Nano/Microrobots Line Up for Gastrointestinal Tract Diseases: Targeted Delivery, Therapy, and Prevention
Abstract
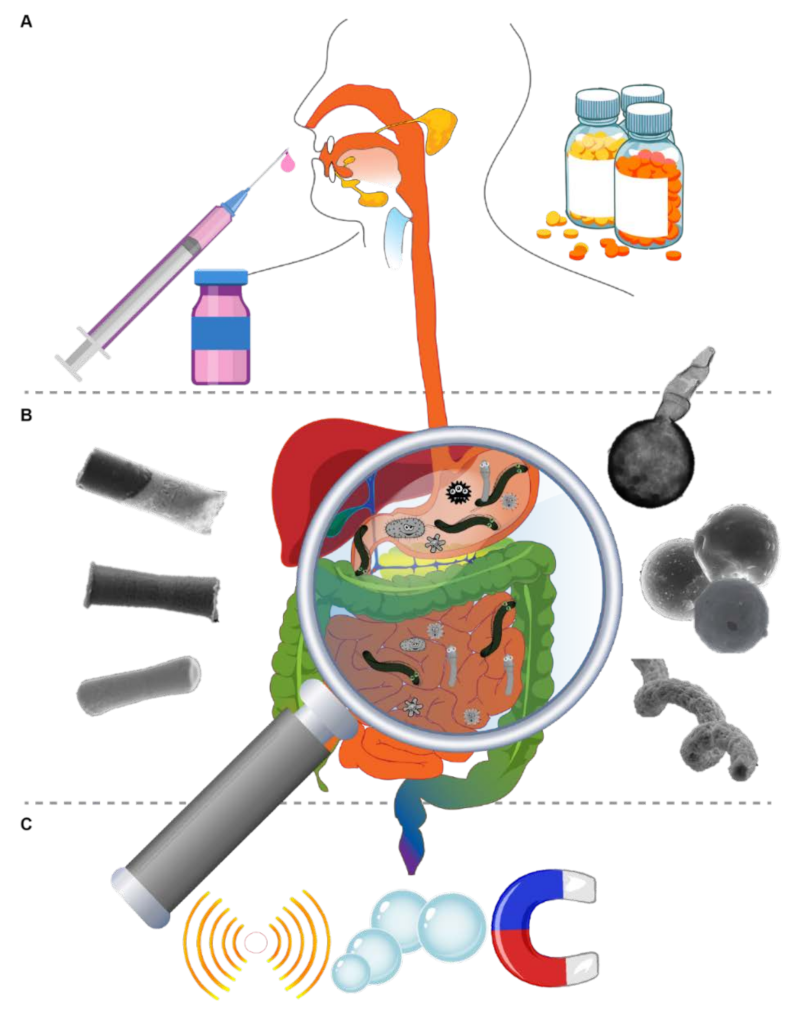
:1. Introduction
2. Nano/Microrobots (NMR) in Action for Stomach Diseases
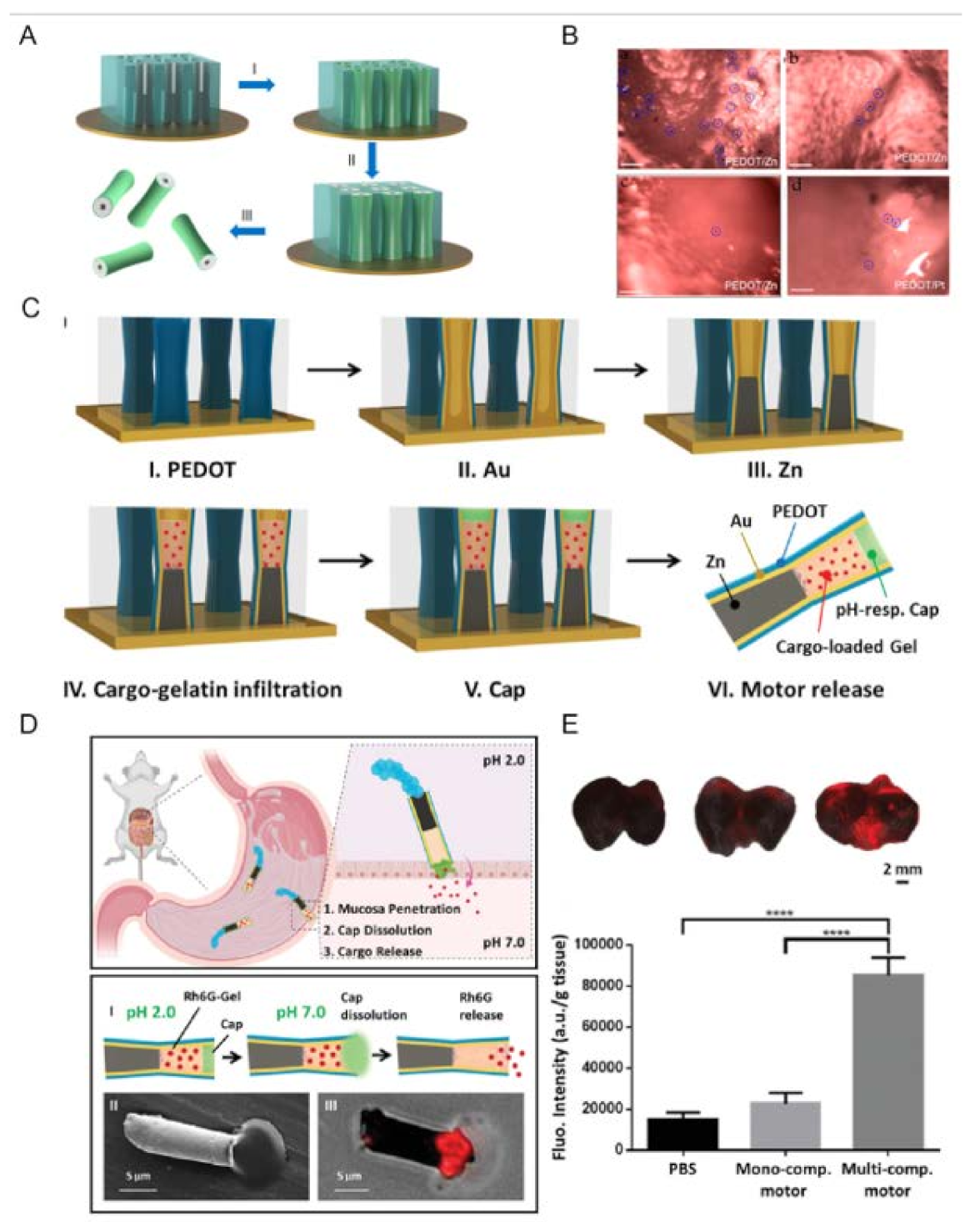
2.1. Zinc (Zn)-Based NMRs
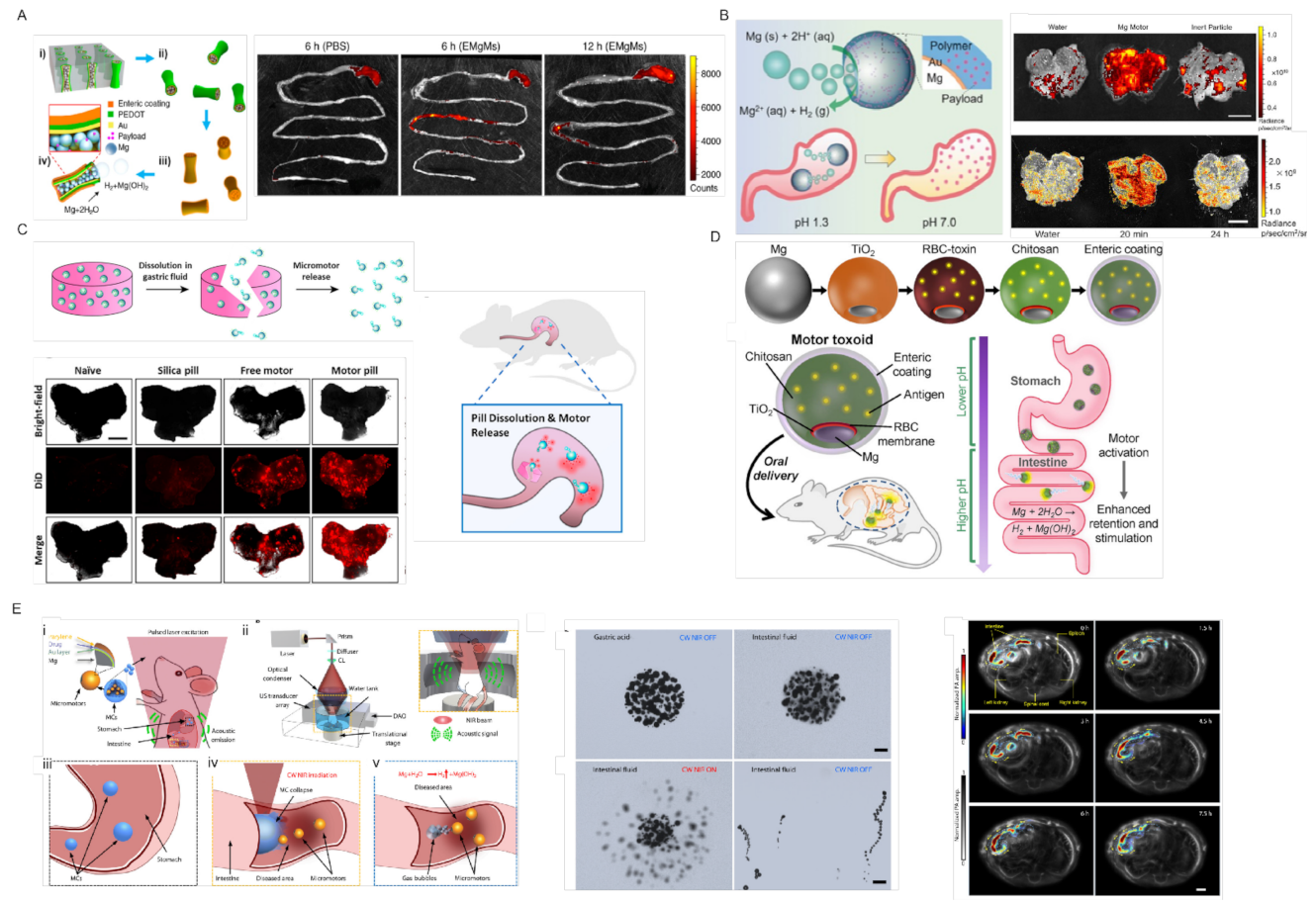
2.2. Magnesium (Mg)-Based NMRs
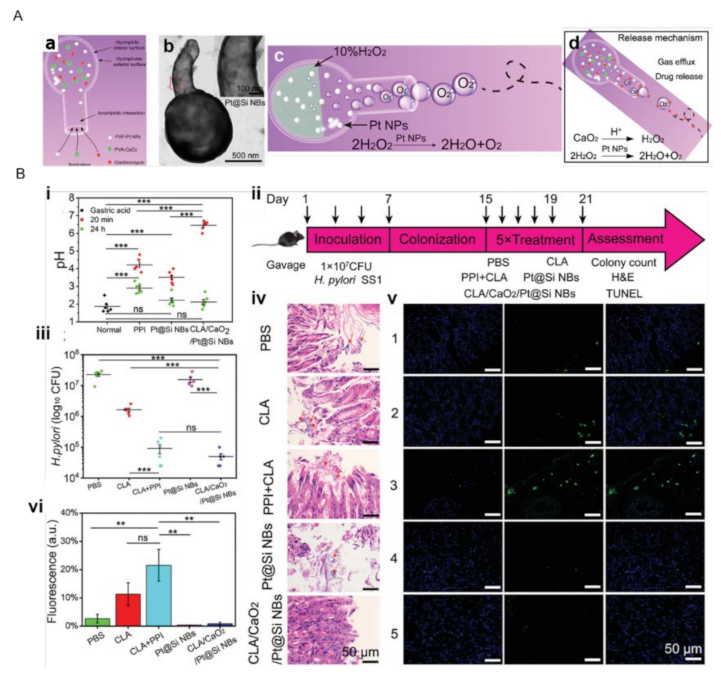
2.3. CaO2/Pt NPs Powered NMRs
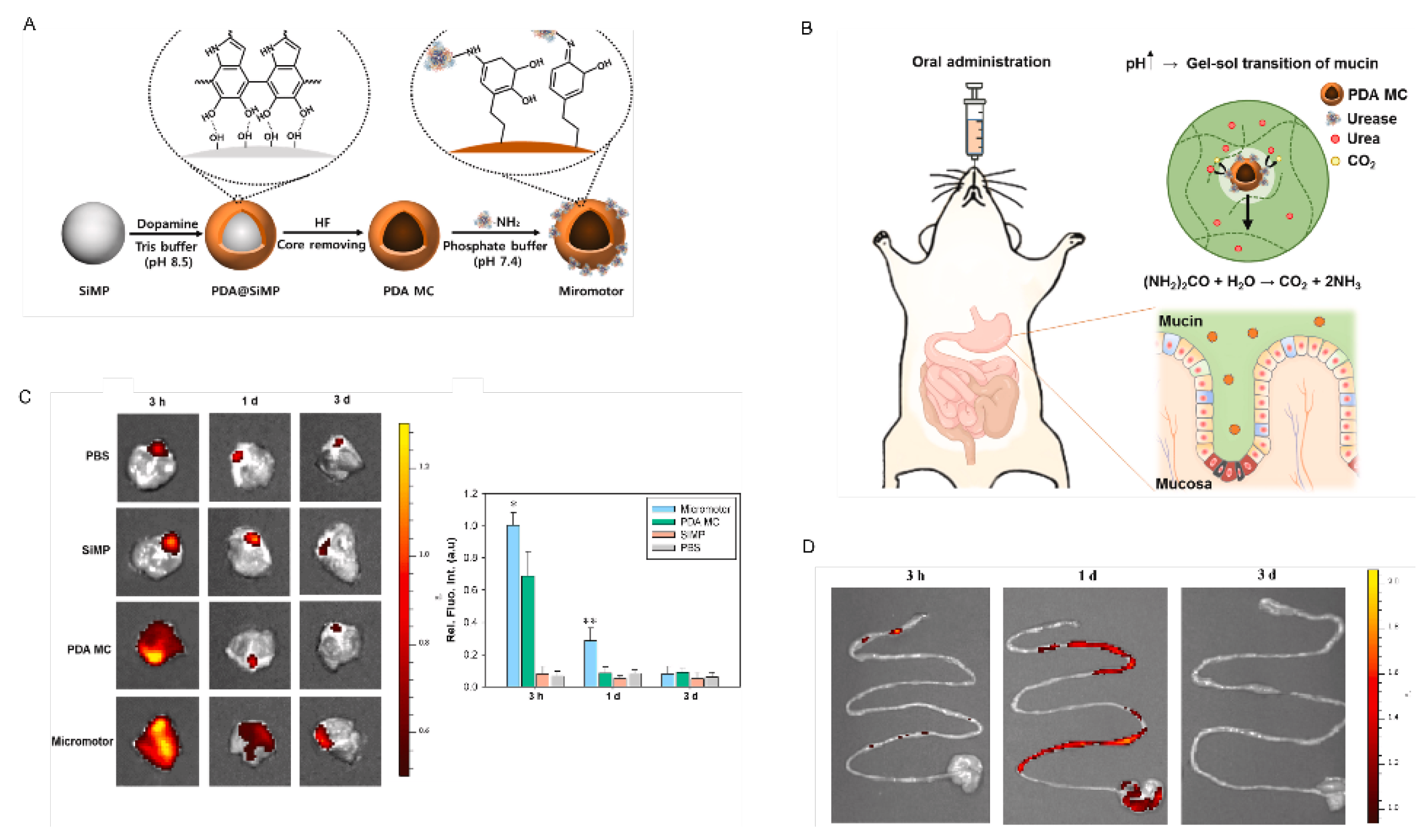
2.4. Enzyme-Powered NMRs
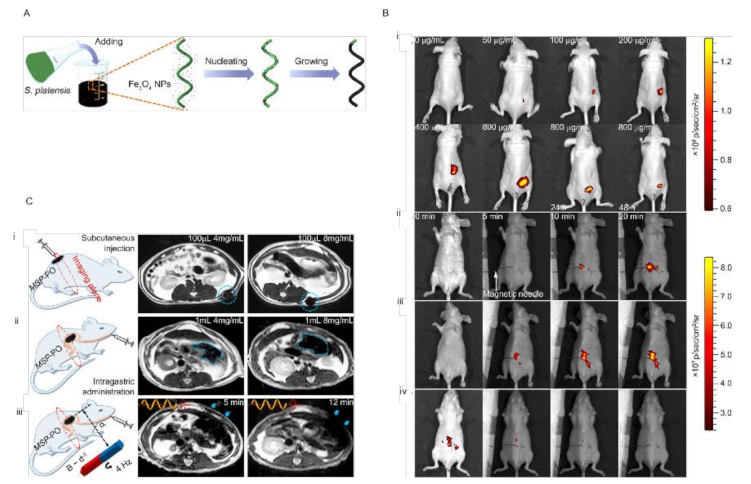
2.5. Biohybrid NMRs
3. Discussion and Conclusions
3.1. Simple Fabrication and Easy Surface Modification
3.2. Biocompatibility and Biodegradability
3.3. Multifunctionality
3.4. Propulsion
3.5. Lifetime
3.6. Imaging
3.7. Size
3.8. Immunogenicity
4. Prospects and Future Direction
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fleischer, R. Fantastic Voyage; 20th Century Fox: Los Angeles, CA, USA, 1966. [Google Scholar]
- Feynman, R.P. There’s plenty of room at the bottom. Resonance 2011, 16, 890. [Google Scholar] [CrossRef] [Green Version]
- Paxton, W.F.; Kistler, K.C.; Olmeda, C.C.; Sen, A.; St. Angelo, S.K.; Cao, Y.; Mallouk, T.E.; Lammert, P.E.; Crespi, V.H. Catalytic Nanomotors: Autonomous Movement of Striped Nanorods. J. Am. Chem. Soc. 2004, 126, 13424–13431. [Google Scholar] [CrossRef]
- Arqué, X.; Romero-Rivera, A.; Feixas, F.; Patiño, T.; Osuna, S.; Sánchez, S. Intrinsic enzymatic properties modulate the self-propulsion of micromotors. Nat. Commun. 2019, 10, 2826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dey, K.K.; Zhao, X.; Tansi, B.M.; Méndez-Ortiz, W.J.; Córdova-Figueroa, U.M.; Golestanian, R.; Sen, A. Micromotors Powered by Enzyme Catalysis. Nano Lett. 2015, 15, 8311–8315. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Karshalev, E.; Guan, J.; Wang, J. Magnesium-Based Micromotors: Water-Powered Propulsion, Multifunctionality, and Biomedical and Environmental Applications. Small 2018, 14, 1704252. [Google Scholar] [CrossRef]
- Koleoso, M.; Feng, X.; Xue, Y.; Li, Q.; Munshi, T.; Chen, X. Micro/nanoscale magnetic robots for biomedical applications. Mater. Today Bio 2020, 8, 100085. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.G.; Al Harraq, A.; Bishop, K.J.M.; Bharti, B. Fabrication and Electric Field-Driven Active Propulsion of Patchy Microellipsoids. J. Phys. Chem. B 2021, 125, 4232–4240. [Google Scholar] [CrossRef]
- Lu, X.; Shen, H.; Zhao, K.; Wang, Z.; Peng, H.; Liu, W. Micro-/Nanomachines Driven by Ultrasonic Power Sources. Chem. Asian J. 2019, 14, 2406–2416. [Google Scholar] [CrossRef]
- Bunea, A.-I.; Martella, D.; Nocentini, S.; Parmeggiani, C.; Taboryski, R.; Wiersma, D.S. Light-Powered Microrobots: Challenges and Opportunities for Hard and Soft Responsive Microswimmers. Adv. Intell. Syst. 2021, 3, 2000256. [Google Scholar] [CrossRef]
