Reuse of Flowback Water from Hydraulic Fracturing for Drilling Mud Preparation and Secondary Hydrocarbon Recovery
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Flowback Collection and Well Information
2.3. Analytical Measurements of Physicochemical Parameters of Flowback Water
2.4. Preparation of Untreated Flowback-Based Drilling Mud
2.5. Coagulation and Filtration as Methods for Flowback Water Treatment
2.6. Flowback Flooding for Enhanced Oil Recovery
3. Results and Discussion
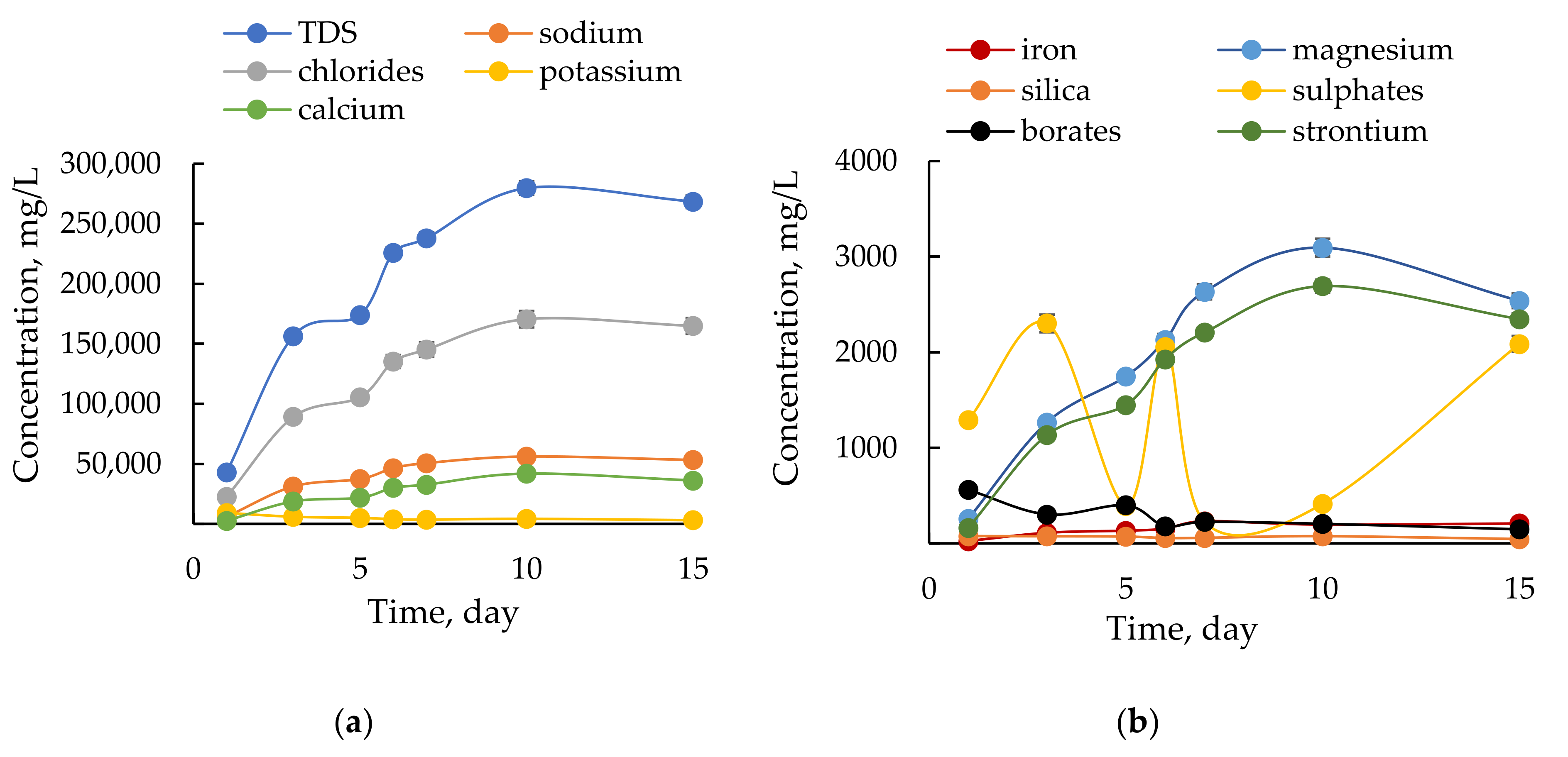
3.1. Chemical Composition of Flowback
3.1.1. Inorganic Constituents in Flowback Water
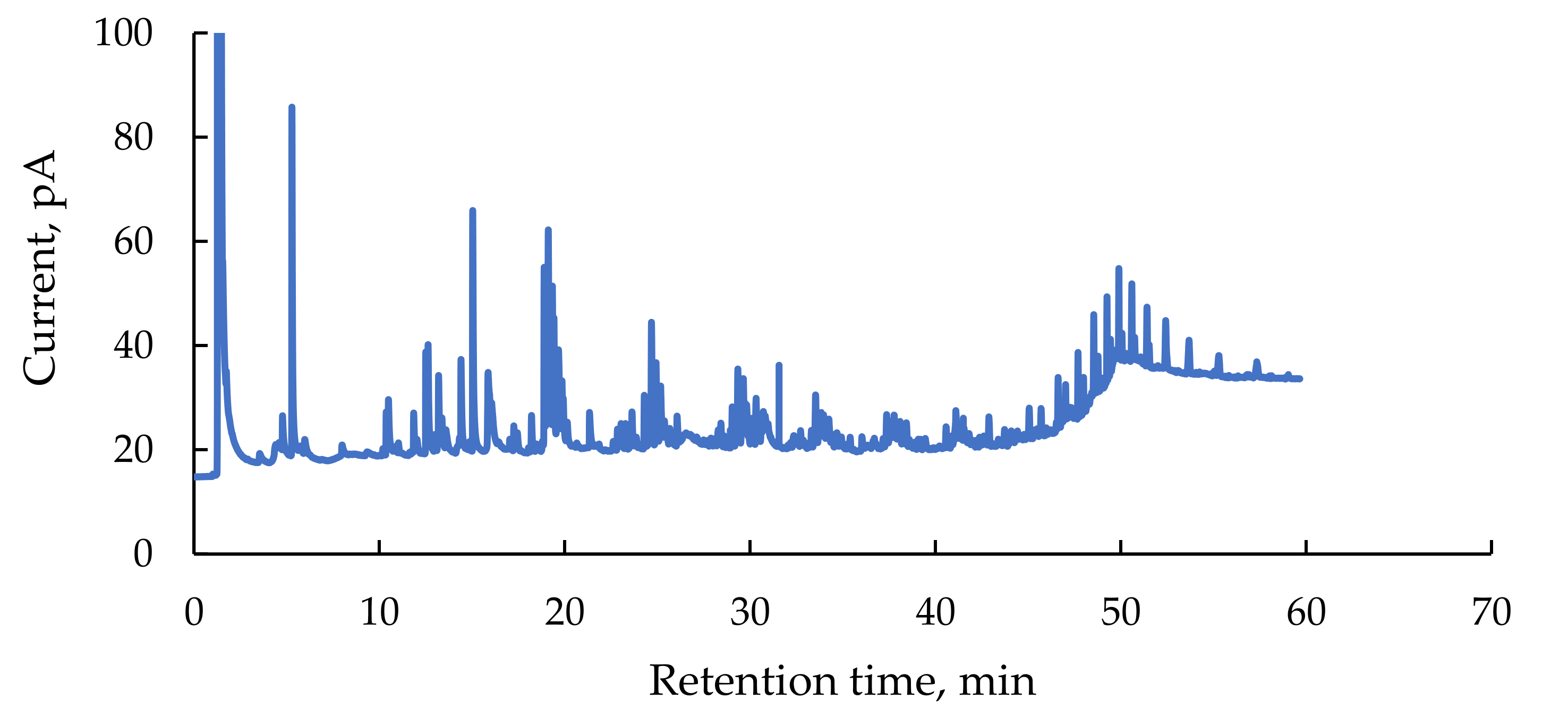
3.1.2. Organic Compounds in Flowback
3.2. Coagulation and Filtration Process
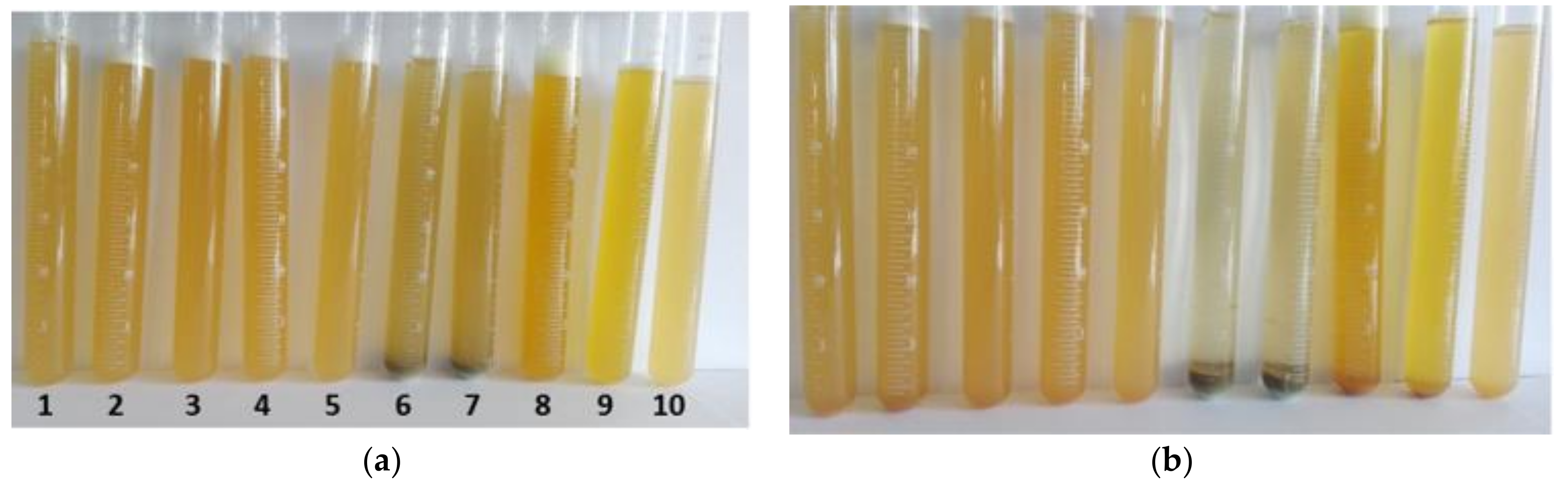
3.2.1. Selection of the Optimal Composition of Coagulant
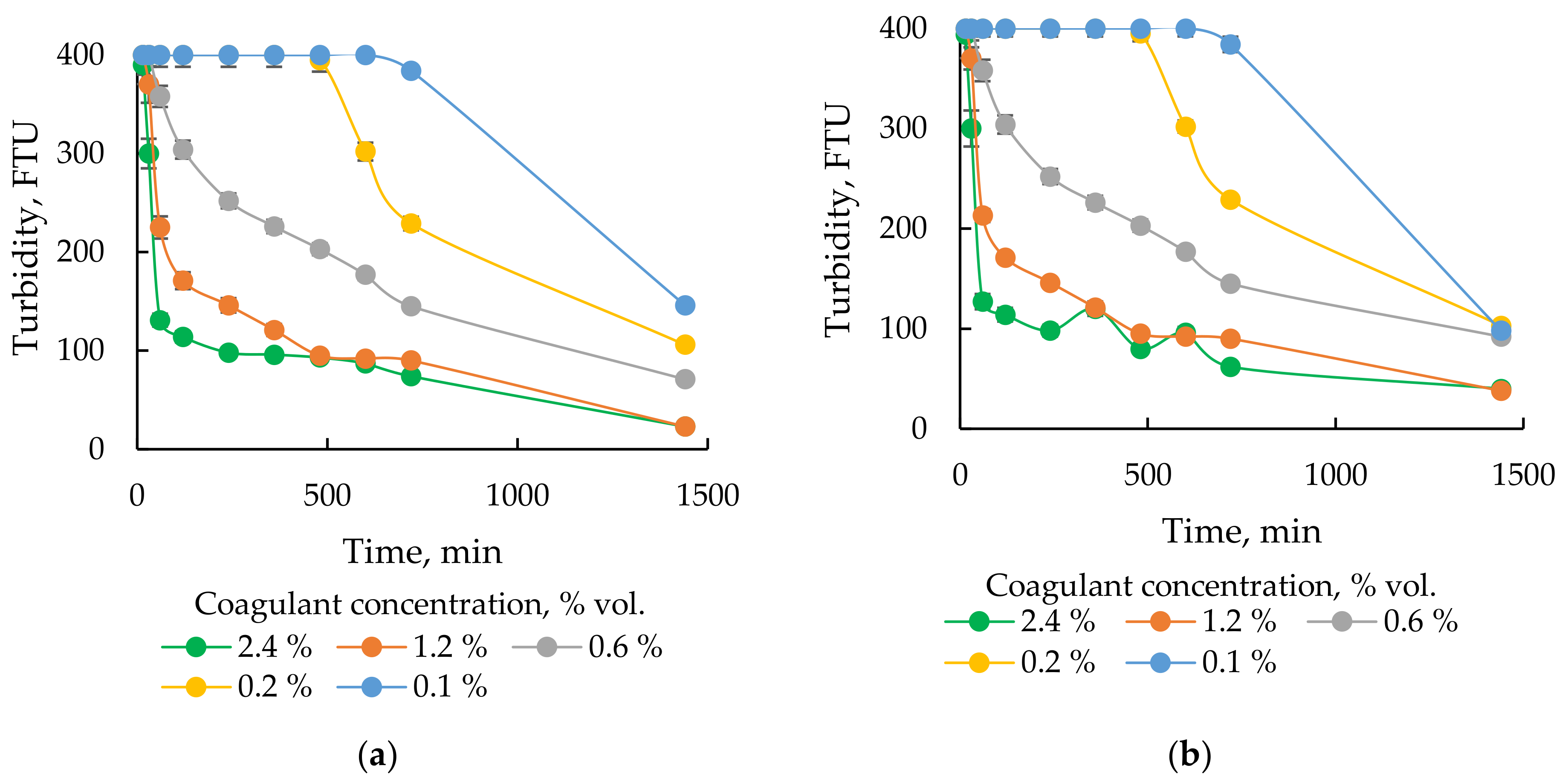
3.2.2. Selection of the Optimal Dose of Coagulant
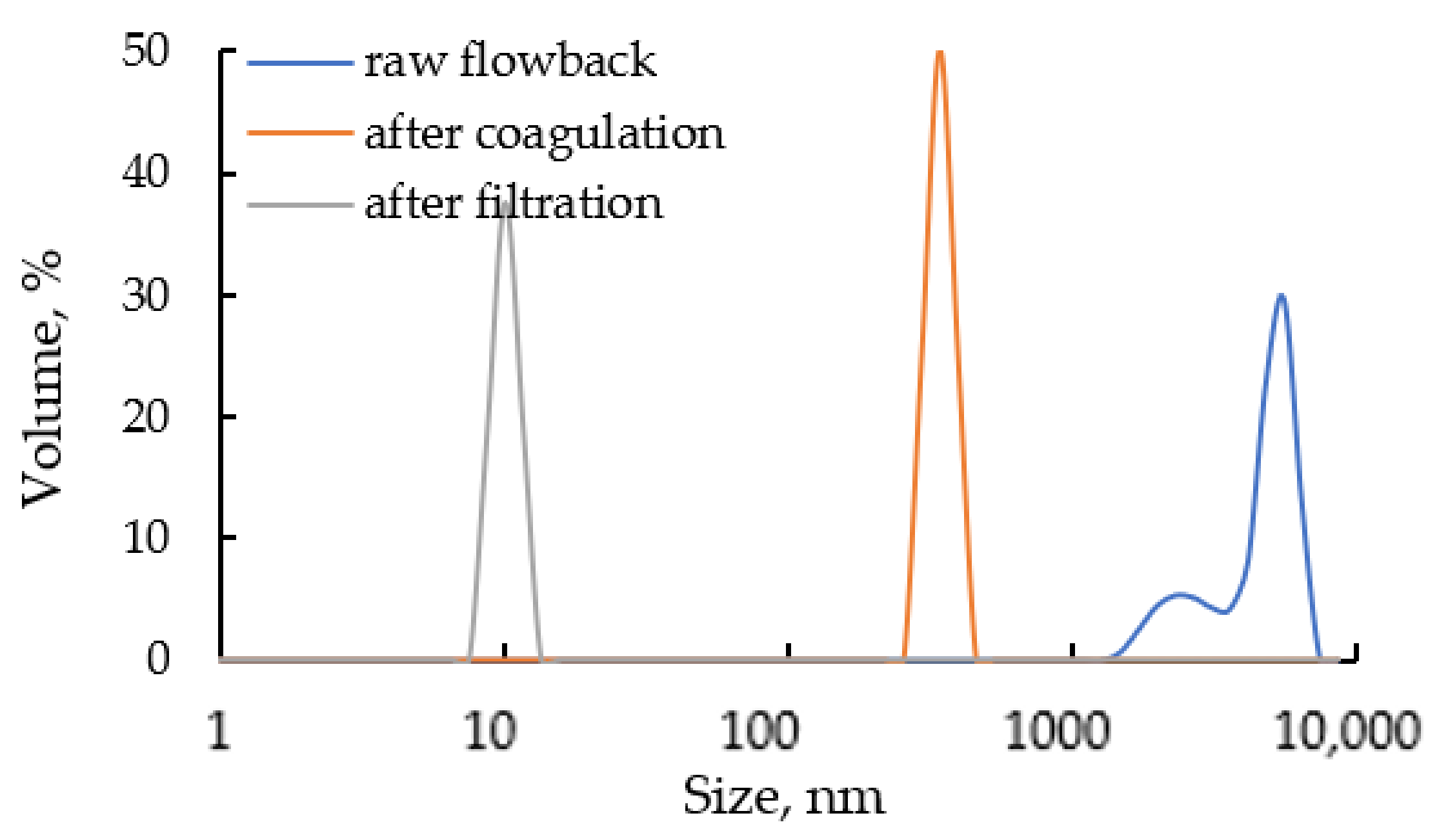
3.2.3. Filtration of Flowback Water after the Coagulation Process
3.3. Flowback Reinjection for EOR
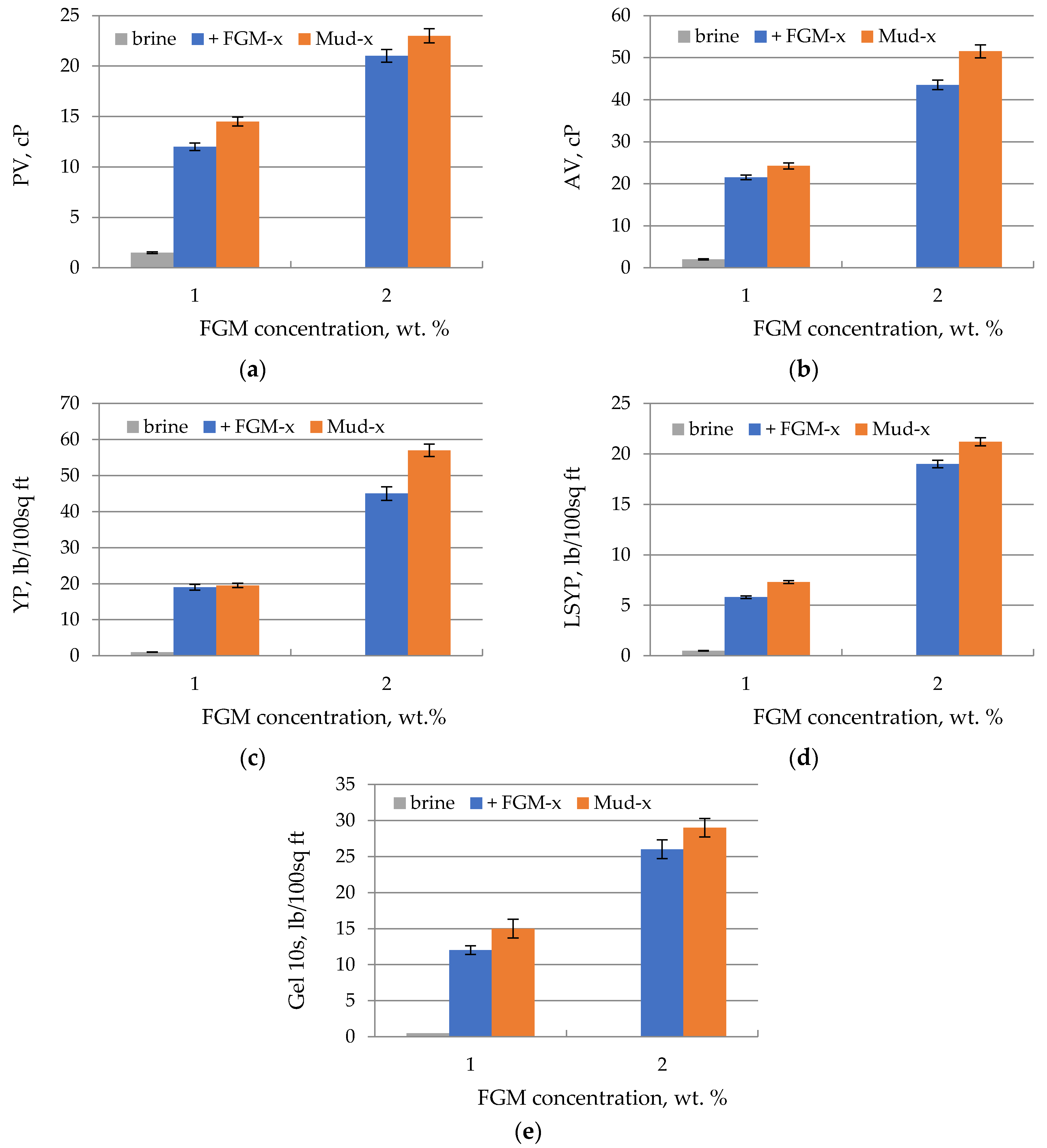
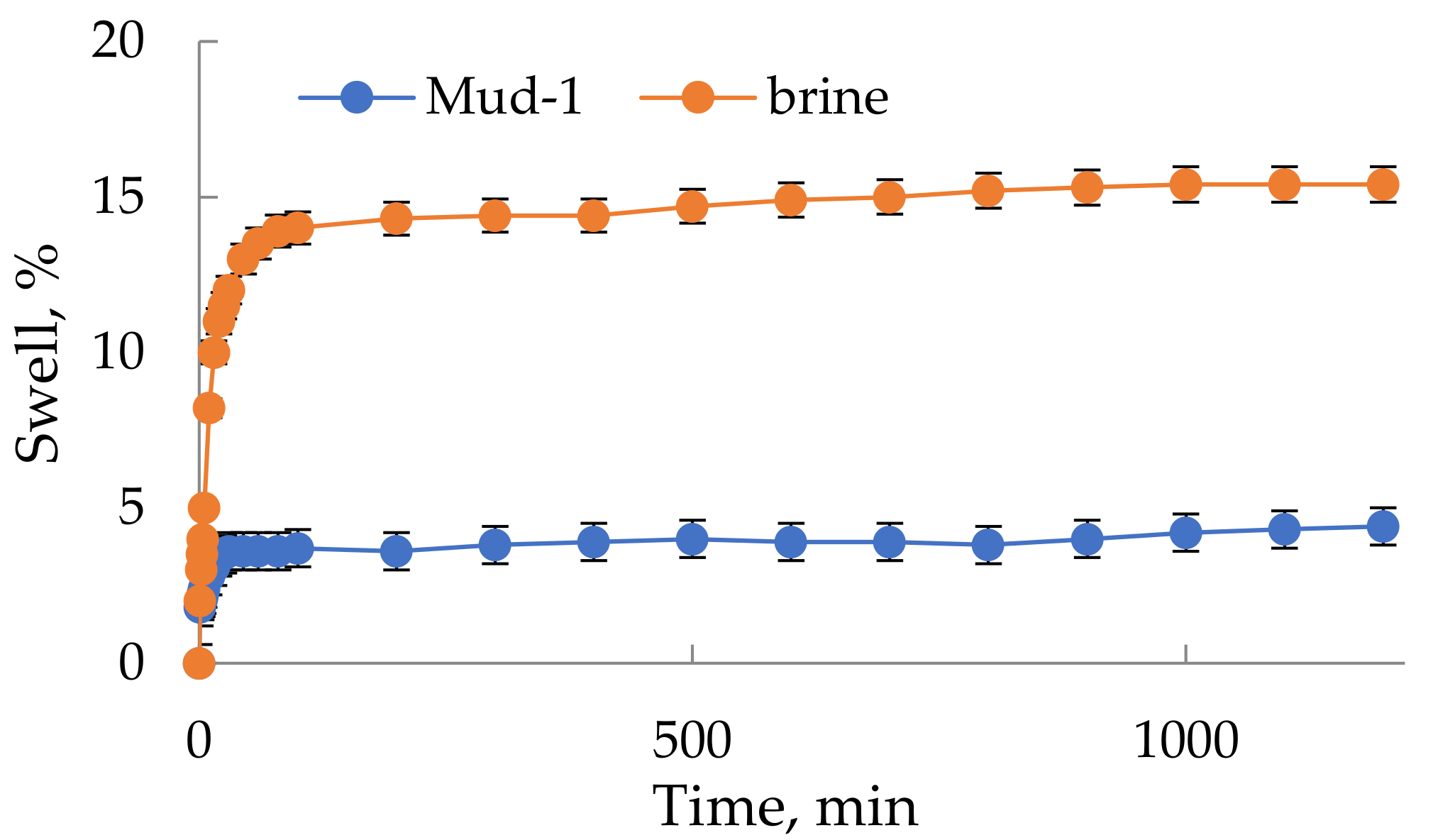
3.4. Filtration and Rheological Parameters of Flowback-Based Mud
4. Conclusions
- (1)
- Coagulation and filtration are suitable treatment technologies for purification of recovered flowback water after completion of hydraulic fracturing before its further beneficial reuse. Due to the applied treatment the TOC in the flowback decreases by 95% and the concentration of scaling ions is reduced at least by half. The turbidity of raw flowback is over 400 FTU and after coagulation with SAX18 commercial coagulant based on NaAlO2 decreases to 23 FTU. The optimal concentration of SAX18 coagulant is 12 mL/L. The residual turbidity is possibly caused by dissolved silica (~22 mg/L) and the DLS measurement confirms the presence of suspended solids with particle size smaller than 10 nm.
- (2)
