Abstract
Shipping accounts for about 3% of global CO2 emissions. In order to achieve the target set by the Paris Agreement, IMO introduced their GHG strategy. This strategy envisages 50% emission reduction from international shipping by 2050, compared with 2008. This target cannot be fulfilled if conventional fuels are used. Amongst others, hydrogen is considered to be one of the strong candidates as a zero-emissions fuel. Yet, concerns around the safety of its storage and usage have been formulated and need to be addressed. “Safety”, in this article, is defined as the control of recognized hazards to achieve an acceptable level of risk. This article aims to propose a new way of comparing two systems with regard to their safety. Since safety cannot be directly measured, fuzzy set theory is used to compare linguistic terms such as “safer”. This method is proposed to be used during the alternative design approach. This approach is necessary for deviations from IMO rules, for example, when hydrogen should be used in shipping. Additionally, the properties of hydrogen that can pose a hazard, such as its wide flammability range, are identified.
1. Introduction
In 2018 shipping accounted for about 1 million tons or 2.89% of global CO2 emissions [1]. Since 80% of global trade is conducted by ship [2], shipping is the most efficient method of transportation. Nonetheless, shipping has to reduce its emissions to reach the defossilization targets set by the Paris Agreement. As a first step, the International Maritime Organization (IMO) regulations will require a reduction in GHG emissions of 50% by 2050 compared with 2008, while pursuing efforts to phase them out [3]. In order to fulfill these requirements, hydrogen, in its different storage forms, such as pressurized, liquified or as LOHC or metal hydride, amongst others, is often discussed as a fossil-free fuel for shipping or as a possibility to import energy from energy-rich countries. Hydrogen does not contain carbon, thus no CO2 or CO is emitted, and the only critical emission is NOX [4].
Despite these discussions, IMO regulations do not include hydrogen, so it may not be used in shipping today. However, the IMO established a mechanism, called the “alternative design approach” [5,6], intended to allow innovation without the corresponding rules being in place. During this “alternative design approach” an equivalent level of technological safety to that of existing rules and technologies should be demonstrated. The “alternative design approach”, on the other hand, does not include methods, how safety can be measured, how to make it comparable or even a definition of safety. That is why this article will propose both approaches.
This article proposes a method utilizing fuzzy set theory to compare hydrogen systems to conventional systems with regard to their safety. Beforehand, the relevant properties and safety aspects of hydrogen that can be used for such a comparison, such as the cryogenic storage temperature, are identified. Then, relevant IMO regulations, which demand such a comparison for hydrogen applications, are introduced. These regulations are then followed by ongoing projects in shipping that already use hydrogen safely.
Definitions of Safety, Hazard and Risk
Although the slogan “safety first” is displayed on most deckhouses on ships, no common definition of the term “safety” in shipping exists. However, in order to compare the safety properties of hydrogen and LNG, the terms “hazard”, “risk” and “safety” have to be defined. This article will use a definition established by McGuiness et al. [7]: ““Safety” can be defined as the control of recognized hazards to achieve an acceptable level of risk”.
On the basis of the ISO/IEC Guide 51:2014 [8], a hazard is defined as a “potential source of harm” to people, property or the environment.
In Engebø et al. [9], risk is defined as the “combination of the probability of occurrence of harm and the severity of that harm”.
2. Materials and Methods
This article compares the properties of hydrogen and LNG relevant to safety and proposes a method to compare their safety rationally. Accordingly, this chapter introduces the literature relevant to the proposed method.
Klebanoff et al. [10] compared hydrogen’s and LNG’s properties for a specific ship. To the authors’ knowledge, a general comparison has not been published before.
After the different criteria that are necessary to compare the safety of the systems are identified, the proposed method uses a pairwise comparison to assign a weighting to those criteria. These criteria can then be used by experts in the field or backed with data from the literature to allow for a comparison of the corresponding safety of the systems to be compared using linguistic terms. These linguistic terms are then translated into fuzzy numbers to account for the uncertainty of linguistic terms. Using the weighting from the pairwise comparison, the fuzzy numbers for each criterion can be combined to obtain a comparison for the whole system.
The pairwise comparison, proposed for the weighting of the criteria, is widely used in engineering for decision making, for example in the analytic hierarchy process, as described by Saaty [11]. It has also found its way into engineering text books, such as that by Feldhusen et al. [12].
After the pairwise comparison, the proposed method also uses the fuzzy set theory to make unsharp linguistic terms such as “a little safer” better comparable. The fuzzy set theory was introduced by Zadeh in 1965 [13,14,15] and has been used in very different fields of engineering. Ehlers et al. [16] used it to compare the safety effects of cooperative intelligent transport systems and Rahman et al. [17] used fuzzy sets to handle uncertainties in failure probabilities for a marine logistics risk model for offshore oil and gas operations.
3. Results
3.1. Properties of Hydrogen Related to Safety
Hydrogen is the most abundant element in the universe, yet hydrogen gas is not directly available as a resource on Earth [18]. It is an odorless, colorless and tasteless gas and is about fourteen times lighter than air [19]. Hydrogen’s low weight allows it to rapidly rise and diffuse in air [20] and its small molecular size allows it to permeate through materials more easily [21].
Hydrogen has a very high gravimetric energy density of 33.3 kWh/kg, but due to its low density the volumetric energy density is only 3 kWh/m3 (at normal temperature and pressure) [22]. By compressing the volumetric energy, the density increases to 776 kWh/m3 at 350 bar or 1.309 kWh/m3 at 700 bar [23]. The energy density can be further increased to 2.358 kWh/m3 by liquefying hydrogen at a temperature of 20 K (−253 °C) [22]. Cryo-compressed storage combines the advantages of compressed and liquified hydrogen by storing liquid hydrogen in an insulated pressure vessel designed for up to 350 bar. This storage offers a high storage density and reduced boil-of-losses [24]. A comparison of the energy density of different energy carriers can be found in Table 1 and Figure 1.

Table 1.
Comparison of the energy density of different energy carriers.
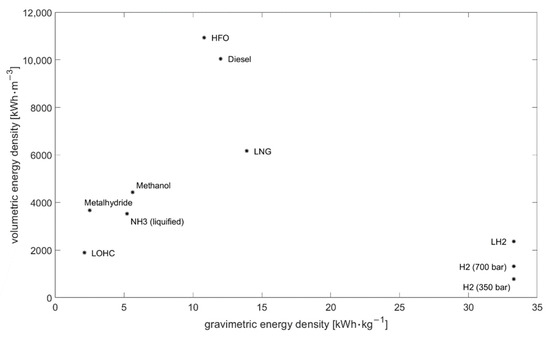
Figure 1.
Energy density of different energy carriers.
The boiling point of hydrogen at normal pressure is 20 K and its melting point is 14 K, both dependent on the pressure [22]. The triple point at which all three phases exist has a temperature of 13.8 and a pressure of 7.2 kPa (72 mbar) [22]. Increasing the pressure above the critical point of 33 K and 1296 MPa (12.96 bar) has no further influence on the boiling point [22]. These points can also be seen in Figure 2.
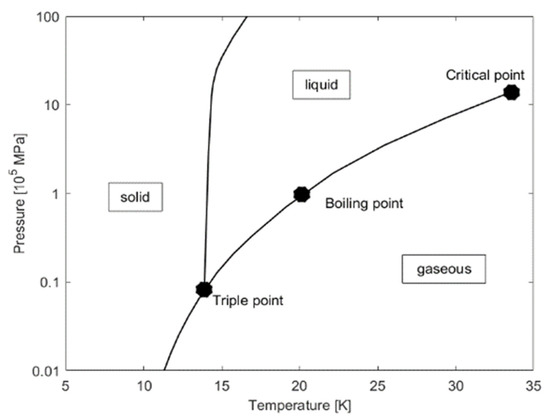
Figure 2.
Phase diagram of hydrogen [21].
Although hydrogen has a high auto-ignition temperature of 858 K [30], it has a very low minimum ignition energy of only 0.02 mJ at the stoichiometric mixture of 29.5% in air [22]. Besides the stochiometric mixture, hydrogen is flammable in concentrations from 4% to 75% [29]. A hydrogen flame has a high temperature of up to 2318 K, depending on the mixture, but emits very little radiation, leading to an invisible flame [22].
The high laminar burning velocity of a maximum of 3.46 m/s results in a flame front velocity of up to 24 m/s. Since most flames are not laminar but turbulent, realistic flames can reach a burning velocity of several hundreds of m/s. The detonation velocity can reach 2000 m/s, and the occurrence of a detonation is favored by the high flame speed, but limited to concentrations between 18 and 59 vol% [22].