- Wan, M.; Li, T.; Chen, H.; Mao, C.; Shen, J. Biosafety, Functionalities, and Applications of Biomedical Micro/nanomotors. Angew. Chem. Int. Ed. 2021, 60, 13158–13176. [Google Scholar] [CrossRef]
- Li, M.; Xi, N.; Wang, Y.; Liu, L. Progress in Nanorobotics for Advancing Biomedicine. IEEE Trans. Biomed. Eng. 2021, 68, 130–147. [Google Scholar] [CrossRef] [PubMed]
- Parmar, J.; Vilela, D.; Villa, K.; Wang, J.; Sánchez, S. Micro- and Nanomotors as Active Environmental Microcleaners and Sensors. J. Am. Chem. Soc. 2018, 140, 9317–9331. [Google Scholar] [CrossRef]
- Singh, V.V.; Wang, J. Nano/micromotors for security/defense applications. A review. Nanoscale 2015, 7, 19377–19389. [Google Scholar] [CrossRef]
- Molinero-Fernández, Á.; Jodra, A.; Moreno-Guzmán, M.; López, M.Á.; Escarpa, A. Magnetic Reduced Graphene Oxide/Nickel/Platinum Nanoparticles Micromotors for Mycotoxin Analysis. Chem. Eur. J. 2018, 24, 7172–7176. [Google Scholar] [CrossRef] [PubMed]
- Bulatov, E.; Khaiboullina, S.; dos Reis, H.J.; Palotás, A.; Venkataraman, K.; Vijayalakshmi, M.; Rizvanov, A. Ubiquitin-Proteasome System: Promising Therapeutic Targets in Autoimmune and Neurodegenerative Diseases. BioNanoScience 2016, 6, 341–344. [Google Scholar] [CrossRef]
- Lin, R.; Yu, W.; Chen, X.; Gao, H. Self-Propelled Micro/Nanomotors for Tumor Targeting Delivery and Therapy. Adv. Healthc. Mater. 2021, 10, 2001212. [Google Scholar] [CrossRef]
- Sun, M.; Fan, X.; Meng, X.; Song, J.; Chen, W.; Sun, L.; Xie, H. Magnetic biohybrid micromotors with high maneuverability for efficient drug loading and targeted drug delivery. Nanoscale 2019, 11, 18382–18392. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Clergeaud, G.; Andresen, T.L.; Boisen, A. Micromotors for drug delivery in vivo: The road ahead. Adv. Drug Deliv. Rev. 2019, 138, 41–55. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Medina-Sánchez, M.; Magdanz, V.; Schwarz, L.; Hebenstreit, F.; Schmidt, O.G. Sperm-Hybrid Micromotor for Targeted Drug Delivery. ACS Nano 2018, 12, 327–337. [Google Scholar] [CrossRef] [Green Version]
- Luo, M.; Feng, Y.; Wang, T.; Guan, J. Micro-/Nanorobots at Work in Active Drug Delivery. Adv. Funct. Mater. 2018, 28, 1706100. [Google Scholar] [CrossRef]
- Yim, S.; Gultepe, E.; Gracias, D.H.; Sitti, M. Biopsy using a magnetic capsule endoscope carrying, releasing, and retrieving untethered microgrippers. IEEE Trans. Biomed. Eng. 2014, 61, 513–521. [Google Scholar]
- Gao, C.; Wang, Y.; Ye, Z.; Lin, Z.; Ma, X.; He, Q. Biomedical Micro-/Nanomotors: From Overcoming Biological Barriers to In Vivo Imaging. Adv. Mater. 2021, 33, 2000512. [Google Scholar] [CrossRef] [PubMed]
