Organic Salt Hydrate as a Novel Paradigm for Thermal Energy Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Calcium Ceftriaxone (CaHS)
2.2. Chemical, Physical, Structural, Morphological, and Thermochemical Characterization of CaHS
2.3. Dehydration–Hydration Cycles
3. Results and Discussion
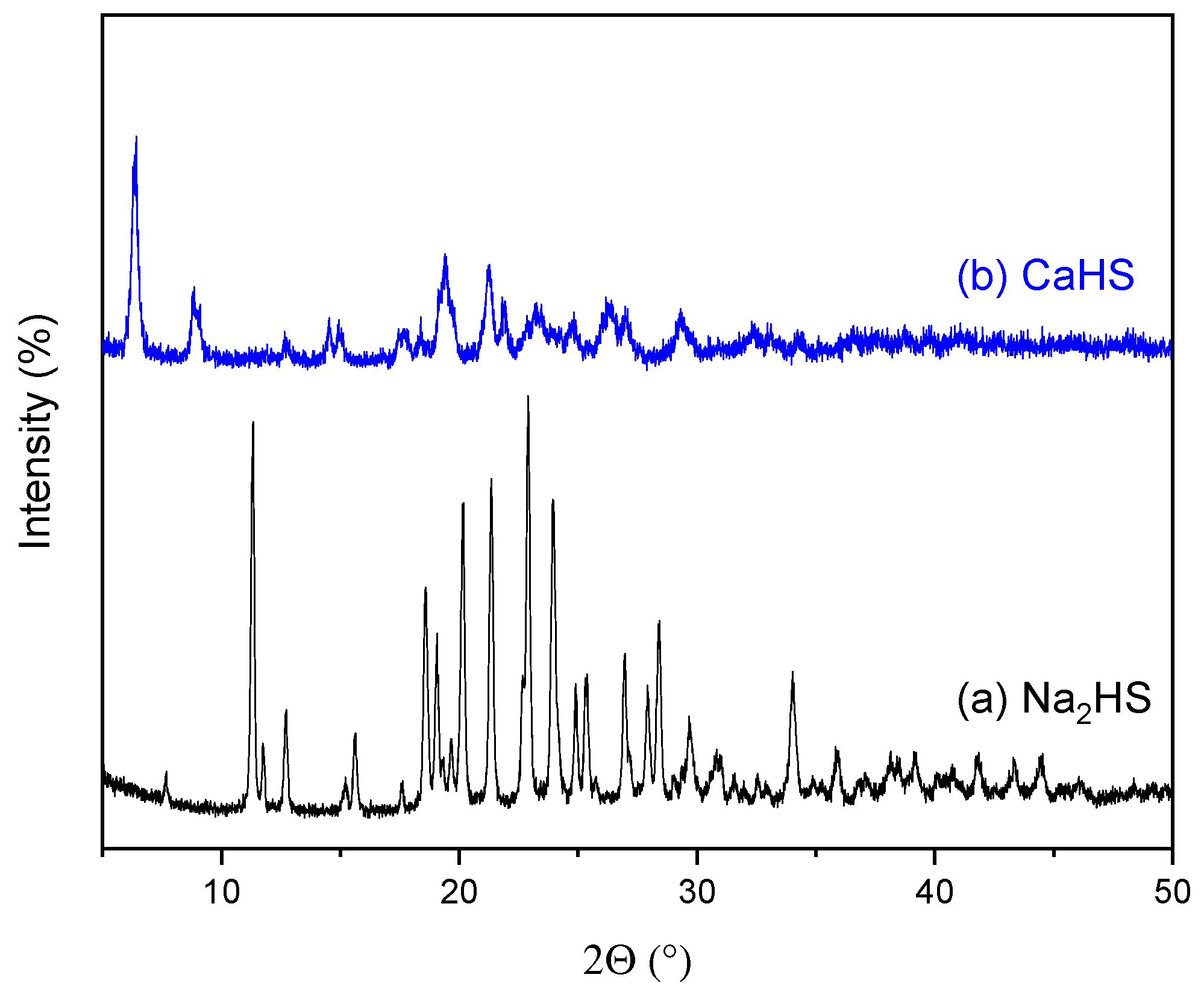
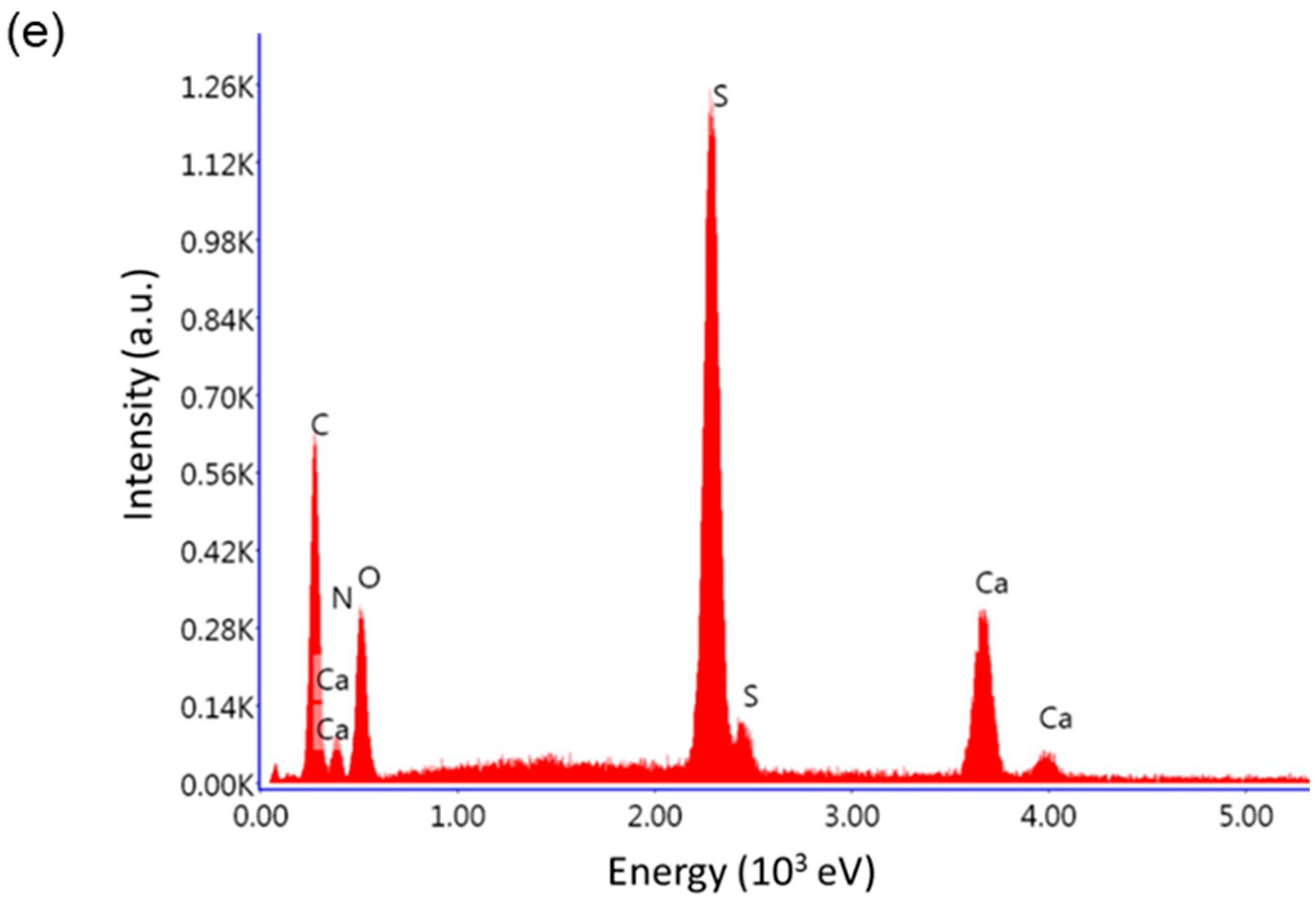
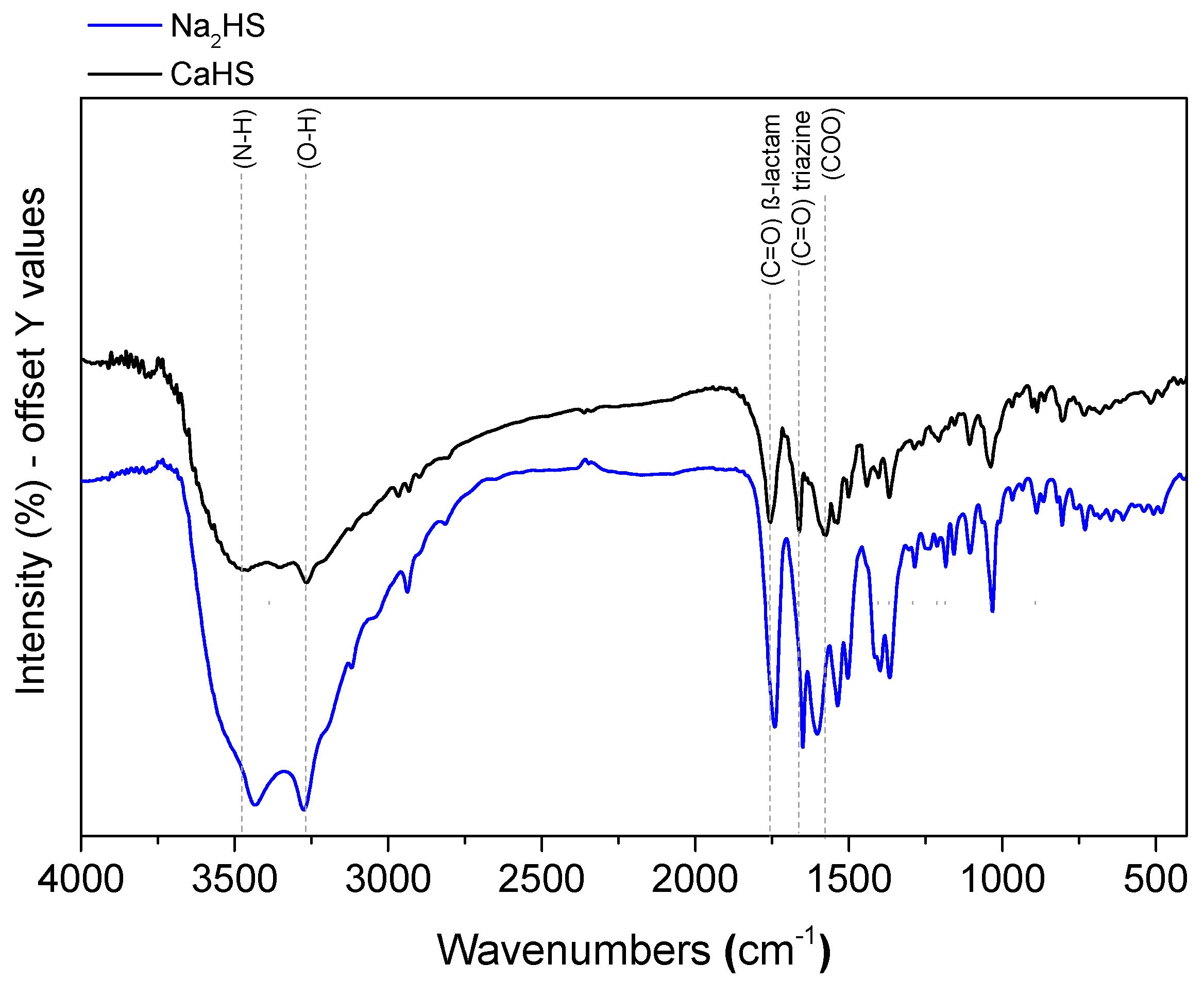
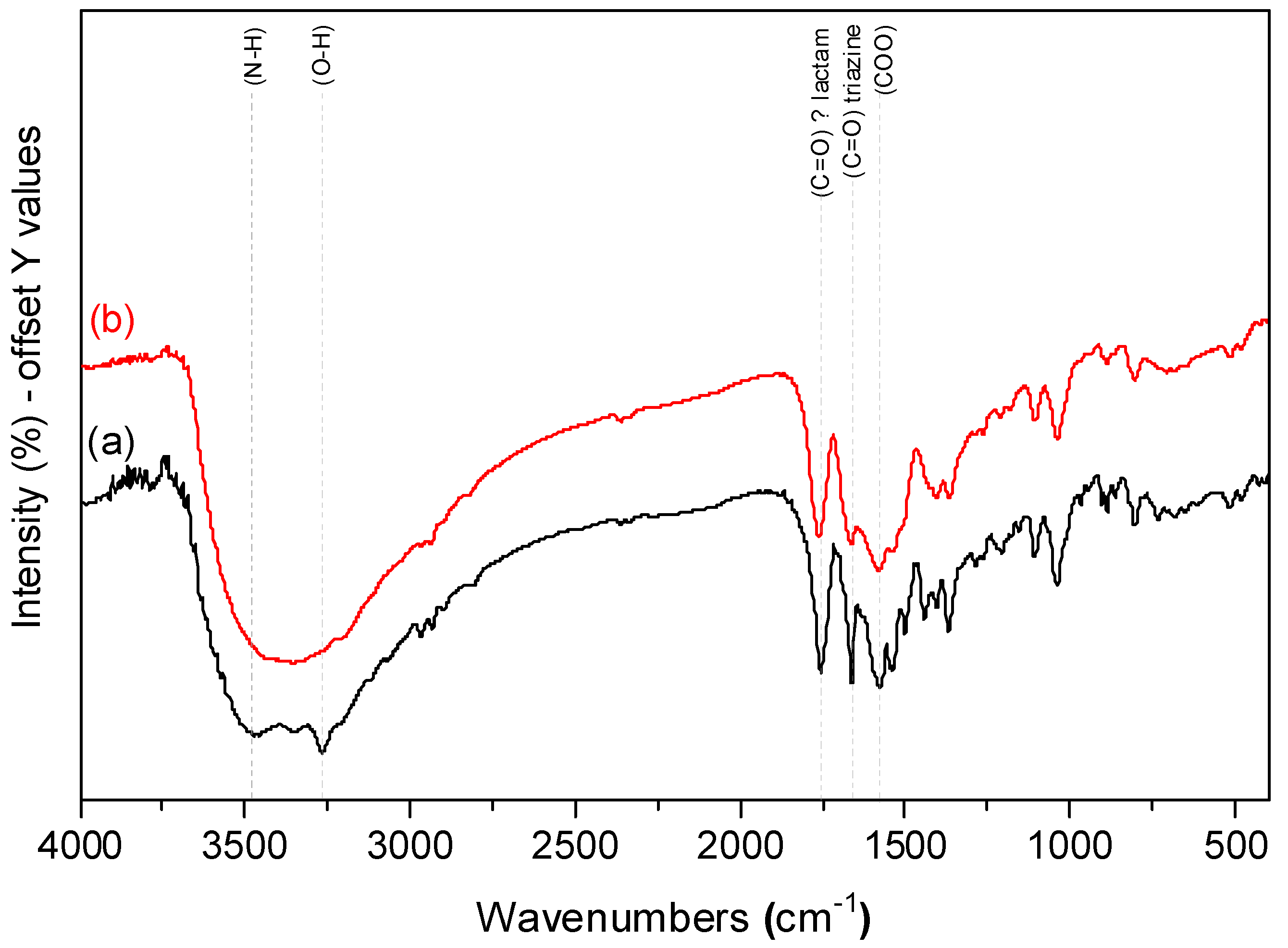
3.1. CaHS Chemical Analysis, Structural, and Morphological Characterization
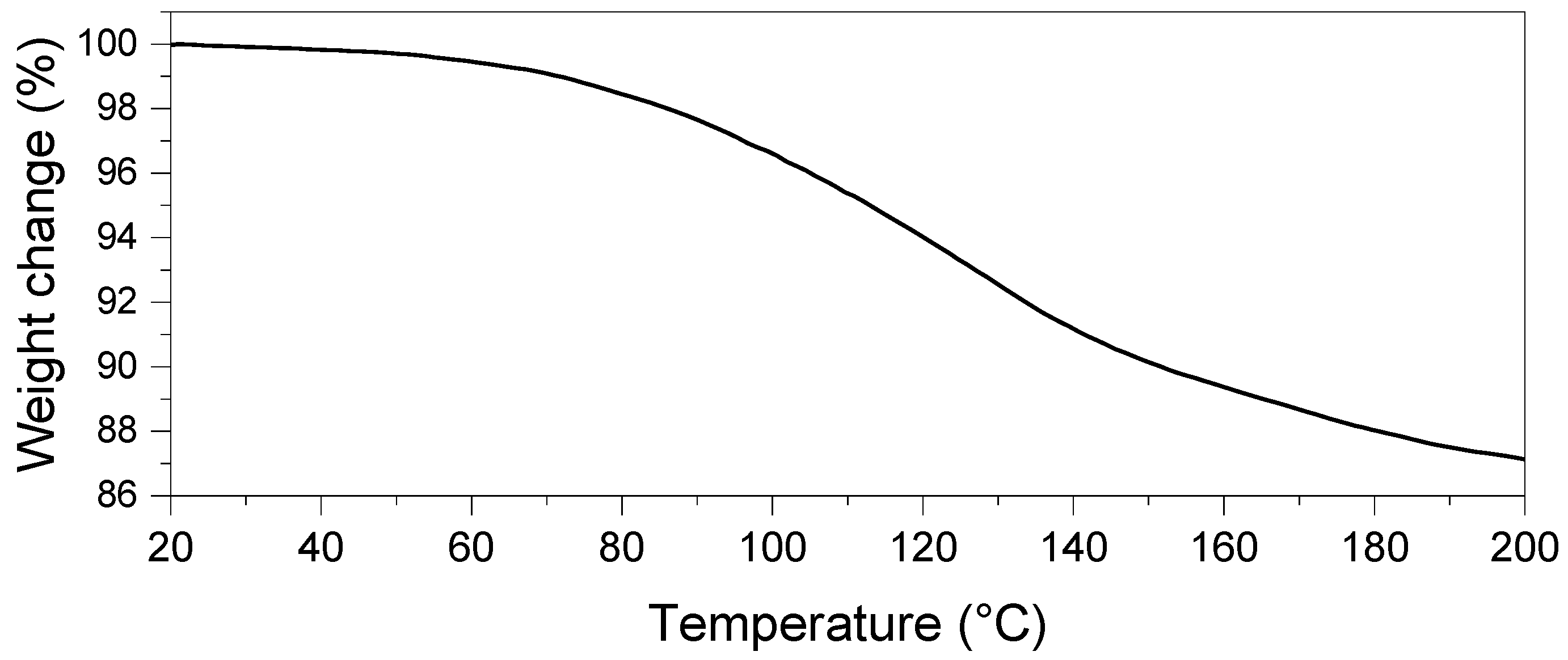
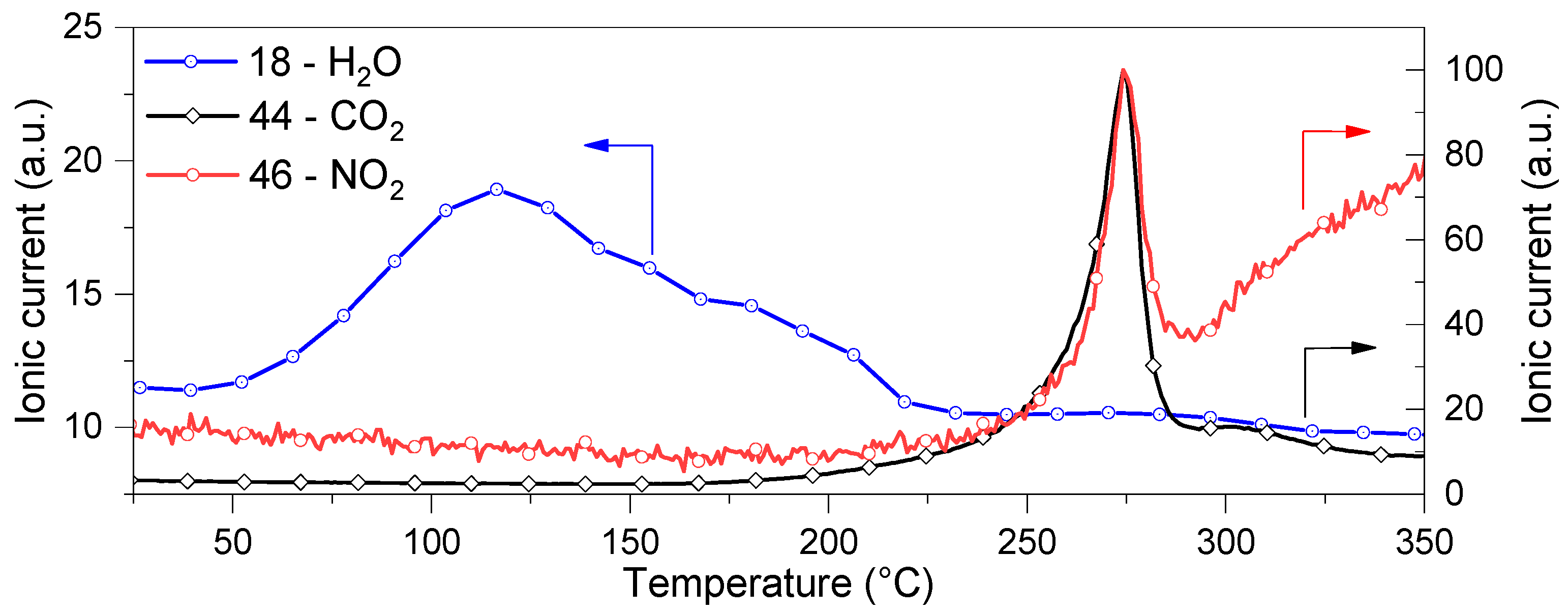
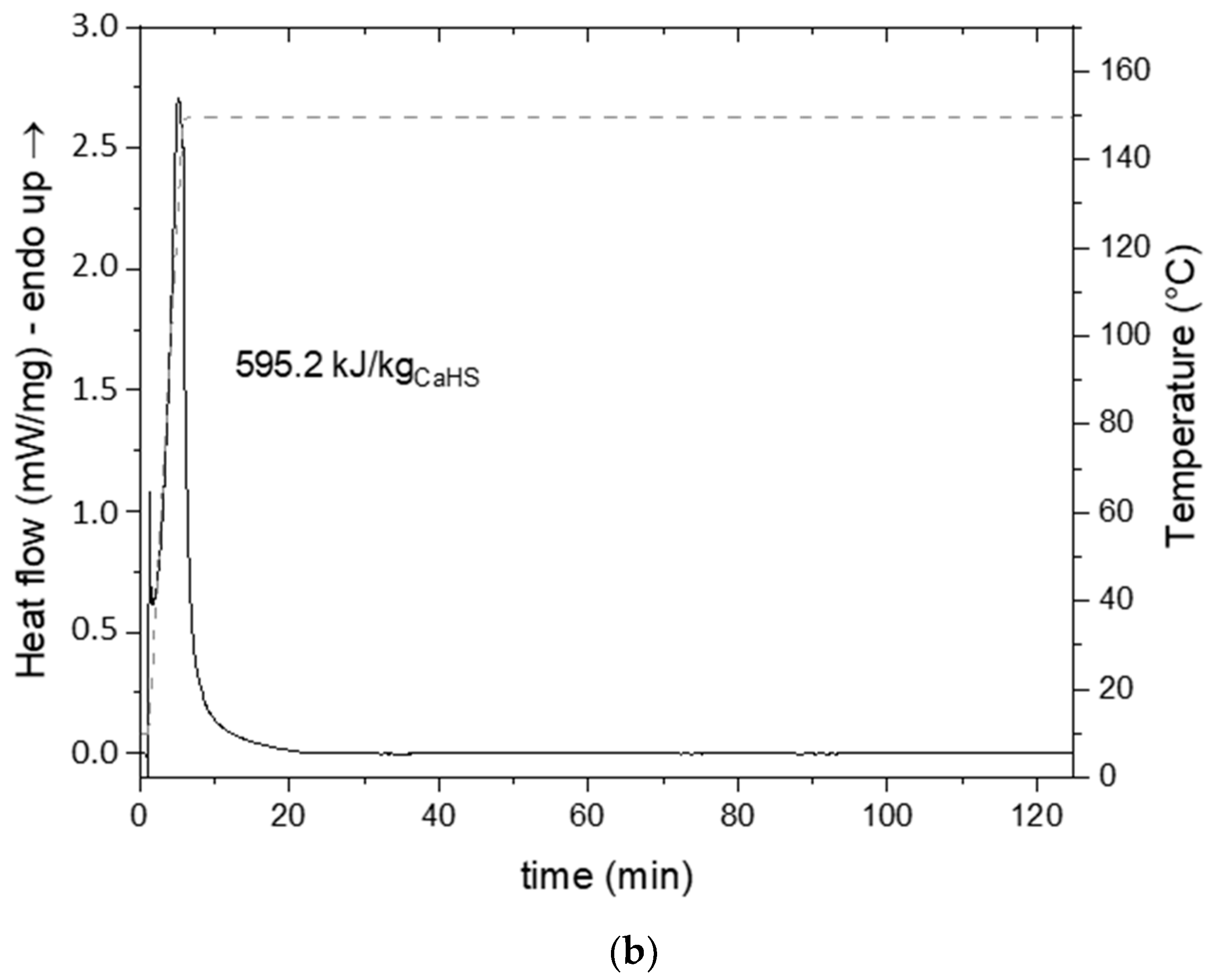
3.2. CaHS Thermochemical Behavior and Thermal Stability
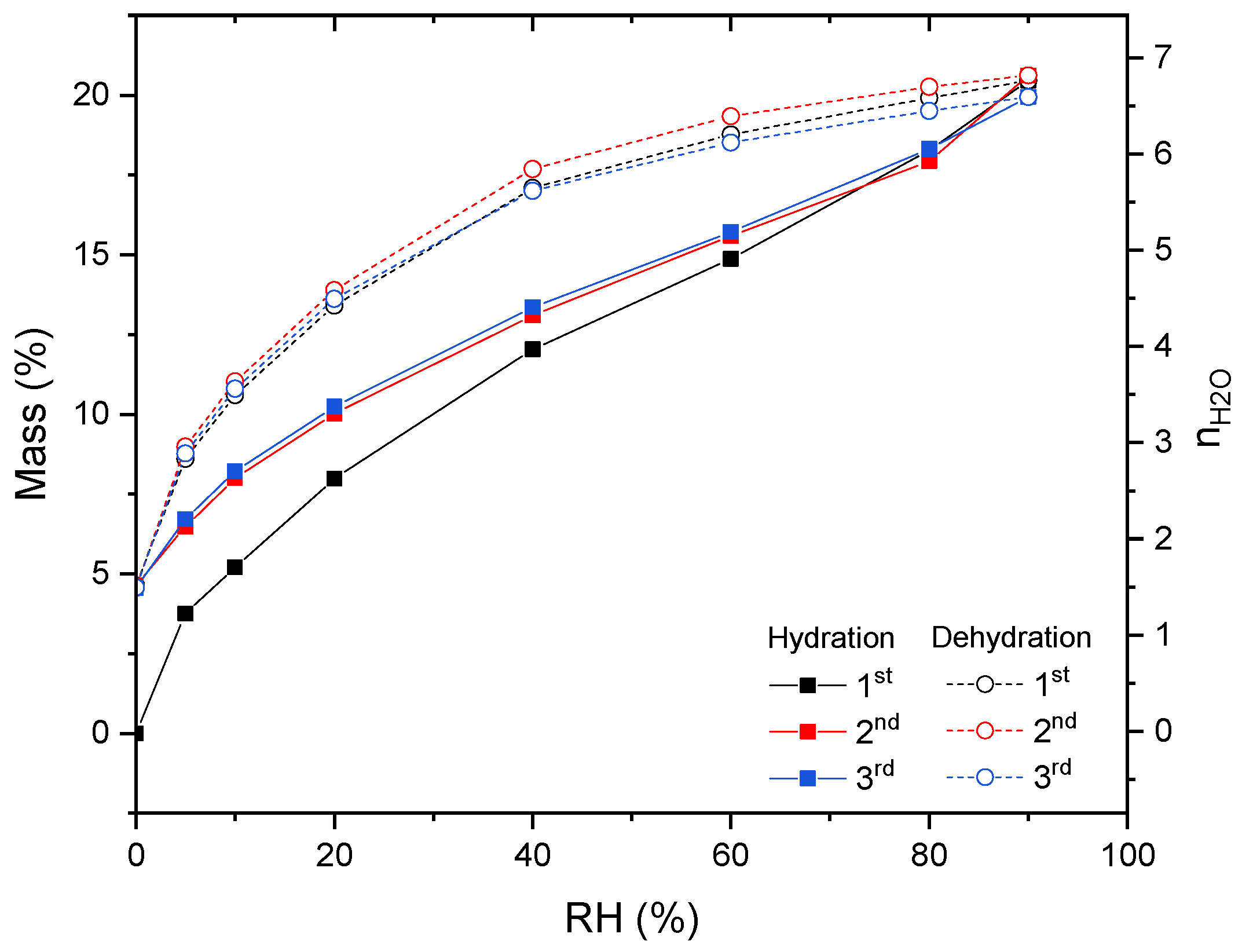
3.3. Dehydration–Hydration Cycles
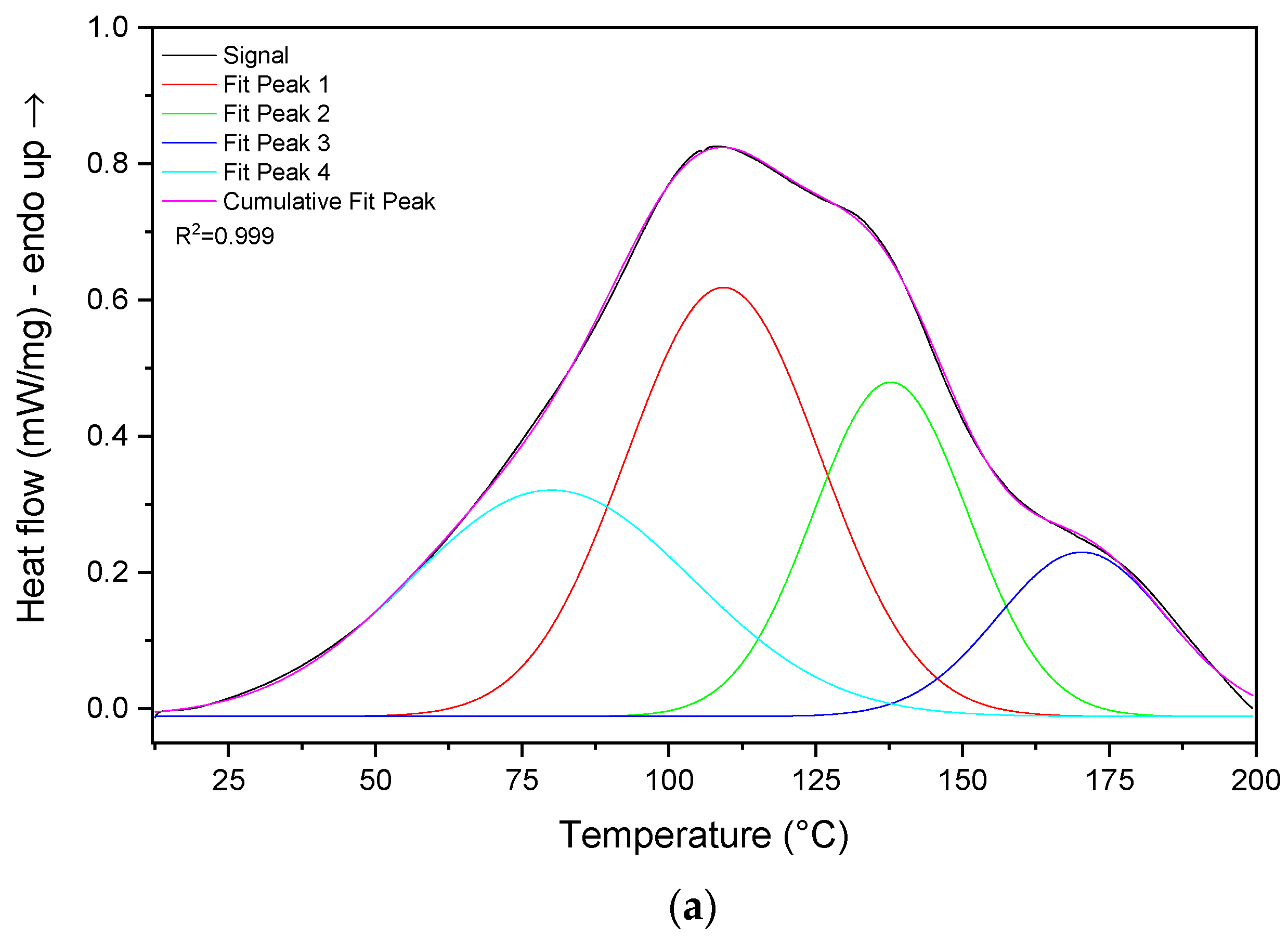
3.4. Heat Storage Capacity Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piperopoulos, E.; Mastronardo, E.; Fazio, M.; Lanza, M.; Galvagno, S.; Milone, C. Synthetic strategies for the enhancement of Mg(OH)2 thermochemical performances as heat storage material. Energy Procedia 2018, 155, 269–279. [Google Scholar] [CrossRef]
