In Situ Combustion of Heavy Oil within a Vuggy Carbonate Reservoir: Part I—Feasibility Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Oil
2.2. Brine
2.3. Core
2.4. Apparatus
2.5. Combustion Test Procedure
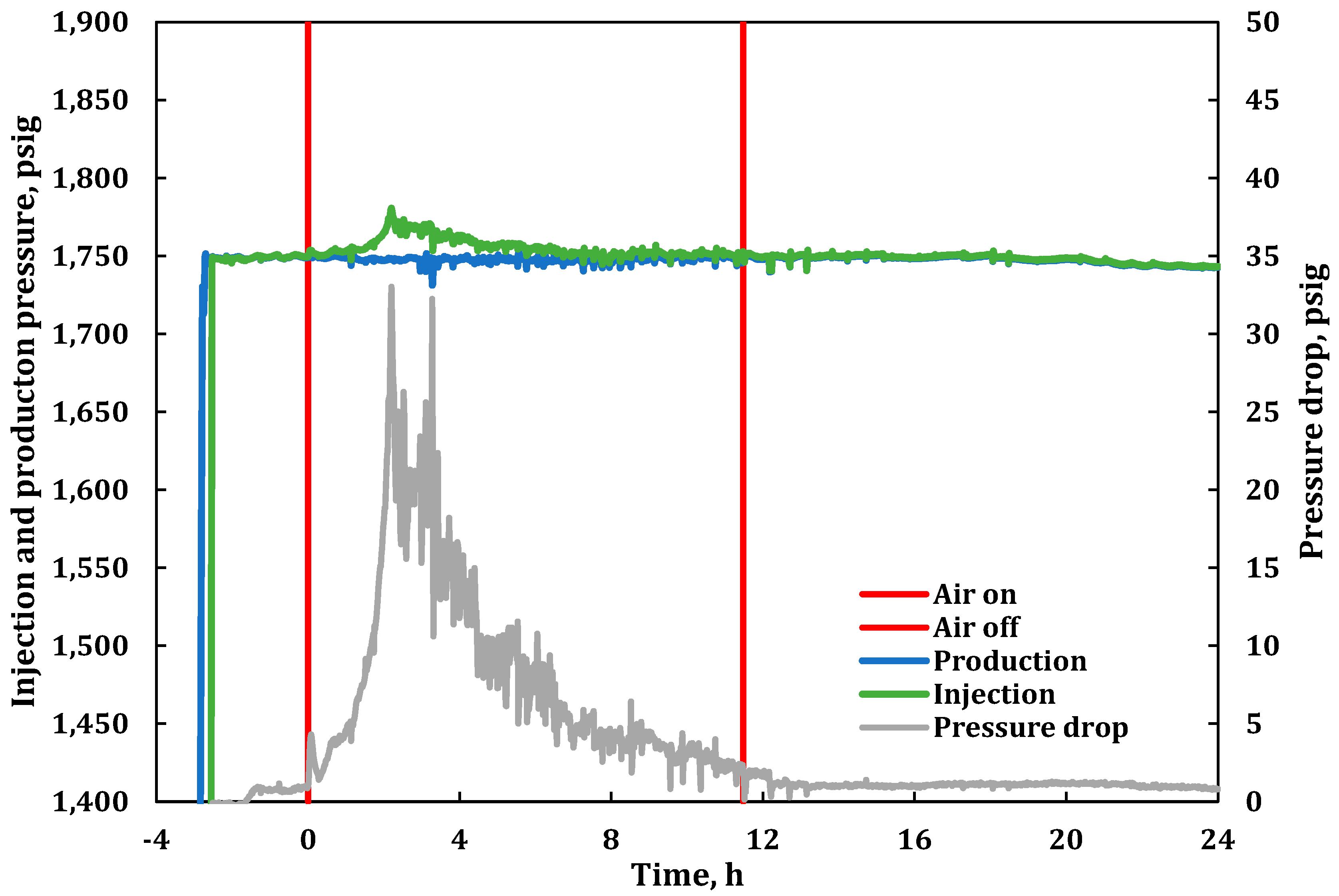
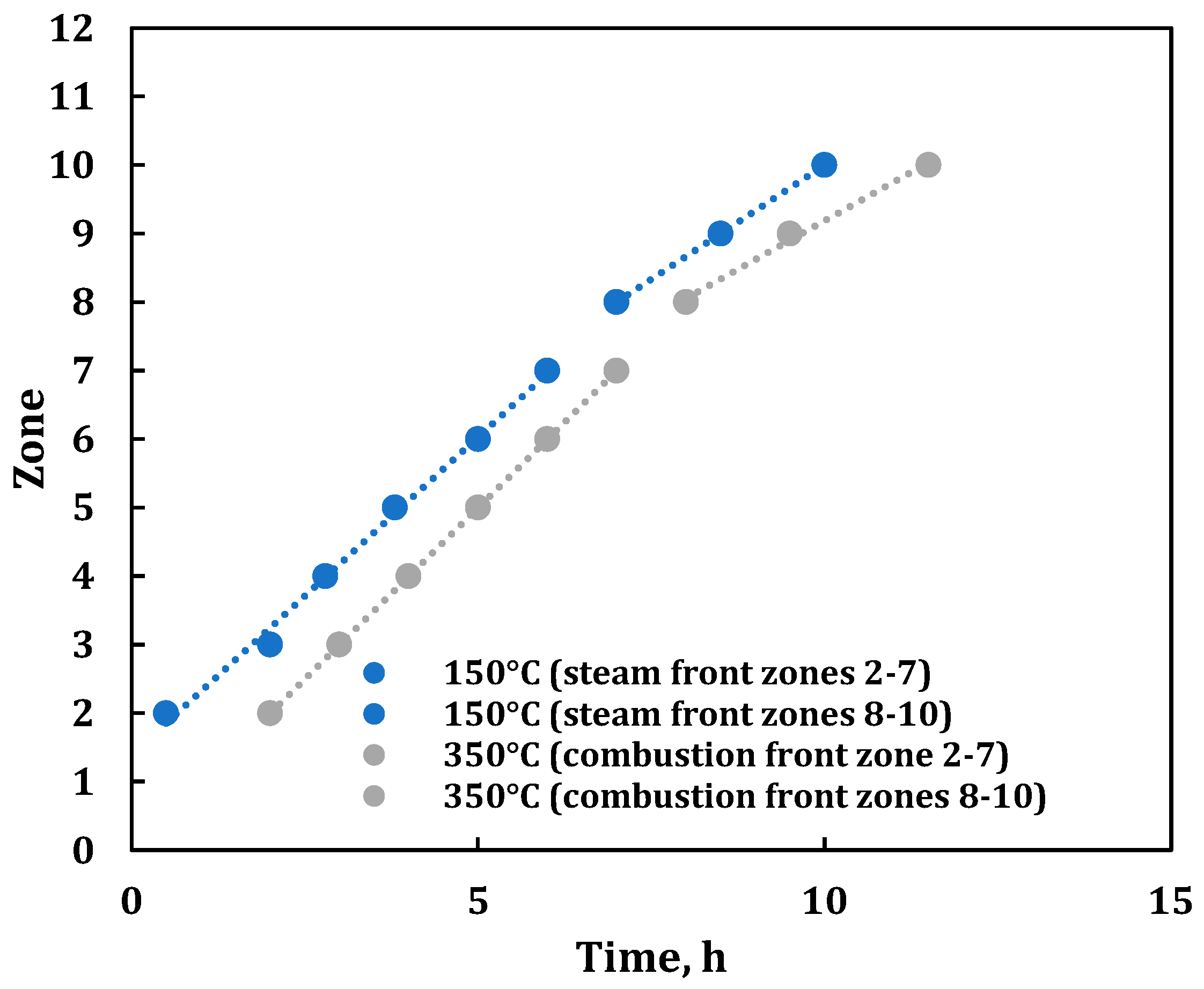
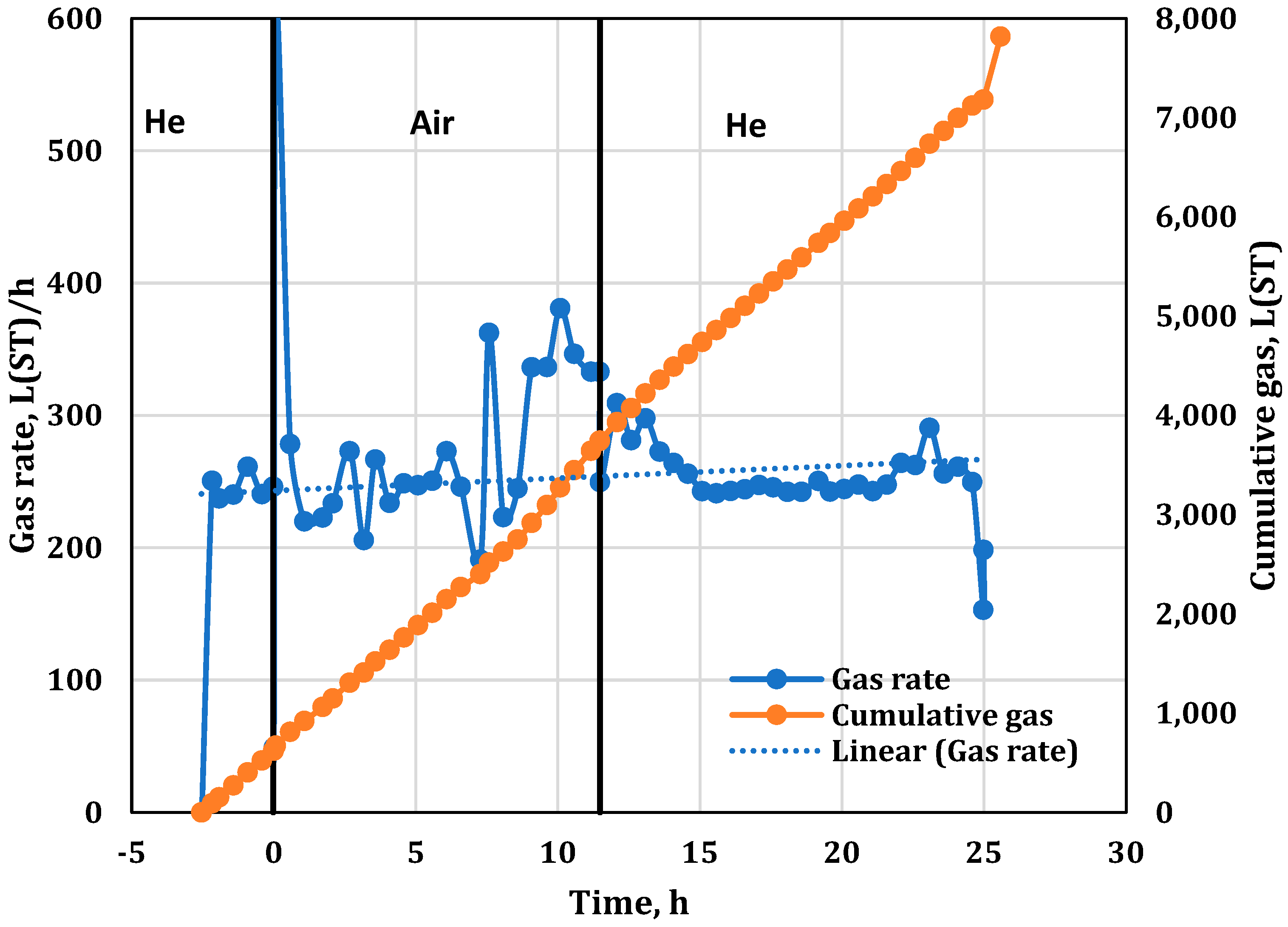
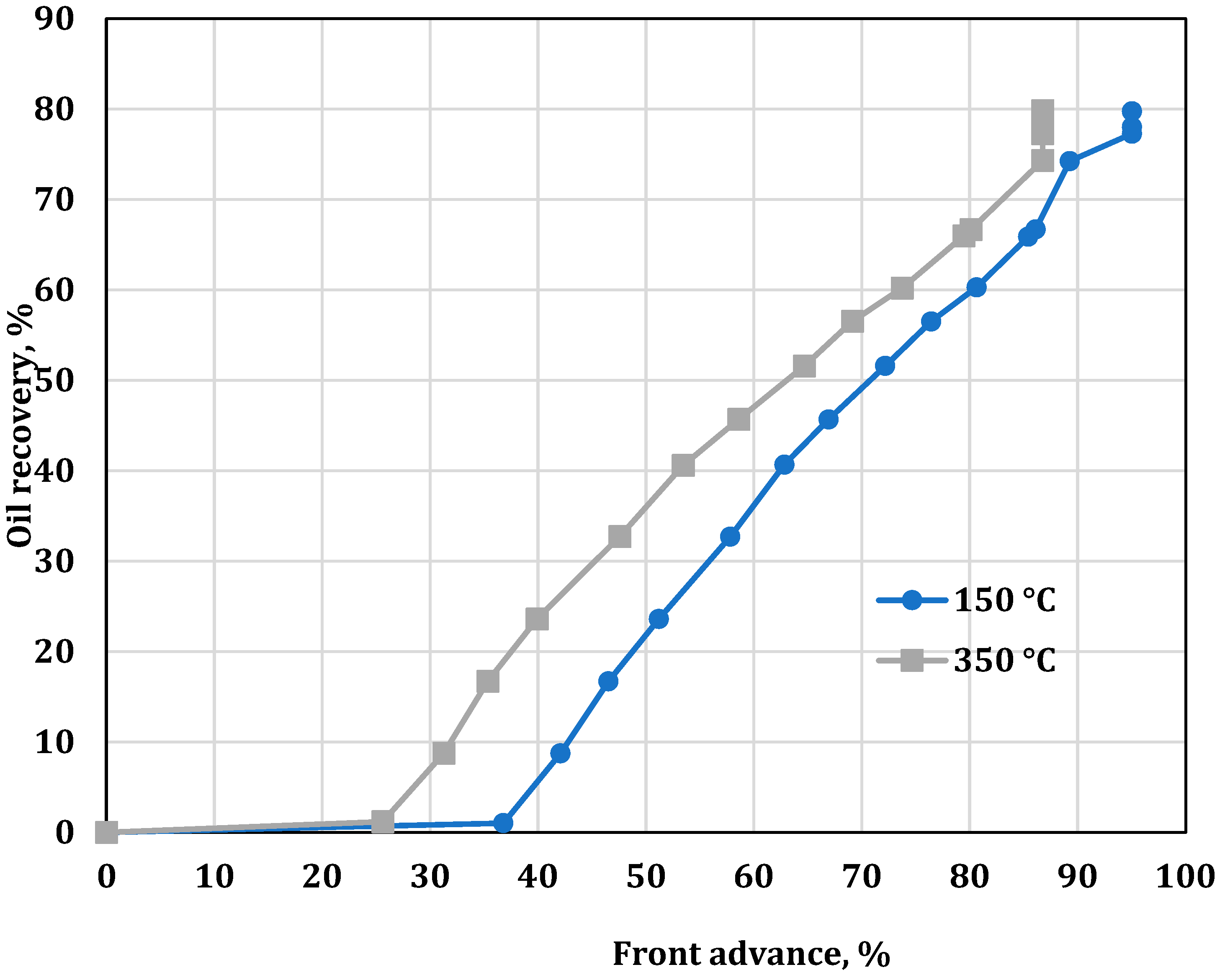
3. Results
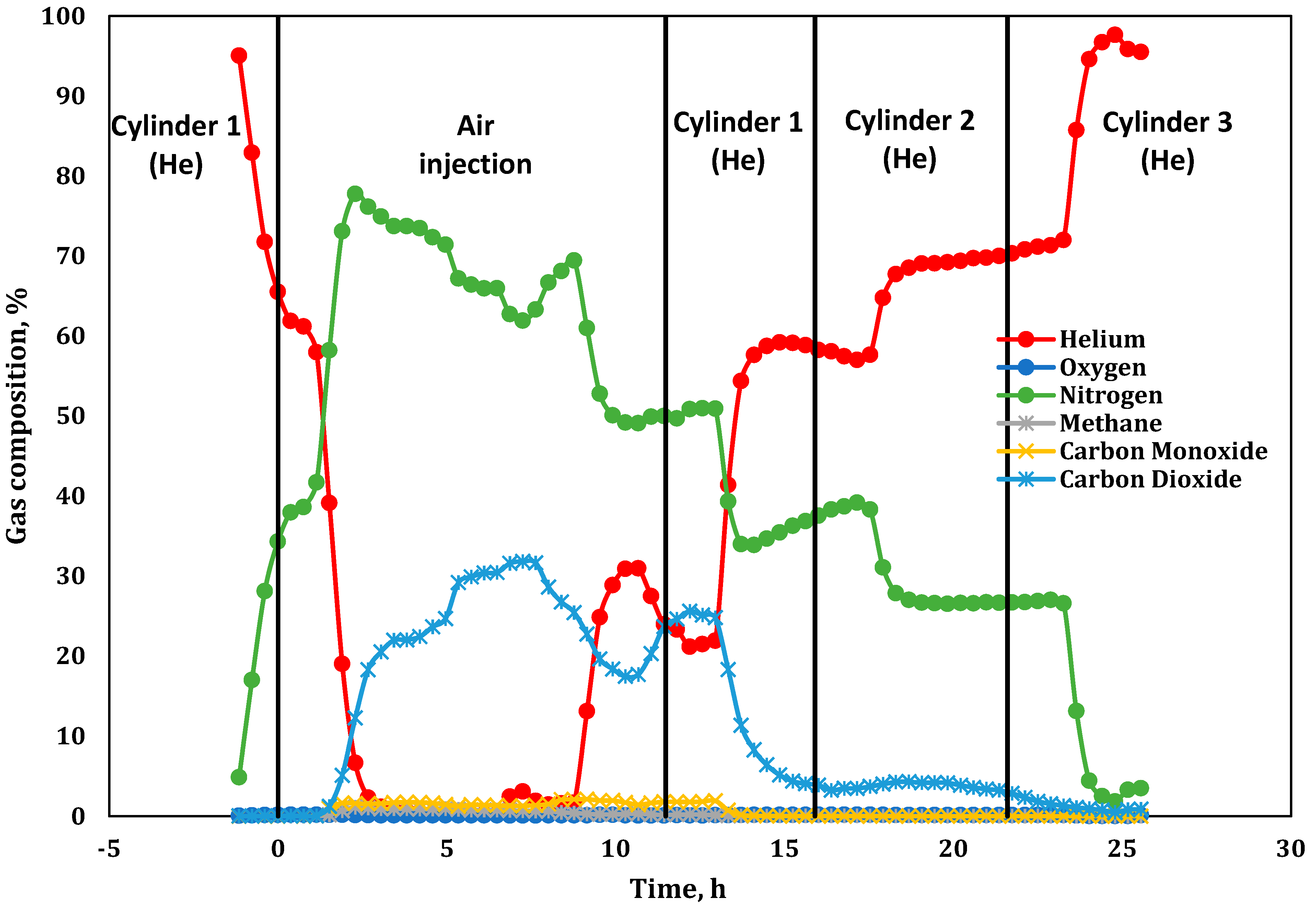
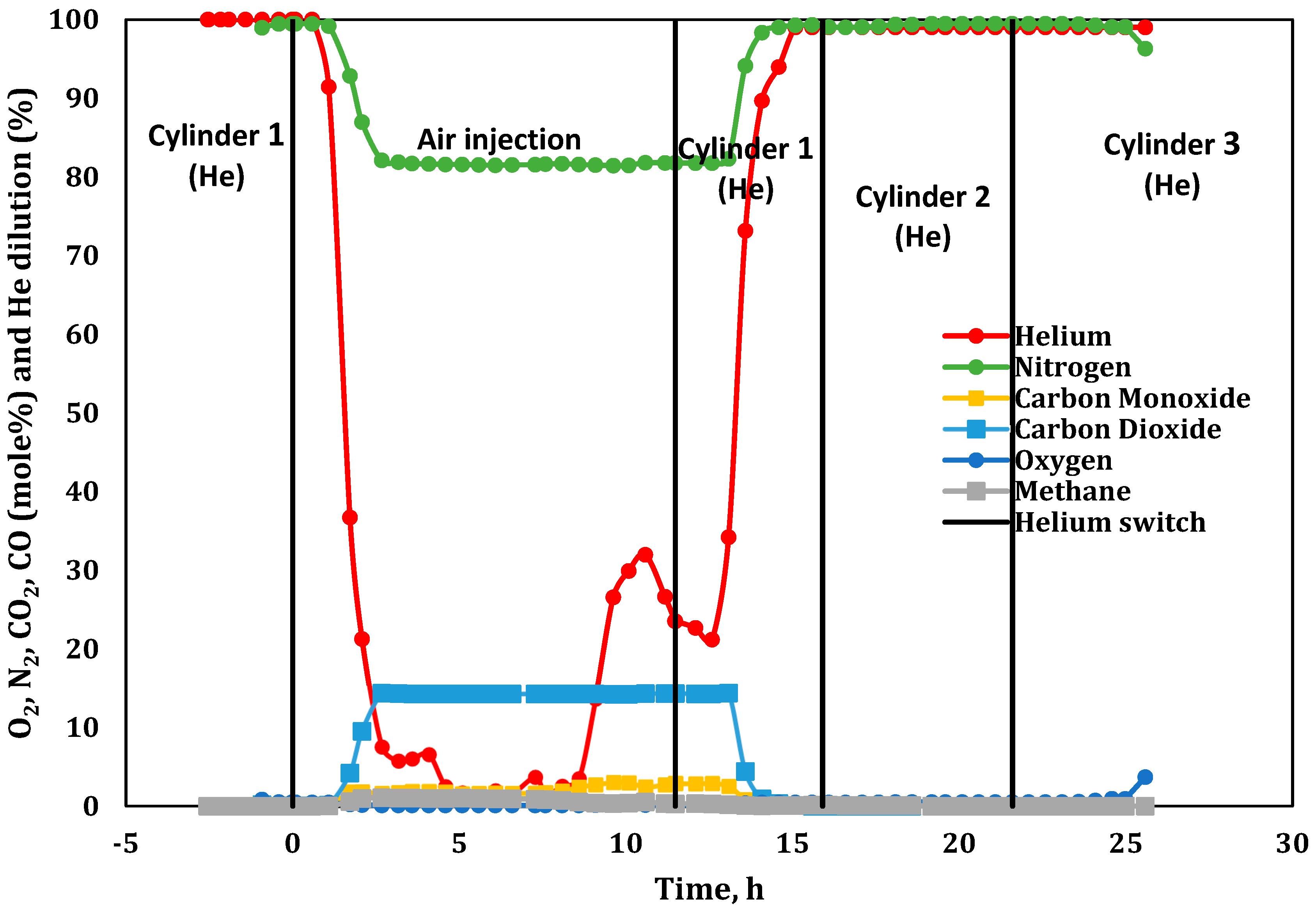
3.1. Product Gas Composition Analysis
3.2. Combustion Parameters Based on Product Gas Analysis
3.3. Post-Test Procedures
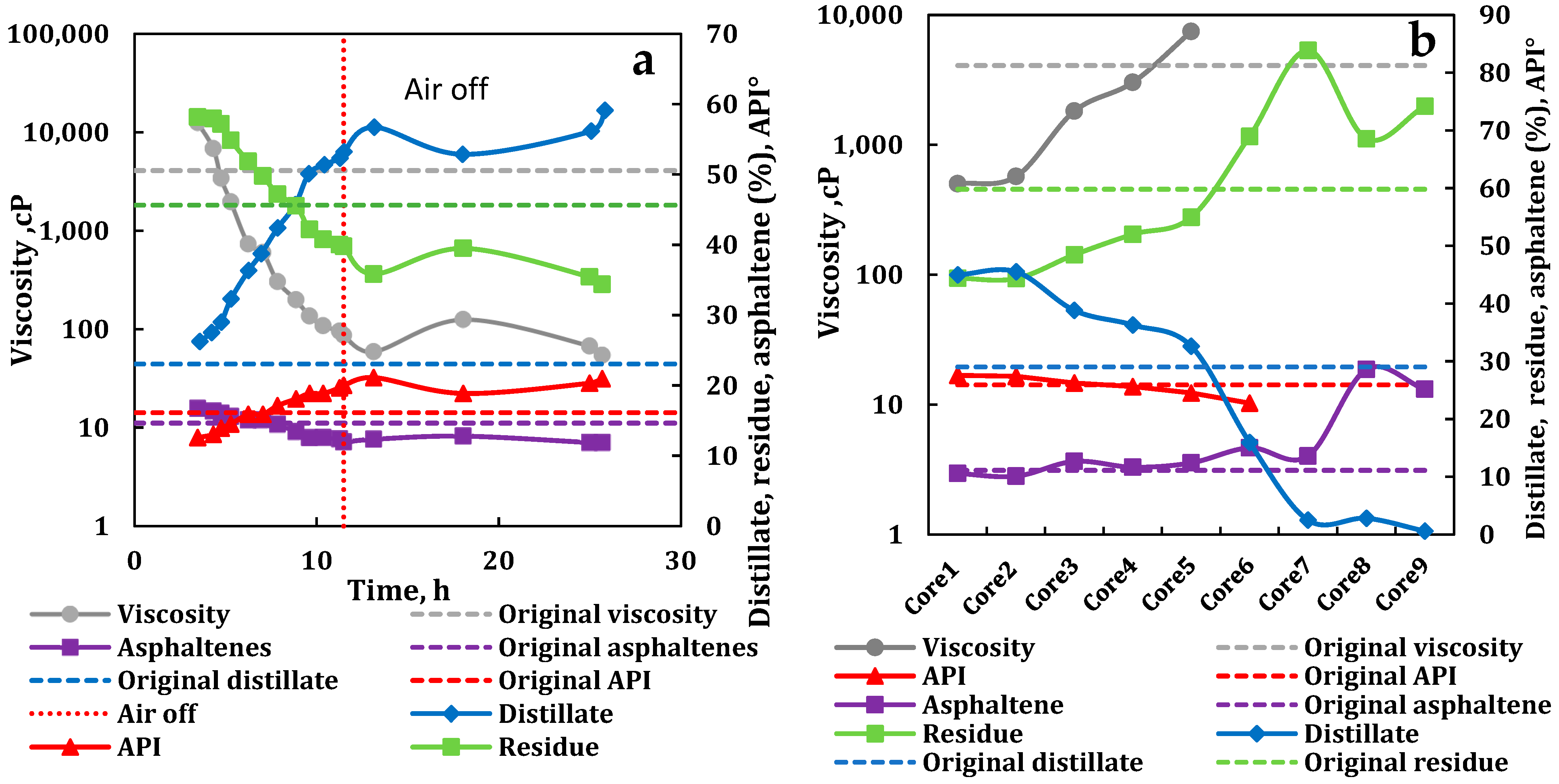
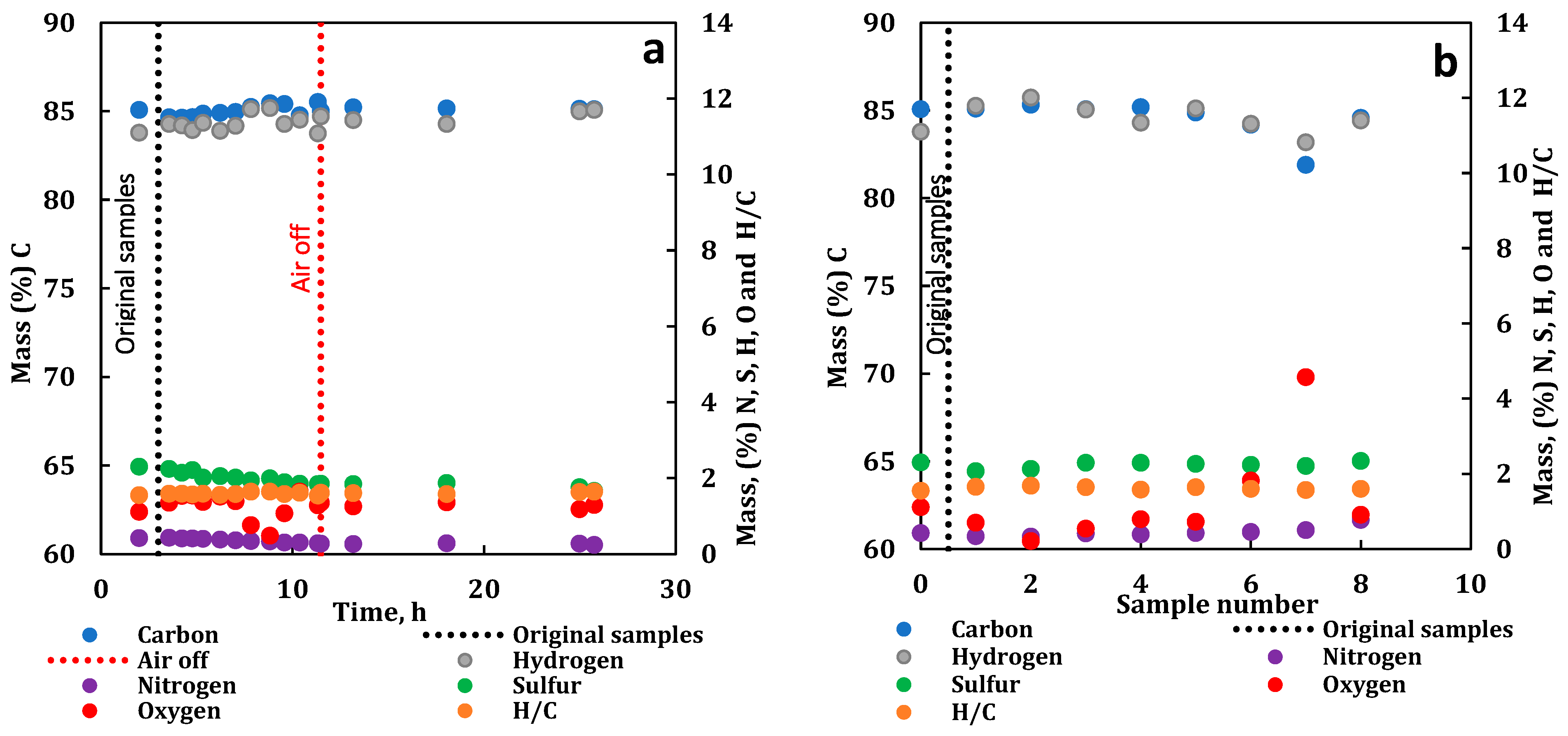
3.4. Compositional Analysis
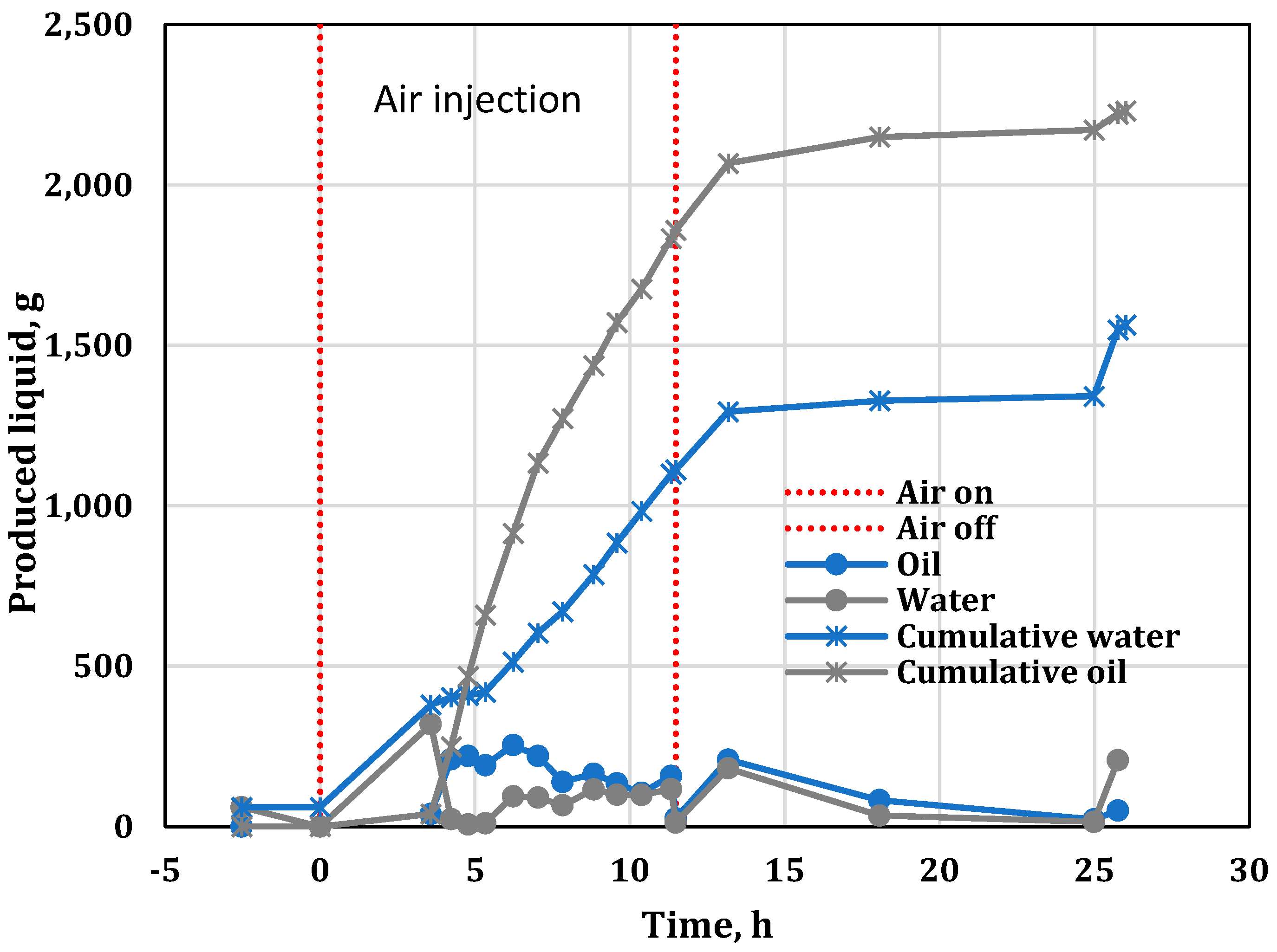
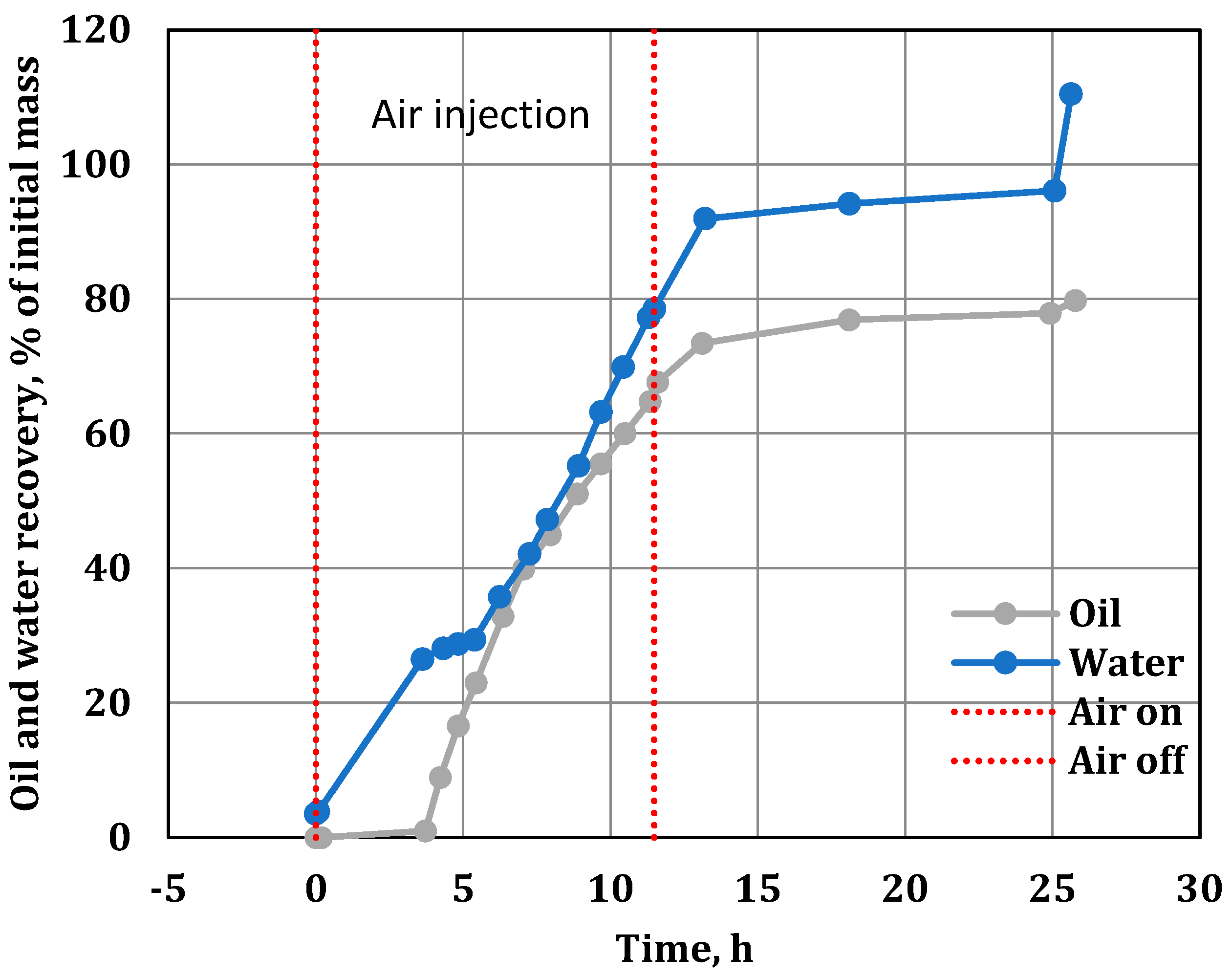
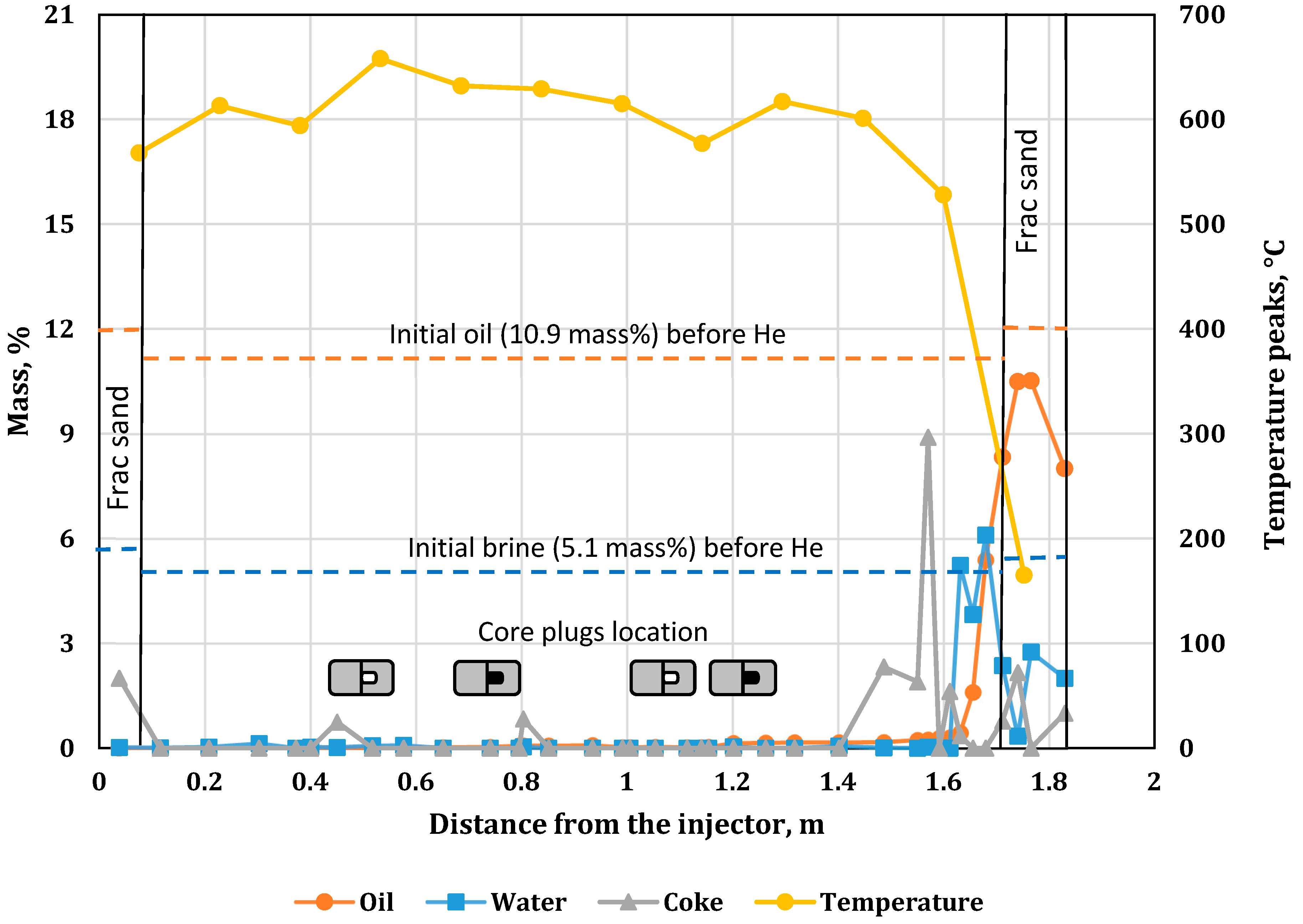
3.5. Water, Oil and Coke Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Green, D.W.; Willhite, G.P. Enhanced Oil Recovery, 4. Nachdr; Henry L. Doherty Memorial Fund of AIME, Society of Petroleum Engineers: Richardson, TX, USA, 2008. [Google Scholar]
- Moore, R.G.; Laureshen, C.J.; Ursenbach, M.G.; Mehta, S.A.; Belgrave, J.D.M. Combustion/Oxidation Behavior of Athabasca Oil Sands Bitumen. SPE/DOE Improv. Oil Recovery Symp. 1999, 2, 8. [Google Scholar] [CrossRef]
- Wu, Z.; Liu, H.; Pang, Z.; Wu, C.; Gao, M. Pore-Scale Experiment on Blocking Characteristics and EOR Mechanisms of Nitrogen Foam for Heavy Oil: A 2D Visualized Study. Energy Fuels 2016, 30, 9106–9113. [Google Scholar] [CrossRef]
- Agosta, F.; Prasad, M.; Aydin, A. Physical properties of carbonate fault rocks, fucino basin (Central Italy): Implications for fault seal in platform carbonates. Geofluids 2007, 7, 19–32. [Google Scholar] [CrossRef]
