Recent Advances in High-Temperature Steam Electrolysis with Solid Oxide Electrolysers for Green Hydrogen Production
Abstract
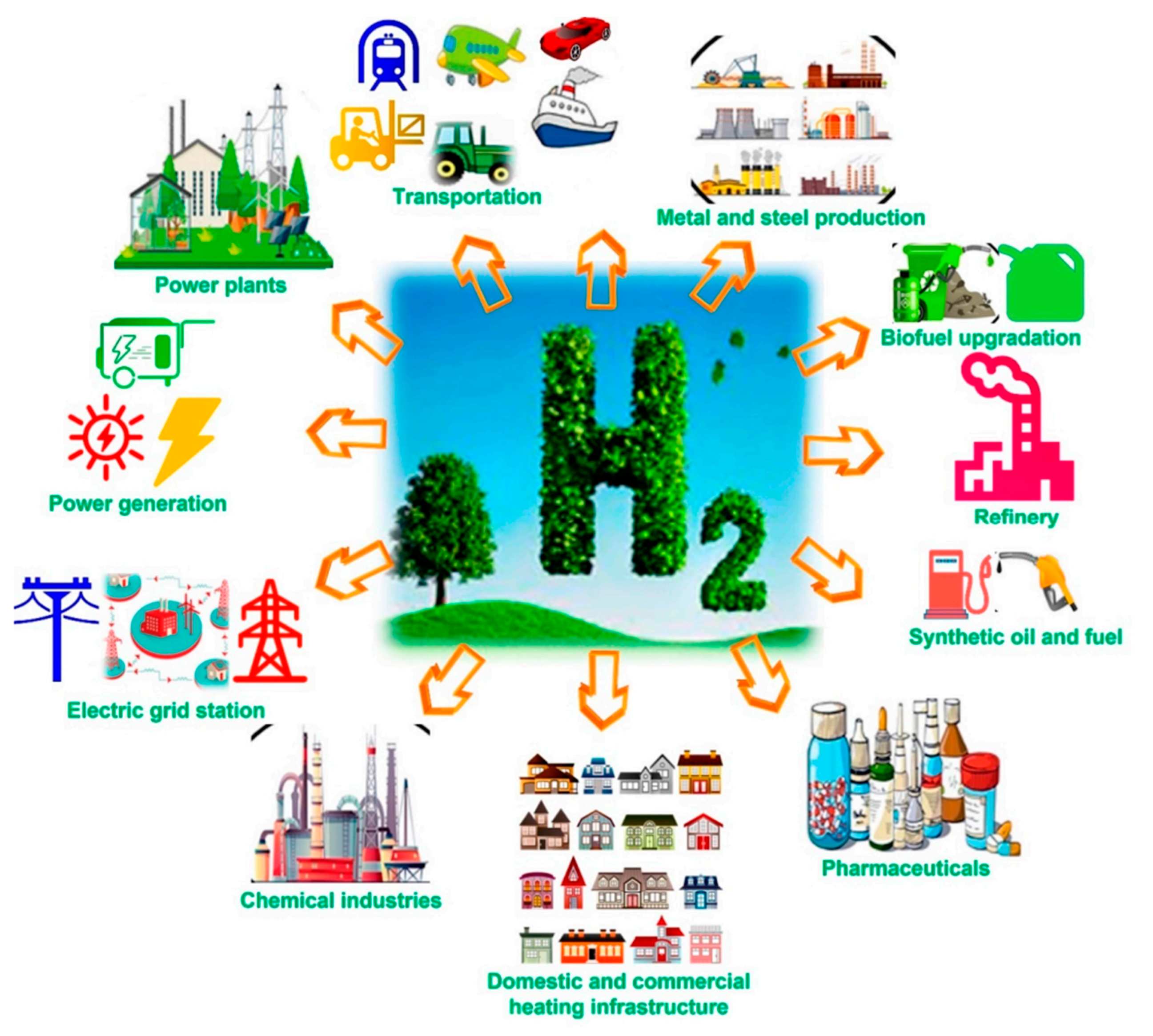
1. Introduction
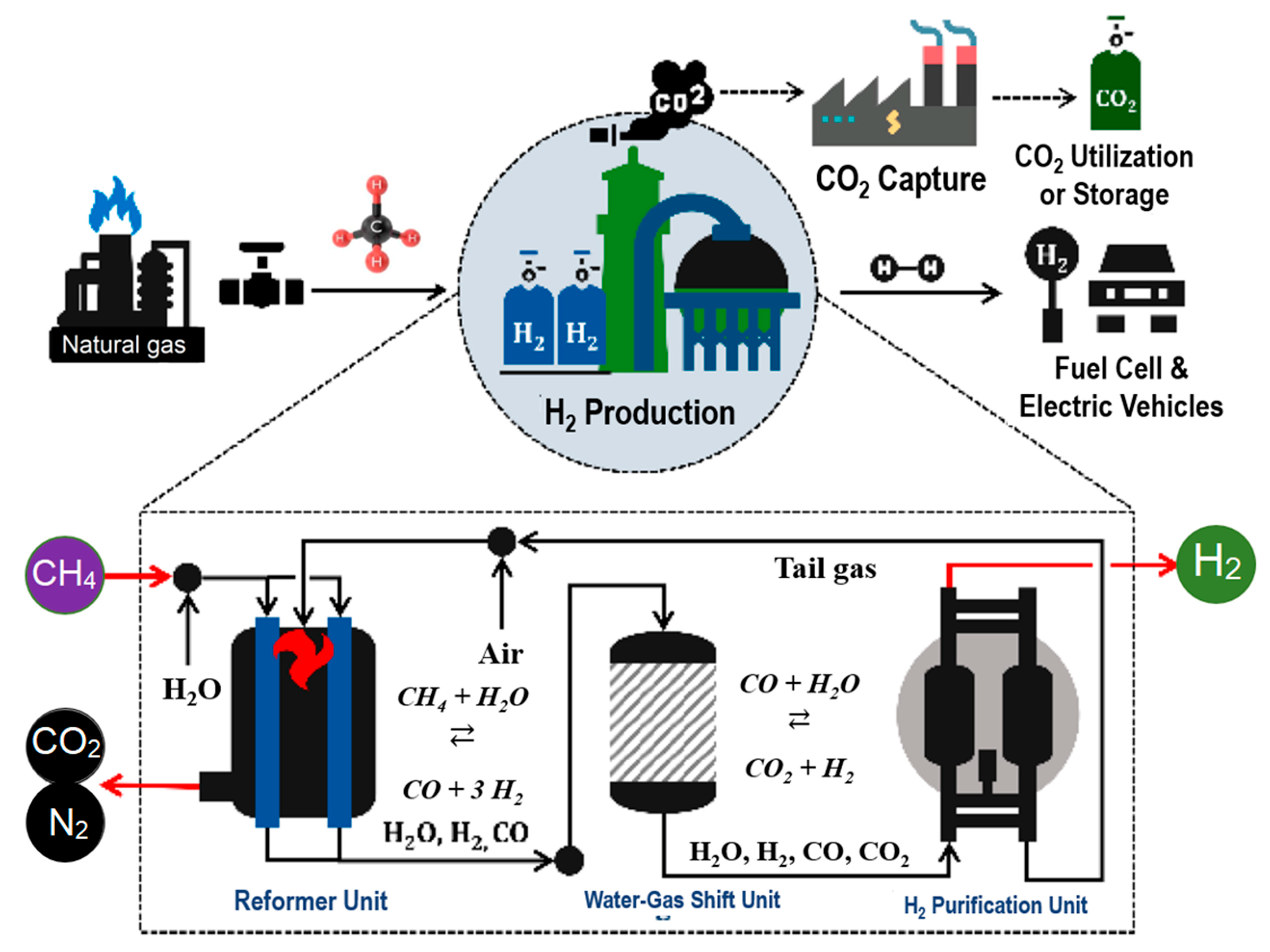
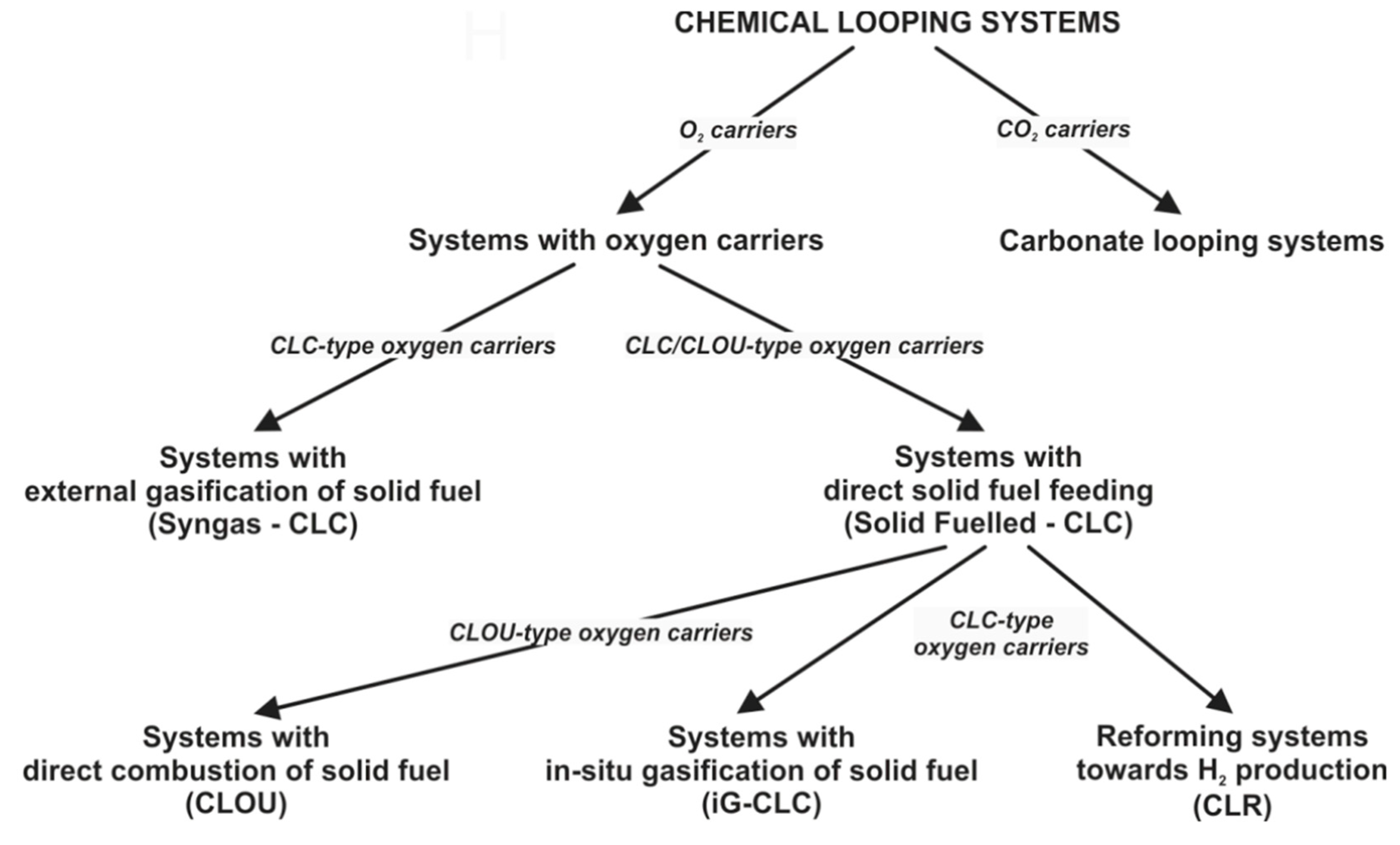
2. Hydrogen Generation Technologies
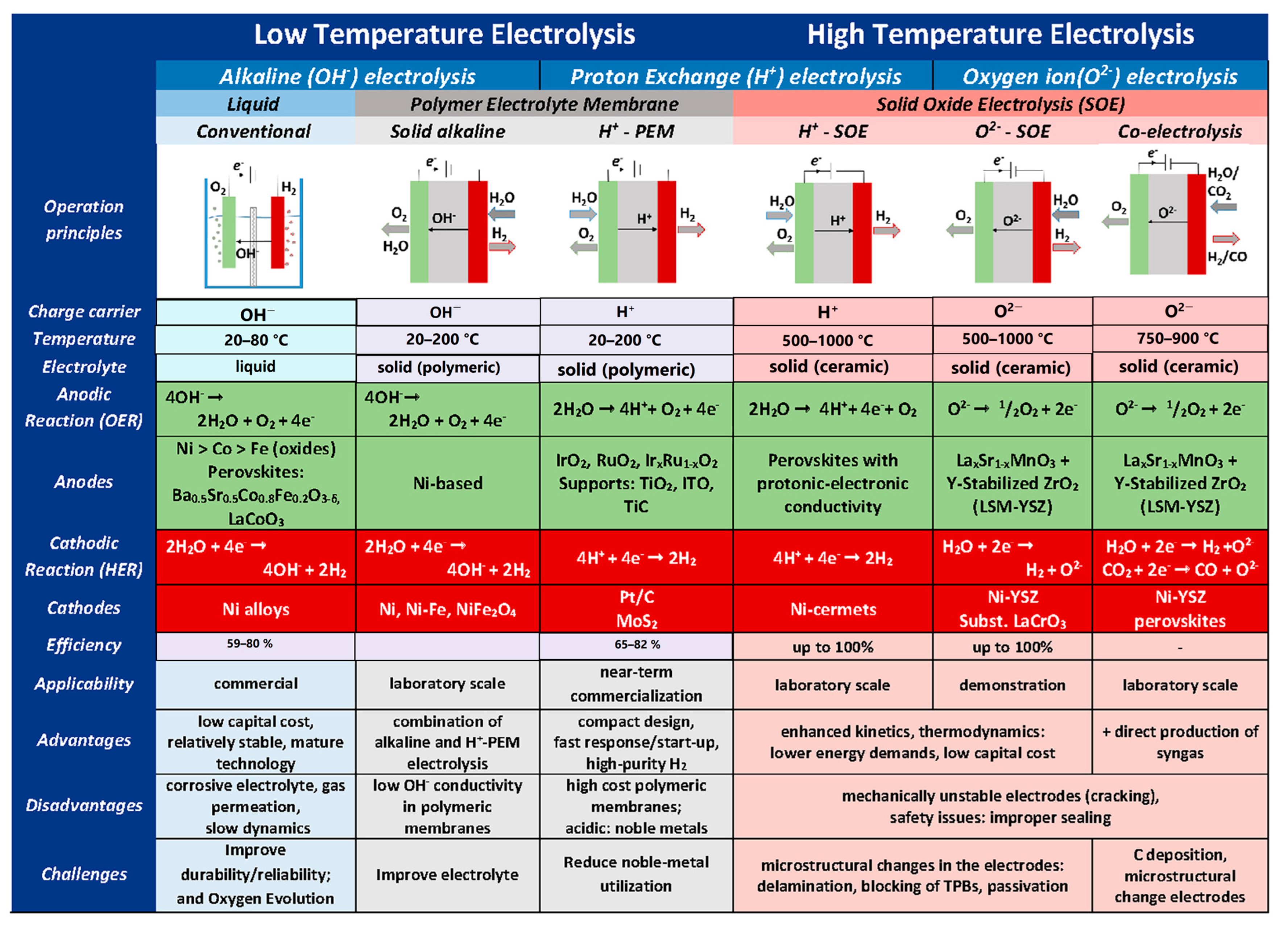
3. Water Electrolysis
3.1. Low-Temperature Water Electrolysis

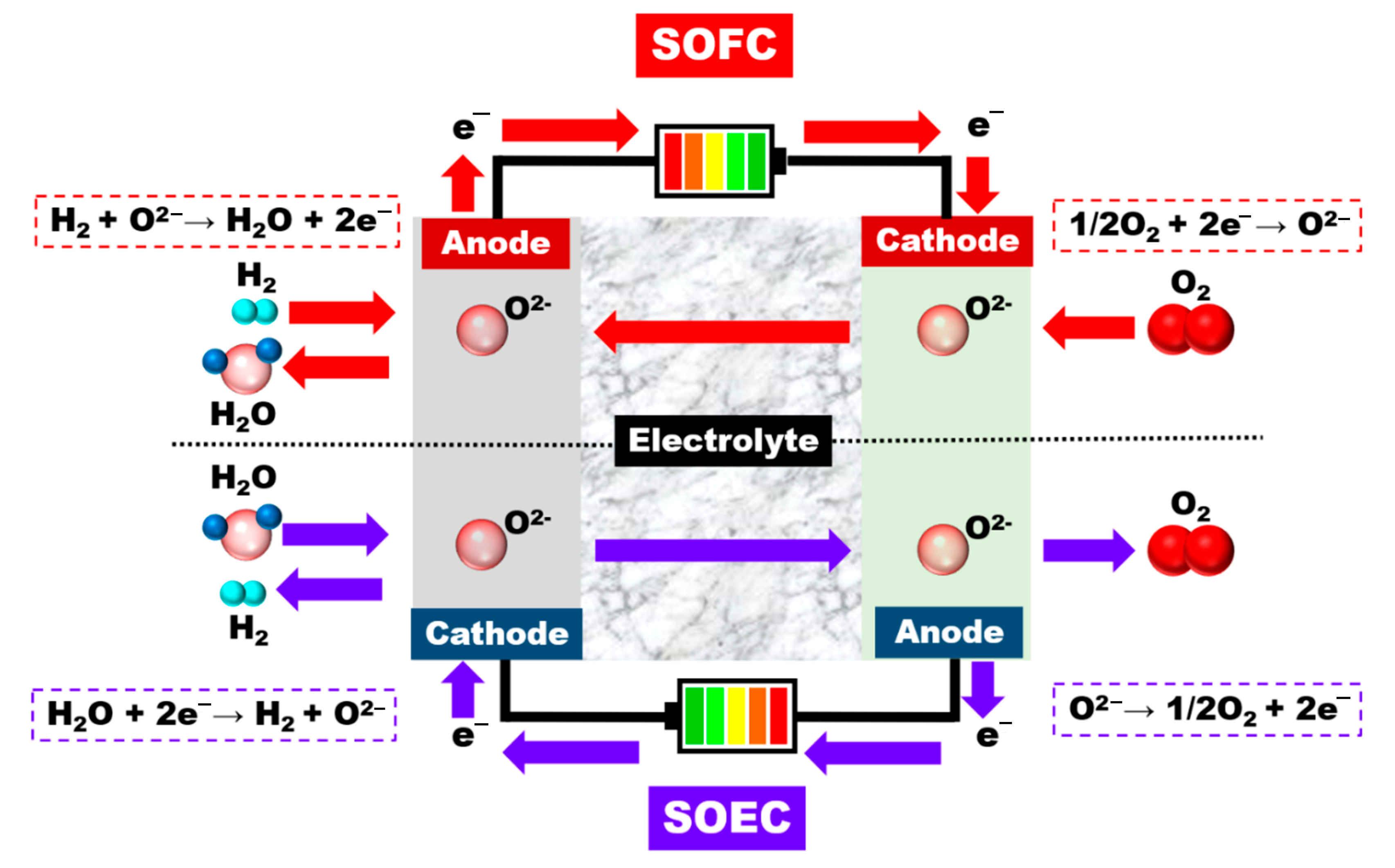
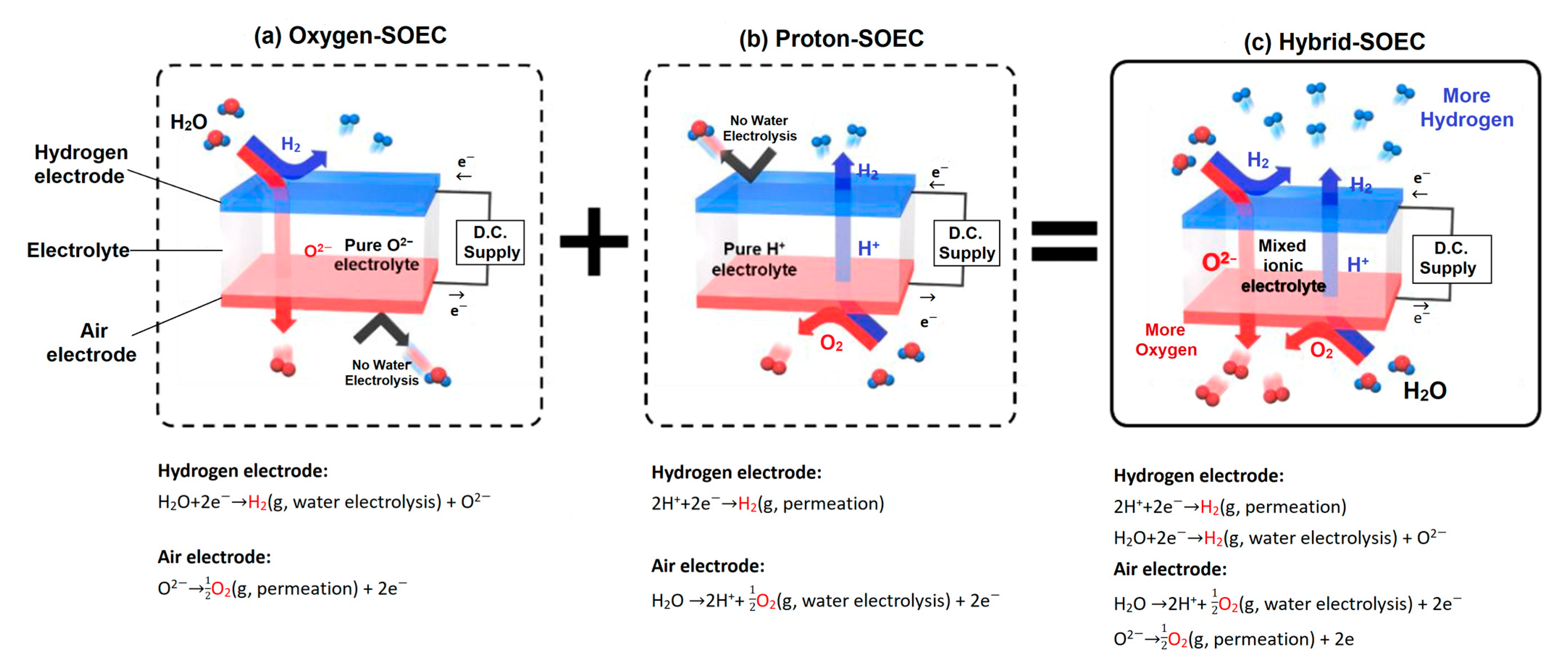
3.2. High-Temperature Steam Electrolysis (HTSE)
- Delamination
- Poisoning
- Microstructure coarsening during sintering
- Decomposition of electrodes
- Formation of an unwanted secondary phase due to the chemical reaction between the components [137].
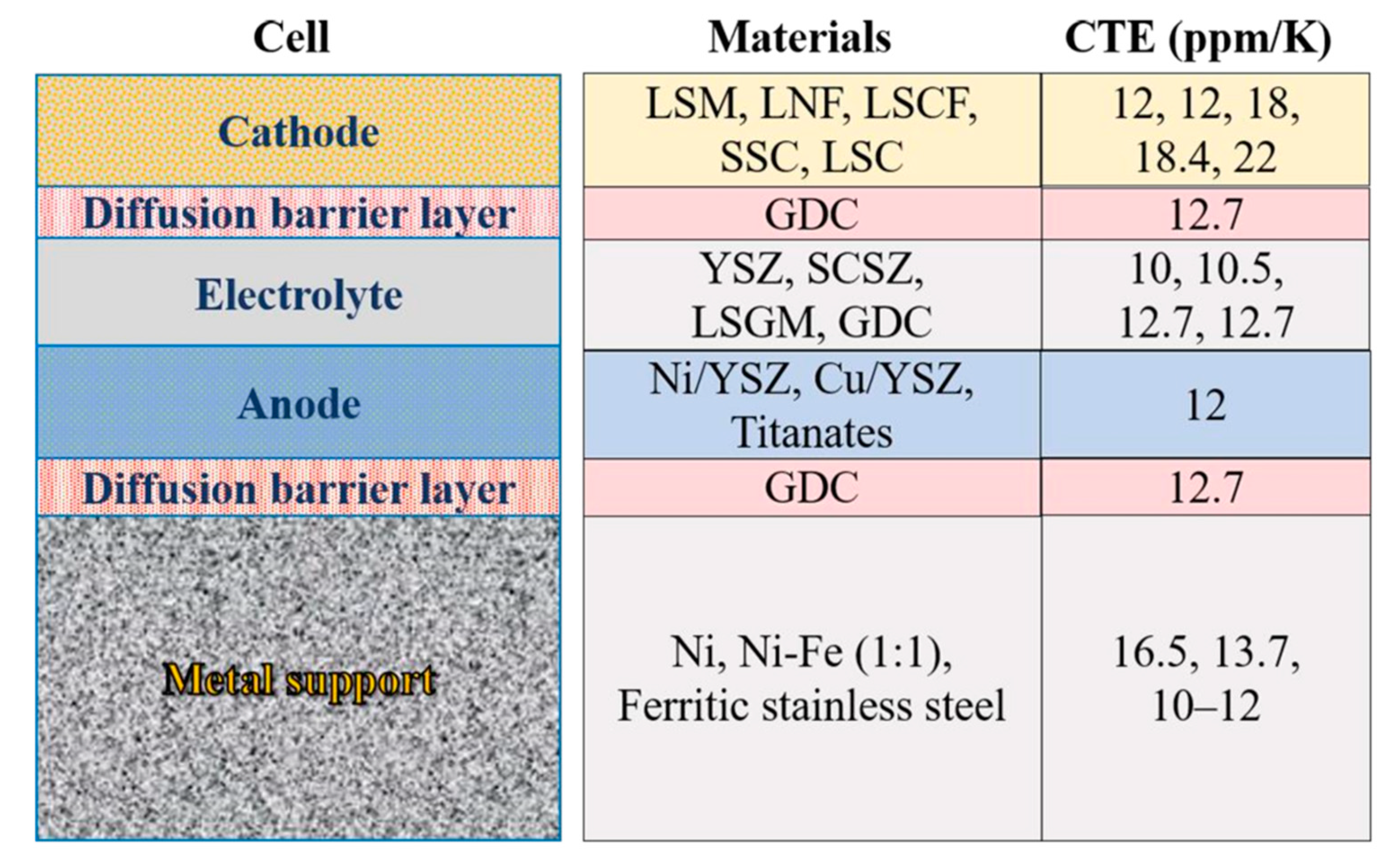
4. Progress in Materials Development for High-Temperature Steam Electrolysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| GHG | Greenhouse Gas | CL-SMR (CLRM) | Chemical Looping Steam Methane Reforming |
| GF | Greenisation Factor | SR-CLC (CLR(s)) | Steam Methane integrated with Chemical Looping Combustion |
| SMR | Steam Methane Reforming | CLC | Chemical Looping Combustion |
| ATR | Auto Thermal Reforming | CLR (a) | Autothermal Chemical Looping Reforming |
| POX | Partial Oxidation | CLG | Chemical Looping Gasification |
| SOE | Solid Oxide Electrolysis | IGCC | Integrated Gasification Combined Cycle |
| SOEC | Solid Oxide Electrolysis Cell | UGC | Underground Coal Gasification |
| SOFC | Solid Oxide Fuel Cell | MIEC | Mixed Ionic-Electronic Conductors |
| HTE | High-Temperature Electrolysis | ASR | Area Specific Resistance |
| HTSE | High-Temperature Steam Electrolysis | TEC | Thermal Expansion Coefficient |
| PEM | Proton-Exchange Membrane | OCAC | Oxygen Carrier Aided Combustion |
| AEM | Anion Exchange Membrane | OC | Oxygen Carrier |
| AEMWT | Anion Exchange Membrane Water Electrolysis | EIS | Electrochemical Impedance Spectroscopy |
| TRL | Technology Readiness Level | MSC | Metal-Supported Cell |
| HER | Hydrogen Evolution Reaction | MS-SOEC | Metal-Supported Solid Oxide Fuel Cell |
| OER | Oxygen Evolution Reaction | OCV | Open Circuit Voltage |
| PCEC(H-SOEC) | Proton-Conducting Solid Oxide Electrolysis Cell | IP | Integrated Planar |
| O-SOEC | Oxygen ion-Conducting Solid Oxide Electrolysis Cell | SLC | Stack Lifetime Capacity |
| TPB | Triple Phase Boundary | EPC | Electrical Power Consumption |
| AC/DC | Alternating Current/Direct Current | SL | Stereolithography |
| DLP | Digital Light Processing | AM | Additive Manufacturing |
| AFL | Anode Functional Layer | PLD | Pulsed Laser Deposition |
| ALD | Atomic Laser Deposition | SPS | Spark Plasma Sintering |
| PCE | Proton-Conducting Electrolyte | SAUP | Self-Architecture Ultra Porous |
| DR/RP-P | Double Perovskite/Ruddlesden-Popper Perovskite | CSA | Ceramic Sealing Agents |
| S-I cycle | Sulphur-Iodine Cycle | Cu-Cl cycle | Copper = Chlorine cycle |
| YSZ | Yttria-Stabilised Zirconia | LSCF | Lanthanum Strontium Cobalt Ferrite |
| YDC | Yttrium-Doped Ceria | GDC (CGO) | Gadolinium-Doped Ceria |
| LSM | Lanthanum Strontium Manganite | BZY | Yttrium-doped Barium Zirconate |
| BZCYYb | Yttrium-and Ytterbium-doped Barium-Zirconium-Cerate | LNF | Lanthanum Nickel Ferrite |
| SSC | Strontium Samarium Cobaltite | LSC | Lanthanum Strontium Cobaltite |
| LSGM | Lanthanum Strontium Gallium Magnesium oxide | SCSZ | Scandia-Ceria-Stabilised Zirconia |
| YbScSZ | Ytterbium Scandium Stabilized Zirconia | CFA | Co-Fe alloy |
| ScSZ | Scandia-Stabilised Zirconia | ESB | Erbium-Stabilised Bismuth oxide |
| μXRD | Synchrotron X-Ray Micro-Diffraction | μXRF | Synchrotron X-Ray Micro-Fluorescence |
| IDL | Inter-Diffusional Layer | CGPO | Pr, Gd co-doped Ceria |
| LSTFO | Iron and Lanthanum-doped Strontium Titanate | LNO | Lanthanum Nickelate |
| LSCN | Nickel-doped Lanthanum Strontium Chromite | BSCF | Barium Strontium Cobalt Ferrite |
| E | actual OCV of the oxygen concentration cell | E 0 | Theoretical OCV of the oxygen concentration cell |
| R | Universal gas constant | T | Temperature (K) |
| F | Faraday constant (96,485 C.mol−1) | oxygen partial pressure of the air electrode | |
| oxygen partial pressure of the fuel electrode | ti | ion transport number | |
| te | electron transport number | η | Faraday efficiency |
| vmeas | calculated hydrogen evolution rate | vtheo | theoretical hydrogen evolution rate |
| I | applied current | Z | electron transport number of steam electrolysis |
| εenergetic | Energetic efficiency | equilibrium cell potential for product k | |
| εk,faradaic | Faradaic efficiency of the product | η | Cell overpotential |
| fi,out | volume flow of produced fuel gas component i | Vi,m | molar volume of reactant component i |
| Pel | consumed electrolysis power |
References
- Hasanuzzaman, M.; Islam, M.A.; Rahim, N.A.; Yuan, Y. Energy Demand. In Energy for Sustainable Development: Demand, Supply, Conversion and Management; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 41–87. ISBN 9780128146453. [Google Scholar]
- Fallah Vostakola, M.; Amini Horri, B. Progress in Material Development for Low-Temperature Solid Oxide Fuel Cells: A Review. Energies 2021, 14, 1280. [Google Scholar] [CrossRef]
- Fallah Vostakola, M.; Mirkazemi, S.M.; Eftekhari Yekta, B. Structural, Morphological, and Optical Properties of W-Doped VO2 Thin Films Prepared by Sol-Gel Spin Coating Method. Int. J. Appl. Ceram. Technol. 2019, 16, 943–950. [Google Scholar] [CrossRef]
- Hooshyari, K.; Amini Horri, B.; Abdoli, H.; Fallah Vostakola, M.; Kakavand, P.; Salarizadeh, P. A Review of Recent Developments and Advanced Applications of High-Temperature Polymer Electrolyte Membranes for PEM Fuel Cells. Energies 2021, 14, 5440. [Google Scholar] [CrossRef]
- Ozcan, H.; El-Emam, R.S.; Horri, B.A. Thermochemical Looping Technologies for Clean Hydrogen Production—Current Status and Recent Advances. J. Clean. Prod. 2022, 382, 135295. [Google Scholar] [CrossRef]
- Calcabrini, A.; Procel, P.; Huang, B.; Calcabrini, A.; Moya, P.P.; Huang, B.; Kambhampati, V. Low-Breakdown-Voltage Solar Cells for Shading-Tolerant Photovoltaic Modules Low-Breakdown-Voltage Solar Cells for Shading-Tolerant Photovoltaic Modules. Cell Rep. Phys. Sci. 2022, 3, 101155. [Google Scholar] [CrossRef]
- Gautier, A.; Wetter, M.; Sulzer, M. Resilient Cooling through Geothermal District Energy System. Appl. Energy 2022, 325, 119880. [Google Scholar] [CrossRef]
- Rezaei, S.E.; Zebarjadi, M.; Esfarjani, K. Effect of Exchange-Correlation Functional Type and Spin-Orbit Coupling on Thermoelectric Properties of ZrTe2. J. Solid State Chem. 2021, 302, 122414. [Google Scholar] [CrossRef]
- Eloffy, M.G.; Elgarahy, A.M.; Saber, A.N.; Hammad, A.; El-Sherif, D.M.; Shehata, M.; Mohsen, A.; Elwakeel, K.Z. Biomass-to-Sustainable Biohydrogen: Insights into the Production Routes, and Technical Challenges. Chem. Eng. J. Adv. 2022, 12, 100410. [Google Scholar] [CrossRef]
- Yilmaz, S.; Cobaner, S.; Yalaz, E.; Horri, B.A. Synthesis and Characterization of Gadolinium-Doped Zirconia as a Potential Electrolyte for Solid Oxide Fuel Cells. Energies 2022, 15, 2826. [Google Scholar] [CrossRef]
- Gebert, F.; Longhini, M.; Conti, F.; Naylor, A.J. An Electrochemical Evaluation of State-of-the-Art Non-Flammable Liquid Electrolytes for High-Voltage Lithium-Ion Batteries. J. Power Source 2023, 556, 232412. [Google Scholar] [CrossRef]
- El-emam, R.S.; Dincer, I.; Zamfirescu, C. Enhanced CANDU Reactor with Heat Upgrade for Combined Power and Hydrogen Production. Int. J. Hydrogen Energy 2019, 44, 23580–23588. [Google Scholar] [CrossRef]
- Vostakola, M.F.; Salamatinia, B.; Horri, B.A. A Review on Recent Progress in the Integrated Green Hydrogen Production Processes. Energies 2022, 15, 1209. [Google Scholar] [CrossRef]
- Qazi, U.Y. Future of Hydrogen as an Alternative Fuel for Next-Generation Industrial Applications; Challenges and Expected Opportunities. Energies 2022, 15, 4741. [Google Scholar] [CrossRef]
- Javaid, R. Catalytic Hydrogen Production, Storage and Application. Catalysts 2021, 11, 836. [Google Scholar] [CrossRef]
- Pereira, C.A.; Coelho, P.M.; Fernandes, J.F.; Gomes, M.H. Study of an Energy Mix for the Production of Hydrogen. Int. J. Hydrogen Energy 2017, 42, 1375–1382. [Google Scholar] [CrossRef]
- El-Emam, R.S.; Khamis, I. Advances in Nuclear Hydrogen Production: Results from an IAEA International Collaborative Research Project. Int. J. Hydrogen Energy 2019, 44, 19080–19088. [Google Scholar] [CrossRef]
- Verfondern, K.; Yan, X.; Nishihara, T.; Allelein, H.J. Safety Concept of Nuclear Cogeneration of Hydrogen and Electricity. Int. J. Hydrogen Energy 2017, 42, 7551–7559. [Google Scholar] [CrossRef]
- Dincer, I. Greenization. Int. J. Energy Res. 2016, 40, 2035–2037. [Google Scholar] [CrossRef]
- Amini Horri, B.; Choolaei, M.; Chaudhry, A.; Qaalib, H. A Highly Efficient Hydrogen Generation Electrolysis System Using Alkaline Zinc Hydroxide Solution. Int. J. Hydrogen Energy 2019, 44, 72–81. [Google Scholar] [CrossRef]
