Seasonal Variation of Lipids and Fatty Acids of the Microalgae Nannochloropsis oculata Grown in Outdoor Large-Scale Photobioreactors
Abstract
:1. Introduction
2. Results and Discussion
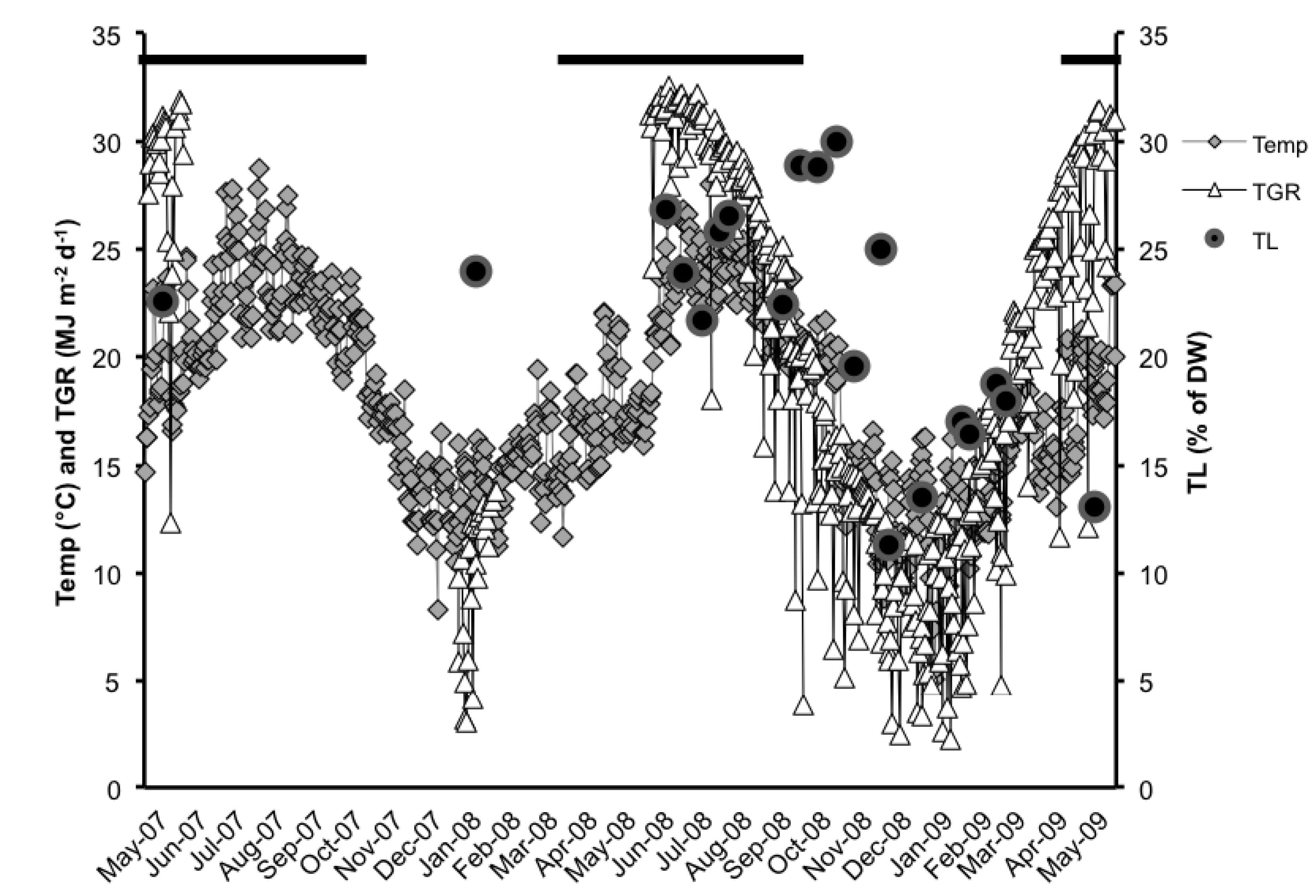
2.1. Total Lipids (TL)
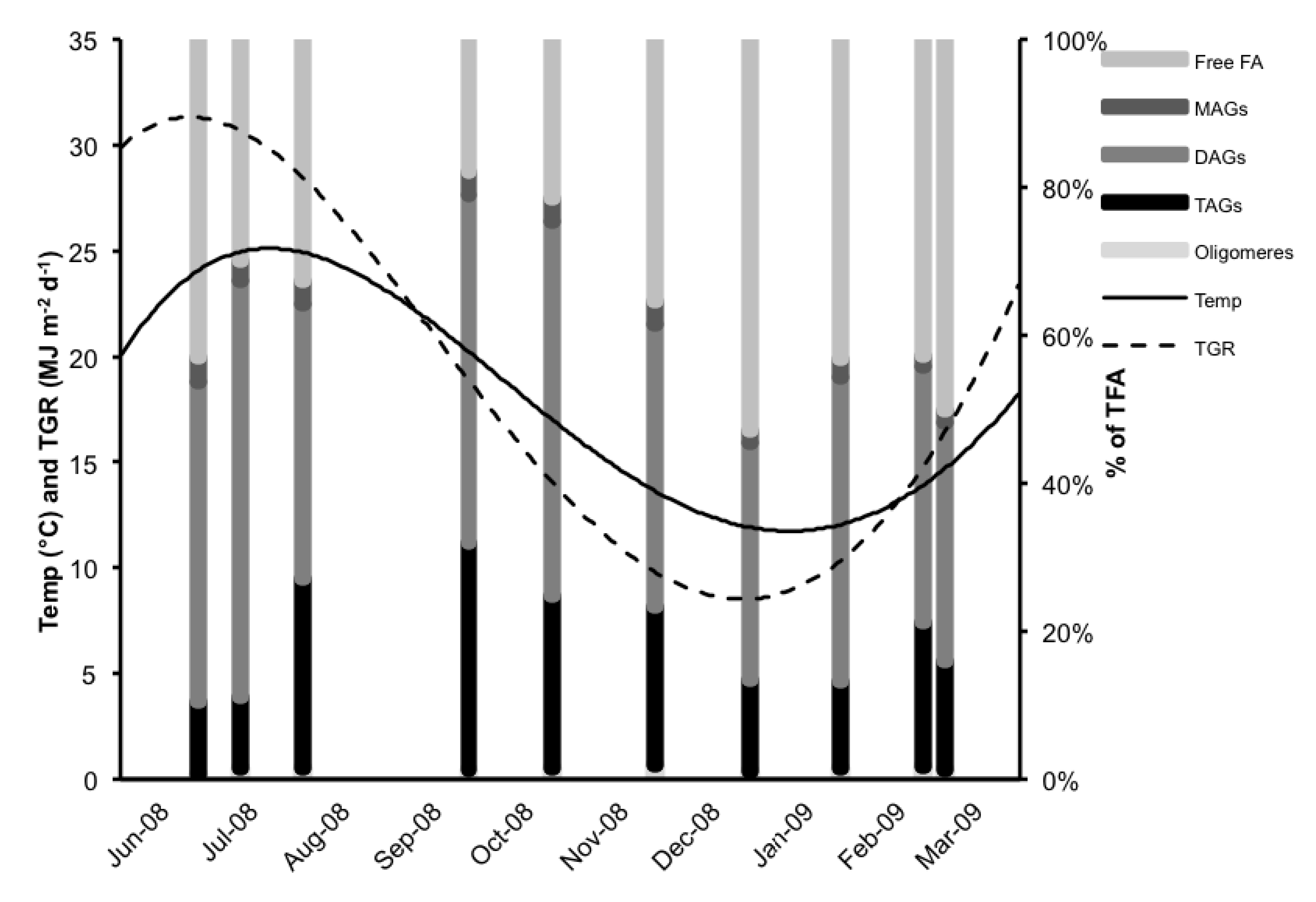
2.2. Neutral Lipid Classes
2.3. Fatty Acid (FA) Profile
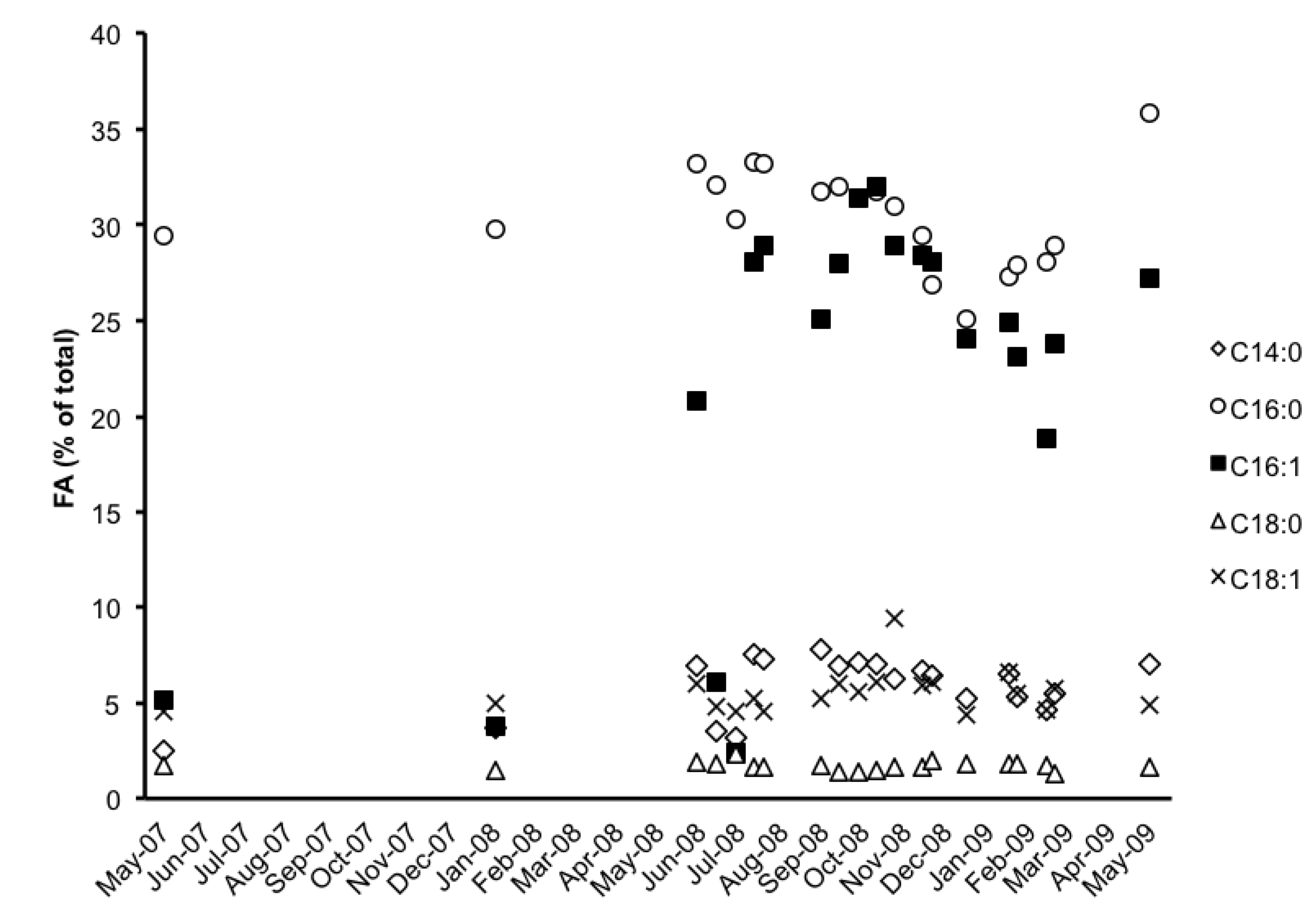
| FA | 13-Jun-08 | 27-Jun-08 | 11-Jul-08 | 25-Jul-08 | 1-Aug-08 | 12-Sep-08 | 26-Sep-08 | 10-Oct-08 | 24-Oct-08 |
| C14:0 | 6.9 | 3.5 | 3.2 | 7.5 | 7.3 | 7.8 | 6.9 | 7.1 | 7 |
| C15:0 | 0.7 | 0.7 | 0.8 | 0.7 | 0.7 | 0.7 | 0.6 | 0.7 | 0.7 |
| C16:0 | 33.2 | 32.1 | 30.3 | 33.3 | 33.2 | 31.7 | 32 | 31.4 | 31.7 |
| C16:1 ω7 | 20.8 | 6.1 | 2.4 | 28.1 | 28.9 | 25.1 | 28 | 31.4 | 32 |
| C16:2 ω4 | 2.1 | 6.5 | 6.7 | 0.7 | 0.7 | 0.9 | 0.6 | 0.6 | - |
| C16:3 ω4 | 1.4 | 4.4 | 6.7 | - | - | 0.5 | - | - | - |
| C18:0 | 1.9 | 1.8 | 2.3 | 1.6 | 1.6 | 1.7 | 1.4 | 1.4 | 1.5 |
| C18:1 ω9 | 5.3 | 3.8 | 3.1 | 4.7 | 4.1 | 4.7 | 5.6 | 5.2 | 5.7 |
| C18:1 ω7 | 0.7 | 1 | 1.4 | 0.5 | 0.4 | 0.5 | 0.4 | 0.4 | 0.4 |
| C18:2 ω6 | 8.4 | 24 | 24.8 | 2.9 | 2.4 | 4.3 | 2.9 | 2.6 | 2.5 |
| C18:3 ω4 | - | - | - | - | 0.5 | 0.3 | 0.6 | - | - |
| C18:3 ω3 | 3.9 | 11.9 | 16.3 | 0.4 | 16.3 | 1.7 | - | - | - |
| C20:4 ω6 | 3.7 | 1.1 | 0.4 | 5.4 | 5.6 | 5.6 | 5.9 | 5.7 | 5.6 |
| C20:5 ω3 | 11.1 | 2.4 | 0.8 | 14.2 | 14.7 | 14.4 | 15.1 | 13.3 | 14.7 |
| C22:3 ω3 | - | 0.6 | 0.8 | - | - | - | - | - | - |
| FA | 7-Nov-08 | 28-Nov-08 | 5-Dec-08 | 30-Dec-08 | 30-Jan-09 | 6-Feb-09 | 27-Feb-09 | 6-Mar-09 | 15-May-09 |