- Croizé, D.; Ehrenberg, S.N.; Bjørlykke, K.; Renard, F.; Jahren, J. Petrophysical properties of bioclastic platform carbonates: Implications for porosity controls during burial. Mar. Pet. Geol. 2010, 27, 1765–1774. [Google Scholar] [CrossRef]
- Heap, M.; Baud, P.; Reuschlé, T.; Meredith, P.G. Stylolites in limestones: Barriers to fluid flow? Geology 2014, 42, 51–54. [Google Scholar] [CrossRef]
- Machel, H.G. Investigations of Burial Diagenesis in Carbonate Hydrocarbon Reservoir Rocks. Geosci. Can. 2005, 32, 27. [Google Scholar]
- Mazzullo, S.J. Overview of Porosity Evolution in Carbonate Reservoirs. 2004. Available online: http://www.searchanddiscovery.com/documents/2004/mazzullo/ (accessed on 17 July 2019).
- Lucia, F.J. Rock-Fabric/Petrophysical Classification of Carbonate Pore Space for Reservoir Characterization. AAPG Bull. 1995, 79, 1275–1300. [Google Scholar] [CrossRef]
- Manrique, E.J.; Muci, V.E.; Gurfinkel, M.E. EOR Field Experiences in Carbonate Reservoirs in the United States. SPE Reserv. Eval. Eng. 2007, 10, 667–686. [Google Scholar] [CrossRef]
- Mayorquin-Ruiz, J.R.; Babadagli, T. Optimal Design of Low Temperature Air Injection for Efficient Recovery of Heavy Oil in Deep Naturally Fractured Reservoirs: Experimental and Numerical Approach. In Proceedings of the SPE Heavy Oil Conference Canada, Calgary, AB, Canada, 12–14 June 2012. [Google Scholar] [CrossRef]