- Rafique, M.; Mubashar, R.; Irshad, M.; Gillani, S.S.A.; Tahir, M.B.; Khalid, N.R.; Yasmin, A.; Shehzad, M.A. A Comprehensive Study on Methods and Materials for Photocatalytic Water Splitting and Hydrogen Production as a Renewable Energy Resource. J. Inorg. Organomet. Polym. Mater. 2020, 30, 3837–3861. [Google Scholar] [CrossRef]
- Yang, W.; Moon, J. Recent Advances in Earth-Abundant Photocathodes for Photoelectrochemical Water Splitting. ChemSusChem 2019, 12, 1889–1899. [Google Scholar] [CrossRef]
- Zaman, N.; Iqbal, N.; Noor, T. Advances and Challenges of MOF Derived Carbon-Based Electrocatalysts and Photocatalyst for Water Splitting: A Review. Arab. J. Chem. 2022, 15, 103906. [Google Scholar] [CrossRef]
- Yu, J.M.; Lee, J.; Kim, Y.S.; Song, J.; Oh, J.; Lee, S.M.; Jeong, M.; Kim, Y.; Kwak, J.H.; Cho, S.; et al. High-Performance and Stable Photoelectrochemical Water Splitting Cell with Organic-Photoactive-Layer-Based Photoanode. Nat. Commun. 2020, 11, 5509. [Google Scholar] [CrossRef] [PubMed]
- Boretti, A.; Nayfeh, J.; Al-Maaitah, A. Hydrogen Production by Solar Thermochemical Water-Splitting Cycle via a Beam Down Concentrator. Front. Energy Res. 2021, 9, 666191. [Google Scholar] [CrossRef]
- Vita, A.; Italiano, C.; Pino, L. Distributed H2 Production from Bioalcohols and Biomethane in Conventional Steam Reforming Units. In Bioenergy Systems for the Future: Prospects for Biofuels and Biohydrogen; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; pp. 279–320. ISBN 9780081010266. [Google Scholar]
- Le Saché, E.; Johnson, S.; Pastor-Pérez, L.; Horri, B.A.; Reina, T.R. Biogas Upgrading via Dry Reforming over a Ni-Sn/CeO2-Al2O3 Catalyst: Influence of the Biogas Source. Energies 2019, 12, 1007. [Google Scholar] [CrossRef]
- Zhang, Y.; Wan, L.; Guan, J.; Xiong, Q.; Zhang, S.; Jin, X. A Review on Biomass Gasification: Effect of Main Parameters on Char Generation and Reaction. Energy Fuels 2020, 34, 13438–13455. [Google Scholar] [CrossRef]
- Lopez, G.; Santamaria, L.; Lemonidou, A.; Zhang, S.; Wu, C.; Ayesha, T.S.; Gao, N. Hydrogen Generation from Biomass by Pyrolysis. Nat. Rev. Methods Prim. 2022, 2, 20. [Google Scholar] [CrossRef]
- Ayodele, B.V.; Mustapa, S.I.; Tuan Abdullah, T.A.R.B.; Salleh, S.F. A Mini-Review on Hydrogen-Rich Syngas Production by Thermo-Catalytic and Bioconversion of Biomass and Its Environmental Implications. Front. Energy Res. 2019, 7, 118. [Google Scholar] [CrossRef]
- Midilli, A.; Kucuk, H.; Topal, M.E.; Akbulut, U.; Dincer, I. A Comprehensive Review on Hydrogen Production from Coal Gasification: Challenges and Opportunities. Int. J. Hydrogen Energy 2021, 46, 25385–25412. [Google Scholar] [CrossRef]
- Damle, A.S. Hydrogen Production by Reforming of Liquid Hydrocarbons in a Membrane Reactor for Portable Power Generation-Experimental Studies. J. Power Source 2009, 186, 167–177. [Google Scholar] [CrossRef]
- Quarton, C.J.; Tlili, O.; Welder, L.; Mansilla, C.; Blanco, H.; Heinrichs, H.; Leaver, J.; Samsatli, N.J.; Lucchese, P.; Robinius, M.; et al. The Curious Case of the Conflicting Roles of Hydrogen in Global Energy Scenarios. Sustain. Energy Fuels 2019, 4, 80–95. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.; Qi, M.; Lee, I.; Moon, I. Process Integration of an Autothermal Reforming Hydrogen Production System with Cryogenic Air Separation and Carbon Dioxide Capture Using Liquefied Natural Gas Cold Energy. Ind. Eng. Chem. Res. 2021, 60, 7257–7274. [Google Scholar] [CrossRef]
- Ismail, M.; Liu, W.; Dunstan, M.T.; Scott, S.A. Development and Performance of Iron Based Oxygen Carriers Containing Calcium Ferrites for Chemical Looping Combustion and Production of Hydrogen. Int. J. Hydrogen Energy 2016, 41, 4073–4084. [Google Scholar] [CrossRef]
- Fazlikeshteli, S.; Vendrell, X.; Llorca, J. Low-Temperature Partial Oxidation of Methane over Pd–Ni Bimetallic Catalysts Supported on CeO2. Int. J. Hydrogen Energy 2022, 48, 12024–12035. [Google Scholar] [CrossRef]
- Sánchez-Bastardo, N.; Schlögl, R.; Ruland, H. Methane Pyrolysis for Zero-Emission Hydrogen Production: A Potential Bridge Technology from Fossil Fuels to a Renewable and Sustainable Hydrogen Economy. Ind. Eng. Chem. Res. 2021, 60, 11855–11881. [Google Scholar] [CrossRef]
- El-emam, R.S.; Ozcan, H.; Zamfirescu, C. Updates on Promising Thermochemical Cycles for Clean Hydrogen Production. J. Clean. Prod. 2020, 262, 121424. [Google Scholar] [CrossRef]
- Crosbie, L.M.; Chapin, D. Hydrogen Production by Nuclear Heat. In Proceedings of the GENES4/ANP2003: International Conference on Global Environment and Advanced Nuclear Power Plants, Kyoto, Japan, 15–19 September 2003; pp. 15–19. [Google Scholar]
- Juárez-Martínez, L.C.; Espinosa-Paredes, G.; Vázquez-Rodríguez, A.; Romero-Paredes, H. Energy Optimization of a Sulfur–Iodine Thermochemical Nuclear Hydrogen Production Cycle. Nucl. Eng. Technol. 2021, 53, 2066–2073. [Google Scholar] [CrossRef]
- O’Brien, J.E.; Stoots, C.M.; Herring, J.S.; Hartvigsen, J.J. Performance of Planar High-Temperature Electrolysis Stacks for Hydrogen Production from Nuclear Energy. Nucl. Technol. 2007, 158, 118–131. [Google Scholar] [CrossRef]
- Fujiwara, N.; Nagase, H.; Tada, S.; Kikuchi, R. Hydrogen Production by Steam Electrolysis in Solid Acid Electrolysis Cells. ChemSusChem 2021, 14, 417–427. [Google Scholar] [CrossRef]
- Schiller, G.; Lang, M.; Monnerie, N.; Sundarraj, P.; Szabo, P. Solid Oxide Steam Electrolysis with Integration of Solar Heat. ECS Trans. 2019, 91, 2553–2564. [Google Scholar] [CrossRef]
- Ajanovic, A.; Sayer, M.; Haas, R. The Economics and the Environmental Benignity of Different Colors of Hydrogen. Int. J. Hydrogen Energy 2022, 47, 24136–24154. [Google Scholar] [CrossRef]
- Boretti, A.; Banik, B.K. Advances in Hydrogen Production from Natural Gas Reforming. Adv. Energy Sustain. Res. 2021, 2, 2100097. [Google Scholar] [CrossRef]
- Kim, J.; El-hameed, A.A.; Soja, R.J.; Ramadhan, H.H.; Nandutu, M. Estimation of the Levelized Cost of Nuclear Hydrogen Production from Light Water Reactors in the United States. Processes 2022, 10, 1620. [Google Scholar] [CrossRef]
- Ingale, G.U.; Kwon, H.; Jeong, S.; Park, D.; Kim, W.; Bang, B.; Lim, Y.; Kim, S.W.; Kang, Y.; Mun, J.; et al. Assessment of Greenhouse Gas Emissions from Hydrogen Production Processes: Turquoise Hydrogen vs. Steam Methane Reforming. Energies 2022, 15, 8679. [Google Scholar] [CrossRef]
- Lee, S.; Kim, H.S.; Park, J.; Kang, B.M.; Cho, C.H.; Lim, H.; Won, W. Scenario-Based Techno-Economic Analysis of Steam Methane Reforming Process for Hydrogen Production. Appl. Sci. 2021, 11, 6021. [Google Scholar] [CrossRef]
- Chen, G.; Tao, J.; Liu, C.; Yan, B.; Li, W.; Li, X. Hydrogen Production via Acetic Acid Steam Reforming: A Critical Review on Catalysts. Renew. Sustain. Energy Rev. 2017, 79, 1091–1098. [Google Scholar] [CrossRef]
- Shiva Kumar, S.; Himabindu, V. Hydrogen Production by PEM Water Electrolysis—A Review. Mater. Sci. Energy Technol. 2019, 2, 442–454. [Google Scholar] [CrossRef]
- Voitic, G.; Pichler, B.; Basile, A.; Iulianelli, A.; Malli, K.; Bock, S.; Hacker, V. Hydrogen Production; Elsevier Inc.: Amsterdam, The Netherlands, 2018; ISBN 9780128114599. [Google Scholar]
- Hildor, F.; Leion, H.; Mattisson, T. Steel Converter Slag as an Oxygen Carrier—Interaction with Sulfur Dioxide. Energies 2022, 15, 5922. [Google Scholar] [CrossRef]
- Tang, M.; Xu, L.; Fan, M. Progress in Oxygen Carrier Development of Methane-Based Chemical-Looping Reforming: A Review. Appl. Energy 2015, 151, 143–156. [Google Scholar] [CrossRef]
- Czakiert, T.; Krzywanski, J.; Zylka, A.; Nowak, W. Chemical Looping Combustion: A Brief Overview. Energies 2022, 15, 1563. [Google Scholar] [CrossRef]
- Krzywanski, J.; Czakiert, T.; Zylka, A.; Nowak, W.; Sosnowski, M.; Grabowska, K.; Skrobek, D.; Sztekler, K.; Kulakowska, A.; Ashraf, W.M.; et al. Modelling of SO2 and NOx Emissions from Coal and Biomass Combustion in Air-Firing, Oxyfuel, IG-CLC, and CLOU Conditions by Fuzzy Logic Approach. Energies 2022, 15, 8095. [Google Scholar] [CrossRef]
- Lee, M.; Kim, Y.; Lim, H.S.; Jo, A.; Kang, D.; Lee, J.W. Reverse Water-Gas Shift Chemical Looping Using a Core-Shell Structured Perovskite Oxygen Carrier. Energies 2020, 13, 5324. [Google Scholar] [CrossRef]
- Wang, T. An Overview of IGCC Systems. In Integrated Gasification Combined Cycle (IGCC) Technologies; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; pp. 1–80. ISBN 9780081001677. [Google Scholar]
- Zhu, Y.; Frey, H.C. Integrated Gasification Combined Cycle (IGCC) Systems. In Combined Cycle Systems for Near-Zero Emission Power Generation; Woodhead Publishing: Raleigh, NC, USA, 2012; pp. 129–161. [Google Scholar]
- Wagner, N.J.; Coertzen, M.; Matjie, R.H.; van Dyk, J.C. Coal Gasification. In Applied Coal Petrology: The Role of Petrology in Coal Utilization; Academic Press: Amsterdam, The Netherlands, 2008; pp. 119–144. ISBN 9780080450513. [Google Scholar]
