Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Kraft Pulp
2.2. Enzymatic Hydrolysis
2.3. Microorganisms
2.4. Pre-Inoculum and Inoculum
2.5. Fermentation
2.6. Analytical Methods
2.7. Calculations
3. Results and Discussion
3.1. Chemical Characterization of the Bark Kraft Pulp
3.2. Enzymatic Hydrolysis
3.3. Fermentation to Bioethanol
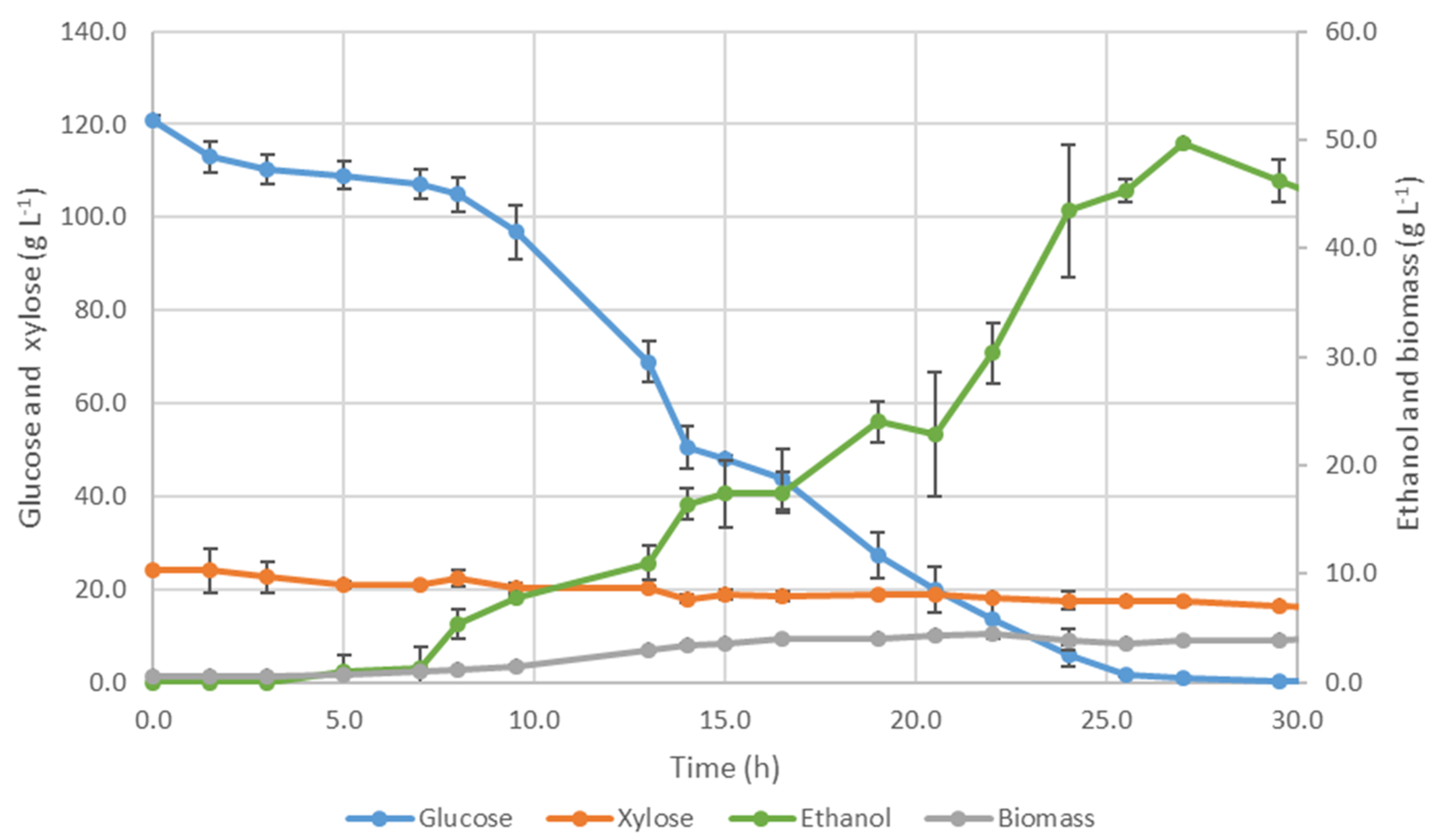
3.3.1. Fermentation by S. cerevisiae
| Feedstock | Yeast Strain | Operation Conditions | Conf | [EtOH]max (g·L−1) | Prod (g·L−1·h−1) | Yethanol/substrate | Ref. |
|---|---|---|---|---|---|---|---|
| E. globulus unbleached kraft pulp | S. cerevisiae ATCC 24860 | 28 c 150 rpm 100 mL | SHF | 19.81 ± 0.15 | 2.01 ± 0.01 | 88.3 ± 1.7% theoretical | [39] |
| E. globulus kraft pulp | S. cerevisiae ATCC 26602 | 38 °C 150 rpm 100 mL | Fed-batch SSF | 55.3 ± 0.3 | 0.70 | 64.9% theoretical | [49] |
| E. globulus kraft pulp | S. cerevisiae IR2T9 | 40 °C 150 rpm 96 h | SSF | 30.0–38.0 | N.S. | 0.168–0.202 g gdry wood−1 | [45] |