| C14:0 | 6.2 | 6.7 | 6.4 | 5.2 | 6.5 | 5.3 | 4.6 | 5.5 | 7 |
| C15:0 | 0.7 | 0.7 | 0.7 | 0.7 | 0.8 | 0.7 | 0.7 | 0.7 | 0.7 |
| C16:0 | 31 | 29.4 | 26.9 | 25.1 | 27.3 | 27.9 | 28.1 | 28.9 | 35.8 |
| C16:1 ω7 | 28.9 | 28.4 | 28.1 | 24.1 | 24.9 | 23.1 | 18.9 | 23.8 | 27.2 |
| C16:2 ω4 | 0.8 | - | 0.9 | 1.2 | 1.2 | 1.3 | 1.3 | 1.1 | - |
| C16:3 ω4 | - | - | 1.1 | 2.4 | 1.4 | 3 | 4.2 | 1.7 | - |
| C18:0 | 1.6 | 1.6 | 2 | 1.8 | 1.8 | 1.8 | 1.7 | 1.3 | 1.6 |
| C18:1 ω9 | 8.9 | 5.5 | 5.5 | 3.8 | 6 | 4.8 | 3.9 | 5.1 | 4.5 |
| C18:1 ω7 | 0.5 | 0.4 | 0.6 | 0.6 | 0.6 | 0.7 | 0.7 | 0.6 | 0.4 |
| C18:2 ω6 | 3.3 | 3.5 | 4.8 | 7.8 | 5.9 | 7.4 | 12.1 | 7.3 | 2.9 |
| C18:3 ω4 | - | - | - | - | - | - | - | - | - |
| C18:3 ω3 | - | 1.1 | 2.6 | 5.6 | 3.2 | 6.7 | 10.5 | 4.2 | - |
| C20:4 ω6 | 5 | 6.3 | 6.1 | 6 | 4.8 | 3.8 | 3.2 | 4.3 | 4.3 |
| C20:5 ω3 | 13.1 | 16.4 | 14.5 | 15.8 | 15.6 | 13.4 | 10 | 15.3 | 15.6 |
| C22:3 ω3 | - | - | - | - | - | - | - | - | - |
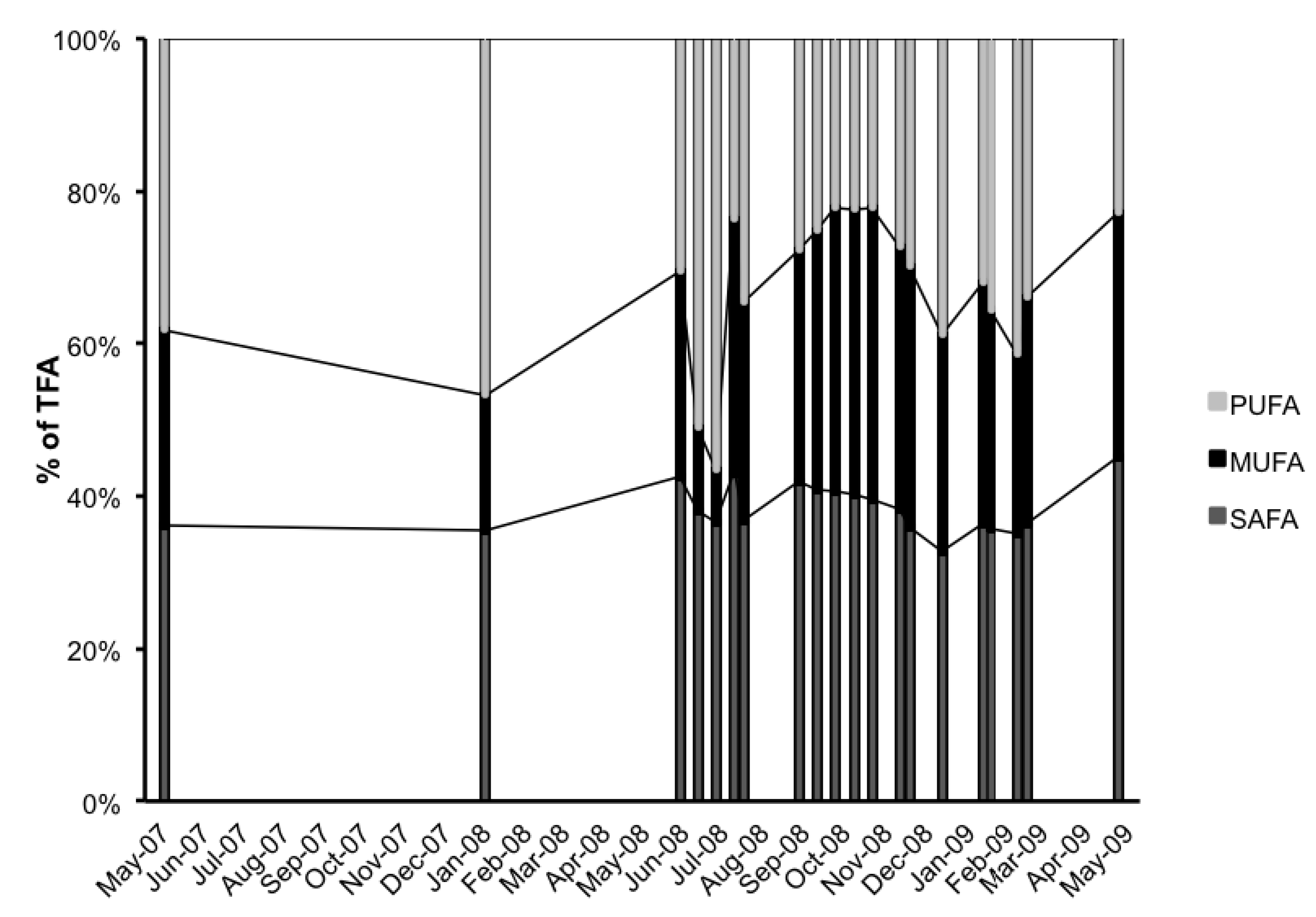

3. Experimental Section
4. Conclusions
Acknowledgments
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef] [PubMed]
- Schenk, P.M.; Thomas-Hall, S.R.; Stephens, E.; Marx, U.C.; Mussgnug, J.H.; Posten, C.; Kruse, O.; Hankamer, B. Second generation biofuels: High-efficiency microalgae for biodiesel production. Bioenergy Res. 2008, 1, 20–43. [Google Scholar] [CrossRef]
- Meng, X.; Yang, J.; Xu, X.; Zhang, L.; Nie, Q.; Xian, M. Biodiesel production from oleaginous microorganisms. Renew. Energy 2009, 34, 1–5. [Google Scholar] [CrossRef]
