Transesterification of Pyrolysed Castor Seed Oil in the Presence of CaCu(OCH3)2 Catalyst
Abstract
1. Introduction
2. Materials and Methods
2.1. Catalyst and Fuel Characterization
2.2. Catalyst Preparation
2.3. Pyrolysis of Castor Oil
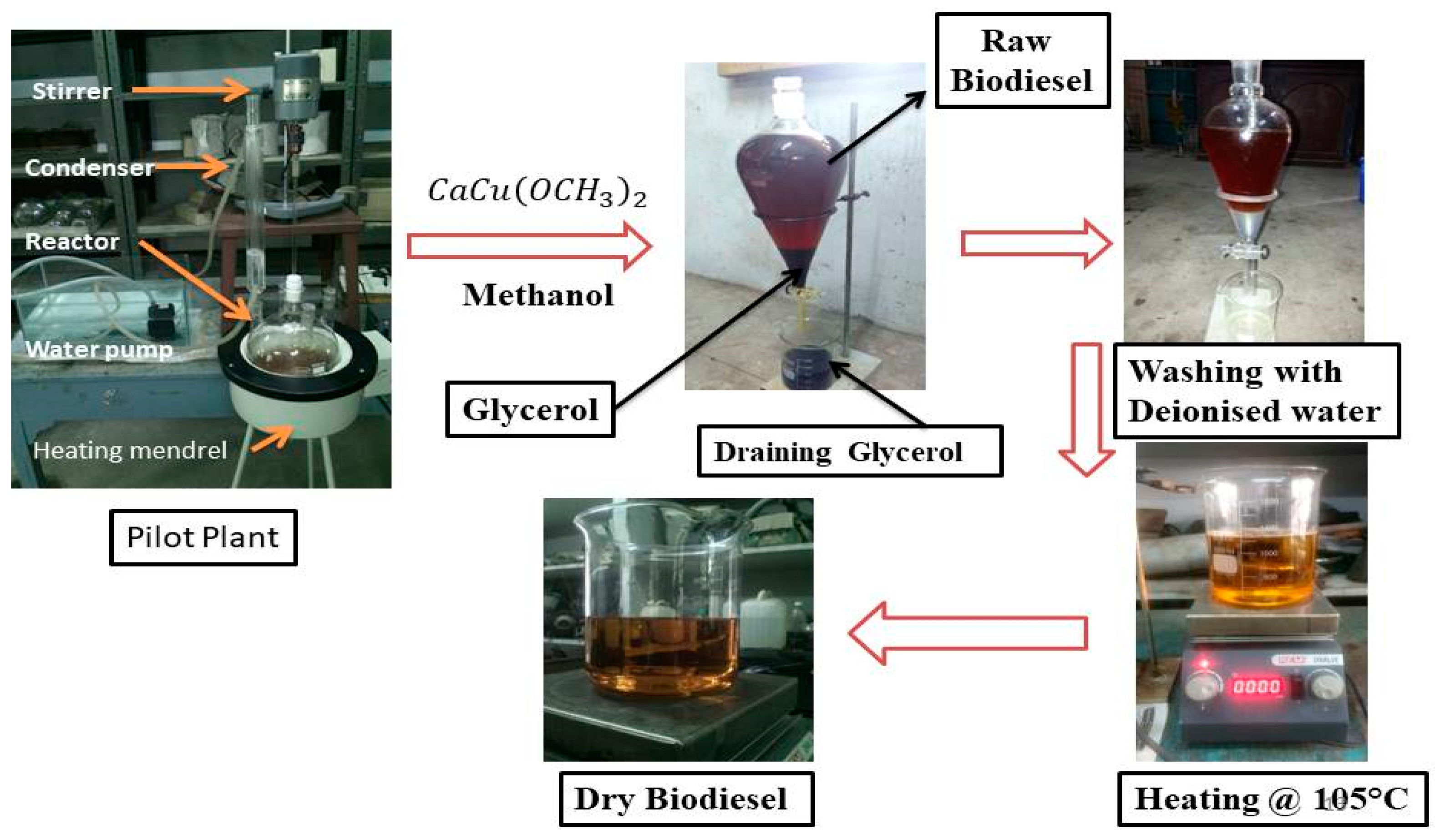
2.4. Biodiesel Production
2.4.1. Esterification of Castor Oil
2.4.2. Transesterification of Esterified Oil
2.4.3. Transesterification of Pyrolysis Castor Oil (PCO)
3. Results and Discussions
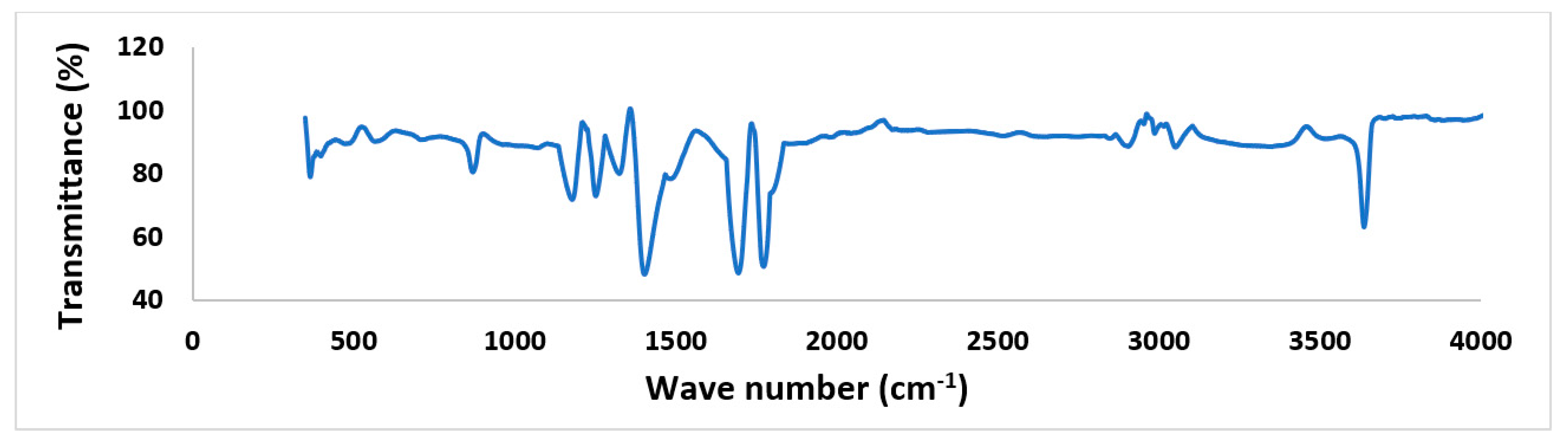
3.1. SEM and FTIR Analysis
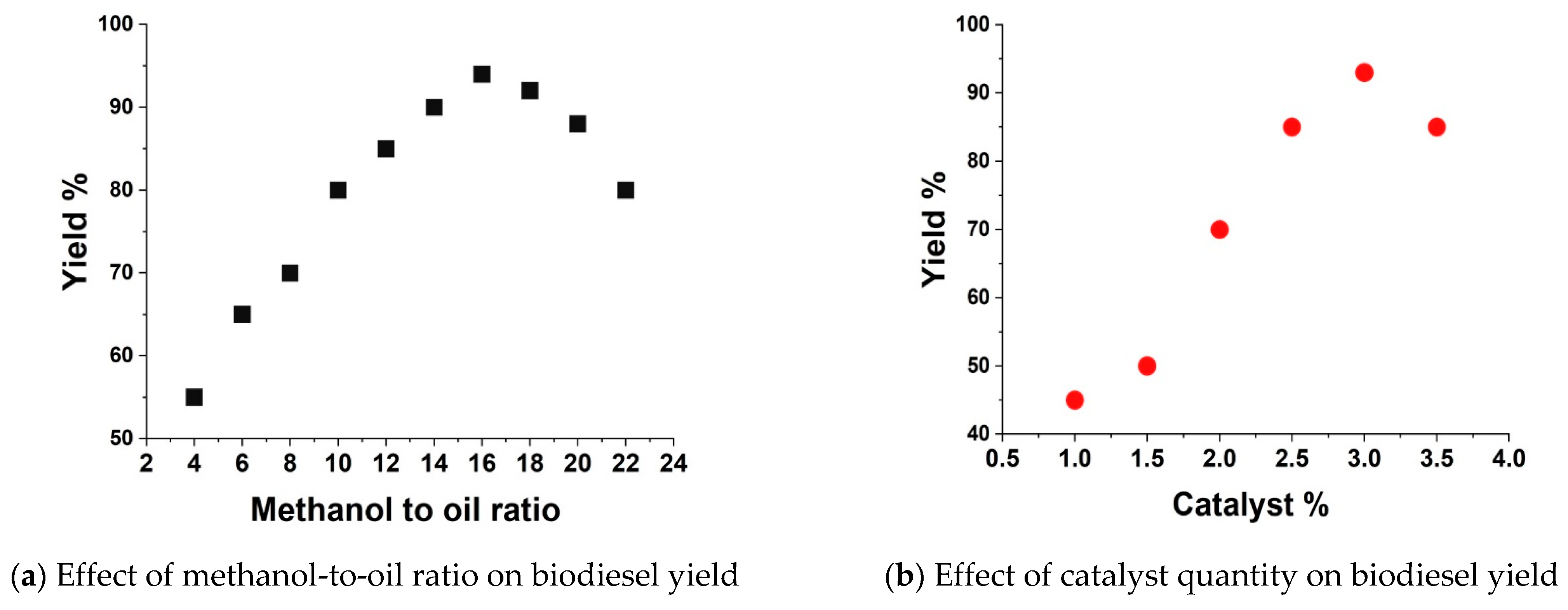
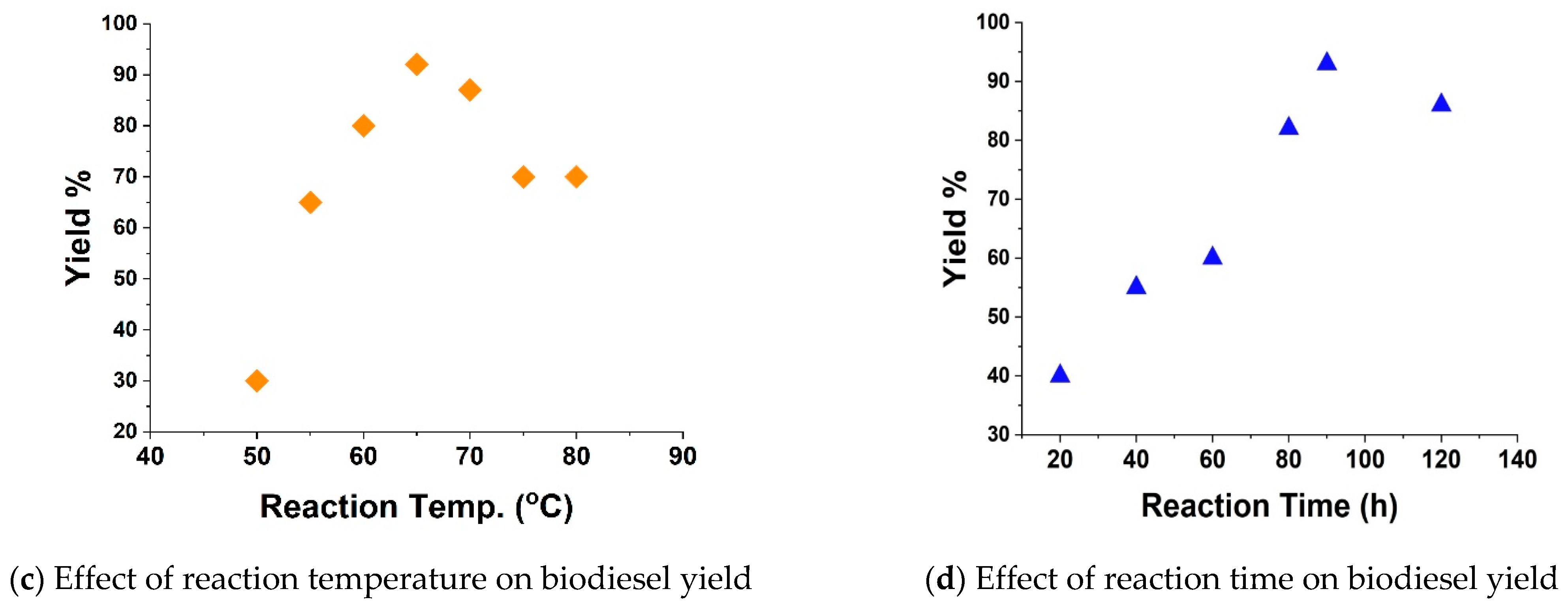
3.2. Optimization of Operating Parameters
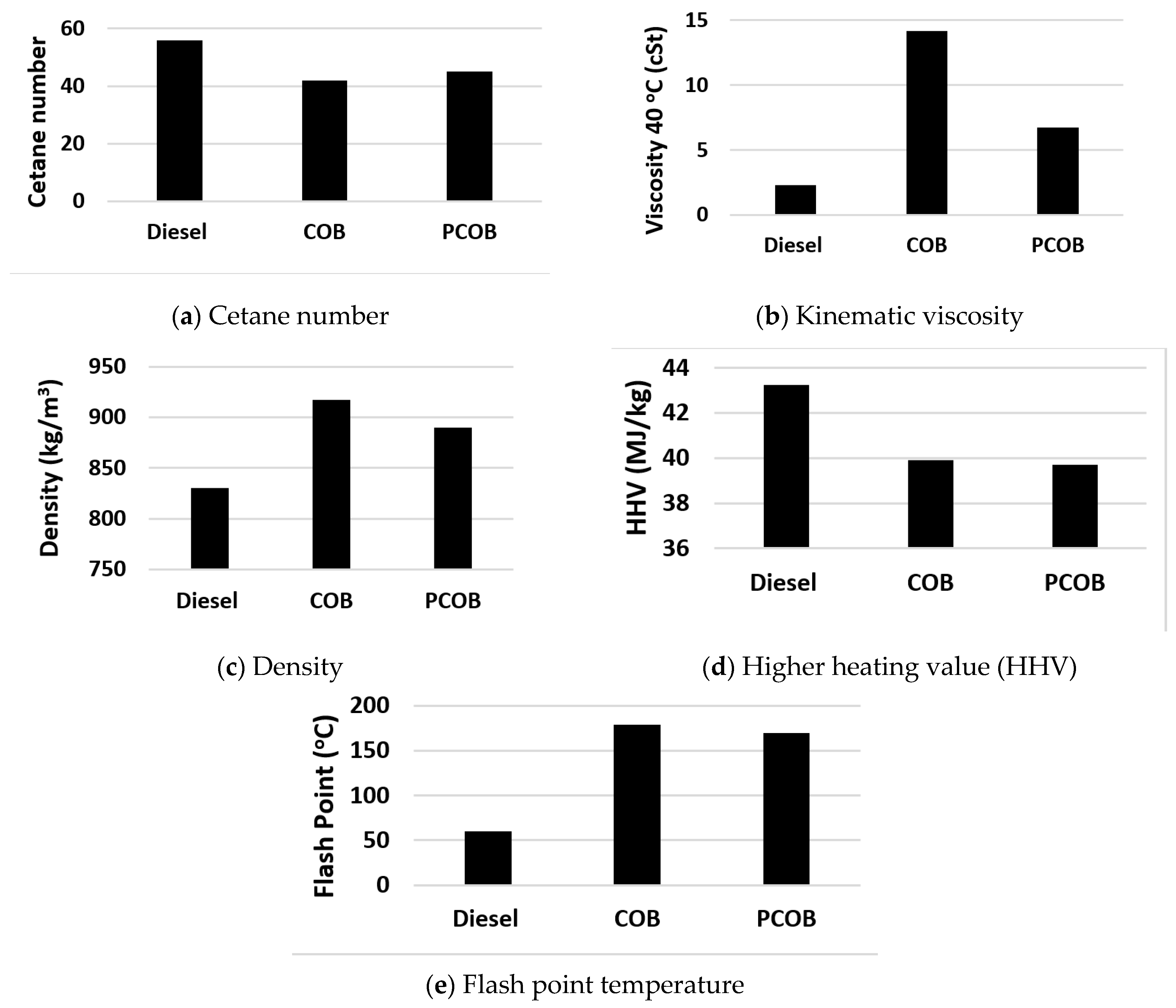
3.3. Biodiesel Properties Analysis
3.3.1. Cetane Number (CN)
3.3.2. Kinematic Viscosity
3.3.3. Density
3.3.4. Higher Heating Value (HHV)