| Jabon bleached kraft pulp | S. cerevisiae | 40 °C 72 h | SSF | N.S. | N.S. | 2.89–16.39% (v/wd.w. pulp) | [46] |
| Seven kraft pulps | S. cerevisiae | 25 °C 48 h 15 L | SHF | N.S. | N.S. | 0.11–0.20 g gd.w. wood−1 | [47] |
| E. grandis sawdust | S. cerevisiae PE-2 | 35 °C 150 rpm 100 mL 48 h | PS-SSF | 57.0 | 1.20 | 81% theoretical | [34] |
| Pinus radiata chips bleached kraft pulp | S. cerevisiae IR2-9a | 40 °C 150 rpm 72 h | SSF | 28 | 0.39 | 62% theoretical | [50] |
| E. globulus unbleached kraft pulp | S. cerevisiae D5A | N.S. | SSF | 5.67 | 0.032 | 0.04227 g gdry wood−1 | [51] |
| Industrial E. globulus bark unbleached kraft pulp | S. cerevisiae ATCC 24860 | 28 °C 150 rpm 100 mL | SHF | 49.7 ± 0.1 | 1.84 ± 0.00 | 76.7 ± 1.1% theoretical | This work |
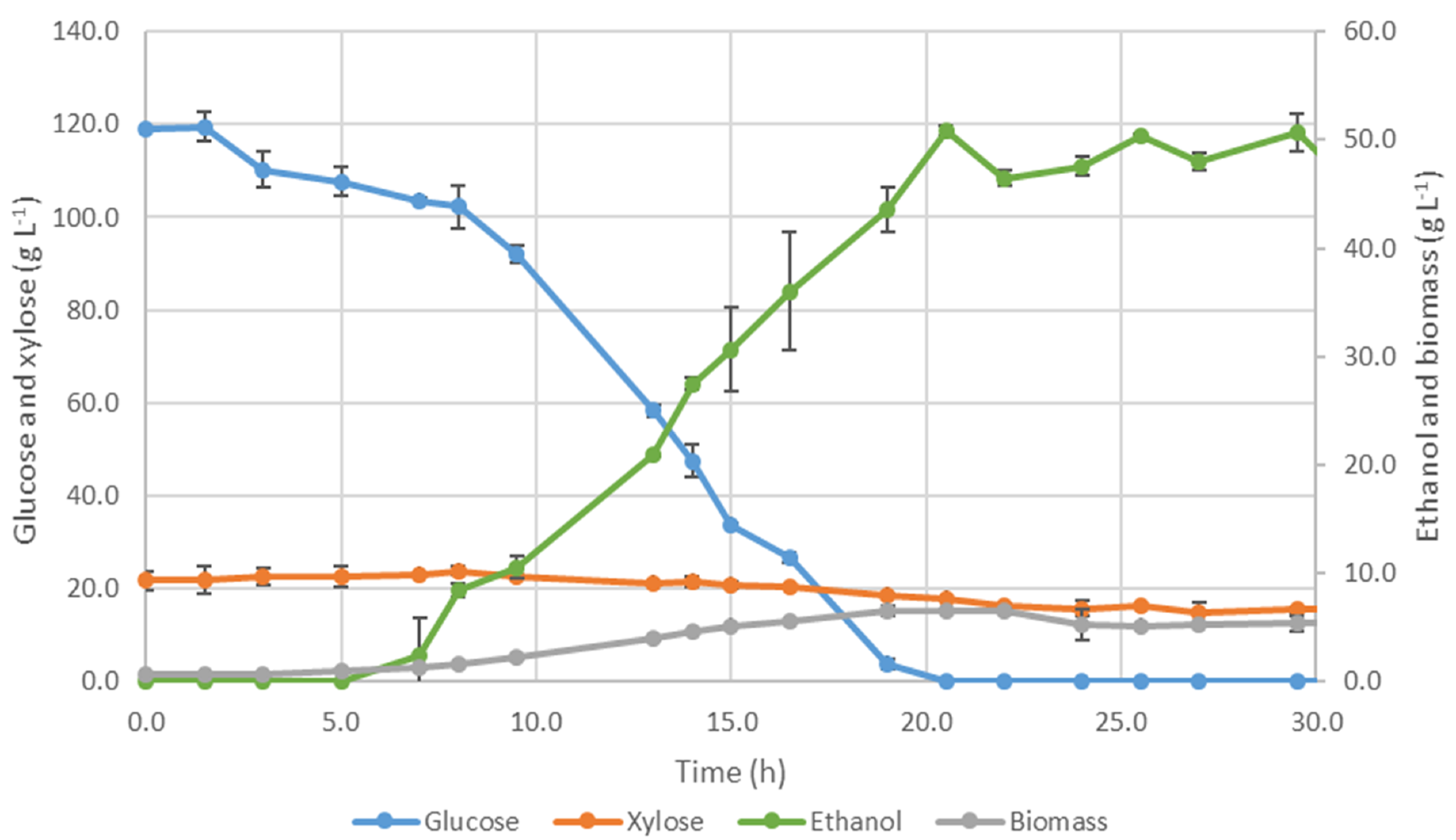
3.3.2. Fermentation by Ethanol Red®
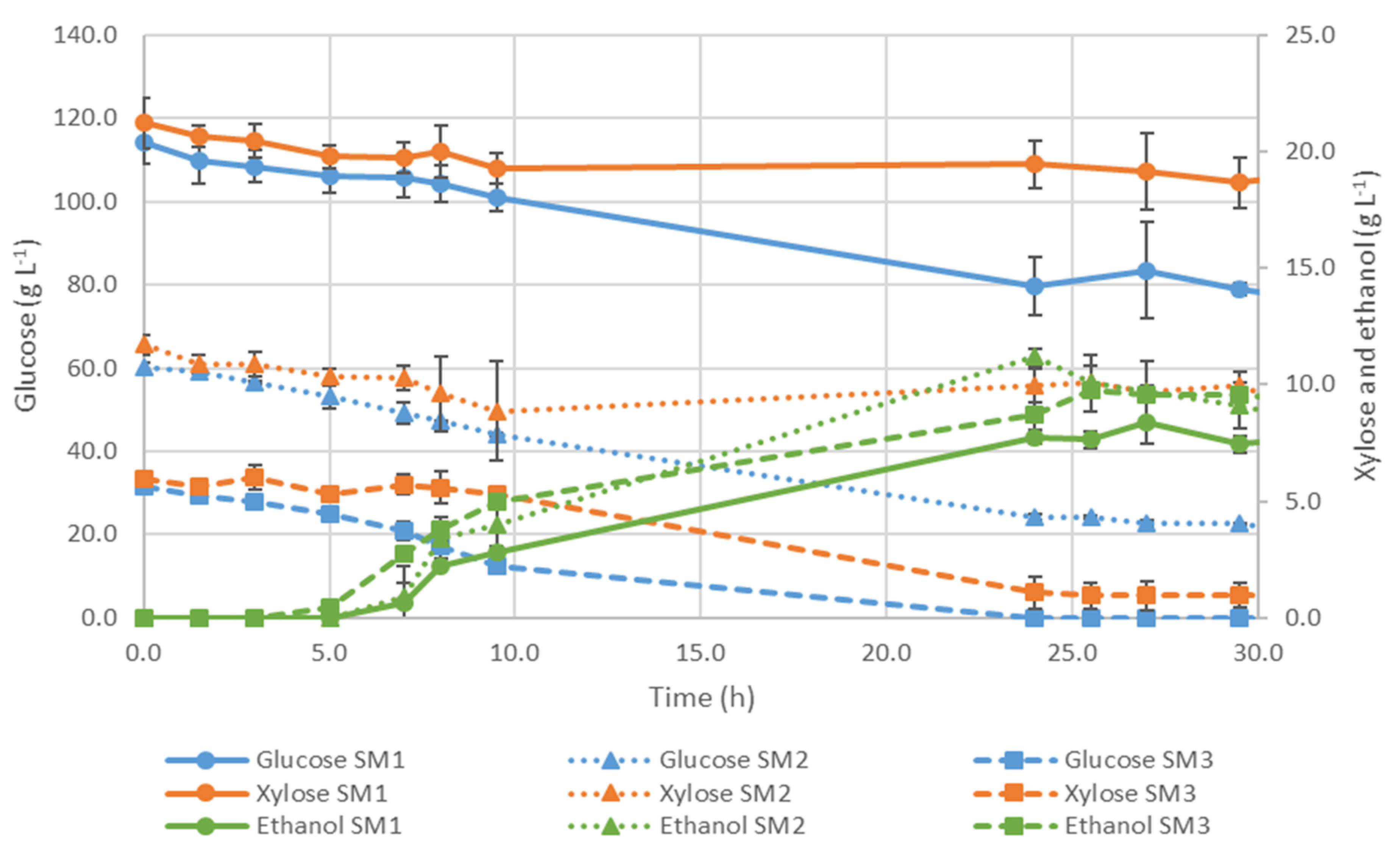
3.3.3. Fermentation Using Simultaneous Co-Culture of Ethanol Red® and S. passalidarum
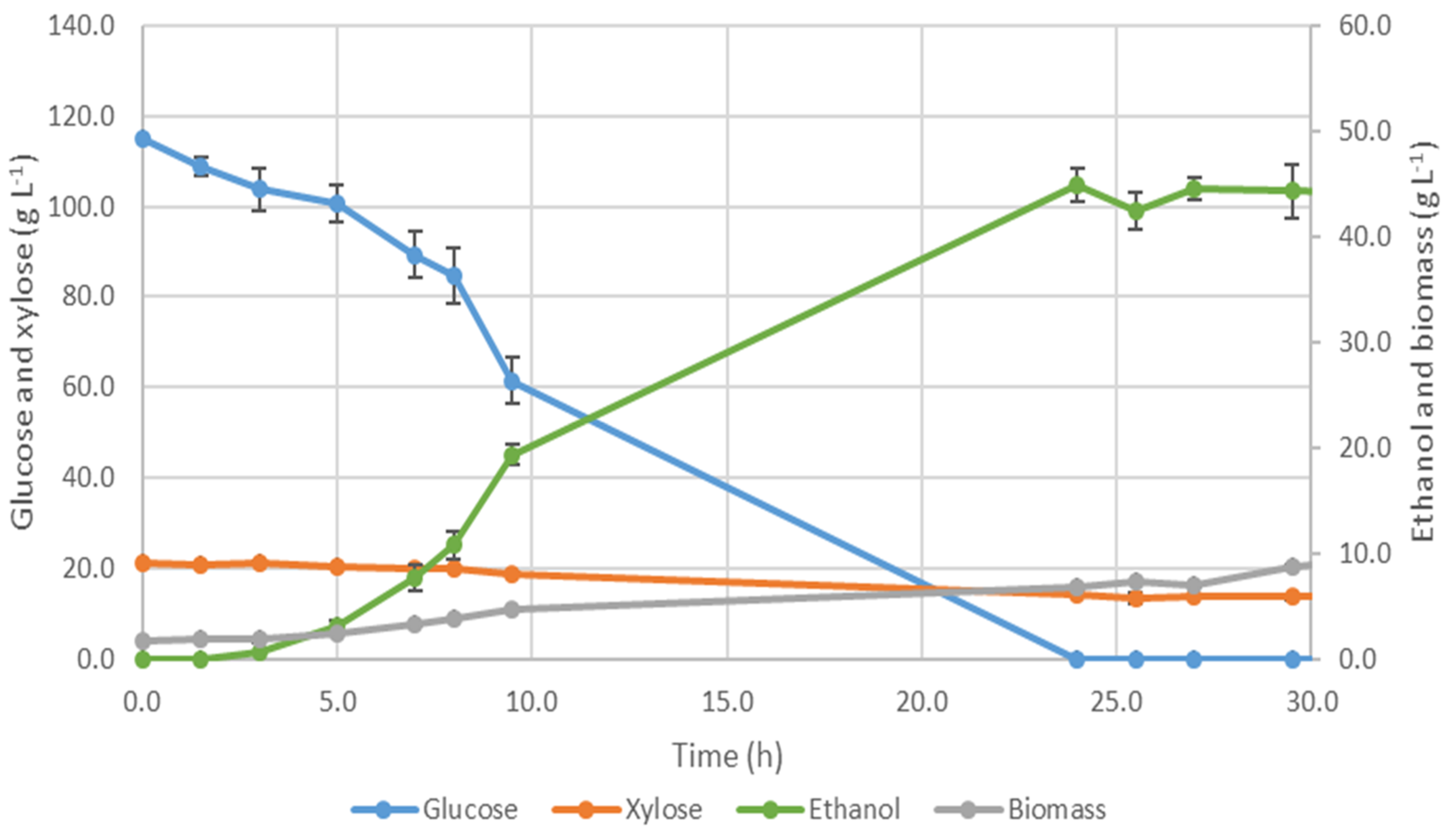
3.3.4. Fermentation Using S. passalidarum Monoculture