- Renaud, S.M.; Parry, D.L.; Thinh, L.V.; Kuo, C.; Padovan, A.; Sammy, N. Effect of light-intensity on the proximate biochemical and fatty-acid composition of Isochrysis sp. and nannochloropsis-oculata for use in tropical aquaculture. J. Appl. Phycol. 1991, 3, 43–53. [Google Scholar] [CrossRef]
- Dhert, P.; Rombaut, G.; Suantika, G.; Sorgeloos, P. Advancement of rotifer culture and manipulation techniques in Europe. Aquaculture 2001, 200, 129–146. [Google Scholar] [CrossRef]
- Rodolfi, L.; Zittelli, G.C.; Barsanti, L.; Rosati, G.; Tredici, M.R. Growth medium recycling in Nannochloropsis sp. mass cultivation. Biomol. Eng. 2003, 20, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Roncarati, A.; Meluzzi, A.; Acciarri, S.; Tallarico, N.; Melotti, P. Fatty acid composition of different microalgae strains (Nannochloropsis sp., Nannochloropsis oculata (Droop) Hibberd, Nannochloris atomus Butcher and Isochrysis sp.) according to the culture phase and the carbon dioxide concentration. J. World Aquacult. Soc. 2004, 35, 401–411. [Google Scholar] [CrossRef]
- Bentley, C.D.; Carroll, P.M.; Watanabe, W.O.; Riedel, A.M. Intensive rotifer production in a pilot-scale continuous culture recirculating system using nonviable microalgae and an ammonia neutralizer. J. World Aquacult. Soc. 2008, 39, 625–635. [Google Scholar] [CrossRef]
