPa | 1575–1750 K/~2.7 MPa | 1873 K/~2.6 MPa | 1473–1873 K/~2.5 MPa |
| Slag removal | Continuous | Lock hopper | Lock hopper | Lock hopper |
| Slag/Char fines recycle | Yes | Yes | Yes | Yes |
| Refractory design | Air cooled refractory | Air cooled refractory | Water cooled membrane wall | Water cooled membrane wall |
| Syngas cooler design | Downflow firetube | Downflow radiant water tube and convective firetube | Downflow concentric circle water tube | Upflow and downflow water tube and convective water tube |
| Particulate removal system | Candle filter at 623 K | Water scrub no filter | Candle filter at 503 K | Candle filter at 513 K |
| Chloride removal | Water scrub | Water scrub | Water Scrub | Water scrub |
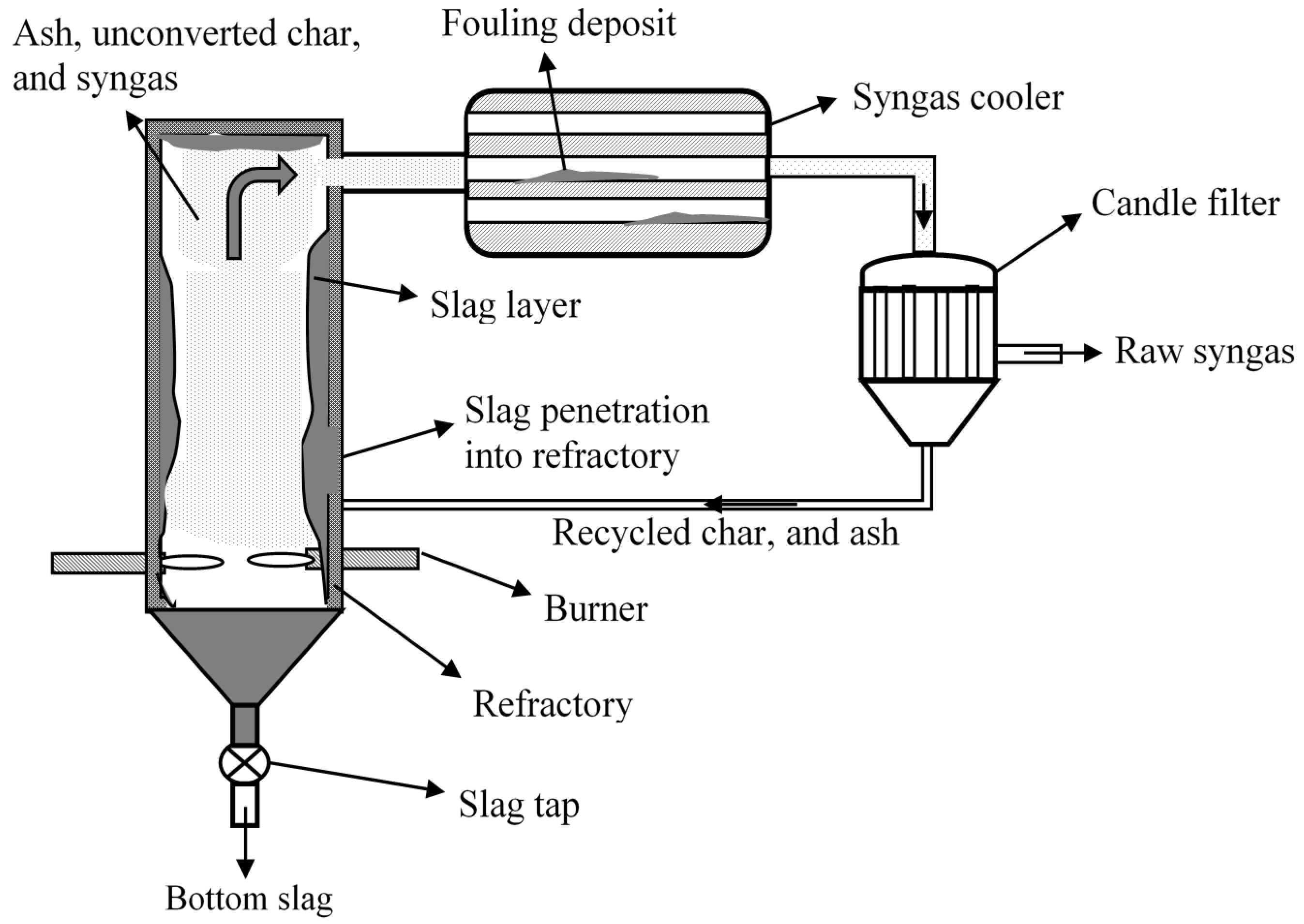
| Gasifier | Wabash River | Tampa electric | Nuon | ELCOGAS |
|---|---|---|---|---|
| E-Gas | GE | Shell | Prenflo | |
| Refractory wear | Life ~2 years | Life ~2 years (more recently 3 years) | Not significant | Not significant |
| Slag tap blockage | Yes | Yes | No | Yes |
| Corrosion and erosion in circulating slag water | Minor | Yes | Yes | Yes |
| Syngas cooler fouling and corrosion | Yes- can be cleaned with in-situ | Only with convective syngas cooler | Minor | Yes- but not significant |
| Candle filter failure | Yes | Not applicable | No | Yes |
4.1. Ash/Slag Formation
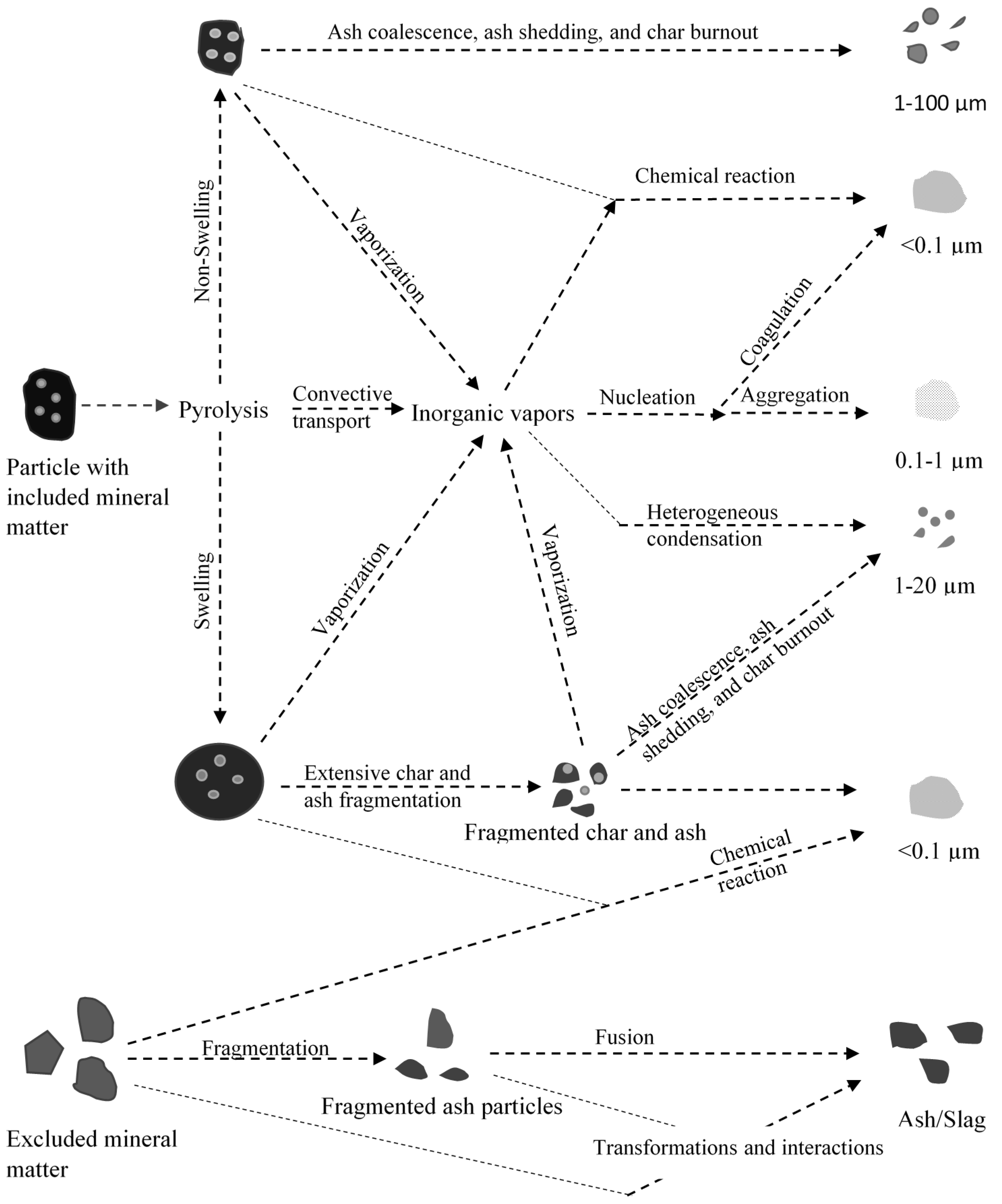
4.2. Slag Mobility
4.3. Slag-Refractory Interactions
| Chemistry (Weight %) | Brick type | ||
|---|---|---|---|
| A | B | C | |
| Cr2O3 | 89.0 | 87.3 | 81.0 |
| Al2O3 | 10.2 | 2.5 | 0.4 |
| ZrO2 | - | 5.2 | Not Reported |
| MgO | 17.0 | ||
| Bulk density (g/cm3) | 4.21 | 4.07 | 3.95 |
| Porosity (vol. %) | 16.7 | 16.5 | 12.0 |
4.4. Fouling
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Harrison, R.M.; Stedman, J.; Derwent, D. New directions: Why are PM 10 concentrations in Europe not falling? Atmos. Environ. 2008, 42, 603–606. [Google Scholar] [CrossRef]