- Van Moolenbroek, G.T.; Patiño, T.; Llop, J.; Sánchez, S. Engineering Intelligent Nanosystems for Enhanced Medical Imaging. Adv. Intell. Syst. 2020, 2, 2000087. [Google Scholar] [CrossRef]
- Pané, S.; Puigmartí-Luis, J.; Bergeles, C.; Chen, X.-Z.; Pellicer, E.; Sort, J.; Počepcová, V.; Ferreira, A.; Nelson, B.J. Imaging Technologies for Biomedical Micro- and Nanoswimmers. Adv. Mater. Technol. 2019, 4, 1800575. [Google Scholar] [CrossRef] [Green Version]
- Martella, D.; Nocentini, S.; Parmeggiani, C.; Wiersma, D.S. Photonic artificial muscles: From micro robots to tissue engineering. Faraday Discuss. 2020, 223, 216–232. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.-M.J.; Fang, R.H.; Copp, J.; Luk, B.T.; Zhang, L. A biomimetic nanosponge that absorbs pore-forming toxins. Nat. Nanotechnol. 2013, 8, 336–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Z.; Li, T.; Gao, W.; Xu, T.; Jurado-Sánchez, B.; Li, J.; Gao, W.; He, Q.; Zhang, L.; Wang, J. Cell-Membrane-Coated Synthetic Nanomotors for Effective Biodetoxification. Adv. Funct. Mater. 2015, 25, 3881–3887. [Google Scholar] [CrossRef] [Green Version]
- Pedram, A.; Pishkenari, H.N. Smart Micro/Nano-robotic Systems for Gene Delivery. Curr. Gene Ther. 2017, 17, 73–79. [Google Scholar] [CrossRef]
- Hu, M.; Ge, X.; Chen, X.; Mao, W.; Qian, X.; Yuan, W.-E. Micro/Nanorobot: A Promising Targeted Drug Delivery System. Pharmaceutics 2020, 12, 665. [Google Scholar] [CrossRef]
- Hwang, G.; Paula, A.J.; Hunter, E.E.; Liu, Y.; Babeer, A.; Karabucak, B.; Stebe, K.; Kumar, V.; Steager, E.; Koo, H. Catalytic antimicrobial robots for biofilm eradication. Sci. Robot. 2019, 4, eaaw2388. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Hu, E.; Xie, R.; Yu, K.; Lu, F.; Bao, R.; Wang, C.; Lan, G.; Dai, F. Magnetically Guided Nanoworms for Precise Delivery to Enhance In Situ Production of Nitric Oxide to Combat Focal Bacterial Infection In Vivo. ACS Appl. Mater. Interfaces 2021, 13, 22225–22239. [Google Scholar] [CrossRef]
- Debraekeleer, A.; Remaut, H. Future perspective for potential Helicobacter pylori eradication therapies. Future Microbiol. 2018, 13, 671–687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chi, Q.; Wang, Z.; Tian, F.; You, J.A.; Xu, S. A Review of Fast Bubble-Driven Micromotors Powered by Biocompatible Fuel: Low-Concentration Fuel, Bioactive Fluid and Enzyme. Micromachines 2018, 9, 537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Dong, R.; Wang, C.; Xu, S.; Chen, D.; Liang, Y.; Ren, B.; Gao, W.; Cai, Y. Glucose-Fueled Micromotors with Highly Efficient Visible-Light Photocatalytic Propulsion. ACS Appl. Mater. Interfaces 2019, 11, 6201–6207. [Google Scholar] [CrossRef]
- Hortelao, A.C.; Simó, C.; Guix, M.; Guallar-Garrido, S.; Julián, E.; Vilela, D.; Rejc, L.; Ramos-Cabrer, P.; Cossío, U.; Gómez-Vallejo, V.; et al. Swarming behavior and in vivo monitoring of enzymatic nanomotors within the bladder. Sci. Robot. 2021, 6, eabd2823. [Google Scholar] [CrossRef]