- Lahmidi, H.; Mauran, S.; Goetz, V. Definition, test and simulation of a thermochemical storage process adapted to solar thermal systems. Sol. Energy 2006, 80, 883–893. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Liu, H.; Le Pierrès, N.; Luo, L. A review on long-term sorption solar energy storage. Renew. Sustain. Energy Rev. 2009, 13, 2385–2396. [Google Scholar] [CrossRef]
- Letcher, T.M. Storing Energy, with Special Reference to Renewable Energy Sources. Chem. Int. 2016, 38, 28. [Google Scholar] [CrossRef]
- Pinel, P.; Cruickshank, C.A.; Beausoleil-Morrison, I.; Wills, A. A review of available methods for seasonal storage of solar thermal energy in residential applications. Renew. Sustain. Energy Rev. 2011, 15, 3341–3359. [Google Scholar] [CrossRef]
- Wang, X.; Li, B.; Qu, Z.; Zhang, J.; Jin, Z. Effects of graphite microstructure evolution on the anisotropic thermal conductivity of expanded graphite/paraffin phase change materials and their thermal energy storage performance. Int. J. Heat Mass Transf. 2020, 155, 119853. [Google Scholar] [CrossRef]
- Farulla, G.A.; Cellura, M.; Guarino, F.; Ferraro, M. A Review of Thermochemical Energy Storage Systems for Power Grid Support. Appl. Sci. 2020, 10, 3142. [Google Scholar] [CrossRef]
- Tescari, S.; Lantin, G.; Lange, M.; Breuer, S.; Agrafiotis, C.; Roeb, M.; Sattler, C. Numerical Model to Design a Thermochemical Storage System for Solar Power Plant. Energy Procedia 2015, 75, 2137–2143. [Google Scholar] [CrossRef] [Green Version]
- Tatsidjodoung, P.; Le Pierrès, N.; Luo, L. A review of potential materials for thermal energy storage in building applications. Renew. Sustain. Energy Rev. 2013, 18, 327–349. [Google Scholar] [CrossRef]
- N’Tsoukpoe, K.E.; Schmidt, T.; Rammelberg, H.U.; Watts, B.A.; Ruck, W.K. A systematic multi-step screening of numerous salt hydrates for low temperature thermochemical energy storage. Appl. Energy 2014, 124, 1–16. [Google Scholar] [CrossRef]
- Mehrabadi, A.; Farid, M. New salt hydrate composite for low-grade thermal energy storage. Energy 2018, 164, 194–203. [Google Scholar] [CrossRef]
