Circular Economy and Green Chemistry: The Need for Radical Innovative Approaches in the Design for New Products
Abstract
1. Introduction
1.1. The Circular Economy
1.2. Green Chemistry
2. The Nexus between Green Chemistry and the Circular Economy
2.1. Green Chemistry in Practice
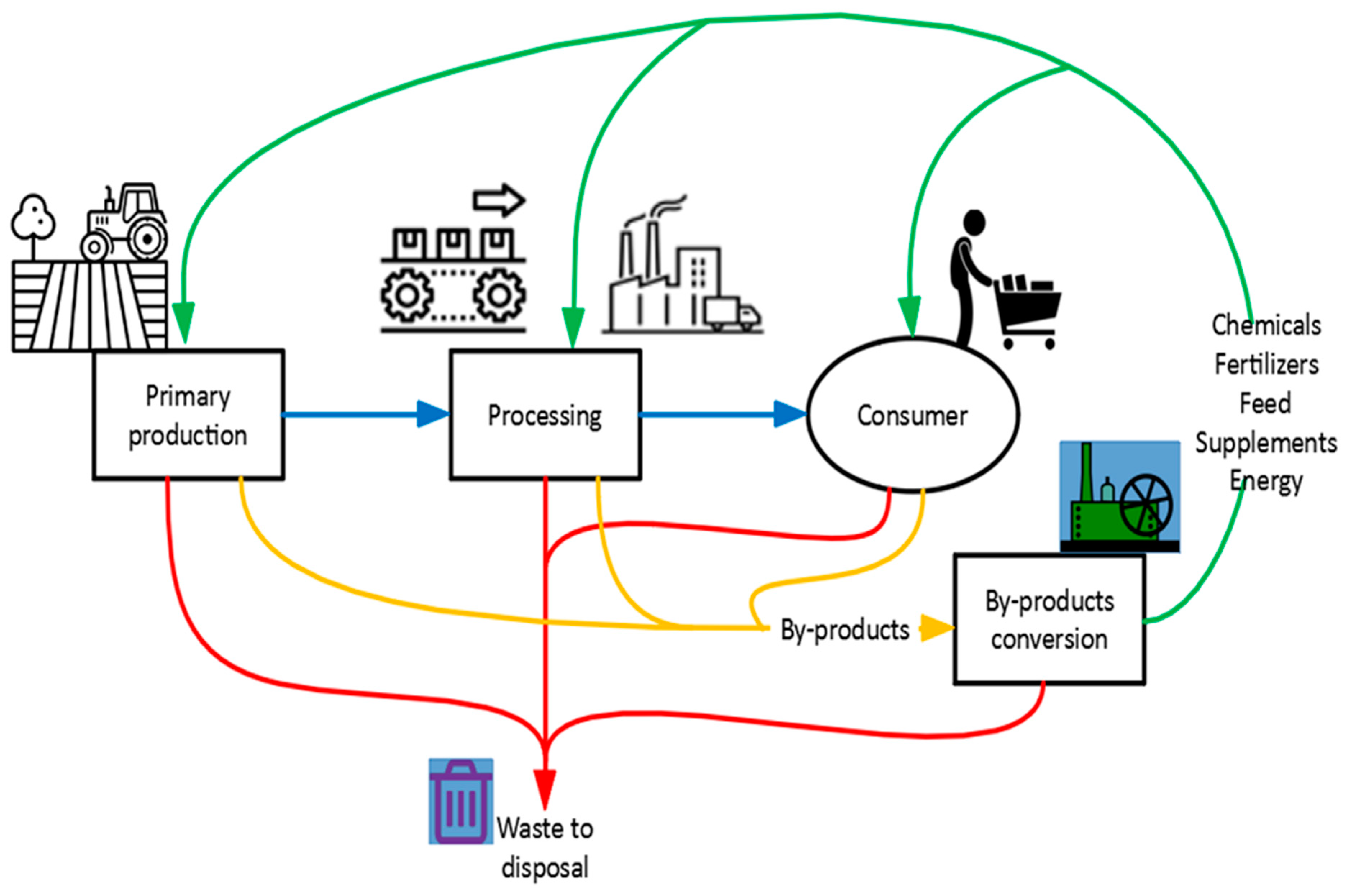
2.1.1. Waste Management Using a GC Approach
2.1.2. Waste Management–Production Stage (Chemical Synthesis Techniques)
2.1.3. Waste Management–Usage Stage (Recycling)
2.1.4. Waste Management–Disposal Stage (Waste to Energy and Value-Added Products)
2.1.5. Utilizing Minimum Energy Requirements
2.1.6. Use of Renewable Feedstocks
2.2. How Do We Move from Theory to Practice?
3. Policy and Structural Issues Related to CE and GC Integration and Adoption
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Commission. Closing the Loop—An EU Action Plan for the Circular Economy. 2015. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52015DC0614 (accessed on 11 January 2023).
- Ellen MacArthur Foundation. Ellen-MacArthur-Foundation-Towards-the-Circular-Economy—Vol. 1. Ellen MacArthur Foundation. 2013, p. 1. Available online: https://emf.thirdlight.com/link/x8ay372a3r11-k6775n/@/preview/1?o (accessed on 11 January 2023).
