Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare
Abstract
1. Introduction
2. Implantable and Wearable Medical Devices in Precision Healthcare
3. Energy Harvesting Techniques
3.1. Human-Centric Energy Harvesting
3.1.1. Biochemical Energy Harvesting
Biofuel Cells
Hydroelectric Generators
3.1.2. Biomechanical Energy Harvesting
Piezoelectric Energy Generators
Electrostatic Transduction
Electromagnetic Induction
Thermoelectric Methods
Triboelectric Energy Harvesting
3.2. Environment-Centric Energy Harvesting
3.2.1. Infrared Radiation
3.2.2. Radio Frequency Signal
3.2.3. Solar Energy Harvesting
3.2.4. Wireless Power Transfer
Ultrasonic Techniques
Capacitive Coupling
Inductive Coupling
3.3. Hybrid Energy Harvesting
4. Extraction of Maximum Power from Energy Harvesters
5. Machine Learning for Emerging Energy Harvesters
6. Use-Cases of Energy Harvesters
6.1. Energy Harvesters in Implantable Medical Devices
6.2. Energy Harvesters in Wearable Medical Devices
7. Interaction of Energy Harvesters with the Human Body
8. Challenges and Future Research Directions
8.1. Efficient Power Management
8.2. Optimizing Power Consumption
8.3. Storage of Harvested Energy
8.4. Energy-Efficient Communication Schemes
8.5. Energy Need for Next-Generation IWM Devices
9. Discussion
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| IWM | Implantable and wearable medical | TENG | Triboelectric nanogenerator |
| PPG | Photoplethysmography | PCB | Printed circuit board |
| WMD | Wearable medical device | MPPT | Maximum power point tracking |
| ECG | Electrocardiogram | PCE | Power conversion efficiency |
| IMD | Implantable medical device | P&O | Perturb and observe |
| RF | Radio frequency | FOCV | Fractional open-circuit voltage |
| PMU | Power management unit | VOC | Open-circuit voltage |
| TEG | Thermoelectric energy generators | SSHI | Synchronized switch harvesting on inductor |
| GBF | Glucose biofuel | SECE | Synchronous electric charge extraction |
| PZT | Polycrystalline lead zirconate titanate | BLE | Bluetooth low energy |
| TKR | Total knee replacement | BFC | Biofuel cells |
| PMN-PT | Lead magnesium niobate–lead titanate | GOx | Glucose oxidase |
| PZN-PT | Lead–zinc niobate–lead titanate | NIR | Near-infrared region |
| ZnO | Zinc oxide | WPT | Wireless power transfer |
| PVDF | Polyvinylidene difluoride | PUEH | Piezoelectric ultrasonic energy harvester |
| MEMS | Microelectromechanical systems | EMG | Electromyography |
| EP | Endocochlear potential | HEG | Hydroelectric generator |
| ECSA | Electrochemically active surface area | rGO | Reduced graphene oxide |
| IME | Implantable electronics | SWG | Single-wire generator |
| SEH | Standard energy harvesting | SCE | Synchronized charge extraction |
| EM | Electromagnetic | EMI | Electromagnetic interference |
| SWCNT | Single wall carbon nanotubes | DWNT | Double-wall nanotube |
| DETA | Diethylenetriamine | TTT | Tetrathiotetracene |
| FOM | Figure of merit | PENG | Piezoelectric nanogenerators |
| ML | Machine learning | ANN | Artificial neural network |
| EMF | Electromagnetic field | DEGS | Dielectric elastomer generator system |
References
- Jeong, I.C.; Bychkov, D.; Searson, P.C. Wearable Devices for Precision Medicine and Health State Monitoring. IEEE Trans. Biomed. Eng. 2019, 66, 1242–1258. [Google Scholar] [CrossRef]
- Aileni, R.M.; Valderrama, A.C.; Strungaru, R. Wearable Electronics for Elderly Health Monitoring and Active Living. In Ambient Assisted Living and Enhanced Living Environments; Elsevier: Amsterdam, The Netherlands, 2017; pp. 247–269. [Google Scholar] [CrossRef]
- Ghamari, M.; Janko, B.; Sherratt, R.; Harwin, W.; Piechockic, R.; Soltanpur, C. A Survey on Wireless Body Area Networks for eHealthcare Systems in Residential Environments. Sensors 2016, 16, 831. [Google Scholar] [CrossRef] [PubMed]
- Shuvo, M.M.H.; Ahmed, N.; Nouduri, K.; Palaniappan, K. A Hybrid Approach for Human Activity Recognition with Support Vector Machine and 1D Convolutional Neural Network. In Proceedings of the 2020 IEEE Applied Imagery Pattern Recognition Workshop (AIPR), Washington, DC, USA, 13–15 October 2020; pp. 1–5. [Google Scholar] [CrossRef]
- Hoyt, R.W.; Reifman, J.; Coster, T.S.; Buller, M.J. Combat medical informatics: Present and future. In Proceedings of the AMIA Symposium, San Antonio, TX, USA, 9–13 November 2002; pp. 335–339. [Google Scholar]
- Friedl, K.E. Military applications of soldier physiological monitoring. J. Sci. Med. Sport 2018, 21, 1147–1153. [Google Scholar] [CrossRef]
- Poh, M.-Z.; Kim, K.; Goessling, A.; Swenson, N.; Picard, R. Cardiovascular Monitoring Using Earphones and a Mobile Device. IEEE Pervasive Comput. 2012, 11, 18–26. [Google Scholar] [CrossRef]
- Teichmann, D.; Kuhn, A.; Leonhardt, S.; Walter, M. The MAIN Shirt: A Textile-Integrated Magnetic Induction Sensor Array. Sensors 2014, 14, 1039–1056. [Google Scholar] [CrossRef]
- di Rienzo, M.; Meriggi, P.; Rizzo, F.; Castiglioni, P.; Lombardi, C.; Ferratini, M.; Parati, G. Textile Technology for the Vital Signs Monitoring in Telemedicine and Extreme Environments. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 711–717. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, F.; Shakhsheer, Y.; Silver, J.D.; Klinefelter, A.; Nagaraju, M.; Boley, J.; Pandey, J.; Shrivastava, A.; Carlson, E.J.; et al. A Batteryless 19 μW MICS/ISM-Band Energy Harvesting Body Sensor Node SoC for ExG Applications. IEEE J. -Solid-State Circ. 2013, 48, 199–213. [Google Scholar] [CrossRef]
- Hossain, Z.; Shuvo, M.M.H.; Sarker, P. Hardware and software implementation of real time electrooculogram (EOG) acquisition system to control computer cursor with eyeball movement. In Proceedings of the 2017 4th International Conference on Advances in Electrical Engineering (ICAEE), Dhaka, Bangladesh, 28–30 September 2017; pp. 132–137. [Google Scholar] [CrossRef]
- Macdonald, A.; Hawkes, L.A.; Corrigan, D.K. Recent advances in biomedical, biosensor and clinical measurement devices for use in humans and the potential application of these technologies for the study of physiology and disease in wild animals. Philos. Trans. R. Soc. B Biol. Sci. 2021, 376, 20200228. [Google Scholar] [CrossRef]
- Yi, N.; Cui, H.; Zhang, L.G.; Cheng, H. Integration of biological systems with electronic-mechanical assemblies. Acta Biomater. 2019, 95, 91–111. [Google Scholar] [CrossRef]
- Shaikh, F.K.; Zeadally, S. Energy harvesting in wireless sensor networks: A comprehensive review. Renew. Sustain. Energy Rev. 2016, 55, 1041–1054. [Google Scholar] [CrossRef]
- Dionisi, A.; Marioli, D.; Sardini, E.; Serpelloni, M. Autonomous Wearable System for Vital Signs Measurement With Energy-Harvesting Module. IEEE Trans. Instrum. Meas. 2016, 65, 1423–1434. [Google Scholar] [CrossRef]
- Paradiso, J.A.; Starner, T. Energy Scavenging for Mobile and Wireless Electronics. IEEE Pervasive Comput. 2005, 4, 18–27. [Google Scholar] [CrossRef]
- Khan, A.S.; Khan, F.U. A survey of wearable energy harvesting systems. Int. J. Energy Res. 2022, 46, 2277–2329. [Google Scholar] [CrossRef]
- Khaligh, A.; Zeng, P.; Zheng, C. Kinetic Energy Harvesting Using Piezoelectric and Electromagnetic Technologies–State of the Art. IEEE Trans. Ind. Electron. 2010, 57, 850–860. [Google Scholar] [CrossRef]
- Beeby, S.P.; Tudor, M.J.; White, N.M. Energy harvesting vibration sources for microsystems applications. Meas. Sci. Technol. 2006, 17, R175–R195. [Google Scholar] [CrossRef]
- Wise, K.D.; Anderson, D.J.; Hetke, J.F.; Kipke, D.R.; Najafi, K. Wireless Implantable Microsystems: High-Density Electronic Interfaces to the Nervous System. Proc. IEEE 2004, 92, 76–97. [Google Scholar] [CrossRef]
- Haerinia, M.; Shadid, R. Wireless Power Transfer Approaches for Medical Implants: A Review. Signals 2020, 1, 12. [Google Scholar] [CrossRef]
- Liu, Y.; Li, B.; Huang, M.; Chen, Z.; Zhang, X. An Overview of Regulation Topologies in Resonant Wireless Power Transfer Systems for Consumer Electronics or Bio-Implants. Energies 2018, 11, 1737. [Google Scholar] [CrossRef]
- Schmidt, C.L.; Scott, E.R. Energy harvesting and implantable medical devices—first order selection criteria. In Proceedings of the 2011 International Electron Devices Meeting, Washington, DC, USA, 5–7 December 2011; pp. 10.5.1–10.5.4. [Google Scholar] [CrossRef]
- Torres, E.O.; Rincon-Mora, G.A. A 0.7-μm BiCMOS Electrostatic Energy-Harvesting System IC. IEEE J. -Solid-State Circ. 2010, 45, 483–496. [Google Scholar] [CrossRef]
- Zhang, Q.; Liang, Q.; Rogers, J.A. Water-soluble energy harvester as a promising power solution for temporary electronic implants. APL Mater. 2020, 8, 120701. [Google Scholar] [CrossRef]
- Olivo, J.; Carrara, S.; de Micheli, G. Energy Harvesting and Remote Powering for Implantable Biosensors. IEEE Sens. J. 2011, 11, 1573–1586. [Google Scholar] [CrossRef]
- Roy, S.; Azad, A.N.M.W.; Baidya, S.; Alam, M.K.; Khan, F. Powering Solutions for Biomedical Sensors and Implants Inside the Human Body: A Comprehensive Review on Energy Harvesting Units, Energy Storage, and Wireless Power Transfer Techniques. IEEE Trans. Power Electron. 2022, 37, 12237–12263. [Google Scholar] [CrossRef]
- Roy, S.; Azad, A.N.M.W.; Baidya, S.; Khan, F. A Comprehensive Review on Rectifiers, Linear Regulators, and Switched-Mode Power Processing Techniques for Biomedical Sensors and Implants Utilizing in-Body Energy Harvesting and External Power Delivery. IEEE Trans. Power Electron. 2021, 36, 12721–12745. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, L.; Jiang, K.; Wei, Z.; Shen, G. Reviews of wearable healthcare systems: Materials, devices and system integration. Mater. Sci. Eng. R Rep. 2020, 140, 100523. [Google Scholar] [CrossRef]
- Jagadeeswari, V.; Subramaniyaswamy, V.; Logesh, R.; Vijayakumar, V. A study on medical Internet of Things and Big Data in personalized healthcare system. Health Inf. Sci. Syst. 2018, 6, 14. [Google Scholar] [CrossRef]
- Lymberis, A. Smart wearable systems for personalised health management: Current R&D and future challenges. In Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (IEEE Cat. No.03CH37439), Cancun, Mexico, 17–21 September 2003; pp. 3716–3719. [Google Scholar] [CrossRef]
- Shuvo, M.M.H.; Ahmed, N.; Islam, H.; Alaboud, K.; Cheng, J.; Mosa, A.S.M.; Islam, S.K. Machine Learning Embedded Smartphone Application for Early-Stage Diabetes Risk Assessment. In Proceedings of the 2022 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Messina, Italy, 22–24 June 2022; pp. 1–6. [Google Scholar] [CrossRef]
- Gatzoulis, L.; Iakovidis, I. Wearable and Portable eHealth Systems. IEEE Eng. Med. Biol. Mag. 2007, 26, 51–56. [Google Scholar] [CrossRef]
- Bayo-Monton, J.-L.; Martinez-Millana, A.; Han, W.; Fernandez-Llatas, C.; Sun, Y.; Traver, V. Wearable Sensors Integrated with Internet of Things for Advancing eHealth Care. Sensors 2018, 18, 1851. [Google Scholar] [CrossRef] [PubMed]
- Findlow, A.; Goulermas, J.Y.; Nester, C.; Howard, D.; Kenney, L.P.J. Predicting lower limb joint kinematics using wearable motion sensors. Gait Posture 2008, 28, 120–126. [Google Scholar] [CrossRef]
- Hester, T.; Sherrill, D.M.; Hamel, M.; Perreault, K.; Boissy, P.; Bonato, P. Identification of Tasks Performed by Stroke Patients Using a Mobility Assistive Device. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 30 August–3 September 2006; pp. 1501–1504. [Google Scholar] [CrossRef]
- Syed, L.; Jabeen, S.; S, M.; Alsaeedi, A. Smart healthcare framework for ambient assisted living using IoMT and big data analytics techniques. Future Gener. Comput. Syst. 2019, 101, 136–151. [Google Scholar] [CrossRef]
- Shuvo, M.M.H.; Hassan, O.; Parvin, D.; Chen, M.; Islam, S.K. An Optimized Hardware Implementation of Deep Learning Inference for Diabetes Prediction. In Proceedings of the 2021 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), Glasgow, UK, 17–21 May 2021; pp. 1–6. [Google Scholar] [CrossRef]
- Connolly, P.; Cotton, C.; Morin, F. Opportunities at the skin interface for continuous patient monitoring: A reverse iontophoresis model tested on lactate and glucose. IEEE Trans. Nanobiosci. 2002, 1, 37–41. [Google Scholar] [CrossRef]
- Hassan, O.; Paul, T.; Thakker, R.; Parvin, D.; Shuvo, M.M.H.; Mosa, A.S.M.; Islam, S.K. A Multi-Sensor Based Automatic Sleep Apnea Detection System for Adults Using Neural Network Inference on FPGA. In Proceedings of the 2022 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Messina, Italy, 22–24 June 2022; pp. 1–6. [Google Scholar] [CrossRef]