- Lele, A.F. A Thermochemical Heat Storage System for Households; Springer: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Kohler, T.; Biedermann, T.; Müller, K. Experimental Study of MgCl2 ⋅ 6 H2 O as Thermochemical Energy Storage Material. Energy Technol. 2018, 6, 1935–1940. [Google Scholar] [CrossRef]
- Frazzica, A.; Cabeza, L.F. Recent Advancements in Materials and Systems for Thermal Energy Storage—An Introduction to Experimental Characterization Methods; Springer International Publishing: New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Mastronardo, E.; Bonaccorsi, L.; Kato, Y.; Piperopoulos, E.; Lanza, M.; Milone, C. Strategies for the enhancement of heat storage materials performances for MgO/H2O/Mg(OH)2 thermochemical storage system. Appl. Therm. Eng. 2017, 120, 626–634. [Google Scholar] [CrossRef]
- Druske, M.-M.; Fopah-Lele, A.; Korhammer, K.; Rammelberg, H.U.; Wegscheider, N.; Ruck, W.; Schmidt, T. Developed Materials for Thermal Energy Storage: Synthesis and Characterization. Energy Procedia 2014, 61, 96–99. [Google Scholar] [CrossRef] [Green Version]
- Zbair, M.; Bennici, S. Survey Summary on Salts Hydrates and Composites Used in Thermochemical Sorption Heat Storage: A Review. Energies 2021, 14, 3105. [Google Scholar] [CrossRef]
- Casey, S.P.; Elvins, J.; Riffat, S.; Robinson, A. Salt impregnated desiccant matrices for ‘open’ thermochemical energy storage—Selection, synthesis and characterisation of candidate materials. Energy Build. 2014, 84, 412–425. [Google Scholar] [CrossRef]
- Michel, B.; Neveu, P.; Mazet, N. Comparison of closed and open thermochemical processes, for long-term thermal energy storage applications. Energy 2014, 72, 702–716. [Google Scholar] [CrossRef] [Green Version]
- Gaeini, M.; Rouws, A.; Salari, J.; Zondag, H.; Rindt, C. Characterization of microencapsulated and impregnated porous host materials based on calcium chloride for thermochemical energy storage. Appl. Energy 2018, 212, 1165–1177. [Google Scholar] [CrossRef]
- Owens, H.M.; Dash, A.K. Ceftriaxone Sodium: Comprehensive Profile. Profiles Drug Subst. Excipients Relat. Methodol. 2003, 30, 21–57. [Google Scholar] [CrossRef]