- Pope, I.C.; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. J. Am. Med. Assoc. 2002, 287, 1132–1141. [Google Scholar] [CrossRef]
- Beer, J.M. Combustion technology developments in power generation in response to environmental challenges. Prog. Energy Combust. Sci. 2000, 26, 301–327. [Google Scholar] [CrossRef]
- Franco, A.; Diaz, A.R. The future challenges for “clean coal technologies”: Joining efficiency increase and pollutant emission control. Energy 2009, 34, 348–354. [Google Scholar] [CrossRef]
- Raask, E. Mineral Impurities in Coal Combustion: Behavior, Problems, and Remedial Measures; Hemisphere Pub. Corp.: Washington, DC, USA, 1985. [Google Scholar]
- Zhang, D.K.; Jackson, P.J.; Vuthaluru, H.B. Low-rank coal and advanced technologies for power generation. In Impact of Mineral Impurities in Solid Fuel Combustion; Gupta, R.P., Wall, T.F., Baxter, L., Eds.; Kluwer Academic Publishers: New York, NY, USA, 1999; pp. 45–64. [Google Scholar]
- Higman, C.; van der Burgt, M. Gasification, 2nd ed.; Gulf Professional Publishing: Burlington, VT, USA, 2008. [Google Scholar]
- Speight, J.G. The Chemistry and Technology of Coal; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Wagner, N.J.; Coertzen, M.; Matjie, R.H.; van Dyk, J.C.; John, C.C. Coal gasification. In Applied Coal Petrology; Isabel, S.R., Crelling, J.C., Eds.; Elsevier: Burlington, VT, USA, 2008; pp. 119–144. [Google Scholar]
- Bartels, M.; Lin, W.; Nijenhuis, J.; Kapteijn, F.; van Ommen, J.R. Agglomeration in fluidized beds at high temperatures: Mechanisms, detection and prevention. Prog. Energy Combust. Sci. 2008, 34, 633–666. [Google Scholar] [CrossRef]
- Bryers, R.W. Fireside slagging, fouling, and high-temperature corrosion of heat-transfer surface due to impurities in steam-raising fuels. Prog. Energy Combust. Sci. 1996, 22, 29–120. [Google Scholar] [CrossRef]