- The treated flowback water can be successfully reused for preparation of drilling muds and for tertiary oil recovery. The reuse system should be designed to meet the particular needs of a given reservoir or drilling operator. For reservoirs at the beginning stage of exploitation, when both new wells are drilled and the existing ones are fractured, the recovered flowback can be used to prepare drilling fluids. For mature fields, where only selected horizons are planned to be fractured, the flowback after fracturing completion may be reinjected into the formation to maintain the reservoir pressure. Treatment requirements are in these cases much lower than for its reuse in agriculture or its discharge to the environment.
- (3)
- Reinjection of reused flowback allows to improve the oil production process. The recovery of oil from sand packs was 7% higher for flooding with treated flowback than with fresh water. This suggests that surfactants present in the flowback water increase its sweep efficiency.
- (4)
- Drilling muds prepared using the untreated flowback exhibit sufficient filtration and rheological properties. Thus prepared drilling fluids can be applied both in vertical and horizontal wells and are especially recommended when drilling in clay rocks and salt domes. Main advantages of the proposed flowback reusing are smaller operational footprints and lowered water demands. Closed-loop water systems are crucial while moving towards a low-emissions oil industry.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix
| Trade Name | Physicochemical Parameters | Chemical Composition | |
|---|---|---|---|
| Aluminium coagulants | SAX13 | Colorless liquid, pH approx. 12.5 | Sodium aluminate (NaAlO2) content: 27–30% |
| SAX18 | Colorless liquid, pH approx. 12.5 | Sodium aluminate (NaAlO2) content: 18–28% | |
| PAX18 | Light yellow liquid, pH approx. 1.0 | Polyaluminium chloride (aluminum content: 9.0 ± 0.3%, chloride content: 21.0 ± 0.3%) | |
| PAXXL19F | Slightly cloudy, light gray liquid, pH approx. 4.0 | Polyaluminium chloride (aluminum content: 8.5 ± 0.3%, chloride content: 5.5 ± 0.5%) | |