- Sandnes, J.M.; Källqvist, T.; Wenner, D.; Gislerød, H.R. Combined influence of light and temperature on growth rates of Nannochloropsis oceanica: Linking cellular responses to large-scale biomass production. J. Appl. Phycol. 2005, 17, 515–525. [Google Scholar] [CrossRef]
- Hu, C.; Li, M.; Li, J.; Zhu, Q.; Liu, Z. Variation of lipid and fatty acid compositions of the marine microalga Pavlova viridis (Prymnesiophyceae) under laboratory and outdoor culture conditions. World J. Microbiol. Biotechnol. 2007, 24, 1209–1214. [Google Scholar] [CrossRef]
- Zittelli, G.C.; Lavista, F.; Bastianini, A.; Rodolfi, L.; Vincenzini, M.; Tredici, M.R. Production of eicosapentaenoic acid by Nannochloropsis sp. cultures in outdoor tubular photobioreactors. J. Biotechnol. 1999, 70, 299–312. [Google Scholar] [CrossRef]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.Y.; Kao, C.Y.; Tsai, M.T.; Ong, S.C.; Chen, C.H.; Lin, C.S. Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour. Technol. 2009, 100, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, L.; Oliveira, A.C. Microalgae as a raw material for biofuels production. World J. Ind. Microbiol. Biotechnol. 2009, 36, 269–274. [Google Scholar] [CrossRef]
- Sukenik, A.; Yamaguchi, Y.; Livne, A. Alterations in lipid molecular-species of the marine eustigmatophyte Nannochloropsis sp. J. Phycol. 1993, 29, 620–626. [Google Scholar] [CrossRef]
- Brown, M.R.; Dunstan, G.A.; Norwood, S.J.; Miller, K.A. Effects of harvest stage and light on the biochemical composition of the diatom Thalassiosira pseudonana. J. Phycol. 1996, 32, 64–73. [Google Scholar] [CrossRef]