- Mancini, E.; Raggi, A. A review of circularity and sustainability in anaerobic digestion processes. J. Environ. Manag. 2021, 291, 112695. [Google Scholar] [CrossRef]
- Pansera, M. The Origins and Purpose of Eco-Innovation. Glob. Environ. 2016, 4, 128–155. [Google Scholar] [CrossRef]
- Stahel, W.R. The circular economy. Nature 2016, 531, 435–438. [Google Scholar] [CrossRef] [PubMed]
- Webster, K. A Circular Economy is about the Economy. Circ. Econ. Sustain. 2021, 1, 115–126. [Google Scholar] [CrossRef]
- WEF. The World Needs a Circular Economy. Help Us Make It Happen; World Economic Forum: Geneva, Switzerland, 2020. [Google Scholar]
- D’Amato, D.; Korhonen, J. Integrating the green economy, circular economy and bioeconomy in a strategic sustainability framework. Ecol. Econ. 2021, 188, 107143. [Google Scholar] [CrossRef]
- Kurowska-Susdorf, A.; Zwierżdżyński, M.; Bevanda, A.M.; Talić, S.; Ivanković, A.; Płotka-Wasylka, J. Green analytical chemistry: Social dimension and teaching. TrAC-Trends Anal. Chem. 2019, 111, 185–196. [Google Scholar] [CrossRef]
- Armenta, S.; Esteve-Turrillas, F.A.; Garrigues, S.; de la Guardia, M. Green Analytical Chemistry: Concepts, evolution, and recent developments. Green Approaches Chem. Anal. 2023, 1–37. [Google Scholar] [CrossRef]
- Raj, A.; Chowdhury, A.; Ali, S.W. Green chemistry: Its opportunities and challenges in colouration and chemical finishing of textiles. Sustain. Chem. Pharm. 2022, 27, 100689. [Google Scholar] [CrossRef]
- Adam, D.H.; Ende Supriyadi, Y.N.; Siregar, Z.M.E. Green manufacturing, green chemistry and environmental sustainability: A review. Int. J. Sci. Technol. Res. 2020, 9, 2209–2211. [Google Scholar]
- Smith, S.L.; Raynes, D.B.; Howard, K.L. National leadership and cross-sector collaboration could help overcome differences in stakeholder definitions of sustainable chemistry. Curr. Res. Green Sustain. Chem. 2022, 5, 100222. [Google Scholar] [CrossRef]
- Zimmerman, J.B.; Anastas, P.T.; Erythropel, H.C.; Leitner, W. Designing for a green chemistry future. Science 2020, 367, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Stahel, W.R.; Reday, G. The Potential for Substituting Manpower for Energy-Research Contract No 76/l3-V/343/78-EN; Commission of the European Communities: Brussels, Belgium, 1977. [Google Scholar]
- Chizaryfard, A.; Trucco, P.; Nuur, C. The transformation to a circular economy: Framing an evolutionary view. J. Evol. Econ. 2021, 31, 475–504. [Google Scholar] [CrossRef]
- Harris, S.; Martin, M.; Diener, D. Circularity for circularity’s sake? Scoping review of assessment methods for environmental performance in the circular economy. Sustain. Prod. Consum. 2021, 26, 172–186. [Google Scholar] [CrossRef]
- Yuan, Z.; Bi, J.; Moriguichi, Y. The circular economy: A new development strategy in China. J. Ind. Ecol. 2006, 10, 4–8. [Google Scholar] [CrossRef]
- McDowall, W.; Geng, Y.; Huang, B.; Barteková, E.; Bleischwitz, R.; Türkeli, S.; Kemp, R.; Doménech, T. Circular Economy Policies in China and Europe. J. Ind. Ecol. 2017, 21, 651–661. [Google Scholar] [CrossRef]
- Calisto Friant, M.; Vermeulen, W.J.V.; Salomone, R. A typology of circular economy discourses: Navigating the diverse visions of a contested paradigm. Resour. Conserv. Recycl. 2020, 161, 104917. [Google Scholar] [CrossRef]
- Hart, J.; Adams, K.; Giesekam, J.; Tingley, D.D.; Pomponi, F. Barriers and drivers in a circular economy: The case of the built environment. Procedia CIRP 2019, 80, 619–624. [Google Scholar] [CrossRef]
- Ghisellini, P.; Cialani, C.; Ulgiati, S. A review on circular economy: The expected transition to a balanced interplay of environmental and economic systems. J. Clean. Prod. 2016, 114, 11–32. [Google Scholar] [CrossRef]
- Genovese, A.; Pansera, M. The Circular Economy at a Crossroads: Technocratic Eco-Modernism or Convivial Technology for Social Revolution? Capital. Nat. Social. 2020, 32, 95–113. [Google Scholar] [CrossRef]
- Clark, J.H. Green chemistry: Challenges and opportunities. Green Chem. 1999, 1, 1–8. [Google Scholar] [CrossRef]
- Linthorst, J.A. An overview: Origins and development of green chemistry. Found. Chem. 2019, 12, 55–68. [Google Scholar] [CrossRef]