- Khadilkar, A.; Rozelle, P.L.; Pisupati, S.V. Models of agglomerate growth in fluidized bed reactors: Critical review, status and applications. Powder Technol. 2014, 264, 216–228. [Google Scholar] [CrossRef]
- Wall, T.F.; Liu, G.-S.; Wu, H.-W.; Roberts, D.G.; Benfell, K.E.; Gupta, S.; Lucas, J.A.; Harris, D.J. The effects of pressure on coal reactions during pulverised coal combustion and gasification. Prog. Energy Combust. Sci. 2002, 28, 405–433. [Google Scholar] [CrossRef]
- Collot, A.G. Matching gasification technologies to coal properties. Int. J. Coal Geol. 2006, 65, 191–212. [Google Scholar] [CrossRef]
- Stubington, J.F.; Sasongko, D. On the heating rate and volatile yield for coal particles injected into fluidised bed combustors. Fuel 1998, 77, 1021–1025. [Google Scholar] [CrossRef]
- Casleton, K.H.; Breault, R.W.; Richards, G.A. System issues and tradeoffs associated with syngas production and combustion. Combust. Sci. Technol. 2008, 180, 1013–1052. [Google Scholar] [CrossRef]
- Soundarrajan, N.; Krishnamurthy, N.; Pisupati, S.V. Characterization of size and density separated fractions of a bituminous coal as a feedstock for entrained slagging gasification. Int. J. Clean Coal Energy 2013, 2, 58–67. [Google Scholar] [CrossRef]
- Van Dyk, J.C.; Waanders, F.B. Manipulation of gasification coal feed in order to increase the ash fusion temperature of the coal enabling the gasifiers to operate at higher temperatures. Fuel 2007, 86, 2728–2735. [Google Scholar] [CrossRef]
- van Dyk, J.C.; Melzer, S.; Sobiecki, A. Mineral matter transformation during sasol-lurgi fixed bed dry bottom gasification—Utilization of HT-XRD and FACTSAGE modelling. Miner. Eng. 2006, 19, 1126–1135. [Google Scholar] [CrossRef]
- Guhl, S. Thermodynamic modelling of the behaviour of mineral matter in the BGL-gasification process. In Proceedings of the GTT (Gesellschaft für Technische Thermochemie und -physik mbH)-Workshop, Herzogenrath, Germany, 11–13 July 2012.