- Lacroix, S.; Delaplace, P.; Bourbiaux, B.; Foulon, D. Simulation of Air Injection in Light-Oil Fractured Reservoirs: Setting-Up a Predictive Dual Porosity Model. In Proceedings of the SPE Annual Technical Conference and Exhibition, Houston, TX, USA, 26–29 September 2004; p. 12. [Google Scholar]
- Schulte, W.M.; de Vries, A.S. In-Situ Combustion in Naturally Fractured Heavy Oil Reservoirs. Soc. Pet. Eng. J. 1985, 25, 67–77. [Google Scholar] [CrossRef]
- Tabasinejad, F.; Karrat, R. Feasibility Study of In-Situ Combustion in Naturally Fractured Heavy Oil Reservoirs. In Proceedings of the International Oil Conference and Exhibition in Mexico, Cancun, Mexico, 31 August–2 September 2006; p. 10. [Google Scholar]
- Stokka, S.; Oesthus, A.; Frangeul, J. Evaluation of Air Injection as an IOR Method for the Giant Ekofisk Chalk Field. In Proceedings of the SPE International Improved Oil Recovery Conference in Asia Pacific, Kuala Lumpur, Malaysia, 5–6 December 2005. [Google Scholar] [CrossRef]
- Fatemi, S.M.; Kharrat, R.; Vossoughi, S. Investigation of Top-Down In-Situ Combustion Process in Complex Fractured Carbonate Models: Effects of Fractures’ Geometrical Properties. In Proceedings of the Canadian Unconventional Resources Conference, Calgary, AB, Canada, 15–17 November 2011. [Google Scholar] [CrossRef]
- Fadaei, H.; Debenest, G.; Kamp, A.M.; Quintard, M.; Renard, G. How the In-Situ Combustion Process Works in a Fractured System: 2D Core- and Block-Scale Simulation. SPE Reserv. Eval. Eng. 2010, 13, 118–130. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Sawatzky, R.P.; Forster, L.M. Alberta’s Bitumen Carbonate Reservoirs—Moving Forward with Advanced R&D. In Proceedings of the World Heavy Oil Congress, Edmonton, AB, Canada, 15 May 2008. [Google Scholar]