- Lymberis, A.; Olsson, S. Intelligent Biomedical Clothing for Personal Health and Disease Management: State of the Art and Future Vision. Telemed. J. e-Health 2003, 9, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhang, J.; Xie, Y.; Gao, F.; Xu, S.; Wu, X.; Ye, Z. Wearable Health Devices in Health Care: Narrative Systematic Review. JMIR Mhealth Uhealth 2020, 8, e18907. [Google Scholar] [CrossRef] [PubMed]
- Elsheikh, M.H.; Shnawah, D.A.; Sabri, M.F.M.; Said, S.B.M.; Hassan, M.H.; Bashir, M.B.A.; Mohamad, M. A review on thermoelectric renewable energy: Principle parameters that affect their performance. Renew. Sustain. Energy Rev. 2014, 30, 337–355. [Google Scholar] [CrossRef]
- Ghomian, T.; Kizilkaya, O.; Choi, J.-W. Lead sulfide colloidal quantum dot photovoltaic cell for energy harvesting from human body thermal radiation. Appl. Energy 2018, 230, 761–768. [Google Scholar] [CrossRef]
- Khaligh, A.; Zeng, P.; Wu, X.; Xu, Y. A hybrid energy scavenging topology for human-powered mobile electronics. In Proceedings of the 2008 34th Annual Conference of IEEE Industrial Electronics, Orlando, FL, USA, 10–13 November 2008; pp. 448–453. [Google Scholar] [CrossRef]
- Akhtar, F.; Rehmani, M.H. Energy replenishment using renewable and traditional energy resources for sustainable wireless sensor networks: A review. Renew. Sustain. Energy Rev. 2015, 45, 769–784. [Google Scholar] [CrossRef]
- Colomer-Farrarons, J.; Miribel-Catala, P.; Saiz-Vela, A.; Samitier, J. A Multiharvested Self-Powered System in a Low-Voltage Low-Power Technology. IEEE Trans. Ind. Electron. 2011, 58, 4250–4263. [Google Scholar] [CrossRef]
- Merrett, G.V.; Huang, H.; White, N.M. Modeling the Effect of Orientation on Human-Powered Inertial Energy Harvesters. IEEE Sens. J. 2015, 15, 434–441. [Google Scholar] [CrossRef]
- Paulo, J.; Gaspar, P.D. Review and future trend of energy harvesting methods for portable medical devices. In Proceedings of the World Congress on Engineering, London, UK, 30 June–2 July 2010; pp. 168–196. [Google Scholar]
- Jia, W.; Valdés-Ramírez, G.; Bandodkar, A.J.; Windmiller, J.R.; Wang, J. Epidermal Biofuel Cells: Energy Harvesting from Human Perspiration. Angew. Chem. Int. Ed. 2013, 52, 7233–7236. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, F.; Rehmani, M.H. Energy Harvesting for Self-Sustainable Wireless Body Area Networks. IT Prof. 2017, 19, 32–40. [Google Scholar] [CrossRef]
- Dong, K.; Jia, B.; Yu, C.; Dong, W.; Du, F.; Liu, H. Microbial fuel cell as power supply for implantable medical devices: A novel configuration design for simulating colonic environment. Biosens. Bioelectron. 2013, 41, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Amar, A.; Kouki, A.; Cao, H. Power Approaches for Implantable Medical Devices. Sensors 2015, 15, 28889–28914. [Google Scholar] [CrossRef]
- Zebda, A.; Alcaraz, J.P.; Vadgama, P.; Shleev, S.; Minteer, S.D.; Boucher, F.; Cinquin, P.; Martin, D.K. Challenges for successful implantation of biofuel cells. Bioelectrochemistry 2018, 124, 57–72. [Google Scholar] [CrossRef]
- Kerzenmacher, S.; Ducrée, J.; Zengerle, R.; von Stetten, F. Energy harvesting by implantable abiotically catalyzed glucose fuel cells. J. Power Sources 2008, 182, 1–17. [Google Scholar] [CrossRef]
- Rapoport, B.I.; Kedzierski, J.T.; Sarpeshkar, R. A Glucose Fuel Cell for Implantable Brain–Machine Interfaces. PLoS ONE 2012, 7, e38436. [Google Scholar] [CrossRef]
- Kwon, C.H.; Ko, Y.; Shin, D.; Kwon, M.; Park, J.; Bae, W.K.; Lee, S.W.; Cho, J. High-power hybrid biofuel cells using layer-by-layer assembled glucose oxidase-coated metallic cotton fibers. Nat. Commun. 2018, 9, 4479. [Google Scholar] [CrossRef]
- Jeerapan, I.; Sempionatto, J.R.; You, J.M.; Wang, J. Enzymatic glucose/oxygen biofuel cells: Use of oxygen-rich cathodes for operation under severe oxygen-deficit conditions. Biosens. Bioelectron. 2018, 122, 284–289. [Google Scholar] [CrossRef]
- Barton, S.C.; Gallaway, J.; Atanassov, P. Enzymatic biofuel cells for implantable and microscale devices. Chem. Rev. 2004, 104, 4867–4886. [Google Scholar] [CrossRef]
- Heller, A. Miniature biofuel cells. Phys. Chem. Chem. Phys. 2004, 6, 209–216. [Google Scholar] [CrossRef]
- Hansen, B.J.; Liu, Y.; Yang, R.; Wang, Z.L. Hybrid nanogenerator for concurrently harvesting biomechanical and biochemical energy. ACS Nano 2010, 4, 3647–3652. [Google Scholar] [CrossRef] [PubMed]
- Katz, E.; MacVittie, K. Implanted biofuel cells operating in vivo—methods, applications and perspectives – feature article. Energy Environ. Sci. 2013, 6, 2791–2803. [Google Scholar] [CrossRef]
- Cinquin, P.; Gondran, C.; Giroud, F.; Mazabrard, S.; Pellissier, A.; Boucher, F.; Alcaraz, J.P.; Gorgy, K.; Lenouvel, F.; Mathé, S.; et al. A glucose biofuel cell implantedin rats. PLoS ONE 2010, 5, e10476. [Google Scholar] [CrossRef] [PubMed]
- Sales, F.C.P.F.; Iost, R.M.; Martins, M.V.A.; Almeida, M.C.; Crespilho, F.N. An intravenous implantable glucose/dioxygen biofuel cell with modified flexible carbon fiber electrodes. Lab. Chip. 2013, 13, 468–474. [Google Scholar] [CrossRef]
- Park, S.I.; Brenner, D.S.; Shin, G.; Morgan, C.D.; Copits, B.A.; Chung, H.U.; Pullen, M.Y.; Noh, K.N.; Davidson, S.; Oh, S.J.; et al. Soft, stretchable, fully implantable miniaturized optoelectronic systems for wireless optogenetics. Nat. Biotechnol. 2015, 33, 1280–1286. [Google Scholar] [CrossRef]
- Milton, R.D.; Giroud, F.; Thumser, A.E.; Minteer, S.D.; Slade, R.C.T. Hydrogen peroxide produced by glucose oxidase affects the performance of laccase cathodes in glucose/oxygen fuel cells: FAD-dependent glucose dehydrogenase as a replacement. Phys. Chem. Chem. Phys. 2013, 15, 19371–19379. [Google Scholar] [CrossRef]
- He, C.; Arora, A.; Kiziroglou, M.E.; Yates, D.C.; O’Hare, D.; Yeatman, E.M. MEMS Energy Harvesting Powered Wireless Biometric Sensor. In Proceedings of the 2009 Sixth International Workshop on Wearable and Implantable Body Sensor Networks, Berkeley, CA, USA, 3–5 June 2009; pp. 207–212. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; You, J.M.; Kim, N.H.; Gu, Y.; Kumar, R.; Mohan, A.V.; Kurniawan, J.; Imani, S.; Nakagawa, T.; Parish, B.; et al. Soft, stretchable, high power density electronic skin-based biofuel cells for scavenging energy from human sweat. Energy Environ. Sci. 2017, 10, 1581–1589. [Google Scholar] [CrossRef]
- Yin, L.; Moon, J.M.; Sempionatto, J.R.; Lin, M.; Cao, M.; Trifonov, A.; Zhang, F.; Lou, Z.; Jeong, J.M.; Lee, S.J.; et al. A passive perspiration biofuel cell: High energy return on investment. Joule 2021, 5, 1888–1904. [Google Scholar] [CrossRef]
- Seok, S.; Wang, C.; Lefeuvre, E.; Park, J. Autonomous Energy Harvester Based on Textile-Based Enzymatic Biofuel Cell for On-Demand Usage. Sensors 2020, 20, 5009. [Google Scholar] [CrossRef] [PubMed]
- Energy-Harvesting Shirt Generates Electricity from Sweat and Movement. Available online: https://newatlas.com/wearables/wearable-microgrid-energy-harvesting-shirt/ (accessed on 25 September 2022).
- Mercier, P.P.; Lysaght, A.C.; Bandyopadhyay, S.; Chandrakasan, A.P.; Stankovic, K.M. Energy extraction from the biologic battery in the inner ear. Nat. Biotechnol. 2012, 30, 1240–1243. [Google Scholar] [CrossRef]
- Gao, Y.; Cho, J.H.; Ryu, J.; Choi, S. A scalable yarn-based biobattery for biochemical energy harvesting in smart textiles. Nano Energy 2020, 74, 104897. [Google Scholar] [CrossRef]
- Yin, J.; Zhou, J.; Fang, S.; Guo, W. Hydrovoltaic Energy on the Way. Joule 2020, 4, 1852–1855. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Yin, J.; Xu, Y.; Fei, W.; Xue, M.; Wang, Q.; Zhou, J.; Guo, W. Emerging hydrovoltaic technology. Nat. Nanotechnol. 2018, 13, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Gao, S.; Xu, W.; Wang, Z. Nanogenerators with Superwetting Surfaces for Harvesting Water/Liquid Energy. Adv. Funct. Mater. 2020, 30, 1908252. [Google Scholar] [CrossRef]
- Shen, D.; Duley, W.W.; Peng, P.; Xiao, M.; Feng, J.; Liu, L.; Zou, G.; Zhou, Y.N. Moisture-Enabled Electricity Generation: From Physics and Materials to Self-Powered Applications. Adv. Mater. 2020, 32, 2003722. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.; Xu, Y.; Ding, T.; Li, J.; Yin, J.; Fei, W.; Cao, Y.; Yu, J.; Yuan, L.; Gong, L.; et al. Water-evaporation-induced electricity with nanostructured carbon materials. Nat. Nanotechnol. 2017, 12, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Wang, Y.; Sun, X.; Li, Y.; Xu, H.; Tan, Y.; Li, Y.; Song, T.; Sun, B. Constant Electricity Generation in Nanostructured Silicon by Evaporation-Driven Water Flow. Angew. Chem. 2020, 132, 10706–10712. [Google Scholar] [CrossRef]
- Han, Y.; Zhang, Z.; Qu, L. Power generation from graphene-water interactions. FlatChem 2019, 14, 100090. [Google Scholar] [CrossRef]
- Shao, C.; Gao, J.; Xu, T.; Ji, B.; Xiao, Y.; Gao, C.; Zhao, Y.; Qu, L. Wearable fiberform hygroelectric generator. Nano Energy 2018, 53, 698–705. [Google Scholar] [CrossRef]
- Zhang, W.; Guan, H.; Zhong, T.; Zhao, T.; Xing, L.; Xue, X. Wearable Battery-Free Perspiration Analyzing Sites Based on Sweat Flowing on ZnO Nanoarrays. Nanomicro Lett. 2020, 12, 1–11. [Google Scholar] [CrossRef]
- Mandal, S.; Roy, S.; Mandal, A.; Ghoshal, T.; Das, G.; Singh, A.; Goswami, D.K. Protein-Based Flexible Moisture-Induced Energy-Harvesting Devices As Self-Biased Electronic Sensors. ACS Appl. Electron. Mater. 2020, 2, 780–789. [Google Scholar] [CrossRef]
- Shen, D.; Xiao, Y.; Zou, G.; Liu, L.; Wu, A.; Xiao, M.; Feng, J.; Hui, Z.; Duley, W.W.; Zhou, Y.N. Exhaling-Driven Hydroelectric Nanogenerators for Stand-Alone Nonmechanical Breath Analyzing. Adv. Mater. Technol. 2020, 5, 1900819. [Google Scholar] [CrossRef]
- Xie, L.; Cai, M. Human Motion: Sustainable Power for Wearable Electronics. IEEE Pervasive Comput. 2014, 13, 42–49. [Google Scholar] [CrossRef]
- Zurbuchen, A.; Pfenniger, A.; Stahel, A.; Stoeck, C.T.; Vandenberghe, S.; Koch, V.M.; Vogel, R. Energy Harvesting from the Beating Heart by a Mass Imbalance Oscillation Generator. Ann. Biomed. Eng. 2013, 41, 131–141. [Google Scholar] [CrossRef]
- Delnavaz, A.; Voix, J. Electromagnetic micro-power generator for energy harvesting from breathing. In Proceedings of the IECON 2012—38th Annual Conference on IEEE Industrial Electronics Society, Montreal, QC, Canada, 25–28 October 2012; pp. 984–988. [Google Scholar] [CrossRef]
- Romero, E.; Warrington, R.O.; Neuman, M.R. Energy scavenging sources for biomedical sensors. Physiol. Meas. 2009, 30, R35–R62. [Google Scholar] [CrossRef] [PubMed]
- Mitcheson, P.D.; Yeatman, E.M.; Rao, G.K.; Holmes, A.S.; Green, T.C. Energy Harvesting From Human and Machine Motion for Wireless Electronic Devices. Proc. IEEE 2008, 96, 1457–1486. [Google Scholar] [CrossRef]
- Ali, F.; Raza, W.; Li, X.; Gul, H.; Kim, K.-H. Piezoelectric energy harvesters for biomedical applications. Nano Energy 2019, 57, 879–902. [Google Scholar] [CrossRef]
- Kymissis, J.; Kendall, C.; Paradiso, J.; Gershenfeld, N. Parasitic power harvesting in shoes. In Proceedings of the Digest of Papers, Second International Symposium on Wearable Computers (Cat. No.98EX215), Pittsburgh, PA, USA, 19–20 October 1998; pp. 132–139. [Google Scholar] [CrossRef]
- Shenck, N.S.; Paradiso, J.A. Energy scavenging with shoe-mounted piezoelectrics. IEEE Micro 2001, 21, 30–42. [Google Scholar] [CrossRef]
- Li, H.; Tian, C.; Deng, Z.D. Energy harvesting from low frequency applications using piezoelectric materials. Appl. Phys. Rev. 2014, 1, 041301. [Google Scholar] [CrossRef]
- Platt, S.R.; Farritor, S.; Haider, H. On Low-Frequency Electric Power Generation With PZT Ceramics. IEEE/ASME Trans. Mechatronics 2005, 10, 240–252. [Google Scholar] [CrossRef]
- Kim, D.H.; Shin, H.J.; Lee, H.; Jeong, C.K.; Park, H.; Hwang, G.T.; Lee, H.Y.; Joe, D.J.; Han, J.H.; Lee, S.H.; et al. In Vivo Self-Powered Wireless Transmission Using Biocompatible Flexible Energy Harvesters. Adv. Funct. Mater. 2017, 27, 1700341. [Google Scholar] [CrossRef]