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Limayem, A.; Ricke, S.C. Lignocellulosic biomass for bioethanol production: Current perspectives, potential issues and future prospects. Prog. Energy Combust. Sci. 2012, 38, 449–467. [Google Scholar] [CrossRef]
- Resquin, F.; Barrichelo, L.E.G.; da Silva, F.G.; Brito, J.O.; Sansigolo, C.A. Wood quality for kraft pulping of Eucalyptus globulus origins planted in Uruguay. Sci. For. Sci. 2006, 72, 57–66. [Google Scholar]
- Hassan, S.S.; Williams, G.A.; Jaiswal, A.K. Lignocellulosic Biorefineries in Europe: Current State and Prospects. Trends Biotechnol. 2019, 37, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Espí, E.; Ribas, Í.; Diáz, C.; Sastrón, Ó. Feedstocks for Advanced Biofuels. In Sustainable Mobility; Llamas, B., Romero, M.F.O., Sillero, E., Eds.; IntechOpen: London, UK, 2020; pp. 1–22. ISBN 9781626239777. [Google Scholar]
- Liguori, R.; Faraco, V. Biological processes for advancing lignocellulosic waste biorefinery by advocating circular economy. Bioresour. Technol. 2016, 215, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Anwar, Z.; Gulfraz, M.; Irshad, M. Agro-industrial lignocellulosic biomass a key to unlock the future bio-energy: A brief review. J. Radiat. Res. Appl. Sci. 2014, 7, 163–173. [Google Scholar] [CrossRef]
- Branco, R.; Serafim, L.; Xavier, A. Second Generation Bioethanol Production: On the Use of Pulp and Paper Industry Wastes as Feedstock. Fermentation 2019, 5, 4. [Google Scholar] [CrossRef]
- Stoklosa, R.J.; Hodge, D.B. Integration of (Hemi)-Cellulosic Biofuels Technologies with Chemical Pulp Production. In Biorefineries: Integrated Biochemical Processes for Liquid Biofuels; Qureshi, N., Hodge, D., Vertes, A., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2014; pp. 73–100. ISBN 9780444595041. [Google Scholar]
- Celpa: Associação da Indústria Papeleira. Boletim Estatístico 2017: Indústria Papeleira Portuguesa. Available online: http://www.celpa.pt/wp-content/uploads/2018/10/Boletim_WEB-2.pdf (accessed on 26 February 2021).