3.2. Hazards Arising from Hydrogen
3.2.1. Hydrogen in General
The most important hazard of hydrogen is its very high flammability. With its high range of flammable concentrations (4–75%), it is often considered more dangerous than natural gas, which is already widely used in shipping. About 30% of all new ship orders use liquefied natural gas (LNG) as a fuel [31]. Compared with the flammability limits of methane (5.3–15% [10]), the main component of LNG hydrogen has a much wider range of flammability and is thus more dangerous in general, but realistically the lower flammability limit (LFL) is more relevant than the upper flammability limit (UFL). Usually, the gas will leak out of the system into the surrounding air, due to overpressure in the gas system. Thus, a leakage of air into the system is unlikely. Gas from a leak will diffuse quickly and reach the LFL first. All safety systems, such as detection and ventilation, are designed to prevent the atmosphere from reaching the LFL. Hydrogen’s LFL of 4% is very close to methane’s LFL of 5.3%, resulting in a similar ignition risk.
Hydrogen has an autoignition temperature of 858 K and a minimum ignition energy of 0.02 mJ [22], whereas methane has an autoignition temperature of 813 K [10] and a minimum ignition energy of 0.29 mJ [32]. The autoignition temperatures are very similar, but the minimum ignition energy seems to be significantly lower for hydrogen. The weak ignition source of electrostatic discharge by a human body contains around 10 mJ [22]. If we consider that such a weak ignition source would ignite both gases, the difference appears less relevant.
The invisible flame of hydrogen, which emits little radiation, causes difficulties in flame detection. Usually, infrared flame detectors are used in safety systems. Since hydrogen flames emit primarily UV radiation, these sensors cannot be used to detect a hydrogen flame. A pure UV sensor can be used to detect hydrogen flame, but is prone to false alarms from other UV sources, such as the sun. To prevent false alarms, multi-spectrum infrared (MIR) sensors that use a combination of software analysis and IR filters are used to detect hydrogen flames [33].
Another related risk is that of explosions. Knowing that there are differences, in this article the term “explosions” will also include deflagrations and detonations. Similar to the flammability, the concentrations in which a gas can explode are given in a range from the lower explosive limit (LEL) to the upper explosive limit (UEL). Again, the lower limit is more relevant, because safety systems have to be designed in a way to prevent the atmosphere from reaching the lower limit. Hydrogen is explosive in range from 18.3 to 59.0 vol%, whereas methane has a range from 6.3 to 13.5 vol% [10]. Again, hydrogen has a wider hazard range, due to its broader explosion limit. However, methane reaches its LEL much earlier than hydrogen, thereby posing a greater danger when an explosive atmosphere is built up in air.
Because of their small size, hydrogen molecules can easily diffuse in other materials. In particular, when hydrogen and ferritic steels are in contact this poses a hazard. Hydrogen will diffuse in the ferritic steel and cause so-called hydrogen embrittlement. The different behavior of brittle and ductile materials in a stress–strain diagram can be seen in Figure 3. In engineering, a ductile behavior is usually preferred, because the component will suffer a plastic deformation before failing, which might be visible or cause vibration. This offers the chance to take measures to prevent a complete failure from occurring. A brittle component, on the other hand, will suddenly fail, without the chance to prevent a failure. Austenitic steels, copper and aluminum do not suffer from hydrogen embrittlement [30]. Interactions of the storage method with materials, such as embrittlement due to cryogenic temperatures, are discussed in the corresponding chapter.
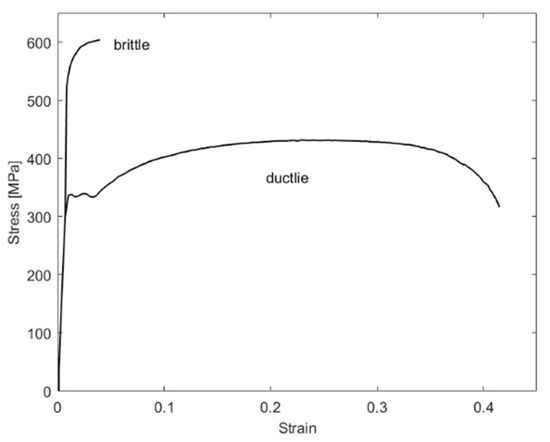
Figure 3.
Brittle vs. ductile behavior in materials.
Hydrogen is 14 times lighter than air and will thus rise quickly in air [34]. If a leak occurs in an open-space environment, this buoyancy is an important safety feature. The hydrogen will quickly rise and disperse below the LFL. In confined spaces, the buoyancy has to be considered during the design of the safety system. Sensors should be arranged above possible leaks or the highest point in a confined space. The roof has to be designed in a way to prevent hydrogen from accumulating. Ventilation outlets should be the highest point of the room. Natural gas is 8 times heavier than hydrogen, but still 2 times lighter than air [34], so it will also rise and disperse but it will take longer than hydrogen to fall below LEL.
Hydrogen is not toxic. The only danger that hydrogen poses directly to humans is a suffocating effect, by displacing oxygen from the environment (asphyxia). When the concentration of oxygen in the air falls below 16%, the first effects, such as an increased breathing rate, are noticeable. Oxygen concentrations below 7% lead to death. Even though hydrogen is an odorless, colorless, and tasteless gas, high concentrations can be detected by the voice becoming high-pitched and squeaky, similar to inhaling helium from balloons. Additionally, no carcinogenic effects from hydrogen are known [35].
There are various different sensor types available for hydrogen. A study found that most commercially available sensors are an electrochemical type [36]. The right sensor type has to be chosen for a specific application with regard to the sensitivity, ambient conditions, aging, drift, response time and reliability. Disadvantages from one sensor type might be compensated for by combining different types [37]. Power consumption, size and cost should also be considered. The cross sensitivity of certain sensor types should also be considered. Electrochemical hydrogen sensors usually have a cross sensitivity to carbon monoxide [38]. Huebert et al. gives an extensive overview of different hydrogen-sensing principles [37].
To mitigate the risks evolving from the gas and make it easily detectible by human senses, natural gas is mixed with odorants [34]. Humans can smell the gas before a hazardous atmosphere occurs and start countermeasures, such as shutting down the gas supply, venting and evacuating the affected area. For hydrogen this will not be suitable. Fuel cells, especially proton exchange membrane (PEM) fuel cells, need very pure hydrogen and the odorant would poison the fuel cell [34]. The molecule’s size is another problem with this solution. The normally used odorant molecules are significantly bigger than the hydrogen molecule. In some cases, small leaks, that are sufficient for hydrogen but too small for the odorant, might be possible [34]. Hence, a hazardous hydrogen atmosphere could form, without being detected by people, causing a false sense of safety.
3.2.2. Pressurized Hydrogen Storage in Vessels
Gaseous hydrogen is usually stored in gas cylinders at a pressure between 350 and 700 bar. Often, 350 bar systems are used for heavy duty, land-based applications, such as busses or trucks [39]. On the other hand, 700 bar systems are more common where higher energy densities are needed, for example in cars [39]. The Elektra, a pusher boat powered by hydrogen, uses a 500 bar system [40].
Gas cylinders are categorized into four types. Type I cylinders are of an all-metal design. They have the highest weight but lowest costs. Still mostly made of metals are Type II cylinders; only a composite overwrap in the hoop direction is added, to improve mechanical strength. They are still cost-competitive, but the weight is reduced only slightly. If the main material is composite and only a metal liner is used, the cylinder is a Type III. Their cost is higher and their weight lower, both significantly. In order to reduce the weight even further, the metal liner can be replaced with a high-density polymer liner. These are the Type IV cylinders, and they also reduce the costs as compared with Type III [29].
A hazard of pressurized storage is the rupture of the vessel, resulting in an explosive release of hydrogen. Said rupture is usually caused by a fire in the vessel’s surroundings. The fire drastically increases the temperature and causes the pressure inside the vessel to increase until the vessel’s material is not able to withstand the load anymore and ruptures [41]. The initial fire can cause the hydrogen to explode. To prevent this from happening, gas cylinders can be equipped with a pressure relief device (PRD). In the case of increased temperatures, pressures, or both it will release some or all of the entire hydrogen in the gas cylinder. A thermally activated pressure relief device (TPRD) will rapidly release all of the hydrogen in the cylinder before a rupture can occur due to weakened walls. TPRDs will not reseal to prevent a re-pressurization [42].
For Type III and IV tanks another failure mechanism can occur. If exposed to fire the fiber-reinforced plastic will deteriorate and the matrix polymer can burn. If the TPRD is non-functional, either because of a failure or because the TPRD’s temperature does not reach the set limit, the tank will fail catastrophically, with an explosion of the tank. In experiments a 14 kg fragment of a Type IV cylinder was thrown 82 m from its original position. Only a marginal increase in the cylinder’s internal temperature (27 to 39 °C) and pressure (345 to 357 bar) was observed during these experiments [43]. A regular PRD would not have actuated. The PRD cannot prevent the rupture in this failure mechanism.
If hydrogen is suddenly released under high pressure into the air, a shockwave will mix hydrogen with air [44], compress the air and heat up to a point where the hydrogen self-ignites [45]. Although self-ignition has been observed at pressures around 40 bar, below a pressure of 200 bar it was difficult to trigger a self-ignition in experiments [45]. Methane can also suffer from self-ignition, but it is even harder to produce [44]. Such high pressures usually occur only in storage systems. The fuel cells and other sources of consumption use much lower pressures. The high-pressure system has to be designed in a way to minimize the risk of a high-pressure leak that might self-ignite.
