Corrosion Behavior of VM12-SHC Steel in Contact with Solar Salt and Ternary Molten Salt in Accelerated Fluid Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Preparation of the Salt Mixtures
2.1.2. Measurement of Physical–Chemical Properties
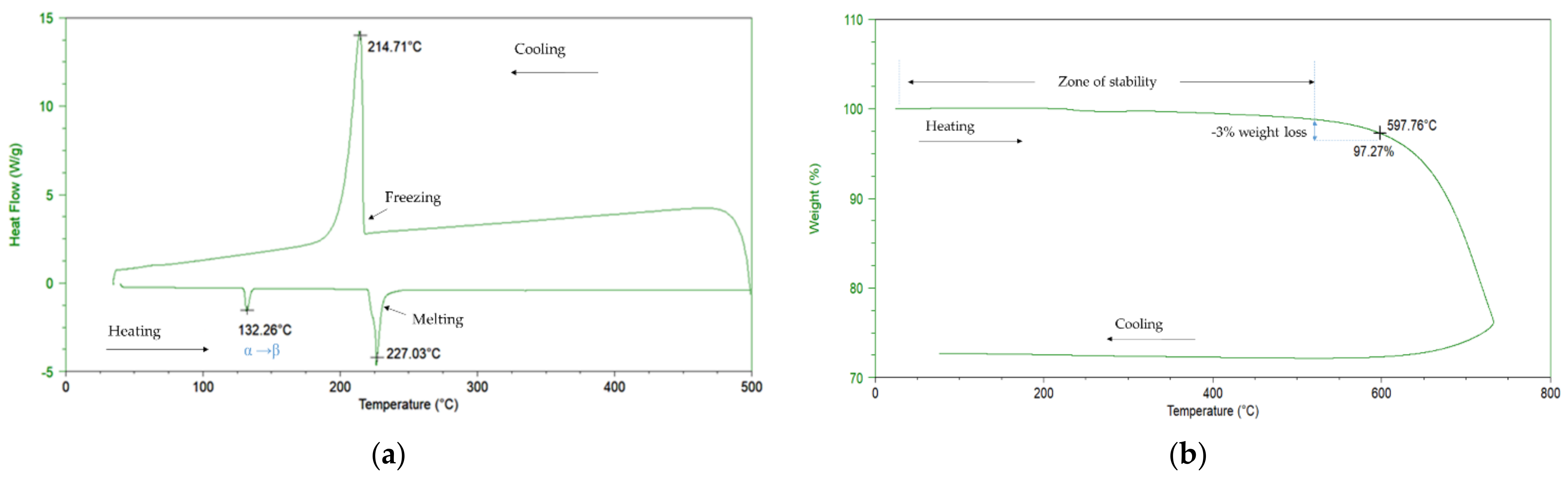
- Melting point and thermal decomposition
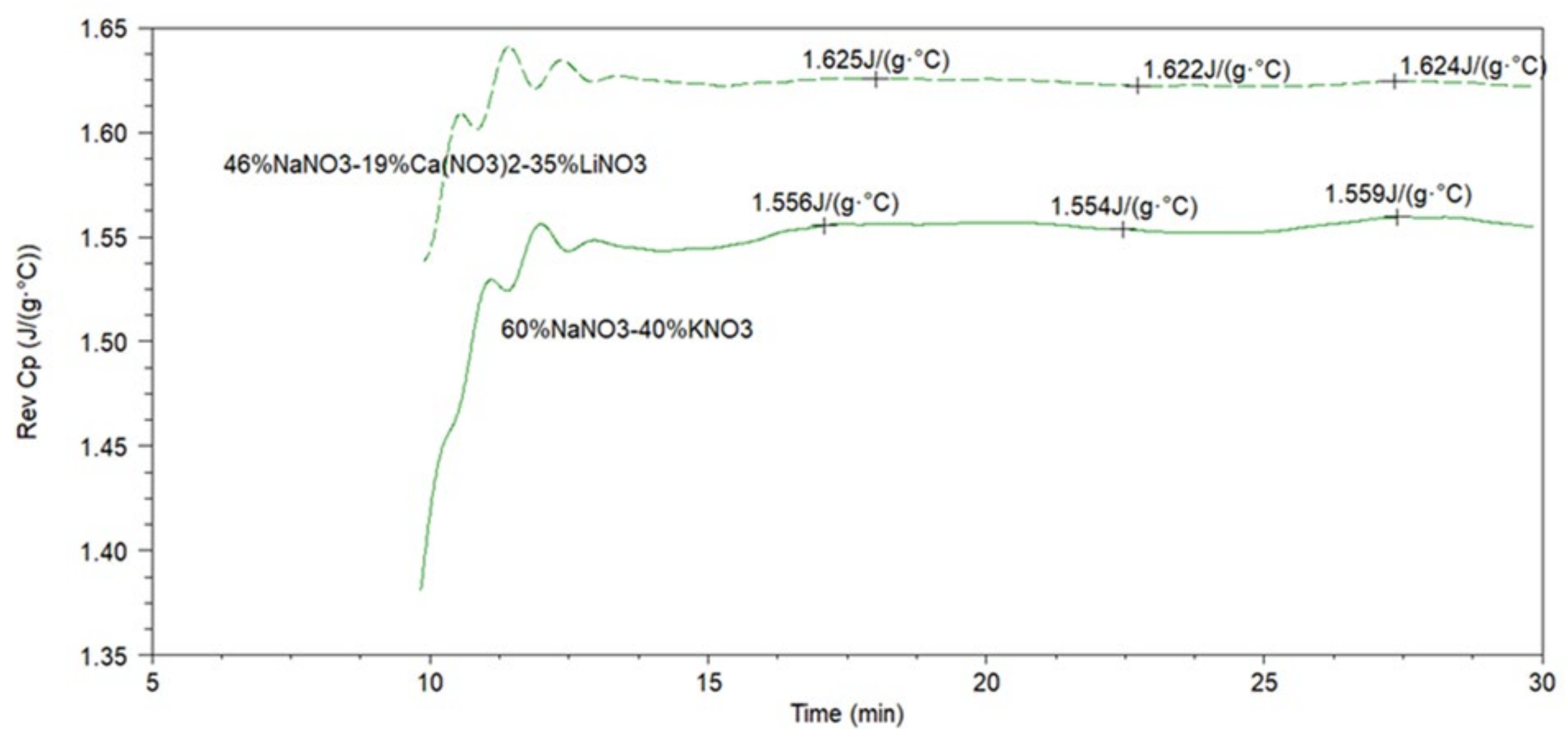
- Specific Heat capacity measurement
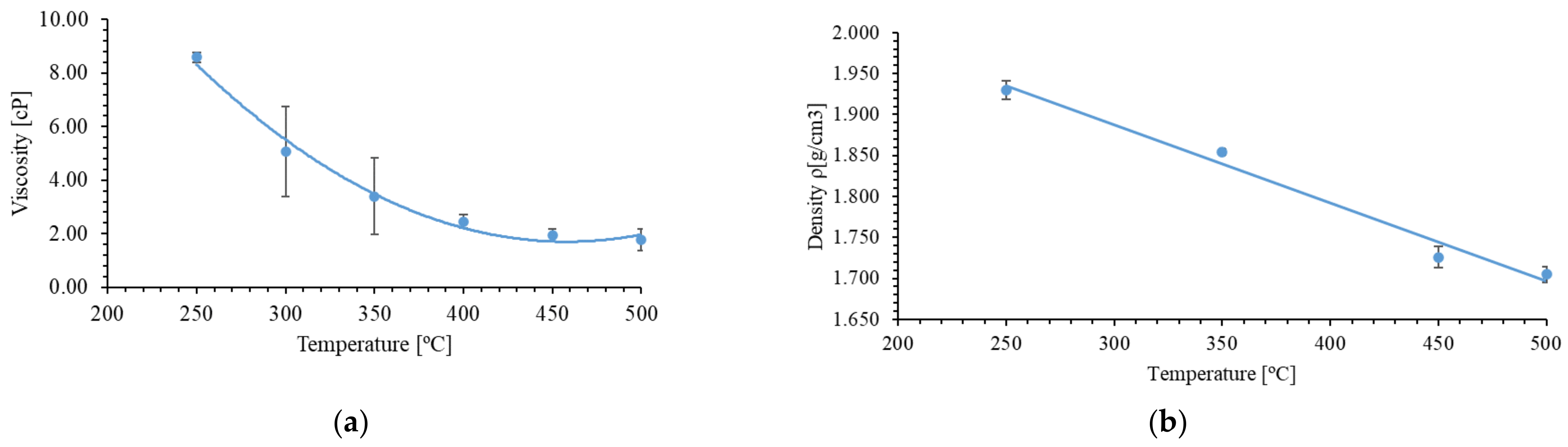
- Density measurement
- Viscosity measurement
2.1.3. Corrosion Study of the Substrates
3. Results and Discussion
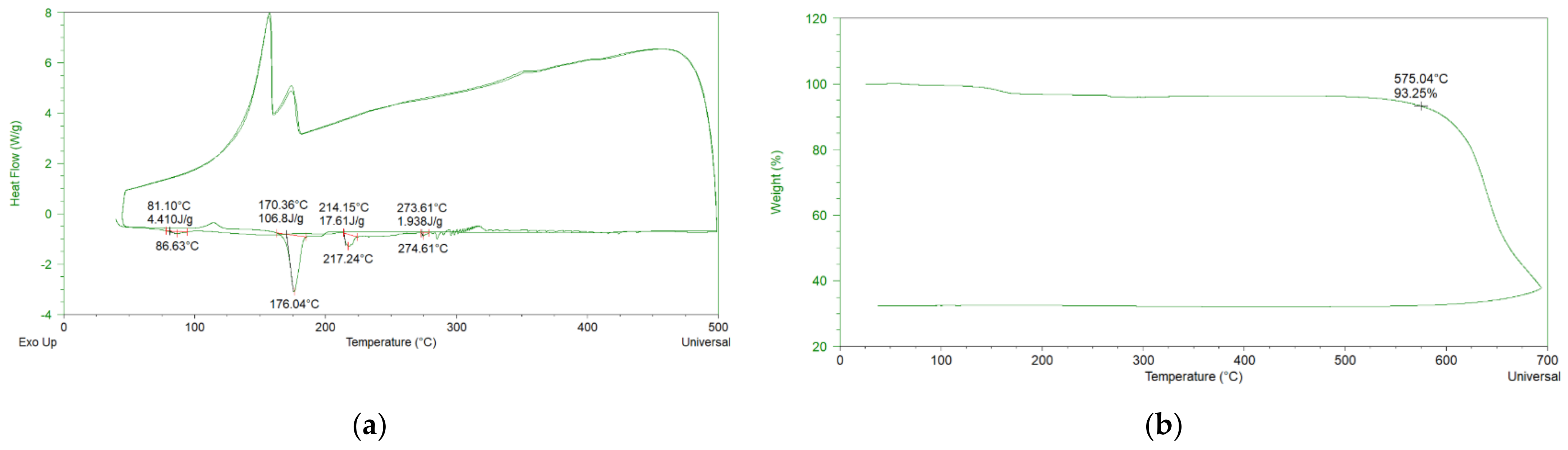
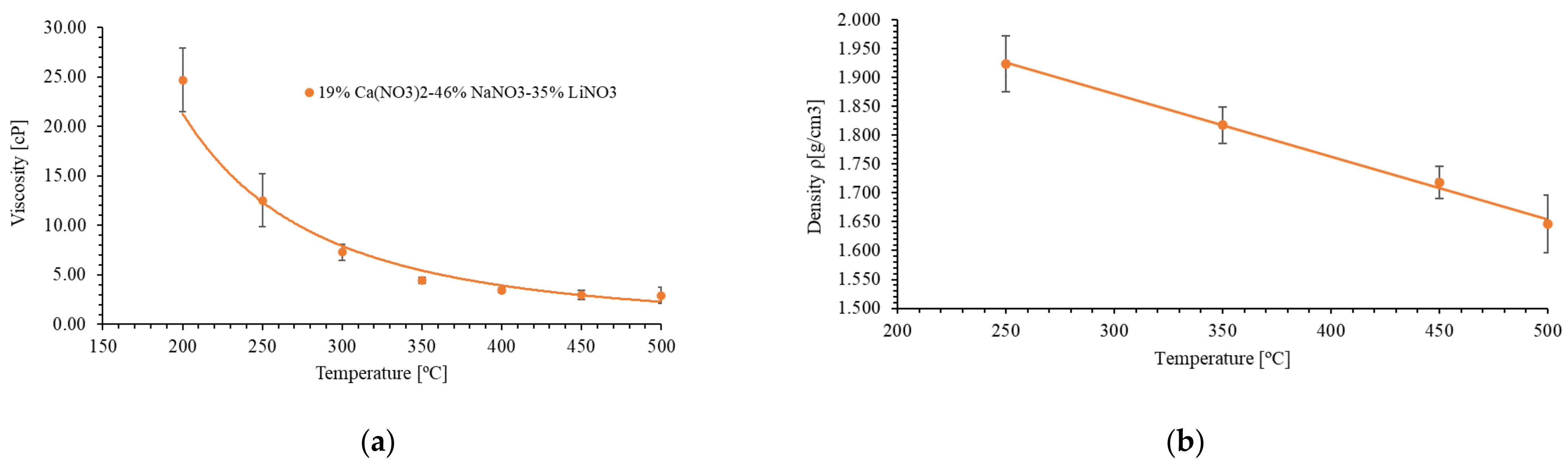
3.1. Characterization of the Salt Mixture
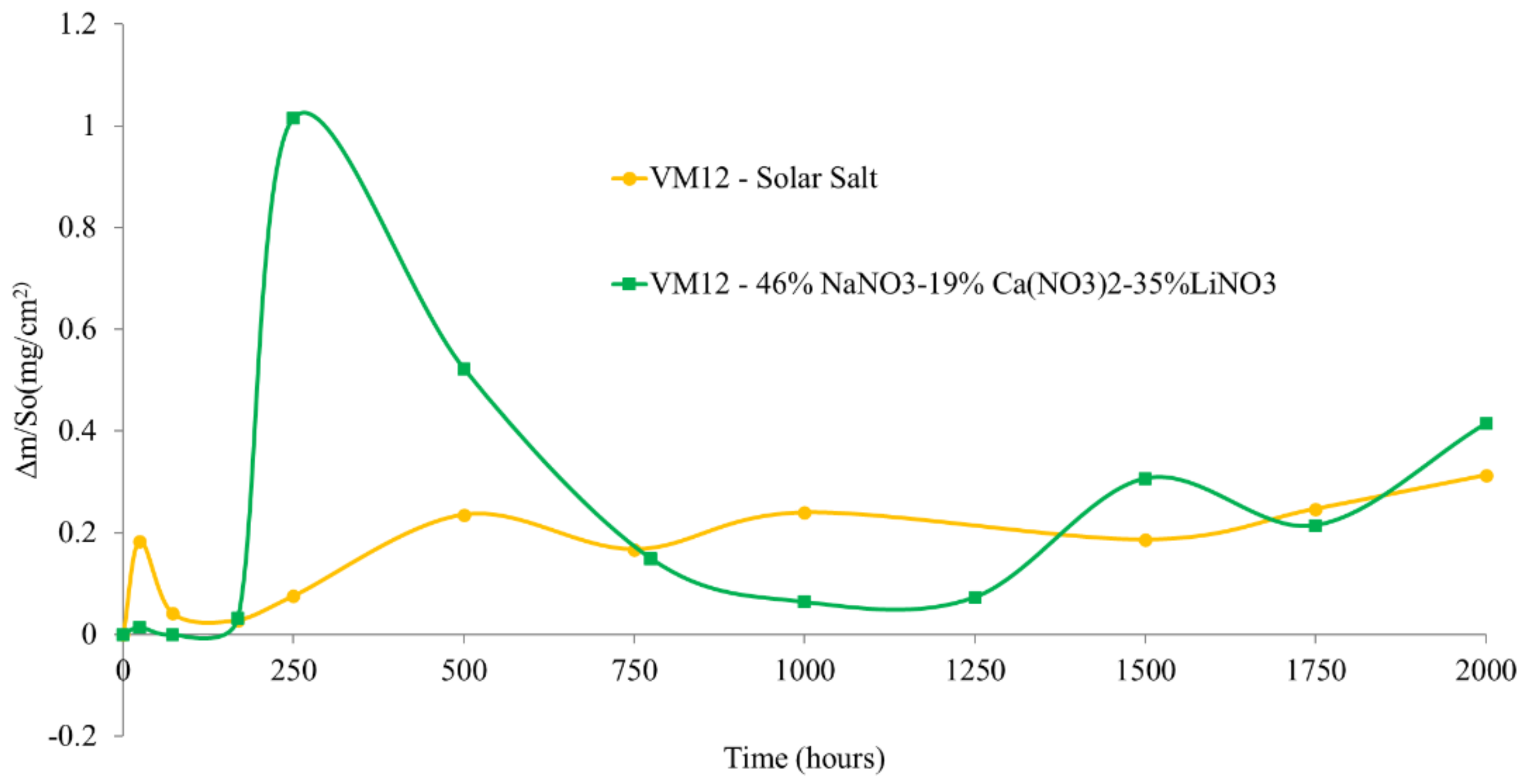
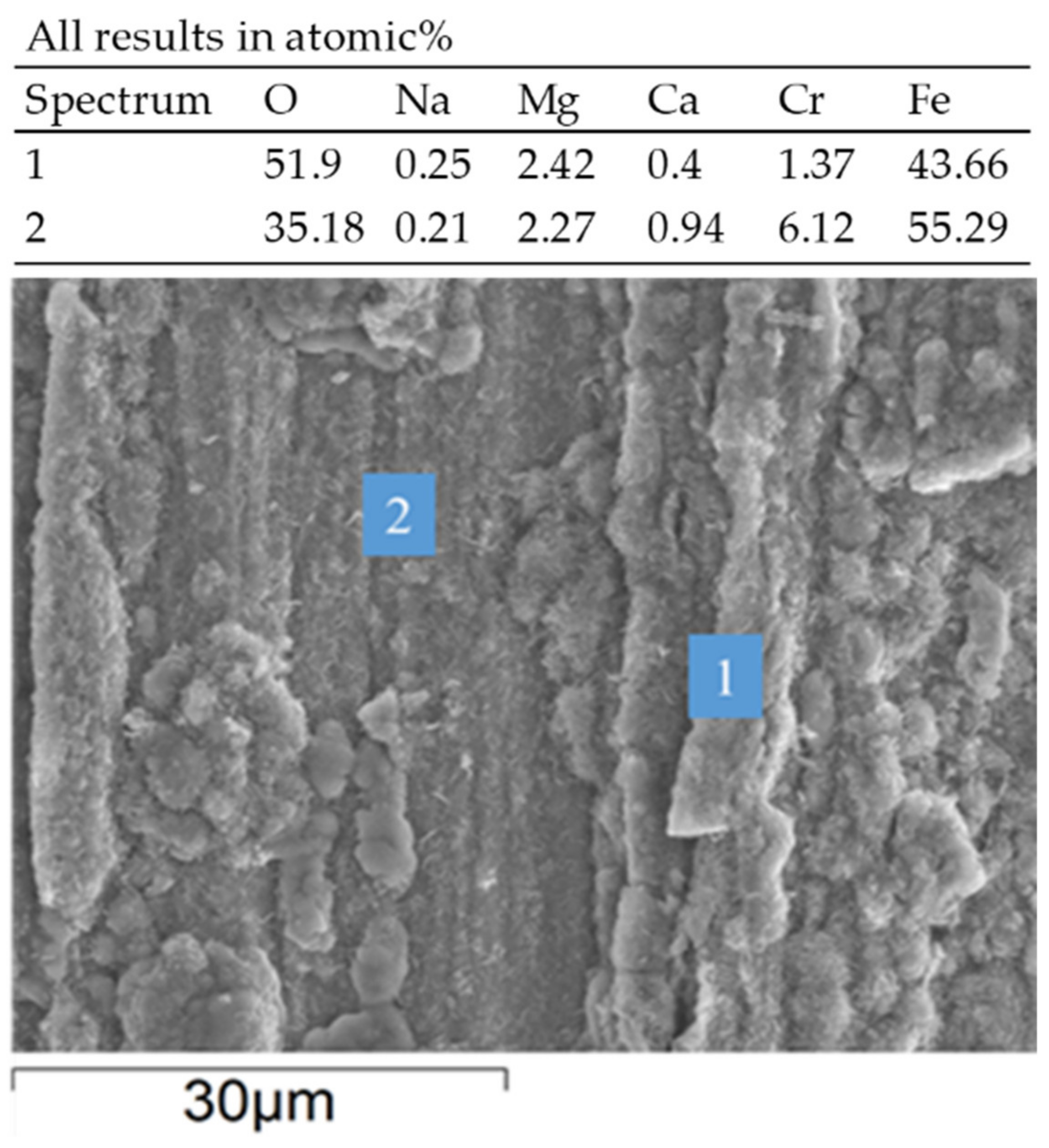
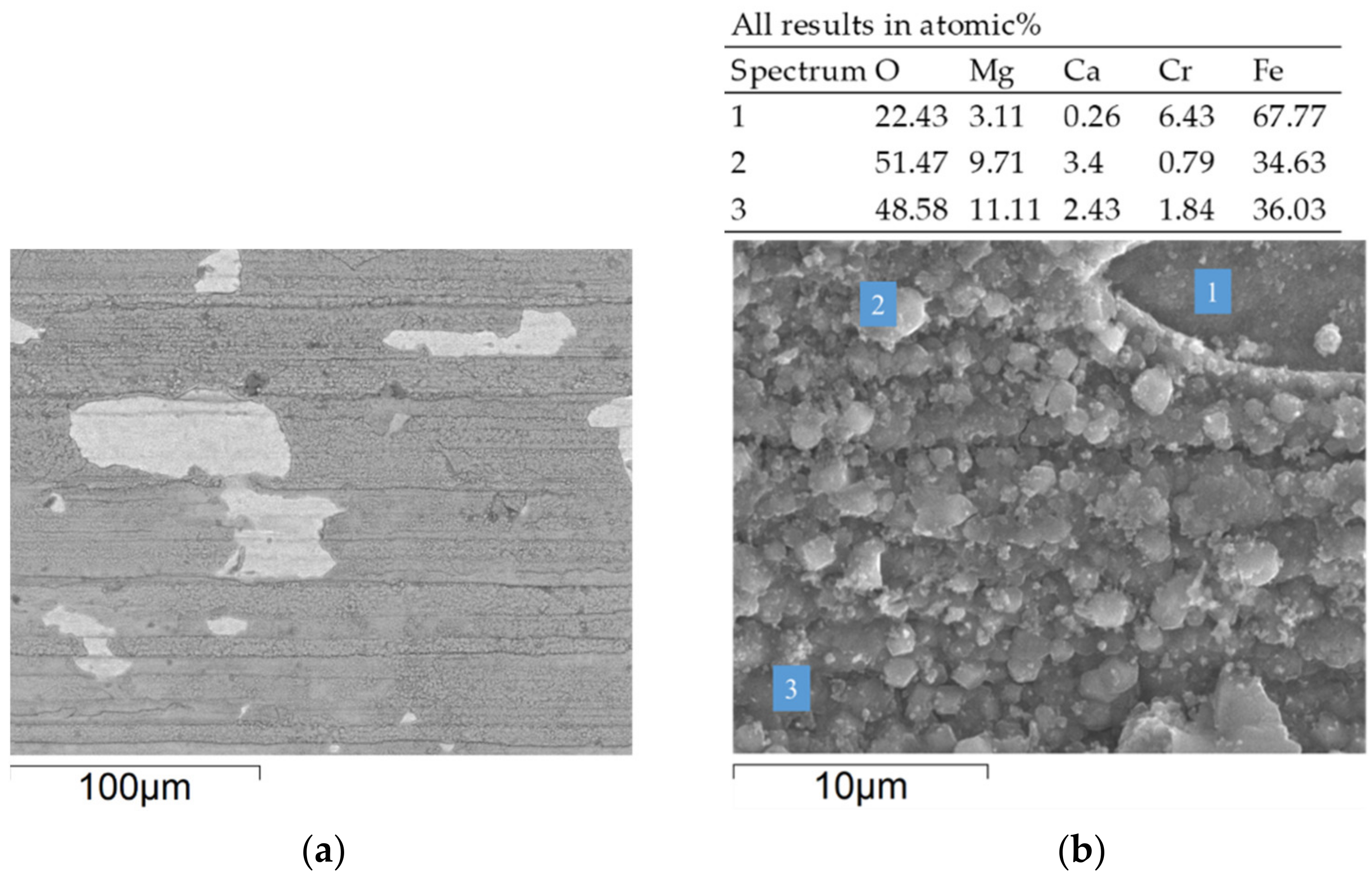
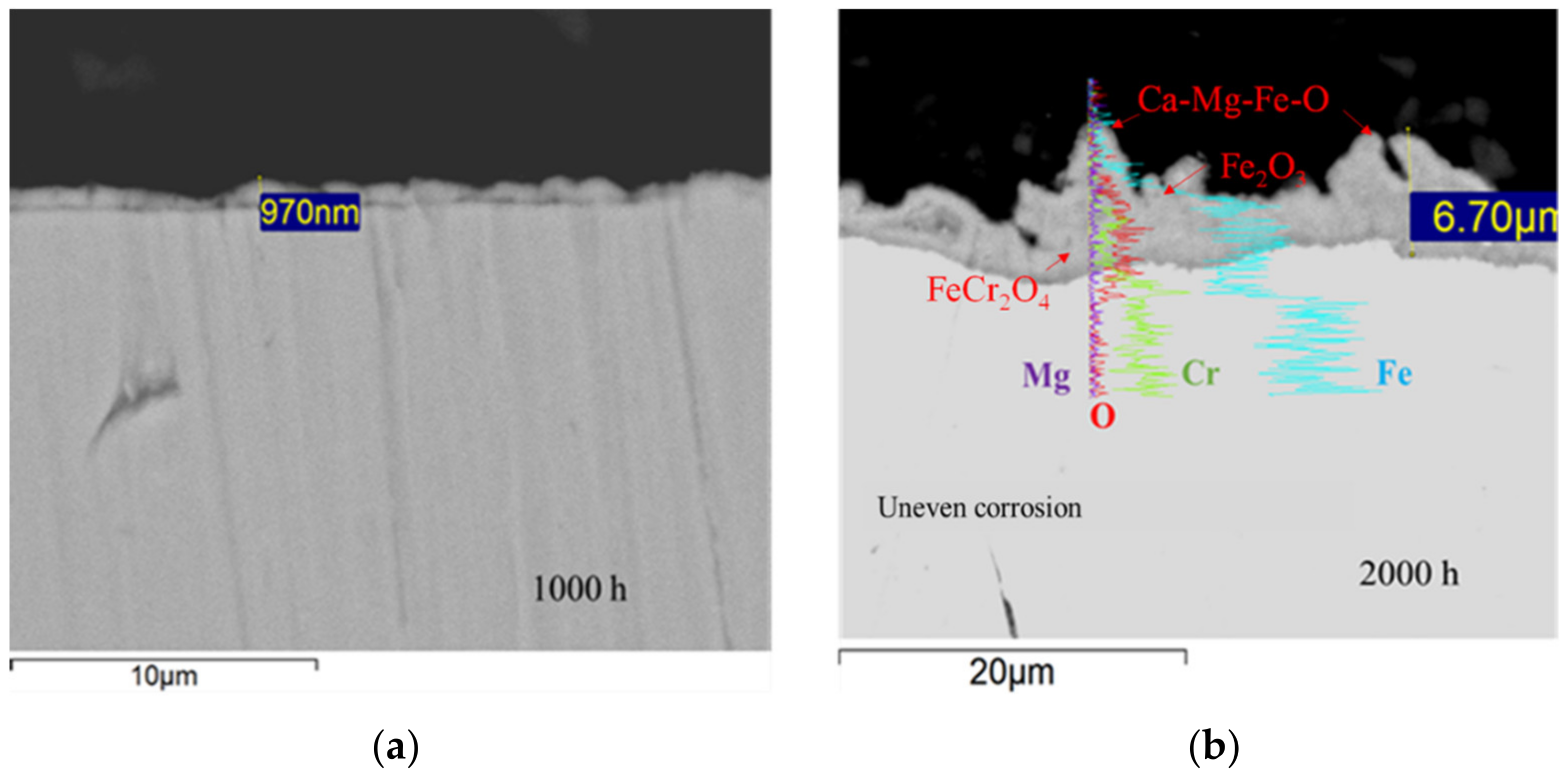
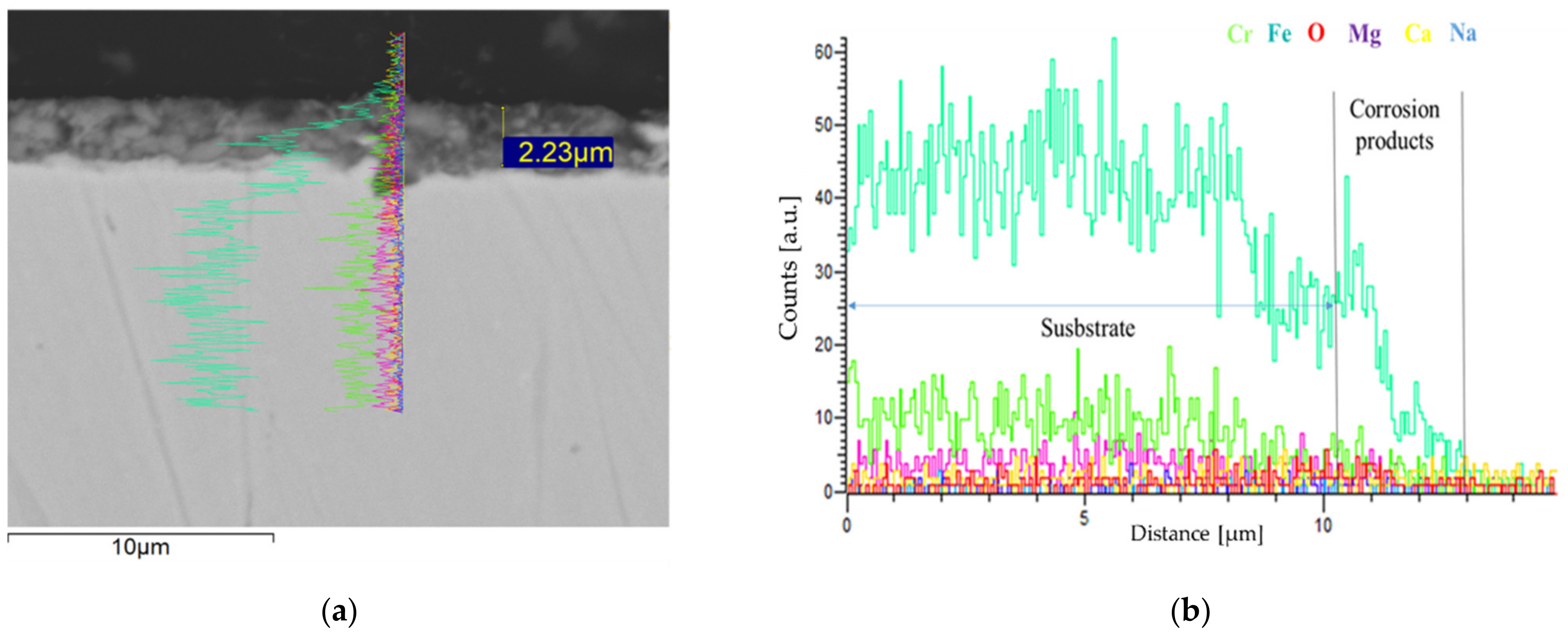
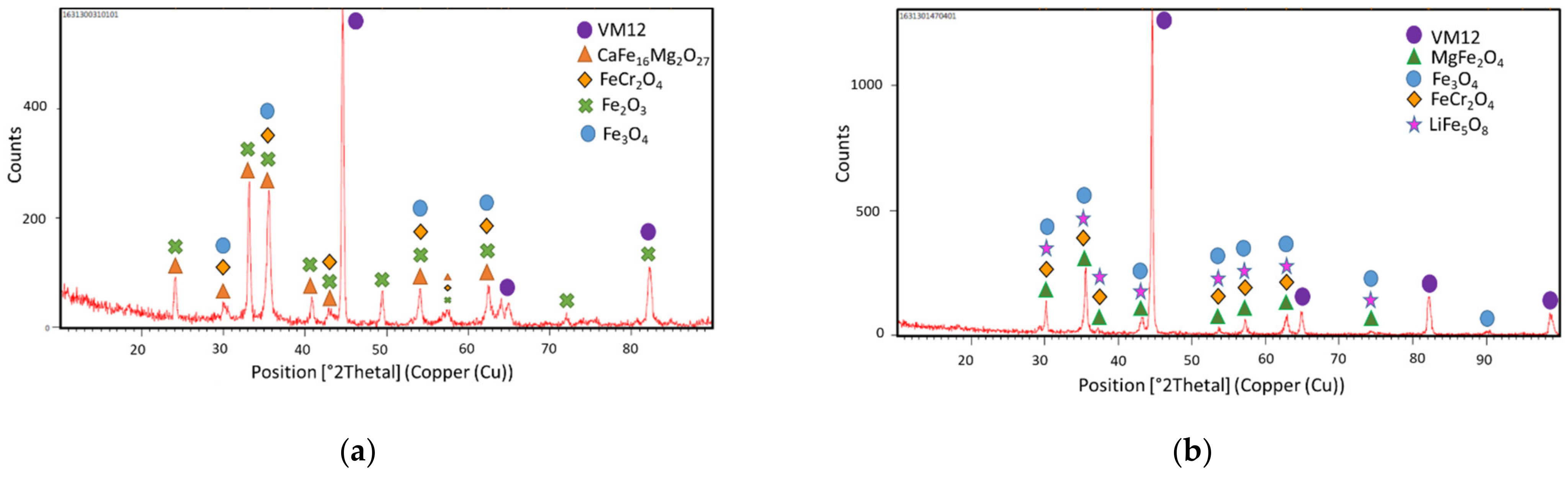