- Refat, M.S.; Altalhi, T.; Fetooh, H.; Alsuhaibani, A.M.; Hassan, R.F. In neutralized medium five new Ca(II), Zn(II), Fe(III), Au(III) and Pd(II) complexity of ceftriaxone antibiotic drug: Synthesis, spectroscopic, morphological and anticancer studies. J. Mol. Liq. 2020, 322, 114816. [Google Scholar] [CrossRef]
- Katz, K.A.; Glusker, J.P.; Beebe, S.A.; Bock, C.W. Calcium Ion Coordination: A Comparison with That of Beryllium, Magnesium, and Zinc. J. Am. Chem. Soc. 1996, 118, 5752–5763. [Google Scholar] [CrossRef]
- Gonzalez, D.; Golab, J.T.; Eilert, J.Y.; Wang, R.; Kaduk, J.A. Crystal structure of ceftriaxone sodium hemiheptahydrate, C18H16N8O7S3Na2(H2O)3.5. Powder Diffr. 2020, 35, 206–212. [Google Scholar] [CrossRef]
- Tieger, E.; Kiss, V.; Pokol, G.; Finta, Z.; Dušek, M.; Rohlíček, J.; Skořepová, E.; Brázda, P. Studies on the crystal structure and arrangement of water in sitagliptinl-tartrate hydrates. CrystEngComm 2016, 18, 3819–3831. [Google Scholar] [CrossRef] [Green Version]