Such a high-pressure leak occurred in 2019 in a hydrogen refueling station for cars in Norway, which exploded and caught fire. The pressure vessels did not explode and no injuries due to the explosion were reported. The explosion, however, triggered the airbags of three cars, causing injuries to two people, who were then hospitalized. The hydrogen leak that led to the explosion was caused by bolts that were not adequately torqued in the plug of a pressure vessel. The manufacturer has since established measures to prevent this human error from reoccurring [46].
Klebanoff et al. calculated the permeation of hydrogen through a 316 (1.4401) stainless steel wall with a thickness of 1/16″ (~1.6 mm). It is assumed that 1200 kg of hydrogen is trapped in a spherical container with a radius of 7 m and at a pressure of 150 psi (~10 bar). At room temperature it would take 60 years to reach hydrogen’s LFL of 4% in a telephone booth, if the telephone booth is hermetically sealed and only hydrogen can enter. Although the pressure in this example is well below the storage pressure in gas cylinders, the risk of the concentration reaching the flammability limit can be easily mitigated by a minimal method of ventilation, e.g., by opening the door to enter the room [10].
3.2.3. Storage as Liquefied Hydrogen at Cryogenic Temperature
The highest volumetric energy density can be achieved by liquifying a gas. The principle is already being used for LNG, which liquifies at 111 K [26]. Hydrogen will liquify at a temperature of 20 K. In addition to the general hazards of hydrogen, the cryogenic temperature of liquid hydrogen poses its own hazards, most of which are also applicable to LNG.
Besides hydrogen embrittlement, as described above, embrittlement can also be caused at cryogenic temperatures. The energy absorbed during the Charpy impact test, which is, for example, specified in ISO 148, is a dimension of a material’s ductility [30]. Figure 4 shows the Charpy impact strength for different materials. It can be seen that the Charpy impact strength, and thus the ductility, of different steels decreases drastically and suddenly at lower temperatures. From the materials shown in Figure 4, only 304 stainless steel and aluminum show a relatively constant behavior and can thus be used for liquid hydrogen applications [30]. In general, austenitic stainless steels, copper and aluminum are suitable for use at cryogenic temperatures [47].
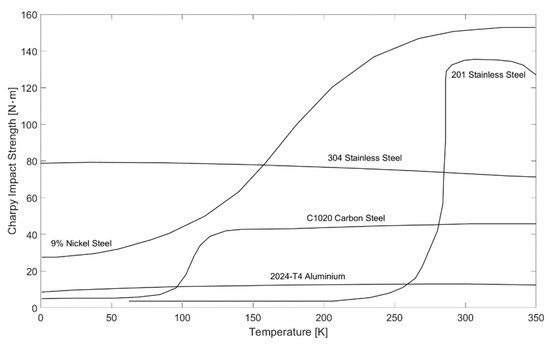
Figure 4.
Charpy impact strength as a function of temperature for various materials based on [30].
Usually, materials will contract when they are cooled down, and the contraction coefficient is different for different materials [22]. The system has to be designed to compensate for these different contractions [30]. Otherwise high stresses can occur [22] and may even cause a structural failure, which can then lead to leakage [30].
Direct contact of liquid hydrogen or cryogenic vapor to the skin can cause severe frostbite [35]. A liquid hydrogen spill can result in an asphyxiating atmosphere, as discussed above.
If the heat transfer into the liquid hydrogen increases significantly, a boiling liquid expanding vapor explosion (BLEVE) might occur. This can be caused by a fire outside of the vessel or damaged insulation due to an accident [48]. Since liquid hydrogen contains no impurities, no nucleation sites are present and it will not start boiling, but the liquid will be superheated until it cannot be superheated any further. At that point microscopic vapor bubbles will be formed, causing a shock wave [30]. The vessel will not be able to withstand the increased pressure and will be ruptured. A pressure wave will follow that further destroys the vessel and expels fragments of it. Both the pressure wave and the fragments can hurt or kill humans or damage structures [48]. However, the shock wave is expected to be drastically smaller than for other liquids [30]. The hydrogen vapor and liquid can ignite and form a fireball, if a sufficient ignition source is present [48].
Hydrogen’s boiling point of 20 K is below every other gas’s boiling and freezing point, except for helium. If a gas, usually air, makes contact with liquid hydrogen, it will condense and solidify. Oxygen has a higher boiling and freezing point than nitrogen and will thus condense earlier. This might lead to a high concentration of up to 50% oxygen in the solidified material [49]. A mixture of solidified oxygen and liquid hydrogen can be detonated by impact and releases more energy than an equal amount of trinitrotoluene (TNT) [47]. If the system is heated up, for example, for maintenance, an oxygen-rich, explosive atmosphere can form inside the system [50]. Solidified air can also form in pipes or valves and plug them or keep valves from closing [49]. Precautions can be taken to prevent solidified air from accumulating. The system should be kept under an overpressure, so if a leak occurs, hydrogen will reach the surrounding area but no air will reach the system. Before the system is filled it should be purged with nitrogen, followed by gaseous hydrogen at ambient temperature [49]. Since LNG’s storage temperature of 111 K is above oxygen’s melting point of 90 K [51], this effect is of no concern for LNG.
The same effect might occur when a large amount of liquid hydrogen is spilled. Oxygen is solidified and an explosive mixture is formed in the pool. When hydrogen and oxygen vaporize a flammable, oxygen-rich atmosphere might be present [49]. The vaporization consumes heat, for which conduction from ground is the dominant source. The other heat sources (atmospheric convection, radiation from flame and radiation from ambient) account for only 10–20% of transferred heat [22]. Compared with LNG, liquid hydrogen takes 3 times less heat to evaporate the same amount of stored energy, resulting in significantly warmer contact surfaces [10].
If liquid hydrogen is spilled, it only needs to be heated up by 2 K to become more buoyant than air, whereas methane becomes more buoyant than air at 164.5 K, a temperature rise of 53.3 K from its boiling point [10]. Because hydrogen’s enthalpy of vaporization of 0.92 kJ/mol is 9.2 times smaller than methane’s, it will not cool down contact areas in the case of a spill as much as LNG will, although it is significantly colder [10]. If liquid hydrogen or LNG is spilled on a ship’s deck, the deck might embrittle and will contract. This contraction might cause the rupture of the deck, which is more likely for brittle materials.
3.2.4. Cryo-Compressed Storage
As described above, cryo-compressed storage uses a vacuum-insulated pressure vessel designed for up to 350 bar. Again, for this storage method the general hazards of hydrogen apply. However, not all hazards from pressurized vessels and cryogenic storage can be combined. Although both vessels have the same internal pressure, it can be shown that the energy released from a sudden hydrogen expansion from a cryo-compressed vessel is about 100 times lower than for a comparable pressure vessel at ambient temperature, without the outer vessel [52]. A potential loss of vacuum in the insulation is also not as hazardous as it is for a cryogenic vessel designed for ambient pressure. Simulations show that the boil-off from a tank vessel, with a compromised vacuum, intended for automotive applications, is limited to the amount of hydrogen the fuel cell can process [52]. This boil-off hydrogen can then be fed to the fuel cell and converted into water and electrical energy. The electricity can then automatically be consumed by internal systems or by a designated resistance. Thus, ventilation to the atmosphere is not necessary, minimizing risks in enclosed spaces.
3.2.5. Chemical Hydrogen Storage as a Metal Hydride
Storage systems based on metal hydrides use a metal alloy to absorb gaseous hydrogen by chemisorption. This effect allows large quantities of hydrogen to be stored reversibly at relatively low pressures (e.g., 30 bar) in moldable pressure bodies. The amount of stored hydrogen corresponds to that of a 700 bar tank. The special feature of chemisorption is that heat is released during hydrogen absorption and must be supplied accordingly during withdrawal [53]. This creates a degree of freedom as an additional control variable for withdrawal. Metal hydrides have a particular advantage from a safety point of view because the storage units require a heat supply to ensure withdrawal. The material compositions of the metal hydrides can be designed and adapted accordingly [29]. As an example, only a certain amount of the flammable gas is released when the valve is ruptured because the spontaneous relief leads to a freezing of the tank, which means that only small amounts or no amount of hydrogen is released. Due to the low pressures in the metal hydride storage tank, it is also possible to design the pressure body differently from typical cylindrical shapes and to better adapt to the local conditions for integration [54]. However, this poses a challenge in that heat integration for the storage tank is necessary and a fault in the heating can also lead to a pressure increase in the storage tank. For this reason, a metal hydride storage still needs a safety device for the pressure.
Metal hydride storage systems are already being used in manned maritime transport [55], such as hydrogen storage systems in submarines, and are bringing the above-mentioned advantages. In this case, the increased weight does not play a role, since the storage tanks are located outside the pressure hull and also support the ship with a certain amount of downforce.