| PAXXL19H | Slightly cloudy, light gray liquid, pH approx. 3.5 | Polyaluminium chloride (aluminum content: 12.5 ± 0.3%, chloride content: 8.5 ± 1.0%) | |
| PAXXL10 | Colorless (to gray) liquid, pH approx. 2.0 | Polyaluminium chloride (aluminum content: 5.0 ± 0.2%, chloride content: 11.5 ± 1.0%) | |
| ALS | Colorless liquid, pH approx. 2.4 | Aluminum sulphate content: 25–28% | |
| Iron coagulants | PIX113 | Dark brown liquid, pH approx. 1 | Iron (III) sulfate content: 50–35%, Manganese (II) sulfate content: <0.25% |
| PIX116 | Brown liquid, pH approx. 1 | Iron (III) chloride content: 36–41% |
| Parameter | Date | Water After Coagulation and Filtration Treatment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Unit | 1st Day 2019-12-10 | 3rd Day 2019-12-12 | 5th Day 2019-12-14 | 6th Day 2020-01-08 | 7th Day 2020-01-09 | 10th Day 2020-01-12 | 15th Day 2020-01-17 | ||
| pH | 6.91 | 5.81 | 5.19 | 4.48 | 5.21 | 4.82 | 4.08 | 5.00 | |
| Conductivity | [mS/cm] | 65.1 | 163.7 | 183 | 204 | 216 | 218 | 219 | 218 |
| Density | [kg/m3] | 1033.24 | 1103.82 | 1122.95 | 1158.37 | 1169.15 | 1181.55 | 1179.47 | 1179.89 |
| Total dissolved solids | [mg/L] | 42,964.2 | 156,412.1 | 173,956.8 | 225,855.2 | 237,974.7 | 279,710.2 | 268,463.8 | 268,788.1 |
| Mineralisation | [mg/L] | 43,322.7 | 156,467.1 | 174,011.8 | 225,855.2 | 238,022.9 | 279,751.2 | 268,484.3 | 268,800.3 |
| General hardness | [mg CaCO3/L] | 7607.7 | 51,788.3 | 61,505.7 | 83,992.6 | 92,443.1 | 117,278.4 | 100,693.9 | 108,589 |
| Non-carbonate hardness | [mval/L] | 140.39 | 1033.96 | 1228.31 | 1679.18 | 1847.8 | 2344.22 | 2013.21 | 2171.79 |
| H2SiO3 | [mg/L] | 100.36 | 97.5 | 93.6 | 73.19 | 76.05 | 98.67 | 60.19 | 28.18 |
| SiO2 | [mg/L] | 77.2 | 75 | 72 | 56.3 | 58.5 | 75.9 | 46.3 | 21.68 |
| Na+ | [mg/L] | 5976 | 31,201 | 37,350 | 46,475 | 50,660 | 56,140 | 53,229 | 53,890 |
| K+ | [mg/L] | 9135.00 | 5931.00 | 5028.00 | 3898.00 | 3527.00 | 4206.00 | 3055 | 3182 |
| Li+ | [mg/L] | 6.09 | 42.70 | 51.97 | 71.70 | 79.54 | 86.80 | 81.2 | 94.31 |
| Ca2+ | [mg/L] | 2623 | 18,667 | 21,770 | 30,156 | 32,710 | 41,902 | 36,172 | 38,990 |