- Sukul, G.; Balaramakrishna, P.V. A review of refractory linings for gasification reactors. J. Aust. Ceram. Soc. 2014, 50, 83–92. [Google Scholar]
- Lee, W.E.; Zhang, S. Melt corrosion of oxide and oxide-carbon refractories. Int. Mater. Rev. 1999, 44, 77–104. [Google Scholar] [CrossRef]
- Skhonde, M.P.; Matjie, R.H.; Bunt, J.R.; Strydom, A.C.; Schobert, H. Sulfur behavior in the sasol-lurgi fixed-bed dry-bottom gasification process. Energy Fuels 2009, 23, 229–235. [Google Scholar] [CrossRef]
- Holt, N. Gasification Technology Status–Technical Update; EPRI Report 1012224; 2006. [Google Scholar]
- Kosminski, A.; Ross, D.P.; Agnew, J.B. Reactions between sodium and silica during gasification of a low-rank coal. Fuel Process. Technol. 2006, 87, 1037–1049. [Google Scholar] [CrossRef]
- Kosminski, A.; Ross, D.P.; Agnew, J.B. Reactions between sodium and kaolin during gasification of a low-rank coal. Fuel Process. Technol. 2006, 87, 1051–1062. [Google Scholar] [CrossRef]
- Hlavac, J. The Technology of Glass and Ceramics; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1983. [Google Scholar]
- Waanders, F.B. The iron mineral changes occurring in lignite coal during gasification. Hyperfine Interact. 2013, 218, 35–40. [Google Scholar] [CrossRef]
- Bryers, R. Factors critically affecting fireside deposits in steam generators. In Impact of Mineral Impurities in Solid Fuel Combustion; Gupta, R.P., Wall, T.F., Baxter, L., Eds.; Kluwer Academic Publishers: New York, USA, 1999; pp. 105–131. [Google Scholar]
- Matjie, R.H.; van Alphen, C.; Pistorius, P.C. Mineralogical characterisation of Secunda gasifier feedstock and coarse ash. Miner. Eng. 2006, 19, 256–261. [Google Scholar] [CrossRef]
- Matjie, R.H.; Li, Z.; Ward, C.R.; French, D. Chemical composition of glass and crystalline phases in coarse coal gasification ash. Fuel 2008, 87, 857–869. [Google Scholar] [CrossRef]
- Matjie, R.H.; Ward, C.R.; Li, Z. Mineralogical transformations in coal feedstocks during carbon conversion, based on packed-bed combustor tests: Part 1. Bulk coal and ash studies. Coal Combust. Gasif. Prod. 2012, 4, 45–54. [Google Scholar]
- Hlatshwayo, T.B.; Matjie, R.H.; Li, Z.; Ward, C.R. Mineralogical characterization of sasol feed coals and corresponding gasification ash constituents. Energy Fuels 2009, 23, 2867–2873. [Google Scholar] [CrossRef]
- ASTM Standard. Standard Test Method for Fusibility of Coal and Coke Ash, Astm International; ASTM Standard: West Conshohocken, PA, USA, 2003; Available online: http://www.astm.org (accessed on 15 June 2014). [CrossRef]
- Huggins, F.E.; Kosmack, D.A.; Huffman, G.P. Correlation between ash-fusion temperatures and ternary equilibrium phase diagrams. Fuel 1981, 60, 577–584. [Google Scholar] [CrossRef]
- Kahraman, H.; Bos, F.; Reifenstein, A.; Coin, C.D.A. Application of a new ash fusion test to theodore coals. Fuel 1998, 77, 1005–1011. [Google Scholar] [CrossRef]
- van Dyk, J.C.; Keyser, M.J. Influence of discard mineral matter on slag-liquid formation and ash melting properties of coal—A FACTSAGEtm simulation study. Fuel 2014, 116, 834–840. [Google Scholar] [CrossRef]
- Slegeir, W.A.; Singletary, J.H.; Kohut, J.F. Application of a microcomputer to the determination of coal ash fusibility characteristics. J. Coal Qual. 1988, 7, 48–54. [Google Scholar]
- van Dyk, J.C.; Waanders, F.B.; Benson, S.A.; Laumb, M.L.; Hack, K. Viscosity predictions of the slag composition of gasified coal, utilizing FACTSAGE equilibrium modelling. Fuel 2009, 88, 67–74. [Google Scholar] [CrossRef]
- Schobert, H.H. Lignites of North America; Elsevier: Amsterdam, The Netherlands, 1995; Volume 23. [Google Scholar]
- Browning, G.J.; Bryant, G.W.; Lucas, J.A.; Wall, T.F. The effects of heterogeneous slag character on viscosity and slag flow in IGCC gasifiers. IFRF Combust. J. 1999, 199901, 1–10. [Google Scholar]
- Gronhovd, G.H.; Harak, A.E.; Fegley, M.M. Some results from slagging, fixed-bed gasification of lignite at pressures to 400 Psig. In Preprints of Papers-American Chemical Society, Division of Fuel Chemistry; American Chemical Society: Philadelphia, PA, USA, 1964. [Google Scholar]
- Schobert, H.H.; Streeter, R.C.; Diehl, E.K. Flow properties of low-rank coal ash slags: Implications for slagging gasification. Fuel 1985, 64, 1611–1617. [Google Scholar] [CrossRef]
- Patterson, J.H.; Hurst, H.J. Ash and slag qualities of Australian bituminous coals for use in slagging gasifiers. Fuel 2000, 79, 1671–1678. [Google Scholar] [CrossRef]
- Basu, P. Combustion and Gasification in Fluidized Beds; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Ariyapadi, S.; Shires, P.; Bhargava, M.; Ebbern, D. KBR’s transport gasifier (TRIG)—An advanced gasification technology for SNG production from low-rank coals. In Proceedings of the 25th Annual International Pittsburgh Coal Conference, Pittsburgh, PA, USA, 29 September–2 October 2008.
