Nano-Iron Oxide-Ethylene Glycol-Water Nanofluid Based Photovoltaic Thermal (PV/T) System with Spiral Flow Absorber: An Energy and Exergy Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. System Description
2.2. Materials
2.3. Mixing Procedure
2.4. Measurements
- HOT DESK Tps 500 (KIJTALEY, Sweden) for measuring the thermal conductivity of emulsions.
- Density tester meter for prepared emulsions density.
- Brookfield Programmer Viscometer (Model: LVDV-III Ultra-programmable) is used to measure the viscosity of emulsions. This instrument is connected to a laptop to collect and store the measured data.
- Nano Zeta-Sizer (ZSN) was used to measure the stability of the prepared emulsions.
- eR: Measurements uncertainty.
- R: An independent variable function V1, V2, …, Vn or
- R = R (V1, V2, …, Vn).
- ei: nth variable uncertainty interval.
- : A single variable measured result sensitivity.
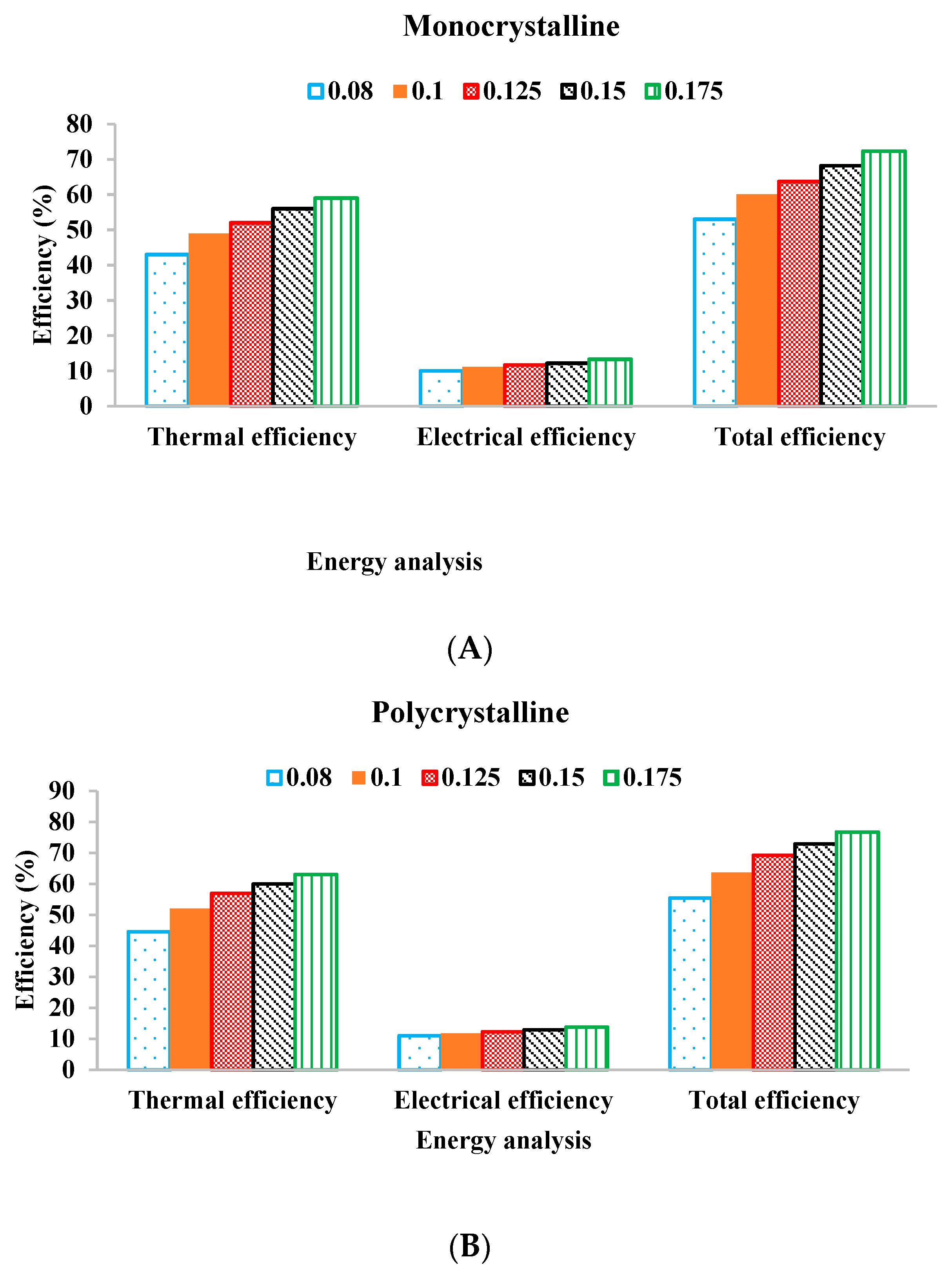
2.5. Energy Analysis
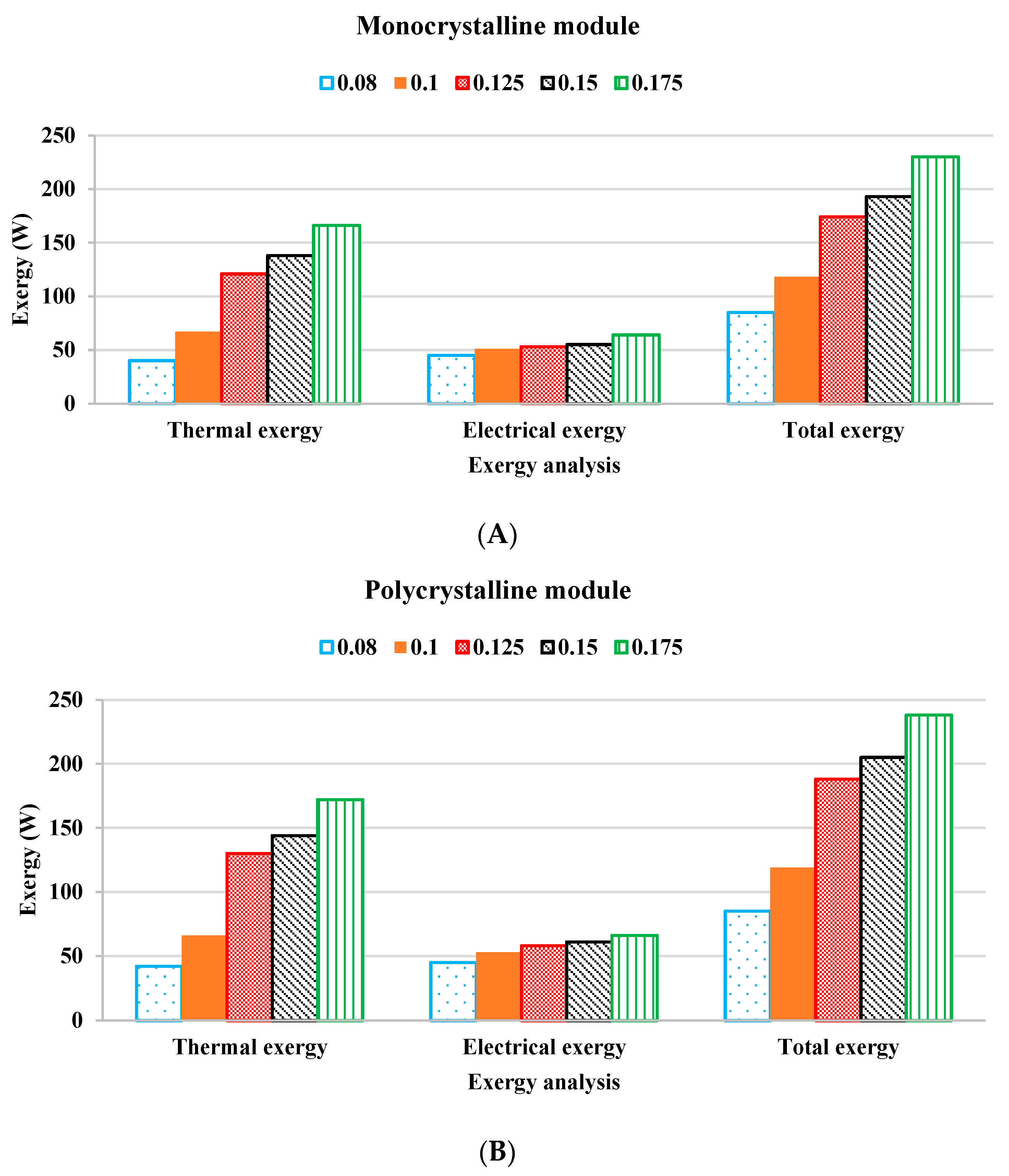
2.6. Exergy Analysis
3. Results and Discussion
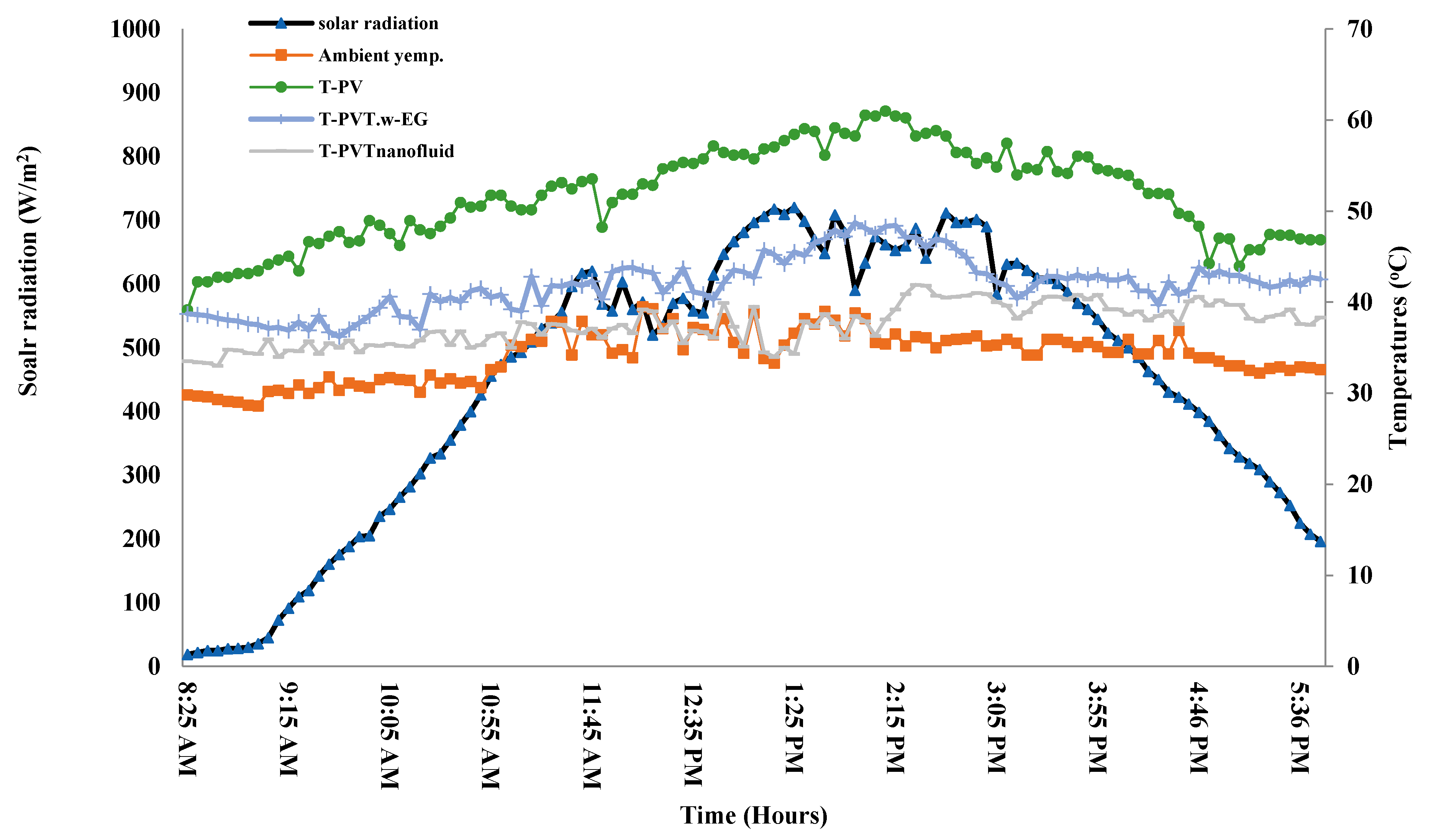
3.1. Climate Conditions
3.2. Thermophysical Properties
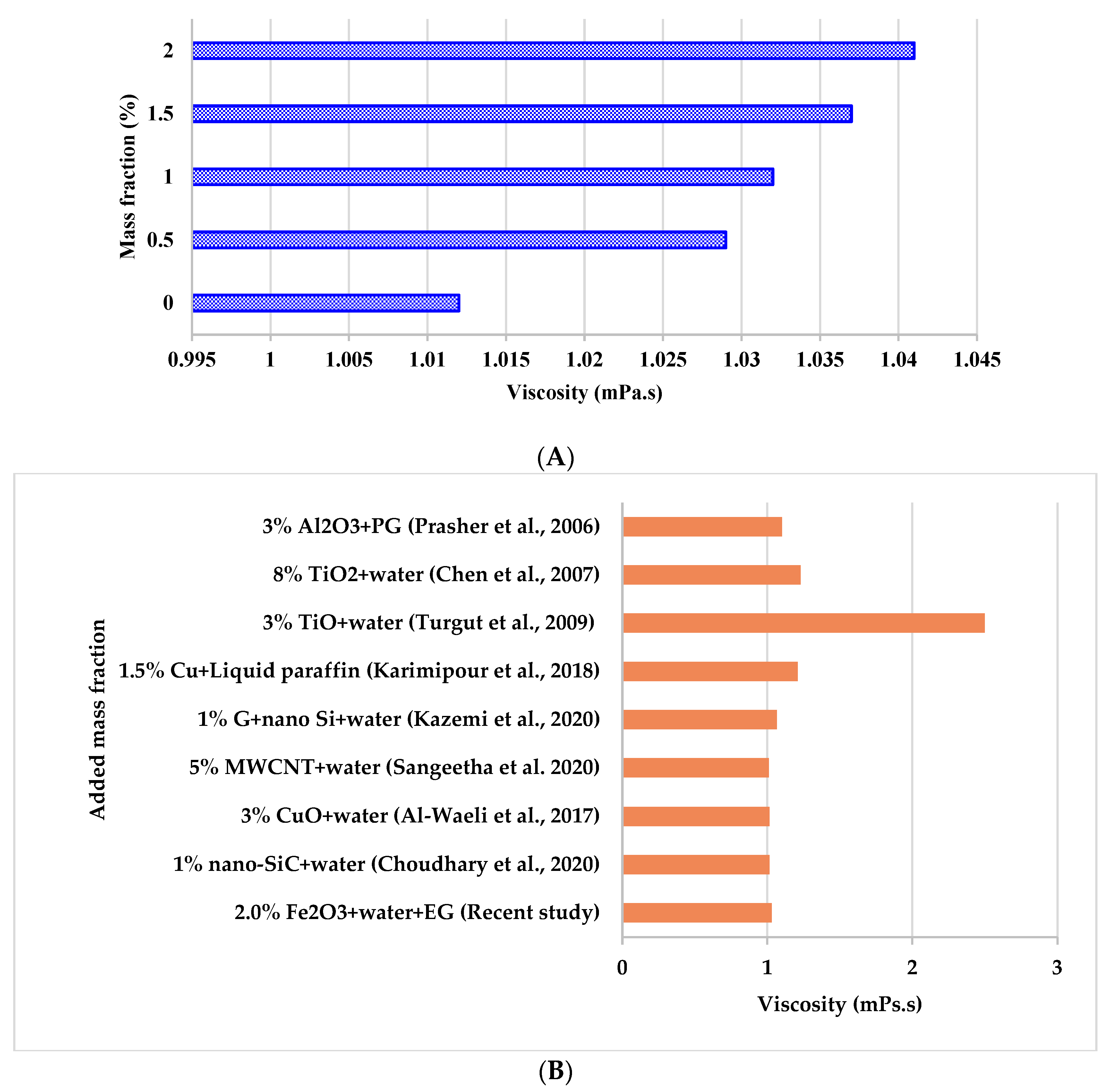
3.2.1. Viscosity
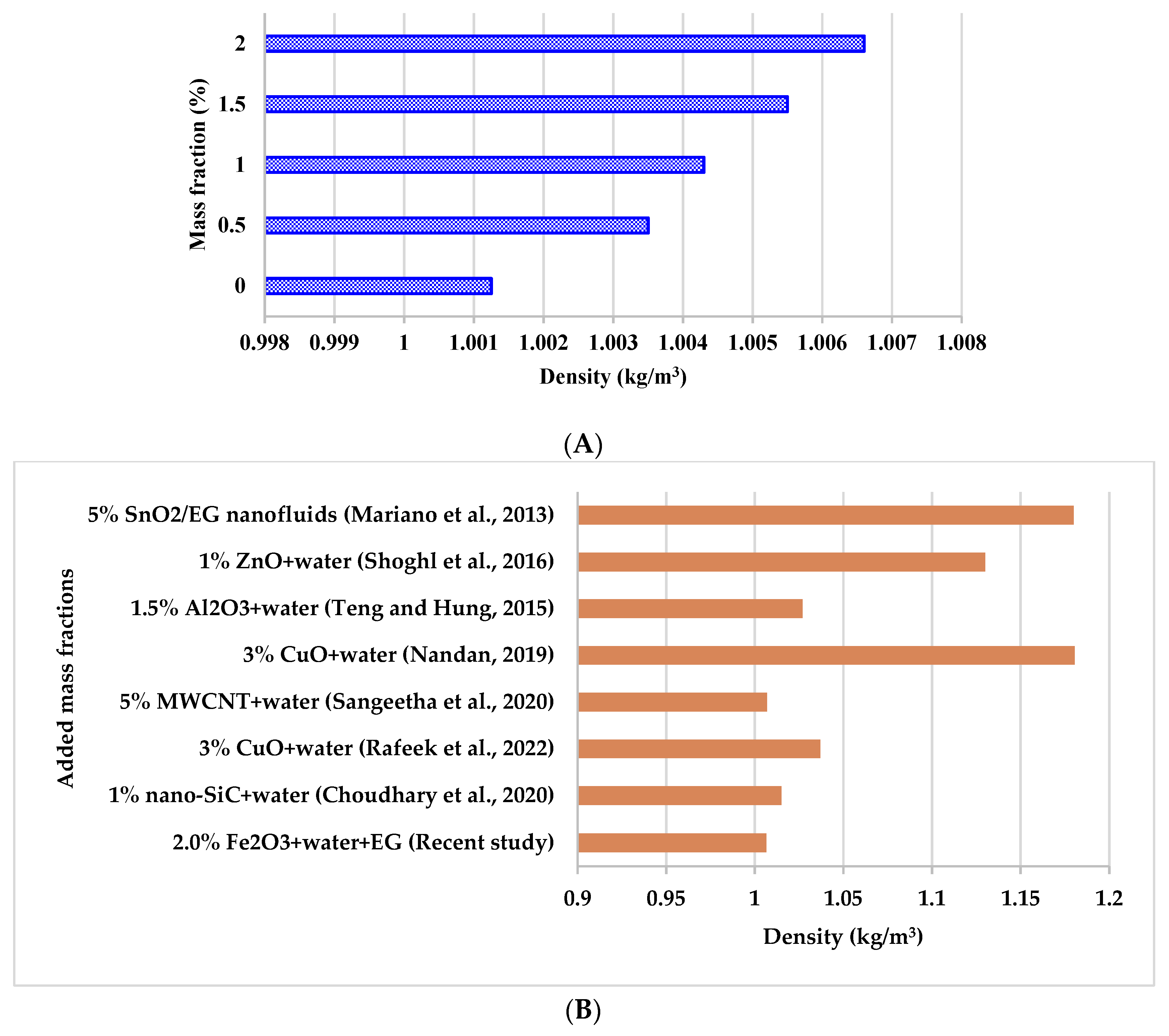
3.2.2. Density
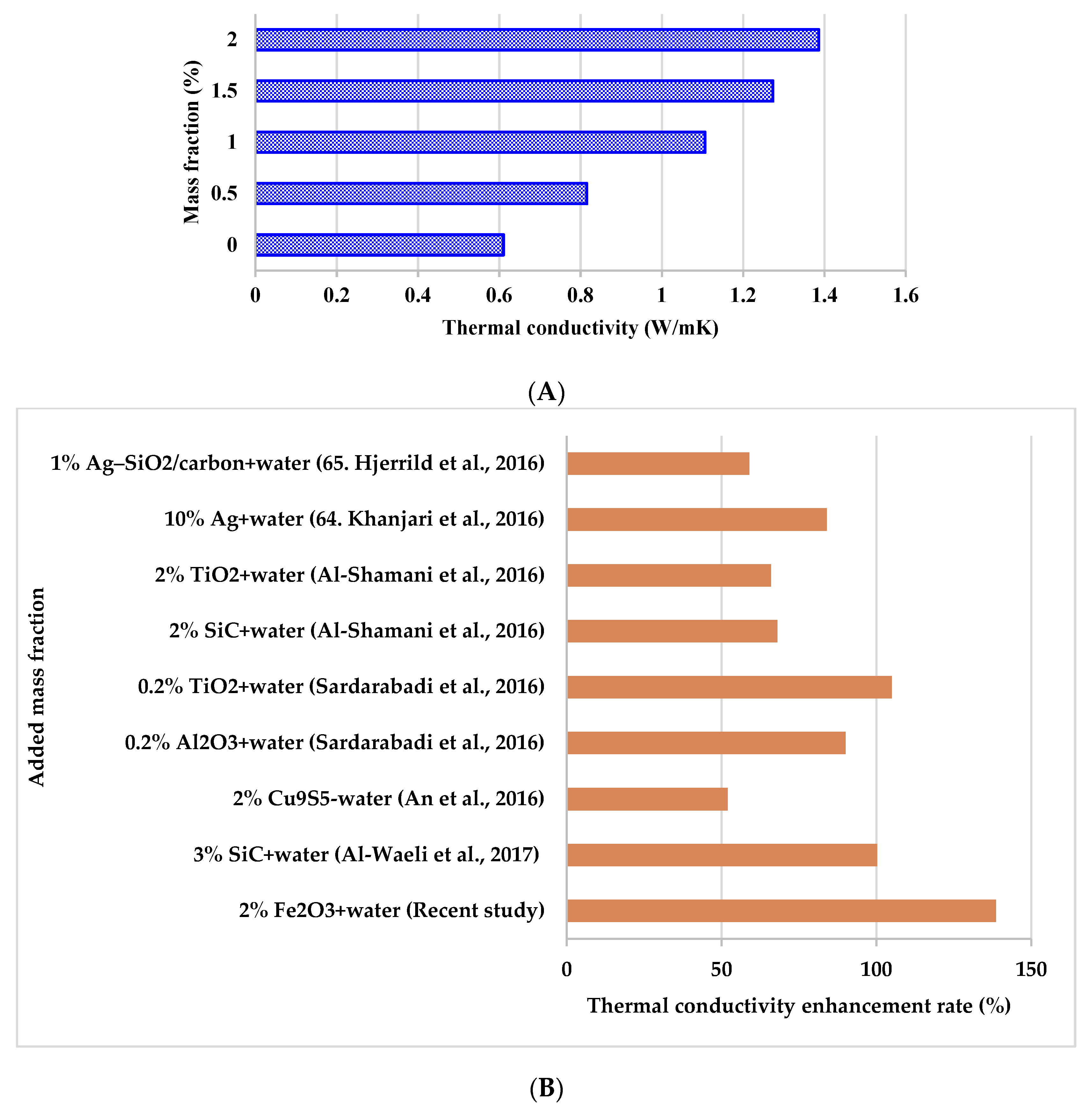
3.2.3. Thermal Conductivity
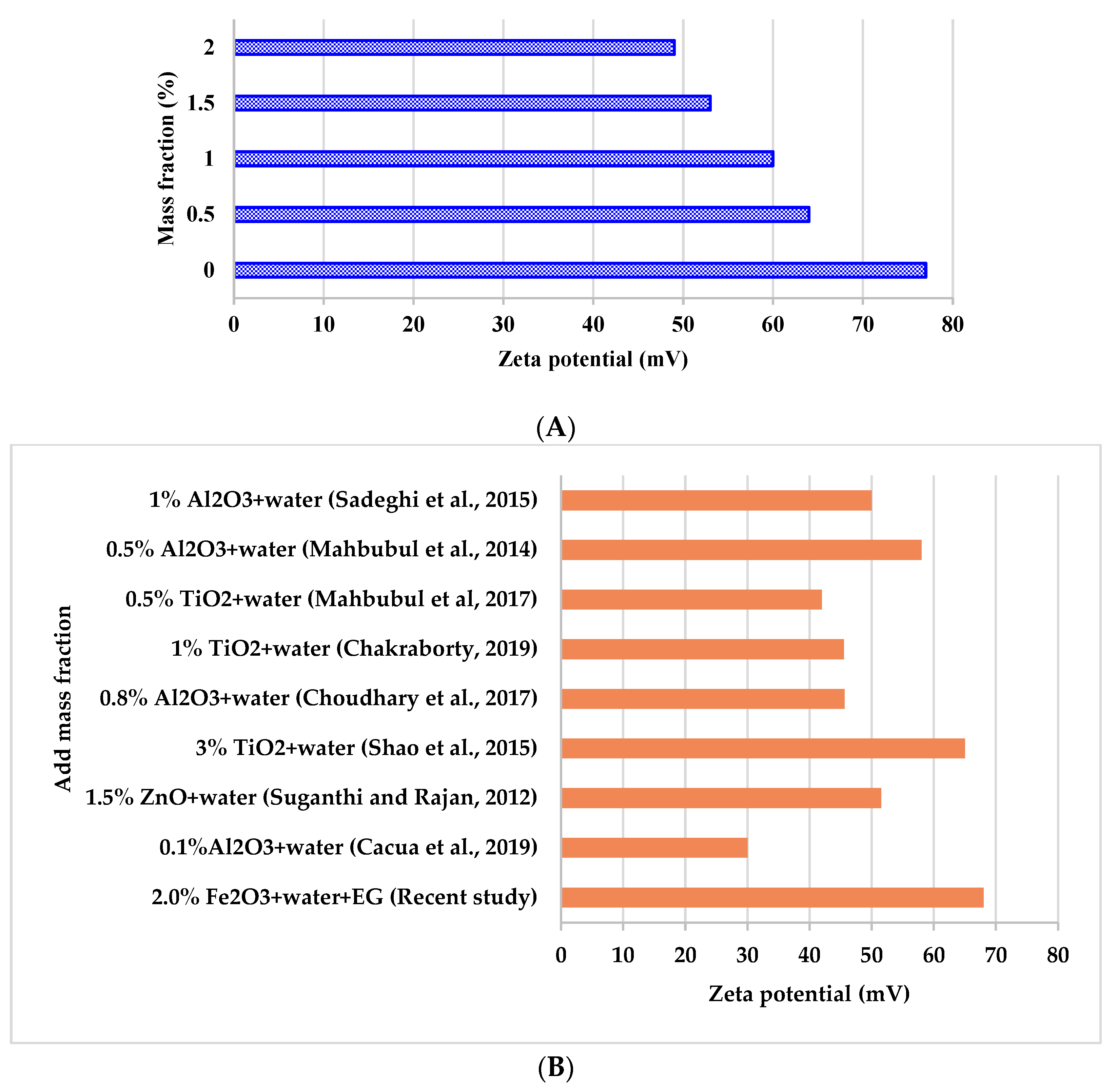
3.2.4. Stability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, Z.; Huang, Z.; Failler, P. Dynamic correlation between crude oil price and investor sentiment in China: Heterogeneous and asymmetric effect. Energies 2022, 15, 687. [Google Scholar] [CrossRef]
- Chaichan, M.T.; Kazem, H.A.; Abed, T.A. Traffic and outdoor air pollution levels near highways in Baghdad, Iraq. Environ. Dev. Sustain. 2018, 20, 589–603. [Google Scholar] [CrossRef]
- Long, E.; Carlsten, C. Controlled human exposure to diesel exhaust: Results illuminate health effects of traffic-related air pollution and inform future directions. Part. Fibre Toxicol. 2022, 19, 11. [Google Scholar] [PubMed]
- Halttunen, K.; Slade, R.; Staffell, I. What if we never run out of oil? From certainty of “peak oil” to “peak demand”. Energy Res. Soc. Sci. 2022, 85, 102407. [Google Scholar] [CrossRef]
- Colasante, A.; D’Adamo, I.; Morone, P. What drives the solar energy transition? The effect of policies, incentives and behavior in a cross-country comparison. Energy Res. Soc. Sci. 2022, 85, 102405. [Google Scholar] [CrossRef]
- Wattana, B.; Aungyut, P. Impacts of solar electricity generation on the Thai electricity industry. Int. J. Renew. Energy Dev. 2022, 11, 156–163. [Google Scholar] [CrossRef]
- Duda, J.; Kusa, R.; Pietruszko, S.; Smol, M.; Suder, M.; Teneta, J.; Wójtowicz, T.; Żdanowicz, T. Development of roadmap for photovoltaic solar technologies and market in Poland. Energies 2022, 15, 174. [Google Scholar] [CrossRef]
- Gupta, B.; Bhalavi, J.; Sharma, S.; Bisen, A. Phase change materials in solar energy applications: A review. Mater. Today Proc. 2021, 46, 5550–5554. [Google Scholar] [CrossRef]
- Meilinger, S.; Herman-Czezuch, A.; Zemo, A.; Bebber, M. Impact of dust storms and urban air pollution on PV-power systems: Case studies from Ghana. In Proceedings of the EMS Annual Meeting Abstracts 2021, No. EMS2021-419, Online, 6–10 September 2021. [Google Scholar] [CrossRef]