3.3.5. Flash Point and Iodine Value
3.3.6. Oxidation Stability
4. Conclusions
- (1)
- Heterogeneous catalyst was prepared from waste eggshell and synthesis with CuO. The size was studied through SEM and found to be larger than 100 nm; therefore, it was used as catalyst instead of nano-catalyst;
- (2)
- Castor biodiesel fuel properties were improved through the combined pyrolysis and transesterification process;
- (3)
- Compared to un-pyrolysed condition, the cetane number and oxidation stability of PCOB were improved, while viscosity, density, flash point, and iodine value were reduced;
- (4)
- The viscosity of PCOB is higher than the automotive biodiesel standard; hence, it can be used in marine engine application.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AOCS | American Oil Chemists’ Society |
| BSFC | brake specific fuel consumption |
| BTE | brake thermal efficiency |
| CN | cetane number |
| COB | castor oil biodiesel |
| CO | castor oil |
| CV | calorific value |
| FP | flash point |
| HHV | higher heating value |
| IV | iodine value |
| NOx | oxides of nitrogen |
| OS | oxidation stability |
| SFA% | saturated fatty acids |
| TCOB | thermal cracked castor oil biodiesel |
| USFA | unsaturated fatty acids |
References
- Singh, D.; Sharma, D.; Soni, S.; Inda, C.S.; Sharma, S.; Sharma, P.K.; Jhalani, A. A comprehensive review of biodiesel production from waste cooking oil and its use as fuel in compression ignition engines: 3rd generation cleaner feedstock. J. Clean. Prod. 2021, 307, 127299. [Google Scholar] [CrossRef]