- Kumar, S.; Wang, Z.; He, Y.; Zhu, Y.; Cen, K. Numerical Analysis for Coal Gasification Performance in a Lab-Scale Gasifier: Effects of the Wall Temperature and Oxygen/Coal Ratio. Energies 2022, 15, 8645. [Google Scholar] [CrossRef]
- Ji, M.; Wang, J. Review and Comparison of Various Hydrogen Production Methods Based on Costs and Life Cycle Impact Assessment Indicators. Int. J. Hydrogen Energy 2021, 46, 38612–38635. [Google Scholar] [CrossRef]
- Thakur, P. CO2 Sequestration and Underground Coal Gasification with Horizontal Wells, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128159972. [Google Scholar]
- Vidas, L.; Castro, R. Recent Developments on Hydrogen Production Technologies: State-of-the-Art Review with a Focus on Green-Electrolysis. Appl. Sci. 2021, 11, 11363. [Google Scholar] [CrossRef]
- Lepage, T.; Kammoun, M.; Schmetz, Q.; Richel, A. Biomass-to-Hydrogen: A Review of Main Routes Production, Processes Evaluation and Techno-Economical Assessment. Biomass Bioenergy 2021, 144, 105920. [Google Scholar] [CrossRef]
- Nikolaidis, P.; Poullikkas, A. A Comparative Overview of Hydrogen Production Processes. Renew. Sustain. Energy Rev. 2017, 67, 597–611. [Google Scholar] [CrossRef]
- Megia, P.J.; Vizcaino, A.J.; Calles, J.A.; Carrero, A. Hydrogen Production Technologies: From Fossil Fuels toward Renewable Sources. A Mini Review. Energy Fuels 2021, 35, 16403–16415. [Google Scholar] [CrossRef]
- Cascioli, A.; Baratieri, M. Enhanced Thermodynamic Modelling for Hydrothermal Liquefaction. Fuel 2021, 298, 120796. [Google Scholar] [CrossRef]
- Zhu, Y.; Biddy, M.J.; Jones, S.B.; Elliott, D.C.; Schmidt, A.J. Techno-Economic Analysis of Liquid Fuel Production from Woody Biomass via Hydrothermal Liquefaction (HTL) and Upgrading. Appl. Energy 2014, 129, 384–394. [Google Scholar] [CrossRef]
- Pinsky, R.; Sabharwall, P.; Hartvigsen, J.; O’Brien, J. Comparative Review of Hydrogen Production Technologies for Nuclear Hybrid Energy Systems. Prog. Nucl. Energy 2020, 123, 103317. [Google Scholar] [CrossRef]
- Chi, J.; Yu, H. Water Electrolysis Based on Renewable Energy for Hydrogen Production. Cuihua Xuebao/Chin. J. Catal. 2018, 39, 390–394. [Google Scholar] [CrossRef]
- Rand, D.A.J. A Journey on the Electrochemical Road to Sustainability. J. Solid State Electrochem. 2011, 15, 1579–1622. [Google Scholar] [CrossRef]
- Dincer, I.; Acar, C. Review and Evaluation of Hydrogen Production Methods for Better Sustainability. Int. J. Hydrogen Energy 2014, 40, 11094–11111. [Google Scholar] [CrossRef]
- Brien, J.E.O.; Mckellar, M.G.; Stoots, C.M.; Herring, J.S.; Hawkes, G.L. Parametric Study of Large-Scale Production of Syngas Via. Int. J. Hydrogen Energy 2009, 34, 4216–4226. [Google Scholar] [CrossRef]
- Cheng, W.; Lee, S. How Green Are the National Hydrogen Strategies? Sustainability 2022, 14, 1930. [Google Scholar] [CrossRef]
- Newborough, M.; Cooley, G. Developments in the Global Hydrogen Market: The Spectrum of Hydrogen Colours. Fuel Cells Bull. 2020, 11, 16–22. [Google Scholar] [CrossRef]
- Yu, M.; Wang, K.; Vredenburg, H. Insights into Low-Carbon Hydrogen Production Methods: Green, Blue and Aqua Hydrogen. Int. J. Hydrogen Energy 2021, 46, 21261–21273. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, S.X.; Yao, R.; Wu, Y.H.; Qiu, J.S. Progress and Prospects of Hydrogen Production: Opportunities and Challenges. J. Electron. Sci. Technol. 2021, 19, 100080. [Google Scholar] [CrossRef]
- Ibrahim, M.D.; Binofai, F.A.S.; Mohamad, M.O.A. Transition to Low-Carbon Hydrogen Energy System in the UAE: Sector Efficiency and Hydrogen Energy Production Efficiency Analysis. Energies 2022, 15, 6663. [Google Scholar] [CrossRef]
- Revankar, S.T. Nuclear Hydrogen Production. In Storage and Hybridization of Nuclear Energy: Techno-Economic Integration of Renewable and Nuclear Energy; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 49–117. ISBN 9780128139752. [Google Scholar]
- Dibyo, S.; Sunaryo, G.R.; Bakhri, S.; Zuhair; Irianto, I.D. Analysis on Operating Parameter Design to Steam Methane Reforming in Heat Application RDE. J. Phys. Conf. Ser. 2018, 962, 012052. [Google Scholar] [CrossRef]
- Milewski, J.; Kupecki, J.; Szczęśniak, A.; Uzunow, N. Hydrogen Production in Solid Oxide Electrolyzers Coupled with Nuclear Reactors. Int. J. Hydrogen Energy 2021, 46, 35765–35776. [Google Scholar] [CrossRef]
- Naterer, G.F.; Suppiah, S.; Stolberg, L.; Lewis, M.; Wang, Z.; Daggupati, V.; Gabriel, K.; Dincer, I.; Rosen, M.A.; Spekkens, P.; et al. Canada’s Program on Nuclear Hydrogen Production and the Thermochemical Cu-Cl Cycle. Int. J. Hydrogen Energy 2010, 35, 10905–10926. [Google Scholar] [CrossRef]
- Brown, N.R.; Oh, S.; Revankar, S.T.; Vierow, K.; Rodriguez, S.; Cole, R.; Gauntt, R. Simulation of Sulfur-Iodine Thermochemical Hydrogen Production Plant Coupled to High-Temperature Heat Source. Nucl. Technol. 2009, 167, 95–106. [Google Scholar] [CrossRef]
- El-Emam, R.S.; Ozcan, H.; Dincer, I. Comparative Cost Evaluation of Nuclear Hydrogen Production Methods with the Hydrogen Economy Evaluation Program (HEEP). Int. J. Hydrogen Energy 2014, 40, 11168–11177. [Google Scholar] [CrossRef]
- Faisal, N.H.; Prathuru, A.; Ahmed, R.; Rajendran, V.; Hossain, M.; Venkatachalapathy, V.; Katiyar, N.K.; Li, J.; Liu, Y.; Cai, Q.; et al. Application of Thermal Spray Coatings in Electrolysers for Hydrogen Production: Advances, Challenges, and Opportunities. ChemNanoMat 2022, 8, 202200384. [Google Scholar] [CrossRef]
- Agyekum, E.B.; Nutakor, C.; Agwa, A.M.; Kamel, S. A Critical Review of Renewable Hydrogen Production Methods: Factors Affecting Their Scale-Up and Its Role in Future Energy Generation. Membranes 2022, 12, 173. [Google Scholar] [CrossRef]
- Deka, D.J.; Gunduz, S.; Kim, J.; Fitzgerald, T.; Shi, Y.; Co, A.C.; Ozkan, U.S. Hydrogen Production from Water in a Solid Oxide Electrolysis Cell: Effect of Ni Doping on Lanthanum Strontium Ferrite Perovskite Cathodes. Ind. Eng. Chem. Res. 2019, 58, 22497–22505. [Google Scholar] [CrossRef]
- Buttler, A.; Spliethoff, H. Current Status of Water Electrolysis for Energy Storage, Grid Balancing and Sector Coupling via Power-to-Gas and Power-to-Liquids: A Review. Renew. Sustain. Energy Rev. 2018, 82, 2440–2454. [Google Scholar] [CrossRef]
- Li, W.; Tian, H.; Ma, L.; Wang, Y.; Liu, X.; Gao, X. Low-Temperature Water Electrolysis: Fundamentals, Progress, and New Strategies. Mater. Adv. 2022, 3, 5598–5644. [Google Scholar] [CrossRef]
- Schalenbach, M.; Zeradjanin, A.R.; Kasian, O.; Cherevko, S.; Mayrhofer, K.J.J. A Perspective on Low-Temperature Water Electrolysis—Challenges in Alkaline and Acidic Technology. Int. J. Electrochem. Sci. 2018, 13, 1173–1226. [Google Scholar] [CrossRef]
- Ozturk, M.; Dincer, I. A Comprehensive Review on Power-to-Gas with Hydrogen Options for Cleaner Applications. Int. J. Hydrogen Energy 2021, 46, 31511–31522. [Google Scholar] [CrossRef]
- Lohmann-Richters, F.P.; Renz, S.; Lehnert, W.; Müller, M.; Carmo, M. Review—Challenges and Opportunities for Increased Current Density in Alkaline Electrolysis by Increasing the Operating Temperature. J. Electrochem. Soc. 2021, 168, 114501. [Google Scholar] [CrossRef]
- Ahmed, K.W.; Jang, M.J.; Habibpour, S.; Chen, Z.; Fowler, M. NiFeOx and NiFeCoOx Catalysts for Anion Exchange Membrane Water Electrolysis. Electrochem 2022, 3, 843–861. [Google Scholar] [CrossRef]
- Mohamed, A.; Ibrahem, H.; Yang, R.; Kim, K. Optimization of Proton Exchange Membrane Electrolyzer Cell Design Using Machine Learning. Energies 2022, 15, 6657. [Google Scholar] [CrossRef]
- Brauns, J.; Turek, T. Alkaline Water Electrolysis Powered by Renewable Energy: A Review. Processes 2020, 8, 248. [Google Scholar] [CrossRef]
- David, M.; Ocampo-Martínez, C.; Sánchez-Peña, R. Advances in Alkaline Water Electrolyzers: A Review. J. Energy Storage 2019, 23, 392–403. [Google Scholar] [CrossRef]
- Obodo, K.O.; Naphtaly, C.; Ouma, M.; Bessarabov, D. Low-Temperature Water Electrolysis. In Power to Fuel; Academic Press: Cambridge, MA, USA, 2021; pp. 17–50. ISBN 9780128228135. [Google Scholar]
- Zeng, K.; Zhang, D. Recent Progress in Alkaline Water Electrolysis for Hydrogen Production and Applications. Prog. Energy Combust. Sci. 2010, 36, 307–326. [Google Scholar] [CrossRef]
- Guo, Y.; Li, G.; Zhou, J.; Liu, Y. Comparison between Hydrogen Production by Alkaline Water Electrolysis and Hydrogen Production by PEM Electrolysis. IOP Conf. Ser. Earth Environ. Sci. 2019, 371, 0420022. [Google Scholar] [CrossRef]
- Sebbahi, S.; Nabilb, N.; Alaoui-Belghiti, A.; Laasri, S.; Rachidi, S.; Hajjaji, A. Assessment of the Three Most Developed Water Electrolysis Technologies: Alkaline Water Electrolysis, Proton Exchange Membrane and Solid-Oxide Electrolysis. Mater. Today Proc. 2022, 66, 140–145. [Google Scholar] [CrossRef]
- Hu, K.; Fang, J.; Ai, X.; Huang, D.; Zhong, Z.; Yang, X.; Wang, L. Comparative Study of Alkaline Water Electrolysis, Proton Exchange Membrane Water Electrolysis and Solid Oxide Electrolysis through Multiphysics Modeling. Appl. Energy 2022, 312, 118788. [Google Scholar] [CrossRef]