| Mg2+ | [mg/L] | 258.2 | 1267.0 | 1747.0 | 2127.0 | 2632.0 | 3094.0 | 2538 | 2748 |
| Ba2+ | [mg/L] | 2.995 | 6.130 | 16.920 | 16.540 | 41.5 | 51.200 | 27.38 | 13.16 |
| Sr2+ | [mg/L] | 163.5 | 1134.0 | 1448.0 | 1926.0 | 2207.0 | 2692.0 | 2347 | 2346 |
| Fe2+ | [mg/L] | 25.6 | 111.7 | 133.6 | 154.9 | 233.3 | 196.7 | 208.7 | 128.33 |
| Mn2+ | [mg/L] | 4.407 | 28.190 | 44.580 | 53.890 | 53.6 | 75.970 | 69.4 | 25.28 |
| Zn2+ | [mg/L] | 0.20 | 5.57 | 14.54 | 7.69 | 20.48 | 9.54 | 10.16 | 14.36 |
| Cu2+ | [mg/L] | 0.102 | 0.010 | 0.399 | 0.010 | <0.005 | <0.01 | <0.01 | 0.070 |
| Ni2+ | [mg/L] | 0.082 | 0.032 | 0.028 | 0.271 | <0.01 | 0.305 | 0.041 | 0.05 |
| Co2+ | [mg/L] | 0.019 | 0.024 | 0.067 | 0.028 | <0.01 | 0.102 | 0.031 | 0.01 |
| Pb2+ | [mg/L] | <0.005 | 0.538 | 0.875 | 0.554 | <0.005 | 0.222 | 0.546 | 0.10 |
| Cd2+ | [mg/L] | <0.005 | 0.040 | 0.051 | 0.008 | 0.07300 | 0.009 | 0.009 | 0.003 |
| Al3+ | [mg/L] | 0.857 | 0.010 | <0.01 | <0.01 | <0.01 | <0.01 | 0.642 | 0.01 |
| Cr3+ | [mg/L] | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | 0.013 | 0.01 |
| V5+ | [mg/L] | 0.180 | 0.309 | 1.100 | 0.334 | 1.385 | 3.250 | 0.371 | 0.20 |
| Ti4+ | [mg/L] | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | 0.29 |
| As3+ | [mg/L] | <0.05 | <0.05 | <0.05 | 0.210 | <0.05 | <0.05 | 0.36 | 0.02 |
| Cl− | [mg/L] | 22,600.0 | 89,120.0 | 105,510.0 | 135,235.0 | 145,290.0 | 170,546.0 | 164,885 | 166,766 |
| SO42− | [mg/L] | 1291.00 | 2301.00 | 393.00 | 2054.00 | 225.80 | 413.00 | 2087 | 424.35 |
| HCO32− | [mg/L] | 717.0 | 110.0 | 110.0 | 41.0 | 96.5 | 82.0 | 41 | 24.4 |
| CO32− | [mg/L] | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| PO43− | [mg/L] | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 |
| BO33− | [mg/L] | 560.32 | 303.01 | 399.30 | 178.98 | 225.60 | 205.09 | 147.97 | 167.95 |
| HBO2 | [mg/L] | 417.44 | 225.74 | 297.48 | 133.34 | 168.07 | 152.79 | 110.24 | 125.12 |
| Identified Name | Retention Time, Min | Relative Amount, % | Quality of Fit, % |
|---|---|---|---|