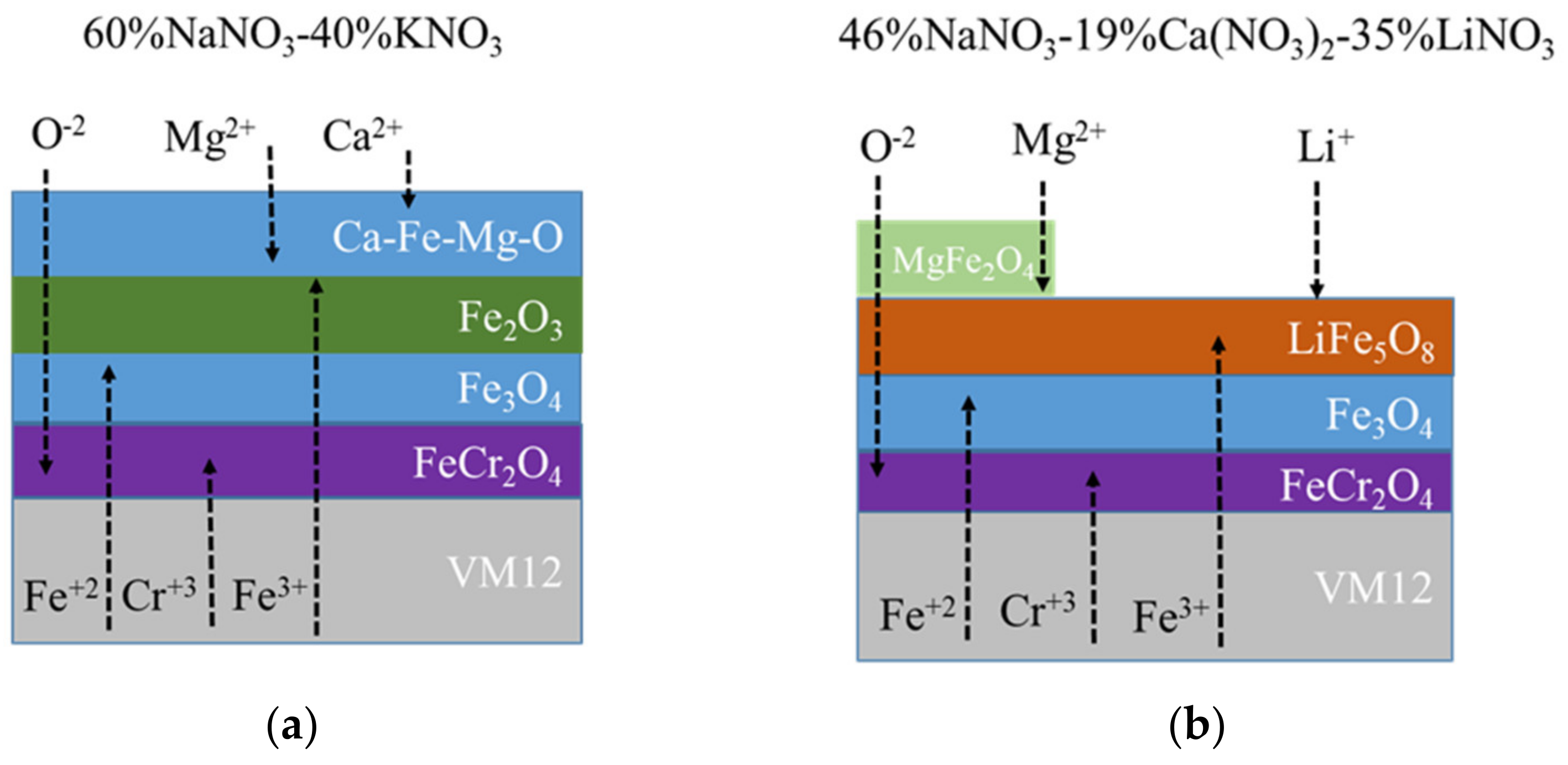
3.2. Corrosion Behavior in VM12
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Renovables, A.d.P.d.E. Impactos Ambientales de la Producción de Electricidad: Estudio Comparativo de Ocho Tecnologías de Generación Eléctrica; Instituto para la Diversificación y Ahorro de la Energía; Ministerio de Ciencia y Tecnología Madrid: Madrid, Spain, 2011. [Google Scholar]
- Blanco, M.J.; Miller, S. Introduction to concentrating solar thermal (CST) technologies. In Advances in Concentrating Solar Thermal Research and Technology; Woodhead Publishing: Sawston, UK, 2017; pp. 3–25. [Google Scholar]
- Pfleger, N.; Braun, M.; Eck, M.; Bauer, T. Assessment of Novel Inorganic Storage Medium with Low Melting Point. Energy Procedia 2015, 69, 988–996. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.Y.; Wu, Z.G. Thermal property characterization of a low melting-temperature ternary nitrate salt mixture for thermal energy storage systems. Sol. Energy Mater. Sol. Cells 2011, 95, 3341–3346. [Google Scholar] [CrossRef]