- Ferrero, D.; Lanzini, A.; Santarelli, M.; Leone, P. A Comparative Assessment on Hydrogen Production from Low- and High-Temperature Electrolysis. Int. J. Hydrogen Energy 2013, 38, 3523–3536. [Google Scholar] [CrossRef]
- Vincent, I.; Lee, E.C.; Kim, H.M. Comprehensive Impedance Investigation of Low-Cost Anion Exchange Membrane Electrolysis for Large-Scale Hydrogen Production. Sci. Rep. 2021, 11, 293. [Google Scholar] [CrossRef]
- Wang, L.; Weissbach, T.; Reissner, R.; Ansar, A.; Gago, A.S.; Holdcroft, S.; Friedrich, K.A. High Performance Anion Exchange Membrane Electrolysis Using Plasma-Sprayed, Non-Precious-Metal Electrodes. ACS Appl. Energy Mater. 2019, 2, 7903–7912. [Google Scholar] [CrossRef]
- Kamlungsua, K.; Su, P.C.; Chan, S.H. Hydrogen Generation Using Solid Oxide Electrolysis Cells. Fuel Cells 2020, 20, 644–649. [Google Scholar] [CrossRef]
- Küngas, R. Review—Electrochemical CO2 Reduction for CO Production: Comparison of Low- and High-Temperature Electrolysis Technologies. J. Electrochem. Soc. 2020, 167, 044508. [Google Scholar] [CrossRef]
- Grim, R.G.; Huang, Z.; Guarnieri, M.T.; Iii, J.R.F.; Tao, L.; Schaidle, J.A. Transforming the Carbon Economy: Challenges and Opportunities in the Convergence of Low-Cost. Energy Environ. Sci. 2020, 13, 472–494. [Google Scholar] [CrossRef]
- El-Emam, R.S.; Özcan, H. Comprehensive Review on the Techno-Economics of Sustainable Large-Scale Clean Hydrogen Production. J. Clean. Prod. 2019, 220, 593–609. [Google Scholar] [CrossRef]
- Zhang, K.; Zhao, Y.; He, W.; Zhao, P.; Zhang, D.; He, T.; Wang, Y.; Liu, T. Pr and Mo Co-Doped SrFeO3−δ as an Efficient Cathode for Pure CO2 Reduction Reaction in a Solid Oxide Electrolysis Cell Kun. Energy Technol. 2020, 8, 2000539. [Google Scholar] [CrossRef]
- Kim, J.; Jun, A.; Gwon, O.; Yoo, S.; Liu, M.; Shin, J.; Lim, T.H.; Kim, G. Hybrid-Solid Oxide Electrolysis Cell: A New Strategy for Efficient Hydrogen Production. Nano Energy 2018, 44, 121–126. [Google Scholar] [CrossRef]
- Nechache, A.; Hody, S. Alternative and Innovative Solid Oxide Electrolysis Cell Materials: A Short Review. Renew. Sustain. Energy Rev. 2021, 149, 111322. [Google Scholar] [CrossRef]
- Laguna-Bercero, M.A. Recent Advances in High Temperature Electrolysis Using Solid Oxide Fuel Cells: A Review. J. Power Source 2012, 203, 4–16. [Google Scholar] [CrossRef]
- Nechache, A.; Cassir, M.; Ringuedé, A. Solid Oxide Electrolysis Cell Analysis by Means of Electrochemical Impedance Spectroscopy: A Review. J. Power Source 2014, 258, 164–181. [Google Scholar] [CrossRef]
- Hauch, A.; Küngas, R.; Blennow, P.; Hansen, A.B.; Hansen, J.B.; Mathiesen, B.V.; Mogensen, M.B. Recent Advances in Solid Oxide Cell Technology for Electrolysis. Science 2020, 370, eaba6118. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.P. Development of Lanthanum Strontium Cobalt Ferrite Perovskite Electrodes of Solid Oxide Fuel Cells—A Review. Int. J. Hydrogen Energy 2019, 44, 7448–7493. [Google Scholar] [CrossRef]
- Tian, Y.; Zheng, H.; Zhang, L.; Chi, B.; Pu, J.; Li, J. Direct Electrolysis of CO2 in Symmetrical Solid Oxide Electrolysis Cell Based on La0.6Sr0.4Fe0.8Ni0.2O3-δ Electrode. J. Electrochem. Soc. 2018, 165, F17–F23. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, Y.; Wu, T.; Zhang, W.; Zhu, J.; Li, Z.; Chen, J.; Yu, B.; Wang, J.; Zhang, J. Oxygen Reduction Kinetic Enhancements of Intermediate-Temperature SOFC Cathodes with Novel Nd0.5Sr0.5CoO3-δ/Nd0.8Sr1.2CoO4±δ Heterointerfaces. Nano Energy 2018, 51, 711–720. [Google Scholar] [CrossRef]
- Bernadet, L.; Moncasi, C.; Torrell, M.; Tarancón, A. High-Performing Electrolyte-Supported Symmetrical Solid Oxide Electrolysis Cells Operating under Steam Electrolysis and Co-Electrolysis Modes. Int. J. Hydrogen Energy 2020, 45, 14208–14217. [Google Scholar] [CrossRef]
- Lei, L.; Zhang, J.; Yuan, Z.; Liu, J.; Ni, M.; Chen, F. Progress Report on Proton Conducting Solid Oxide Electrolysis Cells. Adv. Funct. Mater. 2019, 29, 1903805. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, T.; Li, Q.; Wu, W.; Miao, H.; Xu, C.; Wang, W.G. Achieving 360 NL H-1 Hydrogen Production Rate through 30-Cell Solid Oxide Electrolysis Stack with LSCF-GDC Composite Oxygen Electrode. Fuel Cells 2014, 14, 1066–1070. [Google Scholar] [CrossRef]
- Jiang, S.P. Challenges in the Development of Reversible Solid Oxide Cell Technologies: A Mini Review. Asia-Pac. J. Chem. Eng. 2016, 11, 386–391. [Google Scholar] [CrossRef]
- Gruber, M.; Weinbrecht, P.; Biffar, L.; Harth, S.; Trimis, D.; Brabandt, J.; Posdziech, O.; Blumentritt, R. Power-to-Gas through Thermal Integration of High-Temperature Steam Electrolysis and Carbon Dioxide Methanation—Experimental Results. Fuel Process. Technol. 2018, 181, 61–74. [Google Scholar] [CrossRef]
- Jun, A.; Kim, J.; Shin, J.; Kim, G. Achieving High Efficiency and Eliminating Degradation in Solid Oxide Electrochemical Cells Using High Oxygen-Capacity Perovskite. Angew. Chemie Int. Ed. 2016, 55, 12512–12515. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Ding, H.; Zhang, Y.; Ding, Y.; Katiyar, P.; Majumdar, P.K.; He, T.; Ding, D. 3D Self-Architectured Steam Electrode Enabled Efficient and Durable Hydrogen Production in a Proton-Conducting Solid Oxide Electrolysis Cell at Temperatures Lower Than 600 °C. Adv. Sci. 2018, 5, 1800360. [Google Scholar] [CrossRef]
- Vøllestad, E.; Strandbakke, R.; Tarach, M.; Catalán-Martínez, D.; Fontaine, M.L.; Beeaff, D.; Clark, D.R.; Serra, J.M.; Norby, T. Mixed Proton and Electron Conducting Double Perovskite Anodes for Stable and Efficient Tubular Proton Ceramic Electrolysers. Nat. Mater. 2019, 18, 752–759. [Google Scholar] [CrossRef]
- Sapountzi, F.M.; Gracia, J.M.; Weststrate, C.J.; Fredriksson, H.O.A.; Niemantsverdriet, J.W. Electrocatalysts for the Generation of Hydrogen, Oxygen and Synthesis Gas. Prog. Energy Combust. Sci. 2017, 58, 1–35. [Google Scholar] [CrossRef]
- Fabbri, E.; Pergolesi, D.; Traversa, E. Materials Challenges toward Proton-Conducting Oxide Fuel Cells: A Critical Review. Chem. Soc. Rev. 2010, 39, 4355–4369. [Google Scholar] [CrossRef]
- Yang, Z.; Guo, M.; Wang, N.; Ma, C.; Wang, J.; Han, M. A Short Review of Cathode Poisoning and Corrosion in Solid Oxide Fuel Cell. Int. J. Hydrogen Energy 2017, 42, 24948–24959. [Google Scholar] [CrossRef]
- Jiang, S.P.; Chen, X. Chromium Deposition and Poisoning of Cathodes of Solid Oxide Fuel Cells—A Review. Int. J. Hydrogen Energy 2014, 39, 505–531. [Google Scholar] [CrossRef]
- Duan, C.; Huang, J.; Sullivan, N.; O’Hayre, R. Proton-Conducting Oxides for Energy Conversion and Storage. Appl. Phys. Rev. 2020, 7, 011314. [Google Scholar] [CrossRef]
- Kim, J.; Sengodan, S.; Kwon, G.; Ding, D.; Shin, J.; Liu, M.; Kim, G. Triple-Conducting Layered Perovskites as Cathode Materials for Proton-Conducting Solid Oxide Fuel Cells. ChemSusChem 2014, 7, 2811–2815. [Google Scholar] [CrossRef] [PubMed]
- Callejas, J.F.; Read, C.G.; Popczun, E.J.; McEnaney, J.M.; Schaak, R.E. Nanostructured Co2P Electrocatalyst for the Hydrogen Evolution Reaction and Direct Comparison with Morphologically Equivalent CoP. Chem. Mater. 2015, 27, 3769–3774. [Google Scholar] [CrossRef]
- Kibsgaard, J.; Jaramillo, T.F. Molybdenum Phosphosulfide: An Active, Acid-Stable, Earth-Abundant Catalyst for the Hydrogen Evolution Reaction. Angew. Chemie. Int. Ed. 2014, 53, 14433–14437. [Google Scholar] [CrossRef] [PubMed]
- Naterer, G.F.; Dincer, I.; Zamfirescu, C. Hydrogen Production from Nuclear Energy, 1st ed.; Springer: London, UK, 2013; ISBN 9781447149378. [Google Scholar]
- Develos-Bagarinao, K.; De Vero, J.; Kishimoto, H.; Ishiyama, T.; Yamaji, K.; Horita, T.; Yokokawa, H. Multilayered LSC and GDC: An Approach for Designing Cathode Materials with Superior Oxygen Exchange Properties for Solid Oxide Fuel Cells. Nano Energy 2018, 52, 369–380. [Google Scholar] [CrossRef]
- Cebollero, J.A.; Lahoz, R.; Laguna-Bercero, M.A.; Peña, J.I.; Larrea, A.; Orera, V.M. Characterization of Laser-Processed Thin Ceramic Membranes for Electrolyte-Supported Solid Oxide Fuel Cells. Int. J. Hydrogen Energy 2017, 42, 13939–13948. [Google Scholar] [CrossRef]
- Zhou, Z.; Nadimpalli, V.K.; Pedersen, D.B.; Esposito, V. Degradation Mechanisms of Metal-Supported Solid Oxide Cells and Countermeasures: A Review. Materials 2021, 14, 3139. [Google Scholar] [CrossRef]
- Schefold, J.; Brisse, A.; Poepke, H. 23,000 H Steam Electrolysis with an Electrolyte Supported Solid Oxide Cell. Int. J. Hydrogen Energy 2017, 42, 13415–13426. [Google Scholar] [CrossRef]
- Yang, C.; Yang, Z.; Jin, C.; Liu, M.; Chen, F. High Performance Solid Oxide Electrolysis Cells Using Pr 0.8Sr1.2(Co,Fe)0.8Nb0.2O4+δ-Co-Fe Alloy Hydrogen Electrodes. Int. J. Hydrogen Energy 2013, 38, 11202–11208. [Google Scholar] [CrossRef]
- Ilbas, M.; Kumuk, B. Numerical Modelling of a Cathode-Supported Solid Oxide Fuel Cell (SOFC) in Comparison with an Electrolyte-Supported Model. J. Energy Inst. 2019, 92, 682–692. [Google Scholar] [CrossRef]