3.2.6. Chemical Hydrogen Storage as a Liquid Organic Hydrogen Carrier
Liquid organic hydrogen carriers (LOHCs) are oil-based liquids that can store hydrogen via a chemical reaction with the oil. They can be handled like regular diesel oil and the existing infrastructure can be used [56]. Since hydrogen is chemically bound to the carrier liquid, most of its hazards are mitigated and only apply in the relatively short timeframe between dehydrogenation and usage, when pure hydrogen is present. One promising substance to be used as an LOHC is dibenzyltoluene (DBT), which can store up to 6.2 wt% hydrogen [57]. This paper will use dibenzyltoluene as an example for all LOHCs.
DBT is only poorly flammable, and has a high flash point of 485 K and an auto-ignition temperature of 773 K [58], whereas diesel fuel has a minimum flash point of 329 K and a minimum ignition temperature of 498 K [59]. As most other oils, DBT is hazardous to the aquatic environment [58]. It is also a health hazard if aspirated [58].
3.3. Rules and Regulations
To mitigate risk and set a minimum safety standard, rules and regulations can be adopted. In shipping, the International Maritime Organization (IMO) is responsible for such rules and regulations. The IMO is a specialized agency of the United Nations for the safety, security and environmental protection of international shipping [60]. For this purpose, it issues regulations that are ratified by the 175 member states and are subsequently entered into force. These regulations contain, among other things, safety requirements for ship design and equipment, but also operational requirements, such as the necessary training of crews [61]. Compliance with these regulations is monitored by the flag states [62].
The flag states in turn can have the monitoring carried out by their own authorities or delegate it to classification societies, who are private companies. The authorities or classification societies check all the necessary plans and drawings before construction begins and supervise the construction of the ship at the shipyard. After proving that the ship meets all requirements, the flag state issues all necessary ship certificates and the ship may be operated [63].
If a seagoing vessel is used as a cargo or passenger ship, it always requires classification by the classification societies in addition to the acceptances by the flag states. For this purpose, the classification societies define their own sets of rules, and monitor and certify compliance with them on behalf of the shipowner. In this classification, the seaworthiness of the ship is assessed for certain sailing areas. Classification is usually necessary in order to procure insurance [63].
The rules and regulations adopted by IMO with regard to hydrogen are described below.
Rules and Regulations by the International Maritime Organization
IMO has adopted approximately 50 conventions and more than 1000 codes and recommendations [61]. The first convention was the “International Convention of Safety of Life at Sea” (SOLAS), that was adopted in 1914 after the Titanic disaster [64]. The 1974 version is still in force, but has been updated by amendments on several occasions, last during the 104th Session of the Maritime Safety Committee (MSC104) in October 2021 [65].
SOLAS sets minimum standards for the safety of a ship’s construction, equipment and operation [64]. It consists of 14 chapters and is applicable to all ships engaged in international voyages. Only a few exceptions, such as warships or cargo ships of less than 500 gross tonnage, are possible. Most relevant for hydrogen applications are Chapters II and VII, although hydrogen applications are not yet directly addressed. Standards for the construction of a ship are set in Chapter II, where it refers to the IGF Code for details concerning ships using low-flashpoint fuels. Chapter VII deals with the carriage of dangerous goods and also refers to different codes, containing requirements for different types of dangerous goods. For hydrogen applications, the IGC Code is relevant. Both codes will be discussed in more detail.
In Chapter II-1/55 of SOLAS the approval process for alternative designs and arrangements is described. If a design deviates from the requirements given in SOLAS or other IMO instruments, an engineering analysis has to be performed. If said analysis concludes that the design provides an equivalent level of safety and meets the intent of the regulation that the design deviates from, then the flag state administration can still approve the ship’s design. More details about this process are given in MSC Circulars, viz MSC.1/Circ.1212/Rev.1 [6] for deviation from SOLAS and MSC.1/Circ.1455 [5] for deviation from other IMO instruments.
The “International Code for Safety for Ships Using Gases or Other Low-Flashpoint Fuels” (IGF-Code) [66] was adopted by resolution MSC.391(95) in June 2015 and came into force in 2017. It follows a goal-based approach, according to MSC.1/Circ.1394. The code and most of its chapters begin with the goal, which the regulations in the code or chapter are intended to fulfill. One goal is to provide a safe design of ships, covered by the code. These then lead to functional requirements, for example, to ensure that a single failure does not lead to an unsafe situation. These functional requirements are then followed by detailed regulations that have to be fulfilled. If a loss of power is considered an unsafe situation, one regulation might be to have a redundant fuel supply system, so that a single failure in one can be compensated for by the second system.
The IGF Code is applicable to all ships that have been built after 2017, except for gas carriers, using their cargo or other low-flashpoint gaseous fuels and complying with the IGC Code. Additionally, ships owned or operated by a government and operating in non-commercial service only do not have to comply to the IGF Code. The IGF Code covers both permanently installed and interchangeable storage systems. For now, the IGF Code only covers natural gas as a fuel, but the goal and functional requirements can be used for the alternative design approach according to SOLAS Chapter II-1/55.
Ships carrying liquefied gases as cargo, such as LNG, are covered by the “International Code for the Construction and Equipment of Ships Carrying Liquefied Gases in Bulk” (IGC-Code) [67]. This applies to all ships carrying the products listed in Chapter 19, including those having a gross tonnage of less than 500, but not to Floating Production, Storage and Offloading (FPSO) units. Hydrogen is not covered by the code.
The code categorizes ships by the hazards of the products it is intended to carry into four categories. Type “1G” ships carry the most hazardous products and have the strictest requirements. Ships carrying less hazardous products that only need moderate measures to prevent their escape are classified as “3G” type ships. Type “2G” is an intermediate classification with hazards and preventive measures between “1G” and “3G”. Type “2PG” ships carry products classified for “2G” ships, but have a length of less than 150 m and use type C tanks with a gauge pressure above 7 bar and temperatures above −55 °C.
Similar to the ship types, tanks are also categorized into five different types. If a classical ship-structural analysis based on recognized standards is used during the independent tank’s design process, it will be categorized as Type “A”. If refined analytical tools and analysis methods are used and model tests are performed, the tank will be categorized as a Type “B” independent tank. Type “C” independent tanks, on the other hand, are designed using modified pressure vessel criteria. The code also covers membrane tanks and tanks forming a structural part of the hull, so-called integral tanks.
The use of cargo as a fuel is also regulated by the IGC Code in Chapter 16. The IGF Code allows only methane (LNG) to be used as a fuel. Other fuels, except for toxic products, might be permitted, if an equivalent level of safety can be ensured.
The IMO published the “Interim Recommendations for Carriage of Liquified Hydrogen in Bulk” [68] because they were needed for a pilot project. The experience from this pilot project will be used to include hydrogen in the IGC Code. The Interim Recommendations classify hydrogen carriers as 2G-type ships and specify 29 additional requirements that hydrogen carriers will have to fulfill. One requirement is, for example, that helium or a mixture of hydrogen and nitrogen should be used for the tightness tests. These requirements are then explained in detail and linked to the related hazard.
3.4. Ongoing Hydrogen Projects in Shipping
There are currently various ongoing projects worldwide on the subject of safety on hydrogen-related maritime platforms. A distinction can be made between ships already in function that have already gone into initial operation and development for demonstration purposes. Selected examples are explained below and an overview can be found in Table 2.

Table 2.
Overview of different hydrogen projects.
3.4.1. Suiso Frontier
The tanker “Suiso Frontier” (IMO 9860154) [69] is the result of the “Hydrogen Energy Supply Chain Technology Research Association” (HySTRA) project initiated in 2016 in Japan by different Japanese companies [70]. It will be used to transport liquid hydrogen (LH2) over a distance of 9000 km from Australia to Japan. This vessel, the first of its kind, was delivered by Kawasaki Heavy Industries (KHI) and has been operating successfully since the end of 2021. On board, it holds a vacuum-insulated double-shell-structure stainless-steel LH2 cargo tank, which has a size of 1250 m3. The ship has a diesel–electric propulsion system. The safety requirements have been registered by the Classification Society ClassNK [71]. These inspections are based on the IGC Code (Part N of Rules for the Survey and Construction of Steel Ships), which contains the interim recommendations of the IMO (Interim Recommendations for Carriage of Liquefied Hydrogen in Bulk, as described above). Furthermore, the required risk assessment was based on the Hazard Identification Study (HAZID), with four methods [72,73]. During in-service surveys this project should return experiences to keep the guidelines up to date and to set international standards.
3.4.2. Elektra
The push boat “Elektra” (MMSI 211828680) was also built in 2021 based on a German research project [74,75]. Led by the project consortium ELEKTRA (e.g., HGK Shipping, TU Berlin, and BEHALA), the 20 m-long inland vessel will travel with the loaded heavy-lift carrier “Ursus” from the Berlin area towards the Rhine/Ruhr, Hamburg and Szczecin. On board, three fuel cells will supply the electrical propulsion system (2 × 200 kW). The fuel cells operate with mobile hydrogen tanks (500 bar) containing 750 kg hydrogen. The German “Binnen-Schiffsuntersuchungsordnung” (BinSchUO) has been selected for the orientation, but it currently has no valid regulation for the energy system. For the ship certificate, an approval via the “European Committee for Drawing up Standards in the Field of Inland Navigation” (CESNI) was issued [76].