| Octadecanoic acid 2,3-dihydroxypropyl ester | 35.88 | 14.9 | 98 |
| (+)−2-Bornanone | 18.79 | 3.2 | 97 |
| endo-Borneol | 19.41 | 8.7 | 97 |
| 2,6,10,15-Tetramethylheptadecane | 32.50 | 1.1 | 93 |
| Nonadecane | 34.21 | 1.9 | 93 |
| 2-Hydroxy-3-methyl-2-cyclopenten-1-one | 15.24 | 3.2 | 91 |
| (1S)-1,3,3-trimethylnorbornan-2-ol | 20.11 | 5.2 | 91 |
| Decahydro-1,5,5,8a-tetramethyl-[1R-(1α.,3aβ,4α,8aβ)]-1,4-methanoazulen-9-one | 32.70 | 1.1 | 91 |
| Eicosane | 34.24 | 1.1 | 91 |
| 4-(1-Hydroxy-1-methylethyl)-1-methylcyclohexanol | 23.28 | 12.4 | 90 |
| 4,8,12,16-Tetraoxaeicosan-1-ol | 30.56 | 4.8 | 90 |
| 2-Hydroxy-3-ethyl-2-cyclopenten-1-one | 18.05 | 2.7 | 87 |
| 1-Methyl-4-(1-methylethyl)-3-cyclohexen-1-ol | 18.44 | 3.2 | 87 |
| Tetradecane | 25.43 | 1.8 | 87 |
| 1-Iodoeicosane, | 31.80 | 2.7 | 87 |
| Furyl hydroxymethyl ketone | 16.91 | 10.9 | 86 |
| N-Butylbenzenesulfonamide, | 30.48 | 8.5 | 81 |
| Cyclopentanone | 7.80 | 1.8 | 80 |
| 2-Methyl-2-cyclopenten-1-one | 11.33 | 1.9 | 80 |
| 1-(2-Furanyl)ethanone, | 11.50 | 6.5 | 80 |
| β-Acetylacrylic acid | 12.04 | 2.5 | 80 |
References
- Silva, T.L.S.; Morales-Torres, S.; Castro-Silva, S.; Figueiredo, J.L.; Silva, A.M.T. An overview on exploration and environmental impact of unconventional gas sources and treatment options for produced water. J. Environ. Manag. 2017, 200, 511–529. [Google Scholar] [CrossRef]
- Hannibal, B.; Portney, K. The impact of water scarcity on support for hydraulic fracturing regulation: A water-energy nexus study. Energy Policy 2020, 146, 111718. [Google Scholar] [CrossRef]
- Suboyin, A.; Rahman, M.M.; Haroun, M. Hydraulic fracturing design considerations, water management challenges and insights for Middle Eastern shale gas reservoirs. Energy Rep. 2020, 6, 745–760. [Google Scholar] [CrossRef]
- Candia, E.; Seth, K. Water management. Water recycling enhances well economics. The American Oil&Gas Reporter. Available online: https://www.aogr.com/web-exclusives/exclusive-story/water-recycling-enhances-well-economics (accessed on 30 August 2021).