- Berge, J.P.; Gouygou, J.P.; Dubacq, J.P.; Durand, P. Reassessment of lipid-composition of the diatom, skeletonema-costatum. Phytochemistry 1995, 39, 1017–1021. [Google Scholar] [CrossRef]
- Tonon, T.; Harvey, D.; Larson, T.R.; Graham, I.A. Long chain polyunsaturated fatty acid production and partitioning to triacylglycerols in four microalgae. Phytochemistry 2002, 61, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, A.; Carmeli, Y.; Berner, T. Regulation of fatty-acid composition by irradiance level in the eustigmatophyte Nannochloropsis sp. J. Phycol. 1989, 25, 686–692. [Google Scholar] [CrossRef]
- Suen, Y.; Hubbard, J.S.; Holzer, G.; Tornabene, T.G. Total lipid production of the green-alga Nannochloropsis sp. qii under different nitrogen regimes. J. Phycol. 1987, 23, 289–296. [Google Scholar] [CrossRef]
- Takagi, M.; Watanabe, K.; Yamaberi, K.; Yoshida, T. Limited feeding of potassium nitrate for intracellular lipid and triglyceride accumulation of Nannochloris sp UTEX LB1999. Appl. Microbiol. Biotechnol. 2000, 54, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Weldy, C.S.; Huesemann, M. Lipid production by Dunaliella Salina in batch culture: Effects of nitrogen limitation and light intensity. J. Undergrad. Res. 2007, 115–122. [Google Scholar]
- Takagi, M.; Karseno; Yoshida, T. Effect of salt concentration on intracellular accumulation of lipids and triacylglyceride in marine microalgae Dunaliella cells. J. Biosci. Bioeng. 