- Kusnezoff, M.; Trofimenko, N.; Müller, M.; Michaelis, A. Influence of Electrode Design and Contacting Layers on Performance of Electrolyte Supported SOFC/SOEC Single Cells. Materials 2016, 9, 906. [Google Scholar] [CrossRef]
- Zou, G.; Feng, W.; Song, Y.; Wang, G. Recent Advances in Anode Materials of Solid Oxide Electrolysis Cells. J. Electrochem. 2022. [Google Scholar] [CrossRef]
- Mendoza, R.M.; Mora, J.M.; Cervera, R.B.; Chuang, P.A. Experimental and Analytical Study of an Anode-Supported Solid Oxide Electrolysis Cell. Chem. Eng. Technol. 2020, 43, 2350–2358. [Google Scholar] [CrossRef]
- Gondolini, A.; Mercadelli, E.; Sanson, A. Single Step Anode-Supported Solid Oxide Electrolyzer Cell. J. Eur. Ceram. Soc. 2015, 35, 4617–4621. [Google Scholar] [CrossRef]
- Nechache, A.; Mansuy, A.; Petitjean, M.; Mougin, J.; Mauvy, F.; Boukamp, B.A.; Cassir, M.; Ringuedé, A. Diagnosis of a Cathode-Supported Solid Oxide Electrolysis Cell by Electrochemical Impedance Spectroscopy. Electrochim. Acta 2016, 210, 596–605. [Google Scholar] [CrossRef]
- Nechache, A.; Boukamp, B.A.; Cassir, M.; Ringuedé, A. Premature Degradation Study of a Cathode-Supported Solid Oxide Electrolysis Cell. J. Solid State Electrochem. 2019, 23, 109–123. [Google Scholar] [CrossRef]
- Hjalmarsson, P.; Sun, X.; Liu, Y.L.; Chen, M. Durability of High Performance Ni-Yttria Stabilized Zirconia Supported Solid Oxide Electrolysis Cells at High Current Density. J. Power Source 2014, 262, 316–322. [Google Scholar] [CrossRef]
- Leonard, K.; Okuyama, Y.; Takamura, Y.; Lee, Y.S.; Miyazaki, K.; Ivanova, M.E.; Meulenberg, W.A.; Matsumoto, H. Efficient Intermediate-Temperature Steam Electrolysis with Y:SrZrO3-SrCeO3 and Y:BaZrO3-BaCeO3 Proton Conducting Perovskites. J. Mater. Chem. A 2018, 6, 19113–19124. [Google Scholar] [CrossRef]
- Chelmehsara, M.E.; Mahmoudimehr, J. Techno-Economic Comparison of Anode-Supported, Cathode-Supported, and Electrolyte-Supported SOFCs. Int. J. Hydrogen Energy 2018, 43, 15521–15530. [Google Scholar] [CrossRef]
- Hagen, A.; Caldogno, R.; Capotondo, F.; Sun, X. Metal Supported Electrolysis Cells. Energies 2022, 15, 2045. [Google Scholar] [CrossRef]
- Tucker, M.C. Progress in Metal-Supported Solid Oxide Electrolysis Cells: A Review. Int. J. Hydrogen Energy 2020, 45, 24203–24218. [Google Scholar] [CrossRef]
- Küngas, R.; Blennow, P.; Heiredal-Clausen, T.; Nørby, T.H.; Rass-Hansen, J.; Moses, P.G. Lifetime Capacity—An Important Performance Metric for SOEC Stacks. ECS Trans. 2019, 91, 2601–2611. [Google Scholar] [CrossRef]
- Lei, L.; Tao, Z.; Wang, X.; Lemmon, J.P.; Chen, F. Intermediate-Temperature Solid Oxide Electrolysis Cells with Thin Proton-Conducting Electrolyte and Robust Air Electrode. J. Mater. Chem. A 2017, 5, 22945–22951. [Google Scholar] [CrossRef]
- Lenser, C.; Udomsilp, D.; Menzler, N.H.; Holtappels, P.; Fujisaki, T.; Kwati, L.; Matsumoto, H.; Sabato, A.G.; Smeacetto, F.; Chrysanthou, A.; et al. Solid Oxide Fuel and Electrolysis Cells. In Advanced Ceramics for Energy Conversion and Storage; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; pp. 387–547. ISBN 9780081027264. [Google Scholar]
- Jamil, S.M.; Othman, M.H.D.; Rahman, M.A.; Jaafar, J.; Ismail, A.F.; Li, K. Recent Fabrication Techniques for Micro-Tubular Solid Oxide Fuel Cell Support: A Review. J. Eur. Ceram. Soc. 2015, 35, 1–22. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C. Technological Development of Hydrogen Production by Solid Oxide Electrolyzer Cell (SOEC). Int. J. Hydrogen Energy 2008, 33, 2337–2354. [Google Scholar] [CrossRef]
- Hino, R.; Haga, K.; Aita, H.; Sekita, K. R&D on Hydrogen Production by High-Temperature Electrolysis of Steam. Nucl. Eng. Des. 2004, 233, 363–375. [Google Scholar] [CrossRef]
- Udomsilp, D.; Lenser, C.; Guillon, O.; Menzler, N.H. Performance Benchmark of Planar Solid Oxide Cells Based on Material Development and Designs. Energy Technol. 2021, 9, 2001062. [Google Scholar] [CrossRef]
- Lang, M.; Raab, S.; Lemcke, M.S.; Bohn, C.; Pysik, M. Long-Term Behavior of a Solid Oxide Electrolyzer (SOEC) Stack▴. Fuel Cells 2020, 20, 690–700. [Google Scholar] [CrossRef]
- Toriumi, H.; Jeong, S.; Kitano, S.; Habazaki, H.; Aoki, Y. Enhanced Performance of Protonic Solid Oxide Steam Electrolysis Functional Layer. ACS Omega 2022, 7, 9944–9950. [Google Scholar] [CrossRef] [PubMed]
- Jhong, H.R.M.; Ma, S.; Kenis, P.J. Electrochemical Conversion of CO2 to Useful Chemicals: Current Status, Remaining Challenges, and Future Opportunities. Curr. Opin. Chem. Eng. 2013, 2, 191–199. [Google Scholar] [CrossRef]
- Theis, L.; Babaie, O.; Lyck, A.; Frandsen, L.; Valdemar, J.; Thorvald, H.; Hauch, A.; Kær, K.; Simon, S.; Graves, C.; et al. Electrothermally Balanced Operation of Solid Oxide Electrolysis Cells. J. Power Source 2022, 523, 231040. [Google Scholar] [CrossRef]
- Zhang, Z.; Guan, C.; Xie, L.; Wang, J. Design and Analysis of a Novel Opposite Trapezoidal Flow Channel for Solid Oxide Electrolysis Cell Stack. Energies 2023, 16, 159. [Google Scholar] [CrossRef]
- Minary-Jolandan, M. Formidable Challenges in Additive Manufacturing of Solid Oxide Electrolyzers (SOECs) and Solid Oxide Fuel Cells (SOFCs) for Electrolytic Hydrogen Economy toward Global Decarbonization. Ceramics 2022, 5, 761–779. [Google Scholar] [CrossRef]
- Herradon, C.; Le, L.; Meisel, C.; Huang, J.; Chmura, C.; Kim, Y.D.; Cadigan, C.; O’Hayre, R.; Sullivan, N.P. Proton-Conducting Ceramics for Water Electrolysis and Hydrogen Production at Elevated Pressure. Front. Energy Res. 2022, 10, 1546. [Google Scholar] [CrossRef]
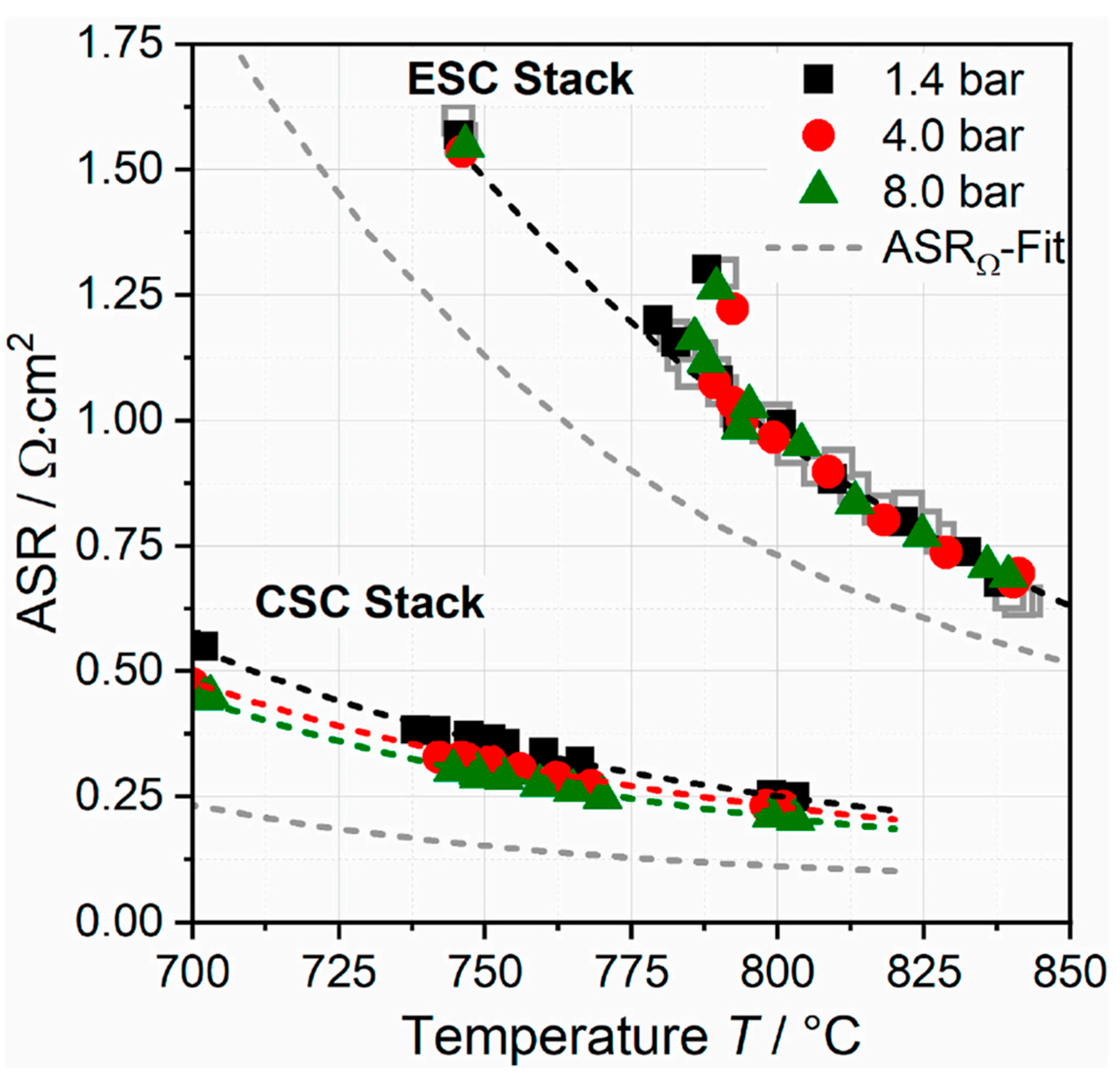
- Riedel, M.; Heddrich, M.P.; Ansar, A.; Fang, Q.; Blum, L.; Friedrich, K.A. Pressurized Operation of Solid Oxide Electrolysis Stacks: An Experimental Comparison of the Performance of 10-Layer Stacks with Fuel Electrode and Electrolyte Supported Cell Concepts. J. Power Source 2020, 475, 228682. [Google Scholar] [CrossRef]
- Chen, K.; Ai, N.; O’Donnell, K.M.; Jiang, S.P. Highly Chromium Contaminant Tolerant BaO Infiltrated La 0.6Sr0.4Co0.2Fe0.8O3−δ Cathodes for Solid Oxide Fuel Cells. Phys. Chem. Chem. Phys. 2015, 17, 4870–4874. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Ebbesen, S.D.; Mogensen, M.B. Carbon Deposition in Solid Oxide Cells during Co-Electrolysis of H2O and CO2. J. Electrochem. Soc. 2014, 161, F337–F343. [Google Scholar] [CrossRef]
- The, D.; Grieshammer, S.; Schroeder, M.; Martin, M.; Al Daroukh, M.; Tietz, F.; Schefold, J.; Brisse, A. Microstructural Comparison of Solid Oxide Electrolyser Cells Operated for 6100 h and 9000 H. J. Power Source 2015, 275, 901–911. [Google Scholar] [CrossRef]
- Léon, A.; Micero, A.; Ludwig, B.; Brisse, A. Effect of Scaling-up on the Performance and Degradation of Long-Term Operated Electrolyte Supported Solid Oxide Cell, Stack and Module in Electrolysis Mode. J. Power Source 2021, 510, 230346. [Google Scholar] [CrossRef]
- Tucker, M.C. Progress in Metal-Supported Solid Oxide Fuel Cells: A Review. J. Power Source 2010, 195, 4570–4582. [Google Scholar] [CrossRef]
- Chi, Y.; Yokoo, K.; Nakajima, H.; Ito, K.; Lin, J.; Song, Y. Optimizing the Homogeneity and Efficiency of a Solid Oxide Electrolysis Cell Based on Multiphysics Simulation and Data-Driven Surrogate Model. J. Power Source 2023, 562, 232760. [Google Scholar] [CrossRef]
- Cui, T.; Xiao, G.; Yan, H.; Zhang, Y.; Wang, J. -Q. Numerical Simulation and Analysis of the Thermal Stresses of a Planar Solid Oxide Electrolysis Cell. Int. J. Green Energy 2023, 20, 432–444. [Google Scholar] [CrossRef]
- Graves, C.; Ebbesen, S.D.; Jensen, S.H.; Simonsen, S.B.; Mogensen, M.B. Eliminating Degradation in Solid Oxide Electrochemical Cells by Reversible Operation. Nat. Mater. 2015, 14, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Hartvigsen, J.; Elangovan, S.; O’Brien, J.; Stoots, C.; Herring, J.S.; Lessing, P. Operation and Analysis of Sofcs in Steam Electrolysis Mode. In Proceedings of the 6th European Solid Oxide Fuel Cell Forum, Lucerne, Switzerland, 28 June–2 July 2004; pp. 378–387. [Google Scholar]
- Tong, X.; Ovtar, S.; Brodersen, K.; Hendriksen, P.V.; Chen, M. A 4×4 cm2 Nanoengineered Solid Oxide Electrolysis Cell for Efficient and Durable Hydrogen Production. ACS Appl. Mater. Interfaces 2019, 11, 25996–26004. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Sun, X.; Chatzichristodoulou, C.; Koch, S.; Hendriksen, P.V.; Mogensen, M.B. Thermoneutral Operation of Solid Oxide Electrolysis Cells in Potentiostatic Mode. ECS Trans. 2017, 78, 3077–3088. [Google Scholar] [CrossRef]
- Hauch, A.; Marchese, M.; Lanzini, A.; Graves, C. Re-Activation of Degraded Nickel Cermet Anodes—Nano-Particle Formation via Reverse Current Pulses. J. Power Source 2018, 377, 110–120. [Google Scholar] [CrossRef]
- Monaco, F.; Ferreira-Sanchez, D.; Hubert, M.; Morel, B.; Montinaro, D.; Grolimund, D.; Laurencin, J. Oxygen Electrode Degradation in Solid Oxide Cells Operating in Electrolysis and Fuel Cell Modes: LSCF Destabilization and Interdiffusion at the Electrode/Electrolyte Interface. Int. J. Hydrogen Energy 2021, 46, 31533–31549. [Google Scholar] [CrossRef]