- Liden, T.; Santos, I.C.; Hildenbrand, Z.L.; Schug, K.A. Treatment modalities for the reuse of produced waste from oil and gas development. Sci. Total Environ. 2018, 643, 107–118. [Google Scholar] [CrossRef]
- Mantell, M. Produced water reuse and recycling challenges and opportunities across major shale plays. In Proceedings of the EPA Technical Workshops for the Hydraulic Fracturing Study: Water Resources Management, Arlington, VA, USA, 29–30 March 2011. [Google Scholar]
- Xiong, B.; Roman-White, S.; Piechowicz, B.; Miller, Z.; Farina, B.; Tasker, T.; Burgos, W.; Zydney, A.L.; Kumar, M. Polyacrylamide in hydraulic fracturing fluid causes severe membrane fouling during flowback water treatment. J. Membr. Sci. 2018, 560, 125–131. [Google Scholar] [CrossRef]
- Sitterley, K.A.; Rosenblum, J.; Ruyle, B.; Keliher, R.; Linden, K.G. Factors impacting electrocoagulation treatment of hydraulic fracturing fluids and removal of common fluid additives and scaling ions. J. Environ. Chem. Eng. 2020, 8, 103728. [Google Scholar] [CrossRef]
- Haghshenas, A.; Nasr-El-Din, H.A. Effect of dissolved solids on reuse of produced water at high temperature in hydraulic fracturing jobs. J. Nat. Gas Sci. Eng. 2014, 21, 316–325. [Google Scholar] [CrossRef]
- Nasim, E.; Clay, T.; Herron, K.; Adam, P.; Kenneth, C. Recycling fracturing flowback water for use in hydraulic fracturing: Influence of organic matter on stability of carboxyl-methyl-cellulose-based fracturing fluids. SPE J. 2016, 21, 1358–1369. [Google Scholar]
- Stewart, D. Beneficial reuse of produced and flowback water. In Proceedings of the US EPA Technical Workshop on Analytical Chemical Methods for Hydraulic Fracturing, Research Triangle Park, NC, USA, 25 February 2013. [Google Scholar]
- Li, R.; Yang, J.; Pan, J.; Zhang, L.; Qin, W. Effect of immobilization on growth and organics removal of chlorella in fracturing flowback fluids treatment. J. Environ. Manag. 2018, 226, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-K.; Kong, F.-X.; Yang, D.-M.; Liu, Q.; Lin, X.-F.; Chen, J.-F.; Zhang, Y. The synergistic effect of electrocoagulation coupled with E-peroxone process for shale gas fracturing flowback water treatment. Chemosphere 2020, 262, 127968. [Google Scholar] [CrossRef]
- Chang, H.; Li, T.; Liu, B.; Vidic, R.D.; Elimelech, M.; Crittenden, J.C. Potential and implemented membrane-based technologies for the treatment and reuse of flowback and produced water from shale gas and oil plays: A review. Desalination 2019, 455, 34–57. [Google Scholar] [CrossRef]
- Siyal, M.I.; Lee, C.K.; Park, C.; Khan, A.A.; Kim, J.O. A review of membrane development in membrane distillation for emulsified industrial or shale gas wastewater treatments with feed containing hybrid impurities. J. Environ. Manag. 2019, 243, 45–66. [Google Scholar] [CrossRef]
- Tavakkoli, S.; Lokare, O.R.; Vidic, R.D.; Khanna, V. A techno-economic assessment of membrane distillation for treatment of Marcellus shale produced water. Desalination 2017, 416, 24–34. [Google Scholar] [CrossRef]
- Kar, A.; Bahadur, V. Using excess natural gas for reverse osmosis-based flowback water treatment in US shale fields. Energy 2020, 196, 117145. [Google Scholar] [CrossRef]
- Kong, F.-X.; Sun, G.-D.; Chen, J.-F.; Han, J.-D.; Guo, C.-M.; Zhang, T.; Lin, X.-F.; Xie, Y.F. Desalination and fouling of NF/low pressure RO membrane for shale gas fracturing flowback water treatment. Sep. Purif. Technol. 2018, 195, 216–223. [Google Scholar] [CrossRef]
- Hickenbottom, K.L.; Hancock, N.T.; Hutchings, N.R.; Appleton, E.W.; Beaudry, E.G.; Xu, P.; Cath, T.Y. Forward osmosis treatment of drilling mud and fracturing wastewater from oil and gas operations. Desalination 2013, 312, 60–66. [Google Scholar] [CrossRef]
- Boschee, P. Produced and flowback water recycling and reuse: Economics, limitations, and technology. Oil Gas Facil. 2014, 3, 16–21. [Google Scholar] [CrossRef]
- Analysis of Hydraulic Fracturing Fluid Data from the FracFocus Chemical Disclosure Registry 1.0; U.S. Environmental Protection Agency, Office of Research and Development: Washington, DC, USA, 2015; EPA/601/R-14/003.
- Fink, J.K. Drilling Muds. In Petroleum Engineer’s Guide to Oil Feld Chemicals and Fuids; Gulf Professional Publishing: Boston, MA, USA, 2012; Volume 1, p. 59. [Google Scholar] [CrossRef]
- Xiao, H.; Liu, S.; Chen, Y.; Han, D.; Wang, D. Impacts of polypropylene glycol (PPG) additive and pH on tribological properties of water-based drilling mud for steel-steel contact. Tribol. Int. 2017, 110, 318–325. [Google Scholar] [CrossRef]
- Oetjen, K.; Giddings, C.G.; McLaughlin, M.; Nell, M.; Blotevogel, J.; Helbling, D.E.; Mueller, D.; Higgins, C.P. Emerging analytical methods for the characterization and quantification of organic contaminants in flowback and produced water. Trends Environ. Anal. Chem. 2017, 15, 12–23. [Google Scholar] [CrossRef]
- Swartwout, R.; Pearcy, R. Design and application of brine-based drilling fluids. In Proceedings of the International Petroleum Conference and Exhibition of Mexico, Villahermosa, Mexico, 5–7 March 1996. [Google Scholar]
- Zhou, H.; Deville, J.P.; Davis, C.L. Novel thermally stable high-density brine-based drill-in fluids for HP/HT applications. In Proceedings of the SPE Middle East Oil and Gas Show and Conference, Manama, Bahrain, 8–11 March 2015. [Google Scholar] [CrossRef]
- Huang, L.; Yu, M.; Miska, S.; Takach, N.; Green, A.; Bloys, B. Determination of safe salinity window in drilling shale formation. In Proceedings of the 46th US Rock Mechanics/Geomechanics Symposium, Chicago, IL, USA, 24–27 June 2012. [Google Scholar]
- Ezzat, A.M.; Gamal, M.; D’Angelo, S. High density brine-based drill-in fluid improved reservoir producibility in gas field offshore Egypt. In Proceedings of the SPE North Africa Technical Conference and Exhibition, Marrakech, Morocco, 12–14 March 2008. [Google Scholar] [CrossRef]
- Ribeiro, L.S.; Dantas, T.N.C.; Dantas-Neto, A.A.; Melo, K.C.; Moura, M.C.P.A.; Aum, P.T.P. The use of produced water in water-based drilling fluids: Influence of calcium and magnesium concentrations. Braz. J. Pet. Gas 2016, 10, 233–245. [Google Scholar] [CrossRef][Green Version]