- Hwang, G.-T.; Park, H.; Lee, J.H.; Oh, S.; Park, K.I.; Byun, M.; Park, H.; Ahn, G.; Jeong, C.K.; No, K.; et al. Self-Powered Cardiac Pacemaker Enabled by Flexible Single Crystalline PMN-PT Piezoelectric Energy Harvester. Adv. Mater. 2014, 26, 4880–4887. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, G.; Yang, R.; Wang, A.C.; Wang, Z.L. Muscle-Driven In Vivo Nanogenerator. Adv. Mater. 2010, 22, 2534–2537. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Qin, Y.; Xu, C.; Wei, Y.; Yang, R.; Wang, Z.L. Self-powered nanowire devices. Nat. Nanotechnol. 2010, 5, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Jeong, C.K.; Baek, C.; Kingon, A.I.; Park, K.; Kim, S. Lead-Free Perovskite Nanowire-Employed Piezopolymer for Highly Efficient Flexible Nanocomposite Energy Harvester. Small 2018, 14, 1704022. [Google Scholar] [CrossRef]
- Todaro, M.T.; Guido, F.; Algieri, L.; Mastronardi, V.M.; Desmaële, D.; Epifani, G.; De Vittorio, M. Biocompatible, Flexible, and Compliant Energy Harvesters Based on Piezoelectric Thin Films. IEEE Trans. Nanotechnol. 2018, 17, 220–230. [Google Scholar] [CrossRef]
- Ramsay, M.J.; Clark, W.W. Piezoelectric energy harvesting for bio-MEMS applications. In Proceedings of the Smart Structures and Materials 2001: Industrial and Commercial Applications of Smart Structures Technologies, Newport Beach, CA, USA, 14 June 2001; pp. 429–438. [Google Scholar] [CrossRef]
- Sohn, J.W.; Choi, S.B.; Lee, D.Y. An investigation on piezoelectric energy harvesting for MEMS power sources. Proc. Inst. Mech. Eng. Part C: J. Mech. Eng. Sci. 2005, 219, 429–436. [Google Scholar] [CrossRef]
- Platt, S.R.; Farritor, S.; Garvin, K.; Haider, H. The Use of Piezoelectric Ceramics for Electric Power Generation Within Orthopedic Implants. IEEE/ASME Trans. Mechatronics 2005, 10, 455–461. [Google Scholar] [CrossRef]
- Chen, H.; Jia, C.; Hao, W.; Zhang, C.; Wang, Z.; Liu, C. Power harvesting with PZT ceramics and circuits design. Analog. Integr. Circ. Signal Process. 2010, 62, 263–268. [Google Scholar] [CrossRef][Green Version]
- Almouahed, S.; Gouriou, M.; Hamitouche, C.; Stindel, E.; Roux, C. Self-powered instrumented knee implant for early detection of postoperative complications. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 5121–5124. [Google Scholar] [CrossRef]
- Renaud, M.; Karakaya, K.; Sterken, T.; Fiorini, P.; van Hoof, C.; Puers, R. Fabrication, modelling and characterization of MEMS piezoelectric vibration harvesters. Sens. Actuators A Phys. 2008, 145–146, 380–386. [Google Scholar] [CrossRef]
- Elfrink, R.; Kamel, T.M.; Goedbloed, M.; Matova, S.; Hohlfeld, D.; Van Andel, Y.; Van Schaijk, R. Vibration energy harvesting with aluminum nitride-based piezoelectric devices. J. Micromech. Microeng. 2009, 19, 094005. [Google Scholar] [CrossRef]
- Gao, S.; Gain, A.K.; Zhang, L. A metamaterial for wearable piezoelectric energy harvester. Smart Mater. Struct. 2020, 30, 015026. [Google Scholar] [CrossRef]
- Beyaz, M.I.; Ahmed, N. A belt-integrated piezoelectric energy harvester for wearable electronic devices. Ferroelectrics 2021, 585, 187–197. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, S.; Jin, Y.M.; Ouyang, H.; Zou, Y.; Wang, X.X.; Xie, L.X.; Li, Z.J.S.S. Flexible piezoelectric nanogenerator in wearable self-powered active sensor for respiration and healthcare monitoring. Semicond. Sci. Technol. 2017, 32, 064004. [Google Scholar] [CrossRef]
- Zhang, Z.; Xiang, H.; Tang, L. Modeling, analysis and comparison of four charging interface circuits for piezoelectric energy harvesting. Mech. Syst. Signal Process. 2021, 152, 107476. [Google Scholar] [CrossRef]
- Long, Z.; Wang, X.; Li, P.; Wang, B.; Zhang, X.; Chung, H.S.H.; Yang, Z. Self-Powered SSDCI Array Interface for Multiple Piezoelectric Energy Harvesters. IEEE Trans. Power Electron. 2021, 36, 9093–9104. [Google Scholar] [CrossRef]
- Chamanian, S.; Muhtaroglu, A.; Kulah, H. A self-adapting synchronized-switch interface circuit for piezoelectric energy harvesters. IEEE Trans. Power Electron. 2020, 35, 901–912. [Google Scholar] [CrossRef]
- Meninger, S.; Mur-Miranda, J.O.; Amirtharajah, R.; Chandrakasan, A.; Lang, J.H. Vibration-to-electric energy conversion. IEEE Trans. Very Large Scale Integr. Syst. 2001, 9, 64–76. [Google Scholar] [CrossRef]
- Khan, F.U.; Usman, M. A power-density-enhanced MEMS electrostatic energy harvester with symmetrized high-aspect ratio comb electrodes You may also like State-of-the-art in vibration-based electrostatic energy harvesting. J. Micromech. Microeng. 2019, 29, 084002. [Google Scholar]
- Lu, Y.; Marty, F.; Galayko, D.; Laheurte, J.M.; Basset, P. A power supply module for autonomous portable electronics: Ultralow-frequency MEMS electrostatic kinetic energy harvester with a comb structure reducing air damping. Microsyst. Nanoeng. 2018, 4, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, R.; Kabei, N.; Katayama, K.; Ishizuka, Y.; Tsuboi, F.; Tsuchiya, K. Development of an Electrostatic Generator that Harnesses the Motion of a Living Body. Use of a Resonant Phenomenon. JSME Int. J. Ser. C 2000, 43, 916–922. [Google Scholar] [CrossRef]
- Miao, P.; Mitcheson, P.D.; Holmes, A.S.; Yeatman, E.M.; Green, T.C.; Stark, B.H. Mems inertial power generators for biomedical applications. Microsyst. Technol. 2006, 12, 1079–1083. [Google Scholar] [CrossRef]
- Daneshvar, S.H.; Maymandi-Nejad, M. A new electro-static micro-generator for energy harvesting from diaphragm muscle. Int. J. Circuit Theory Appl. 2017, 45, 2307–2328. [Google Scholar] [CrossRef]
- Moretti, G.; Rosset, S.; Vertechy, R.; Anderson, I.; Fontana, M. A Review of Dielectric Elastomer Generator Systems. Adv. Intell. Syst. 2020, 2, 2000125. [Google Scholar] [CrossRef]
- Leese, H.S.; Tejkl, M.; Vilar, L.; Georgi, L.; Yau, H.C.; Rubio, N.; Reixach, E.; Buk, J.; Jiang, Q.; Bismarck, A.; et al. High-k dielectric screen-printed inks for mechanical energy harvesting devices. Mater. Adv. 2022, 3, 1780–1790. [Google Scholar] [CrossRef]
- Krupenkin, T.; Taylor, J.A. Reverse electrowetting as a new approach to high-power energy harvesting. Nat. Commun. 2011, 2, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Panchadar, K.; West, D.; Taylor, J.A.; Krupenkin, T. Mechanical energy harvesting using a liquid metal vortex magnetohydrodynamic generator. Appl. Phys. Lett. 2019, 114, 093901. [Google Scholar] [CrossRef]
- Tanaka, Y.; Miyoshi, T.; Suzuki, Y. Stochastic Model of Human Arm Swing Toward Standard Testing for Rotational Energy Harvester. J. Phys. Conf. Ser. 2019, 1407, 012033. [Google Scholar] [CrossRef]
- Miyoshi, T.; Adachi, M.; Suzuki, K.; Liu, Y.; Suzuki, Y. Low-profile rotational electret generator using print circuit board for energy harvesting from arm swing. In Proceedings of the IEEE International Conference on Micro Electro Mechanical Systems (MEMS), Seoul, Korea, 21–25 January 2018; pp. 230–232. [Google Scholar]
- Miyoshi, T.; Adachi, M.; Tanaka, Y.; Suzuki, Y. Low-profile rotational electret energy harvester for batteryless wearable device. In Proceedings of the IEEE/ASME International Conference on Advanced Intelligent Mechatronics, Auckland, New Zealand, 9–12 July 2018; pp. 391–394. [Google Scholar]
- Zhang, Y.; Wang, T.; Luo, A.; Hu, Y.; Li, X.; Wang, F. Micro electrostatic energy harvester with both broad bandwidth and high normalized power density. Appl. Energy 2018, 212, 362–371. [Google Scholar] [CrossRef]
- Choi, Y.-M.; Lee, M.; Jeon, Y. Wearable Biomechanical Energy Harvesting Technologies. Energies 2017, 10, 1483. [Google Scholar] [CrossRef]
- Nasiri, A.; Zabalawi, S.A.; Jeutter, D.C. A Linear Permanent Magnet Generator for Powering Implanted Electronic Devices. IEEE Trans. Power Electron. 2011, 26, 192–199. [Google Scholar] [CrossRef]
- Zurbuchen, A.; Haeberlin, A.; Pfenniger, A.; Bereuter, L.; Schaerer, J.; Jutzi, F.; Huber, C.; Fuhrer, J.; Vogel, R. Towards Batteryless Cardiac Implantable Electronic Devices–The Swiss Way. IEEE Trans. Biomed. Circ. Syst. 2017, 11, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Yu, C.-H.; Ishiyama, K. Rotary-type electromagnetic power generator using a cardiovascular system as a power source for medical implants. IEEE/ASME Trans. Mechatronics 2015, 21, 122–129. [Google Scholar] [CrossRef]
- Franzina, N.; Zurbuchen, A.; Zumbrunnen, A.; Niederhauser, T.; Reichlin, T.; Burger, J.; Haeberlin, A. A miniaturized endocardial electromagnetic energy harvester for leadless cardiac pacemakers. PLoS ONE 2020, 15, e0239667. [Google Scholar]
- Haeberlin, A.; Rösch, Y.; Tholl, M.V.; Gugler, Y.; Okle, J.; Heinisch, P.P.; Reichlin, T.; Burger, J.; Zurbuchen, A. Intracardiac Turbines Suitable for Catheter-Based Implantation-An Approach to Power Battery and Leadless Cardiac Pacemakers? IEEE Trans. Biomed. Eng. 2020, 67, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Secord, T.W.; Audi, M.C. A Tunable Resonance Cantilever for Cardiac Energy Harvesting. Cardiovasc. Eng. Technol. 2019, 10, 380–393. [Google Scholar] [CrossRef]
- Samad, F.A.; Karim, M.F.; Paulose, V.; Ong, L.C. A Curved Electromagnetic Energy Harvesting System for Wearable Electronics. IEEE Sens. J. 2016, 16, 1969–1974. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Y.; Kim, E.S. Power generation from human body motion through magnet and coil arrays with magnetic spring. J. Appl. Phys. 2014, 115, 064908. [Google Scholar] [CrossRef]
- Roberts, P.; Stanley, G.; Morgan, J.M. Abstract 2165: Harvesting the Energy of Cardiac Motion to Power a Pacemaker. Circulation 2008, 118, S_679–S_680. [Google Scholar] [CrossRef]
- Lueke, J.; Moussa, W.A. MEMS-Based Power Generation Techniques for Implantable Biosensing Applications. Sensors 2011, 11, 1433–1460. [Google Scholar] [CrossRef]
- Li, W.J.; Ho, T.C.H.; Chan, G.M.H.; Leong, P.H.W.; Wong, H.Y. Infrared signal transmission by a laser-micromachined, vibration-induced power generator. In Proceedings of the 43rd IEEE Midwest Symposium on Circuits and Systems (Cat.No.CH37144), Lansing, MI, USA, 8–11 August 2000; pp. 236–239. [Google Scholar]
- Romero, E.; Warrington, R.O.; Neuman, M.R. Body motion for powering biomedical devices. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 2–6 September 2009; pp. 2752–2755. [Google Scholar]
- Lasheras, A.; Gutiérrez, J.; Reis, S.; Sousa, D.; Silva, M.; Martins, P.; Lanceros-Mendez, S.; Barandiarán, J.M.; Shishkin, D.A.; Potapov, A.P. Energy harvesting device based on a metallic glass/PVDF magnetoelectric laminated composite. Smart Mater. Struct. 2015, 24, 065024. [Google Scholar] [CrossRef]
- Zhang, J.; Kang, Y.; Gao, Y.; Weng, G.J. Experimental Investigation of the Magnetoelectric Effect in NdFeB-Driven A-Line Shape Terfenol-D/PZT-5A Structures. Materials 2019, 12, 1055. [Google Scholar] [CrossRef]
- Toshiyoshi, H.; Ju, S.; Honma, H.; Ji, C.H.; Fujita, H. MEMS vibrational energy harvesters. Sci. Technol. Adv. Mater. 2019, 20, 124–143. [Google Scholar] [CrossRef]
- Zurbuchen, A.; Haeberlin, A.; Bereuter, L.; Pfenniger, A.; Bosshard, S.; Kernen, M.; Heinisch, P.P.; Fuhrer, J.; Vogel, R. Endocardial energy harvesting by electromagnetic induction. IEEE Trans. Biomed. Eng. 2018, 65, 424–430. [Google Scholar] [CrossRef]
- Gupta, R.K.; Shi, Q.; Dhakar, L.; Wang, T.; Heng, C.H.; Lee, C. Broadband Energy Harvester Using Non-linear Polymer Spring and Electromagnetic/Triboelectric Hybrid Mechanism. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Arroyo, E.; Badel, A.; Formosa, F. Energy harvesting from ambient vibrations: Electromagnetic device and synchronous extraction circuit. Orig. Artic. J. Intell. Mater. Syst. Struct. 2013, 24, 2023–2035. [Google Scholar] [CrossRef]
- Li, K.; He, Q.; Wang, J.; Zhou, Z.; Li, X. Wearable energy harvesters generating electricity from low-frequency human limb movement. Microsyst. Nanoeng. 2018, 4, 1–13. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, D.; Luo, J.; Xie, S.; Pu, H.; Li, Z. Power density improvement based on investigation of initial relative position in an electromagnetic energy harvester with self-powered applications. Smart Mater. Struct. 2021, 30, 065005. [Google Scholar] [CrossRef]
- Ramadass, Y.K.; Chandrakasan, A.P. A Battery-Less Thermoelectric Energy Harvesting Interface Circuit With 35 mV Startup Voltage. IEEE J. -Solid-State Circ. 2011, 46, 333–341. [Google Scholar] [CrossRef]
- Wang, W.; Deng, J.; Liu, Y.; Lu, Y.; Jia, Y.; Yu, C. Recent advances in power supply strategies for untethered neural implants. J. Micromech. Microeng. 2021, 31, 104003. [Google Scholar] [CrossRef]
- Zou, Y.; Bo, L.; Li, Z. Recent progress in human body energy harvesting for smart bioelectronic system. Fundam. Res. 2021, 1, 364–382. [Google Scholar] [CrossRef]