- Dahlin, R.S.; Peng, W.; Nelson, M.; Vimalchand, P.; Liu, G. Formation and prevention of agglomerated deposits during the gasification of high-sodium lignite. Energy Fuels 2006, 20, 2465–2470. [Google Scholar] [CrossRef]
- Kosminski, A.; Manzoori, A.R. Inorganic Matter Behavior in the Gasification of Southern Australian Coals for Combined Cycle Power Generation Technical Report; Technical Services Department, Electricity Trust of Southern Australia: Adelaide, Australia, 1990. [Google Scholar]
- Lin, C.L.; Peng, T.H.; Wang, W.J. Effect of particle size distribution on agglomeration/defluidization during fluidized bed combustion. Powder Technol. 2011, 207, 290–295. [Google Scholar] [CrossRef]
- Manzoori, A.R.; Agarwal, P.K. The role of inorganic matter in coal in the formation of agglomerates in circulating fluid-bed combustors. Fuel 1993, 72, 1069–1075. [Google Scholar] [CrossRef]
- Vuthaluru, H.B.; Zhang, D.-K.; Linjewile, T.M. Behaviour of inorganic constituents and ash characteristics during fluidised-bed combustion of several Australian low-rank coals. Fuel Process. Technol. 2000, 67, 165–176. [Google Scholar] [CrossRef]
- Kuo, J.-H.; Lin, C.-L.; Wey, M.-Y. Effect of alkali concentrations and operating conditions on agglomeration/defluidization behavior during fluidized bed air gasification. Powder Technol. 2011, 214, 443–446. [Google Scholar] [CrossRef]
- Schafer, H.N.S. Pyrolysis of brown coals. 2. Decomposition of acidic groups on heating in the range 100–900 °C. Fuel 1979, 58, 673–679. [Google Scholar] [CrossRef]
- Murray, J.B. Changes in state of combination of inorganic constituents during carbonization of Victorian brown coal. Fuel 1973, 52, 105–111. [Google Scholar] [CrossRef]
- Kosminski, A.; Ross, D.P.; Agnew, J.B. Transformations of sodium during gasification of low-rank coal. Fuel Process. Technol. 2006, 87, 943–952. [Google Scholar] [CrossRef]
- van Eyk, P.J.; Ashman, P.J.; Alwahabi, Z.T.; Nathan, G.J. The release of water-bound and organic sodium from Loy Yang coal during the combustion of single particles in a flat flame. Combust. Flame 2011, 158, 1181–1192. [Google Scholar] [CrossRef]
- Li, C.-Z. Some recent advances in the understanding of the pyrolysis and gasification behaviour of Victorian brown coal. Fuel 2007, 86, 1664–1683. [Google Scholar] [CrossRef]
- Marinov, V.; Marinov, S.P.; Lazarov, L.; Stefanova, M. Ash agglomeration during fluidized bed gasification of high sulphur content lignites. Fuel Process. Technol. 1992, 31, 181–191. [Google Scholar] [CrossRef]
- Williamson, J.; West, S.S.; Laughlin, M.K. The behaviour of bed material during fluidized bed gasification: The effects of mineral matter interactions. Fuel 1994, 73, 1039–1045. [Google Scholar] [CrossRef]
- Mason, D.M.; Patel, J.G. Chemistry of ash agglomeration in the U-Gas® process. Fuel Process. Technol. 1980, 3, 181–206. [Google Scholar] [CrossRef]
- Khadilkar, A.B.; Rozelle, P.L.; Pisupati, S.V. A study on initiation of ash agglomeration in fluidized bed gasification systems. Fuel 2015, 152, 48–57. [Google Scholar] [CrossRef]
- Anthony, E.J.; Iribarne, A.P.; Iribarne, J.V.; Talbot, R.; Jia, L.; Granatstein, D.L. Fouling in a 160 MWe FBC boiler firing coal and petroleum coke. Fuel 2001, 80, 1009–1014. [Google Scholar] [CrossRef]
- Kreutzkam, B.; Wieland, C.; Spliethoff, H. Improved numerical prediction of ash formation and deposition using a novel developed char fragmentation model. Fuel 2012, 98, 103–110. [Google Scholar] [CrossRef]
- Vuthaluru, H.B.; Zhang, D.-K. Control methods for remediation of ash-related problems in fluidised-bed combustors. Fuel Process. Technol. 1999, 60, 145–156. [Google Scholar] [CrossRef]
- Butler, C.J.; Green, A.M.; Chaffee, A.L. The fate of trace elements during MTE and HTD dewatering of latrobe valley brown coals. Coal Prep. 2007, 27, 210–229. [Google Scholar] [CrossRef]
- Artanto, Y.; Chaffee, A.L. Dewatering low rank coals by mechanical thermal expression (MTE) and its influence on organic carbon and inorganic removal. Coal Prep. 2005, 25, 251–267. [Google Scholar] [CrossRef]
- Vuthaluru, H.B.; Zhang, D.K. Effect of coal blending on particle agglomeration and defluidisation during spouted-bed combustion of low-rank coals. Fuel Process. Technol. 2001, 70, 41–51. [Google Scholar] [CrossRef]
- Tillman, D.A.; Duong, D.N.B.; Harding, N.S. Chapter 3—Blending coal on coal. In Solid Fuel Blending; Butterworth-Heinemann: Boston, MA, USA, 2012; pp. 71–123. [Google Scholar]
- Kyi, S.; Chadwick, B.L. Screening of potential mineral additives for use as fouling preventatives in Victorian brown coal combustion. Fuel 1999, 78, 845–855. [Google Scholar] [CrossRef]
- Vuthaluru, H.B.; Zhang, D.K. Effect of Ca- and Mg-bearing minerals on particle agglomeration defluidisation during fluidised-bed combustion of a south Australian lignite. Fuel Process. Technol. 2001, 69, 13–27. [Google Scholar] [CrossRef]
- McCullough, D.P.; van Eyk, P.J.; Ashman, P.J.; Mullinger, P.J. Investigation of agglomeration and defluidization during spouted-bed gasification of high-sodium, high-sulfur south australian lignite. Energy Fuels 2011, 25, 2772–2781. [Google Scholar] [CrossRef]
- Wabash River Coal Gasification Repowering Project: A DOE Assessment; U.S. Department of Energy: Washington, DC, USA, 2002.