- Neiva, D.M.; Araújo, S.; Gominho, J.; Carneiro, A.C.; Pereira, H. Potential of Eucalyptus globulus industrial bark as a biorefinery feedstock: Chemical and fuel characterization. Ind. Crop. Prod. 2018, 123, 262–270. [Google Scholar] [CrossRef]
- Rodrigues, A.E.; Pinto, P.C.O.R.; Barreiro, M.F.; Costa, C.A.E.; Mota, M.I.F.; Fernandes, I. Chemical Pulp Mills as Biorefineries. In An Integrated Approach for Added-Value Products from Lignocellulosic Biorefineries: Vanillin, Syringaldehyde, Polyphenols, and Polyurethane; Springer Nature Switzerland AG: Cham, Switzerland, 2018; pp. 1–51. ISBN 9783319993126. [Google Scholar]
- Pinto, F.; Gominho, J.; André, R.N.; Gonçalves, D.; Miranda, M.; Varela, F.; Neves, D.; Santos, J.; Lourenço, A.; Pereira, H. Improvement of gasification performance of Eucalyptus globulus stumps with torrefaction and densification pre-treatments. Fuel 2017, 206, 289–299. [Google Scholar] [CrossRef]
- Souza, A.G.; Lima, G.F.; Rodrigues, R.C.L.B.; Cesarino, I.; Leão, A.L.; Rosa, D.S. A New Approach for Conversion of Eucalyptus Lignocellulosic Biomass into Cellulose Nanostructures: A Method that Can Be Applied in Industry. J. Nat. Fibers 2019, 1–11. [Google Scholar] [CrossRef]
- Vea, E.B.; Romeo, D.; Thomsen, M. Biowaste Valorisation in a Future Circular Bioeconomy. In Proceedings of the 25th CIRP Life Cycle Engineering (LCE) Conference, Copenhagen, Denmark, 30 April–2 May 2018; Volume 69, pp. 591–596. [Google Scholar]
- Celpa—Associação da Indústria Papeleira. Boletim Estatístico Indústria Papeleira Portuguesa 2019. Lisboa, 2020. Available online: http://www.celpa.pt/category/boletins-estatisticos/ (accessed on 26 February 2021).
- Faubert, P.; Barnabé, S.; Bouchard, S.; Côté, R.; Villeneuve, C. Pulp and paper mill sludge management practices: What are the challenges to assess the impacts on greenhouse gas emissions? Resour. Conserv. Recycl. 2016, 108, 107–133. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Freire, C.S.R.; Domingues, M.R.M.; Silvestre, A.J.D.; Neto, C.P. Characterization of phenolic components in polar extracts of Eucalyptus globulus labill. Bark by high-performance liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2011, 59, 9386–9393. [Google Scholar] [CrossRef] [PubMed]
- Romaní, A.; Michelin, M.; Domingues, L.; Teixeira, J.A. Valorization of wastes from agrofood and pulp and paper industries within the biorefinery concept: Southwestern Europe scenario. In Waste Biorefinery: Potential and Perspectives; Bhaskar, T., Pandey, A., Mohan, S.V., Lee, D.-J., Khanal, S.K., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2018; pp. 487–504. ISBN 9780444639936. [Google Scholar]
- Domingues, R.M.A.; Sousa, G.D.A.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P. Eucalyptus globulus biomass residues from pulping industry as a source of high value triterpenic compounds. Ind. Crop. Prod. 2010, 31, 65–70. [Google Scholar] [CrossRef]
- Ek, M.; Gellerstedt, G. Pulping Chemistry and Technology. In Pulp and Paper Chemistry and Technology; Ek, M., Gellerstedt, G., Henriksson, G., Eds.; Walter de Gruyter: Berlin, Germany, 2009; Volume 2. [Google Scholar]
- Ressel, J.B. Wood Yard Operations. In Handbook of Pulp; Sixta, H., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; Volume 1, pp. 69–107. ISBN 3527309993. [Google Scholar]
- Neiva, D.M.; Costa, R.A.; Gominho, J.; Ferreira-Dias, S.; Pereira, H. Fractionation and valorization of industrial bark residues by autohydrolysis and enzymatic saccharification. Bioresour. Technol. Reports 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Isikgor, F.H.; Becer, C.R. Lignocellulosic biomass: A sustainable platform for the production of bio-based chemicals and polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef]
- Guerrero, M. Portugal Biofuels Standing Report 2015; USDA Foreign Agricultural Service: Global Agricultural Information Network: Madrid, Spain, 2015.
- Neiva, D.M.; Luís, Â.; Gominho, J.; Domingues, F.; Duarte, A.P.; Pereira, H. Bark residues valorization potential regarding antioxidant and antimicrobial extracts. Wood Sci. Technol. 2020, 54, 559–585. [Google Scholar] [CrossRef]
- Kemppainen, K.; Inkinen, J.; Uusitalo, J.; Nakari-Setälä, T.; Siika-aho, M. Hot water extraction and steam explosion as pretreatments for ethanol production from spruce bark. Bioresour. Technol. 2012, 117, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Mota, M.I.F.; Pinto, P.C.O.R.; Novo, C.C.; Sousa, G.D.A.; Guerreiro, O.; Guerra, Â.; Duarte, M.F.P.; Rodrigues, A.E. Eucalyptus globulus bark as a source of polyphenolic compounds with biological activity. O Pap. 2013, 74, 57–64. [Google Scholar]
- Koutinas, A.A.; Vlysidis, A.; Pleissner, D.; Kopsahelis, N.; Lopez Garcia, I.; Kookos, I.K.; Papanikolaou, S.; Kwan, T.H.; Lin, C.S.K. Valorization of industrial waste and by-product streams via fermentation for the production of chemicals and biopolymers. Chem. Soc. Rev. 2014, 43, 2587–2627. [Google Scholar] [CrossRef]
- Gomes, D.G.; Michelin, M.; Romaní, A.; Domingues, L.; Teixeira, J.A. Co-production of biofuels and value-added compounds from industrial Eucalyptus globulus bark residues using hydrothermal treatment. Fuel 2021, 285, 119265. [Google Scholar] [CrossRef]