2006, 101, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, A.; Bennett, J.; Mortain-Bertrand, A.; Falkowski, P.G. Adaptation of the photosynthetic apparatus to irradiance in Dunaliella tertiolecta: A kinetic study. Plant Physiol. 1990, 92, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Fabregas, J.; Maseda, A.; Dominguez, A.; Otero, A. The cell composition of Nannochloropsis sp. changes under different irradiances in semicontinuous culture. World J. Microbiol. Biotechnol. 2004, 20, 31–35. [Google Scholar] [CrossRef]
- Lynch, D.V.; Thompson, G.A. Low temperature-induced alterations in the chloroplast and microsomal-membranes of Dunaliella salina. Plant Physiol. 1982, 69, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.A.; Guo, M.X.; Harrison, P.J.; Whyte, J.N.C. Effects of variation in temperature. 2. On the fatty-acid composition of 8 species of marine-phytoplankton. J. Phycol. 1992, 28, 488–497. [Google Scholar] [CrossRef]
- Aatola, H.; Larmi, M.; Sarjovaara, T. Hydrotreated Vegetable Oil (HVO) as a renewable diesel fuel: Trade-off between NOx, particulate emission, and fuel consumption of a heavy duty engine. SAE Int. J. Engines 2009, 1, 1251–1262. [Google Scholar]
- Herrera-Valencia, V.A.; Contreras-Pool, P.Y.; Lopez-Adrian, S.J.; Peraza-Echeverria, S.; Barahona-Perez, L.F. The green microalga chlorella saccharophila as a suitable source of oil for biodiesel production. Curr. Microbiol. 2011, 63, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G. “Designer” biodiesel: Optimizing fatty ester composition to improve fuel properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Ramos, M.J.; Fernandez, C.M.; Casas, A.; Rodriguez, L.; Perez, A. Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour. Technol. 2009, 100, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sotelo-Boyás, R.; Murata, K.; Minowa, T.; Sakanishi, K. Hydrotreatment of vegetable oils to produce bio-hydrogenated diesel and liquefied petroleum gas fuel over catalysts containing sulfided Ni–Mo and solid acids. Energy Fuels 2011, 25, 4675–4685. [Google Scholar] [CrossRef]
- Renaud, S.M.; Thinh, L.V.; Lambrinidis, G.; Parry, D.L. Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 2002, 211, 195–214. [Google Scholar] [CrossRef]
- Israelachvili, J.N.; Marcelja, S.; Horn, R.G. Physical principles of membrane organization. Q. Rev. Biophys. 1980, 13, 121–200. [Google Scholar] [CrossRef] [PubMed]
- Fisher, T.; Berner, T.; Iluz, D.; Dubinsky, Z. The kinetics of the photoacclimation response of Nannochloropsis sp. (Eustigmatophyceae): A study of changes in ultrastructure and PSU density. J. Phycol. 1998, 34, 818–824. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sustain. Energy Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Dunstan, G.A.; Volkman, J.K.; Barrett, S.M.; Garland, C.D. Changes in the lipid-composition and maximization of the polyunsaturated fatty-acid content of 3 microalgae grown in mass-culture. J. Appl. Phycol. 1993, 5, 71–83. [Google Scholar] [CrossRef]
- Shanklin, J.; Cahoon, E.B. Desaturation and related modifications of fatty acids. Annu. Rev. Plant Phys. 1998, 49, 611–641. [Google Scholar] [CrossRef]