- Wang, J.; Xu, F.; Han, H.; Zeng, D. Thermodynamic Modeling and Experimental Verification of Eutectic Point in the LiNO3-NaNO3-Ca(NO3)2 Ternary System. J. Phase Equilibria Diffus. 2015, 36, 606–612. [Google Scholar] [CrossRef]
- Mantha, D.; Wang, T.; Reddy, R.G. Thermodynamic modeling of eutectic point in the LiNO3–NaNO3–KNO3–NaNO2 quaternary system. Sol. Energy Mater. Sol. Cells 2013, 118, 18–21. [Google Scholar] [CrossRef]
- Cheng, W.-J.; Chen, D.-J.; Wang, C.-J. High-temperature corrosion of Cr–Mo steel in molten LiNO3–NaNO3–KNO3 eutectic salt for thermal energy storage. Sol. Energy Mater. Sol. Cells 2015, 132, 563–569. [Google Scholar] [CrossRef]
- Bonk, A.; Sau, S.; Uranga, N.; Hernaiz, M. Advanced heat transfer fluids for direct molten salt line-focusing CSP plants. Prog. Energy Combust. Sci. 2018, 67, 69–87. [Google Scholar] [CrossRef]
- Parrado, C.; Marzo, A.; Fuentealba, E.; Fernández, A.G. 2050 LCOE improvement using new molten salts for thermal energy storage in CSP plants. Renew. Sustain. Energy Rev. 2016, 57, 505–514. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Zhao, C.Y. Thermophysical properties of Ca(NO3)2-NaNO3-KNO3 mixtures for heat transfer and thermal storage. Sol. Energy 2017, 146, 172–179. [Google Scholar] [CrossRef]
- Shackelford, J.F. Introducción a la Ciencia de Materiales Para Ingenieros, 6th ed.; Pearson-Prentice Hall: London, UK, 2005. [Google Scholar]
- Walczak, M.; Pineda, F.; Fernández, Á.G.; Mata-Torres, C.; Escobar, R.A. Materials corrosion for thermal energy storage systems in concentrated solar power plants. Renew. Sustain. Energy Rev. 2018, 86, 22–44. [Google Scholar] [CrossRef]