- Clark, R.-J.; Mehrabadi, A.; Farid, M. State of the art on salt hydrate thermochemical energy storage systems for use in building applications. J. Energy Storage 2019, 27, 101145. [Google Scholar] [CrossRef]
- Donkers, P.; Sögütoglu, L.; Huinink, H.; Fischer, H.; Adan, O. A review of salt hydrates for seasonal heat storage in domestic applications. Appl. Energy 2017, 199, 45–68. [Google Scholar] [CrossRef]


| Compound | n | ΔH (kJ/mol) | QM (kJ/kg) | QV (kWh/m3) | Tdeh (°C) | Thyd (°C) | Ref. |
|---|---|---|---|---|---|---|---|
| CaHS · 7H2O | 4.3 | 427.8 | 595.2 | 278.6 | 150 | 30 | this study |
| SrBr2 · 6H2O | 5 | 337.0 | 947.9 | 433.5 | 52 | 45 | [3,4,5] |
| SrCl2 · 2H2O | 1 | 59.0 | 302.4 | 164.1 | 52 | 46 | [3,4,5] |
| MgSO4 · 6H2O | 4 | 225.1 | 986.4 | 558.0 | 91–123 | 10 | [3,4,5] |
| MgCl2 · 6H2O | 1.3 | 71.5 | 351.7 | 153.3 | 104 | 61 | [4,8] |
| CaCl2 · 2H2O | 2 | 125.0 | 837.0 | 542.5 | 111 | 63 | [4,8] |
| LiCl · H2O | 1 | 62.9 | 1041.2 | 486.0 | 80 | 73 | [4] |
| K2CO3 · 1.5H2O | 1.5 | 95.5 | 579.6 | 355.6 | 65 | 59 | [4] |