- Ravindra, B. Forecasting solar radiation during dust storms using deep learning. arXiv 2018, arXiv:1808.10854. [Google Scholar]
- Charfi, W.; Chaabane, M.; Mhiri, H.; Bournot, P. Performance evaluation of a solar photovoltaic system. Energy Rep. 2018, 4, 400–406. [Google Scholar] [CrossRef]
- Mamun, M.A.A.; Islam, M.M.; Hasanuzzaman, M.; Selvaraj, J. Effect of tilt angle on the performance and electrical parameters of a PV module: Comparative indoor and outdoor experimental investigation. Energy Built Environ. 2021, 3, 278–290. [Google Scholar] [CrossRef]
- Alnasser, T.M.; Mahdy, A.M.; Abass, K.I.; Chaichan, M.T.; Kazem, H.A. Impact of dust ingredient on photovoltaic performance: An experimental study. Sol. Energy 2020, 195, 651–659. [Google Scholar] [CrossRef]
- Kazem, H.A.; Chaichan, M.T.; Al-Waeli, A.H.; Sopian, K. A review of dust accumulation and cleaning methods for solar photovoltaic systems. J. Clean. Prod. 2020, 276, 123187. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Sopian, K.; Kazem, H.A.; Chaichan, M.T. Photovoltaic solar thermal (PV/T) collectors past, present and future: A review. Int. J. Appl. Eng. Res. 2016, 11, 10757–10765. [Google Scholar]
- Esfe, M.H.; Kamyab, M.H.; Valadkhani, M. Application of nanofluids and fluids in photovoltaic thermal system: An updated review. Sol. Energy 2020, 199, 796–818. [Google Scholar] [CrossRef]
- Shrivastava, A.; Jose, J.P.A.; Borole, Y.D.; Saravanakumar, R.; Sharifpur, M.; Harasi, H.; Razak, R.A.; Afzal, A. A study on the effects of forced air-cooling enhancements on a 150 W solar photovoltaic thermal collector for green cities. Sustain. Energy Technol. Assess. 2022, 49, 101782. [Google Scholar] [CrossRef]
- Gomaa, M.R.; Ahmed, M.; Rezk, H. Temperature distribution modeling of PV and cooling water PV/T collectors through thin and thick cooling cross-fined channel box. Energy Rep. 2022, 8, 1144–1153. [Google Scholar] [CrossRef]
- Parsa, S.M.; Yazdani, A.; Aberoumand, H.; Farhadi, Y.; Ansari, A.; Aberoumand, S.; Karimi, N.; Afrand, M.; Cheraghian, G.; Ali, H.M. A critical analysis on the energy and exergy performance of photovoltaic/thermal (PV/T) system: The role of nanofluids stability and synthesizing method. Sustain. Energy Technol. Assess. 2022, 51, 101887. [Google Scholar]
- Kong, X.; Zhang, L.; Li, H.; Wang, Y.; Fan, M. Experimental thermal and electrical performance analysis of a concentrating photovoltaic/thermal system integrated with phase change material (PV/T-CPCM). Sol. Energy Mater. Sol. Cells 2022, 234, 111415. [Google Scholar] [CrossRef]
- Azimi, N.; Davoodbeygi, Y.; Rahimi, M.; Ahmadi, S.; Karami, E.; Roshani, M. Optimization of thermal and electrical efficiencies of a photovoltaic module using combined PCMs with a thermo-conductive filler. Sol. Energy 2022, 231, 283–296. [Google Scholar] [CrossRef]
- Sathyamurthy, R.; Kabeel, A.E.; Chamkha, A.; Karthick, A.; Manokar, A.M.; Sumithra, M.G. Experimental investigation on cooling the photovoltaic panel using hybrid nanofluids. Appl. Nanosci. 2021, 11, 363–374. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Chaichan, M.T.; Kazem, H.A.; Sopian, K. Evaluation and analysis of nanofluid and surfactant impact on photovoltaic-thermal systems. Case Stud. Therm. Eng. 2019, 13, 100392. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Chaichan, M.T.; Sopian, K.; Kazem, H.A. Influence of the base fluid on the thermo-physical properties of PV/T nanofluids with surfactant. Case Stud. Therm. Eng. 2019, 13, 100340. [Google Scholar] [CrossRef]