- Peacock, P. Beneficial use of produced water in the Indian Basin field: Eddy County, NM. In Proceedings of the Ground Water Protection Council Produced Water Conference, Colorado Springs, CO, USA, 16–17 October 2002. [Google Scholar]
- Michaelsen, J.; Bergu, B.; Marrelli, J.; Theobald, M. Subsea water injection-water quality management. In Proceedings of the Offshore Technology Conference, Houston, TX, USA, 2–5 May 2005. [Google Scholar] [CrossRef]
- Bader, M.S.H. Seawater versus produced water in oil-fields water injection operations. Desalination 2007, 208, 159–168. [Google Scholar] [CrossRef]
- He, M.; Lai, X.; Li, N.; Xiao, Y.; Shen, L.; Liu, X.; Ma, Z.; Wang, Y. Recovery and treatment of fracturing flowback fluids in the Sulige Gasfield, Ordos Basin. Nat. Gas Ind. B 2015, 2, 467–472. [Google Scholar] [CrossRef]
- Burnstad, R.G.; Martin, A.N.; Stemberger, D.J.; Purwanto, B. A case study of a mature field redevelopment using propped hydraulic fracturing. In Proceedings of the SPE Asia Pacific Oil and Gas Conference and Exhibition, Perth, Australia, 18–20 October 2004. [Google Scholar]
- Salah, M.; El-Sebaee, M.; Batmaz, T. Best practices and lessons learned from more than 1,000 treatments: Revival of mature fields by hydraulic fracturing in Khalda Ridge, Egypt’s Western Desert. In Proceedings of the SPE Annual Technical Conference and Exhibition, Dubai, United Arab Emirates, 26–28 September 2016. [Google Scholar] [CrossRef]
- Dai, C.; Wang, K.; Liu, Y.; Li, H.; Wei, Z.; Zhao, M. Reutilization of fracturing flowback fluids in surfactant flooding for enhanced oil recovery. Energy Fuels 2015, 29, 2304–2311. [Google Scholar] [CrossRef]
- Samanta, A.; Ojha, K.; Mandal, A. Enhanced recovery of oil by eco-friendly natural surfactant and polymer. In Proceedings of the Recent Trends in Engineering & Education, NITTTR, Kolkata, India, 28–29 January 2010. [Google Scholar]
- Mohd, T.A.T.; Taib, N.M.; Adzmi, A.F.; Nik Ab Lah, N.K.I.; Sauki, A.; Jaafar, M.Z. Evaluation of polymer properties for potential selection in enhanced oil recovery. Chem. Eng. Trans. 2018, 65, 343–348. [Google Scholar] [CrossRef]
- Puleo, V.; Sambito, M.; Freni, G. An environmental analysis of the effect of energy saving, production and recovery measures on water supply systems under scarcity conditions. Energies 2015, 8, 5937–5951. [Google Scholar] [CrossRef]
- Piazza, S.; Sambito, M.; Feo, R.; Freni, G.; Puleo, V. Optimal positioning of water quality sensors in water distribution networks: Comparison of numerical and experimental results. In Proceedings of the CCWI 2017–Computing and Control for the Water Industry, Sheffield, UK, 5–7 September 2017. [Google Scholar]
- Shubair, T.; Eljamal, O.; Khalil, A.; Matsunaga, N. Nitrate removal in porous media using nanoscale zero valent iron: Column experiment. In Proceedings of the International Exchange and Innovation Conference on Engineering & Sciences (IEICES), Fukuoka, Japan, 19–20 October 2017. [Google Scholar] [CrossRef]
- Eljamal, O.; Eljamal, R.; Maamoun, I.; Khalil, A.; Shubair, T.; Falyouna, O.; Sugihara, Y. Efficient treatment of ammonia-nitrogen contaminated waters by nano zero-valent iron/zeolite composite. Chemosphere 2021, 287, 131990. [Google Scholar] [CrossRef]
- Recommended Practice for Field Testing Water-Based Drilling Fluids; API Publishing Services: Washington, DC, USA, 2017.
- Welch, S.A.; Sheets, J.M.; Daly, R.A.; Hanson, A.; Sharma, S.; Darrah, T.; Olesik, J.; Lutton, A.; Mouser, P.J.; Wrighton, K.C.; et al. Comparative geochemistry of flowback chemistry from the Utica/Point Pleasant and Marcellus formations. Chem. Geol. 2021, 564, 120041. [Google Scholar] [CrossRef]
- Wang, H.; Lu, L.; Chen, X.; Bian, Y.; Ren, Z.J. Geochemical and microbial characterizations of flowback and produced water in three shale oil and gas plays in the central and western United States. Water Res. 2019, 164, 114942. [Google Scholar] [CrossRef]
- Haluszczak, L.O.; Rose, A.W.; Kump, L.R. Geochemical evaluation of flowback brine from Marcellus gas wells in Pennsylvania, USA. Appl. Geochem. 2013, 28, 55–61. [Google Scholar] [CrossRef]
- Fu, Y.; Jiang, Y.; Hu, Q.; Luo, T.; Li, Y.; Zhian, L.; Wang, Z.; Yin, X. Fracturing flowback fluids from shale gas wells in western chongqing: Geochemical analyses and relevance for exploration & development. J. Nat. Gas Sci. Eng. 2021, 88, 103821. [Google Scholar] [CrossRef]
- Lester, Y.; Ferrer, I.; Thurman, E.M.; Sitterley, K.A.; Korak, J.; Aiken, G.; Linden, K.G. Characterization of hydraulic fracturing flowback water in Colorado: Implications for water treatment. Sci. Total Environ. 2015, 512–513, 637–644. [Google Scholar] [CrossRef]
- Burnside, N.M.; Westaway, R.; Banks, D.; Zimmermann, G.; Hofmann, H.; Boyce, A.J. Rapid water-rock interactions evidenced by hydrochemical evolution of flowback fluid during hydraulic stimulation of a deep geothermal borehole in granodiorite: Pohang, Korea. Appl. Geochem. 2019, 111, 104445. [Google Scholar] [CrossRef]
- Jeng, A.S. Weathering of some Norwegian Alum Shales, II. Laboratory simulations to study the influence of aging, acidification and liming on heavy metal release. Acta Agric. Scand. B-Plant Soil Sci. 1992, 2, 76–87. [Google Scholar] [CrossRef]
- Chorghe, D.; Sari, M.A.; Chellam, S. Boron removal from hydraulic fracturing wastewater by aluminum and iron coagulation: Mechanisms and limitations. Water Res. 2017, 126, 481–487. [Google Scholar] [CrossRef]
- Ozkan, A.; Kaplan, B.M. Investigation of the effects on rheological and filtration properties of water-based drillingmud of boron minerals: An experimental study. Pamukkale Üniversitesi Mühendislik Bilimleri Dergisi 2019, 25, 884–888. [Google Scholar] [CrossRef]
- Sun, C.; Zhang, Y.; Alessi, D.S.; Martin, J.W. Nontarget profiling of organic compounds in a temporal series of hydraulic fracturing flowback and produced waters. Environ. Int. 2019, 131, 104944. [Google Scholar] [CrossRef] [PubMed]
- Kahrilas, G.A.; Blotevogel, J.; Corrin, E.R.; Borch, T. Downhole transformation of the hydraulic fracturing fluid biocide glutaraldehyde: Implications for flowback and produced water quality. Environ. Sci. Technol. 2016, 50, 11414–11423. [Google Scholar] [CrossRef]
- Jiang, Q.; Rentschler, J.; Perrone, R.; Liu, K. Application of ceramic membrane and ion-exchange for the treatment of the flowback water from Marcellus shale gas production. J. Membr. Sci. 2013, 431, 55–61. [Google Scholar] [CrossRef]