- Craig, F.J.; Parrish, D.R. A Multipilot Evaluation of the COFCAW Process. J. Pet. Technol. 1974, 26, 659–666. [Google Scholar] [CrossRef]
- Qi, J.; Yuan, J.-Y. History Matching Grosmont C Carbonate Thermal Production Performance. In Proceedings of the SPE Heavy Oil Conference-Canada, Calgary, AB, Canada, 11–13 June 2013. [Google Scholar] [CrossRef]
- Yuan, J.-Y.; Jiang, Q.; Russel-Houston, J.; Thornton, B.; Putnam, P. Evolving Recovery Technologies Directed Towards Commercial Development of the Grosmont Carbonate Reservoirs. In Proceedings of the Canadian Unconventional Resources and International Petroleum Conference, Calgary, AB, Canada, 19–21 October 2010. [Google Scholar] [CrossRef]
- Kovscek, A.R.; Castanier, L.M.; Gerritsen, M.G. Improved Predictability of In-Situ-Combustion Enhanced Oil Recovery. SPE Reserv. Eval. Eng. 2013, 16, 172–182. [Google Scholar] [CrossRef]
- Izgec, O. Reactive Flow in Uggy Carbonates: Methods and Models Applied to Matrix Acidizing of Carbonates; Texas A&M University: College Station, TX, USA, 2009. [Google Scholar]
- Barenblatt, G.; Zheltov, I.; Kochina, I. Basic concepts in the theory of seepage of homogeneous liquids in fissured rocks [strata]. J. Appl. Math. Mech. 1960, 24, 1286–1303. [Google Scholar] [CrossRef]
- Warren, J.; Root, P. The Behavior of Naturally Fractured Reservoirs. Soc. Pet. Eng. J. 1963, 3, 245–255. [Google Scholar] [CrossRef]
- Kazemi, H. Pressure Transient Analysis of Naturally Fractured Reservoirs with Uniform Fracture Distribution. Soc. Pet. Eng. J. 1969, 9, 451–462. [Google Scholar] [CrossRef]