- Xu, C.; Wang, S.; Wang, H.; Liu, K.; Zhang, S.; Chen, B.; Liu, H.; Tong, F.; Peng, F.; Tu, Y.; et al. Magnesium-Based Micromotors as Hydrogen Generators for Precise Rheumatoid Arthritis Therapy. Nano Lett. 2021, 21, 1982–1991. [Google Scholar] [CrossRef]
- Guo, M.; Wang, S.; Guo, Q.; Hou, B.; Yue, T.; Ming, D.; Zheng, B. NIR-Responsive Spatiotemporally Controlled Cyanobacteria Micro-Nanodevice for Intensity-Modulated Chemotherapeutics in Rheumatoid Arthritis. ACS Appl. Mater. Interfaces 2021, 13, 18423–18431. [Google Scholar] [CrossRef]
- Wang, S.; Liu, K.; Zhou, Q.; Xu, C.; Gao, J.; Wang, Z.; Wang, F.; Chen, B.; Ye, Y.; Ou, J.; et al. Hydrogen-Powered Microswimmers for Precise and Active Hydrogen Therapy Towards Acute Ischemic Stroke. Adv. Funct. Mater. 2021, 31, 2009475. [Google Scholar] [CrossRef]
- Blaser, M.J. Helicobacter pylori and the pathogenesis of gastroduodenal inflammation. J. Infect. Dis. 1990, 161, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Rotimt, O.; Nduduba, D.A.; Otegbeye, F.M. Helicobacter pylori and the pathogenesis of gastroduodenal disease: Implications for the management of peptic ulcer disease. Niger. Postgrad. Med. J. 2005, 12, 289–298. [Google Scholar] [PubMed]
- Asaka, M.; Sugiyama, T.; Kato, M.; Satoh, K.; Kuwayama, H.; Fukuda, Y.; Fujioka, T.; Takemoto, T.; Kimura, K.; Shimoyama, T.; et al. A multicenter, double-blind study on triple therapy with lansoprazole, amoxicillin and clarithromycin for eradication of Helicobacter pylori in Japanese peptic ulcer patients. Helicobacter 2001, 6, 254–261. [Google Scholar] [CrossRef]
- Tsay, F.-W.; Hsu, P.-I.H. pylori infection and extra-gastroduodenal diseases. J. Biomed. Sci. 2018, 25, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sugiyama, A.; Maruta, F.; Ikeno, T.; Ishida, K.; Kawasaki, S.; Katsuyama, T.; Shimizu, N.; Tatematsu, M. Helicobacter pylori infection enhances N-methyl-N-nitrosourea-induced stomach carcinogenesis in the Mongolian gerbil. Cancer Res. 1998, 58, 2067–2069. [Google Scholar] [PubMed]
- Kalisperati, P.; Spanou, E.; Pateras, I.S.; Korkolopoulou, P.; Varvarigou, A.; Karavokyros, I.; Gorgoulis, V.G.; Vlachoyiannopoulos, P.G.; Sougioultzis, S. Inflammation, DNA Damage, Helicobacter pylori and Gastric Tumorigenesis. Front. Genet. 2017, 8, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaynes, M.; Kumar, A.B. The risks of long-term use of proton pump inhibitors: A critical review. Ther. Adv. Drug Saf. 2018, 10, 2042098618809927. [Google Scholar] [CrossRef] [PubMed]
- De Ávila, B.E.-F.; Angsantikul, P.; Li, J.; Angel Lopez-Ramirez, M.; Ramírez-Herrera, D.E.; Thamphiwatana, S.; Chen, C.; Delezuk, J.; Samakapiruk, R.; Ramez, V.; et al. Micromotor-enabled active drug delivery for in vivo treatment of stomach infection. Nat. Commun. 2017, 8, 272. [Google Scholar] [CrossRef] [Green Version]