- Sangeetha, M.; Manigandan, S.; Chaichan, M.T.; Kumar, V. Progress of MWCNT, Al2O3, and CuO with water in enhancing the photovoltaic thermal system. Int. J. Energy Res. 2020, 44, 821–832. [Google Scholar] [CrossRef]
- Al-Ghezi, M.K.; Abass, K.I.; Salam, A.Q.; Jawad, R.S.; Kazem, H.A. The possibilities of using nano-CuO as coolants for PVT system: An experimental study. J. Phys. Conf. Ser. 2021, 1973, 012123. [Google Scholar] [CrossRef]
- Kazem, H.A.; Chaichan, M.T.; Al-Waeli, A.H.; Sopian, K. Investigation of a nanofluid-based photovoltaic thermal system using single-wall carbon nanotubes: An experimental study. Int. J. Energy Res. 2021, 45, 10285–10303. [Google Scholar] [CrossRef]
- Fu, Z.; Liang, X.; Li, Y.; Li, L.; Zhu, Q. Performance improvement of a PVT system using a multilayer structural heat exchanger with PCMs. Renew. Energy 2021, 169, 308–317. [Google Scholar] [CrossRef]
- Siddiqui, M.U.; Siddiqui, O.K.; Al-Sarkhi, A.; Arif, A.F.M.; Zubair, S.M. A novel heat exchanger design procedure for photovoltaic panel cooling application: An analytical and experimental evaluation. Appl. Energy 2019, 239, 41–56. [Google Scholar] [CrossRef]
- Yadav, S.; Panda, S.K.; Tiwari, G.N.; Al-Helal, I.M.; Alsadon, A.A.; Shady, M.R.; Tiwari, A. Semi-transparent photovoltaic thermal greenhouse system combined with earth air heat exchanger for hot climatic condition. J. Therm. Sci. Eng. Appl. 2022, 14, 081007. [Google Scholar]
- Kazem, H.A.; Al-Waeli, A.H.; Chaichan, M.T.; Al-Waeli, K.H.; Al-Aasam, A.B.; Sopian, K. Evaluation and comparison of different flow configurations PVT systems in Oman: A numerical and experimental investigation. Sol. Energy 2020, 208, 58–88. [Google Scholar] [CrossRef]
- Al-Ghezi, M.K.; Mahmoud, B.K.; Alnasser, T.; Chaichan, M.T. A comparative study of regression models and meteorological parameters to estimate the global solar radiation on a horizontal surface for Baghdad City, Iraq. Int. J. Renew. Energy Dev. 2022, 11, 71–81. [Google Scholar] [CrossRef]
- Chaichan, M.T.; Kazem, H.A. Generating Electricity Using Photovoltaic Solar Plants in Iraq; Springer: Berlin/Heidelberg, Germany, 2018; ISBN 978-3-319-75030-9. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Kazem, H.A.; Sopian, K.; Chaichan, M.T. Techno-economical assessment of grid connected PV/T using nanoparticles and water as base-fluid systems in Malaysia. Int. J. Sustain. Energy 2018, 37, 558–575. [Google Scholar] [CrossRef]
- Kong, X.; Ohadi, M.M. November. Applications of micro and nano technologies in the oil and gas industry-an overview of the recent progress. In Proceedings of the Abu Dhabi International Petroleum Exhibition and Conference, OnePetro, Abu Dhabi, United Arab Emirates, 1 November 2010. [Google Scholar]
- Zhao, B.; Wang, Q.; Jin, L. Heat-resistant antiflaming and friction mechanisms in nano-Fe2O3-reinforced silicon rubber. Sci. Eng. Compos. Mater. 2013, 20, 331–335. [Google Scholar] [CrossRef]
- Nikolic, V.N.; Spasojevic, V.; Panjan, M.; Kopanja, L.; Mrakovic, A.; Tadic, M. Re-formation of metastable ε-Fe2O3 in post-annealing of Fe2O3/SiO2 nanostructure: Synthesis, computational particle shape analysis in micrographs and magnetic properties. Ceram. Int. 2017, 43, 7497–7507. [Google Scholar] [CrossRef]