- Kim, S.; Woo, D.; Lee, D.; Lee, J.; Sung, H.; Zubair, M.; Eun, J.; Lee, S.; Joo, S.; Song, R.; et al. Microstructure Tailoring of Solid Oxide Electrolysis Cell Air Electrode to Boost Performance and Long-Term Durability. Chem. Eng. J. 2021, 410, 128318. [Google Scholar] [CrossRef]
- Han, D.; Okumura, Y.; Nose, Y.; Uda, T. Synthesis of La1−xSrxSc1−yFeyO3−δ (LSSF) and Measurement of Water Content in LSSF, LSCF and LSC Hydrated in Wet Artificial Air at 300 °C. Solid State Ion. 2010, 181, 1601–1606. [Google Scholar] [CrossRef]
- Ovtar, S.; Tong, X.; Bentzen, J.J.; Thydén, K.T.S.; Simonsen, S.B.; Chen, M. Boosting the Performance and Durability of Ni/YSZ Cathode for Hydrogen Production at High Current Densities: Via Decoration with Nano-Sized Electrocatalysts. Nanoscale 2019, 11, 4394–4406. [Google Scholar] [CrossRef]
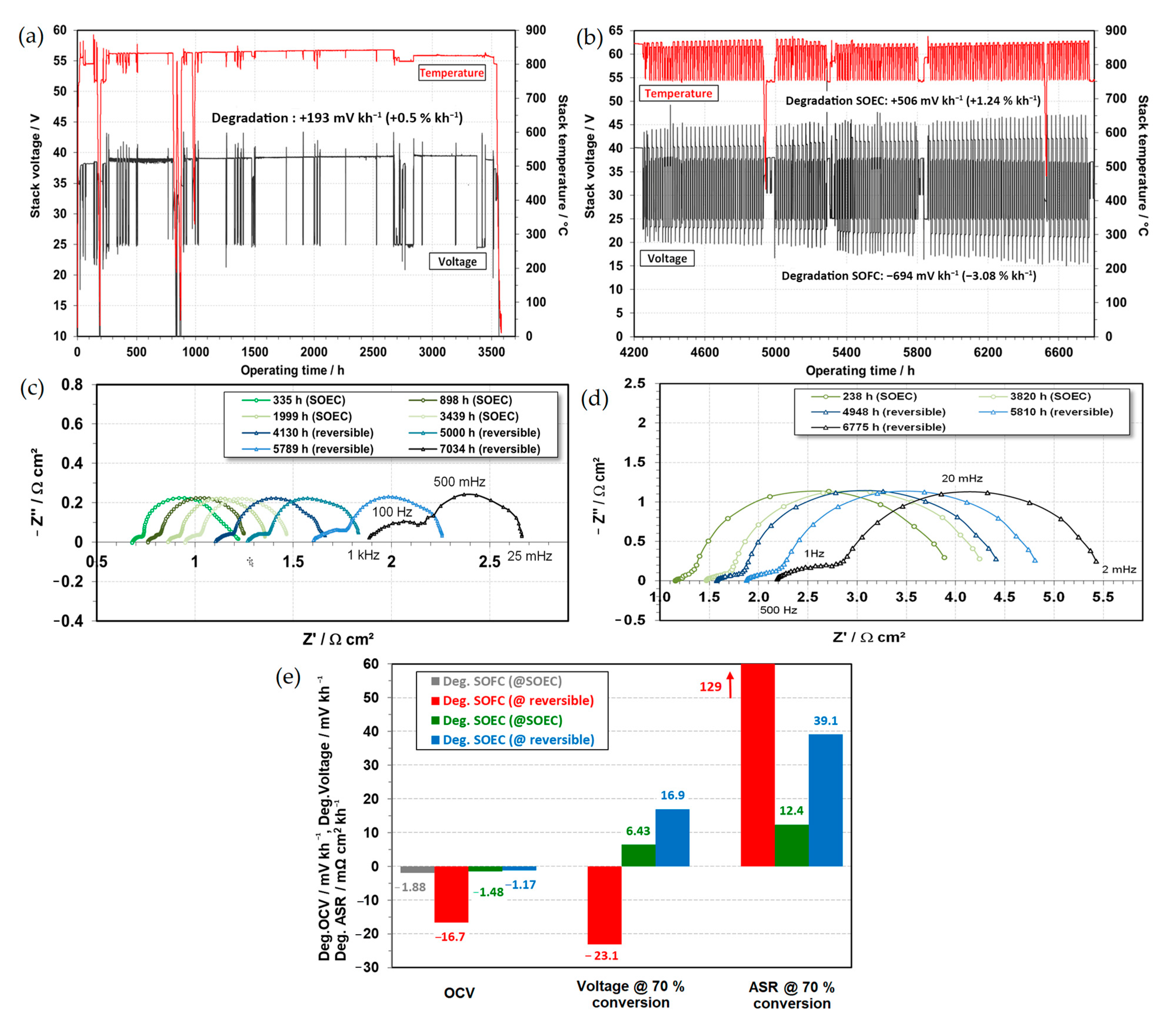
- Chen, K.; Jiang, S.P. Review—Materials Degradation of Solid Oxide Electrolysis Cells. J. Electrochem. Soc. 2016, 163, F3070–F3083. [Google Scholar] [CrossRef]
- Wang, Y.; Li, W.; Ma, L.; Li, W.; Liu, X. Degradation of Solid Oxide Electrolysis Cells: Phenomena, Mechanisms, and Emerging Mitigation Strategies—A Review. J. Mater. Sci. Technol. 2020, 55, 35–55. [Google Scholar] [CrossRef]
- Moçoteguy, P.; Brisse, A. A Review and Comprehensive Analysis of Degradation Mechanisms of Solid Oxide Electrolysis Cells. Int. J. Hydrogen Energy 2013, 38, 15887–15902. [Google Scholar] [CrossRef]
- Wolf, S.E.; Vibhu, V.; Tröster, E.; Vinke, I.C.; Eichel, R.-A.; Aart, L.G.J. (Bert) de Steam Electrolysis vs. Co-Electrolysis: Mechanistic Studies of Long-Term Solid Oxide Electrolysis Cells Stephanie. Energies 2022, 15, 5449. [Google Scholar] [CrossRef]
- Park, B.; Zhang, Q.; Voorhees, P.W.; Barnett, S.A. Conditions for Stable Operation of Solid Oxide Electrolysis Cells: Oxygen Electrode Effects. Energy Environ. Sci. 2019, 12, 3053–3062. [Google Scholar] [CrossRef]
- Königshofer, B.; Höber, M.; Nusev, G.; Boškoski, P.; Juričić, Ð.; Margaritis, N.; Hochenauer, C.; Subotić, V. Towards Strategies to Mitigate Degradation and Regenerate Performance of a Solid Oxide Electrolyzer during Co-Electrolysis Operation. J. Power Source 2023, 556, 232404. [Google Scholar] [CrossRef]
- Königshofer, B.; Höber, M.; Nusev, G.; Boškoski, P.; Hochenauer, C.; Subotić, V. Accelerated Degradation for Solid Oxide Electrolysers: Analysis and Prediction of Performance for Varying Operating Environments. J. Power Source 2022, 523, 230982. [Google Scholar] [CrossRef]
- Ebbesen, S.D.; Graves, C.; Hauch, A.; Jensen, S.H.; Mogensen, M. Poisoning of Solid Oxide Electrolysis Cells by Impurities. J. Electrochem. Soc. 2010, 157, B1419. [Google Scholar] [CrossRef]
- Papaefthimiou, V.; Niakolas, D.K.; Paloukis, F.; Teschner, D.; Knop-Gericke, A.; Haevecker, M.; Zafeiratos, S. Operando Observation of Nickel/Ceria Electrode Surfaces during Intermediate Temperature Steam Electrolysis. J. Catal. 2017, 352, 305–313. [Google Scholar] [CrossRef]
- Chen, D.; Mewafy, B.; Paloukis, F.; Zhong, L.; Papaefthimiou, V.; Dintzer, T.; Papazisi, K.M.; Balomenou, S.P.; Tsiplakides, D.; Teschner, D.; et al. Revising the Role of Chromium on the Surface of Perovskite Electrodes: Poison or Promoter for the Solid Oxide Electrolysis Cell Performance? J. Catal. 2020, 381, 520–529. [Google Scholar] [CrossRef]
- Tao, Y.; Ebbesen, S.D.; Mogensen, M.B. Degradation of Solid Oxide Cells during Co-Electrolysis of Steam and Carbon Dioxide at High Current Densities. J. Power Source 2016, 328, 452–462. [Google Scholar] [CrossRef]
- Tietz, F.; Sebold, D.; Brisse, A.; Schefold, J. Degradation Phenomena in a Solid Oxide Electrolysis Cell after 9000 h of Operation. J. Power Source 2013, 223, 129–135. [Google Scholar] [CrossRef]
- Zohourian, R.; Merkle, R.; Raimondi, G.; Maier, J. Mixed-Conducting Perovskites as Cathode Materials for Protonic Ceramic Fuel Cells: Understanding the Trends in Proton Uptake. Adv. Funct. Mater. 2018, 28, 1801241. [Google Scholar] [CrossRef]
- Zhang, J.; Lenser, C.; Menzler, N.H.; Guillon, O. Comparison of Solid Oxide Fuel Cell (SOFC) Electrolyte Materials for Operation at 500 °C. Solid State Ion. 2020, 344, 115138. [Google Scholar] [CrossRef]
- Kreuer, K.D. Proton-Conducting Oxides. Annu. Rev. Mater. Res. 2003, 33, 333–359. [Google Scholar] [CrossRef]
- Peltzer, D.; Múnera, J.; Cornaglia, L. Study of the Sorption Properties of Alkali Zirconate-Based Sorbents at High Temperature in the Presence of Water and Low CO2 Concentration. J. Alloys Compd. 2022, 895, 162419. [Google Scholar] [CrossRef]
- Hossain, M.K.; Hasan, S.M.K.; Hossain, M.I.; Das, R.C.; Bencherif, H.; Rubel, M.H.K.; Rahman, M.F.; Emrose, T.; Hashizume, K. A Review of Applications, Prospects, and Challenges of Proton-Conducting Zirconates in Electrochemical Hydrogen Devices. Nanomaterials 2022, 12, 3581. [Google Scholar] [CrossRef]
- Tanaka, M.; Ohshima, T. Recovery of Hydrogen from Gas Mixture by an Intermediate-Temperature Type Proton Conductor. Fusion Eng. Des. 2010, 85, 1038–1043. [Google Scholar] [CrossRef]
- Pergolesi, D.; Fabbri, E.; D’Epifanio, A.; Di Bartolomeo, E.; Tebano, A.; Sanna, S.; Licoccia, S.; Balestrino, G.; Traversa, E. High Proton Conduction in Grain-Boundary-Free Yttrium-Doped Barium Zirconate Films Grown by Pulsed Laser Deposition. Nat. Mater. 2010, 9, 846–852. [Google Scholar] [CrossRef]
- Assabumrungrat, S.; Sangtongkitcharoen, W.; Laosiripojana, N.; Arpornwichanop, A.; Charojrochkul, S.; Praserthdam, P. Effects of Electrolyte Type and Flow Pattern on Performance of Methanol-Fuelled Solid Oxide Fuel Cells. J. Power Source 2005, 148, 18–23. [Google Scholar] [CrossRef]
- Hossain, M.K.; Chanda, R.; El-Denglawey, A.; Emrose, T.; Rahman, M.T.; Biswas, M.C.; Hashizume, K. Recent Progress in Barium Zirconate Proton Conductors for Electrochemical Hydrogen Device Applications: A Review. Ceram. Int. 2021, 47, 23725–23748. [Google Scholar] [CrossRef]
- Choi, S.; Davenport, T.C.; Haile, S.M. Protonic Ceramic Electrochemical Cells for Hydrogen Production and Electricity Generation: Exceptional Reversibility, Stability, and Demonstrated Faradaic Efficiency. Energy Environ. Sci. 2019, 12, 206–215. [Google Scholar] [CrossRef]
- Leng, Z.; Huang, Z.; Zhou, X.; Zhang, B.; Bai, H.; Zhou, J.; Wang, S. The Effect of Sintering Aids on BaCe0.7Zr0.1Y0.1Yb0.1O3-δ as the Electrolyte of Proton-Conducting Solid Oxide Electrolysis Cells. Int. J. Hydrogen Energy 2022, 47, 33861–33871. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhou, W.; Ran, R.; Chen, Y.; Shao, Z.; Liu, M. Promotion of Oxygen Reduction by Exsolved Silver Nanoparticles on a Perovskite Scaffold for Low-Temperature Solid Oxide Fuel Cells. Nano Lett. 2016, 16, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, Q.; Luo, J.L. Highly Stable and Efficient Catalyst with in Situ Exsolved Fe-Ni Alloy Nanospheres Socketed on an Oxygen Deficient Perovskite for Direct CO2 Electrolysis. ACS Catal. 2016, 6, 6219–6228. [Google Scholar] [CrossRef]
- Qin, Q.; Wu, G.; Chen, S.; Doherty, W.; Xie, K.; Wu, Y. Perovskite Titanate Cathode Decorated by In-Situ Grown Iron Nanocatalyst with Enhanced Electrocatalytic Activity for High-Temperature Steam Electrolysis. Electrochim. Acta 2014, 127, 215–227. [Google Scholar] [CrossRef]
- Amaya-Dueñas, D.M.; Riegraf, M.; Nenning, A.; Opitz, A.K.; Costa, R.; Friedrich, K.A. Operational Aspects of a Perovskite Chromite-Based Fuel Electrode in Solid Oxide Electrolysis Cells (SOEC). ACS Appl. Energy Mater. 2022, 5, 8143–8156. [Google Scholar] [CrossRef]
- Vibhu, V.; Vinke, I.C.; Zaravelis, F.; Neophytides, S.G.; Niakolas, D.K.; Eichel, R.; Haart, L.G.J.B. De Cell Composed of Mo-Au-Ni / GDC Fuel Electrode and LSCF Oxygen Electrode during High Temperature Steam Electrolysis. Energies 2022, 15, 2726. [Google Scholar] [CrossRef]
- Dogdibegovic, E.; Shen, F.; Wang, R.; Robinson, I.; Lau, G.Y.; Tucker, M.C. Progress in Metal-Supported Solid Oxide Fuel Cells and Electrolyzers with Symmetric Metal Supports and Infiltrated Electrodes. ECS Trans. 2019, 91, 877–885. [Google Scholar] [CrossRef]
- Lee, D.; Lee, Y.L.; Hong, W.T.; Biegalski, M.D.; Morgan, D.; Shao-Horn, Y. Oxygen Surface Exchange Kinetics and Stability of (La,Sr)2CoO4±δ/La1-XSrxMO3-δ (M= Co and Fe) Hetero-Interfaces at Intermediate Temperatures. J. Mater. Chem. A 2015, 3, 2144–2157. [Google Scholar] [CrossRef]
- Feng, Z.; Yacoby, Y.; Gadre, M.J.; Lee, Y.; Hong, W.T.; Zhou, H.; Biegalski, M.D.; Christen, H.M.; Adler, S.B.; Morgan, D.; et al. Anomalous Interface and Surface Strontium Segregation in (La1−ySry)2CoO4±δ/La1−xSrxCoO3−δ Heterostructured Thin Films. J. Phys. Chem. Lett. 2014, 5, 1027–1034. [Google Scholar] [CrossRef]
- Xi, X.; Fan, Y.; Zhang, J.; Luo, J.L.; Fu, X.Z. In Situ Construction of Hetero-Structured Perovskite Composites with Exsolved Fe and Cu Metallic Nanoparticles as Efficient CO2 Reduction Electrocatalysts for High Performance Solid Oxide Electrolysis Cells. J. Mater. Chem. A 2022, 10, 2509–2518. [Google Scholar] [CrossRef]