- Michel, M.; Rzeczek, L. Pre-treatment of flowback water to desalination. In Membranes and Membrane Processes in Environmental Protection. Monographs of the Environmental Engineering Committee; Polish Academy of Sciences: Warsaw, Poland, 2014; pp. 309–321. [Google Scholar]
- Thurman, E.M.; Ferrer, I.; Blotevogel, J.; Borch, T. Analysis of hydraulic fracturing flowback and produced waters using accurate mass: Identification of ethoxylated surfactants. Anal. Chem. 2014, 86, 9653–9661. [Google Scholar] [CrossRef]
- Luek, J.L.; Harir, M.; Schmitt-Kopplin, P.; Mouser, P.J.; Gonsior, M. Temporal dynamics of halogenated organic compounds in Marcellus Shale flowback. Water Res. 2018, 136, 200–206. [Google Scholar] [CrossRef]
- Murali-Mohan, A.; Hartsock, A.; Bibby, K.J.; Hammack, R.W.; Vidic, R.D.; Gregory, K.B. Microbial community changes in hydraulic fracturing fluids and produced water from shale gas extraction. Environ. Sci. Technol. 2013, 47, 13141–13150. [Google Scholar] [CrossRef] [PubMed]
- Pontius, F.W. Water Quality and Treatment, 4th ed.; McGraw-Hill Inc: New York, NY, USA, 1990. [Google Scholar]
- Leopold, P.; Freese, S. A Simple Guide to the Chemistry, Selection and Use of Chemicals for Water and Wastewater Treatment; WRC-Water Research Commission: Pretoria, South Africa, 2009; pp. 45–47. [Google Scholar]
- Dastgheib, S.A.; Knutson, C.; Yang, Y.; Salih, H.H. International Journal of Greenhouse Gas Control Treatment of produced water from an oilfield and selected coal mines in the Illinois Basin. Int. J. Greenh. Gas Control 2016, 54, 513–523. [Google Scholar] [CrossRef]
- Agarwal, P.; Gupta, A.; Dasgupta, S.; Thummar, D.; Satyarthi, R.S.; Joshi, M.; Chauhan, S.S.; Mishra, J.; Parasher, A.; Tiwari, S.; et al. Waste water treatment by chemical coagulation method-increasing output and reducing environmental impact. In Proceedings of the SPE International Conference and Exhibition on Health, Safety, Security, Environment, and Social Responsibility, Abu Dhabi, United Arab Emirates, 16–18 April 2018. [Google Scholar] [CrossRef]
- Hosny, R.; Fathy, M.; Ramzi, M.; Abdel Moghny, T.; Desouky, S.E.M.; Shama, S.A. Treatment of the oily produced water (OPW) using coagulant mixtures. Egypt. J. Pet. 2016, 25, 391–396. [Google Scholar] [CrossRef]
- Raymond, D.L. Water Quality and Treatment-A Handbook of Community Water Supplies, 5th ed.; McGraw-Hill, Inc: New York, NY, USA, 1999. [Google Scholar]
- Chen, P.; Willingham, T.; Al Sowaidi, A.; Stojkovic, D.; Brown, J. Solids loading assessment for produced water reinjection in a carbonate reservoir. In Proceedings of the Paper presented at the Abu Dhabi International Petroleum Exhibition & Conference, Abu Dhabi, United Arab Emirates, 18 November 2018. [Google Scholar] [CrossRef]
- Dai, C.; Wang, K.; Liu, Y.; Fang, J.; Zhao, M. Study on the reutilization of clear fracturing flowback fluids in surfactant flooding with additives for enhanced oil recovery (EOR). PLoS ONE 2014, 9, e113723. [Google Scholar] [CrossRef] [PubMed]

| Function | Component | Amount of Additive, Per 1 m3 of Water |
|---|---|---|
| Bacteria control | reaction products of paraformaldehyde and 2-hydroxypropylamine (ratio 3:2), [MBO] | 0.1 L |
| Clay control | KCl | 20 kg |
| Buffering agent | Ammonium acetate, acetic acid | 1 L |
| Gelling agent | Contains no hazardous substances–guar gum | 3.6 kg |
| Function | Component | Amount of Additive, Per 1 m3 of Water |
|---|---|---|
| Bacteria control | Reaction products of paraformaldehyde and 2-hydroxypropylamine (ratio 3:2); [MBO] | 0.10 L |
| Clay control | KCl | 20 kg |
| Buffering agent | Ammonium acetate, acetic acid | 1 L |
| Gelling agent | Contains no hazardous substances: guar gum | 3.6 kg |
| Surfactant | Isopropyl alcohol, orange sweet, ext. (citrus terpenes) | 2.0 L |
| Buffering agent | Potassium carbonate | 3.2 L |
| Breaker 1 | Ammonium persulfate | 0.48–1.0 kg |
| Breaker 2 | Sodium persulfate | 0.2–2.0 kg |
| Delayed x-linker | Triethanolamine zirconate, propanol, glycerine | 0.6 L |
| Instant X–linker | Disodium octaboratetetrahydrate | 3.6 L |
| Chemicals | Brine | +FGM-1 | +FGM-2 | Mud-1 | Mud-2 | |
|---|---|---|---|---|---|---|
| Flowback brine | 1000 | 1000 | 1000 | 1000 | 1000 | [L] |
| FGM | - | 10 | 20 | 10 | 20 | [g/L] |
| Nano-Amine 016 | - | - | - | 1 | 1 | [g/L] |
| Block | - | - | - | 100 | 100 | [g/L] |
| K2CO3 | - | - | - | to pH~9 | to pH~9 | |
| Antykor | - | - | - | 5 | 5 | [g/L] |
| Parameter | Fresh Water | Flowback Water |
|---|---|---|
| Sand pack porosity, % | 15.6 ± 0.9 | 14.5 ± 0.8 |
| Residual water saturation, % | 21.2 ± 1.2 | 22.3 ± 0.9 |
| Primary oil recovery (under the gravitational force), % | 18.7 ± 1.1 | 19.6 ± 1.3 |
| Oil recovery after flooding, % | 33.5 ± 1.5 | 39.3 ± 1.5 |
| Total oil recovery, % | 52.2 ± 3.5 | 58.9 ± 2.9 |
| Parameter | FGM Solution in Fresh Water | FGM Solution in Brine (Flowback) |
|---|---|---|
| Plastic viscosity, mPa × s | 15 | 10 |
| Apparent viscosity, mPa × s | 27 | 20 |
| Yield point, Pa | 23 | 20 |
| Low shear rate yield point, Pa | 8 | 6 |
| Gel 10 s, Pa | 8 | 6.5 |
| Gel 10 min, Pa | 9 | 7.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knapik, E.; Chruszcz-Lipska, K.; Łukańko, Ł.; Wysocki, S. Reuse of Flowback Water from Hydraulic Fracturing for Drilling Mud Preparation and Secondary Hydrocarbon Recovery. Energies 2021, 14, 5921. https://doi.org/10.3390/en14185921
Knapik E, Chruszcz-Lipska K, Łukańko Ł, Wysocki S. Reuse of Flowback Water from Hydraulic Fracturing for Drilling Mud Preparation and Secondary Hydrocarbon Recovery. Energies. 2021; 14(18):5921. https://doi.org/10.3390/en14185921
Chicago/Turabian StyleKnapik, Ewa, Katarzyna Chruszcz-Lipska, Łukasz Łukańko, and Sławomir Wysocki. 2021. "Reuse of Flowback Water from Hydraulic Fracturing for Drilling Mud Preparation and Secondary Hydrocarbon Recovery" Energies 14, no. 18: 5921. https://doi.org/10.3390/en14185921
APA StyleKnapik, E., Chruszcz-Lipska, K., Łukańko, Ł., & Wysocki, S. (2021). Reuse of Flowback Water from Hydraulic Fracturing for Drilling Mud Preparation and Secondary Hydrocarbon Recovery. Energies, 14(18), 5921. https://doi.org/10.3390/en14185921