- Gao, W.; Dong, R.; Thamphiwatana, S.; Li, J.; Gao, W.; Zhang, L.; Wang, J. Artificial Micromotors in the Mouse’s Stomach: A Step toward in Vivo Use of Synthetic Motors. ACS Nano 2015, 9, 117–123. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Thamphiwatana, S.; Liu, W.; Esteban-Fernández de Ávila, B.; Angsantikul, P.; Sandraz, E.; Wang, J.; Xu, T.; Soto, F.; Ramez, V.; et al. Enteric Micromotor Can Selectively Position and Spontaneously Propel in the Gastrointestinal Tract. ACS Nano 2016, 10, 9536–9542. [Google Scholar] [CrossRef]
- Li, J.; Angsantikul, P.; Liu, W.; Esteban-Fernández de Ávila, B.; Thamphiwatana, S.; Xu, M.; Sandraz, E.; Wang, X.; Delezuk, J.; Gao, W.; et al. Micromotors Spontaneously Neutralize Gastric Acid for pH-Responsive Payload Release. Angew. Chem. Int. Ed. 2017, 56, 2156–2161. [Google Scholar] [CrossRef] [Green Version]
- Karshalev, E.; Esteban-Fernández de Ávila, B.; Beltrán-Gastélum, M.; Angsantikul, P.; Tang, S.; Mundaca-Uribe, R.; Zhang, F.; Zhao, J.; Zhang, L.; Wang, J. Micromotor Pills as a Dynamic Oral Delivery Platform. ACS Nano 2018, 12, 8397–8405. [Google Scholar] [CrossRef]
- Esteban-Fernández de Ávila, B.; Lopez-Ramirez, M.A.; Mundaca-Uribe, R.; Wei, X.; Ramírez-Herrera, D.E.; Karshalev, E.; Nguyen, B.; Fang, R.H.; Zhang, L.; Wang, J. Multicompartment Tubular Micromotors Toward Enhanced Localized Active Delivery. Adv. Mater. 2020, 32, 2000091. [Google Scholar] [CrossRef]
- Yan, X.; Zhou, Q.; Vincent, M.; Deng, Y.; Yu, J.; Xu, J.; Xu, T.; Tang, T.; Bian, L.; Wang, Y.-X.J.; et al. Multifunctional biohybrid magnetite microrobots for imaging-guided therapy. Sci. Robot. 2017, 2, eaaq1155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, Y.; Song, Z.; Deng, G.; Jiang, K.; Wang, H.; Zhang, X.; Han, H. Gastric Acid Powered Nanomotors Release Antibiotics for In Vivo Treatment of Helicobacter pylori Infection. Small 2021, 17, 2006877. [Google Scholar] [CrossRef]
- Choi, H.; Jeong, S.H.; Kim, T.Y.; Yi, J.; Hahn, S.K. Bioinspired urease-powered micromotor as an active oral drug delivery carrier in stomach. Bioact. Mater. 2021, 9, 54–62. [Google Scholar] [CrossRef]
- Wei, X.; Beltrán-Gastélum, M.; Karshalev, E.; Esteban-Fernández de Ávila, B.; Zhou, J.; Ran, D.; Angsantikul, P.; Fang, R.H.; Wang, J.; Zhang, L. Biomimetic Micromotor Enables Active Delivery of Antigens for Oral Vaccination. Nano Lett. 2019, 19, 1914–1921. [Google Scholar] [CrossRef]
- Wu, Z.; Li, L.; Yang, Y.; Hu, P.; Li, Y.; Yang, S.-Y.; Wang, L.V.; Gao, W. A microrobotic system guided by photoacoustic computed tomography for targeted navigation in intestines in vivo. Sci. Robot. 2019, 4, eaax0613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, H.; Liu, X.; Wang, L.; Ma, X. Fundamentals and applications of enzyme powered micro/nano-motors. Bioact. Mater. 2021, 6, 1727–1749. [Google Scholar] [CrossRef]