- Delise, T.; Tizzoni, A.; Ferrara, M.; Corsaro, N.; D’Ottavi, C.; Sau, S.; Licoccia, S. Thermophysical, environmental, and compatibility properties of nitrate and nitrite containing molten salts for medium temperature CSP applications: A critical review. J. Eur. Ceram. Soc. 2019, 39, 92–99. [Google Scholar] [CrossRef]
- Fernández, Á.G.; Gomez-Vidal, J.C. Thermophysical properties of low cost lithium nitrate salts produced in northern Chile for thermal energy storage. Renew. Energy 2017, 101, 120–125. [Google Scholar] [CrossRef] [Green Version]
- McConohy, G.; Kruizenga, A. Molten nitrate salts at 600 and 680 °C: Thermophysical property changes and corrosion of high-temperature nickel alloys. Sol. Energy 2014, 103, 242–252. [Google Scholar] [CrossRef]
- Martín, G.G.; Lasanta, M.; Encinas-Sánchez, V.; de Miguel, M.; Trujillo, F.J.P. Evaluation of corrosion resistance of A516 Steel in a molten nitrate salt mixture using a pilot plant facility for application in CSP plants. Sol. Energy Mater. Sol. Cells 2017, 161, 226–231. [Google Scholar] [CrossRef]