- Murphy, M.A. Early Industrial Roots of Green Chemistry and the history of the BHC Ibuprofen process invention and its Quality connection. Found. Chem. 2018, 20, 121–165. [Google Scholar] [CrossRef]
- De Marco, B.A.; Rechelo, B.S.; Tótoli, E.G.; Kogawa, A.C.; Salgado, H.R.N. Evolution of green chemistry and its multidimensional impacts: A review. Saudi Pharm. J. 2019, 27, 1–8. [Google Scholar] [CrossRef]
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Armstrong, L.B.; Rivas, M.C.; Douskey, M.C.; Baranger, A.M. Teaching students the complexity of green chemistry and assessing growth in attitudes and understanding. Curr. Opin. Green Sustain. Chem. 2018, 13, 61–67. [Google Scholar] [CrossRef]
- Anastas, P.T.; Bartlett, L.B.; Kirchhoff, M.M.; Williamson, T.C. The role of catalysis in the design, development, and implementation of green chemistry. Catal. Today 2000, 55, 11–22. [Google Scholar] [CrossRef]
- Rodriguez-padron, A.D.; Puente-santiago, A.; Alina, M.; Munoz-batista, M.; Luque, R. Environmental Catalysis: Present and Future. ChemCatChem 2019, 11, 18–38. [Google Scholar] [CrossRef]
- Abdussalam-Mohammed, W.; Ali, A.Q.; Errayes, A.O. Green Chemistry: Principles, Applications, and Disadvantages. Chem. Methodol. 2020, 4, 408–423. [Google Scholar] [CrossRef]
- Kühlborn, J.; Groß, J.; Opatz, T. Making natural products from renewable feedstocks: Back to the roots? Nat. Prod. Rep. 2020, 37, 380–424. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. ScienceDirect The greening of solvents: Towards sustainable organic synthesis. Curr. Opin. Green Sustain. 2019, 18, 13–19. [Google Scholar] [CrossRef]
- Chatel, G.; Varma, R.S. Ultrasound and microwave irradiation: Contributions of alternative physicochemical activation methods to Green Chemistry. Green Chem. 2019, 21, 6043–6050. [Google Scholar] [CrossRef]
- Ahmed, S.N.; Haider, W. Heterogeneous photocatalysis and its potential applications in water and wastewater treatment: A review. Nanotechnology 2018, 29, 342001. [Google Scholar] [CrossRef]
- Al-mamun, M.R.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Woodley, J.M. Role of Biocatalysis in Sustainable Chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef] [PubMed]
- Erythropel, H.C.; Zimmerman, J.B.; de Winter, T.M.; Petitjean, L.; Melnikov, F.; Lam, C.H.; Lounsbury, A.W.; Mellor, K.E.; Janković, N.Z.; Tu, Q.; et al. The Green ChemisTREE: 20 years after taking root with the 12 principles. Green Chem. 2018, 20, 1929–1961. [Google Scholar] [CrossRef]
- Jamarani, R.; Erythropel, H.C.; Nicell, J.A.; Leask, R.L.; Marić, M. How Green is Your Plasticizer? Polymers 2018, 10, 834. [Google Scholar] [CrossRef] [PubMed]
- Wasserbaur, R.; Sakao, T.; Milios, L. Interactions of governmental policies and business models for a circular economy: A systematic literature review. J. Clean. Prod. 2022, 337, 130329. [Google Scholar] [CrossRef]
- Arun, K.B.; Madhavan, A.; Anoopkumar, A.N.; Surendhar, A.; Liz Kuriakose, L.; Tiwari, A.; Sirohi, R.; Kuddus, M.; Rebello, S.; Kumar Awasthi, M.; et al. Integrated biorefinery development for pomegranate peel: Prospects for the production of fuel, chemicals and bioactive molecules. Bioresour. Technol. 2022, 362, 127833. [Google Scholar] [CrossRef]
- Mesa, J.; González-Quiroga, A.; Maury, H. Developing an indicator for material selection based on durability and environmental footprint: A Circular Economy perspective. Resour. Conserv. Recycl. 2020, 160, 104887. [Google Scholar] [CrossRef]
- Pivnenko, K. Waste Material Recycling: Assessment of Contaminants Limiting Recycling; Department of Environmental Engineering, Technical University of Denmark (DTU): Lyngby, Denmark, 2016. [Google Scholar]
- Hurst, G.A. Systems thinking approaches for international green chemistry education. Curr. Opin. Green Sustain. Chem. 2020, 21, 93–97. [Google Scholar] [CrossRef]
- Yilan, G.; Cordella, M.; Morone, P. Evaluating and managing the sustainability of investments in green and sustainable chemistry: An overview of sustainable finance approaches and tools. Curr. Opin. Green Sustain. Chem. 2022, 36, 100635. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, A.; Wang, W.Y.; Zhao, Y.L.; Chiang, P.C. Implementing Green Chemistry Principles for Circular Economy towards Sustainable Development Goals. Chem. Eng. Trans. 2021, 88, 955–960. [Google Scholar] [CrossRef]