3.4.3. Hydra
NORLEDs ship of the year 2021 [77], “Hydra” (IMO 9887530), is based in Stavanger, Norway. The ferry has connected the three ports of Hjelmeland, Nesvik and Skipavik since 2021. It is also the world’s first operating hydrogen ferry. The 82-metre-long vessel can carry 80 cars and 290 passengers and is classified by the Norwegian Maritime Directorate, DSB and DNV Ship Register [78,79]. The used liquid hydrogen fuel is imported by the company Linde from Leuna (Germany) [80].
3.4.4. Topeka
“Topeka” is the result of a EU-funded project called HyShip, which pursues the target of a zero-emission prototype vessel. Led by Norwegian company Wilh. Wilhelmsen ASA, a RoRo freighter is scheduled to be launched in 2024. The ship, which will operate on the Norwegian west coast, is planned with a 3 MW PEM fuel cell. The needed liquid (green) hydrogen will be obtained through the national grid. Combined with the 1 MWh battery pack the 125 m-long ship will replace up to 25,000 trucks per year from the roads. It should help to close the gap in experience with liquid hydrogen in a decentralized energy system, as well as the hurdles of the required infrastructure [81,82].
3.4.5. LH2 FGSS (MAN Cryo)
MAN announced in 2020 that they are currently working in Sweden on liquid-hydrogen fuel-gas-supply systems (LH2 FGSSs). This system has been designed to be compliant with the IGF Code and has, for example, a holding time for the contained liquified hydrogen of 15 days. It has a scalable design, which allows it to be used in different ship types [83]. In 2021 MAN delivered the 175 m3 FGSS, including a tank connection space (TCS), bunker station and regasification. According to the IGF code, it works on a 9 bar-design which allows a usable filling level volume of 69%. In future this filling level should be improved. Regarding the safety measures, to be approved by different class societies, a double-hull tank, double-walled piping, H2 detection and automatic controls are installed [84].
3.5. Proposal of a Method for the Alternative Design Approach
As described in Section 3.3, there are currently no rules for hydrogen in shipping available, but the alternative design approach allows the approval of hydrogen systems, if an equivalent level of safety is demonstrated. However, many criteria, such as the detectability of a gas or flame, cannot be described properly by definite numbers. Other criteria, that can be described by definite numbers, such as the storage temperature of the liquified gas, may show a misleading ratio to each other. Hydrogen is stored at 20 K, whereas LNG is stored at 111 K, which is 5.55 times warmer. However, LNG’s temperature is not automatically 5.55 times more safe. For example, most materials embrittle at both temperatures. So, both temperatures could be considered more similar than their ratio expresses. Additionally, not all criteria are equally relevant to the safety of a system. The flammability of a gas is usually more relevant than its enthalpy of vaporization, for example. Considering this, this chapter will propose a method, based on fuzzy set theory, that can compensate for uncertainties from verbal formulations such as “significantly safer” and translate them into a value to make two systems better comparable.
The method should be executed by experts from all relevant fields working on the project. The more experts participate, the more accurate the results become.
As a first step, all criteria of the investigated system have to be identified for a relevant comparison. For example, the flammability or the temperature of the liquid gas are possible criteria. These criteria will then be weighted for their relevance to safety by using a pairwise comparison. This method uses a simplified pairwise comparison, as proposed in [12]. Table 3 shows an exemplary pairwise comparison for four criteria. All identified criteria are filled in the rows and columns of the table. Then, every criterion written in a row is compared to the criterion in the column. If the criterion in the row is more relevant than the one in the column the corresponding field is filled with a “2”; if they are equally relevant the field will be filled with a “1”; and if the criterion in the column is more important the field will be filled with a “0”. The diagonal, marked in grey in Table 3, always has to be filled with a “1”, because a criterion is compared with itself. The blue marked area above the diagonal has to be filled as well. The green marked area under the diagonal can be automatically filled using Equation (1) and the yellow marked area can be automatically calculated using the equations shown in Table 3.

Table 3.
Pairwise comparison for four aspects.
When the table is filled completely, the values in every row are summed up. If the sums for each row are again summed up, the result has to be n2. This value can be used as a control mechanism. The sums for each row are then divided by n2, the result of which is the weighting of every aspect. The sum of all weightings has to be 1.
In the next step, the newly designed system is compared with a system designed following existing rules. In this case, a hydrogen system would be compared with an LNG system. In order to compare these two systems another table is needed. In this table the experts can rate, for each criterion, if the new or the existing system is “significantly safer”, “a little safer” or if both systems are equally safe. If the novel system is examined, the terms “a little safer” and “significantly safer” appointed to the existing system need to be translated into “a little unsafer” and “significantly unsafer” and vice versa. Table 4 is an example of such a table.

Table 4.
Comparison table.
In order to translate verbal terms such as “significantly safer” into comparable information, fuzzy sets will be used. Zadeh introduced his fuzzy set theory in 1965 to describe imprecise or incomplete information, such as “beautiful people” [13]. Instead of appointing a single, sharp number to a linguistic term, a fuzzy number, described by a membership function , is used to translate the unsharpness of linguistic terms. The value of at describes the grade of membership for each point in the fuzzy set on the real unit interval [0;1]. Different shapes for the membership function can be used. If each term is described by a single number, the membership function has the value 1 for this number and 0 everywhere else. In more detail, fuzzy sets have been described by Zadeh [13,14,15].
Different shapes for a membership function are possible, but in engineering mostly triangular and trapezoidal are used [16,85]. The proposed method will also use triangular fuzzy numbers, which are shown in Figure 5 and can described by the membership function given in Equation (2):
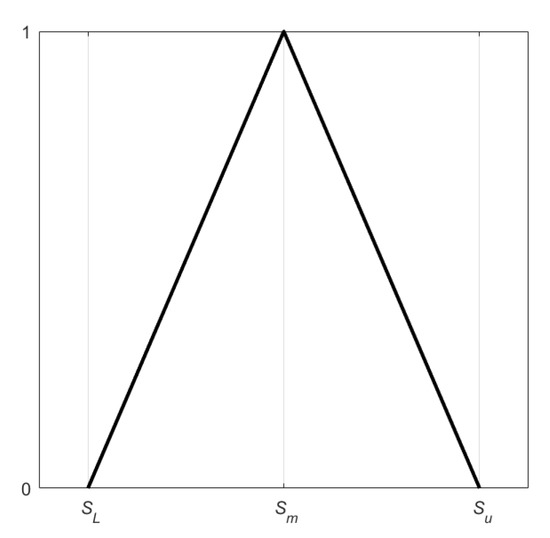
Figure 5.
Triangular fuzzy number.
In Equation (2), represents the lower boundary, the most likely value and the upper boundary. Now, the terms used in Table 4 are translated into the fuzzy number with the values given in Table 5. The result is shown in Figure 6. The comparison factors in five steps in Table 4 and the corresponding values in Table 5 are a suggestion and can be adapted if necessary for the specific project.

Table 5.
Linguistic statements translated into fuzzy numbers.
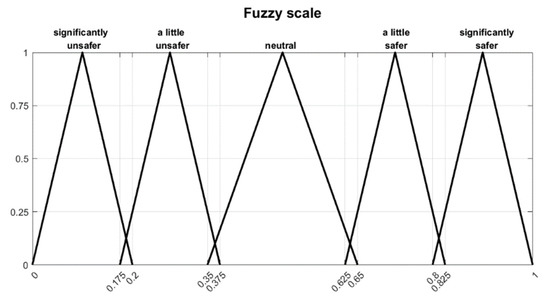
Figure 6.
Fuzzy scale.
The fuzzy numbers obtained for every expert separately can then be merged using Equation (3) for each criterion separately, or using Equation (4) for the whole system.
In Equations (3) and (4) m is the number of experts participating in the process and n is the number of criterions.
After Equation (4) has been used to assess both systems, they can be easily compared by visualizing both fuzzy numbers in one graph, similar to that presented in Figure 6.
4. Discussion
Many properties and hazards from hydrogen cannot directly be compared to those of LNG. The material compatibility, for example, can be described in linguistic terms, but a numerical value cannot be assigned, which makes it harder to compare. Other values, such as the minimum ignition energy, have fixed numerical values, but a comparison of these properties by the relationship of their values may still not represent the real effect on safety. The minimum ignition energy of hydrogen is 0.02 mJ and methane’s is 0.29 mJ, so the ratio between both is 14.5. If we consider that the discharge from a human body contains around 10 mJ energy, it can be argued that methane (LNG) is not 14.5 times safer than hydrogen, but that they are on a more similar level.
Now, these hard-to-compare hazards should be used to demonstrate an equivalent level of safety to IMO instruments, such as the IGF Code. In order to make these hazards better comparable, a new method is proposed.