- Stark, I.; Stordeur, M. New micro thermoelectric devices based on bismuth telluride-type thin solid films. In Proceedings of the Eighteenth International Conference on Thermoelectrics, Proceedings, ICT’99 (Cat. No.99TH8407), Baltimore, MD, USA, 29 August–2 September 1999; pp. 465–472. [Google Scholar] [CrossRef]
- Strasser, M.; Aigner, R.; Lauterbach, C.; Sturm, T.F.; Franosch, M.; Wachutka, G. Micromachined CMOS thermoelectric generators as on-chip power supply. Sens. Actuators A Phys. 2004, 114, 362–370. [Google Scholar] [CrossRef]
- Leonov, V. Thermoelectric Energy Harvesting of Human Body Heat for Wearable Sensors. IEEE Sens. J. 2013, 13, 2284–2291. [Google Scholar] [CrossRef]
- Hoang, D.C.; Tan, Y.K.; Chng, H.B.; Panda, S.K. Thermal energy harvesting from human warmth for wireless body area network in medical healthcare system. In Proceedings of the 2009 International Conference on Power Electronics and Drive Systems (PEDS), 2–5 November 2009; pp. 1277–1282. [Google Scholar] [CrossRef]
- Moriarty, G.P.; De, S.; King, P.J.; Khan, U.; Via, M.; King, J.A.; Coleman, J.N.; Grunlan, J.C. Thermoelectric behavior of organic thin film nanocomposites. J. Polym. Sci. B Polym. Phys. 2013, 51, 119–123. [Google Scholar] [CrossRef]
- Hong, C.T.; Kang, Y.H.; Ryu, J.; Cho, S.Y.; Jang, K.S. Spray-printed CNT/P3HT organic thermoelectric films and power generators. J. Mater. Chem. A Mater. 2015, 3, 21428–21433. [Google Scholar] [CrossRef]
- Cho, C.; Wallace, K.L.; Tzeng, P.; Hsu, J.H.; Yu, C.; Grunlan, J.C. Outstanding Low Temperature Thermoelectric Power Factor from Completely Organic Thin Films Enabled by Multidimensional Conjugated Nanomaterials. Adv. Energy Mater. 2016, 6, 1502168. [Google Scholar] [CrossRef]
- Wu, G.; Gao, C.; Chen, G.; Wang, X.; Wang, H. High-performance organic thermoelectric modules based on flexible films of a novel n-type single-walled carbon nanotube. J. Mater. Chem. A Mater. 2016, 4, 14187–14193. [Google Scholar] [CrossRef]
- Pudzs, K.; Vembris, A.; Rutkis, M.; Woodward, S. Thin Film Organic Thermoelectric Generator Based on Tetrathiotetracene. Adv. Electron. Mater. 2017, 3, 1600429. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, W.; Kang, Y.H.; Cho, S.Y.; Jang, K.S. Wet-spinning and post-treatment of CNT/PEDOT:PSS composites for use in organic fiber-based thermoelectric generators. Carbon N. Y. 2018, 133, 293–299. [Google Scholar] [CrossRef]
- Peng, J.; Witting, I.; Geisendorfer, N.; Wang, M.; Chang, M.; Jakus, A.; Kenel, C.; Yan, X.; Shah, R.; Snyder, G.J.; et al. 3D extruded composite thermoelectric threads for flexible energy harvesting. Nat. Commun. 2019, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wen, N.; Fan, Z.; Yang, S.; Zhao, Y.; Cong, T.; Xu, S.; Zhang, H.; Wang, J.; Huang, H.; Li, C.; et al. Highly conductive, ultra-flexible and continuously processable PEDOT:PSS fibers with high thermoelectric properties for wearable energy harvesting. Nano Energy 2020, 78, 105361. [Google Scholar] [CrossRef]
- Kuai, Q.; Leung, H.Y.; Wan, Q.; Mok, P.K.T. A High-Efficiency Dual-Polarity Thermoelectric Energy-Harvesting Interface Circuit With Cold Startup and Fast-Searching ZCD. IEEE J. Solid-State Circ. 2022, 57, 1899–1912. [Google Scholar] [CrossRef]
- Hu, Z.; Mu, E.; Wu, Z. MEMS thermoelectric power chip for large scale thermal energy harvesting. In Proceedings of the 2020 IEEE 8th Electronics System-Integration Technology Conference, ESTC 2020, Vestfold, Norway, 15–18 September 2020. [Google Scholar] [CrossRef]
- Kumar, P.M.; Jagadeesh Babu, V.; Subramanian, A.; Bandla, A.; Thakor, N.; Ramakrishna, S.; Wei, H. The Design of a Thermoelectric Generator and Its Medical Applications. Designs 2019, 3, 22. [Google Scholar] [CrossRef]
- Selvam, C.; Manikandan, S.; Krishna, N.V.; Lamba, R.; Kaushik, S.C.; Mahian, O. Enhanced thermal performance of a thermoelectric generator with phase change materials. Int. Commun. Heat Mass Transf. 2020, 114, 104561. [Google Scholar] [CrossRef]
- Fan, F.-R.; Tian, Z.-Q.; Wang, Z.L. Flexible triboelectric generator. Nano Energy 2012, 1, 328–334. [Google Scholar] [CrossRef]
- Zhu, G.; Pan, C.; Guo, W.; Chen, C.Y.; Zhou, Y.; Yu, R.; Wang, Z.L. Triboelectric-Generator-Driven Pulse Electrodeposition for Micropatterning. Nano Lett. 2012, 12, 4960–4965. [Google Scholar] [CrossRef]
- Zhang, X.-S.; Han, M.; Kim, B.; Bao, J.-F.; Brugger, J.; Zhang, H. All-in-one self-powered flexible microsystems based on triboelectric nanogenerators. Nano Energy 2018, 47, 410–426. [Google Scholar] [CrossRef]
- Parida, K.; Bhavanasi, V.; Kumar, V.; Wang, J.; Lee, P.S. Fast charging self-powered electric double layer capacitor. J. Power Sources 2017, 342, 70–78. [Google Scholar] [CrossRef]
- Kumar, V.; Park, S.; Parida, K.; Bhavanasi, V.; Lee, P.S. Multi-responsive supercapacitors: Smart solution to store electrical energy. Mater. Today Energy 2017, 4, 41–57. [Google Scholar] [CrossRef]
- Pu, X.; Guo, H.; Chen, J.; Wang, X.; Xi, Y.; Hu, C.; Wang, Z.L. Eye motion triggered self-powered mechnosensational communication system using triboelectric nanogenerator. Sci. Adv. 2017, 3, e1700694. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Guo, H.; Chen, L.; Wang, X.; Wei, D.; Hu, C. Double-induced-mode integrated triboelectric nanogenerator based on spring steel to maximize space utilization. Nano Res. 2016, 9, 3355–3363. [Google Scholar] [CrossRef]
- Hwang, H.J.; Jung, Y.; Choi, K.; Kim, D.; Park, J.; Choi, D. Comb-structured triboelectric nanogenerators for multi-directional energy scavenging from human movements. Sci. Technol. Adv. Mater. 2019, 20, 725–732. [Google Scholar] [CrossRef]
- Pan, R.; Xuan, W.; Chen, J.; Dong, S.; Jin, H.; Wang, X.; Li, H.; Luo, J. Fully biodegradable triboelectric nanogenerators based on electrospun polylactic acid and nanostructured gelatin films. Nano Energy 2018, 45, 193–202. [Google Scholar] [CrossRef]
- Lin, Z.; Chen, J.; Li, X.; Zhou, Z.; Meng, K.; Wei, W.; Yang, J.; Wang, Z.L. Triboelectric Nanogenerator Enabled Body Sensor Network for Self-Powered Human Heart-Rate Monitoring. ACS Nano 2017, 11, 8830–8837. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Dai, K.; Yi, F.; Han, Y.; Wang, X.; You, Z. Optimization of triboelectric nanogenerator load characteristics considering the air breakdown effect. Nano Energy 2018, 53, 706–715. [Google Scholar] [CrossRef]
- Hazarika, A.; Deka, B.K.; Jeong, C.; Park, Y.b.; Park, H.W. Biomechanical Energy-Harvesting Wearable Textile-Based Personal Thermal Management Device Containing Epitaxially Grown Aligned Ag-Tipped-NiCo Nanowires/Reduced Graphene Oxide. Adv. Funct. Mater. 2019, 29, 1903144. [Google Scholar] [CrossRef]
- Sagan, C.; Mullen, G. Earth and Mars: Evolution of Atmospheres and Surface Temperatures. Science 1972, 177, 52–56. [Google Scholar] [CrossRef]
- Murakawa, K.; Kobayashi, M.; Nakamura, O.; Kawata, S. A wireless near-infrared energy system for medical implants. IEEE Eng. Med. Biol. Mag. 1999, 18, 70–72. [Google Scholar] [CrossRef]
- Goto, K.; Nakagawa, T.; Nakamura, O.; Kawata, S. An implantable power supply with an optically rechargeable lithium battery. IEEE Trans. Biomed. Eng. 2001, 48, 830–833. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A.; Kochubey, V.I.; Tuchin, V.v. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D Appl. Phys. 2005, 38, 2543. [Google Scholar] [CrossRef]
- Sordillo, L.A.; Pu, Y.; Pratavieira, S.; Budansky, Y.; Alfano, R.R. Deep optical imaging of tissue using the second and third near-infrared spectral windows. J. Biomed. Opt. 2014, 19, 056004. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Mancini, M.C.; Nie, S. Second window for in vivo imaging. Nat. Nanotechnol. 2009, 4, 710. [Google Scholar] [CrossRef] [PubMed]
- Kayes, B.M.; Nie, H.; Twist, R.; Spruytte, S.G.; Reinhardt, F.; Kizilyalli, I.C.; Higashi, G.S. 27.6% Conversion efficiency, a new record for single-junction solar cells under 1 sun illumination. In Proceedings of the Conference Record of the IEEE Photovoltaic Specialists Conference, Seattle, WA, USA, 19–24 June 2011; pp. 000004–000008. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, A.; Green, M.A.; Ferrazza, F. 19.8% efficient ‘honeycomb’ textured multicrystalline and 24.4% monocrystalline silicon solar cells. Appl. Phys. Lett. 1998, 73, 1991. [Google Scholar] [CrossRef]
- Haeberlin, A.; Zurbuchen, A.; Schaerer, J.; Wagner, J.; Walpen, S.; Huber, C.; Haeberlin, H.; Fuhrer, J.; Vogel, R. Successful pacing using a batteryless sunlight-powered pacemaker. Europace 2014, 16, 1534–1539. [Google Scholar] [CrossRef] [PubMed]
- Bereuter, L.; Williner, S.; Pianezzi, F.; Bissig, B.; Buecheler, S.; Burger, J.; Vogel, R.; Zurbuchen, A.; Haeberlin, A. Energy Harvesting by Subcutaneous Solar Cells: A Long-Term Study on Achievable Energy Output. Ann. Biomed. Eng. 2017, 45, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Yang, Z.; Meacham, K.; Cvetkovic, C.; Corbin, E.A.; Vázquez-Guardado, A.; Xue, M.; Yin, L.; Boroumand, J.; Pakeltis, G.; et al. Biodegradable Monocrystalline Silicon Photovoltaic Microcells as Power Supplies for Transient Biomedical Implants. Adv. Energy Mater. 2018, 8, 1703035. [Google Scholar] [CrossRef]
- Kim, J.; Seo, J.; Jung, D.; Lee, T.; Ju, H.; Han, J.; Kim, N.; Jeong, J.; Cho, S.; Seol, J.H.; et al. Active photonic wireless power transfer into live tissues. Proc. Natl. Acad. Sci. USA 2020, 117, 16856–16863. [Google Scholar] [CrossRef]
- Moon, E.; Blaauw, D.; Phillips, J.D. Subcutaneous Photovoltaic Infrared Energy Harvesting for Bio-implantable Devices. IEEE Trans. Electron Devices 2017, 64, 2432–2437. [Google Scholar] [CrossRef]
- Song, K.; Han, J.H.; Lim, T.; Kim, N.; Shin, S.; Kim, J.; Choo, H.; Jeong, S.; Kim, Y.C.; Wang, Z.L.; et al. Subdermal Flexible Solar Cell Arrays for Powering Medical Electronic Implants. Adv. Healthc. Mater. 2016, 5, 1572–1580. [Google Scholar] [CrossRef]
- Hu, F.; Li, W.; Zou, M.; Li, Y.; Chen, F.; Lin, N.; Guo, W.; Liu, X.Y. Subcutaneous Energy/Signal Transmission Based on Silk Fibroin Up-Conversion Photonic Amplification. ACS Nano 2021, 15, 9559–9567. [Google Scholar] [CrossRef]
- Kendall, C.J. Parasitic Power Collection in Shoe Mounted Devices. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 1998. [Google Scholar]
- Moorthy, B.; Baek, C.; Wang, J.E.; Jeong, C.K.; Moon, S.; Park, K.I.; Kim, D.K. Piezoelectric energy harvesting from a PMN–PT single nanowire. RSC Adv. 2017, 7, 260–265. [Google Scholar] [CrossRef]
- Wang, W.; Cao, J.; Bowen, C.R.; Zhou, S.; Lin, J. Optimum resistance analysis and experimental verification of nonlinear piezoelectric energy harvesting from human motions. Energy 2017, 118, 221–230. [Google Scholar] [CrossRef]
- Daneshvar, S.H.; Maymandi-Nejad, M.; Sachdev, M.; Redoute, J.-M. A Charge-Depletion Study of an Electrostatic Generator With Adjustable Output Voltage. IEEE Sens. J. 2019, 19, 1028–1039. [Google Scholar] [CrossRef]
- Risquez, S.; Woytasik, M.; Wei, J.; Parrain, F.; Lefeuvre, E. Design of a 3D multilayer out-of-plane overlap electrostatic energy harvesting MEMS for medical implant applications. In Proceedings of the 2015 Symposium on Design, Test, Integration and Packaging of MEMS/MOEMS (DTIP), Montpellier, France, 27–30 April 2015; pp. 1–5. [Google Scholar] [CrossRef]
- Naruse, Y.; Matsubara, N.; Mabuchi, K.; Izumi, M.; Suzuki, S. Electrostatic micro power generation from low-frequency vibration such as human motion. J. Micromech. Microeng. 2009, 19, 094002. [Google Scholar] [CrossRef]
- Huang, W.; Tzeng, K.; Cheng, M.; Huang, R. A silicon mems micro power generator for wearable micro devices. J. Chin. Inst. Eng. 2007, 30, 133–140. [Google Scholar] [CrossRef]
- Chen, C.; Chau, L.Y.; Liao, W.-H. A knee-mounted biomechanical energy harvester with enhanced efficiency and safety. Smart Mater. Struct. 2017, 26, 065027. [Google Scholar] [CrossRef]
- Ylli, K.; Hoffmann, D.; Willmann, A.; Becker, P.; Folkmer, B.; Manoli, Y. Energy harvesting from human motion: Exploiting swing and shock excitations. Smart Mater. Struct. 2015, 24, 025029. [Google Scholar] [CrossRef]
- Hyland, M.; Hunter, H.; Liu, J.; Veety, E.; Vashaee, D. Wearable thermoelectric generators for human body heat harvesting. Appl. Energy 2016, 182, 518–524. [Google Scholar] [CrossRef]
- Kim, M.K.; Kim, M.S.; Jo, S.E.; Kim, Y.J. Flexible thermoelectric generator for human body heat energy harvesting. Electron. Lett. 2012, 48, 1015–1017. [Google Scholar] [CrossRef]