- De Swaan, O.A. Analytic Solutions for Determining Naturally Fractured Reservoir Properties by Well Testing. Soc. Pet. Eng. J. 1976, 16, 117–122. [Google Scholar] [CrossRef]
- Liu, C. Exact solution for the compressible flow equations through a medium with triple-porosity. Appl. Math. Mech. 1981, 2, 457–462. [Google Scholar] [CrossRef]
- Abdassah, D.; Ershaghi, I. Triple-Porosity Systems for Representing Naturally Fractured Reservoirs. SPE Form. Eval. 1986, 1, 113–127. [Google Scholar] [CrossRef]
- Jalali, Y.; Ershaghi, I. Pressure Transient Analysis of Heterogeneous Naturally Fractured Reservoirs. In Proceedings of the SPE California Regional Meeting, Ventura, CA, USA, 8–10 April 1987; p. 14. [Google Scholar] [CrossRef]
- Al-Ghamdi, A.; Ershaghi, I. Pressure Transient Analysis of Dually Fractured Reservoirs. SPE J. 1996, 1, 93–100. [Google Scholar] [CrossRef]
- Aguilera, R. Analysis of Naturally Fractured Reservoirs From Conventional Well Logs. J. Pet. Technol. 1976, 28, 764–772. [Google Scholar] [CrossRef]
- Aguilera, R.F.; Aguilera, R. A Triple Porosity Model for Petrophysical Analysis of Naturally Fractured Reservoirs. Petrophysics 2004, 45, SPWLA-2004-v45n2a4. [Google Scholar]
- Moore, R.; Mehta, S.; Belgrave, J.; Ursenbach, M.; Laureshen, C.; Weissman, J.; Kessler, R. A Downhole Catalytic Upgrading Process for Heavy Oil Using In Situ Combustion. J. Can. Pet. Technol. 1999, 38, PETSOC-99-13-44. [Google Scholar] [CrossRef]
- Feizabadi, S.E. The Challenge of Proving Vapor Phase Burning in Porous Media in an In Situ Combustion Heavy Oil Recovery Process Based on Laboratory Modeling. 2017. Available online: http://hdl.handle.net/11023/4269 (accessed on 17 December 2022).