- To, M.H.; Uisan, K.; Ok, Y.S.; Pleissner, D.; Lin, C.S.K. Recent trends in green and sustainable chemistry: Rethinking textile waste in a circular economy. Curr. Opin. Green Sustain. Chem. 2019, 20, 1–10. [Google Scholar] [CrossRef]
- Ali, S.S.; Elsamahy, T.; Abdelkarim, E.A.; Al-tohamy, R.; Kornaros, M.; Ruiz, A.; Zhao, T.; Li, F.; Sun, J. Biowastes for biodegradable bioplastics production and end-of-life scenarios in circular bioeconomy and biorefinery concept. Bioresour. Technol. 2022, 363, 127869. [Google Scholar] [CrossRef] [PubMed]
- Kaza, S.; Yao, L.; Bhada-Tata, P.; van Woerden, F. What a Waste 2.0; The World Bank: New York, NY, USA, 2018. [Google Scholar]
- Annamalai, S.; Chandrasekaran, K.; Shin, W.S.; Sundaram, M.; Khaleel, T.M. Beyond dumping: New strategies in the separation of preservative salt from tannery waste mixed salt and its reuse for tannery industrial application. Environ. Res. 2022, 214 Pt 2, 113885. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; Norton, M. Green chemistry and the plastic pollution challenge: Towards a circular economy. Green Chem. 2020, 22, 6310–6322. [Google Scholar] [CrossRef]
- He, R.; Sandoval-Reyes, M.; Scott, I.; Semeano, R.; Ferrão, P.; Matthews, S.; Small, M.J. Global knowledge base for municipal solid waste management: Framework development and application in waste generation prediction. J. Clean. Prod. 2022, 377, 134501. [Google Scholar] [CrossRef]
- Chavez-Rico, V.S.; Bodelier, P.L.E.; van Eekert, M.; Sechi, V.; Veeken, A.; Buisman, C. Producing organic amendments: Physicochemical changes in biowaste used in anaerobic digestion, composting, and fermentation. Waste Manag. 2022, 149, 177–185. [Google Scholar] [CrossRef]
- Anastas, P.T. Beyond Reductionist Thinking in Chemistry for Sustainability. Trends Chem. 2019, 1, 145–148. [Google Scholar] [CrossRef]
- Maria, C.; Góis, J.; Leitão, A. Challenges and perspectives of greenhouse gases emissions from municipal solid waste management in Angola. Energy Rep. 2020, 6, 364–369. [Google Scholar] [CrossRef]
- Pires, A.; Martinho, G. Waste hierarchy index for circular economy in waste management. Waste Manag. 2019, 95, 298–305. [Google Scholar] [CrossRef]
- Anastas, P.; Eghbali, N. Green Chemistry: Principles and Practice. Chem. Soc. Rev. 2010, 39, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Ardila-fierro, K.J.; Hernández, J.G. Sustainability Assessment of Mechanochemistry by Using the Twelve Principles of Green Chemistry. ChemSusChem 2021, 14, 2145–2162. [Google Scholar] [CrossRef]
- López-Lorente, Á.I.; Pena-Pereira, F.; Pedersen-Bjergaard, S.; Zuin, V.G.; Ozkan, S.A.; Psillakis, E. The ten principles of green sample preparation. TrAC Trends Anal. Chem. 2022, 148, 116530. [Google Scholar] [CrossRef]
- Sadatshojaei, E.; Wood, D.A. Water, the most accessible eco-friendly solvent, and extraction and separation agent. In Green Sustainable Process for Chemical and Environmental Engineering and Science: Green Solvents for Environmental Remediation; Elsevier: Amsterdam, The Netherlands, 2021; pp. 283–292. [Google Scholar] [CrossRef]
- Kim, D.; Nunes, S.P. Green solvents for membrane manufacture: Recent trends and perspectives. Curr. Opin. Green Sustain. Chem. 2021, 28, 100427. [Google Scholar] [CrossRef]
- Gulzar, T.; Farooq, T.; Kiran, S.; Ahmad, I.; Hameed, A. Green chemistry in the wet processing of textiles. In The Impact and Prospects of Green Chemistry for Textile Technology; Elsevier Ltd.: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Vandeponseele, A.; Chatel, G.; Draye, M.; Piot, C. Subcritical water and supercritical carbon dioxide: Efficient and selective eco-compatible solvents for coffee and coffee by-products valorization. Green Chem. 2020, 22, 8544–8571. [Google Scholar] [CrossRef]
- Ivanković, A.; Dronjić, A.; Bevanda, A.M.; Talić, S. Review of 12 Principles of Green Chemistry in Practice. Int. J. Sustain. Green Energy 2017, 6, 39–48. [Google Scholar] [CrossRef]
- Hairprint. Biological Hair Color. Hairprint. 2017. Available online: https://www.myhairprint.com/ (accessed on 11 January 2023).
- Elevance. Elevance Biorefinery Metathesis Technology. 2022. Available online: https://elevance.com/technology/ (accessed on 11 January 2023).