- Chen, G.-B.; Li, Y.-H.; Wu, W.-T. Effects of catalysts on pyrolysis of castor meal. Energy 2017, 119, 1–9. [Google Scholar] [CrossRef]
- Manojkumar, N.; Muthukumaran, C.; Sharmila, G. A comprehensive review on the application of response surface methodology for optimization of biodiesel production using different oil sources. J. King Saud Univ.-Eng. Sci. 2020. [Google Scholar] [CrossRef]
- Renish, R.R.; Selvam, M.A.J. A critical review on production process, physicochemical properties, performance, and emission characteristics of sea mango biodiesel-diesel blends. Mater. Today Proc. 2021, 44, 2600–2605. [Google Scholar] [CrossRef]
- Estevez, R.; Deblas, L.M.A.; Bautista, F.M.; Luna, D.; Luna, C.; Calero, J.; Posadillo, A.; Romero, A.A. Biodiesel at the Crossroads: A Critical Review. Catalysts 2019, 9, 1033. [Google Scholar] [CrossRef]
- Kochaphum, C.; Gheewala, S.H.; Vinitnantharat, S. Does biodiesel demand affect palm oil prices in Thailand? Energy Sustain. Dev. 2013, 17, 658–670. [Google Scholar] [CrossRef]
- Bin Mohiddin, M.N.; Tan, Y.H.; Seow, Y.X.; Kansedo, J.; Mubarak, N.; Abdullah, M.O.; Chan, Y.S.; Khalid, M. Evaluation on feedstock, technologies, catalyst and reactor for sustainable biodiesel production: A review. J. Ind. Eng. Chem. 2021, 98, 60–81. [Google Scholar] [CrossRef]
- Parvizsedghy, R.; Sadrameli, S.M. Thermal Cracking Approach Investigation to Improve Biodiesel Properties. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2015, 9, 805–809. [Google Scholar] [CrossRef]
- India National Biofuel Policy 2018. 2020. Available online: http://www.cbip.org/Policies2019/PD_07_Dec_2018_Policies/1_MNRE/2-Biofuel/2OrderBio-FuelPolicy.pdf (accessed on 6 March 2020).