- Leonov, V.; Fiorini, P.; Sedky, S.; Torfs, T.; van Hoof, C. Thermoelectric mems generators as a power supply for a body area network. In Proceedings of the 13th International Conference on Solid-State Sensors, Actuators and Microsystems, Seoul, Korea, 5–9 June 2005; pp. 291–294. [Google Scholar] [CrossRef]
- Sahatiya, P.; Kannan, S.; Badhulika, S. Few layer MoS2 and in situ poled PVDF nanofibers on low cost paper substrate as high performance piezo-triboelectric hybrid nanogenerator: Energy harvesting from handwriting and human touch. Appl. Mater. Today 2018, 13, 91–99. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, G.; Yang, W.; Jing, Q.; Bai, P.; Yang, Y.; Hou, T.C.; Wang, Z.L. Harmonic-Resonator-Based Triboelectric Nanogenerator as a Sustainable Power Source and a Self-Powered Active Vibration Sensor. Adv. Mater. 2013, 25, 6094–6099. [Google Scholar] [CrossRef]
- Zhang, R.; Örtegren, J.; Hummelgård, M.; Olsen, M.; Andersson, H.; Olin, H. Harvesting triboelectricity from the human body using non-electrode triboelectric nanogenerators. Nano Energy 2018, 45, 298–303. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, H.; Shi, B.; Xue, X.; Liu, Z.; Jin, Y.; Ma, Y.; Zou, Y.; Wang, X.; An, Z.; et al. In Vivo Self-Powered Wireless Cardiac Monitoring via Implantable Triboelectric Nanogenerator. ACS Nano 2016, 10, 6510–6518. [Google Scholar] [CrossRef]
- Wu, F.; Li, C.; Yin, Y.; Cao, R.; Li, H.; Zhang, X.; Zhao, S.; Wang, J.; Wang, B.; Xing, Y.; et al. A Flexible, Lightweight, and Wearable Triboelectric Nanogenerator for Energy Harvesting and Self-Powered Sensing. Adv. Mater. Technol. 2019, 4, 1800216. [Google Scholar] [CrossRef]
- Ho, J.S.; Yeh, A.J.; Neofytou, E.; Kim, S.; Tanabe, Y.; Patlolla, B.; Beygui, R.E.; Poon, A.S. Wireless power transfer to deep-tissue microimplants. Proc. Natl. Acad. Sci. USA 2014, 111, 7974–7979. [Google Scholar] [CrossRef] [PubMed]
- Oh, T.; Islam, S.K.; To, G.; Mahfouz, M. Powering wearable sensors with a low-power CMOS piezoelectric energy harvesting circuit. In Proceedings of the 2017 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Rochester, MN, USA, 7–10 May 2017; pp. 308–313. [Google Scholar] [CrossRef]
- Singh, N.; Kanaujia, B.K.; Beg, M.T.; Khan, T.; Kumar, S. A dual polarized multiband rectenna for RF energy harvesting. AEU—Int. J. Electron. Commun. 2018, 93, 123–131. [Google Scholar] [CrossRef]
- Singh, N.; Kumar, S.; Kanaujia, B.K.; Beg, M.T.; Kumar, S. A compact and efficient graphene FET based RF energy harvester for green communication. AEU—Int. J. Electron. Commun. 2020, 115, 153059. [Google Scholar] [CrossRef]
- Agrawal, S.; Parihar, M.S.; Kondekar, P.N. Broadband Rectenna for Radio Frequency Energy Harvesting Application. IETE J. Res. 2018, 64, 347–353. [Google Scholar] [CrossRef]
- Vital, D.; Bhardwaj, S.; Volakis, J.L. Textile-Based Large Area RF-Power Harvesting System for Wearable Applications. IEEE Trans. Antennas Propag. 2020, 68, 2323–2331. [Google Scholar] [CrossRef]
- Krikidis, I.; Timotheou, S.; Nikolaou, S.; Zheng, G.; Ng, D.W.K.; Schober, R. Simultaneous wireless information and power transfer in modern communication systems. IEEE Commun. Mag. 2014, 52, 104–110. [Google Scholar] [CrossRef]
- Akkermans, J.A.G.; van Beurden, M.C.; Doodeman, G.J.N.; Visser, H.J. Analytical models for low-power rectenna design. IEEE Antennas Wirel. Propag. Lett. 2005, 4, 187–190. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, H.; Zhang, L.; Zhang, C.; Wang, Z.; Chen, X. An Energy-Efficient ASIC for Wireless Body Sensor Networks in Medical Applications. IEEE Trans. Biomed. Circ. Syst. 2010, 4, 11–18. [Google Scholar] [CrossRef]
- Xia, L.; Cheng, J.; Glover, N.E.; Chiang, P. 0.56 V, –20 dBm RF-Powered, Multi-Node Wireless Body Area Network System-on-a-Chip With Harvesting-Efficiency Tracking Loop. IEEE J. -Solid-State Circ. 2014, 49, 1345–1355. [Google Scholar] [CrossRef]
- Chung, W.Y.; Le, G.T.; Tran, T.V.; Nguyen, N.H. Novel proximal fish freshness monitoring using batteryless smart sensor tag. Sens. Actuators B Chem. C 2017, 248, 910–916. [Google Scholar] [CrossRef]
- Lam, M.B.; Dang, N.T.; Nguyen, T.H.; Chung, W.Y. A Neural Network-Based Model of Radio Frequency Energy Harvesting Characteristics in a Self-Powered Food Monitoring System. Undefined 2019, 19, 8813–8823. [Google Scholar] [CrossRef]
- Roundy, S.; Steingart, D.; Frechette, L.; Wright, P.; Rabaey, J. Power Sources for Wireless Sensor Networks; Springer: Berlin/Heidelberg, Germany, 2004; pp. 1–17. [Google Scholar]
- Grätzel, M. Photoelectrochemical cells. In Materials for Sustainable Energy; Co-Published with Macmillan Publishers Ltd.: London, UK, 2010; pp. 26–32. [Google Scholar]
- Dieffenderfer, J.P.; Beppler, E.; Novak, T.; Whitmire, E.; Jayakumar, R.; Randall, C.; Qu, W.; Rajagopalan, R.; Bozkurt, A. Solar powered wrist worn acquisition system for continuous photoplethysmogram monitoring. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 3142–3145. [Google Scholar] [CrossRef]
- Ostfeld, A.E.; Gaikwad, A.M.; Khan, Y.; Arias, A.C. High-performance flexible energy storage and harvesting system for wearable electronics. Sci. Rep. 2016, 6, 26122. [Google Scholar] [CrossRef]
- Toh, W.Y.; Tan, Y.K.; Koh, W.S.; Siek, L. Autonomous Wearable Sensor Nodes With Flexible Energy Harvesting. IEEE Sens. J. 2014, 14, 2299–2306. [Google Scholar] [CrossRef]
- Wu, T.; Wu, F.; Redoute, J.-M.; Yuce, M.R. An Autonomous Wireless Body Area Network Implementation Towards IoT Connected Healthcare Applications. IEEE Access 2017, 5, 11413–11422. [Google Scholar] [CrossRef]
- Lechêne, B.P.; Cowell, M.; Pierre, A.; Evans, J.W.; Wright, P.K.; Arias, A.C. Organic solar cells and fully printed super-capacitors optimized for indoor light energy harvesting. Nano Energy 2016, 26, 631–640. [Google Scholar] [CrossRef]
- Ayazian, S.; Akhavan, V.A.; Soenen, E.; Hassibi, A. A Photovoltaic-Driven and Energy-Autonomous CMOS Implantable Sensor. IEEE Trans. Biomed. Circ. Syst. 2012, 6, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Haeberlin, A.; Zurbuchen, A.; Walpen, S.; Schaerer, J.; Niederhauser, T.; Huber, C.; Tanner, H.; Servatius, H.; Seiler, J.; Haeberlin, H.; et al. The first batteryless, solar-powered cardiac pacemaker. Heart Rhythm. 2015, 12, 1317–1323. [Google Scholar] [CrossRef]
- Wu, T.; Redoute, J.-M.; Yuce, M.R. A Wireless Implantable Sensor Design With Subcutaneous Energy Harvesting for Long-Term IoT Healthcare Applications. IEEE Access 2018, 6, 35801–35808. [Google Scholar] [CrossRef]
- Phillips, W.B.; Towe, B.C.; Larson, P.J. An ultrasonically-driven piezoelectric neural stimulator. In Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (IEEE Cat. No.03CH37439), Cancun, Mexico, 17–21 September 2003; pp. 1983–1986. [Google Scholar] [CrossRef]
- Towe, B.C.; Larson, P.J.; Gulick, D.W. Wireless ultrasound-powered biotelemetry for implants. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 2–6 September 2009; pp. 5421–5424. [Google Scholar] [CrossRef]
- Zhu, Y.; Moheimani, S.O.R.; Yuce, M.R. Ultrasonic Energy Transmission and Conversion Using a 2-D MEMS Resonator. IEEE Electron Device Lett. 2010, 31, 374–376. [Google Scholar] [CrossRef]
- Shi, Q.; Wang, T.; Lee, C. MEMS Based Broadband Piezoelectric Ultrasonic Energy Harvester (PUEH) for Enabling Self-Powered Implantable Biomedical Devices. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Zhang, T.; Liang, H.; Wang, Z.; Qiu, C.; Peng, Y.B.; Zhu, X.; Li, J.; Ge, X.; Xu, J.; Huang, X.; et al. Piezoelectric ultrasound energy–harvesting device for deep brain stimulation and analgesia applications. Sci. Adv. 2022, 8, 159. [Google Scholar] [CrossRef]
- Jian, L.G.; Yang, Y.; Chen, R.; Lu, G.; Li, R.; Li, D.; Humayun, M.S.; Shung, K.K.; Zhu, J.; Chen, Y.; et al. Flexible piezoelectric ultrasonic energy harvester array for bio-implantable wireless generator. Nano Energy 2019, 56, 216–224. [Google Scholar] [CrossRef]
- Wan, X.; Chen, P.; Xu, Z.; Mo, X.; Jin, H.; Yang, W.; Wang, S.; Duan, J.; Hu, B.; Luo, Z.; et al. Hybrid-Piezoelectret Based Highly Efficient Ultrasonic Energy Harvester for Implantable Electronics. Adv. Funct. Mater. 2022, 32, 2200589. [Google Scholar] [CrossRef]
- Culurciello, E.; Andreou, A.G. Capacitive Inter-Chip Data and Power Transfer for 3-D VLSI. IEEE Trans. Circ. Syst. II Express Briefs 2006, 53, 1348–1352. [Google Scholar] [CrossRef]
- Sambas, A.; Mamat, M.; Arafa, A.A.; Mahmoud, G.M.; Mohamed, M.A.; Sanjaya, W.S.M. A New Design of Capacitive Power Transfer Based on Hybrid Approach for Biomedical Implantable Device. Int. J. Electr. Comput. Eng. 2019, 9, 2365–2376. [Google Scholar] [CrossRef]
- Sha, X.; Zheng, P.; Karimi, Y.; Stanacevic, M. Capacitive link for data communication between free floating mm-sized brain implants. In Proceedings of the 2021 IEEE International Symposium on Medical Measurements and Applications, MeMeA 2021—Conference Proceedings, Lausanne, Switzerland, 23–25 June 2021. [Google Scholar] [CrossRef]
- Riistama, J.; Väisänen, J.; Heinisuo, S.; Harjunpää, H.; Arra, S.; Kokko, K.; Mäntylä, M.; Kaihilahti, J.; Heino, P.; Kellomäki, M.; et al. Wireless and inductively powered implant for measuring electrocardiogram. Med. Biol. Eng. Comput. 2007, 45, 1163–1174. [Google Scholar] [CrossRef] [PubMed]
- Hsia, M.-L.; Tsai, Y.-S.; Chen, O.T.-C. An UHF Passive RFID Transponder Using A Low-Power Clock Generator without Passive Components. In Proceedings of the 2006 49th IEEE International Midwest Symposium on Circuits and Systems, San Juan, Puerto Rico, 6–9 August 2006; pp. 11–15. [Google Scholar] [CrossRef]
- Parramon, J.; Doguet, P.; Marin, D.; Verleyssen, M.; Munoz, R.; Leija, L.; Valderrama, E. ASIC-based batteryless implantable telemetry microsystem for recording purposes. In Proceedings of the 19th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Magnificent Milestones and Emerging Opportunities in Medical Engineering (Cat. No.97CH36136), Chicago, IL, USA, 30 October–2 November 1997; pp. 2225–2228. [Google Scholar] [CrossRef]
- Sauer, C.; Stanacevic, M.; Cauwenberghs, G.; Thakor, N. Power harvesting and telemetry in CMOS for implanted devices. IEEE Trans. Circ. Syst. I Regul. Pap. 2005, 52, 2605–2613. [Google Scholar] [CrossRef]
- Hannan, M.A.; Mutashar, S.; Samad, S.A.; Hussain, A. Energy harvesting for the implantable biomedical devices: Issues and challenges. Biomed. Eng. Online 2014, 13, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.; Kim, J.A.; Gurgu, A.; Khawaja, M.; Cozma, D.; Chelu, M.G. Innovations in Cardiac Implantable Electronic Devices. Cardiovasc. Drugs Ther. 2021, 36, 1–13. [Google Scholar] [CrossRef]
- Lyu, H.; Wang, J.; La, J.H.; Chung, J.M.; Babakhani, A. An Energy-Efficient Wirelessly Powered Millimeter-Scale Neurostimulator Implant Based on Systematic Codesign of an Inductive Loop Antenna and a Custom Rectifier. IEEE Trans. Biomed. Circ. Syst 2018, 12, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Mirbozorgi, S.A.; Yeon, P.; Ghovanloo, M. Robust Wireless Power Transmission to mm-Sized Free-Floating Distributed Implants. IEEE Trans. Biomed. Circ. Syst. 2017, 11, 692–702. [Google Scholar] [CrossRef]
- Ghomian, T.; Mehraeen, S. Survey of energy scavenging for wearable and implantable devices. Energy 2019, 178, 33–49. [Google Scholar] [CrossRef]
- Jokic, P.; Magno, M. Powering smart wearable systems with flexible solar energy harvesting. In Proceedings of the 2017 IEEE International Symposium on Circuits and Systems (ISCAS), Baltimore, MD, USA, 28–31 May 2017; pp. 1–4. [Google Scholar] [CrossRef]
- Ahnood, A.; Fox, K.E.; Apollo, N.V.; Lohrmann, A.; Garrett, D.J.; Nayagam, D.A.; Karle, T.; Stacey, A.; Abberton, K.M.; Morrison, W.A.; et al. Diamond encapsulated photovoltaics for transdermal power delivery. Biosens. Bioelectron. 2016, 77, 589–597. [Google Scholar] [CrossRef]
- Laube, T.; Brockmann, C.; Buß, R.; Lau, C.; Höck, K.; Stawski, N.; Stieglitz, T.; Richter, H.A.; Schilling, H. Optical energy transfer for intraocular microsystems studied in rabbits. Graefe’s Arch. Clin. Exp. Ophthalmol. 2004, 242, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, Y.Z.; Chen, H.C.; Liu, C.W.; Chen, Y.S.; Lo, Y.C.; Tsao, C.S.; Huang, Y.C.; Liu, S.W.; Wong, K.T.; et al. Unveiling the underlying mechanism of record-high efficiency organic near-infrared photodetector harnessing a single-component photoactive layer. Mater. Horizons 2020, 7, 1171–1179. [Google Scholar] [CrossRef]