- Beghetto, V.; Sole, R.; Buranello, C.; Al-abkal, M. Recent Advancements in Plastic Packaging Recycling: A Mini-Review. Materials 2021, 14, 4782. [Google Scholar] [CrossRef]
- Bracquené, E.; Martinez, M.G.; Wagner, E.; Wagner, F.; Boudewijn, A.; Peeters, J.; Duflou, J. Quantifying the environmental impact of clustering strategies in waste management: A case study for plastic recycling from large household appliances. Waste Manag. 2021, 126, 497–507. [Google Scholar] [CrossRef]
- Rahimi, A.R.; Garciá, J.M. Chemical recycling of waste plastics for new materials production. Nat. Rev. Chem. 2017, 1, 46. [Google Scholar] [CrossRef]
- Lee, A.; Liew, M.S. Tertiary recycling of plastics waste: An analysis of feedstock, chemical and biological degradation methods. J. Mater. Cycles Waste Manag. 2021, 23, 32–43. [Google Scholar] [CrossRef]
- Thiounn, T.; Smith, R.C. Advances and approaches for chemical recycling of plastic waste. J. Polym. Sci. 2020, 58, 1347–1364. [Google Scholar] [CrossRef]
- Huang, J.; Veksha, A.; Chan, W.P.; Giannis, A.; Lisak, G. Chemical recycling of plastic waste for sustainable material management: A prospective review on catalysts and processes. Renew. Sustain. Energy Rev. 2022, 154, 111866. [Google Scholar] [CrossRef]
- Achilias, D.S.; Karayannidis, G.P. The Chemical Recycling of Pet in the Framework of Sustainable Development. Water Air Soil Pollut. Focus 2004, 4, 385–396. [Google Scholar] [CrossRef]
- Assadi, R.; Colin, X.; Verdu, J. Irreversible structural changes during PET recycling by extrusion. Polymer 2004, 45, 4403–4412. [Google Scholar] [CrossRef]
- WAT. A World Where Resources Are Kept in Constant Circulation, DRIVING Economic, Social and Environmental Benefits. Worn again Technologies. 2021. Available online: https://wornagain.co.uk/ (accessed on 11 January 2023).
- APK. Newcycling®—An Economic and Ecological Recycling Technology for a Real Circular Economy for Plastics. 2022. Available online: https://www.apk.group/en/newcycling/ (accessed on 11 January 2023).
- CreaCycle. The CreaSolv® Process. CreaCycleGmbH. 2010. Available online: https://www.creacycle.de/en/the-process.html (accessed on 11 January 2023).
- Li, H.; Marie Kirkelund, G. Pulsed stirring for energy efficiency improvements during electrodialytic extraction of As, Cd, Cr, Cu, Pb, and Zn from municipal solid waste incineration fly ash and air pollution control residue. Sep. Purif. Technol. 2022, 290, 120835. [Google Scholar] [CrossRef]
- Li, H.; Aguirre-Villegas, H.A.; Allen, R.D.; Bai, X.; Benson, C.H.; Beckham, G.T.; Bradshaw, S.L.; Brown, J.L.; Brown, R.C.; Cecon, V.S.; et al. Expanding plastics recycling technologies: Chemical aspects, technology status and challenges. Green Chem. 2022, 24, 8899–9002. [Google Scholar] [CrossRef]
- European Chemical Agency. Annual Report. 2021. Available online: https://echa.europa.eu/documents/10162/11872732/mb_05_2022_2_annual_report_2021_mb65_en.pdf/7688a1e9-5d23-59fb-213c-2bd940c052ff?t=1660039291431 (accessed on 11 January 2023).
- De Boni, A.; Melucci, F.M.; Acciani, C.; Roma, R. Community composting: A multidisciplinary evaluation of an inclusive, participative, and eco-friendly approach to biowaste management. Clean. Environ. Syst. 2022, 6, 100092. [Google Scholar] [CrossRef]
- Zuin, V.G. Circularity in green chemical products, processes and services: Innovative routes based on integrated eco-design and solution systems. Curr. Opin. Green Sustain. Chem. 2016, 2, 40–44. [Google Scholar] [CrossRef]
- Orejuela-Escobar, L.M.; Landázuri, A.C.; Goodell, B. Second generation biorefining in Ecuador: Circular bioeconomy, zero waste technology, environment and sustainable development: The nexus. J. Bioresour. Bioprod. 2021, 6, 83–107. [Google Scholar] [CrossRef]
- Nair, L.G.; Agrawal, K.; Verma, P. An overview of sustainable approaches for bioenergy production from agro-industrial wastes. Energy Nexus 2022, 6, 100086. [Google Scholar] [CrossRef]
- Kefalew, T.; Lami, M. Biogas and bio-fertilizer production potential of abattoir waste: Implication in sustainable waste management in Shashemene City, Ethiopia. Heliyon 2021, 7, e08293. [Google Scholar] [CrossRef] [PubMed]
- Masilela, P.; Pradhan, A. Systematic literature review of the sustainability and environmental performance of dark fermentative biohydrogen production. J. Clean. Prod. 2022, 372, 133541. [Google Scholar] [CrossRef]
- Zhou, P.; Gao, S.; Wang, B.; Wang, Y.; Li, C.; Wang, Y.; Sun, B. Influence of hydrogen fuel cell temperature safety on bus driving characteristics and stack heating mode. Int. J. Hydrogen Energy, 2022, in press. [CrossRef]
- Ozawa, A.; Kudoh, Y.; Murata, A.; Honda, T.; Saita, I.; Takagi, H. Hydrogen in low-carbon energy systems in Japan by 2050: The uncertainties of technology development and implementation. Int. J. Hydrogen Energy 2018, 43, 18083–18094. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, H.; Shui, X.; Li, Y.; Zhang, Z. Research progress of additives in photobiological hydrogen production system to enhance biohydrogen. Bioresour. Technol. 2022, 362, 127787. [Google Scholar] [CrossRef]