This method assigns a weighting to the different criteria using a pairwise comparison. Different experts from all relevant fields then compare the fuels for all criteria using simple linguistic terms such as “a little safer”. These linguistic terms are then translated into fuzzy numbers, which are then, together with the previously obtained weighting, brought together to produce a total score for each fuel or system, which can be directly compared.
5. Conclusions
This article introduced a novel method to compare the safety of fuel alternatives. A possible application was shown for a comparison of hydrogen and LNG systems. The method suggests a straightforward comparability and simplifies IMO’s alternative design approach for all stakeholders, such as authorities and ship designers. The proposed method can help to enable a wider, but still safe, use of hydrogen in shipping.
To allow for the easier application of the proposed method, all relevant properties of hydrogen and LNG were introduced and compared where applicable. It was shown that a simple comparison of different values is often not possible, which explains the necessity of the proposed method.
Author Contributions
Conceptualization, J.D. and A.D.; investigation, J.D., A.D. and L.R.; writing—original draft preparation, J.D., A.D. and L.R.; writing—review and editing, J.D.; supervision, S.E. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
Special thanks to all our colleagues at the Institute for Maritime Energy Systems for productive discussion on the topic and their willingness to proofread this paper.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| BinSchUO | Binnen-Schiffsuntersuchungsordnung |
| BLEVE | boiling liquid expanding vapor explosion |
| CESNI | European Committee for Drawing up Standards in the Field of Inland Navigation |
| CO | carbon monoxide |
| CO2 | carbon dioxide |
| DBT | dibenzyltoluene |
| DSB | Direktoratet for samfunnssikkerhet og beredskap (Norwegian Maritime Directorate) |
| EU | European Union |
| FPSO | Floating Production, Storage and Offloading |
| GHG | greenhouse gas |
| H2 | hydrogen |
| HAZID | Hazard Identification Study |
| HySTRA | Hydrogen Energy Supply Chain Technology Research Association |
| IEC | International Electrotechnical Commission |
| IGC Code | International Code for the Construction and Equipment of Ships Carrying Liquefied Gases in Bulk |
| IGF Code | International Code for Safety for Ships Using Gases or Other Low-Flashpoint Fuels |
| IMO | International Maritime Organization |
| IR | infrared |
| ISO | International Organization for Standardization |
| KHI | Kawasaki Heavy Industries |
| LEL | lower explosive limit |
| LFL | lower flammability limit |
| LH2 | liquified hydrogen |
| LH2 FGSS | liquid-hydrogen fuel-gas-supply systems |
| LNG | liquified natural gas |
| LOHC | liquid organic hydrogen carrier |
| MIR | multi-spectrum-infrared |
| MSC | Maritime Safety Committee |
| MSC104 | 104th Session of the Maritime Safety Committee |
| NOX | nitrogen oxide |
| PEM | proton exchange membrane |
| PRD | pressure relief device |
| RoRo | Roll on Roll off |
| SOLAS | International Convention of Safety of Life at Sea |
| TNT | trinitrotoluene |
| TPRD | thermally activated pressure relief device |
| UEL | upper explosive limit |
| UFL | upper flammability limit |
| UV | ultraviolet |
References
- Faber, J.; Hanayama, S.; Zhang, S.; Pereda, P.; Comer, B.; Hauerhof, E.; van der Loeff, W.S.; Smith, T.; Zhang, Y.; Kosaka, H.; et al. Fourth IMO GHG Study 2020: Full Report, London. 2021. Available online: https://wwwcdn.imo.org/localresources/en/OurWork/Environment/Documents/Fourth%20IMO%20GHG%20Study%202020%20-%20Full%20report%20and%20annexes.pdf (accessed on 7 December 2021).
- UNCTAD United Nations Conference on Trade and Development. Review of Maritime Transport 2019, New York, United States of America. 2019. Available online: https://unctad.org/system/files/official-document/rmt2019_en.pdf (accessed on 7 December 2021).
- International Maritime Organization (IMO) Resolution MEPC.304(72) Initial IMO Strategy on Reduction of GHG Emissions from Ships: MEPC.304(72), 2018. Available online: https://wwwcdn.imo.org/localresources/en/KnowledgeCentre/IndexofIMOResolutions/MEPCDocuments/MEPC.304(72).pdf (accessed on 10 March 2022).
- Wallner, T.; Lohse-Busch, H.; Shidore, N. Operating strategy for a hydrogen engine for improved drive-cycle efficiency and emissions behavior. Int. J. Hydrogen Energy 2009, 34, 4617–4625. [Google Scholar] [CrossRef]
- International Maritime Organization (IMO) Guidelines for the Approval of Alternatives and Equivalents as Provided for in Various IMO Instruments: MSC.1/Circ.1455, 2013. Available online: https://puc.overheid.nl/nsi/doc/PUC_2017_14/1/ (accessed on 10 March 2022).
- International Maritime Organization (IMO) Revised Guidelines on Alternative Design and Arrangements for SOLAS Chapters II-1 and III: MSC.1/Circ.1212/Rev.1, 2019. Available online: https://puc.overheid.nl/nsi/doc/PUC_1648_14/1/ (accessed on 10 March 2022).
- McGuinness, E.; Utne, I.B. A systems engineering approach to implementation of safety management systems in the Norwegian fishing fleet. Reliab. Eng. Syst. Saf. 2014, 121, 221–239. [Google Scholar] [CrossRef]
- ISO International Organization for Standardization. Safety Aspects—Guidelines for Their Inclusion in Standards, 2014 (ISO/IEC Guide 51:2014). Available online: https://www.iso.org/obp/ui/#iso:std:iso-iec:guide:51:ed-3:v1:en (accessed on 15 March 2022).
- Engebø, A.; Nilsen, S.; Kirchsteiger, C.; Perrette, L.; Merad, M.; Andersen, H.B. Biennal Report On Hydrogen Safety: Chapter IV: Risk Assessment, 2007. Available online: http://www.hysafe.org/download/1199/BRHS_Chap4_V1p2.pdf (accessed on 15 March 2022).
- Klebanoff, L.E.; Pratt, J.W.; LaFleur, C.B. Comparison of the safety-related physical and combustion properties of liquid hy-drogen and liquid natural gas in the context of the SF-BREEZE high-speed fuel-cell ferry. Int. J. Hydrogen Energy 2017, 42, 757–774. [Google Scholar] [CrossRef] [Green Version]
- Saaty, T.L. Decision making with the analytic hierarchy process. Int. J. Serv. Sci. 2008, 1, 83–98. [Google Scholar] [CrossRef] [Green Version]
- Feldhusen, J.; Grote, K.-H. (Eds.) Konstruktionslehre: Methoden und Anwendung Erfolgreicher Produktentwicklung, 8. Vollständig überarbeitete Auflage; Springer Vieweg: Berlin/Heidelberg, Germany, 2013; ISBN 978-3-642-29569-0. [Google Scholar]
- Zadeh, L.A. Fuzzy sets. Inf. Control 1965, 1965, 338–353. [Google Scholar] [CrossRef] [Green Version]
- Zadeh, L.A. Quantitative fuzzy semantics. Inf. Sci. 1971, 3, 159–176. [Google Scholar] [CrossRef]
- Zadeh, L.A. The concept of a linguistic variable and its application to approximate reasoning—I. Inf. Sci. 1975, 8, 199–249. [Google Scholar] [CrossRef]
- Ehlers, U.C.; Ryeng, E.O.; McCormack, E.; Khan, F.; Ehlers, S. Assessing the safety effects of cooperative intelligent transport systems: A bowtie analysis approach. Accid. Anal. Prev. 2017, 99, 125–141. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S.; Khan, F.; Shaikh, A.; Ahmed, S.; Imtiaz, S. Development of risk model for marine logistics support to offshore oil and gas operations in remote and harsh environments. Ocean Eng. 2019, 174, 125–134. [Google Scholar] [CrossRef]
- van Hoecke, L.; Laffineur, L.; Campe, R.; Perreault, P.; Verbruggen, S.W.; Lenaerts, S. Challenges in the use of hydrogen for maritime applications. Energy Environ. Sci. 2021, 14, 815–843. [Google Scholar] [CrossRef]
- EIGA European Industrial Gases Association AISBL. Gaseous Hydrogen Installations: EIGA Doc 15/21 15/21, Brussels, 2021. Available online: https://www.eiga.eu/uploads/documents/DOC015.pdf (accessed on 23 November 2021).
- EIGA European Industrial Gases Association AISBL. Safety in Storage, Handling and Distribution of Liquid Hydrogen: Doc 06/19 06/19, Brussels, 2019. Available online: https://www.eiga.eu/uploads/documents/DOC006.pdf (accessed on 23 November 2021).
- Verfondern, K. Biennal Report on Hydrogen Safety: Chapter I: Hydrogen Fundamentals, 2007. Available online: http://www.hysafe.org/download/1196/BRHS_Chap1_V1p2.pdf (accessed on 7 December 2021).