- Biswas, S.; Kaur, G.; Paul, G.; Giddey, S. A Critical Review on Cathode Materials for Steam Electrolysis in Solid Oxide Electrolysis. Int. J. Hydrogen Energy 2023, in press. [CrossRef]
- Wang, Y.; Wang, Z.; Jin, C.; Li, C.; Li, X.; Li, Y.; Yang, R.; Liu, M. Enhanced Overall Water Electrolysis on a Bifunctional Perovskite Oxide through Interfacial Engineering. Electrochim. Acta 2019, 318, 120–129. [Google Scholar] [CrossRef]
- Chen, T.; Liu, M.; Yuan, C.; Zhou, Y.; Ye, X.; Zhan, Z.; Xia, C.; Wang, S. High Performance of Intermediate Temperature Solid Oxide Electrolysis Cells Using Nd2NiO4+δ Impregnated Scandia Stabilized Zirconia Oxygen Electrode. J. Power Source 2015, 276, 1–6. [Google Scholar] [CrossRef]
- Zhang, X.; Ye, L.; Hu, J.; Li, J.; Jiang, W.; Tseng, C.J.; Xie, K. Perovskite LSCM Impregnated with Vanadium Pentoxide for High Temperature Carbon Dioxide Electrolysis. Electrochim. Acta 2016, 212, 32–40. [Google Scholar] [CrossRef]
- Liu, Q.; Li, R.; Feng, W.; Li, J.; Zhang, X.; Lv, H.; Shen, Y.; Song, Y.; Wang, G.; Bao, X. Promoting High-Temperature Oxygen Evolution Reaction via Infiltration of PrCoO3−δ Nanoparticles. ACS Appl. Energy Mater. 2022, 5, 11604–11612. [Google Scholar] [CrossRef]
- Zhang, C.; Lu, B.; Xiong, H.; Lin, C.; Fang, L.; Fu, J.; Deng, D.; Fan, X.; Li, Y.; Wu, Q.-H. Cobalt-Based Perovskite Electrodes for Solid Oxide Electrolysis Cells. Inorganics 2022, 10, 187. [Google Scholar] [CrossRef]
- Harrison, C.M.; Slater, P.R.; Steinberger-Wilckens, R. Lanthanum Nickelates and Their Application in Solid Oxide Cells—The LaNi1-XFexO3 System and Other ABO3-Type Nickelates. Solid State Ion. 2021, 373, 115799. [Google Scholar] [CrossRef]
- Prasopchokkul, P.; Seeharaj, P.; Kim-Lohsoontorn, P. Ba0.5Sr0.5(Co0.8Fe0.2)1-XTaxO3-δ Perovskite Anode in Solid Oxide Electrolysis Cell for Hydrogen Production from High-Temperature Steam Electrolysis. Int. J. Hydrogen Energy 2021, 46, 7023–7036. [Google Scholar] [CrossRef]
- Zhu, C.; Hou, L.; Li, S.; Gan, L.; Xie, K. Efficient Carbon Dioxide Electrolysis with Metal Nanoparticles Loaded La0.75Sr0.25Cr0.5Mn0.5O3-δ Cathodes. J. Power Source 2017, 363, 177–184. [Google Scholar] [CrossRef]
- Vibhu, V.; Yildiz, S.; Vinke, I.C.; Eichel, R.-A.; Bassat, J.-M.; de Haart, L.G.J. High Performance LSC Infiltrated LSCF Oxygen Electrode for High Temperature Steam Electrolysis Application. J. Electrochem. Soc. 2019, 166, F102–F108. [Google Scholar] [CrossRef]
- Zheng, H.; Tian, Y.; Zhang, L.; Chi, B.; Pu, J.; Jian, L. La0.8Sr0.2Co0.8Ni0.2O3-δ Impregnated Oxygen Electrode for H2O/CO2 Co-Electrolysis in Solid Oxide Electrolysis Cells. J. Power Source 2018, 383, 93–101. [Google Scholar] [CrossRef]
- Li, Z.; Shan, P.; Tang, W.; Ni, Q.; Qian, B.; Wang, S.; Zheng, Y.; Ge, L.; Chen, H.; Zhang, C. Enhancing the Catalytic Activity and Stability of the Pr2NiO4+δ Ruddlesden-Popper Perovskite Air Electrode for High-Temperature Steam Electrolysis with Barium Doping. J. Alloys Compd. 2023, 932, 167646. [Google Scholar] [CrossRef]
- Pei, K.; Zhou, Y.; Xu, K.; Zhang, H.; Ding, Y.; Zhao, B.; Yuan, W.; Sasaki, K.; Choi, Y.M.; Chen, Y.; et al. Surface Restructuring of a Perovskite-Type Air Electrode for Reversible Protonic Ceramic Electrochemical Cells. Nat. Commun. 2022, 13, 2207. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Kee, R.; Zhu, H.; Sullivan, N.; Zhu, L.; Bian, L.; Jennings, D.; O’Hayre, R. Highly Efficient Reversible Protonic Ceramic Electrochemical Cells for Power Generation and Fuel Production. Nat. Energy 2019, 4, 230–240. [Google Scholar] [CrossRef]
- Papac, M.; Stevanović, V.; Zakutayev, A.; O’Hayre, R. Triple Ionic–Electronic Conducting Oxides for next-Generation Electrochemical Devices. Nat. Mater. 2021, 20, 301–313. [Google Scholar] [CrossRef]
- Kasyanova, A.V.; Tarutina, L.R.; Rudenko, A.O.; Lyagaeva, J.G.; Medvedev, D.A. Ba(Ce,Zr)O3-Based Electrodes for Protonic Ceramic Electrochemical Cells: Towards Highly Compatible Functionality and Triple-Conducting Behaviour. Russ. Chem. Rev. 2020, 89, 667–692. [Google Scholar] [CrossRef]
- Ding, H.; Wu, W.; Jiang, C.; Ding, Y.; Bian, W.; Hu, B.; Singh, P.; Orme, C.J.; Wang, L.; Zhang, Y.; et al. Self-Sustainable Protonic Ceramic Electrochemical Cells Using a Triple Conducting Electrode for Hydrogen and Power Production. Nat. Commun. 2020, 11, 1907. [Google Scholar] [CrossRef]
- Wang, N.; Toriumi, H.; Sato, Y.; Tang, C.; Nakamura, T.; Amezawa, K.; Sho Kitano, H.; Habazaki, H.; Aok, Y. La0.8 Sr0.2Co1-xNixO3-δ as the Efficient Triple Conductor Air Electrode for Protonic Ceramic Cells. ACS Appl. Energy Mater. 2021, 4, 554–563. [Google Scholar] [CrossRef]
- Mah, J.C.W.; Muchtar, A.; Somalu, M.R.; Ghazali, M.J. Metallic Interconnects for Solid Oxide Fuel Cell: A Review on Protective Coating and Deposition Techniques. Int. J. Hydrogen Energy 2017, 42, 9219–9229. [Google Scholar] [CrossRef]
- Mahato, N.; Banerjee, A.; Gupta, A.; Omar, S.; Balani, K. Progress in Material Selection for Solid Oxide Fuel Cell Technology: A Review. Prog. Mater. Sci. 2015, 72, 141–337. [Google Scholar] [CrossRef]
- Fergus, J.W. Sealants for Solid Oxide Fuel Cells. J. Power Source 2005, 147, 46–57. [Google Scholar] [CrossRef]
- Duquette, J.; Petric, A. Silver Wire Seal Design for Planar Solid Oxide Fuel Cell Stack. J. Power Source 2004, 137, 71–75. [Google Scholar] [CrossRef]
- Li, R.; Lu, Y.; Yu, Y.; Ren, X.; Ding, F.; Guan, C.; Wang, J. Investigation on Long-Term Stability of Vermiculite Seals for Reversible Solid Oxide Cell. Molecules 2023, 28, 1462. [Google Scholar] [CrossRef]
- Batfalsky, P.; Haanappel, V.A.C.; Malzbender, J.; Menzler, N.H.; Shemet, V.; Vinke, I.C.; Steinbrech, R.W. Chemical Interaction between Glass-Ceramic Sealants and Interconnect Steels in SOFC Stacks. J. Power Source 2006, 155, 128–137. [Google Scholar] [CrossRef]
- Javed, H.; Zanchi, E.; D’Isanto, F.; Bert, C.; Ferrero, D.; Santarelli, M.; Smeacetto, F. Novel SrO-Containing Glass-Ceramic Sealants for Solid Oxide Electrolysis Cells (SOEC): Their Design and Characterization under Relevant Conditions. Materials 2022, 15, 5805. [Google Scholar] [CrossRef]
- Sreedhar, I.; Agarwal, B.; Goyal, P.; Agarwal, A. An Overview of Degradation in Solid Oxide Fuel Cells-Potential Clean Power Sources. J. Solid State Electrochem. 2020, 24, 1239–1270. [Google Scholar] [CrossRef]
- Javed, H.; Sabato, A.G.; Mansourkiaei, M.; Ferrero, D.; Santarelli, M.; Herbrig, K.; Walter, C.; Smeacetto, F. Glass-Ceramic Sealants for SOEC: Thermal Characterization and Electrical Resistivity in Dual Atmosphere. Energies 2020, 13, 3682. [Google Scholar] [CrossRef]
- Da Silva, M.J.; Bartolomé, J.F.; De Aza, A.H.; Mello-Castanho, S. Glass Ceramic Sealants Belonging to BAS (BaO-Al2O3-SiO2) Ternary System Modified with B2O3 Addition: A Different Approach to Access the SOFC Seal Issue. J. Eur. Ceram. Soc. 2016, 36, 631–644. [Google Scholar] [CrossRef]
- Chou, Y.S.; Stevenson, J.W.; Singh, P. Effect of Pre-Oxidation and Environmental Aging on the Seal Strength of a Novel High-Temperature Solid Oxide Fuel Cell (SOFC) Sealing Glass with Metallic Interconnect. J. Power Source 2008, 184, 238–244. [Google Scholar] [CrossRef]
- Wang, X.; Ou, D.R.; Zhao, Z.; Cheng, M. Stability of SrO-La2O3-Al2O3-SiO2 Glass Sealants in High-Temperature Air and Steam. Ceram. Int. 2016, 42, 7514–7523. [Google Scholar] [CrossRef]
- Javed, H.; Sabato, A.G.; Herbrig, K.; Ferrero, D.; Walter, C.; Salvo, M.; Smeacetto, F. Design and Characterization of Novel Glass-Ceramic Sealants for Solid Oxide Electrolysis Cell (SOEC) Applications. Int. J. Appl. Ceram. Technol. 2018, 15, 999–1010. [Google Scholar] [CrossRef]
- Javed, H.; Sabato, A.G.; Dlouhy, I.; Halasova, M.; Bernardo, E.; Salvo, M.; Herbrig, K.; Walter, C.; Smeacetto, F. Shear Performance at Room and High Temperatures of Glass-Ceramic Sealants for Solid Oxide Electrolysis Cell Technology. Materials 2019, 12, 298. [Google Scholar] [CrossRef] [PubMed]
- Kiebach, R.; Agersted, K.; Zielke, P.; Ritucci, I.; Brock, M.; Hendriksen, P.V. A Novel SOFC/SOEC Sealing Glass with a Low SiO2 Content and a High Thermal Expansion Coefficient. ECS Trans. 2017, 78, 1739–1747. [Google Scholar] [CrossRef]
- Nakane, H.; Shimada, H.; Sagata, K.; Yamaguchi, Y. Effective Ceramic Sealing Agents for Solid Oxide Cells by Low Temperature Curing below 200 °C. Ceram. Int. 2022, 48, 12988–12995. [Google Scholar] [CrossRef]
- Tulyaganov, D.U.; Reddy, A.A.; Kharton, V.V.; Ferreira, J.M.F. Aluminosilicate-Based Sealants for SOFCs and Other Electrochemical Applications—A Brief Review. J. Power Source 2013, 242, 486–502. [Google Scholar] [CrossRef]
- Sabato, A.G.; Cempura, G.; Montinaro, D.; Chrysanthou, A.; Salvo, M.; Bernardo, E.; Secco, M.; Smeacetto, F. Glass-Ceramic Sealant for Solid Oxide Fuel Cells Application: Characterization and Performance in Dual Atmosphere. J. Power Source 2016, 328, 262–270. [Google Scholar] [CrossRef]