- Nabhani, N.; Emami, M.; Moghadam, A.T. Application of nanotechnology and nanomaterials in oil and gas industry. In AIP Conference Proceedings 2011; American Institute of Physics: College Park, MD, USA, 2011; Volume 1415, pp. 128–131. [Google Scholar]
- Shirini, F.; Abedini, M. Application of nanocatalysts in multi-component reactions. J. Nanosci. Nanotechnol. 2013, 13, 4838–4860. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Chaichan, M.T.; Kazem, H.A.; Sopian, K.; Ibrahim, A.; Mat, S.; Ruslan, M.H. Comparison study of indoor/outdoor experiments of a photovoltaic thermal PV/T system containing SiC nanofluid as a coolant. Energy 2018, 151, 33–44. [Google Scholar] [CrossRef]
- Choudhary, S.; Sachdeva, A.; Kumar, P. Investigation of the stability of MgO nanofluid and its effect on the thermal performance of flat plate solar collector. Renew. Energy 2020, 147, 1801–1814. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Sopian, K.; Kazem, H.A.; Chaichan, M.T. Evaluation of the electrical performance of a photovoltaic thermal system using nano-enhanced paraffin and nanofluids. Case Stud. Therm. Eng. 2020, 21, 100678. [Google Scholar] [CrossRef]
- Chen, Z.; Shahsavar, A.; Al-Rashed, A.A.; Afrand, M. The impact of sonication and stirring durations on the thermal conductivity of alumina-liquid paraffin nanofluid: An experimental assessment. Powder Technol. 2020, 360, 1134–1142. [Google Scholar] [CrossRef]
- Mohtar, H.; Chesse, P.; Chalet, D. Describing uncertainties encountered during laboratory turbocharger compressor tests. Exp. Tech. 2012, 36, 53–61. [Google Scholar] [CrossRef]
- Madu, K.; Uyaelumuo, A.E. Water based photovoltaic thermal (PVT) collector with spiral flow absorber: An energy and exergy evaluation. Equat. J. Eng. 2018, 51–58. [Google Scholar]
- Zhang, X.; Zhao, X.; Smith, S.; Xu, J.; Yu, X. Review of R&D progress and practical application of the solar photovoltaic/thermal (PV/T) technologies. Renew. Sustain. Energy Rev. 2012, 16, 599–617. [Google Scholar]
- Radziemska, E. Performance analysis of a photovoltaic-thermal integrated system. Int. J. Photo-Energy 2009, 2009, 732093. [Google Scholar] [CrossRef]
- Islam, M.M.; Hasanuzzaman, M.; Rahim, N.A.; Pandey, A.K.; Rawa, M.; Kumar, L. Real time experimental performance investigation of a NePCM based photovoltaic thermal system: An energetic and exergetic approach. Renew. Energy 2021, 172, 71–87. [Google Scholar] [CrossRef]
- Shahsavar, A. Experimental evaluation of energy and exergy performance of a nanofluid-based photovoltaic/thermal system equipped with a sheet-and-sinusoidal serpentine tube collector. J. Clean. Prod. 2021, 287, 125064. [Google Scholar] [CrossRef]
- Rafeek, M.T.M.; Muthu, V.; Athikesavan, M.M.; Sathyamurthy, R.; Kabeel, A.E. Experimental investigation of an active inclined solar panel absorber solar still-energy and exergy analysis. Environ. Sci. Pollut. Res. 2022, 29, 14005–14018. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Chaichan, M.T.; Kazem, H.A.; Sopian, K. Comparative study to use nano-(Al2O3, CuO, and SiC) with water to enhance photovoltaic thermal PV/T collectors. Energy Convers. Manag. 2017, 148, 963–973. [Google Scholar] [CrossRef]
- Kazemi, I.; Sefid, M.; Afrand, M. A novel comparative experimental study on rheological behavior of mono & hybrid nanofluids concerned graphene and silica nano-powders: Characterization, stability and viscosity measurements. Powder Technol. 2020, 366, 216–229. [Google Scholar]