- Lo, Y.K.; Chen, K.; Gad, P.; Liu, W. A Fully-Integrated High-Compliance Voltage SoC for Epi-Retinal and Neural Prostheses. IEEE Trans. Biomed. Circ. Syst. 2013, 7, 761–772. [Google Scholar] [CrossRef]
- Lee, S.Y.; Hsieh, C.H.; Yang, C.M. Wireless Front-End With Power Management for an Implantable Cardiac Microstimulator. IEEE Trans. Biomed. Circ. Syst. 2012, 6, 28–38. [Google Scholar] [CrossRef]
- Kelly, S.K.; Shire, D.B.; Chen, J.; Doyle, P.; Gingerich, M.D.; Cogan, S.F.; Drohan, W.A.; Behan, S.; Theogarajan, L.; Wyatt, J.L.; et al. A Hermetic Wireless Subretinal Neurostimulator for Vision Prostheses. IEEE Trans. Biomed. Eng. 2011, 58, 3197–3205. [Google Scholar] [CrossRef] [PubMed]
- Knecht, O.; Kolar, J.W. Performance Evaluation of Series-Compensated IPT Systems for Transcutaneous Energy Transfer. IEEE Trans. Power Electrons 2019, 34, 438–451. [Google Scholar] [CrossRef]
- Jegadeesan, R.; Nag, S.; Agarwal, K.; Thakor, N.v.; Guo, Y.-X. Enabling Wireless Powering and Telemetry for Peripheral Nerve Implants. IEEE J. Biomed. Health Inform. 2015, 19, 958–970. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Deng, Y.; Wang, Y.; Shen, S.; Gulfam, R. High-performance photovoltaic-thermoelectric hybrid power generation system with optimized thermal management. Energy 2016, 100, 91–101. [Google Scholar] [CrossRef]
- Li, G.; Shittu, S.; Diallo, T.M.O.; Yu, M.; Zhao, X.; Ji, J. A review of solar photovoltaic-thermoelectric hybrid system for electricity generation. Energy 2018, 158, 41–58. [Google Scholar] [CrossRef]
- Kraemer, D.; Hu, L.; Muto, A.; Chen, X.; Chen, G.; Chiesa, M. Photovoltaic-thermoelectric hybrid systems: A general optimization methodology. Appl. Phys. Lett. 2008, 92, 243503. [Google Scholar] [CrossRef]
- Magno, M.; Brunelli, D.; Sigrist, L.; Andri, R.; Cavigelli, L.; Gomez, A.; Benini, L. InfiniTime: Multi-sensor wearable bracelet with human body harvesting. Sustain. Comput. Inform. Syst. 2016, 11, 38–49. [Google Scholar] [CrossRef]
- Park, K.-T.; Shin, S.M.; Tazebay, A.S.; Um, H.D.; Jung, J.Y.; Jee, S.W.; Oh, M.W.; Park, S.D.; Yoo, B.; Yu, C.; et al. Lossless hybridization between photovoltaic and thermoelectric devices. Sci. Rep. 2013, 3, 2123. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zi, Y.; He, X.; Guo, H.; Lai, Y.C.; Wang, J.; Zhang, S.L.; Wu, C.; Cheng, G.; Wang, Z.L. Concurrent Harvesting of Ambient Energy by Hybrid Nanogenerators for Wearable Self-Powered Systems and Active Remote Sensing. ACS Appl. Mater. Interfaces 2018, 10, 14708–14715. [Google Scholar] [CrossRef]
- Chung, J.; Yong, H.; Moon, H.; Duong, Q.V.; Choi, S.T.; Kim, D.; Lee, S. Hand-Driven Gyroscopic Hybrid Nanogenerator for Recharging Portable Devices. Adv. Sci. 2018, 5, 1801054. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Liu, X.; Wang, X.; Li, Q. An energy extraction enhanced interface circuit for piezoelectric and thermoelectric energy harvesting. IEICE Electron. Express 2019, 16. [Google Scholar] [CrossRef]
- Xie, Y.; Zhang, Z.; Zhou, H.; Wang, Z.; Lin, Y.; Chen, Y.; Lv, Y.; Chen, Y.; Zhang, C. Multisource Energy Harvester with Coupling Structure and Multiplexing Mechanism. Adv. Mater. Interfaces 2022, 9, 2200468. [Google Scholar] [CrossRef]
- Zhang, Z.; He, T.; Zhao, J.; Liu, G.; Wang, Z.L.; Zhang, C. Tribo-thermoelectric and tribovoltaic coupling effect at metal-semiconductor interface. Mater. Today Phys. 2021, 16, 100295. [Google Scholar] [CrossRef]
- Mitcheson, P.D.; Toh, T.T. Power Management Electronics. In Energy Harvesting for Autonomous Systems; Artech House: Norwood, MA, USA, 2010. [Google Scholar]
- Newell, D.; Duffy, M. Review of Power Conversion and Energy Management for Low-Power, Low-Voltage Energy Harvesting Powered Wireless Sensors. IEEE Trans. Power Electron. 2019, 34, 9794–9805. [Google Scholar] [CrossRef]
- Kong, N.; Ha, D.S. Low-Power Design of a Self-powered Piezoelectric Energy Harvesting System With Maximum Power Point Tracking. IEEE Trans. Power Electron. 2012, 27, 2298–2308. [Google Scholar] [CrossRef]
- Szarka, G.D.; Burrow, S.G.; Proynov, P.P.; Stark, B.H. Maximum Power Transfer Tracking for Ultralow-Power Electromagnetic Energy Harvesters. IEEE Trans. Power Electron. 2014, 29, 201–212. [Google Scholar] [CrossRef]
- Kim, H.; Kim, S.; Kwon, C.-K.; Min, Y.-J.; Kim, C.; Kim, S.-W. An Energy-Efficient Fast Maximum Power Point Tracking Circuit in an 800-µW Photovoltaic Energy Harvester. IEEE Trans. Power Electron. 2013, 28, 2927–2935. [Google Scholar] [CrossRef]
- Sankman, J.; Ma, D. A 12 µW to 1.1 mW AIM Piezoelectric Energy Harvester for Time-Varying Vibrations With 450 nA IQ. IEEE Trans. Power Electron. 2015, 30, 632–643. [Google Scholar] [CrossRef]
- Chew, Z.J.; Zhu, M. Adaptive Maximum Power Point Finding Using Direct V OC /2 Tracking Method With Microwatt Power Consumption for Energy Harvesting. IEEE Trans. Power Electron. 2018, 33, 8164–8173. [Google Scholar] [CrossRef]
- Fang, S.; Xia, H.; Xia, Y.; Ye, Y.; Shi, G.; Wang, X.; Chen, Z. An Efficient Piezoelectric Energy Harvesting Circuit With Series-SSHI Rectifier and FNOV-MPPT Control Technique. IEEE Trans. Ind. Electron. 2021, 68, 7146–7155. [Google Scholar] [CrossRef]
- Shim, M.; Kim, J.; Jeong, J.; Park, S.; Kim, C. Self-Powered 30 µW to 10 mW Piezoelectric Energy Harvesting System With 9.09 ms/V Maximum Power Point Tracking Time. IEEE J. -Solid-State Circ. 2015, 50, 2367–2379. [Google Scholar] [CrossRef]
- Fan, S.; Wei, R.; Zhao, L.; Yang, X.; Geng, L.; Feng, P.X.-L. An Ultralow Quiescent Current Power Management System With Maximum Power Point Tracking (MPPT) for Batteryless Wireless Sensor Applications. IEEE Trans. Power Electron. 2018, 33, 7326–7337. [Google Scholar] [CrossRef]
- Shrivastava, A.; Roberts, N.E.; Khan, O.U.; Wentzloff, D.D.; Calhoun, B.H. A 10 mV-Input Boost Converter With Inductor Peak Current Control and Zero Detection for Thermoelectric and Solar Energy Harvesting With 220 mV Cold-Start and -14.5 dBm, 915 MHz RF Kick-Start. IEEE J. -Solid-State Circ. 2015, 50, 1820–1832. [Google Scholar] [CrossRef]
- Carreon-Bautista, S.; Erbay, C.; Han, A.; Sanchez-Sinencio, E. Power Management System With Integrated Maximum Power Extraction Algorithm for Microbial Fuel Cells. IEEE Trans. Energy Convers. 2015, 30, 262–272. [Google Scholar] [CrossRef]
- Doms, I.; Merken, P.; van Hoof, C.; Mertens, R.P. Capacitive Power Management Circuit for Micropower Thermoelectric Generators With a 1.4 μA Controller. IEEE J. -Solid-State Circ. 2009, 44, 2824–2833. [Google Scholar] [CrossRef]
- Liu, X.; Huang, L.; Ravichandran, K.; Sanchez-Sinencio, E. A Highly Efficient Reconfigurable Charge Pump Energy Harvester With Wide Harvesting Range and Two-Dimensional MPPT for Internet of Things. IEEE J. -Solid-State Circ. 2016, 51, 1302–1312. [Google Scholar] [CrossRef]
- Shuvo, M.M.H. Edge AI: Leveraging the Full Potential of Deep Learning. In Recent Innovations in Artificial Intelligence and Smart Applications, Studies in Computational Intelligence; Springer: Berlin, Germany, 2022; Volume 1061. [Google Scholar]
- Li, X.; Ning, S.; Liu, Z.; Yan, Z.; Luo, C.; Zhuang, Z. Designing phononic crystal with anticipated band gap through a deep learning based data-driven method. Comput. Methods Appl. Mech. Eng. 2020, 361, 112737. [Google Scholar] [CrossRef]
- Luo, C.; Ning, S.; Liu, Z.; Zhuang, Z. Interactive inverse design of layered phononic crystals based on reinforcement learning. Extrem. Mech. Lett. 2020, 36, 100651. [Google Scholar] [CrossRef]
- Jin, Y.; Wang, W.; Wen, Z.; Torrent, D.; Djafari-Rouhani, B. Topological states in twisted pillared phononic plates. Extrem. Mech. Lett. 2020, 39, 100777. [Google Scholar] [CrossRef]
- Wahba, M.A.; Ashour, A.S.; Ghannam, R. Prediction of Harvestable Energy for Self-Powered Wearable Healthcare Devices: Filling a Gap. IEEE Access 2020, 8, 170336–170354. [Google Scholar] [CrossRef]
- Kwan, J.C.; Chaulk, J.M.; Fapojuwo, A.O. A Coordinated Ambient/Dedicated Radio Frequency Energy Harvesting Scheme Using Machine Learning. IEEE Sens. J. 2020, 20, 13808–13823. [Google Scholar] [CrossRef]
- Hussein, D.; Bhat, G.; Doppa, J.R. Adaptive Energy Management for Self-Sustainable Wearables in Mobile Health. Proc. AAAI 2022. [Google Scholar]
- Akinaga, H. Recent advances and future prospects in energy harvesting technologies. Jpn. J. Appl. Phys. 2020, 59, 110201. [Google Scholar] [CrossRef]
- Ye, Y.; Azmat, F.; Adenopo, I.; Chen, Y.; Shi, R. RF energy modelling using machine learning for energy harvesting communications systems. Int. J. Commun. Syst. 2021, 34, e4688. [Google Scholar] [CrossRef]
- Xiao, L.; Wu, K.; Tian, X.; Luo, J. Activity-specific caloric expenditure estimation from kinetic energy harvesting in wearable devices. Pervasive Mob. Comput. 2020, 67, 101185. [Google Scholar] [CrossRef]
- Guo, X.; He, T.; Zhang, Z.; Luo, A.; Wang, F.; Ng, E.J.; Zhu, Y.; Liu, H.; Lee, C. Artificial Intelligence-Enabled Caregiving Walking Stick Powered by Ultra-Low-Frequency Human Motion. ACS Nano 2021, 15, 19054–19069. [Google Scholar] [CrossRef] [PubMed]
- Wen, F.; Sun, Z.; He, T.; Shi, Q.; Zhu, M.; Zhang, Z.; Li, L.; Zhang, T.; Lee, C. Machine Learning Glove Using Self-Powered Conductive Superhydrophobic Triboelectric Textile for Gesture Recognition in VR/AR Applications. Adv. Sci. 2020, 7, 2000261. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Sun, Z.; Zhang, Z.; Shi, Q.; He, T.; Liu, H.; Chen, T.; Lee, C. Haptic-feedback smart glove as a creative human-machine interface (HMI) for virtual/augmented reality applications. Sci. Adv. 2020, 6, eaaz8693. [Google Scholar] [CrossRef]
- Zhu, J.; Zhu, M.; Shi, Q.; Wen, F.; Liu, L.; Dong, B.; Haroun, A.; Yang, Y.; Vachon, P.; Guo, X.; et al. Progress in TENG technology—A journey from energy harvesting to nanoenergy and nanosystem. EcoMat 2020, 2, e12058. [Google Scholar] [CrossRef]
- Zhang, Z.; He, T.; Zhu, M.; Shi, Q.; Lee, C. Smart Triboelectric Socks for Enabling Artificial Intelligence of Things (AIoT) Based Smart Home and Healthcare. In Proceedings of the 2020 IEEE 33rd International Conference on Micro Electro Mechanical Systems (MEMS), Vancouver, BC, Canada, 18–22 January 2020; pp. 80–83. [Google Scholar] [CrossRef]
- Ji, X.; Zhao, T.; Zhao, X.; Lu, X.; Li, T. Triboelectric Nanogenerator Based Smart Electronics via Machine Learning. Adv. Mater Technol. 2020, 5, 1900921. [Google Scholar] [CrossRef]
- Politi, B.; Foucaran, A.; Camara, N. Low-Cost Sensors for Indoor PV Energy Harvesting Estimation Based on Machine Learning. Energies 2022, 15, 1144. [Google Scholar] [CrossRef]
- Park, Y.; Cho, K.; Kim, S. Performance Prediction of Hybrid Energy Harvesting Devices Using Machine Learning. ACS Appl. Mater. Interfaces 2022, 14, 11248–11254. [Google Scholar] [CrossRef]
- Zhu, J.; Cho, M.; Li, Y.; He, T.; Ahn, J.; Park, J.; Ren, T.L.; Lee, C.; Park, I. Machine learning-enabled textile-based graphene gas sensing with energy harvesting-assisted IoT application. Nano Energy 2021, 86, 106035. [Google Scholar] [CrossRef]
- Hamdi, R.; Chen, M.; Said, A.b.; Qaraqe, M.; Poor, H.V. Federated Learning over Energy Harvesting Wireless Networks. IEEE Internet Things J. 2022, 9, 92–103. [Google Scholar] [CrossRef]
- Guler, B.; Yener, A. Energy-Harvesting Distributed Machine Learning. In Proceedings of the 2021 IEEE International Symposium on Information Theory, Virtual, 11–16 July 2021; pp. 320–325. [Google Scholar] [CrossRef]
- Pervez, I.; Antoniadis, C.; Massoud, Y. A Reduced Search Space Exploration Metaheuristic Algorithm for MPPT. IEEE Access 2022, 10, 26090–26100. [Google Scholar] [CrossRef]
- Peng, Y.; Wang, Y.; Liu, Y.; Gao, K.; Yin, T.; Yu, H. Few-shot learning based multi-weather-condition impedance identification for MPPT-controlled PV converters. IET Renew. Power Gener. 2022, 16, 1345–1353. [Google Scholar] [CrossRef]
- Singh, Y.; Pal, N. Reinforcement learning with fuzzified reward approach for MPPT control of PV systems. Sustain. Energy Technol. Assessments 2021, 48, 101665. [Google Scholar] [CrossRef]
- Zafar, M.H.; Khan, N.M.; Mansoor, M.; Khan, U.A. Towards green energy for sustainable development: Machine learning based MPPT approach for thermoelectric generator. J. Clean. Prod. 2022, 351, 131591. [Google Scholar] [CrossRef]
- Ouyang, H.; Liu, Z.; Li, N.; Shi, B.; Zou, Y.; Xie, F.; Ma, Y.; Li, Z.; Li, H.; Zheng, Q.; et al. Symbiotic cardiac pacemaker. Nat. Commun. 2019, 10, 1821. [Google Scholar] [CrossRef]