- Jönsson, L.J.; Martín, C. Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 2016, 199, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.A.; Taherzadeh, M.J. Improving the economy of lignocellulose-based biorefineries with organosolv pretreatment. Bioresour. Technol. 2020, 299, 122695. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Bagley, D.M.; Leung, K.T.; Liss, S.N.; Liao, B.Q. Recent advances in membrane technologies for biorefining and bioenergy production. Biotechnol. Adv. 2012, 30, 817–858. [Google Scholar] [CrossRef]
- Kang, Q.; Appels, L.; Tan, T.; Dewil, R. Bioethanol from lignocellulosic biomass: Current findings determine research priorities. Sci. World J. 2014. [Google Scholar] [CrossRef]
- Guigou, M.; Cabrera, M.N.; Vique, M.; Bariani, M.; Guarino, J.; Ferrari, M.D.; Lareo, C. Combined pretreatments of eucalyptus sawdust for ethanol production within a biorefinery approach. Biomass Convers. Biorefin. 2019, 9, 293–304. [Google Scholar] [CrossRef]
- Chen, H.Z.; Liu, Z.H. Enzymatic hydrolysis of lignocellulosic biomass from low to high solids loading. Eng. Life Sci. 2017, 17, 489–499. [Google Scholar] [CrossRef]
- Gomes, A.C.; Moysés, D.N.; Santa Anna, L.M.M.; Castro, A.M. Fed-batch strategies for saccharification of pilot-scale mild-acid and alkali pretreated sugarcane bagasse: Effects of solid loading and surfactant addition. Ind. Crop. Prod. 2018, 119, 283–289. [Google Scholar] [CrossRef]
- He, L.; Han, Q.; Jameel, H.; Chang, H.M.; Phillips, R.; Wang, Z. Comparison of One-Stage Batch and Fed-Batch Enzymatic Hydrolysis of Pretreated Hardwood for the Production of Biosugar. Appl. Biochem. Biotechnol. 2018, 184, 1441–1452. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Yunyun, L.; Jingliang, X.; Zhanhong, Y.; Wei, Q.; Xinshu, Z.; MinChao, H. High solid and low enzyme loading based saccharification of agricultural biomass. BioResources 2012, 7, 345–353. [Google Scholar] [CrossRef]
- Branco, R.H.R.; Amândio, M.S.T.; Serafim, L.S.; Xavier, A.M.R.B. Ethanol production from hydrolyzed kraft pulp by mono- and co-cultures of yeasts: The challenge of C6 and C5 sugars consumption. Energies 2020, 13, 744. [Google Scholar] [CrossRef]
- Radler, F.; Schütz, H. Glycerol production of various strains of Saccharomyces. Am. J. Enol. Vitic. 1982, 33, 36–40. [Google Scholar]
- Scanes, K.T.; Hohmann, S.; Prior, B.A. Glycerol Production by the Yeast Saccharomyces cerevisiae and its Relevance to Wine: A Review. S. Afr. J. Enol. Vitic. 1998, 19, 17–24. [Google Scholar] [CrossRef]
- Zhang, L.; Tang, Y.; Guo, Z.; Ding, Z.; Shi, G. Improving the ethanol yield by reducing glycerol formation using cofactor regulation in Saccharomyces cerevisiae. Biotechnol. Lett. 2011, 33, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.H.; Chang, K.S.; Huang, C.W.; Hsu, C.L.; Jang, H. Der Comparison of batch and fed-batch fermentations using corncob hydrolysate for bioethanol production. Fuel 2012, 97, 166–173. [Google Scholar] [CrossRef]
- Coote, N.; Kirsop, B.H. Factors responsible for the decrease in pH during beer fermentations. J. Inst. Brew. 1976, 82, 149–153. [Google Scholar] [CrossRef]
- Monrroy, M.; Garcia, J.-R.; Mendonça, R.T.; Baeza, J.; Freer, J. Kraft pulping of Eucalyptus globulus as a pretreatment for bioethanol production by simultaneous saccharification and fermentation. J. Chil. Chem. Soc. 2012, 57, 1113–1117. [Google Scholar] [CrossRef]
- Wistara, N.J.; Pelawi, R.; Fatriasari, W. The Effect of Lignin Content and Freeness of Pulp on the Bioethanol Productivity of Jabon Wood. Waste Biomass Valorization 2016, 7, 1141–1146. [Google Scholar] [CrossRef]
- Przybysz Buzała, K.; Kalinowska, H.; Małachowska, E.; Przybysz, P. The utility of selected kraft hardwood and softwood pulps for fuel ethanol production. Ind. Crops Prod. 2017, 108, 824–830. [Google Scholar] [CrossRef]
- Frankó, B.; Galbe, M.; Wallberg, O. Bioethanol production from forestry residues: A comparative techno-economic analysis. Appl. Energy 2016, 184, 727–736. [Google Scholar] [CrossRef]
- Mendes, C.V.T.; Rocha, J.M.S.; Menezes, F.F.; Carvalho, M.G.V.S. Batch and fed-batch simultaneous saccharification and fermentation of primary sludge from pulp and paper mills. Environ. Technol. 2017, 38, 1498–1506. [Google Scholar] [CrossRef] [PubMed]
- Edgardo, A.; Carolina, P.; Manuel, R.; Juanita, F.; Baeza, J. Selection of thermotolerant yeast strains Saccharomyces cerevisiae for bioethanol production. Enzyme Microb. Technol. 2008, 43, 120–123. [Google Scholar] [CrossRef]
- Ko, C.H.; Wang, Y.N.; Chang, F.C.; Chen, J.J.; Chen, W.H.; Hwang, W.S. Potentials of lignocellulosic bioethanols produced from hardwood in Taiwan. Energy 2012, 44, 329–334. [Google Scholar] [CrossRef]
- Kossatz, H.L.; Rose, S.H.; Viljoen-Bloom, M.; van Zyl, W.H. Production of ethanol from steam exploded triticale straw in a simultaneous saccharification and fermentation process. Process. Biochem. 2017, 53, 10–16. [Google Scholar] [CrossRef]
- Demeke, M.M.; Dietz, H.; Li, Y.; Foulquié-Moreno, M.R.; Mutturi, S.; Deprez, S.; Den Abt, T.; Bonini, B.M.; Liden, G.; Dumortier, F.; et al. Development of a D-xylose fermenting and inhibitor tolerant industrial Saccharomyces cerevisiae strain with high performance in lignocellulose hydrolysates using metabolic and evolutionary engineering. Biotechnol. Biofuels 2013, 6, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Leaf by Lesaffre Industrial Ethanol. Available online: https://lesaffreadvancedfermentations.com/ethanol_yeast/ (accessed on 2 February 2021).