- Karimipour, A.; Ghasemi, S.; Darvanjooghi, M.H.K.; Abdollahi, A. A new correlation for estimating the thermal conductivity and dynamic viscosity of CuO/liquid paraffin nanofluid using neural network method. Int. Commun. Heat Mass Transf. 2018, 92, 90–99. [Google Scholar] [CrossRef]
- Turgut, A.; Tavman, I.; Chirtoc, M.; Schuchmann, H.P.; Sauter, C.; Tavman, S. Thermal conductivity and viscosity measurements of water-based TiO2 nanofluids. Int. J. Thermophys. 2009, 30, 1213–1226. [Google Scholar] [CrossRef]
- Chen, H.; Ding, Y.; He, Y.; Tan, C. Rheological behavior of ethylene glycol based titania nanofluids. Chem. Phys. Lett. 2007, 444, 333–337. [Google Scholar] [CrossRef]
- Prasher, R.; Song, D.; Wang, J.; Phelan, P. Measurements of nanofluid viscosity and its implications for thermal applications. Appl. Phys. Lett. 2006, 89, 133108. [Google Scholar] [CrossRef]
- Nandan, G. Performance of solar photovoltaic panel using forced convection of water-based CuO nanofluid: An understanding. In IOP Conference Series: Materials Science and Engineering 2019; IOP Publishing: Bristol, UK, 2019; Volume 691, p. 012088. [Google Scholar]
- Teng, T.P.; Hung, Y.H. Estimation and experimental study of the density and specific heat for alumina nanofluid. J. Exp. Nanosci. 2014, 9, 707–718. [Google Scholar] [CrossRef]
- Shoghl, S.N.; Jamali, J.; Moraveji, M.K. Electrical conductivity, viscosity, and density of different nanofluids: An experimental study. Exp. Therm. Fluid Sci. 2016, 74, 339–346. [Google Scholar] [CrossRef]
- Mariano, A.; Pastoriza-Gallego, M.J.; Lugo, L.; Camacho, A.; Canzonieri, S.; Piñeiro, M.M. Thermal conductivity, rheological behavior and density of non-Newtonian ethylene glycol-based SnO2 nanofluids. Fluid Phase Equilibria 2013, 337, 119–124. [Google Scholar] [CrossRef]
- An, W.; Wu, J.; Zhu, T.; Zhu, Q. Experimental investigation of a concentrating PV/T collector with Cu9S5 nanofluid spectral splitting filter. Appl. Energy 2016, 184, 197–206. [Google Scholar] [CrossRef]
- Sardarabadi, M.; Passandideh-Fard, M.; Heris, S.Z. Experimental investigation of the effects of silica/water nanofluid on PV/T (photovoltaic thermal units). Energy 2014, 66, 264–272. [Google Scholar] [CrossRef]
- Al-Shamani, A.N.; Sopian, K.; Mat, S.; Hasan, H.A.; Abed, A.M.; Ruslan, M.H. Experimental studies of rectangular tube absorber photovoltaic thermal collector with various types of nanofluids under the tropical climate conditions. Energy Convers. Manag. 2016, 124, 528–542. [Google Scholar] [CrossRef]
- Khanjari, Y.; Pourfayaz, F.; Kasaeian, A.B. Numerical investigation on using of nanofluid in a water-cooled photovoltaic thermal system. Energy Convers. Manag. 2016, 122, 263–278. [Google Scholar] [CrossRef]
- Hjerrild, N.E.; Mesgari, S.; Crisostomo, F.; Scott, J.A.; Amal, R.; Taylor, R.A. Hybrid PV/T enhancement using selectively absorbing Ag–SiO2/carbon nanofluids. Sol. Energy Mater. Sol. Cells 2016, 147, 281–287. [Google Scholar] [CrossRef]
- Chen, T.; Qi, C.; Tang, J.; Wang, G.; Yan, Y. Numerical and experimental study on optimization of CPU system cooled by nanofluids. Case Stud. Therm. Eng. 2021, 24, 100848. [Google Scholar] [CrossRef]
- Aberoumand, S.; Woodfield, P.; Shabani, B.; Dao, D.V. Advances in electrode and electrolyte improvements in vanadium redox flow batteries with a focus on the nanofluidic electrolyte approach. Phys. Rep. 2020, 881, 1–49. [Google Scholar] [CrossRef]