- Patiño, T.; Arqué, X.; Mestre, R.; Palacios, L.; Sánchez, S. Fundamental Aspects of Enzyme-Powered Micro- and Nanoswimmers. Acc. Chem. Res. 2018, 51, 2662–2671. [Google Scholar] [CrossRef] [PubMed]
- Mathesh, M.; Sun, J.; Wilson, D.A. Enzyme catalysis powered micro/nanomotors for biomedical applications. J. Mater. Chem. B 2020, 8, 7319–7334. [Google Scholar] [CrossRef] [PubMed]
- Alapan, Y.; Yasa, O.; Yigit, B.; Yasa, I.C.; Erkoc, P.; Sitti, M. Microrobotics and Microorganisms: Biohybrid Autonomous Cellular Robots. Annu. Rev. Control Robot. Auton. Syst. 2019, 2, 205–230. [Google Scholar] [CrossRef]
- Lin, Z.; Jiang, T.; Shang, J. The emerging technology of biohybrid micro-robots: A review. Bio-Des. Manuf. 2021. Available online: https://link.springer.com/article/10.1007/s42242-021-00135-6#citeas (accessed on 16 May 2021). [CrossRef]
- Zhong, D.; Du, Z.; Zhou, M. Algae: A natural active material for biomedical applications. VIEW 2021. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/VIW.20200189 (accessed on 23 February 2021).
- Wang, H.-D.; Li, X.-C.; Lee, D.-J.; Chang, J.-S. Potential biomedical applications of marine algae. Bioresour. Technol. 2017, 244, 1407–1415. [Google Scholar] [CrossRef]
- Yan, X.; Zhou, Q.; Yu, J.; Xu, T.; Deng, Y.; Tang, T.; Feng, Q.; Bian, L.; Zhang, Y.; Ferreira, A.; et al. Magnetite Nanostructured Porous Hollow Helical Microswimmers for Targeted Delivery. Adv. Funct. Mater. 2015, 25, 5333–5342. [Google Scholar] [CrossRef]
- Wang, B.; Kostarelos, K.; Nelson, B.J.; Zhang, L. Trends in Micro-/Nanorobotics: Materials Development, Actuation, Localization, and System Integration for Biomedical Applications. Adv. Mater. 2021, 33, 2002047. [Google Scholar] [CrossRef]
- Sun, J.; Tan, H.; Lan, S.; Peng, F.; Tu, Y. Progress on the fabrication strategies of self-propelled micro/nanomotors. JCIS Open 2021, 2, 100011. [Google Scholar] [CrossRef]
- Liu, M.; Zhao, K. Engineering Active Micro and Nanomotors. Micromachines 2021, 12, 687. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, X.; Wang, Y.; Xu, D.; Liang, C.; Guo, J.; Ma, X. Biocompatibility of artificial micro/nanomotors for use in biomedicine. Nanoscale 2019, 11, 14099–14112. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, C. A Journey of Nanomotors for Targeted Cancer Therapy: Principles, Challenges, and a Critical Review of the State-of-the-Art. Adv. Healthc. Mater. 2021, 10, 2001236. [Google Scholar] [CrossRef] [PubMed]
- Soto, F.; Karshalev, E.; Zhang, F.; Esteban Fernandez de Avila, B.; Nourhani, A.; Wang, J. Smart Materials for Microrobots. Chem. Rev. 2021. Available online: https://pubs.acs.org/doi/10.1021/acs.chemrev.0c00999 (accessed on 1 February 2021). [CrossRef]