- Mishra, S.; Anand, K.; Mehta, P.S. Predicting the Cetane Number of Biodiesel Fuels from Their Fatty Acid Methyl Ester Composition. Energy Fuels 2016, 30, 10425–10434. [Google Scholar] [CrossRef]
- Abdelfattah, M.S.H.; Abu-Elyazeed, O.S.M.; El Mawla, E.A.; Abdelazeem, M.A. On biodiesels from castor raw oil using catalytic pyrolysis. Energy 2018, 143, 950–960. [Google Scholar] [CrossRef]
- Karmakar, B.; Dhawane, S.H.; Halder, G. Optimization of biodiesel production from castor oil by Taguchi design. J. Environ. Chem. Eng. 2018, 6, 2684–2695. [Google Scholar] [CrossRef]
- Lei, S.; Qin, S.; Li, B.; Zhao, C. Pt/HAP catalyzed direct decarboxylation of lipid to alkanes via stabilization and synergism effect. J. Catal. 2021, 400, 244–254. [Google Scholar] [CrossRef]
- Aboelazayem, O.; El-Gendy, N.S.; Abdel-Rehim, A.A.; Ashour, F.; Sadek, M.A. Biodiesel production from castor oil in Egypt: Process optimisation, kinetic study, diesel engine performance and exhaust emissions analysis. Energy 2018, 157, 843–852. [Google Scholar] [CrossRef]
- Kumar, N. Oxidative stability of biodiesel: Causes, effects and prevention. Fuel 2017, 190, 328–350. [Google Scholar] [CrossRef]
- Figueiredo, M.-K.; Romeiro, G.; Damasceno, R. Low temperature conversion (LTC) of castor seeds—A study of the oil fraction (pyrolysis oil). J. Anal. Appl. Pyrolysis 2009, 86, 53–57. [Google Scholar] [CrossRef]
- Singh, R.; Shadangi, K. Liquid fuel from castor seeds by pyrolysis. Fuel 2011, 90, 2538–2544. [Google Scholar] [CrossRef]
- Koul, M.; Shadangi, K.P.; Mohanty, K. Effect of catalytic vapour cracking on fuel properties and composition of castor seed pyrolytic oil. J. Anal. Appl. Pyrolysis 2016, 120, 103–109. [Google Scholar] [CrossRef]
- Vallinayagam, R.; Vedharaj, S.; Yang, W.; Roberts, W.; Dibble, R. Feasibility of using less viscous and lower cetane (LVLC) fuels in a diesel engine: A review. Renew. Sustain. Energy Rev. 2015, 51, 1166–1190. [Google Scholar] [CrossRef]
- Aguado-Deblas, L.; Hidalgo-Carrillo, J.; Bautista, F.M.; Luna, D.; Luna, C.; Calero, J.; Posadillo, A.; Romero, A.A.; Estevez, R. Diethyl Ether as an Oxygenated Additive for Fossil Diesel/Vegetable Oil Blends: Evaluation of Performance and Emission Quality of Triple Blends on a Diesel Engine. Energies 2020, 13, 1542. [Google Scholar] [CrossRef]
- Tsai, S.-Y.; Tseng, W.-S.; Wu, C.-T.; Lin, C.-P. Dwarf-castor Oil made into a Suitable Biodiesel. Procedia Eng. 2014, 84, 940–947. [Google Scholar] [CrossRef][Green Version]
- Das, M.; Sarkar, M.; Datta, A.; Santra, A.K. An experimental study on the combustion, performance and emission characteristics of a diesel engine fuelled with diesel-castor oil biodiesel blends. Renew. Energy 2018, 119, 174–184. [Google Scholar] [CrossRef]
- Thakkar, K.; Kachhwaha, S.S.; Kodgire, P.; Srinivasan, S. Combustion investigation of ternary blend mixture of biodiesel/n-butanol/diesel: CI engine performance and emission control. Renew. Sustain. Energy Rev. 2021, 137, 110468. [Google Scholar] [CrossRef]