- Jin, H.; Cheol, I.; Onoe, J. Characteristic stability of bare Au-water nanofluids fabricated by pulsed laser ablation in liquids. Opt. Laser Eng. 2009, 47, 532–538. [Google Scholar] [CrossRef]
- Zhu, D.; Li, X.; Wang, N.; Wang, X.; Gao, J.; Li, H. Dispersion behavior and thermal conductivity characteristics of Al2O3-H2O nanofluids. Curr. Appl. Phys. 2009, 9, 131–139. [Google Scholar] [CrossRef]
- Ding, Z.; Qi, C.; Wang, Y.; Tu, J.; Sun, L.; Wang, C. Preparation and photothermal conversion performance of carbon-silica nanocomposite films for spectrally selective solar absorbers. Sol. Energy Mater. Sol. Cells. 2021, 233, 111391. [Google Scholar] [CrossRef]
- Qi, C.; Li, C.; Li, K.; Han, D. Natural convection of nanofluids in solar energy collectors based on a two-phase lattice Boltzmann model. J. Therm. Anal. Calorim. 2021, 147, 2417–2438. [Google Scholar] [CrossRef]
- Huang, J.; Wang, X.; Long, Q.; Wen, X.; Zhou, Y.; Li, L. Influence of pH on the stability characteristics of nanofluids. In Proceedings of the 2009 Symposium Photonics Optoelectron, SOPO, Wuhan, China, 14–16 August 2009; pp. 2–4. [Google Scholar] [CrossRef]
- Li, X.; Zhu, D.; Wang, X. Evaluation on dispersion behavior of the aqueous copper nano-suspensions. J. Colloid Interface Sci. 2007, 310, 456–463. [Google Scholar] [CrossRef]
- Utomo, A.T.; Poth, H.; Robbins, P.T.; Pacek, A.W. Experimental and theoretical studies of thermal conductivity, viscosity and heat transfer coefficient of titania and alumina nanofluids. Int. J. Heat Mass Transf. 2012, 55, 7772–7781. [Google Scholar] [CrossRef]
- Cacua, K.; Ordoñez, F.; Zapata, C.; Herrera, B.; Pabón, E.; Buitrago-Sierra, R. Surfactant concentration and pH effects on the zeta potential values of alumina nanofluids to inspect stability. Colloids Surf. A Physicochem. Eng. Asp. 2019, 583, 123960. [Google Scholar] [CrossRef]
- Suganthi, K.S.; Rajan, K.S. Temperature induced changes in ZnO–water nanofluid: Zeta potential, size distribution and viscosity profiles. Int. J. Heat Mass Transf. 2012, 55, 7969–7980. [Google Scholar] [CrossRef]
- Shao, X.; Chen, Y.; Mo, S.; Cheng, Z.; Yin, T. Dispersion stability of TiO2-H2O nanofluids containing mixed nanotubes and nano-sheets. Energy Procedia 2015, 75, 2049–2054. [Google Scholar] [CrossRef][Green Version]
- Choudhary, R.; Khurana, D.; Kumar, A.; Subudhi, S. Stability analysis of Al2O3/water nanofluids. J. Exp. Nanosci. 2017, 12, 140–151. [Google Scholar] [CrossRef]
- Chakraborty, S. An investigation on the long-term stability of TiO2 nanofluid. Mater. Today Proc. 2019, 11, 714–718. [Google Scholar] [CrossRef]
- Mahbubul, I.M.; Elcioglu, E.B.; Saidur, R.; Amalina, M.A. Optimization of ultrasonication period for better dispersion and stability of TiO2–water nanofluid. Ultrason. Sonochem. 2017, 37, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Mahbubul, I.M.; Chong, T.H.; Khaleduzzaman, S.S.; Shahrul, I.M.; Saidur, R.; Long, B.D.; Amalina, M.A. Effect of ultrasonication duration on colloidal structure and viscosity of alumina–water nanofluid. Ind. Eng. Chem. Res. 2014, 53, 6677–6684. [Google Scholar] [CrossRef]
- Sadeghi, R.; Etemad, S.G.; Keshavarzi, E.; Haghshenasfard, M. Investigation of alumina nanofluid stability by UV–vis spectrum. Microfluid. Nanofluid. 2015, 18, 1023–1030. [Google Scholar] [CrossRef]