- Wang, B.; Zhang, Y.; Zhang, L. Recent progress on micro- and nano-robots: Towards in vivo tracking and localization. Quant. Imaging Med. Surg. 2018, 8, 461–479. [Google Scholar] [CrossRef]
- Wang, L.; Meng, Z.; Chen, Y.; Zheng, Y. Engineering Magnetic Micro/Nanorobots for Versatile Biomedical Applications. Adv. Intell. Syst. 2021, 3, 2000267. [Google Scholar] [CrossRef]
- Aziz, A.; Pane, S.; Iacovacci, V.; Koukourakis, N.; Czarske, J.; Menciassi, A.; Medina-Sánchez, M.; Schmidt, O.G. Medical Imaging of Microrobots: Toward In Vivo Applications. ACS Nano 2020, 14, 10865–10893. [Google Scholar] [CrossRef]
- Fu, D.; Wang, Z.; Tu, Y.; Peng, F. Interactions between Biomedical Micro-/Nano-Motors and the Immune Molecules, Immune Cells, and the Immune System: Challenges and Opportunities. Adv. Healthc. Mater. 2021, 10, 2001788. [Google Scholar] [CrossRef] [PubMed]
| NMR | Organ (In Vivo) | Propulsion Mode | Responsibility | Size | Velocity (µms−1) | Ref |
|---|---|---|---|---|---|---|
| PEDOT/Zn | Stomach | Acid powered (gastric juice) | cargo delivery | 5 µm | 60 | [48] |
| Enteric coating/ PEDOT/Au/Mg | Intestine | Intestinal fluid powered | Deliver the payload to a particular location to localize tissue penetration and retention. | 5 µm | 60 | [49] |
| pH-sensitive polymer/Au/Mg | Stomach | Acid powered (gastric juice) | The neutralization of gastric environment and cargo delivery | 20 ± 5 µm | 60 | [50] |
| Chit/PLGA/TiO2/Mg | Stomach | Acid powered (gastric juice) | Antibiotic delivery for H. pylori infection and neutralization of the acidic environment | 20 ± 5 µm | 120 | [47] |
| Mg/TiO2 | Stomach | Acid powered (gastric juice) | Targeted cargo delivery | 20 ± 5 µm | ~300 | [51] |
| pH-sensitive polymer/PEDOT/Au/Zn/ | Stomach | Acid powered (gastric juice) | Targeted cargo delivery | 5 µm | 70 | [52] |
| Spirulina platensis/Fe3O4 | intraperitoneal cavity | Magnetic field | Imaging | ~200 µm | - | [53] |
| Si Nanobottles | Stomach | Acid powered (gastric juice) | Antibiotic delivery for H. pylori infection and neutralization of the acidic environment | 576 ± 50 nm | - | [54] |
| PDA MC | Stomach | Urea powered | Penetration and retention in the stomach wall | 1 µm | 0.78–1.9 | [55] |
| External Source | Considerations |
|---|---|
| Light | UV light damages the function of the immune system and living organisms. UV and visible light have a poor penetration ability into human tissues. X-ray has an adverse effect on human health (hair loss, increased possibility of cancer and genetic mutation, infertility). NMRs operating with high-power lasers may generate overheat in biological tissues. Light-driven NMRs usually cannot move in high-ionic-strength environments. Light-driven NMRs usually require a chemical fuel to trigger their activity, such as H2O2, which is toxic and cannot be used for medical applications. |