- U.S. Department of Energy. Gasifier Database. Available online: http://www.netl.doe.gov/research/Coal/energy-systems/gasification /gasifipedia/ (accessed on 7 July 2015).
- Tampa Electric Polk Power Station Integrated Gasification Combined Cycle Project: Final Technical Report; U.S. Department of Energy: Washington, DC, USA, 2002.
- Shannon, G.N.; Rozelle, P.L.; Pisupati, Sarma V.; Sridhar, S. Conditions for entrainment into a FeOx containing slag for a carbon-containing particle in an entrained coal gasifier. Fuel Process. Technol. 2008, 89, 1379–1385. [Google Scholar] [CrossRef]
- Wabash River Coal Gasification Repowering Project: Final Technical Report; U.S. Department of Energy: Washington, DC, USA, 2000.
- Mondal, P.; Dang, G.S.; Garg, M.O. Syngas production through gasification and cleanup for downstream applications—Recent developments. Fuel Process. Technol. 2011, 92, 1395–1410. [Google Scholar] [CrossRef]
- Wang, P.; Massoudi, M. Slag behavior in gasifiers. Part I: Influence of coal properties and gasification conditions. Energies 2013, 6, 784–806. [Google Scholar]
- Baxter, L.L.; DeSollar, R.W. A mechanistic description of ash deposition during pulverized coal combustion: Predictions compared with observations. Fuel 1993, 72, 1411–1418. [Google Scholar] [CrossRef]
- Wall, T.F. Mineral matter transformations and ash deposition in pulverised coal combustion. Symp. (Int.) Combust. 1992, 24, 1119–1126. [Google Scholar] [CrossRef]
- Mueller, C.; Selenius, M.; Theis, M.; Skrifvars, B.-J.; Backman, R.; Hupa, M.; Tran, H. Deposition behaviour of molten alkali-rich fly ashes—Development of a submodel for CFD applications. Proc. Combust. Inst. 2005, 30, 2991–2998. [Google Scholar] [CrossRef]
- Li, S.; Wu, Y.; Whitty, K.J. Ash deposition behavior during char—Slag transition under simulated gasification conditions. Energy Fuels 2010, 24, 1868–1876. [Google Scholar] [CrossRef]
- Koyama, S.; Morimoto, T.; Ueda, A.; Matsuoka, H. A microscopic study of ash deposits in a two-stage entrained-bed coal gasifier. Fuel 1996, 75, 459–465. [Google Scholar] [CrossRef]
- Khanna, S.K.; Wu, C.; Sahajwalla, V. Estimation of critical carbon particle engulfment velocity in metallurgical melts. High Temp. Mater. Process. 1998, 17, 193–202. [Google Scholar] [CrossRef]
- Wu, T.; Gong, M.; Lester, E.; Wang, F.; Zhou, Z.; Yu, Z. Characterisation of residual carbon from entrained-bed coal water slurry gasifiers. Fuel 2007, 86, 972–982. [Google Scholar] [CrossRef]
- Shannon, G.N.; Matsuura, H.; Rozelle, P.; Fruehan, R.J.; Pisupati, S.; Sridhar, S. Effect of size and density on the thermodynamic predictions of coal particle phase formation during coal gasification. Fuel Process. Technol. 2009, 90, 1114–1121. [Google Scholar] [CrossRef]
- Wu, H.; Bryant, G.; Wall, T. The effect of pressure on ash formation during pulverized coal combustion. Energy Fuels 2000, 14, 745–750. [Google Scholar] [CrossRef]
- Huang, L.Y.; Norman, J.S.; Pourkashanian, M.; Williams, A. Prediction of ash deposition on superheater tubes from pulverized coal combustion. Fuel 1996, 75, 271–279. [Google Scholar] [CrossRef]
- Bennett, J.; Kwong, K.-S. Failure mechanisms in high chrome oxide gasifier refractories. Metall. Mater. Trans. A 2011, 42, 888–904. [Google Scholar] [CrossRef]
- Trapp, B.; Moock, N.; Denton, D. Coal gasification: Ready for prime time. Available online: http://www.clean-energy.us/projects/eastman_power_magazine.htm (accessed on 30 December 2014).