- Barzin, Y.; Moore, R.G.; Mehta, S.A.; Mallory, D.G.; Ursenbach, M.G.; Tabasinejad, F. Role of Vapor Phase in Oxidation/Combustion Kinetics of High-Pressure Air Injection (HPAI). In Proceedings of the SPE Annual Technical Conference and Exhibition, Florence, Italy, 19–22 September 2010; p. SPE-135641-MS. [Google Scholar] [CrossRef]
- Ambastha, A.; Kumar, M. New Insights Into In-Situ Combustion Simulation for Heavy Oil Reservoirs. In Proceedings of the SPE Annual Technical Conference and Exhibition, Houston, TX, USA, 3–6 October 1999. [Google Scholar] [CrossRef]
- Fazlyeva, R.R.; Ursenbach, M.G.; Mehta, S.A.; Moore, R.G.; Mallory, D.G. University of Calgary Vug Test No. 1; University of Calgary: Calgary, AB, Canada, 2021. [Google Scholar]
- Teramoto, T.; Uematsu, H.; Takabayashi, K.; Onishi, T. Air-Injection EOR in Highly Water-Saturated Light-Oil Reservoir. In Proceedings of the SPE Europec/EAGE Annual Conference and Exhibition, Vienna, Austria, 12–15 June 2006. [Google Scholar] [CrossRef]
- Sequera-Dalton, B.M.; Aminfar, E.; Moore, R.G.; Mehta, S.A.R.; Ursenbach, M.G. Compositional Changes in Athabasca Bitumen During Air Injection into Mature SAGD Chambers—Observations from 3-D Large Scale Experiments. In Proceedings of the SPE Canada Heavy Oil Conference, Virtual, 28 September–2 October 2020. [Google Scholar] [CrossRef]
- Askarova, A.G.; Popov, E.Y.; Maerle, K.V.; Cheremisin, A.N. Comparative Study of In-Situ Combustion Tests on Consolidated and Crushed Cores. SPE Reserv. Eval. Eng. 2022, 26, 167–179. [Google Scholar] [CrossRef]


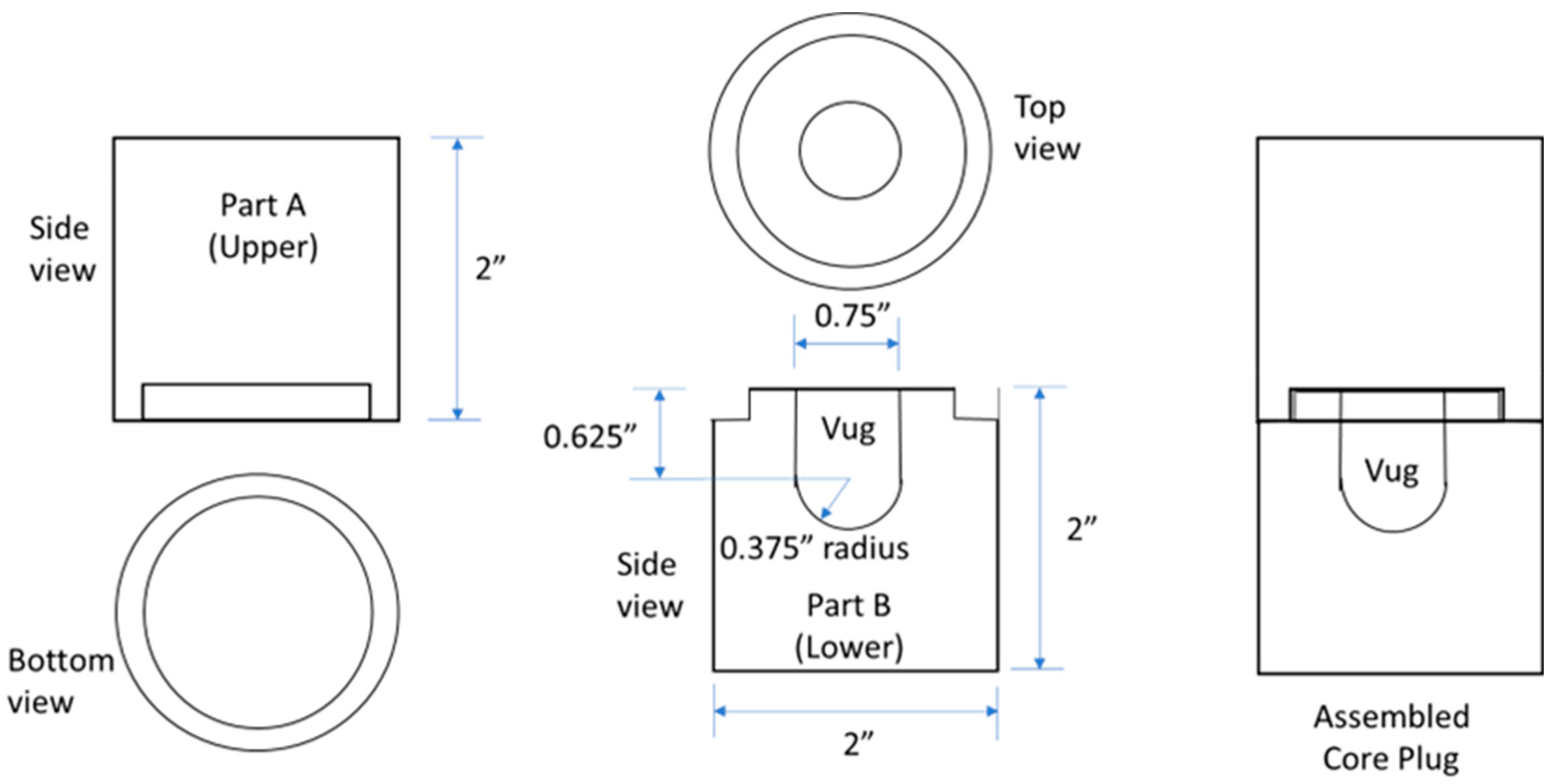
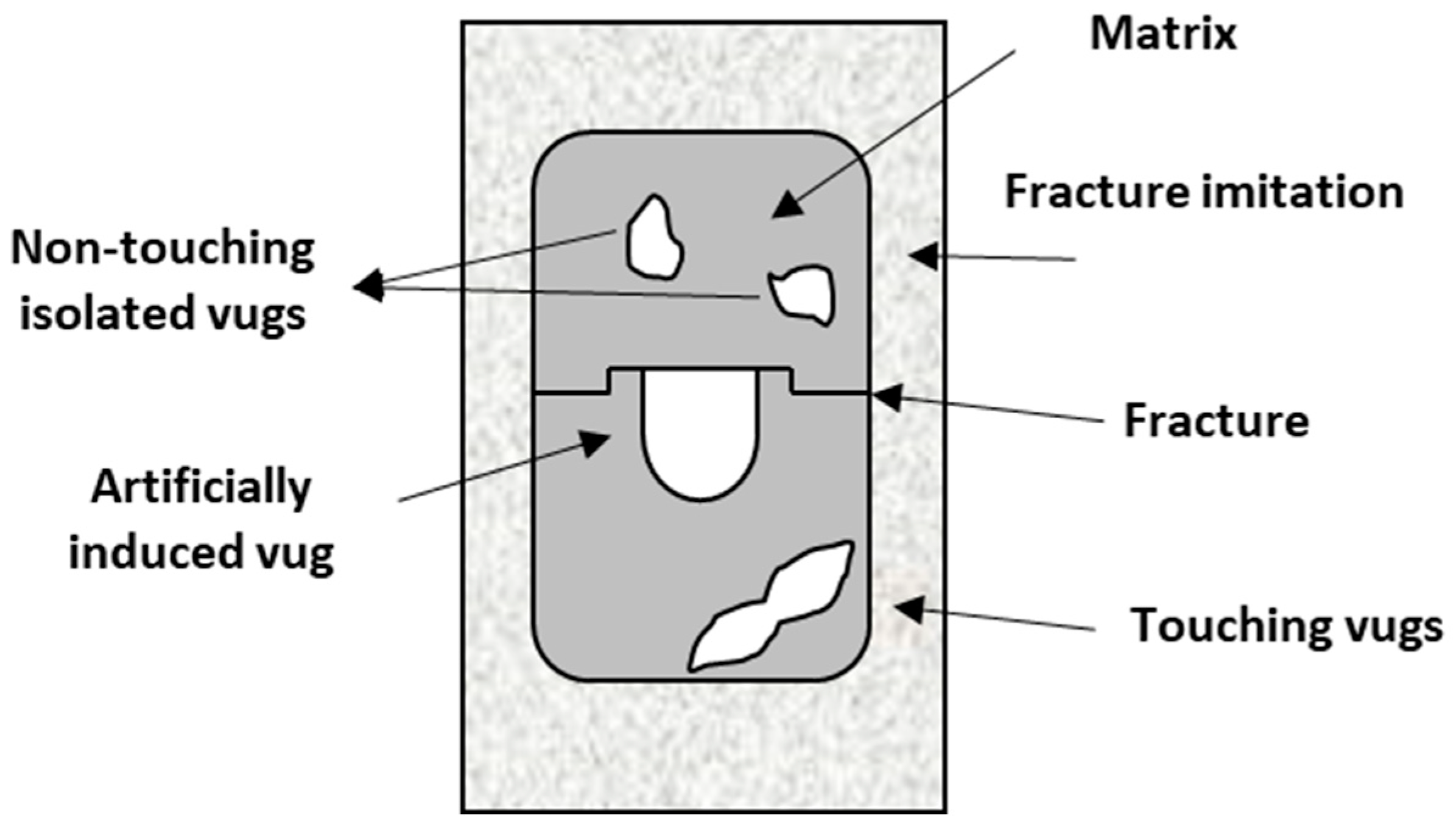
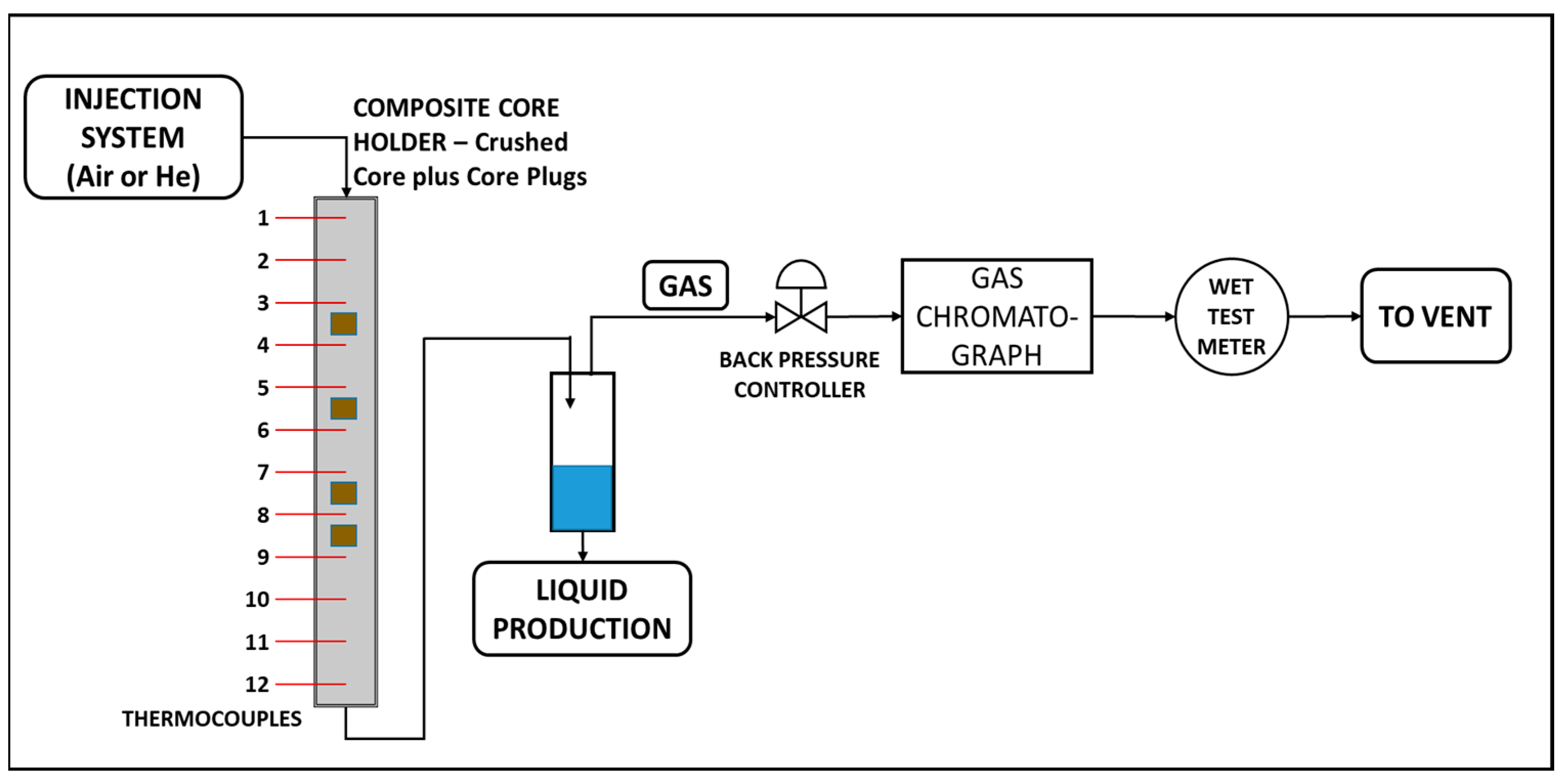
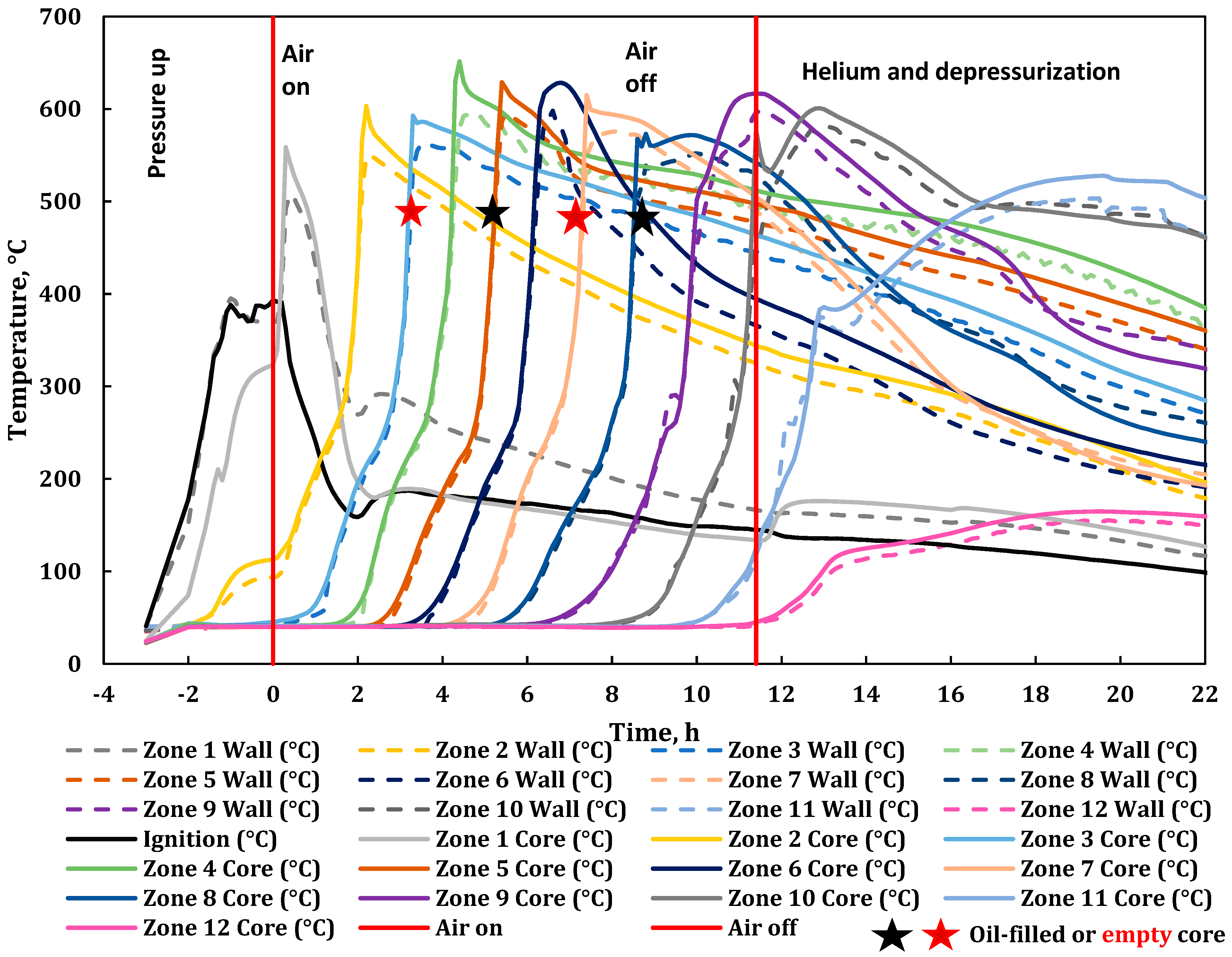




| Property | Oil |
|---|---|
| MW, g/mol | 375 |
| Carbon (C), wt% | 85.76 |
| Hydrogen (H2), wt% | 11.17 |
| Atomic H/C | 1.56 |
| Nitrogen (N2), wt% | 0.51 |
| Sulphur (S), wt% | 1.81 |
| Density @25 °C, kg/m3 | 972 |
| Viscosity @25 °C, cP | 4091 |
| Saturates, wt% | 31.58 |
| Aromatics, wt% | 32.29 |
| Resins, wt% | 25.00 |
| Asphaltenes, wt% | 11.13 |
| Components | Molar Mass (g/mol) | Concentration (mol/L) | Concentration (g/L) | Concentration (g/10 L) |
|---|---|---|---|---|
| MgCl2 • 6H2O | 203.30 | 0.102016 | 20.740 | 207.398 |
| CaCl2 • 2H2O | 147.01 | 0.137226 | 20.174 | 201.735 |
| FeCl3 • 6H2O | 270.30 | 0.000000 | 0.000 | 0.000 |
| KCl | 74.55 | 0.056346 | 4.201 | 42.006 |
| NaHCO3 | 84.01 | 0.004998 | 0.420 | 4.199 |
| Na2SO4 | 142.04 | 0.010285 | 1.461 | 14.609 |
| NaCl | 58.44 | 0.799164 | 46.703 | 467.031 |
| Material or Property | Value | Description |
|---|---|---|
| Oil, g | 2788.4 | Including the oil used for the core plug saturation and filling the vugs, as well as the crushed core premix. |