- Ananthi, V.; Ramesh, U.; Balaji, P.; Kumar, P.; Govarthanan, M.; Arun, A. A review on the impact of various factors on biohydrogen production. Int. J. Hydrogen Energy, 2022, in press. [CrossRef]
- Ranieri, L.; Mossa, G.; Pellegrino, R.; Digiesi, S. Energy recovery from the organic fraction of municipal solid waste: A real options-based facility assessment. Sustainability 2018, 10, 368. [Google Scholar] [CrossRef]
- Tampio, E.; Ervasti, S.; Rintala, J. Characteristics and agronomic usability of digestates from laboratory digesters treating food waste and autoclaved food waste. J. Clean. Prod. 2015, 94, 86–92. [Google Scholar] [CrossRef]
- Thiriet, P.; Bioteau, T.; Tremier, A. Optimization method to construct micro-anaerobic digesters networks for decentralized biowaste treatment in urban and peri-urban areas. J. Clean. Prod. 2020, 243, 118478. [Google Scholar] [CrossRef]
- Rena; Mohammed Bin Zacharia, K.; Yadav, S.; Machhirake, N.P.; Kim, S.H.; Lee, B.D.; Jeong, H.; Singh, L.; Kumar, S.; Kumar, R. Bio-hydrogen and bio-methane potential analysis for production of bio-hythane using various agricultural residues. Bioresour. Technol. 2020, 309, 123297. [Google Scholar] [CrossRef]
- Prashanth Kumar, C.; Rena Meenakshi, A.; Khapre, A.S.; Kumar, S.; Anshul, A.; Singh, L.; Kim, S.H.; Lee, B.D.; Kumar, R. Bio-Hythane production from organic fraction of municipal solid waste in single and two stage anaerobic digestion processes. Bioresour. Technol. 2019, 294, 122220. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, G.; Ripa, M.; Ulgiati, S. Chemicals from biomass: Technological versus environmental feasibility. A review. Biofuels Bioprod. Biorefining 2017, 11, 195–214. [Google Scholar] [CrossRef]
- López-Gómez, J.P.; Pérez-Rivero, C.; Venus, J. Valorisation of solid biowastes: The lactic acid alternative. Process Biochem. 2020, 99, 222–235. [Google Scholar] [CrossRef]
- Santana-Mayor, Á.; Rodríguez-Ramos, R.; Herrera-Herrera, A.V.; Socas-Rodríguez, B.; Rodríguez-Delgado, M.Á. Deep eutectic solvents. The new generation of green solvents in analytical chemistry. TrAC-Trends Anal. Chem. 2021, 134, 116108. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Bystrzejewski, M.; De Adhikari, A.; Huczko, A.; Wang, N. Methods for the conversion of biomass waste into value-added carbon nanomaterials: Recent progress and applications. Prog. Energy Combust. Sci. 2022, 92, 101023. [Google Scholar] [CrossRef]
- Kaya, S.I.; Cetinkaya, A.; Ozkan, S.A. Green analytical chemistry approaches on environmental analysis. Trends Environ. Anal. Chem. 2022, 33, e00157. [Google Scholar] [CrossRef]
- Wang, Z.; Shen, D.; Wu, C.; Gu, S. State-of-the-art on the production and application of carbon nanomaterials from biomass. Green Chem. 2018, 20, 5031–5057. [Google Scholar] [CrossRef]
- Goswami, L.; Kayalvizhi, R.; Dikshit, P.K.; Sherpa, K.C.; Roy, S.; Kushwaha, A.; Kim, B.S.; Banerjee, R.; Jacob, S.; Rajak, R.C. A critical review on prospects of bio-refinery products from second and third generation biomasses. Chem. Eng. J. 2022, 448, 137677. [Google Scholar] [CrossRef]
- Awasthi, M.K.; Sarsaiya, S.; Wainaina, S.; Rajendran, K.; Awasthi, S.K.; Liu, T.; Duan, Y.; Jain, A.; Sindhu, R.; Binod, P.; et al. Techno-economics and life-cycle assessment of biological and thermochemical treatment of bio-waste. Renew. Sustain. Energy Rev. 2021, 144, 110837. [Google Scholar] [CrossRef]
- Wen, C.; Yin, A.; Dai, W.L. Recent advances in silver-based heterogeneous catalysts for green chemistry processes. Appl. Catal. B Environ. 2014, 160–161, 730–741. [Google Scholar] [CrossRef]
- Verma, C.; Quraishi, M.A.; Ebenso, E.E. Microwave and ultrasound irradiations for the synthesis of environmentally sustainable corrosion inhibitors: An overview. Sustain. Chem. Pharm. 2018, 10, 134–147. [Google Scholar] [CrossRef]
- Ratti, R. Industrial applications of green chemistry: Status, Challenges and Prospects. SN Appl. Sci. 2020, 2, 263. [Google Scholar] [CrossRef]
- Anastas, P.T.; Beach, E.S. Green chemistry: The emergence of a transformative framework. Green Chem. Lett. Rev. 2007, 1, 9–24. [Google Scholar] [CrossRef]
- Collins, J.; Gourdin, G.; Qu, D. Modern Applications of Green Chemistry: Renewable Energy. Green Chem. Incl. Approach 2018, 771–860. [Google Scholar] [CrossRef]
- Leitner, W.; Quadrelli, E.A.; Schlögl, R. Harvesting renewable energy with chemistry. Green Chem. 2017, 3, 2015–2016. [Google Scholar] [CrossRef]
- Çelik, D.; Yıldız, M. Investigation of hydrogen production methods in accordance with green chemistry principles. Int. J. Hydrogen Energy 2017, 42, 23395–23401. [Google Scholar] [CrossRef]
- Dyecoo. DyeCoo. 2022. Available online: https://dyecoo.com/ (accessed on 11 January 2023).