- Habibullah, M.; Masjuki, H.; Alam, M.A.; Fattah, I.M.R.; Ashraful, A.; Mobarak, H. Biodiesel production and performance evaluation of coconut, palm and their combined blend with diesel in a single-cylinder diesel engine. Energy Convers. Manag. 2014, 87, 250–257. [Google Scholar] [CrossRef]
- Patel, C.; Chandra, K.; Hwang, J.; Agarwal, R.A.; Gupta, N.; Bae, C.; Gupta, T.; Agarwal, A.K. Comparative compression ignition engine performance, combustion, and emission characteristics, and trace metals in particulates from Waste cooking oil, Jatropha and Karanja oil derived biodiesels. Fuel 2019, 236, 1366–1376. [Google Scholar] [CrossRef]
- Dhanasekaran, R.; Ganesan, S.; Kumar, B.R.; Saravanan, S. Utilization of waste cooking oil in a light-duty DI diesel engine for cleaner emissions using bio-derived propanol. Fuel 2019, 235, 832–837. [Google Scholar] [CrossRef]
- Rashed, M.; Kalam, A.; Masjuki, H.; Mofijur, M.; Rasul, M.; Zulkifli, N. Performance and emission characteristics of a diesel engine fueled with palm, jatropha, and moringa oil methyl ester. Ind. Crop. Prod. 2016, 79, 70–76. [Google Scholar] [CrossRef]
- Auti, S.; Rathod, W. Effect of hybrid blends of raw tyre pyrolysis oil, karanja biodiesel and diesel fuel on single cylinder four stokes diesel engine. Energy Rep. 2021, 7, 2214–2220. [Google Scholar] [CrossRef]
- Raman, L.A.; Deepanraj, B.; Rajakumar, S.; Sivasubramanian, V. Experimental investigation on performance, combustion and emission analysis of a direct injection diesel engine fuelled with rapeseed oil biodiesel. Fuel 2019, 246, 69–74. [Google Scholar] [CrossRef]
- Tamilselvan, P.; Sassykova, L.R.; Prabhahar, M.; Bhaskar, K.; Kannayiram, G.; Subramanian, S.; Prakash, S. Influence of saturated fatty acid material composition in biodiesel on its performance in internal combustion engines. Mater. Today Proc. 2020, 33, 1181–1186. [Google Scholar] [CrossRef]
- Kamaronzaman, M.F.F.; Kahar, H.; Hassan, N.; Hanafi, M.F.; Sapawe, N. Biodiesel production from waste cooking oil using nickel doped onto eggshell catalyst. Mater. Today Proc. 2020, 31, 342–346. [Google Scholar] [CrossRef]
- Teo, S.H.; Rashid, U.; Taufiq-Yap, Y.H. Green nano-catalyst for methanolysis of non-edible Jatropha oil. Energy Convers. Manag. 2014, 87, 618–627. [Google Scholar] [CrossRef]
- Kirubakaran, M.; Selvan, V.A.M. Experimental investigation on the effects of micro eggshell and nano-eggshell catalysts on biodiesel optimization from waste chicken fat. Bioresour. Technol. Rep. 2021, 14, 100658. [Google Scholar] [CrossRef]
- Sharma, V.; Duraisamy, G. Production and characterization of bio-mix fuel produced from a ternary and quaternary mixture of raw oil feedstock. J. Clean. Prod. 2019, 221, 271–285. [Google Scholar] [CrossRef]
- Niazmahmud. Cetane Number Prediction 2012. 2020. Available online: https://www.slideshare.net/NIAZMAHMUD/aniline-point-for-petroleum-productes (accessed on 10 September 2020).