| Brine, g | 1253.3 | Used for the crushed core premix. An additional 342 g was used in the lines and pressure taps and is not included in this table. |
| Crushed core and frac sand, g | 20,279.0 | Dolomite core with 16- and 20/30-mesh frac sand. |
| Core plugs, g | 1732.3 | Prior to saturation. |
| Calculated porosity, % | 45.1 | Whole composite core. |
| Average Soil, % | 44.3 | Core saturation at the start of air injection based on oil and brine densities at 25 °C and atmospheric pressure. They provide an “order of magnitude” estimation of the core saturation under the actual conditions of 40 °C and 1740 psig (120 bar). Note that these calculations include the combined frac sand, crushed dolomite core and core plug (including vugs) components of the composite core pack. |
| Average Sbrine, % | 18.4 | |
| Average Sgas, % | 37.3 | |
| Initial temperature, °C | 40 | Represents a reservoir temperature. The ignition temperature was set as 325 °C. |
| Operating back pressure, psig | 1740 | Maintained throughout the test. |
| Run Time, (Hour) | Activity |
|---|---|
| −3.43 | Started pressurizing of the system to 1740 psig using He injection into the core and annulus. |
| −3.22 | All zones set to 30 °C. Started the core preheating to 40 °C (reservoir temperature). |
| −3.07 | All zones set to 40 °C. |
| −2.73 | Reached the target pressure of 1740 psig (120 bar). |
| −2.53 | Started He injection at 234 L(ST)/h (flux of 30.0 m3(ST)/m2h). Later, we found that the He cylinder was contaminated with 42% N2. |
| −2.35 | Start of ignition heating by setting Zone 1 to 100 °C. |
| 0.00 | Zone 1 at 325 °C (wall temperature 370 °C); start of air injection at 234 L(ST)/h; flux of 30.0 m3 (ST)/m2h. |
| 0.08 | Indication of ignition. |
| 3.57 | Produced first liquid (oil). |
| 5.92 | Installed flue gas scrubber (caustic) due to H2S/mercaptans in the produced gas. |
| 11.48 | Stopped air injection, reaching Zone 10; switched to He purge at 234 L(ST)/h (flux of 30 m3(ST)/m2h). Same He cylinder with 42% N2. |
| 15.82 | Switched to the second He cylinder. Determined that this cylinder contained 27% N2. |
| 21.58 | Identified the problem of He cylinder contamination. Changed to the third cylinder containing pure He. |
| 24.98 | Completed He purge; started depressurizing the system. |
| 25.58 | Test completion. |
| As Measured by the H2/He GC in Mole Percent | Injection Rate and Start Time | |||
|---|---|---|---|---|
| Temperature | He | N2 | O2 | L(ST)/h and (h) |
| Cylinder 1 (He) | 57.48 | 42.45 | 0.06 | 234.80; (−2.53 and 11.48) |
| Cylinder 2 (He) | 72.52 | 26.71 | 0.10 | 250.8; (15.8) |
| Cylinder 3 (He) | 100 | 0 | 0 | 292.6; (21.6) |