- Frankó, B.; Galbe, M.; Wallberg, O. Influence of bark on fuel ethanol production from steam-pretreated spruce. Biotechnol. Biofuels 2015, 8, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Collograi, K.C.; da Costa, A.C.; Ienczak, J.L. Effect of contamination with Lactobacillus fermentum I2 on ethanol production by Spathaspora passalidarum. Appl. Microbiol. Biotechnol. 2019, 103, 5039–5050. [Google Scholar] [CrossRef]
- Farias, D.; Maugeri-Filho, F. Sequential fed batch extractive fermentation for enhanced bioethanol production using recycled Spathaspora passalidarum and mixed sugar composition. Fuel 2020, 288, 119673. [Google Scholar] [CrossRef]
- Bonan, C.I.D.G.; Biazi, L.E.; Dionísio, S.R.; Soares, L.B.; Tramontina, R.; Sousa, A.S.; de Oliveira Filho, C.A.; Costa, A.C.; Ienczak, J.L. Redox potential as a key parameter for monitoring and optimization of xylose fermentation with yeast Spathaspora passalidarum under limited-oxygen conditions. Bioprocess. Biosyst. Eng. 2020, 43, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Farias, D.; Maugeri Filho, F. Co-culture strategy for improved 2G bioethanol production using a mixture of sugarcane molasses and bagasse hydrolysate as substrate. Biochem. Eng. J. 2019, 147, 29–38. [Google Scholar] [CrossRef]
- Chen, Y. Development and application of co-culture for ethanol production by co-fermentation of glucose and xylose: A systematic review. J. Ind. Microbiol. Biotechnol. 2011, 38, 581–597. [Google Scholar] [CrossRef]
- Neitzel, T.; Lima, C.S.; Biazi, L.E.; Collograi, K.C.; Carvalho da Costa, A.; Vieira dos Santos, L.; Ienczak, J.L. Impact of the Melle-Boinot process on the enhancement of second-generation ethanol production by Spathaspora passalidarum. Renew. Energy 2020, 160, 1206–1216. [Google Scholar] [CrossRef]
- Su, Y.K.; Willis, L.B.; Jeffries, T.W. Effects of aeration on growth, ethanol and polyol accumulation by Spathaspora passalidarum NRRL Y-27907 and Scheffersomyces stipitis NRRL Y-7124. Biotechnol. Bioeng. 2015, 112, 457–469. [Google Scholar] [CrossRef]
- Fátima Rodrigues Souza, R.; Dutra, E.D.; Leite, F.C.B.; Cadete, R.M.; Rosa, C.A.; Stambuk, B.U.; Stamford, T.L.M.; Morais, M.A. Production of ethanol fuel from enzyme-treated sugarcane bagasse hydrolysate using d-xylose-fermenting wild yeast isolated from Brazilian biomes. 3 Biotech 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Hou, X.; Yao, S. Improved inhibitor tolerance in xylose-fermenting yeast Spathaspora passalidarum by mutagenesis and protoplast fusion. Appl. Microbiol. Biotechnol. 2012, 93, 2591–2601. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Li, Y.; Zhao, X.; Pei, X.; Yuan, W.; Bai, F.; Jiang, Y. The production of ethanol from lignocellulosic biomass by Kluyveromyces marxianus CICC 1727-5 and Spathaspora passalidarum ATCC MYA-4345. Appl. Microbiol. Biotechnol. 2019, 103, 2845–2855. [Google Scholar] [CrossRef]
- Nakanishi, S.C.; Soares, L.B.; Biazi, L.E.; Nascimento, V.M.; Costa, A.C.; Rocha, G.J.M.; Ienczak, J.L. Fermentation strategy for second generation ethanol production from sugarcane bagasse hydrolyzate by Spathaspora passalidarum and Scheffersomyces stipitis. Biotechnol. Bioeng. 2017, 114, 2211–2221. [Google Scholar] [CrossRef]
- Morales, P.; Gentina, J.C.; Aroca, G.; Mussatto, S.I. Development of an acetic acid tolerant Spathaspora passalidarum strain through evolutionary engineering with resistance to inhibitors compounds of autohydrolysate of Eucalyptus globulus. Ind. Crop. Prod. 2017, 106, 5–11. [Google Scholar] [CrossRef]
- Soares, L.B.; Bonan, C.I.D.G.; Biazi, L.E.; Dionísio, S.R.; Bonatelli, M.L.; Andrade, A.L.D.; Renzano, E.C.; Costa, A.C.; Ienczak, J.L. Investigation of hemicellulosic hydrolysate inhibitor resistance and fermentation strategies to overcome inhibition in non-saccharomyces species. Biomass Bioenergy 2020, 137, 105549. [Google Scholar] [CrossRef]