- Melero, J.A.; Iglesias, J.; Garcia, A. Biomass as renewable feedstock in standard refinery units. Feasibility, opportunities and challenges. Energy Environ. Sci. 2012, 5, 7393–7420. [Google Scholar] [CrossRef]
- Bukartyk, M.; Zholobko, O.; Wu, X. Green Synthesis of Soy Protein Nanocomposites: E ff ects of Cross- Linking and Clay Nanoparticles on the Mechanical Performance. ACS Omega 2022, 7, 5883–5893. [Google Scholar] [CrossRef]
- Swain, S.N.; Biswal, S.M.; Nanda, P.K.; Nayak, P.L. Biodegradable Soy-Based Plastics: Opportunities and Challenges. J. Environ. Polym. Degrad. 2004, 12, 35–42. [Google Scholar] [CrossRef]
- Chen, N.; Lin, Q.; Zheng, P.; Rao, J.; Zeng, Q.; Sun, J. A sustainable bio-based adhesive derived from defatted soy flour and epichlorohydrin. Wood Sci. Technol. 2019, 53, 801–817. [Google Scholar] [CrossRef]
- Samson, D.O.; Mat Jafri, M.Z.; Hashim, R.; Sulaiman, O.; Aziz, M.Z.A.; Yusof, M.F.M.; Shukri, A. Rhizophora spp. Particleboards incorporating defatted soy flour bonded with NaOH/IA-PAE: Towards a water equivalent phantom material. Radiat. Phys. Chem. 2020, 176, 109057. [Google Scholar] [CrossRef]
- Pfaltzgra, L.A.; De, M.; Cooper, E.C.; Budarin, V.; Clark, H. Food waste biomass: A resource for high-value chemicals. Green Chem. 2013, 15, 307–314. [Google Scholar] [CrossRef]
- Kazzaz, A.E.; Feizi, Z.H.; Fatehi, P. Grafting strategies for hydroxy groups of lignin for producing materials. Green Chem. 2019, 21, 5714–5752. [Google Scholar] [CrossRef]
- Österberg, M.; Sipponen, M.H.; Mattos, B.D.; Rojas, O.J. Spherical lignin particles: A review on their sustainability and applications. Green Chem. 2020, 22, 2712–2733. [Google Scholar] [CrossRef]
- Shirvan, A.R.; Shakeri, M.; Bashari, A. Recent advances in application of chitosan and its derivatives in functional finishing of textiles. In The Impact and Prospects of Green Chemistry for Textile Technology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 107–133. [Google Scholar] [CrossRef]
- Raj, T.; Chandrasekhar, K.; Morya, R.; Kumar Pandey, A.; Jung, J.H.; Kumar, D.; Singhania, R.R.; Kim, S.H. Critical challenges and technological breakthroughs in food waste hydrolysis and detoxification for fuels and chemicals production. Bioresour. Technol. 2022, 360, 127512. [Google Scholar] [CrossRef]
- NatureWorks LLC. NatureWorks. 2022. Available online: https://www.natureworksllc.com/About-NatureWorks (accessed on 11 January 2023).
- The Coca-Cola Company. Coca-Cola Collaborates with Tech Partners to Create Bottle Prototype Made from 100% Plant-Based Sources. Thecocacolacompany. 2021. Available online: https://www.coca-colacompany.com/news/100-percent-plant-based-plastic-bottle (accessed on 11 January 2023).
- Brzeska, J. A Brief Introduction to the Polyurethanes According to the Principles of Green Chemistry. Processes 2021, 9, 1929. [Google Scholar] [CrossRef]
- Preka, R.; Fiorentino, G.; De Carolis, R.; Barberio, G. The challenge of plastics in a circular perspective. Front. Sustain. Cities 2022, 4, 920242. [Google Scholar] [CrossRef]
- Domínguez de María, P. Biocatalysis, sustainability, and industrial applications: Show me the metrics. Curr. Opin. Green Sustain. Chem. 2021, 31, 100514. [Google Scholar] [CrossRef]
- NewLight. From Greenhouse Gas to Regenerative Materials That Improve the World. 2022. Available online: https://www.newlight.com/ (accessed on 11 January 2023).