- Verfondern, K. Safety Considerations on Liquid Hydrogen; Forschungszentrum Zentralbibliothek: Jülich, Germany, 2008; ISBN 978-3-89336-530-2. [Google Scholar]
- Raucci, C.; Calleya, J.; Suárez de la Fuente, S.; Pawling, R. Hydrogen on board ship: A first analysis of key parameters and implications. In Proceedings of the International Conference on Shipping in Changing Climates 2015, Glasgow, UK, 24–26 November 2015. [Google Scholar]
- Aceves, S.M.; Petitpas, G.; Espinosa-Loza, F.; Matthews, M.J.; Ledesma-Orozco, E. Safe, long range, inexpensive and rapidly refuelable hydrogen vehicles with cryogenic pressure vessels. Int. J. Hydrogen Energy 2013, 38, 2480–2489. [Google Scholar] [CrossRef]
- Engineering ToolBox. Fuels-Higher and Lower Calorific Values: Higher and Lower Calorific Values (Heating Values) for Fuels Like Coke, Oil, Wood, Hydrogen and Others. Available online: https://www.engineeringtoolbox.com/fuels-higher-calorific-values-d_169.html (accessed on 16 December 2021).
- Balcombe, P.; Brierley, J.; Lewis, C.; Skatvedt, L.; Speirs, J.; Hawkes, A.; Staffell, I. How to decarbonise international shipping: Options for fuels, technologies and policies. Energy Convers. Manag. 2019, 182, 72–88. [Google Scholar] [CrossRef]
- Cheliotis, M.; Boulougouris, E.; Trivyza, N.L.; Theotokatos, G.; Livanos, G.; Mantalos, G.; Stubos, A.; Stamatakis, E.; Ve-netsanos, A. Review on the safe use of ammonia fuel cells in the maritime industry. Energies 2021, 14, 3023. [Google Scholar] [CrossRef]
- Müller, K.; Stark, K.; Emel’yanenko, V.N.; Varfolomeev, M.A.; Zaitsau, D.H.; Shoifet, E.; Schick, C.; Verevkin, S.P.; Arlt, W. Liquid organic hydrogen carriers: Thermophysical and thermochemical studies of benzyl- and dibenzyl-toluene derivatives. Ind. Eng. Chem. Res. 2015, 54, 7967–7976. [Google Scholar] [CrossRef]
- Rivard, E.; Trudeau, M.; Zaghib, K. Hydrogen storage for mobility: A review. Materials 2019, 12, 1973. [Google Scholar] [CrossRef] [Green Version]
- Rigas, F. Hydrogen Safety, Online-Ausg; CRC Press/Taylor & Francis Group: Boca Raton, FL, USA, 2012; ISBN 978-1-4398-6232-2. [Google Scholar]
- SEA-LNG Ltd. LNG-Fuelled Vessels Approaching 30% of Orders. Available online: https://sea-lng.org/2021/09/lng-fuelled-vessels-approaching-30-of-orders/ (accessed on 30 November 2021).
- IFA Institute for Occupational Safety and Health of the German Social Accident Insurance. Methane: GESTIS-Substance Database. Available online: https://gestis.dguv.de/data?name=010000&lang=en (accessed on 23 December 2021).
- Drägerwerk AG & Co. KGaA. Hydrogen: How to Meet the Safety Challenges, 2021. Available online: https://www.draeger.com/Library/Content/hydrogen-safety-challenges-ebk-11064-en-master.pdf (accessed on 23 December 2021).
- HyResponder. Properties of Hydrogen Relevant to Safety: Lecture 2. Level IV Specialist Officer, 2021. Available online: https://hyresponder.eu/e-platform/training-materials/educational-training/lecture-2-properties-of-hydrogen-relevant-to-safety/ (accessed on 9 December 2021).
- IFA Institute for Occupational Safety and Health of the German Social Accident Insurance. Hydrogen: GESTIS-Substance Database. Available online: https://gestis.dguv.de/data?name=007010&lang=en (accessed on 3 December 2021).
- Boon-Brett, L.; Bousek, J.; Black, G.; Moretto, P.; Castello, P.; Hübert, T.; Banach, U. Identifying performance gaps in hydrogen safety sensor technology for automotive and stationary applications. Int. J. Hydrogen Energy 2010, 35, 373–384. [Google Scholar] [CrossRef]
- Hübert, T.; Boon-Brett, L.; Black, G.; Banach, U. Hydrogen sensors—A review. Sens. Actuators B Chem. 2011, 157, 329–352. [Google Scholar] [CrossRef]
- Buttner, W.J.; Post, M.B.; Burgess, R.; Rivkin, C. An overview of hydrogen safety sensors and requirements. Int. J. Hydrogen Energy 2011, 36, 2462–2470. [Google Scholar] [CrossRef]
- H2 MOBILITY Deutschland GmbH & Co. KG. FAQs. Available online: https://h2-mobility.de/en/faq/ (accessed on 27 December 2021).
- BEHALA Berliner Hafen- und Lagerhausgesellschaft mbH. Versorgung der “ELEKTRA” mit grünem Wasserstoff bis Ende 2024 vertraglich gesichert, 2021. Available online: https://www.behala.de/wp-content/uploads/2021/07/PM_ELEKTRA_Wasserstoffversorgung_14_07_2021.pdf (accessed on 12 January 2022).
- Weyandt, N.; Janssens, M.L. Analysis of Induced Catastrophic Failure of a 5000 PSIG Type IV Hydrogen Cylinder: Final Report, San Antonio, Texas, USA, 2005. Available online: https://www.mvfri.org/contracts/Final%20Reports/SwRI%20Final%20Type%203%20H2%20Tank%20Test.pdf (accessed on 13 December 2021).
- HyResponder. Hydrogen Storage: Lecture 3. Level IV Specialist Officer, 2021. Available online: https://hyresponder.eu/e-platform/training-materials/educational-training/lecture-3-hydrogen-storage/ (accessed on 14 December 2021).
- Zalosh, R. Blast Waves and Fireballs Generated by Hydrogen Fuel Tank Rupture During Fire Exposure, Wellesley, MA, USA, 2007. Available online: https://www.mvfri.org/contracts/Final%20Reports/Zalosh-HydrogenTankFirePaper.pdf (accessed on 14 December 2021).
- Dryer, F.L.; Chaos, M.; Zhao, Z.; Stein, J.N.; Alpert, J.Y.; Homer, C.J. Spontaneous ignition of pressur-ized releases of hydrogen and natural gas into air. Combust. Sci. Technol. 2007, 179, 663–694. [Google Scholar] [CrossRef]
- Mogi, T.; Wada, Y.; Ogata, Y.; Koichi Hayashi, A. Self-ignition and flame propagation of high-pressure hydrogen jet during sudden discharge from a pipe. Int. J. Hydrogen Energy 2009, 34, 5810–5816. [Google Scholar] [CrossRef]
- Pagliaro, M.; Iulianelli, A. Hydrogen Refueling Stations: Safety and Sustainability. Gen. Chem. 2019, 6, 190029. [Google Scholar] [CrossRef]
- Venetsanos, A.; Benard, P.; Papanikolaou, E.; Verfondern, K.; Gallego, E.; Martin-Valdepeñas, J.; Jimenez, M. Biennal Report on Hydrogen Safety: Chapter III: Accidental Phenomena and Consequences, 2007. Available online: http://www.hysafe.org/download/1198/BRHS_Chap3_V1p2.pdf (accessed on 16 December 2021).
- Ustolin, F.; Paltrinieri, N.; Landucci, G. An innovative and comprehensive approach for the consequence analysis of liquid hydrogen vessel explosions. J. Loss Prev. Process Ind. 2020, 68, 104323. [Google Scholar] [CrossRef]
- Prichard, D.K.; Rattigan, W.M. Hazards of Liquid Hydrogen: Position Paper, 2010. Available online: https://www.hse.gov.uk/research/rrpdf/rr769.pdf (accessed on 22 December 2021).
- Aziz, M. Liquid hydrogen: A review on liquefaction, storage, transportation, and safety. Energies 2021, 14, 5917. [Google Scholar] [CrossRef]
- IFA Institute for Occupational Safety and Health of the German Social Accident Insurance. Oxygen: GESTIS-Substance Da-tabase. Available online: https://gestis.dguv.de/data?name=007080&lang=en (accessed on 12 January 2022).