| Ultrasound | The choice of material is limited. Heat generation. |
| Magnetic field | Construction of a large magnetic field with precise control. Costly. Biocompatible magnetic materials should be employed to fabricate NMRs. |
| Electrical field | Safety concern. Electrodes are required to reach the potential and need to be close to the NMRs, which implies difficulties. |
| Imaging Technique | Consideration |
|---|---|
| FI | This technique requires embedding a fluorescent dye in the fabrication of NMRs without any change in their fluorescent characteristics. Most fluorescent dyes are toxic, and only a few are allowed to be used in biosystems. Fluorescent imaging is generally based on visible light, which cannot penetrate deep tissues because of weak power. In addition, they have poor stability in acid, salt, alkali, and other media. Self-quenching is also another concern. |
| MRI | Size limitation is the main challenge in using this technique. Moreover, magnetic resonance imaging is a slow imaging method that limits its application as a tracker in vivo. |
| RI | Safety issues due to exposure to ionizing radiation (X-rays and radionuclides). |
| USI | However, USI is considered a promising imaging method for in vivo application of NMRs, and USI still faces some limitations such as errors in locating artifacts and background signals. Moreover, it should be considered that USI is not an appropriate technique for the imaging of gas-containing organs. |
| PACT | Imaging depth is limited (several centimeters). The imaging speed is also limited due to the repetition rate of laser pulses |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Děkanovský, L.; Li, J.; Zhou, H.; Sofer, Z.; Khezri, B. Nano/Microrobots Line Up for Gastrointestinal Tract Diseases: Targeted Delivery, Therapy, and Prevention. Energies 2022, 15, 426. https://doi.org/10.3390/en15020426
Děkanovský L, Li J, Zhou H, Sofer Z, Khezri B. Nano/Microrobots Line Up for Gastrointestinal Tract Diseases: Targeted Delivery, Therapy, and Prevention. Energies. 2022; 15(2):426. https://doi.org/10.3390/en15020426
Chicago/Turabian StyleDěkanovský, Lukáš, Jinhua Li, Huaijuan Zhou, Zdenek Sofer, and Bahareh Khezri. 2022. "Nano/Microrobots Line Up for Gastrointestinal Tract Diseases: Targeted Delivery, Therapy, and Prevention" Energies 15, no. 2: 426. https://doi.org/10.3390/en15020426
APA StyleDěkanovský, L., Li, J., Zhou, H., Sofer, Z., & Khezri, B. (2022). Nano/Microrobots Line Up for Gastrointestinal Tract Diseases: Targeted Delivery, Therapy, and Prevention. Energies, 15(2), 426. https://doi.org/10.3390/en15020426