- Sabato, A.G.; Rost, A.; Schilm, J.; Kusnezoff, M.; Salvo, M.; Chrysanthou, A.; Smeacetto, F. Effect of Electric Load and Dual Atmosphere on the Properties of an Alkali Containing Diopside-Based Glass Sealant for Solid Oxide Cells. J. Power Source 2019, 415, 15–24. [Google Scholar] [CrossRef]
- Smeacetto, F.; Zanchi, E.; Meena Narayana Menon, D.; Janner, D.; Lamnini, S.; Salvo, M.; De La Pierre, S.; Javed, H.; Ferraris, M. Torsional Behaviour of Glass-Joined, Laser-Processed Crofer 22 APU Interconnect: Unravelling the Effect of Surface Roughness on the Shear Strength. Ceram. Int. 2022, 48, 32837–32843. [Google Scholar] [CrossRef]
- Li, R.; Peng, L.; Wang, X.; Yang, J.; Yan, D.; Pu, J.; Chi, B.; Li, J. Investigating the Performance of Glass/Al2O3 Composite Seals in Planar Solid Oxide Fuel Cells. Compos. Part B Eng. 2020, 192, 107984. [Google Scholar] [CrossRef]
- Ritucci, I.; Agersted, K.; Zielke, P.; Wulff, A.C.; Khajavi, P.; Smeacetto, F.; Sabato, A.G.; Kiebach, R. A Ba-Free Sealing Glass with a High Coefficient of Thermal Expansion and Excellent Interface Stability Optimized for SOFC/SOEC Stack Applications. Int. J. Appl. Ceram. Technol. 2018, 15, 1011–1022. [Google Scholar] [CrossRef]
- Lee, H.; Kim, U.S.; Kim, S.D.; Woo, S.K.; Chung, W.J. SiO2–B2O3-BaO-WO3 Glasses with Varying Al2O3 Content as a Sealing Material for Reversible Solid Oxide Fuel Cells. Ceram. Int. 2020, 46, 18256–18261. [Google Scholar] [CrossRef]
- Sudireddy, B.R.; Nielsen, J.; Persson, Å.H.; Thyden, K.; Brodersen, K.; Ramousse, S.; Neagu, D.; Stefan, E.; Irvine, J.T.S.; Geisler, H.; et al. Development of Robust Metal Supported SOFCs and Stack Components in EUMETSAPP Consortium. Fuel Cells 2017, 17, 508–516. [Google Scholar] [CrossRef]
- Si, X.; Cao, J.; Talic, B.; Ritucci, I.; Li, C.; Qi, J.; Feng, J.; Kiebach, R. A Novel Ag Based Sealant for Solid Oxide Cells with a Fully Tunable Thermal Expansion. J. Alloys Compd. 2020, 831, 154608. [Google Scholar] [CrossRef]
- Kiebach, R.; Engelbrecht, K.; Grahl-Madsen, L.; Sieborg, B.; Chen, M.; Hjelm, J.; Norrman, K.; Chatzichristodoulou, C.; Hendriksen, P.V. An Ag Based Brazing System with a Tunable Thermal Expansion for the Use as Sealant for Solid Oxide Cells. J. Power Source 2016, 315, 339–350. [Google Scholar] [CrossRef]



| Fuel | Energy Content (MJ.kg−1) |
|---|---|
| Hydrogen | 120 |
| Liquefied Natural Gas (LNG) | 54.4 |
| Propane | 49.6 |
| Gasoline | 45.6–46.8 |
| Ethanol | 29.6 |
| Methanol | 19.7 |
| Coke | 27 |
| Dry wood | 16.2 |
| Bagasse | 9.6 |
| Process | Feedstock | Operating Condition | Level of Development | Efficiency (%) |
|---|---|---|---|---|
| Steam reforming | Light hydrocarbons | 700–1000 °C | Commercial | 74–85 |
| Partial oxidation | Hydrocarbons, coal, and heavy fuel oil | 800–1000 °C | Commercial | 60–75 |
| Autothermal refining | Light hydrocarbons | 700–1000 °C | Early commercial | 60–75 |
| Pyrolysis | Hydrocarbons | 1000–1400 °C | Commercial | 35–50 |
| Coal gasification | Coal | 700–1200 °C | Commercial | - |
| Ammonia reforming | Ammonia | 800–900 °C | Early commercial | 28 |
| Biomass gasification | Biomass | 800–1000 °C | Commercial | 30–60 |
| Thermolysis | - | >2500 °C (1000 °C < for thermochemical cycles) | R&D | 20–50 |
| Photoelectrolysis | Water and sunlight | Ambient conditions | R&D | 0.06–14 |
| Biophotolysis of microorganisms | - | Ambient conditions | R&D | 10–15 |
| Dark fermentation of | Carbohydrate-rich substrates | Anoxic conditions | R&D | 60–80 |
| Photofermentative | Small organic molecules and sunlight | Anaerobic conditions at ambient temperature | R&D | 0.1–12 |
| Pyrolysis of biomass | Biomass | 300–1000 °C (under an oxygen-free atmosphere) | Commercial | 21.9–75 |
| Alkaline electrolysis | Water and electricity | 40–90 °C | Commercial | 62–82 |
| PEM electrolysis | Water and electricity | 20–100 °C | Commercial | 67–82 |
| SOEC | Water, electricity, and heat | 700–1000 °C | R&D | ~100 |
| Hydrothermal liquefaction | Wet biomass | 4–22 MPa 250–370 °C | R&D | 85–90 |
| Thermochemical water splitting | Heat and water | 500–1000 °C | R&D | 20–45 |
| Cell Geometry | Strength | Weakness |
|---|---|---|
| Anode-supported |
|
|
| Electrolyte-supported |
|
|
| Cathode-supported |
|
|
| Metal-supported |
|
|
| Region | Degradation | Test Conditions |
|---|---|---|
| Physical, chemical, and microstructure change | ||
| LSM-YSZ//YSZ | LSM-YSZ: formation of small YSZ grains; YSZ: intergranular fracture and hole/pore formation along the grain boundaries of YSZ close to the air electrode. | 1–2 A.cm−2, 850 °C, 50%H2O/50%H2, 80–900 h |
| LSM//YSZ | LSM: delamination, disintegration of LSM particles at the interface; YSZ: grain boundary widening. | 500 mA.cm−2, 800 °C, 48 h. 895 °C |
| LSM-YSZ//YSZ | YSZ: void formation; no phase change; LSM-YSZ: delamination but no cation diffusion. | 895 °C up to 2.5 A.cm−2, 70% H2O |
| LSM-YSZ//YSZ | LSM: delamination, disintegration of LSM particles at the interface; YSZ: surface roughening. | 500 mA.cm−2, 800 °C, 100 h |
| LSM-YSZ//YSZ | LSM-YSZ: delamination. | 0.5 A.cm−2, 750 °C, 640 h |
| LSM-YSZ//YSZ | LSM-YSZ: La2Zr2O7 formation | 1.5–1.7 V, 800–950 °C |
| LSCF//YSZ | Delamination | 800 mA.cm−2, 800 °C, 50 h |
| LSCF/GDC//YSZ | LSCF: phase change from rhombohedral to cubic; GDC interlayer: pore formation | 800 mA.cm−2, 800 °C, 100 h |
| LSCF/GDC//YSZ | LSCF: SrZrO3 formation within GDC layer | As prepared |
| LSCF/GDC//YSZ | LSCF: SrZrO3 formation within GDC layer; YSZ: pore formation near the interface; GDC: Y and Zr diffusion into GDC interlayer, forming a dense reaction layer. | 0.75–1.0 A.cm−2, 780 °C, 9000 h. |
| LSCF/GDC//YSZ | LSCF: reduction of Co4+ to Co3+; GDC: Sr enrichment. | 0.8 A.cm−2 for 1000 h |
| LSCF-GDC/GDC//YSZ | LSCF-GDC: no delamination. | 0.5 A.cm−2, 750 °C, 640 h |
| LSC-GDC/GDC//YSZ | LSC-GDC: delamination at air inlet. | 0.5 A.cm−2, 750 °C, 640 h |
| Ni-YSZ//YSZ | Ni-YSZ: agglomeration of Ni particles, no change in YSZ phase. | 0.8 A.cm−2, 800 °C, 90%H2O/10%H2, 1000 h |
| Ni-YSZ//YSZ | Ni-YSZ: 2–4 μm dense Ni/YSZ layer | 2 A.cm−2, 950 °C, 90%H2O |
| Ni-YSZ//YSZ | Ni-YSZ: Ni agglomeration and depletion at the interface; YSZ: pore formation at the interface | 0.75–1.0 Acm−2, 780 °C, 80%H2O, 9000 h. |
| //10Sc1CeSZ | 10Sc1CeSZ: presence of Ce3+, phase change from cubic to rhombohedral | over 2 V. |
| Effect of Contaminants/Impurities | ||
| Ni-YSZ (porosity: 25%)//YSZ | Ni-YSZ: formation of carbon nano-fibre in Ni-YSZ electrode close to YSZ; Delamination | 2.25 A.cm−2, 875 °C, 45%H2O/45%CO2/10%H2, 11 h |
| Ni-YSZ (porosity: 40%)//YSZ | Ni-YSZ: No carbon deposition in Ni-YSZ electrode at the interface; No delamination | 2.0 A.cm−2, 875 °C, 45%H2O/45%CO2/10%H2, 677 h |
| Ni-infiltrated GDC//YSZ Ni-YSZ//YSZ: | Ni-YSZ: carbon deposition | 0.1 A.cm−2, 600 °C, 5%CO/95%CO2, 5 h |
| Ni-YSZ//YSZ: glass | Ni-YSZ: SiO2 deposition at region close to YSZ electrolyte. | 0.5 A.cm−2, 850 °C in 50%H2O/50%H2 for 1316 h |
| LSM//YSZ: Fe-Cr alloy | LSM: Cr deposition and SrCrO4 formation at the interface and in the electrode bulk. | 0.2–0.5 A.cm−2, 800 °C, air |
| LSCF//GDC: Fe-Cr alloy | LSCF: Cr deposition mainly on the outmost surface of the electrodes in the form of SrCrO4, CrO2.5, and Cr2O3 | 0.2 A.cm−2, 800–900 °C, air |
| LSC | LSC contact layer: Cr deposition mainly on the LSC contact layer. | 2000 h. |
| LSM//YSZ: glass | LSM: accelerated Sr segregation and boron deposition at the interface, forming lanthanum borates | 0.2 A.cm−2, 800 °C, air |
| Interconnect Material | Coating | Coating Technique | Test Conditions | ASR (mΩ) |
|---|---|---|---|---|
| SUS430 | Mn-Co | PVD | 800 °C/air/1250 h | 28.6 |
| AISI 430 | Mn-Co | DGPA | 800 °C/air | 29 (480 h) |
| Crofer22 APU | MnCo1.7Fe0.3O4 | EPD | 800 °C/air/1000 h | - |
| Crofer22 APU | MnCo1.7Fe0.3O4 | APS | 700 °C/air/1000 h | 50 |
| Sanergy HT | (Mn, Co)3O4 | Screen printing | 800 °C/air/1000 h | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fallah Vostakola, M.; Ozcan, H.; El-Emam, R.S.; Amini Horri, B. Recent Advances in High-Temperature Steam Electrolysis with Solid Oxide Electrolysers for Green Hydrogen Production. Energies 2023, 16, 3327. https://doi.org/10.3390/en16083327
Fallah Vostakola M, Ozcan H, El-Emam RS, Amini Horri B. Recent Advances in High-Temperature Steam Electrolysis with Solid Oxide Electrolysers for Green Hydrogen Production. Energies. 2023; 16(8):3327. https://doi.org/10.3390/en16083327
Chicago/Turabian StyleFallah Vostakola, Mohsen, Hasan Ozcan, Rami S. El-Emam, and Bahman Amini Horri. 2023. "Recent Advances in High-Temperature Steam Electrolysis with Solid Oxide Electrolysers for Green Hydrogen Production" Energies 16, no. 8: 3327. https://doi.org/10.3390/en16083327
APA StyleFallah Vostakola, M., Ozcan, H., El-Emam, R. S., & Amini Horri, B. (2023). Recent Advances in High-Temperature Steam Electrolysis with Solid Oxide Electrolysers for Green Hydrogen Production. Energies, 16(8), 3327. https://doi.org/10.3390/en16083327