- Can, Ö. Combustion characteristics, performance and exhaust emissions of a diesel engine fueled with a waste cooking oil biodiesel mixture. Energy Convers. Manag. 2014, 87, 676–686. [Google Scholar] [CrossRef]
- Sharma, V.; Duraisamy, G. Production and characterization of bio-mix fuel produced from the mixture of raw oil feedstock, and its effects on performance and emission analysis in DICI diesel engine. Environ. Sci. Pollut. Res. 2019, 26, 16742–16761. [Google Scholar] [CrossRef]
- Wang, W.; Liu, H.; Li, F.; Wang, H.; Ma, X.; Li, J.; Zhou, L.; Xiao, Q. Effects of unsaturated fatty acid methyl esters on the oxidation stability of biodiesel determined by gas chromatography-mass spectrometry and information entropy methods. Renew. Energy 2021, 175, 880–886. [Google Scholar] [CrossRef]





| Pyrolysis Process | Viscosity (cSt) | Ref. |
|---|---|---|
| LTC pyrolysis at 380 °C | 84.14 | [16] |
| Fast Pyrolysis at 600 °C | 83.19 | [17] |
| Catalysis Pyrolysis (Zeolite ZSM-5) at 300 °C | 43.47 | [11] |
| Catalysis Pyrolysis (Zeolite ZSM-5) at 400 °C | 29.29 | [2] |
| Catalytic Upgrading (ZnO) at 550 °C | 28 | [18] |
| Biodiesel & Blends | BSFC | BTE | CO | HC | NOx | Smoke | Ref. |
|---|---|---|---|---|---|---|---|
| Castor biodiesel | 9.7% | 2.9% | 12.3% | 28.9% | 11.6% | 4.5% | [22,23] |
| Coconut biodiesel | 6.9% | 1.3% | 5.2% | 53% | 8% | 11.3% | [24] |
| Waste oil biodiesel | 6.7% | 1.5% | 12% | 32.8% | 14.6% | 2.3% | [25,26] |
| Jatropha Biodiesel | 2.4% | 0.49% | 13% | 36.8% | 10.5% | 3% | [25,27] |
| Karanja Biodiesel | 3.9 | 1.9% | 11% | 23.6% | 9.2% | 5.2% | [25,28] |
| Rape seed biodiesel | 8.5% | 3% | 7.6% | 33% | 14% | 5.6% | [29] |
| Mahua Biodiesel | 5% | 2.2% | 12% | 32.7% | 12% | 2.8% | [30] |
| Fuel Properties | Name of the Instrument | Standards |
|---|---|---|
| Density | Pycnometer | ASTM D4892-14 |
| Kinematic viscosity | Ubbelohde viscometer | ASTM D4603-18 |
| Calorific Value | Bomb Calorimeter | ASTM D240-19 |
| Oxidation stability | Rancimat Method (AOCS Cd 12b-92) | EN 14112 |
| Iodine value | Titration method | EN 14111 |
| Flash Point | Closed cup | EN 3679 |
| Material | Specifications |
|---|---|
| Stainless steel cylinder | 219 mm diameter, 400 mm height and 5 mm thickness |
| Copper condenser tube | 1-inch ID, 2-inch OD |
| Electric heater | 6 kW |
| Capacity | 10 kg |
| Pressure gauge | Range 0–7 bar 1 bar |
| Thermocouple (K-type) | Range 0–1500 °C 1 °C |
| Biodiesel | Viscosity 40 °C (cSt) | Density (kg/m3) | CN | HHV (MJ/kg) | Flash Point (°C) | I.V (gI2/100 g) | Oxidation Stability (h) |
|---|---|---|---|---|---|---|---|
| COB 100 | 14.2 | 917.6 | 42 | 39.9 | 179 | 85.2 | 13 |
| PCOB 100 | 6.72 | 890 | 45 | 39.7 | 170 | 80 | 18.5 |
| Diesel | 2.3 | 830 | 56 | 43,250 | 60 | - | - |
| EN biodiesel Standard limits EN 14214;2003 [34] | 3.5–5.0 | 860–900 | 51 min | - | 120 min | 120 | 6 |
| Petroleum diesel standard EN 590;1999 [34] | 2–4.5 | 820–845 | 51 min | - | 55 min | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, V.; Hossain, A.K.; Duraisamy, G.; Vijay, M. Transesterification of Pyrolysed Castor Seed Oil in the Presence of CaCu(OCH3)2 Catalyst. Energies 2021, 14, 6064. https://doi.org/10.3390/en14196064
Sharma V, Hossain AK, Duraisamy G, Vijay M. Transesterification of Pyrolysed Castor Seed Oil in the Presence of CaCu(OCH3)2 Catalyst. Energies. 2021; 14(19):6064. https://doi.org/10.3390/en14196064
Chicago/Turabian StyleSharma, Vikas, Abul Kalam Hossain, Ganesh Duraisamy, and Murugan Vijay. 2021. "Transesterification of Pyrolysed Castor Seed Oil in the Presence of CaCu(OCH3)2 Catalyst" Energies 14, no. 19: 6064. https://doi.org/10.3390/en14196064
APA StyleSharma, V., Hossain, A. K., Duraisamy, G., & Vijay, M. (2021). Transesterification of Pyrolysed Castor Seed Oil in the Presence of CaCu(OCH3)2 Catalyst. Energies, 14(19), 6064. https://doi.org/10.3390/en14196064