- Zhu, J.; Kaneko, T.K.; Mu, H.; Bennett, J.P.; Sridhar, S. Effects of measurement materials and oxygen partial pressure on the viscosity of synthetic eastern and western united states coal slags. Energy Fuels 2012, 26, 4465–4474. [Google Scholar] [CrossRef]
- Kinaev, N. A Review of Mineral Matter Issues in Coal Gasification; Cooperative Research Center for Coal in Sustainable Development: Pullenvale, QLD, Australia, 2006. [Google Scholar]
- Bockelie, M.; Denison, M.; Chen, Z.; Linjewile, T.; Senior, C.; Sarofim, A.; Holt, N. CFD Modelling of Entrained Flow Gasifiers. In Proceedings the Gasification Technologies Conference, San Francisco, CA, USA, 27–30 October 2002.
- Oh, M.S.; Brooker, D.D.; de Paz, E.F.; Brady, J.J.; Decker, T.R. Effect of crystalline phase formation on coal slag viscosity. Fuel Process. Technol. 1995, 44, 191–199. [Google Scholar] [CrossRef]
- Sridhar, S.; Rozzelle, P.; Morreale, B.; Alman, D. Materials challenges for advanced combustion and gasification fossil energy systems. Metall. Mater. Trans. A 2011, 42, 871–877. [Google Scholar] [CrossRef]
- Yuan, H.; Liang, Q.; Gong, X. Crystallization of coal ash slags at high temperatures and effects on the viscosity. Energy Fuels 2012, 26, 3717–3722. [Google Scholar] [CrossRef]
- Kondratiev, A.; Jak, E. Predicting coal ash slag flow characteristics (viscosity model for the Al2O3-CaO-FeO-SiO2 system). Fuel 2001, 80, 1989–2000. [Google Scholar] [CrossRef]
- Song, W.; Sun, Y.; Wu, Y.; Zhu, Z.; Koyama, S. Measurement and simulation of flow properties of coal ash slag in coal gasification. AIChE J. 2011, 57, 801–818. [Google Scholar] [CrossRef]
- Song, W.; Tang, L.; Zhu, X.; Wu, Y.; Rong, Y.; Zhu, Z.; Koyama, S. Fusibility and flow properties of coal ash and slag. Fuel 2009, 88, 297–304. [Google Scholar] [CrossRef]
- Li, X.; Li, G.; Cao, Z.; Xu, S. Research on flow characteristics of slag film in a slag tapping gasifier. Energy Fuels 2010, 24, 5109–5115. [Google Scholar] [CrossRef]
- Kong, L.; Bai, J.; Bai, Z.; Guo, Z.; Li, W. Effects of CaCO3 on slag flow properties at high temperatures. Fuel 2013, 109, 76–85. [Google Scholar] [CrossRef]
- Yu, G.; Zhu, Q.; Chi, G.; Guo, Q.; Zhou, Z. Study on slag composition and flow property in a bench-scale OMB gasifier. Fuel Process. Technol. 2012, 104, 136–143. [Google Scholar] [CrossRef]
- Kwong, K.; Petty, A.; Bennett, J.; Krabbe, R.; Thomas, H. Wear mechanisms of chromia refractories in slagging gasifiers. Int. J. Appl. Ceram. Technol. 2007, 4, 503–513. [Google Scholar] [CrossRef]
- Bennett, J.P. Refractory liner materials used in slagging gasifiers. Refractories Appl. News 2004, 9, 20–24. [Google Scholar]
- Dogan, C.P.; Kwong, K.S.; Benett, J.P.; Chinn, R.E.; Dahlin, C.L. New develpments in gasifier refractories. In Proceedings of the Gasification Technologies Conference, San Francisco, CA, USA, 27–30 October 2002.
- Keeler, C.G. Operating experience at the Wabash river repowering project. In Proceedings of the Gasification Technologies Conference, San Francisco, CA, USA, 27–30 October 2003.
- Kwong, K.; Bennett, J.; Nakano, J. Gasifier refractories, coal slags, and their interaction. In Proceedings of the 9th International Conference on Molten Slags, Fluxes and Salts, Beijing, China, 27–30 May 2012.