- Guan, H.; Lv, D.; Zhong, T.; Dai, Y.; Xing, L.; Xue, X.; Zhang, Y.; Zhan, Y. Self-powered, wireless-control, neural-stimulating electronic skin for in vivo characterization of synaptic plasticity. Nano Energy 2020, 67, 104182. [Google Scholar] [CrossRef]
- Yao, G.; Kang, L.; Li, J.; Long, Y.; Wei, H.; Ferreira, C.A.; Jeffery, J.J.; Lin, Y.; Cai, W.; Wang, X. Effective weight control via an implanted self-powered vagus nerve stimulation device. Nat. Commun. 2018, 9, 5349. [Google Scholar] [CrossRef]
- Lee, S.; Wang, H.; Peh, W.Y.X.; He, T.; Yen, S.C.; Thakor, N.V.; Lee, C. Mechano-neuromodulation of autonomic pelvic nerve for underactive bladder: A triboelectric neurostimulator integrated with flexible neural clip interface. Nano Energy 2019, 60, 449–456. [Google Scholar] [CrossRef]
- Hassani, F.A.; Mogan, R.P.; Gammad, G.G.; Wang, H.; Yen, S.C.; Thakor, N.V.; Lee, C. Toward Self-Control Systems for Neurogenic Underactive Bladder: A Triboelectric Nanogenerator Sensor Integrated with a Bistable Micro-Actuator. ACS Nano 2018, 12, 3487–3501. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, H.; He, T.; He, B.; Thakor, N.v.; Lee, C. Investigation of Low-Current Direct Stimulation for Rehabilitation Treatment Related to Muscle Function Loss Using Self-Powered TENG System. Adv. Sci. 2019, 6, 1900149. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Thakor, N.v.; Lee, C. Self-Powered Direct Muscle Stimulation Using a Triboelectric Nanogenerator (TENG) Integrated with a Flexible Multiple-Channel Intramuscular Electrode. ACS Nano 2019, 13, 3589–3599. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Xue, X.; Ma, Y.; Han, M.; Zhang, W.; Xu, Z.; Zhang, H.; Zhang, H. Implantable and self-powered blood pressure monitoring based on a piezoelectric thinfilm: Simulated, in vitro and in vivo studies. Nano Energy 2016, 22, 453–460. [Google Scholar] [CrossRef]
- Southcott, M.; MacVittie, K.; Halámek, J.; Halámková, L.; Jemison, W.D.; Lobel, R.; Katz, E. A pacemaker powered by an implantable biofuel cell operating under conditions mimicking the human blood circulatory system—Battery not included. Phys. Chem. Chem. Phys. 2013, 15, 6278. [Google Scholar] [CrossRef] [PubMed]
- MacVittie, K.; Halámek, J.; Halámková, L.; Southcott, M.; Jemison, W.D.; Lobel, R.; Katz, E. From ‘cyborg’ lobsters to a pacemaker powered by implantable biofuel cells. Energy Environ. Sci. 2013, 6, 81–86. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, A.C.; Liu, Y.; Zhou, Y.; Wang, Z.L. Functional Electrical Stimulation by Nanogenerator with 58 V Output Voltage. Nano Lett. 2012, 12, 3086–3090. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.-T.; Kim, Y.; Lee, J.H.; Oh, S.; Jeong, C.K.; Park, D.Y.; Ryu, J.; Kwon, H.; Lee, S.G.; Joung, B.; et al. Self-powered deep brain stimulation via a flexible PIMNT energy harvester. Energy Environ. Sci. 2015, 8, 2677–2684. [Google Scholar] [CrossRef]
- Lee, S.; Wang, H.; Wang, J.; Shi, Q.; Yen, S.C.; Thakor, N.V.; Lee, C. Battery-free neuromodulator for peripheral nerve direct stimulation. Nano Energy 2018, 50, 148–158. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; He, T.; Li, Z.; Lee, C. Direct muscle stimulation using diode-amplified triboelectric nanogenerators (TENGs). Nano Energy 2019, 63, 103844. [Google Scholar] [CrossRef]
- Alam, M.; Li, S.; Ahmed, R.U.; Yam, Y.M.; Thakur, S.; Wang, X.Y.; Tang, D.; Ng, S.; Zheng, Y.P. Development of a battery-free ultrasonically powered functional electrical stimulator for movement restoration after paralyzing spinal cord injury. J. Neuroeng. Rehabil. 2019, 16, 36. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ma, Y.; Ouyang, H.; Shi, B.; Li, N.; Jiang, D.; Xie, F.; Qu, D.; Zou, Y.; Huang, Y.; et al. Transcatheter Self-Powered Ultrasensitive Endocardial Pressure Sensor. Adv. Funct. Mater. 2019, 29, 1807560. [Google Scholar] [CrossRef]
- Ma, Y.; Zheng, Q.; Liu, Y.; Shi, B.; Xue, X.; Ji, W.; Liu, Z.; Jin, Y.; Zou, Y.; An, Z.; et al. Self-Powered, One-Stop, and Multifunctional Implantable Triboelectric Active Sensor for Real-Time Biomedical Monitoring. Nano Lett. 2016, 16, 6042–6051. [Google Scholar] [CrossRef]
- Curry, E.J.; Ke, K.; Chorsi, M.T.; Wrobel, K.S.; Miller III, A.N.; Patel, A.; Kim, I.; Feng, J.; Yue, L.; Wu, Q.; et al. Biodegradable Piezoelectric Force Sensor. Proc. Natl. Acad. Sci. USA 2018, 115, 909–914. [Google Scholar] [CrossRef]
- Jeerapan, I.; Sempionatto, J.R.; Pavinatto, A.; You, J.-M.; Wang, J. Stretchable biofuel cells as wearable textile-based self-powered sensors. J. Mater. Chem. A Mater. 2016, 4, 18342–18353. [Google Scholar] [CrossRef]
- Jiang, D.; Ouyang, H.; Shi, B.; Zou, Y.; Tan, P.; Qu, X.; Chao, S.; Xi, Y.; Zhao, C.; Fan, Y.; et al. A wearable noncontact free-rotating hybrid nanogenerator for self-powered electronics. InfoMat 2020, 2, 1191–1200. [Google Scholar] [CrossRef]
- Ouyang, H.; Tian, J.; Sun, G.; Zou, Y.; Liu, Z.; Li, H.; Zhao, L.; Shi, B.; Fan, Y.; Fan, Y.; et al. Self-Powered Pulse Sensor for Antidiastole of Cardiovascular Disease. Adv. Mater. 2017, 29, 1703456. [Google Scholar] [CrossRef]
- Park, D.Y.; Joe, D.J.; Kim, D.H.; Park, H.; Han, J.H.; Jeong, C.K.; Park, H.; Park, J.G.; Joung, B.; Lee, K.J. Self-Powered Real-Time Arterial Pulse Monitoring Using Ultrathin Epidermal Piezoelectric Sensors. Adv. Mater. 2017, 29, 1702308. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.-C.; Deng, J.; Niu, S.; Peng, W.; Wu, C.; Liu, R.; Wen, Z.; Wang, Z.L. Electric Eel-Skin-Inspired Mechanically Durable and Super-Stretchable Nanogenerator for Deformable Power Source and Fully Autonomous Conformable Electronic-Skin Applications. Adv. Mater. 2016, 28, 10024–10032. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Bandodkar, A.J.; Valdés-Ramírez, G.; Windmiller, J.R.; Yang, Z.; Ramírez, J.; Chan, G.; Wang, J. Electrochemical Tattoo Biosensors for Real-Time Noninvasive Lactate Monitoring in Human Perspiration. Anal. Chem. 2013, 85, 6553–6560. [Google Scholar] [CrossRef]
- Teymourian, H.; Moonla, C.; Tehrani, F.; Vargas, E.; Aghavali, R.; Barfidokht, A.; Tangkuaram, T.; Mercier, P.P.; Dassau, E.; Wang, J. Microneedle-Based Detection of Ketone Bodies along with Glucose and Lactate: Toward Real-Time Continuous Interstitial Fluid Monitoring of Diabetic Ketosis and Ketoacidosis. Anal. Chem. 2020, 92, 2291–2300. [Google Scholar] [CrossRef]
- Kim, J.; Khan, S.; Wu, P.; Park, S.; Park, H.; Yu, C.; Kim, W. Self-charging wearables for continuous health monitoring. Nano Energy 2021, 79, 105419. [Google Scholar] [CrossRef]
- Kim, C.S.; Yang, H.M.; Lee, J.; Lee, G.S.; Choi, H.; Kim, Y.J.; Lim, S.H.; Cho, S.H.; Cho, B.J. Self-Powered Wearable Electrocardiography Using a Wearable Thermoelectric Power Generator. ACS Energy Lett. 2018, 3, 501–507. [Google Scholar] [CrossRef]
- Vivekananthan, V.; Chandrasekhar, A.; Alluri, N.R.; Purusothaman, Y.; Kim, W.J.; Kang, C.N.; Kim, S.J. A flexible piezoelectric composite nanogenerator based on doping enhanced lead-free nanoparticles. Mater. Lett. 2019, 249, 73–76. [Google Scholar] [CrossRef]
- Mohsen, S.; Zekry, A.; Youssef, K.; Abouelatta, M. A Self-powered Wearable Wireless Sensor System Powered by a Hybrid Energy Harvester for Healthcare Applications. Wirel Pers Commun. 2021, 116, 3143–3164. [Google Scholar] [CrossRef]
- Wagih, M.; Hillier, N.; Yong, S.; Weddell, A.S.; Beeby, S. RF-Powered Wearable Energy Harvesting and Storage Module Based on E-Textile Coplanar Waveguide Rectenna and Supercapacitor. IEEE Open J. Antennas Propag. 2021, 2, 302–314. [Google Scholar] [CrossRef]
- Huang, X.; Wang, L.; Wang, H.; Zhang, B.; Wang, X.; Stening, R.Y.; Sheng, X.; Yin, L. Materials Strategies and Device Architectures of Emerging Power Supply Devices for Implantable Bioelectronics. Small 2020, 16, 1902827. [Google Scholar] [CrossRef]
- Dinis, H.; Mendes, P.M. A comprehensive review of powering methods used in state-of-the-art miniaturized implantable electronic devices. Biosens. Bioelectron. 2021, 172, 112781. [Google Scholar] [CrossRef]
- Sheng, H.; Zhou, J.; Li, B.; He, Y.; Zhang, X.; Liang, J.; Zhou, J.; Su, Q.; Xie, E.; Lan, W.; et al. A thin, deformable, high-performance supercapacitor implant that can be biodegraded and bioabsorbed within an animal body. Sci. Adv. 2021, 7, eabe3097. [Google Scholar] [CrossRef]
- Guan, Y.-S.; Thukral, A.; Zhang, S.; Sim, K.; Wang, X.; Zhang, Y.; Ershad, F.; Rao, Z.; Pan, F.; Wang, P.; et al. Air/water interfacial assembled rubbery semiconducting nanofilm for fully rubbery integrated electronics. Sci. Adv. 2020, 6, eabb3656. [Google Scholar] [CrossRef]
- Sheng, H.; Zhang, X.; Liang, J.; Shao, M.; Xie, E.; Yu, C.; Lan, W. Recent Advances of Energy Solutions for Implantable Bioelectronics. Adv. Healthc. Mater. 2021, 10, 2100199. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Koo, J.; Rogers, J.A. Inorganic materials for transient electronics in biomedical applications. MRS Bull. 2020, 45, 103–112. [Google Scholar] [CrossRef]
- Wang, P.; Hu, M.; Wang, H.; Chen, Z.; Feng, Y.; Wang, J.; Ling, W.; Huang, Y. The Evolution of Flexible Electronics: From Nature, Beyond Nature, and To Nature. Adv. Sci. 2020, 7, 2001116. [Google Scholar] [CrossRef]
- Ershad, F.; Thukral, A.; Yue, J.; Comeaux, P.; Lu, Y.; Shim, H.; Sim, K.; Kim, N.I.; Rao, Z.; Guevara, R.; et al. Ultra-conformal drawn-on-skin electronics for multifunctional motion artifact-free sensing and point-of-care treatment. Nat. Commun. 2020, 11, 3823. [Google Scholar] [CrossRef] [PubMed]
- Chao, S.; Ouyang, H.; Jiang, D.; Fan, Y.; Li, Z. Triboelectric nanogenerator based on degradable materials. EcoMat 2021, 3, e12072. [Google Scholar] [CrossRef]
- Niu, Q.; Huang, L.; Lv, S.; Shao, H.; Fan, S.; Zhang, Y. Pulse-driven bio-triboelectric nanogenerator based on silk nanoribbons. Nano Energy 2020, 74, 104837. [Google Scholar] [CrossRef]
- Peng, X.; Dong, K.; Ye, C.; Jiang, Y.; Zhai, S.; Cheng, R.; Liu, D.; Gao, X.; Wang, J.; Wang, Z.L. A breathable, biodegradable, antibacterial, and self-powered electronic skin based on all-nanofiber triboelectric nanogenerators. Sci. Adv. 2020, 6, eaba9624. [Google Scholar] [CrossRef] [PubMed]
- Dagdeviren, C.; Hwang, S.W.; Su, Y.; Kim, S.; Cheng, H.; Gur, O.; Haney, R.; Omenetto, F.G.; Huang, Y.; Rogers, J.A. Transient, Biocompatible Electronics and Energy Harvesters Based on ZnO. Small 2013, 9, 3398–3404. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Guo, H.; Ribera, J.; Wu, C.; Tu, K.; Binelli, M.; Panzarasa, G.; Schwarze, F.W.; Wang, Z.L.; Burgert, I. Sustainable and Biodegradable Wood Sponge Piezoelectric Nanogenerator for Sensing and Energy Harvesting Applications. ACS Nano 2020, 14, 14665–14674. [Google Scholar] [CrossRef]
- Lin, J.C. A new IEEE standard for safety levels with respect to human exposure to radio-frequency radiation. IEEE Antennas Propag. Mag. 2006, 48, 157–159. [Google Scholar] [CrossRef]
- Lu, D.; Yan, Y.; Avila, R.; Kandela, I.; Stepien, I.; Seo, M.H.; Bai, W.; Yang, Q.; Li, C.; Haney, C.R.; et al. Bioresorbable, Wireless, Passive Sensors as Temporary Implants for Monitoring Regional Body Temperature. Adv. Healthc. Mater. 2020, 9, 2000942. [Google Scholar] [CrossRef]
- Guo, Q.; Koo, J.; Xie, Z.; Avila, R.; Yu, X.; Ning, X.; Zhang, H.; Liang, X.; Kim, S.B.; Yan, Y.; et al. A Bioresorbable Magnetically Coupled System for Low-Frequency Wireless Power Transfer. Adv. Funct. Mater. 2019, 29, 1905451. [Google Scholar] [CrossRef]
- Zhao, J.; Ghannam, R.; Htet, K.O.; Liu, Y.; Law, M.K.; Roy, V.A.; Michel, B.; Imran, M.A.; Heidari, H. Self-Powered Implantable Medical Devices: Photovoltaic Energy Harvesting Review. Adv. Healthc. Mater. 2020, 9, 2000779. [Google Scholar] [CrossRef]
- Kim, T.B.; Ho, C.-T.B. Validating the moderating role of age in multi-perspective acceptance model of wearable healthcare technology. Telemat. Inform. 2021, 61, 101603. [Google Scholar] [CrossRef]
- Huarng, K.-H.; Yu, T.H.-K.; Lee, C.F. Adoption model of healthcare wearable devices. Technol. Forecast. Soc. Chang. 2022, 174, 121286. [Google Scholar] [CrossRef]
- Yin, Z.; Yan, J.; Fang, S.; Wang, D.; Han, D. User acceptance of wearable intelligent medical devices through a modified unified theory of acceptance and use of technology. Ann. Transl. Med. 2022, 10, 629. [Google Scholar] [CrossRef]
- Zhao, S.; Fang, C.; Yang, J.; Sawan, M. Emerging Energy-Efficient Biosignal-Dedicated Circuit Techniques: A Tutorial Brief. IEEE Trans. Circ. Syst. II Express Briefs 2022, 69, 2592–2597. [Google Scholar] [CrossRef]