| Na2S · 5H2O | 3 | 188.7 | 1120.3 | 780.6 | 73 | 66 | [3,4] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mastronardo, E.; La Mazza, E.; Palamara, D.; Piperopoulos, E.; Iannazzo, D.; Proverbio, E.; Milone, C. Organic Salt Hydrate as a Novel Paradigm for Thermal Energy Storage. Energies 2022, 15, 4339. https://doi.org/10.3390/en15124339
Mastronardo E, La Mazza E, Palamara D, Piperopoulos E, Iannazzo D, Proverbio E, Milone C. Organic Salt Hydrate as a Novel Paradigm for Thermal Energy Storage. Energies. 2022; 15(12):4339. https://doi.org/10.3390/en15124339
Chicago/Turabian StyleMastronardo, Emanuela, Emanuele La Mazza, Davide Palamara, Elpida Piperopoulos, Daniela Iannazzo, Edoardo Proverbio, and Candida Milone. 2022. "Organic Salt Hydrate as a Novel Paradigm for Thermal Energy Storage" Energies 15, no. 12: 4339. https://doi.org/10.3390/en15124339
APA StyleMastronardo, E., La Mazza, E., Palamara, D., Piperopoulos, E., Iannazzo, D., Proverbio, E., & Milone, C. (2022). Organic Salt Hydrate as a Novel Paradigm for Thermal Energy Storage. Energies, 15(12), 4339. https://doi.org/10.3390/en15124339