| As measured by the combustion GC * in mole percent | ||||
| Cylinder 1 (He) | - | 99.37 | 0.60 | |
| Cylinder 2 (He) | - | 99.12 | 0.83 | |
| Cylinder 3 (He) | - | - | - | |
| Air Cylinder | 78.11 | 21.89 | ||
| Total Fuel Consumed (g) | 237.9 |
| Mass of Carbon in Combustion Products (g) | 200.6 |
| Mass of Hydrogen in Combustion Products (g) | 37.4 |
| Total Air Required (liters (ST)) | 2690.8 |
| Measured Oxygen Feed (liters (ST)) | 589.0 |
| Measured Air Feed (liters (ST)) | 2690.8 |
| Total Volume of Produced Gas (inert flood, He-free) (liters (ST)) | 2539.4 |
| Total Volume of Produced Gas (He included) (liters (ST)) | 7824.8 |
| Overall Oxygen and Fuel Requirement Parameters: | |
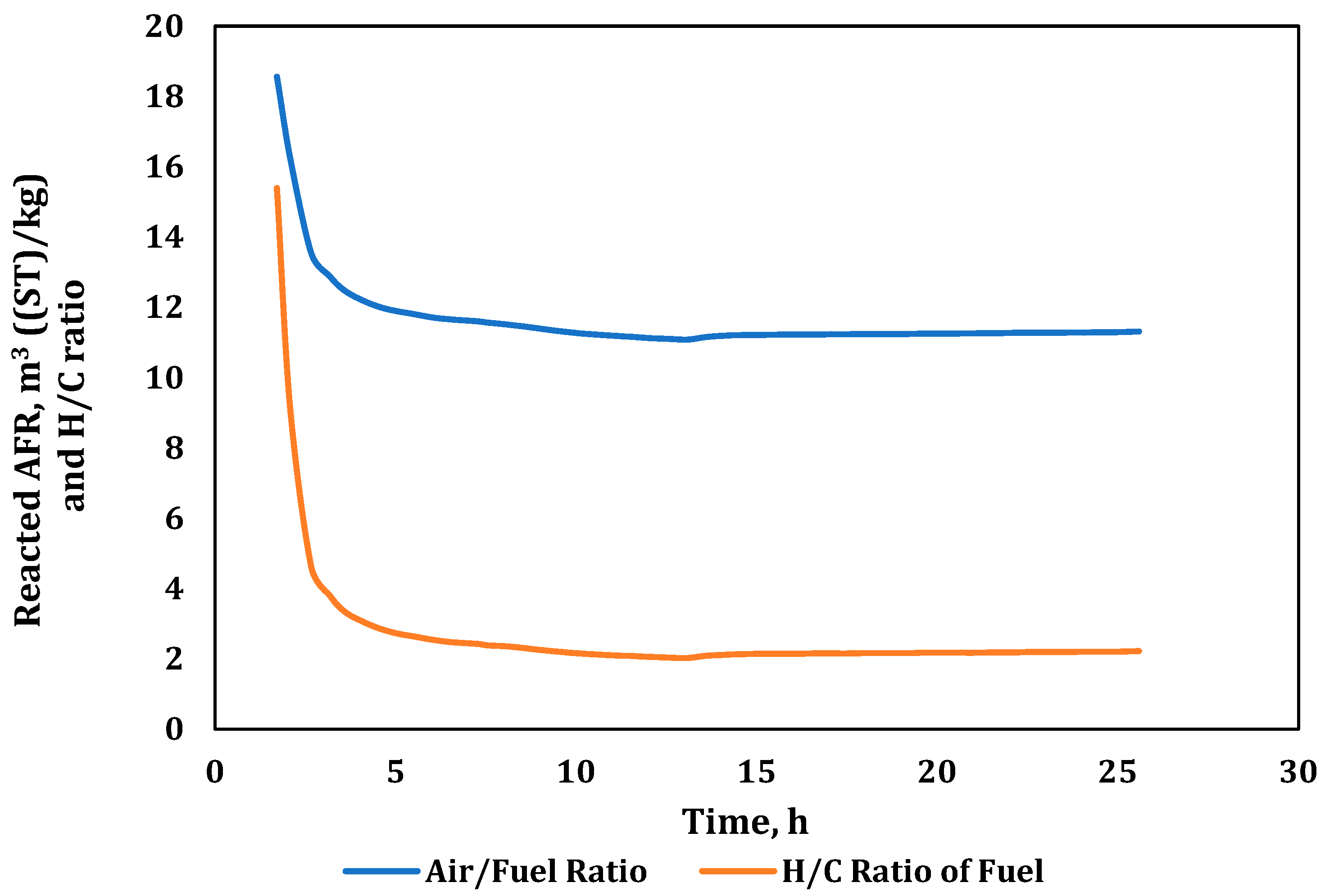
| Air to Fuel Ratio (m3(ST)/kg) | 11.31 |
| Oxygen to Fuel Ratio (m3(ST)/kg) | 2.47 |
| Air Requirement (m3(ST)/m3) | 238.7 |
| Fuel Requirement (kg/m3) | 21.11 |
| Air and Fuel Requirements Based on a (Burned) Tube Volume of 0.0113 (m3) | |
| Hydrogen to Carbon Ratio | 2.22 |
| Overall Oxygen Utilization (percent) | 99.3 |
| Overall (CO2 + CO)/CO Ratio | 7.21 |
| Overall (CO2 + CO)/N2 Ratio | 0.19 |
| Reacted Oxygen Forming COx (percent) | 62.7 |
| Total Fuel Gas Production (g) | 44.6 |
| Mass of Carbon in Fuel Gas (g) | 35.1 |
| Mass of Hydrogen in Fuel Gas (g) | 8.6 |
| Mass of Sulfur in Fuel Gas (g) | 0.9 |
| Total Mass of Oil Produced as Gas (g) | 282.5 |
| Component Production litres (ST) | |
| O2 | 4.3 |
| N2 | 2101.7 |
| CO | 54.6 |
| CO2 | 339.0 |
| CH4 | 17.1 |
| C2H4 | 1.8 |
| C2H6 | 5.5 |
| C3H6 | 3.1 |
| C3H8 | 4.7 |
| C4+ | 3.3 |
| H2 | 3.6 |
| H2S | 0.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fazlyeva, R.; Ursenbach, M.; Mallory, D.; Mehta, S.; Cheremisin, A.; Moore, G.; Spasennykh, M. In Situ Combustion of Heavy Oil within a Vuggy Carbonate Reservoir: Part I—Feasibility Study. Energies 2023, 16, 2233. https://doi.org/10.3390/en16052233
Fazlyeva R, Ursenbach M, Mallory D, Mehta S, Cheremisin A, Moore G, Spasennykh M. In Situ Combustion of Heavy Oil within a Vuggy Carbonate Reservoir: Part I—Feasibility Study. Energies. 2023; 16(5):2233. https://doi.org/10.3390/en16052233
Chicago/Turabian StyleFazlyeva, Rita, Matthew Ursenbach, Donald Mallory, Sudarshan (Raj) Mehta, Alexey Cheremisin, Gordon Moore, and Mikhail Spasennykh. 2023. "In Situ Combustion of Heavy Oil within a Vuggy Carbonate Reservoir: Part I—Feasibility Study" Energies 16, no. 5: 2233. https://doi.org/10.3390/en16052233
APA StyleFazlyeva, R., Ursenbach, M., Mallory, D., Mehta, S., Cheremisin, A., Moore, G., & Spasennykh, M. (2023). In Situ Combustion of Heavy Oil within a Vuggy Carbonate Reservoir: Part I—Feasibility Study. Energies, 16(5), 2233. https://doi.org/10.3390/en16052233