- Strobel, T.M.; Hurley, J.P. Coal-ash corrosion of monolithic silicon carbide-based refractories. Fuel Process. Technol. 1995, 44, 201–211. [Google Scholar] [CrossRef]
- Kim, H.B.; Oh, M.S. Changes in microstructure of a high chromia refractory due to interaction with infiltrating coal slag in a slagging gasifier environment. Ceram. Int. 2008, 34, 2107–2116. [Google Scholar] [CrossRef]
- Guo, Z.-Q.; Han, B.-Q.; Dong, H. Effect of coal slag on the wear rate and microstructure of the ZrO2-bearing chromia refractories. Ceram. Int. 1997, 23, 489–496. [Google Scholar] [CrossRef]
- Nakano, J.; Sridhar, S.; Bennett, J.; Kwong, K.-S.; Moss, T. Interactions of refractory materials with molten gasifier slags. Int. J. Hydrog. Energy 2011, 36, 4595–4604. [Google Scholar] [CrossRef]
- Atwell, M. GE/Bechtel alliance IGCC reference plant update. In Proceedings of the Gasification Technologies Conference, San Francisco, CA, USA, 14–17 October 2007.
- Brooker, D. Chemistry of deposit formation in a coal gasification syngas cooler. Fuel 1993, 72, 665–670. [Google Scholar] [CrossRef]
- Bakker, W. High temperature corrosion in gasifiers. Mater. Res. 2004, 7, 53–59. [Google Scholar] [CrossRef]
- Brooker, D.; Oh, M.S. Iron sulfide deposition during coal gasification. Fuel Process. Technol. 1995, 44, 181–190. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Vassileva, C.G.; Baxter, D.; Andersen, L.K. A new approach for the combined chemical and mineral classification of the inorganic matter in coal. 2. Potential applications of the classification systems. Fuel 2009, 88, 246–254. [Google Scholar] [CrossRef]
- Ai, W.; Kuhlman, J.M. Simulation of coal ash particle deposition experiments. Energy Fuels 2011, 25, 708–718. [Google Scholar] [CrossRef]
- Slezak, A.; Kuhlman, J.M.; Shadle, L.J.; Spenik, J.; Shi, S. CFD simulation of entrained-flow coal gasification: Coal particle density/sizefraction effects. Powder Technol. 2010, 203, 98–108. [Google Scholar] [CrossRef]
- Lee, H.-H.; Lee, J.-C.; Joo, Y.-J.; Oh, M.; Lee, C.-H. Dynamic modeling of shell entrained flow gasifier in an integrated gasification combined cycle process. Appl. Energy 2014, 131, 425–440. [Google Scholar] [CrossRef]
- Casella, F.; Colonna, P. Dynamic modeling of IGCC power plants. Appl. Therm. Eng. 2012, 35, 91–111. [Google Scholar] [CrossRef]
- Wieland, C.; Kreutzkam, B.; Balan, G.; Spliethoff, H. Evaluation, comparison and validation of deposition criteria for numerical simulation of slagging. Appl. Energy 2012, 93, 184–192. [Google Scholar] [CrossRef]
- Chen, L.; Yong, S.Z.; Ghoniem, A.F. Modeling the slag behavior in three dimensional CFD simulation of a vertically-oriented oxy-coal combustor. Fuel Process. Technol. 2013, 112, 106–117. [Google Scholar] [CrossRef]
- Gibson, L.; Soundarrajan, N.; Spenik, J.; Ma, J.; Shadle, L.; Pisupati, S.V. Application of particle population model to determine the contribution to slag, flyash, and syngas in entrained flow gasification from particle size distribution. Energy Fuels 2013, 27, 7681–7695. [Google Scholar] [CrossRef]
- Gibson, L.M.; Gopalan, B.; Pisupati, S.V.; Shadle, L.J. Image analysis measurements of particle coefficient of restitution for coal gasification applications. Powder Technol. 2013, 247, 30–43. [Google Scholar] [CrossRef]
- Gibson, L.M.; Shadle, L.J.; Pisupati, S.V. Determination of sticking probability based on the critical velocity derived from a visco-elastoplastic model to characterize ash deposition in an entrained flow gasifier. Energy Fuels 2004, 28, 5307–5317. [Google Scholar] [CrossRef]
- Garcia Pena, F. Operating experience and current status of Puertollano IGCC power plant. In Proceedings of the 1st International Freiberg Conference on IGCC & XtL Technologies, Freiberg, Germany, 15–18 June 2005.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krishnamoorthy, V.; Pisupati, S.V. A Critical Review of Mineral Matter Related Issues during Gasification of Coal in Fixed, Fluidized, and Entrained Flow Gasifiers. Energies 2015, 8, 10430-10463. https://doi.org/10.3390/en80910430
Krishnamoorthy V, Pisupati SV. A Critical Review of Mineral Matter Related Issues during Gasification of Coal in Fixed, Fluidized, and Entrained Flow Gasifiers. Energies. 2015; 8(9):10430-10463. https://doi.org/10.3390/en80910430
Chicago/Turabian StyleKrishnamoorthy, Vijayaragavan, and Sarma V. Pisupati. 2015. "A Critical Review of Mineral Matter Related Issues during Gasification of Coal in Fixed, Fluidized, and Entrained Flow Gasifiers" Energies 8, no. 9: 10430-10463. https://doi.org/10.3390/en80910430
APA StyleKrishnamoorthy, V., & Pisupati, S. V. (2015). A Critical Review of Mineral Matter Related Issues during Gasification of Coal in Fixed, Fluidized, and Entrained Flow Gasifiers. Energies, 8(9), 10430-10463. https://doi.org/10.3390/en80910430