- Damaschke, J.M. Design of a low-input-voltage converter for thermoelectric generator. IEEE Trans. Ind. Appl. 1997, 33, 1203–1207. [Google Scholar] [CrossRef]
- Thielen, M.; Sigrist, L.; Magno, M.; Hierold, C.; Benini, L. Human body heat for powering wearable devices: From thermal energy to application. Energy Convers. Manag. 2017, 131, 44–54. [Google Scholar] [CrossRef]
- Mitcheson, P.D. Energy harvesting for human wearable and implantable bio-sensors. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, Buenos Aires, Argentina, 31 August–4 September 2010; pp. 3432–3436. [Google Scholar]
- Borges, L.M.; Chávez-Santiago, R.; Barroca, N.; Velez, F.J.; Balasingham, I. Radio-frequency energy harvesting for wearable sensors. Healthc. Technol. Lett. 2015, 2, 22–27. [Google Scholar] [CrossRef]
- Shigeta, R.; Sasaki, T.; Quan, D.M.; Kawahara, Y.; Vyas, R.J.; Tentzeris, M.M.; Asami, T. Ambient RF Energy Harvesting Sensor Device With Capacitor-Leakage-Aware Duty Cycle Control. IEEE Sens. J. 2013, 13, 2973–2983. [Google Scholar] [CrossRef]
- Karlen, W.; Floreano, D. Adaptive Sleep–Wake Discrimination for Wearable Devices. IEEE Trans. Biomed. Eng. 2011, 58, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Jelicic, V.; Magno, M.; Brunelli, D.; Bilas, V.; Benini, L. Benefits of Wake-Up Radio in Energy-Efficient Multimodal Surveillance Wireless Sensor Network. IEEE Sens. J. 2014, 14, 3210–3220. [Google Scholar] [CrossRef]
- Anastasi, G.; Conti, M.; di Francesco, M.; Passarella, A. Energy conservation in wireless sensor networks: A survey. Ad. Hoc. Netw. 2009, 7, 537–568. [Google Scholar] [CrossRef]
- Hsu, J.; Zahedi, S.; Kansal, A.; Srivastava, M.; Raghunathan, V. Adaptive duty cycling for energy harvesting systems. In Proceedings of the 2006 International Symposium on Low Power Electronics and Design—ISLPED ’06, Bavaria, Germany, 4–6 October 2006; p. 180. [Google Scholar]
- Mallela, V.S.; Ilankumaran, V.; Rao, N.S. Trends in cardiac pacemaker batteries. Indian Pacing Electrophysiol. J. 2004, 4, 201–212. [Google Scholar] [PubMed]
- Pu, X.; Li, L.; Song, H.; Du, C.; Zhao, Z.; Jiang, C.; Cao, G.; Hu, W.; Wang, Z.L. A Self-Charging Power Unit by Integration of a Textile Triboelectric Nanogenerator and a Flexible Lithium-Ion Battery for Wearable Electronics. Adv. Mater. 2015, 27, 2472–2478. [Google Scholar] [CrossRef]
- Gaikwad, A.M.; Khau, B.v.; Davies, G.; Hertzberg, B.; Steingart, D.A.; Arias, A.C. A High Areal Capacity Flexible Lithium-Ion Battery with a Strain-Compliant Design. Adv. Energy Mater. 2015, 5, 1401389. [Google Scholar] [CrossRef]
- Lee, Y.-H.; Kim, J.S.; Noh, J.; Lee, I.; Kim, H.J.; Choi, S.; Seo, J.; Jeon, S.; Kim, T.S.; Lee, J.Y.; et al. Wearable Textile Battery Rechargeable by Solar Energy. Nano Lett. 2013, 13, 5753–5761. [Google Scholar] [CrossRef]
- Li, N.; Chen, Z.; Ren, W.; Li, F.; Cheng, H.-M. Flexible graphene-based lithium ion batteries with ultrafast charge and discharge rates. Proc. Natl. Acad. Sci. USA 2012, 109, 17360–17365. [Google Scholar] [CrossRef]
- IEEE Std 802.15.1-2005 (Revision of IEEE Std 802.15.1-2002); IEEE Standard for Information technology—Local and metropolitan area networks—Specific requirements– Part 15.1a: Wireless Medium Access Control (MAC) and Physical Layer (PHY) specifications for Wireless Personal Area Networks (WPAN). IEEE: New York, NY, USA, 14 June 2005; pp. 1–700.
- Baker, N. ZigBee and Bluetooth strengths and weaknesses for industrial applications. IEEE Comput. Control Eng. 2005, 16, 20–25. [Google Scholar] [CrossRef]
- Hsu, J.M.; Chiang, T.C.; Yu, Y.C.; Teng, W.G.; Hou, T.W. A New Energy Efficient and Reliable MedRadio Scheme Based on Cooperative Communication for Implanted Medical Devices. Int. J. Distrib. Sens. Netw. 2015, 11, 239597. [Google Scholar] [CrossRef]
- Li, H.; Tan, J. Heartbeat-Driven Medium-Access Control for Body Sensor Networks. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 44–51. [Google Scholar] [PubMed]
- Hassan, O.; Paul, T.; Shuvo, M.M.H.; Parvin, D.; Thakker, R.; Chen, M.; Mosa, A.S.M.; Islam, S.K. Energy Efficient Deep Learning Inference Embedded on FPGA for Sleep Apnea Detection. J. Sign. Process. Syst. 2022, 94, 609–619. [Google Scholar] [CrossRef]
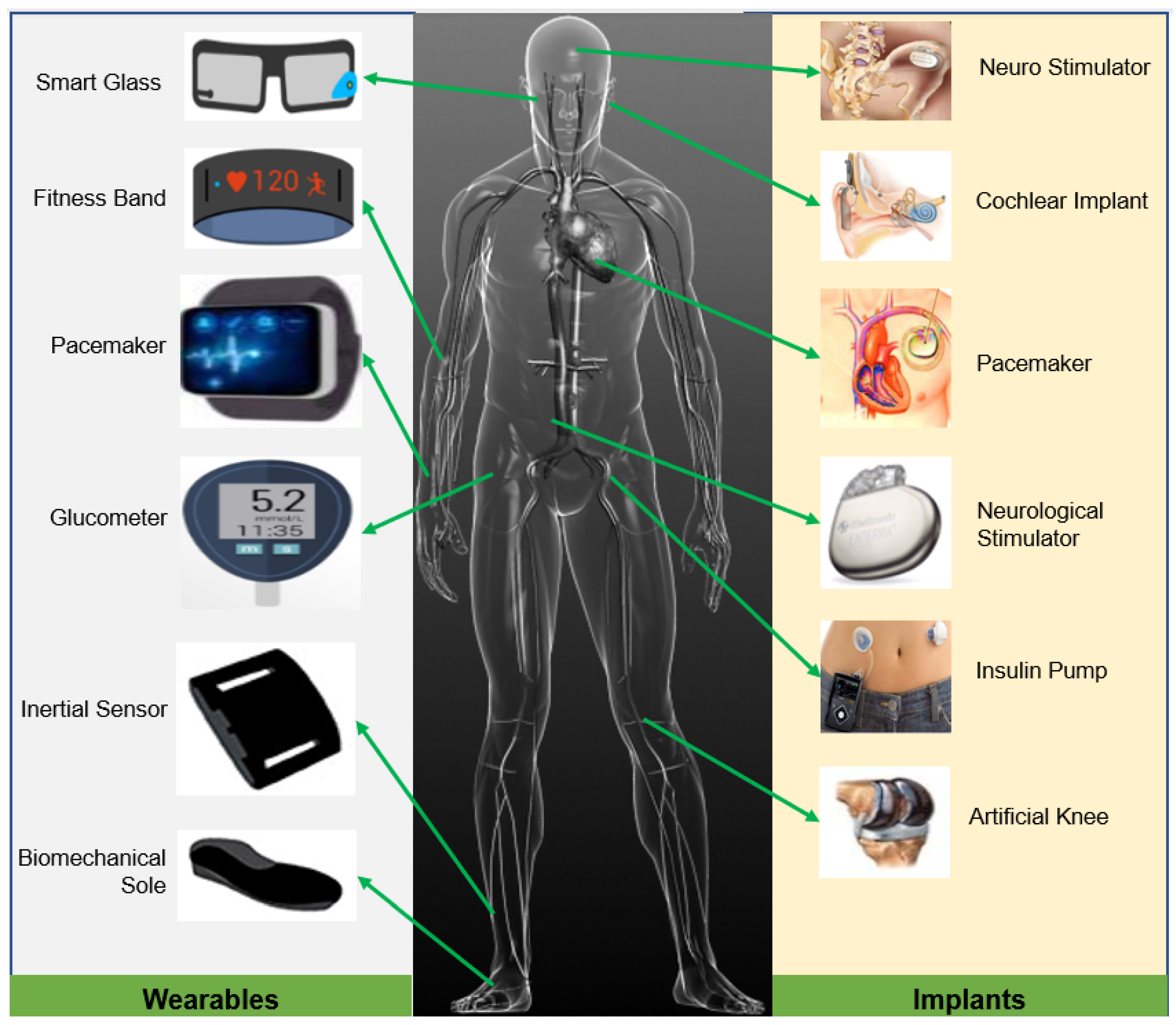

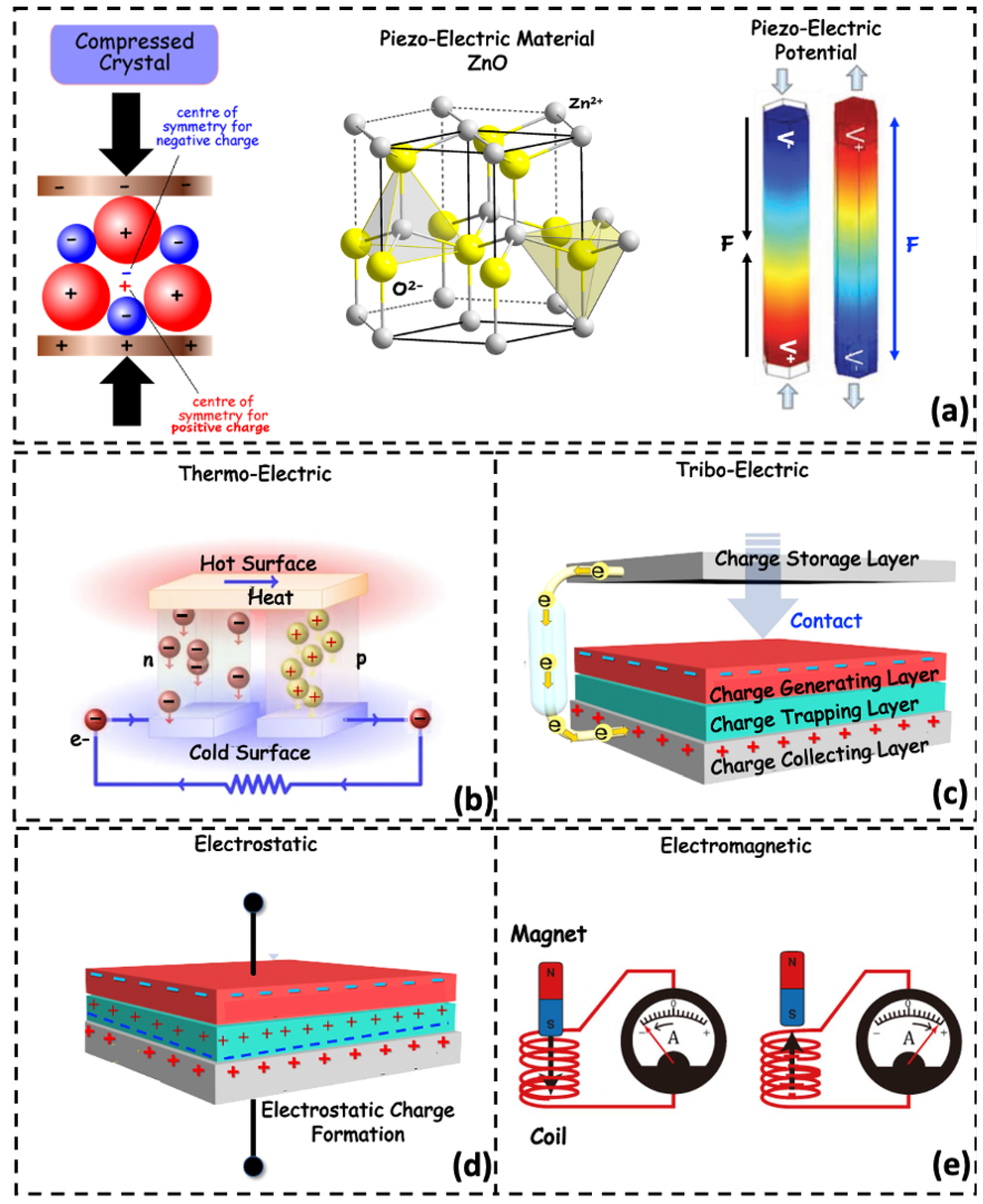
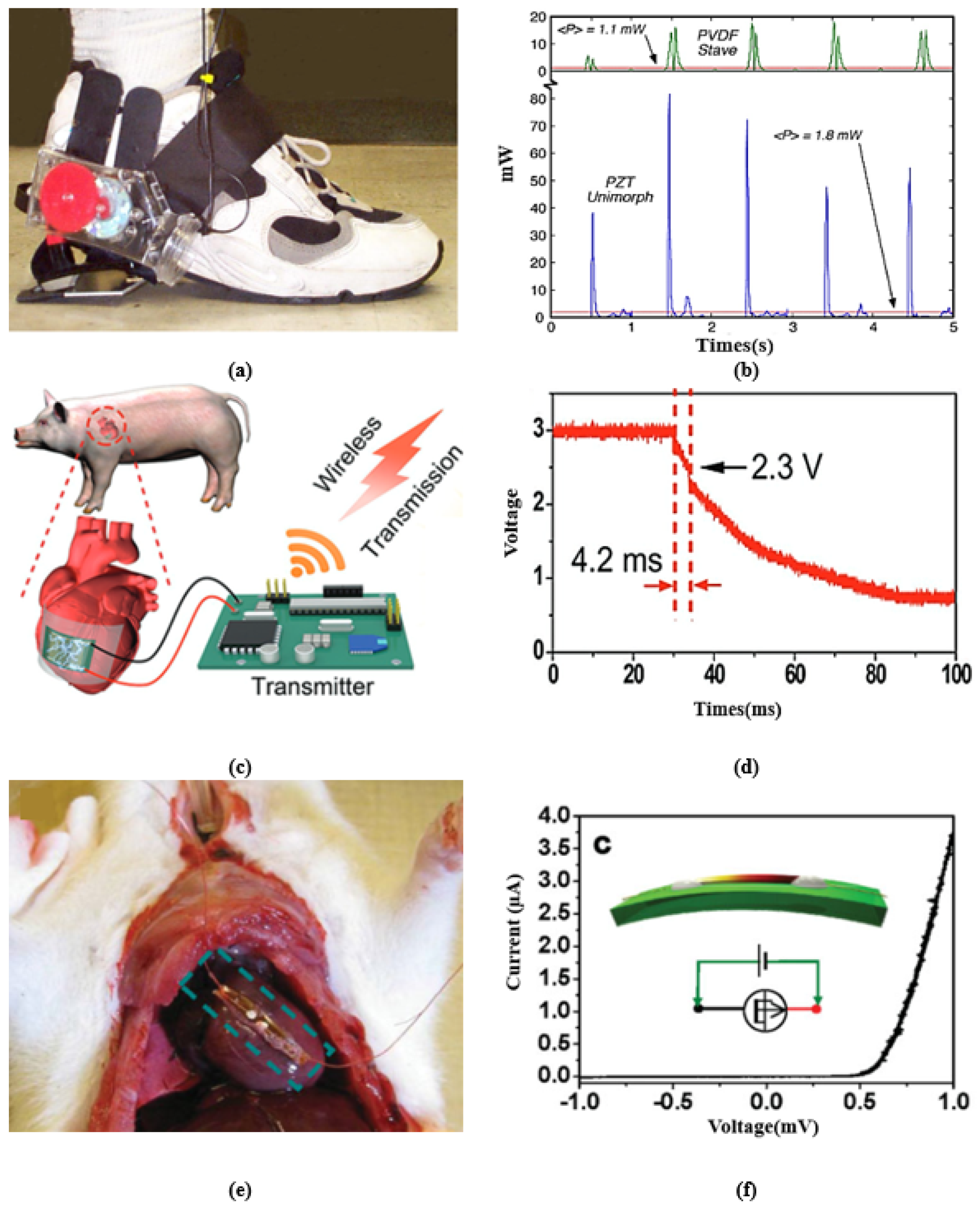
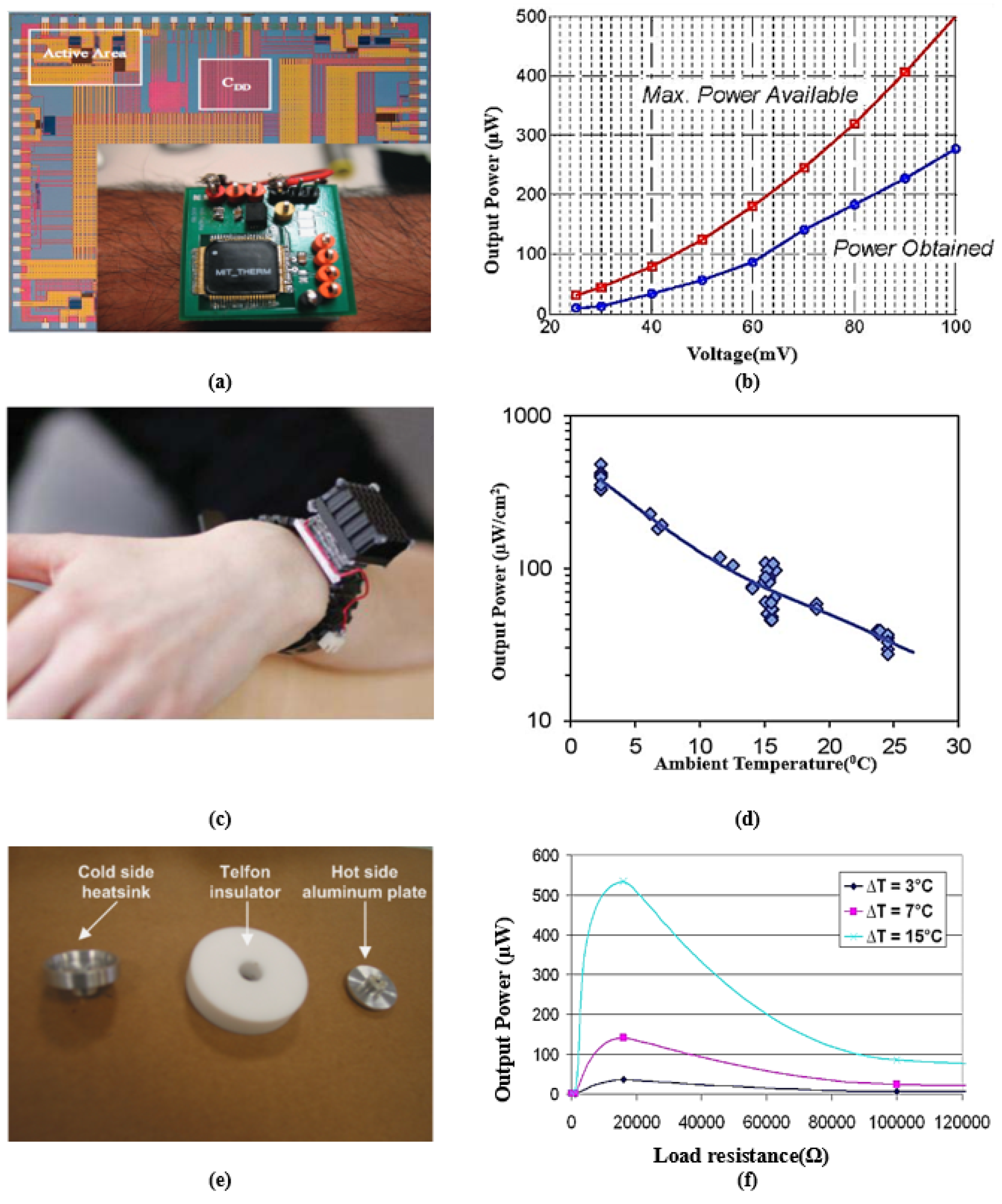
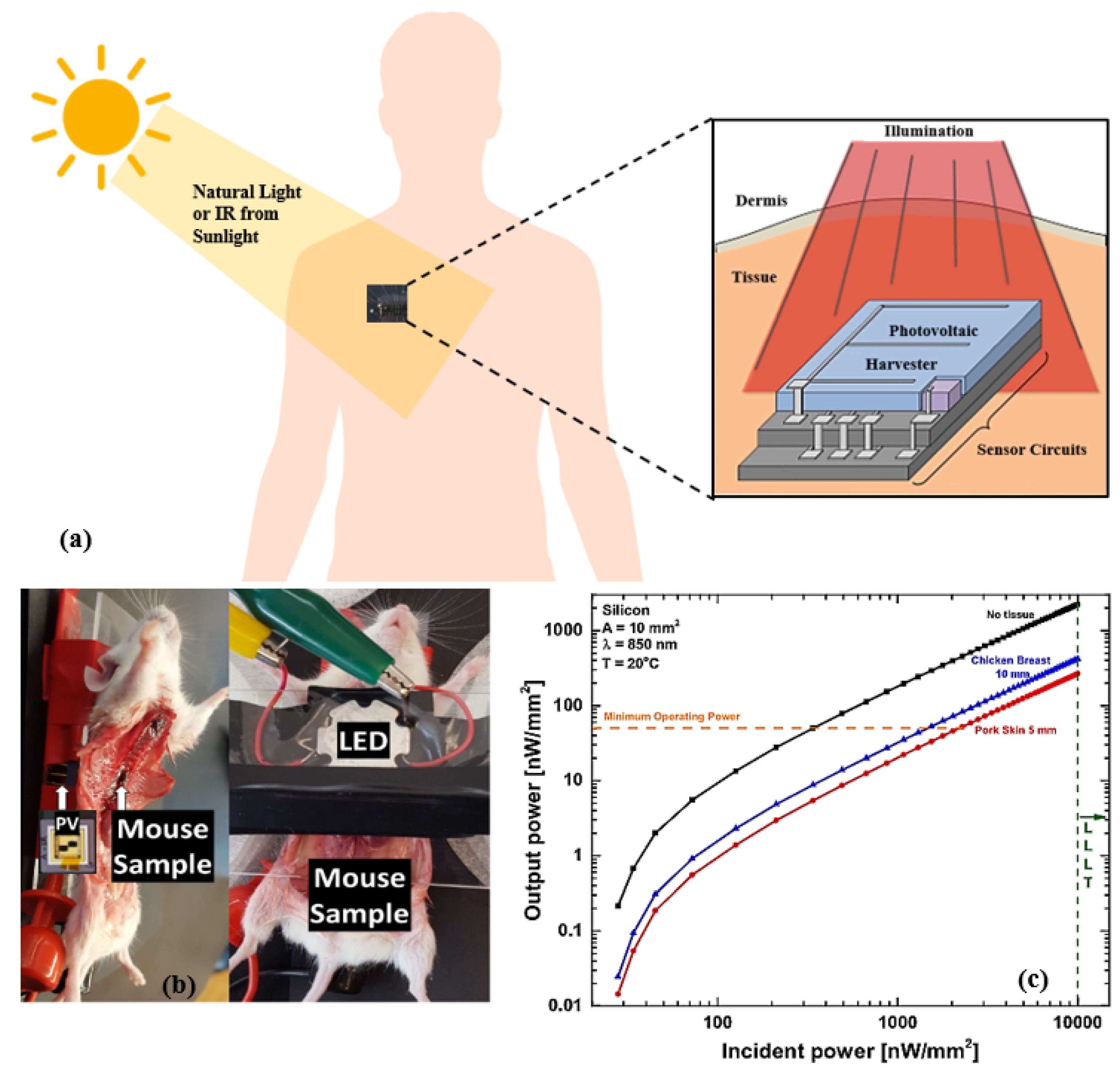


|
Energy Harvesting Techniques | Energy Source | Output Power (µW) | a Output Power Density (µW/mm) | b Size (mm) |
|---|---|---|---|---|
| Piezoelectric | Hand motion [99] | 15.2 | 3.8 W/mm | 4 cm |
| Human walking [195] | 2000 | 0.25 W/mm | mm | |
| Finger tapping [196] | – | 140 nm m | ||
| Porcine heart motion [95] | 0.73 | 100 mm | ||
| Elbow bending [96] | 0.167 | 24.276 | ||
| Human walking [197] | 30.55 | – | – | |
| Human knee [94] | 4800 | 0.8 | 2000 | |
| Electrostatic | Knee joint motion [198] | 78 | 125 | |
| Heartbeat vibration [199] | 10 | 0.074 | – | |
| Chest motion [119] | 1840 | 125 | ||
| Waist motion [200] | 40 | 0.044 | 900 mm | |
| Electromagnetic | Blood flow and pressure [131] | 3400 | 1.076 | |
| Finger rubbing [201] | 0.32 | 36 | ||
| Knee [202] | 5.614 | |||
| Foot [203] | 0.086 | |||
| Heart muscle [86] | 16.7 | 0.1429 | 116.86 mm | |
| Thoracic diaphragm [129] | 1100 | 0.069 | ||
| Thermoelectric | Human wrist [204] | 100 | 0.20 W/mm | mm |
| Human body [205] | 2.1 | W/mm | mm | |
| Radial artery [206] | 100 | W/mm | 175.2 | |
| Triboelectric | Handwriting [207] | – | 1.8 W/mm | cm |
| Human vibration [208] | – | 0.7261 W/mm | – | |
| Human body [209] | – | 3.3 W/mm | – | |
| Cardiac motion [210] | 5 | – | cm | |
| Body motion [211] | – | W/mm | cm |
|
Energy Harvesting Techniques | IWM Device/ Placement/ Application | Output Power (mW) | Output Power Density (mW/cm) | Size/ Diameter (mm) |
|---|---|---|---|---|
| Infrared | Pacemaker [182] | 4 | 22 | 5 × 4 × 0.7 cm |
| Porcine skin and fat [190] | 0.06 | – | 390 × 410 × 1.5 m | |
| Skin-attachable patch [191] | 0.0082 | 0.0741 | 11.1 mm | |
| Radio Frequency | WMDs [252] | – | – | 25–100 m distance |
| Solar | Outdoor/ Cardiac [188] | – | 4.94 | – |
| WMDs [253] | 16 (Outdoor) 0.21 (Indoor) | – | – | |
| Cochlear [254] | – | 9.35 | – | |
| Retina [255] | – | 44.12 | – | |
| Retina [256] | – | 3.84 | – | |
| Ultrasonic | Skin [237] | – | 0.00375 | |
| Brain [238] | 0.28 | 1100 | 13.4 × 9.6 × 2.1 mm | |
| Nerve [240] | 13.13 | – | – | |
| Inductive | Retina [257] | 98.8 | 31.49 | 20 |
| Pacemaker [258] | 0.040 | 0.0905 | 7.5 | |
| Retina [259] | 50 | 17.63 | 19 | |
| Heart [260] | – | 78–780 | 70 | |
| Nerve [261] | 105–127 | 33.4–40.4 | 20 |
|
MPPT Methods | Energy Harvesting Techniques | Power Consumption (µW) |
|---|---|---|
| FOCV | Piezoelectric [278] | 5.16 |
| Piezoelectric [280] | 10 | |
| Electromagnetic [281] | 0.15 | |
| Thermoelectric + photovoltaic [282] | 0.3 | |
| Glucose biofuel [283] | 0.5 | |
| P&O | Piezoelectric [274] | 408 |
| Electromagnetic [275] | 0.75 | |
| Photovoltaic [276] | 4.6 | |
| Thermoelectric [284] | 2.1 | |
| Thermoelectric + photovoltaic [285] | 3.48 |
| Energy Harvesting Techniques | Biochemical | Piezoelectric | Electrostatic | Electromagnetic | Thermoelectric | Triboelectric |
| Potential Applications | Biosensors; IMDs | Orthopedic implants; pacemaker; pressure sensors; accelerometers; pulse sensor | Pacemaker; watch; neural stimulator | Pacemaker; WMDs | Pulse oximeter; Pacemaker; hearing aid; ECG; EMG; EEG | Textiles; touchpad; glucose biosensor |
| Energy Harvesting Techniques | Radio Frequency | Solar | Ultrasonic | Inductive |
| Potential Applications | Wireless optogenetics; WMDs | Pacemaker; watch; neural stimulator; WMDs | IMDs; cell stimulants; drug delivery acoustic sensors; catalysis | Cardioverter– defibrillator; neuro-stimulants; cochlear and retinal IMDs |
| Energy Harvesting Techniques | Typical Output Power Density (mW/cm) | Features | Limitations | Potential Applications |
|---|---|---|---|---|
| Biochemical | –0.194 | Uses glucose, oxygen, and biofluids, which are clean and renewable; abundant amount of biofuel; mostly biocompatible; reasonable energy density | Unstable output power; limited life-time of enzymes; low power density; possibility of biofouling | Self-powered biosensors |
| Piezoelectric | – mW/cm | Compact size; high output voltage; high sensitivity; high power density; simple excitation; fabrication process is well-known; microscale and nanoscale fabrication possible; easy tuning to specific frequency | High impedance and low current at output; minimal efficiency at low frequency; some materials are poisonous; frequency-dependent; complex fabrication; expensive; complex MPPT | Orthopedic implants; pacemaker; pressure sensors; inertial sensors; pulse sensor |
| Electrostatic | – | Compatible to microscale fabrication; high energy density; low frequency; linear change of output with frequency; adjustable coupling coefficient | Separate voltage source needed; parasitic elements and leakage current decreases efficiency; complicated MPPT; low energy density; high output impedance | Pacemaker; wrist watch; neural stimulator |
| Electromagnetic | –5.614 | High output current; high efficiency; high power density; robust; simple MPPT | Low efficiency at low frequency; efficiency decreases with miniaturization; source of EMI; complicated MEMS integration; low output voltage | Cardiac pacemaker; wearable medical devices |
| Thermoelectric | 0.084–0.571 | Cost effective; low maintenance; lightweight; high reliability and accessibility; long life; frequency independent; simplified MPPT; scalability | Low power density; low energy conversion efficiency; harvested power is unpredictable; requires start-up circuit; need boost converter; expensive | Pulse oximeter; pacemaker; hearing aid; ECG; EMG; EEG |
| Triboelectric | 0.0022–50 | High output power density; flexible; easy fabrication; scalable; cost effective; wide variety of materials; frequency independent; compatible in microscale; | Need high voltage insulation; complex fabrication process; high erosion of material; limited lifetime; high output impedance; complicated interface circuitry | Wearable textiles; self-powered touchpad devices; glucose biosensor |
| Energy Harvesting Techniques | Typical Output Power Density (mW/cm) | Features | Limitations | Potential Applications |
|---|---|---|---|---|
| Radio frequency | 0.45–84 nW/cm | Availability of RF sources; wide variety of operating frequencies | Distance dependent output; low output power; unpredictable; low efficiency | wireless optogenetics |
| Solar | 0.059–6.7 | High power density; cost effective; easy fabrication; low maintenance; high reliability and availability; easy scalability; mature technology | Not continuous; dependence on weather and lighting conditions; location dependent; low efficiency; expensive initial setup; efficiency degrades with miniaturization | Cardiac pacemaker; watch; neural stimulator; wearable medical sensors |
| Ultrasonic | 1.2–290 | High efficiency for small IMDs; increased penetration depth; compact design; minimal tissue heating | Low power harvesting; poor data transmission; low efficiency in large IMDs; requires propagation medium | Implantable medical devices |
| Inductive | 0.0764–780 | Higher data rate; batteryless operation; bidirectional communication; continuous supply; efficient in small distance; energy transfer through tissue; high power density | Tissue absorption; potential source of EMI; complicated microscale fabrication; require precise alignment of coils | Implantable medical devices |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shuvo, M.M.H.; Titirsha, T.; Amin, N.; Islam, S.K. Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare. Energies 2022, 15, 7495. https://doi.org/10.3390/en15207495
Shuvo MMH, Titirsha T, Amin N, Islam SK. Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare. Energies. 2022; 15(20):7495. https://doi.org/10.3390/en15207495
Chicago/Turabian StyleShuvo, Md Maruf Hossain, Twisha Titirsha, Nazmul Amin, and Syed Kamrul Islam. 2022. "Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare" Energies 15, no. 20: 7495. https://doi.org/10.3390/en15207495
APA StyleShuvo, M. M. H., Titirsha, T., Amin, N., & Islam, S. K. (2022). Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare. Energies, 15(20), 7495. https://doi.org/10.3390/en15207495