- Volkman, J.K.; Brown, M.R.; Dunstan, G.A.; Jeffrey, S.W. The biochemical-composition of marine microalgae from the class eustigmatophyceae. J. Phycol. 1993, 29, 69–78. [Google Scholar] [CrossRef]
- European Standard EN 14214. Automotive fuels—Fatty acid methyl esters (FAME) for diesel engines—Requirements and test methods. AFNOR: Saint-Denis, France, 2004. [Google Scholar]
- Wijffels, R.H.; Barbosa, M.J.; Eppink, M.H.M. Microalgae for the production of bulk chemicals and biofuels. Biofuels Bioprod. Biorefin. 2010, 4, 287–295. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Olofsson, M.; Lamela, T.; Nilsson, E.; Bergé, J.P.; Del Pino, V.; Uronen, P.; Legrand, C. Seasonal Variation of Lipids and Fatty Acids of the Microalgae Nannochloropsis oculata Grown in Outdoor Large-Scale Photobioreactors. Energies 2012, 5, 1577-1592. https://doi.org/10.3390/en5051577
Olofsson M, Lamela T, Nilsson E, Bergé JP, Del Pino V, Uronen P, Legrand C. Seasonal Variation of Lipids and Fatty Acids of the Microalgae Nannochloropsis oculata Grown in Outdoor Large-Scale Photobioreactors. Energies. 2012; 5(5):1577-1592. https://doi.org/10.3390/en5051577
Chicago/Turabian StyleOlofsson, Martin, Teresa Lamela, Emmelie Nilsson, Jean Pascal Bergé, Victória Del Pino, Pauliina Uronen, and Catherine Legrand. 2012. "Seasonal Variation of Lipids and Fatty Acids of the Microalgae Nannochloropsis oculata Grown in Outdoor Large-Scale Photobioreactors" Energies 5, no. 5: 1577-1592. https://doi.org/10.3390/en5051577