- Keijer, T.; Bakker, V.; Slootweg, J.C. Circular chemistry to enable a circular economy. Nat. Chem. 2019, 11, 190–195. [Google Scholar] [CrossRef]
- Scafà, M.; Carbonari, S.; Papetti, A.; Rossi, M.; Germani, M. A new method for Product Service System: The case of urban waste management. Procedia CIRP 2018, 73, 67–72. [Google Scholar] [CrossRef]
- Kurpiela, S.; Teuteberg, F. Strategic planning of product-service systems: A systematic literature review. J. Clean. Prod. 2022, 338, 130528. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Lathifah, A. Ordering and waste reuse decisions in a make-to-order system under demand uncertainty. Eur. J. Oper. Res. 2022, 303, 1290–1303. [Google Scholar] [CrossRef]
- Meglin, R.; Kytzia, P.S.; Habert, P.G. Regional environmental-economic assessment of building materials to promote circular economy: Comparison of three Swiss cantons. Resour. Conserv. Recycl. 2022, 181, 106247. [Google Scholar] [CrossRef]
- Palmer, M.; Truong, Y. The Impact of Technological Green New Product Introductions on Firm Profitability. Ecol. Econ. 2017, 136, 86–93. [Google Scholar] [CrossRef]
- Gaona, S.D. The Utility of the Toxic Release Inventory (TRI) in Tracking Implementation and Environmental Impact of Industrial Green Chemistry Practices in the United States. In Green Chemistry; Saleh, H.E.-D.M., Koller, M., Eds.; IntechOpen: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef]
- Veleva, V.R.; Cue, B.W. The role of drivers, barriers, and opportunities of green chemistry adoption in the major world markets. Curr. Opin. Green Sustain. Chem. 2019, 19, 30–36. [Google Scholar] [CrossRef]
- Jiahuey, Y.; Liu, Y.; Yu, Y. Measuring green growth performance of China’s chemical industry. Resour. Conserv. Recycl. 2019, 149, 160–167. [Google Scholar] [CrossRef]
- Karl, H.; Jim, L.; Jane, C.; Meet the 2020 Consumers Driving Change. IBM Institute for Business Value. 2020. Available online: https://www.ibm.com/downloads/cas/EXK4XKX8 (accessed on 11 January 2023).
- Veleva, V.R.; Cue, B.W.; Todorova, S.; Thakor, H.; Mehta, N.H.; Padia, K.B. Benchmarking green chemistry adoption by the Indian pharmaceutical supply chain. Green Chem. Lett. Rev. 2018, 11, 439–456. [Google Scholar] [CrossRef]
- Fernandez Rivas, D.; Cintas, P. On an intensification factor for green chemistry and engineering: The value of an operationally simple decision-making tool in process assessment. Sustain. Chem. Pharm. 2022, 27, 100651. [Google Scholar] [CrossRef]
- Borchert, F.; Beronius, A.; Ågerstrand, M. Characterisation and analysis of key studies used to restrict substances under REACH. Environ. Sci. Eur. 2022, 34, 83. [Google Scholar] [CrossRef]
- Goh, H.Y.; Wen, W.; Wong, C.; Ong, Y.Y. A Study To Reduce Chemical Waste Generated in Chemistry Teaching Laboratories. J. Chem. Educ. 2020, 97, 87–96. [Google Scholar] [CrossRef]
- Yadav, R.; Pathak, G.S. Young consumers’ intention towards buying green products in a developing nation: Extending the theory of planned behavior. J. Clean. Prod. 2016, 135, 732–739. [Google Scholar] [CrossRef]
- Licence, P.; Ke, J.; Sokolova, M.; Ross, S.K.; Poliakoff, M. Chemical reactions in supercritical carbon dioxide: From laboratory to commercial plant. Green Chem. 2003, 5, 99–104. [Google Scholar] [CrossRef]
- Han, X.; Poliakoff, M. Continuous reactions in supercritical carbon dioxide: Problems, solutions and possible ways forward. Chem. Soc. Rev. 2012, 41, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- IMF. World Economic Outlook Countering the Cost of Living Crisis; IMF: Washington, DC, USA, 2022. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ncube, A.; Mtetwa, S.; Bukhari, M.; Fiorentino, G.; Passaro, R. Circular Economy and Green Chemistry: The Need for Radical Innovative Approaches in the Design for New Products. Energies 2023, 16, 1752. https://doi.org/10.3390/en16041752
Ncube A, Mtetwa S, Bukhari M, Fiorentino G, Passaro R. Circular Economy and Green Chemistry: The Need for Radical Innovative Approaches in the Design for New Products. Energies. 2023; 16(4):1752. https://doi.org/10.3390/en16041752
Chicago/Turabian StyleNcube, Amos, Sandile Mtetwa, Mahak Bukhari, Gabriella Fiorentino, and Renato Passaro. 2023. "Circular Economy and Green Chemistry: The Need for Radical Innovative Approaches in the Design for New Products" Energies 16, no. 4: 1752. https://doi.org/10.3390/en16041752
APA StyleNcube, A., Mtetwa, S., Bukhari, M., Fiorentino, G., & Passaro, R. (2023). Circular Economy and Green Chemistry: The Need for Radical Innovative Approaches in the Design for New Products. Energies, 16(4), 1752. https://doi.org/10.3390/en16041752