- Su, Y.K.; Willis, L.B.; Rehmann, L.; Smith, D.R.; Jeffries, T.W. Spathaspora passalidarum selected for resistance to AFEX hydrolysate shows decreased cell yield. FEMS Yeast Res. 2018, 18. [Google Scholar] [CrossRef]
- Yu, H.; Guo, J.; Chen, Y.; Fu, G.; Li, B.; Guo, X.; Xiao, D. Efficient utilization of hemicellulose and cellulose in alkali liquor-pretreated corncob for bioethanol production at high solid loading by Spathaspora passalidarum U1-58. Bioresour. Technol. 2017, 232, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Rodrussamee, N.; Sattayawat, P.; Yamada, M. Highly efficient conversion of xylose to ethanol without glucose repression by newly isolated thermotolerant Spathaspora passalidarum CMUWF1-2. BMC Microbiol. 2018, 18, 73. [Google Scholar] [CrossRef] [PubMed]
- Hou, X. Anaerobic xylose fermentation by Spathaspora passalidarum. Appl. Microbiol. Biotechnol. 2012, 94, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Long, T.M.; Su, Y.K.; Headman, J.; Higbee, A.; Willis, L.B.; Jeffries, T.W. Cofermentation of glucose, xylose, and cellobiose by the beetle-associated yeast Spathaspora passalidarum. Appl. Environ. Microbiol. 2012, 78, 5492–5500. [Google Scholar] [CrossRef] [PubMed]
- Tolosa, L.; Kostov, Y.; Harms, P.; Rao, G. Noninvasive measurement of dissolved oxygen in shake flasks. Biotechnol. Bioeng. 2002, 80, 594–597. [Google Scholar] [CrossRef]
- Veras, H.C.T.; Parachin, N.S.; Almeida, J.R.M. Comparative assessment of fermentative capacity of different xylose-consuming yeasts. Microb. Cell Fact. 2017, 16, 153. [Google Scholar] [CrossRef]
- Selim, K.A.; Easa, S.M.; El-Diwany, A.I. The xylose metabolizing yeast Spathaspora passalidarum is a promising genetic treasure for improving bioethanol production. Fermentation 2020, 6, 33. [Google Scholar] [CrossRef]
- Mendes, C.V.T.; Carvalho, M.G.V.S.; Rocha, J.M.S. Bioethanol production from cellulosic fibers: Comparison between batch and fed-batch saccharification. Cellul. Chem. Technol. 2017, 51, 291–299. [Google Scholar]
- Raposo, S.; Constantino, A.; Rodrigues, F.; Rodrigues, B.; Lima-Costa, M.E. Nitrogen Sources Screening for Ethanol Production Using Carob Industrial Wastes. Appl. Biochem. Biotechnol. 2017, 181, 827–843. [Google Scholar] [CrossRef]
- Pereira, F.B.; Guimarães, P.M.R.; Teixeira, J.A.; Domingues, L. Optimization of low-cost medium for very high gravity ethanol fermentations by Saccharomyces cerevisiae using statistical experimental designs. Bioresour. Technol. 2010, 101, 7856–7863. [Google Scholar] [CrossRef] [PubMed]
- Kadam, K.L.; Newman, M.M. Development of a low-cost fermentation medium for ethanol production from biomass. Appl. Microbiol. Biotechnol. 1997, 47, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Pejó, E.; Negro, M.J.; Sáez, F.; Ballesteros, M. Effect of nutrient addition on preinoculum growth of S. cerevisiae for application in SSF processes. Biomass Bioenergy 2012, 45, 168–174. [Google Scholar] [CrossRef]
- Feng, S.; Cheng, S.; Yuan, Z.; Leitch, M.; Xu, C. Valorization of bark for chemicals and materials: A review. Renew. Sustain. Energy Rev. 2013, 26, 560–578. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amândio, M.S.T.; Rocha, J.M.S.; Serafim, L.S.; Xavier, A.M.R.B. Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment. Energies 2021, 14, 2185. https://doi.org/10.3390/en14082185
Amândio MST, Rocha JMS, Serafim LS, Xavier AMRB. Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment. Energies. 2021; 14(8):2185. https://doi.org/10.3390/en14082185
Chicago/Turabian StyleAmândio, Mariana S. T., Jorge M. S. Rocha, Luísa S. Serafim, and Ana M. R. B. Xavier. 2021. "Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment" Energies 14, no. 8: 2185. https://doi.org/10.3390/en14082185
APA StyleAmândio, M. S. T., Rocha, J. M. S., Serafim, L. S., & Xavier, A. M. R. B. (2021). Cellulosic Bioethanol from Industrial Eucalyptus globulus Bark Residues Using Kraft Pulping as a Pretreatment. Energies, 14(8), 2185. https://doi.org/10.3390/en14082185