- Petitpas, G.; Aceves, S.M. Modeling of sudden hydrogen expansion from cryogenic pressure vessel failure. Int. J. Hydrogen Energy 2013, 38, 8190–8198. [Google Scholar] [CrossRef] [Green Version]
- Modi, P.; Aguey-Zinsou, K.-F. Room temperature metal hydrides for stationary and heat storage applications: A review. Front. Energy Res. 2021, 9, 616115. [Google Scholar] [CrossRef]
- Bellosta von Colbe, J.; Ares, J.-R.; Barale, J.; Baricco, M.; Buckley, C.; Capurso, G.; Gallandat, N.; Grant, D.M.; Guzik, M.N.; Jacob, I.; et al. Application of hydrides in hydrogen storage and compression: Achievements, outlook and perspectives. Int. J. Hydrogen Energy 2019, 44, 7780–7808. [Google Scholar] [CrossRef]
- Krummrich, S. Fuel cell methanol reformer system for submarines. In Proceedings of the 18th World Hydrogen Energy Conference 2010—WHEC 2010, Essen, Germany, 16–21 May 2010; Forschungszentrum IEF-3: Jülich, Germany, 2010. ISBN 978-3-89336-653-8. [Google Scholar]
- Teichmann, D.; Arlt, W.; Wasserscheid, P.; Freymann, R. A future energy supply based on liquid organic hydrogen carriers (LOHC). Energy Environ. Sci. 2011, 4, 2767. [Google Scholar] [CrossRef]
- Brückner, N.; Obesser, K.; Bösmann, A.; Teichmann, D.; Arlt, W.; Dungs, J.; Wasserscheid, P. Evaluation of industrially applied heat-transfer fluids as liquid organic hydrogen carrier systems. ChemSusChem 2014, 7, 229–235. [Google Scholar] [CrossRef]
- IFA Institute for Occupational Safety and Health of the German Social Accident Insurance. Dibenzyltoluene, Isomers: GES-TIS-Substance Database. Available online: https://gestis.dguv.de/data?name=492182&lang=en (accessed on 12 January 2022).
- IFA Institute for Occupational Safety and Health of the German Social Accident Insurance. Diesel Fuel: GESTIS-Substance Database. Available online: https://gestis.dguv.de/data?name=536303&lang=en (accessed on 12 January 2022).
- IMO International Maritime Organization. Introduction to IMO. Available online: https://www.imo.org/en/About/Pages/Default.aspx (accessed on 15 December 2021).
- IMO International Maritime Organization. IMO What It Is, London, UK, 2013. Available online: https://wwwcdn.imo.org/localresources/en/About/Documents/What%20it%20is%20Oct%202013_Web.pdf (accessed on 15 December 2021).
- Deutsche Flagge. BG Verkehr. Available online: https://www.deutsche-flagge.de/en/german-flag/flag-state/bg-verkehr/bg-verkehr?set_language=en (accessed on 23 December 2021).
- Deutsche Flagge. Classification Societies. Available online: https://www.deutsche-flagge.de/en/german-flag/flag-state/classification-societies-2/classification-societies?set_language=en (accessed on 23 December 2021).
- IMO International Maritime Organization. International Convention for the Safety of Life at Sea (SOLAS), 1974. Available online: https://www.imo.org/en/About/Conventions/Pages/International-Convention-for-the-Safety-of-Life-at-Sea-(SOLAS),-1974.aspx (accessed on 27 December 2021).
- IMO International Maritime Organization. Maritime Safety Committee (MSC), 104th Session, 4–8 October 2021. Available online: https://www.imo.org/en/MediaCentre/MeetingSummaries/Pages/MSC-104th-session.aspx (accessed on 27 December 2021).
- International Maritime Organization (IMO) International Code of Safety for Ships Using Gases or Other Low-Flashpoint Fuels: IGF-Code, 2015. Available online: https://www.imo.org/en/OurWork/Safety/Pages/IGF-Code.aspx (accessed on 10 March 2022).
- International Maritime Organization (IMO) International Code for the Construction and Equipment of Ships Carrying Liquefied Gases in Bulk: IGC-Code, 2014. Available online: https://www.imo.org/en/OurWork/Environment/Pages/IGCCode.aspx (accessed on 10 March 2022).
- International Maritime Organization (IMO) Resolution MSC.420(97) Interim Recommendations for Carriage of Liquefied Hydrogen in Bulk: MSC.420(97), 2016. Available online: https://wwwcdn.imo.org/localresources/en/KnowledgeCentre/IndexofIMOResolutions/MSCResolutions/MSC.420(97).pdf (accessed on 10 March 2022).
- FleetMon. Suiso Frontier. Available online: https://www.fleetmon.com/vessels/suiso-frontier_9860154_2165466/ (accessed on 24 January 2022).
- Kudou, J.; Ota, S.; Senga, Y. Safety requirements for liquedied hydrogen tankers. In Proceedings of the International Conference on Hydrogen Safety, Yokohama, Japan, 19–21 October 2015. [Google Scholar]
- ClassNK. ClassNK Adds Liquefied Hydrogen Carrier to Its Register: World’s First Liquefied Hydrogen Carrier Built by Kawasaki Heavy Industries; ClassNK: Tokyo, Japan, 2021; Available online: https://www.classnk.or.jp/hp/en/hp_news.aspx?id=6982&type=press_release (accessed on 25 January 2022).
- Kawasaki Heavy Industries, Ltd. Kawasaki Develops Cargo Containment System for Large Liquefied Hydrogen Carrier with World’s Highest Carrying Capacity—AiP Obtained from ClassNK; Kawasaki Heavy Industries, Ltd.: Tokyo, Japan, 2021; Available online: https://global.kawasaki.com/en/corp/newsroom/news/detail/?f=20210506_9983 (accessed on 24 January 2022).
- ShipInsight. ClassNK Gives AiP for Liquid Hydrogen Containment System. ShipInsight [Online], 10 May 2021. Available online: https://shipinsight.com/articles/classnk-gives-aip-for-liquid-hydrogen-cargo-containment-system (accessed on 24 January 2022).
- BEHALA Berliner Hafen- und Lagerhausgesellschaft mbH. The Push Boat with a Whole New Energy System: Lighthouse Project: ELEKTRA. Available online: https://www.behala.de/en/the-push-boat-with-a-whole-new-energy-system/ (accessed on 24 January 2022).
- Lichtfuß, K.-G. Erfolgreicher Stapellauf der “ELEKTRA” in Derben; BEHALA—Berliner Hafen- und Lagerhausgesellschaft mbH: Berlin, Germany, 2021; Available online: https://www.behala.de/wp-content/uploads/2021/05/BEHALA_PM_ELEKTRA_Stapellauf_27052021.pdf (accessed on 26 April 2022).
- Kräft, M.; Andreas, J.; Segieth, P. Vorzeige-Projekt für Wasserstoff-Technologie. Binnenschifffahrt [Online], 1 September 2020. Available online: https://binnenschifffahrt-online.de/2020/09/schiffstechnik/16744/ (accessed on 24 January 2022).
- Larsen, S.; Markussen, H.M. MF ‘Hydra’ Named Ship of The Year: Minister of Transport and Communications Knut Arild Hareide Hands Out This Year’s Ship of the Year on Board MF ‘Hydra’. Skipsrevyen [Online], 9 September 2021. Available online: https://www.skipsrevyen.no/article/mf-hydra-named-ship-of-the-year/ (accessed on 25 January 2022).
- Norway: MF “Hydra”, the World’s First Hydrogen Operated Ferry Wins Ship of the Year 2021. FuelCellWorks [Online], 28 De-cember 2021. Available online: https://fuelcellsworks.com/news/norway-mf-hydra-the-worlds-first-hydrogen-operated-ferry-wins-ship-of-the-year-2021/ (accessed on 25 January 2022).
- LMG Marin AS. Hydra. Available online: https://www.lmgmarin.no/references/485/hydra (accessed on 25 January 2022).
- Radowitz, B. World’s First Hydrogen-Powered Ferry in Norway to Run on Green Gas from Germany: Linde to Provide H2 from an Electrolyser at Facility in Leuna Chemicals Complex Despite Cheaper Power Prices in Norway. Recharge [Online], 9 March 2021. Available online: https://www.rechargenews.com/technology/worlds-first-hydrogen-powered-ferry-in-norway-to-run-on-green-gas-from-germany/2-1-976939 (accessed on 25 January 2022).
- Turner, J. HySHIP: Inside Europe’s Flagship Hydrogen Ship Demonstrator Project. Ship Technology [Online], 22 December 2020. Available online: https://www.ship-technology.com/features/hydrogen-vessel/ (accessed on 25 January 2022).
- Wilh. Wilhelmsen Holding ASA. Topeka’s Hydrogen Vessels One Step Closer to Reality, 2021. Available online: https://www.wilhelmsen.com/media-news-and-events/press-releases/2021/topekas-hydrogen-vessels-one-step-closer-to-reality/ (accessed on 25 January 2022).
- Søholt, N. MAN Cryo Announces Series of Hydrogen Projects: Company Takes Lead in Fuel-Gas-Supply Systems for Alternative Fuel with Great Potential; MAN Energy Solutions: Augsburg, Germany, 2020. [Google Scholar]
- MAN Energy Solutions. Hydrogen in Shipping; MAN Energy Solutions: Augsburg, Germany, 2021. [Google Scholar]
- Ferdous, R.; Khan, F.; Sadiq, R.; Amyotte, P.; Veitch, B. Handling and updating uncertain information in bow-tie analysis. J. Loss Prev. Process Ind. 2012, 25, 8–19. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).