- Dubey, S.; Tiwari, G.N. Analysis of PV/T flat plate water collectors connected in series. Sol. Energy 2009, 83, 1485–1494. [Google Scholar] [CrossRef]
- Huang, B.J.; Lin, T.H.; Hung, W.C.; Sun, F.S. Performance evaluation of solar photovoltaic/thermal systems. Sol. Energy 2001, 70, 443–448. [Google Scholar] [CrossRef]
- Chow, T.T.; Ji, J.; He, W. Photovoltaic thermal collector system for domestic application. J. Sol. Energy Eng. 2007, 129, 205–209. [Google Scholar] [CrossRef]
- He, W.; Chow, T.T.; Ji, J.; Lu, J.P.; Pei, G.; Chan, L.S. Hybrid photovoltaic and thermal solar collector designed for natural circulation of water. Appl. Energy 2006, 83, 199–210. [Google Scholar] [CrossRef]
- Ji, J.; Lu, J.P.; Chow, T.T.; He, W.; Pei, G. A sensitivity study of a hybrid photovoltaic/thermal water-heating system with natural circulation. Appl. Energy 2007, 84, 222–237. [Google Scholar] [CrossRef]
- Gangadevi, R.; Vinayagam, B.K.; Senthilraja, S. Experimental investigations of hybrid PV/Spiral flow thermal collector system performance using Al2O3/water nanofluid. IOP Conf. Ser. Mater. Sci. Eng. 2017, 197, 012041. [Google Scholar] [CrossRef]
- Al-Waeli, A.H.; Kazem, H.A.; Chaichan, M.T.; Sopian, K. Experimental investigation of using nano-PCM/nanofluid on a photovoltaic thermal system (PVT): Technical and economic study. Therm. Sci. Eng. Prog. 2019, 11, 213–230. [Google Scholar] [CrossRef]

| Module Type | Polycrystalline | Monocrystalline |
|---|---|---|
| Company name | A star | Nuru Tech Fzco |
| Peak power (Pmax) | 100 W | 100 W |
| Open circuit voltage (Voc) | 22.5 V | 22.6 V |
| Short circuit current (Isc) | 5.81 A | 5.76 A |
| Maximum power voltage (Vmp) | 18.0 V | 17.96 V |
| Maximum power current (Imp) | 5.56 A | 5.57 A |
| Power tolerance | ±3% | ±3% |
| Dimension (mm) | 1012 × 660 × 30 | 1010 × 660 × 34 |
| January | February | March | April | May | June | July | August | September | October | November | December | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Max. Temp (°C) | 15.5 | 17 | 21 | 30 | 37 | 41 | 45 | 43 | 40 | 33 | 25 | 17 |
| Min. Temp. (°C) | 5 | 6 | 8 | 14 | 20 | 22 | 25 | 24 | 22.5 | 16 | 10 | 5 |
| Shinning hours (H) | 196 | 200 | 248 | 256 | 300 | 355 | 350 | 360 | 310 | 285 | 212 | 200 |
| Precipitation (mm) | 23 | 19 | 22 | 10 | 3 | 0 | 0 | 0 | 0 | 3 | 13 | 23 |
| Rainy days | 6 | 6 | 7 | 5 | 3 | 1 | 0 | 0 | 3 | 4 | 7 | 8 |
| Humidity (%) | 70 | 60 | 54 | 49 | 32 | 21 | 20 | 22 | 27 | 38 | 56 | 68 |
| Wind speed (m/s) | 1 | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 1 | 1 | 1 | 1 |
| Particle | ρp (kg/m3) | kp (W/m·°C) | Purity | dp (nm) | Color | Source |
|---|---|---|---|---|---|---|
| Fe2O3 | 5240 | 30 | 99.0% | 20–40 | Red nanopowder | Sky Spring Nanomaterials Inc. (Houston, TX, USA) |
| Base fluid | ρf (kg/m3) | kf (W/m·°C) | cpf (J/kg·°C) | μf (nm) | ||
| De-ionized water (DIW) (75%) + Ethaline glycol (25%) | 1007.1 | 0.6117 | 4773 | 0.00997 | Steam Lap. + Merck KGaA, Darmstadt, Germany | |
| No. | Measured Parameter | Measuring Devise | Uncertainty (%) |
|---|---|---|---|
| 1 | Voltage and current | Multi-meter | 0.9 |
| 2 | Coolants flow rate | Flowmeter | 0.34 |
| 3 | Thermocouples | Temperature | 0.27 |
| 4 | Irradiance | Solar radiation intensity meter | 0.98 |
| 5 | Nanoparticle mass fraction weight | Sensitive weight | 0.001 |
| 6 | Nanofluids density | Density tester | 0.28 |
| 7 | Viscosity | Brookfield Programmer Viscometer (Model: LVDV-III Ultra-programmable) | 0.3 |
| 8 | Thermal conductivity and capacity | Hot desk Tps 500 | 1.2 |
| No. | Parameter | Equation | Parameters | Ref. |
|---|---|---|---|---|
| (1) | Thermal efficiency | , | [45] | |
| (2) | Useful gained heat | , | [45] | |
| (3) | Electrical power | , | [46] | |
| (4) | Electrical efficiency | , , , | [47] | |
| (5) | Total PV/T system efficiency | , , | [47] | |
| (6) | Primary energy saving efficiency | , , , | [46] |
| No. | Parameter | Equation | Parameter | Ref. |
|---|---|---|---|---|
| 1 | The general exergy balance | , , | [48,49,50] | |
| 2 | The general exergy balance | , , , , | [48,49,50] | |
| 3 | The input exergy | , , , , , | [48,49,50] | |
| 4 | The thermal exergy | , , , | [48,49,50] | |
| 5 | The PV exergy | , , , , | [48,49,50] | |
| 6 | The photovoltaic thermal exergy | , , | [48,49,50] | |
| 7 | The exergy destruction or irreversibility | , , | [48,49,50] | |
| 8 | The exergy efficiency | , , | [48,49,50] |
| Ref. No. | Electrical Efficiency | Thermal Efficiency | Total Efficiency | Cooling Fluid | Collector Design |
|---|---|---|---|---|---|
| [83] | 9.5 | 50 | 59.5 | Water | Flat plate |
| [84] | 9 | 38 | 47 | Water | Corrugated polycarbonate panel |
| [85] | 11 | 51 | 62 | Water | Aluminum-alloy flat-box |
| [86] | - | - | 64.9 | Water | Flat-box absorber |
| [87] | 9.87 | 40 | 49.87 | Water | Flat-box Al-alloy absorber plate |
| [88] | 13 | 45 | 58 | Nano-Al2O3-Water | Spiral flow absorber |
| [25] | 17.2 | 54.8 | 72 | MWCNT-water | Copper sheet and tube |
| [63] | 9.9 | 54.28 | 64.18 | Nano-SiC—water | Direct-flow configuration |
| [89] | 16 | 70 | 86 | Nano-SiC—water +Nano-paraffin | Copper tubes in heat storage tank |
| Current study (Monocrystalline) | 13.3 | 59 | 72.3 | Nano-Fe2O3-water-EG | Spiral flow absorber |
| Current study (Polycrystalline) | 13.75 | 63 | 76.75 | Nano-Fe2O3-water-EG | Spiral flow absorber |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Ezzi, A.; Chaichan, M.T.; Majdi, H.S.; Al-Waeli, A.H.A.; Kazem, H.A.; Sopian, K.; Fayad, M.A.; Dhahad, H.A.; Yusaf, T. Nano-Iron Oxide-Ethylene Glycol-Water Nanofluid Based Photovoltaic Thermal (PV/T) System with Spiral Flow Absorber: An Energy and Exergy Analysis. Energies 2022, 15, 3870. https://doi.org/10.3390/en15113870
Al Ezzi A, Chaichan MT, Majdi HS, Al-Waeli AHA, Kazem HA, Sopian K, Fayad MA, Dhahad HA, Yusaf T. Nano-Iron Oxide-Ethylene Glycol-Water Nanofluid Based Photovoltaic Thermal (PV/T) System with Spiral Flow Absorber: An Energy and Exergy Analysis. Energies. 2022; 15(11):3870. https://doi.org/10.3390/en15113870
Chicago/Turabian StyleAl Ezzi, Amged, Miqdam T. Chaichan, Hasan S. Majdi, Ali H. A. Al-Waeli, Hussein A. Kazem, Kamaruzzaman Sopian, Mohammed A. Fayad, Hayder A. Dhahad, and Talal Yusaf. 2022. "Nano-Iron Oxide-Ethylene Glycol-Water Nanofluid Based Photovoltaic Thermal (PV/T) System with Spiral Flow Absorber: An Energy and Exergy Analysis" Energies 15, no. 11: 3870. https://doi.org/10.3390/en15113870
APA StyleAl Ezzi, A., Chaichan, M. T., Majdi, H. S., Al-Waeli, A. H. A., Kazem, H. A., Sopian, K., Fayad, M. A., Dhahad, H. A., & Yusaf, T. (2022). Nano-Iron Oxide-Ethylene Glycol-Water Nanofluid Based Photovoltaic Thermal (PV/T) System with Spiral Flow Absorber: An Energy and Exergy Analysis. Energies, 15(11), 3870. https://doi.org/10.3390/en15113870