- Prieto, C.; Gallardo-Gonzalez, J.; Ruiz-Cabañas, F.J.; Barreneche, C.; Martínez, M.; Segarra, M.; Fernández, A.I. Study of corrosion by Dynamic Gravimetric Analysis (DGA) methodology. Influence of chloride content in solar salt. Sol. Energy Mater. Sol. Cells 2016, 157, 526–532. [Google Scholar] [CrossRef] [Green Version]
- Federsel, K.; Wortmann, J.; Ladenberger, M. High-temperature and Corrosion Behavior of Nitrate Nitrite Molten Salt Mixtures Regarding their Application in Concentrating Solar Power Plants. Energy Procedia 2015, 69, 618–625. [Google Scholar] [CrossRef] [Green Version]
- Audigié, P.; Bizien, N.; Baráibar, I.; Rodríguez, S.; Pastor, A.; Hernández, M.; Agüero, A. Aluminide slurry coatings for protection of ferritic steel in molten nitrate corrosion for concentrated solar power technology. AIP Conf. Proc. 2017, 1850, 70002. [Google Scholar]
- Vallourec. VM12-SHC. Technical Datasheet. 2018. Available online: http://www.vallourec.com/fossilpower/EN/Products/Pages/vm12-shc.aspx (accessed on 15 May 2020).
- Marulanda, J.L.; Castañeda, S.I.; Pérez, F.J. Improvement in Resistance to Steam Oxidation of Aluminide-Coated AISI 304 and AISI 316 Steel Produced by Chemical Vapor Deposition in a Fluidized Bed Reactor. Oxid. Met. 2015, 84, 429–445. [Google Scholar] [CrossRef]
- Malinowski, S.; Jaroszyńska-Wolińska, J.; Herbert, T. Theoretical predictions of anti-corrosive properties of THAM and its derivatives. J. Mol. Modeling 2018, 24, 1–12. [Google Scholar] [CrossRef]
- Sanchez, L.; Bolívar, F.; Hierro, M.; Trilleros, J.; Pérez, F. Effects of reactive gaseous mixture and time on the growth rate and composition of aluminium diffusion coatings by CVD-FBR on 12Cr-ferritic steel. Surf. Coat. Technol. 2007, 201, 7626–7634. [Google Scholar] [CrossRef]
- Agüero, A.; Audigié, P.; Rodríguez, S.; Encinas-Sánchez, V.; De Miguel, M.T.; Perez-Trujillo, F.J. Protective coatings for high temperature molten salt heat storage systems in solar concentration power plants. AIP Conf. Proc. 2018, 2033, 090001. [Google Scholar]
- Fähsing, D.; Oskay, C.; Meißner, T.; Galetz, M. Corrosion testing of diffusion-coated steel in molten salt for concentrated solar power tower systems. Surf. Coat. Technol. 2018, 354, 46–55. [Google Scholar] [CrossRef]
- Reoyo-Prats, R.; Plaza, A.C.; Faugeroux, O.; Claudet, B.; Soum-Glaude, A.; Hildebrandt, C.; Binyamin, Y.; Agüero, A.; Meißner, T. Accelerated aging of absorber coatings for CSP receivers under real high solar flux—Evolution of their optical properties. Sol. Energy Mater. Sol. Cells 2019, 193, 92–100. [Google Scholar] [CrossRef]
- Li, X.L.; Wei, X.; Lu, J.; Ding, J.; Wang, W. Corrosion resistance of 310S and 316L austenitic stainless steel in a quaternary molten salt for concentrating solar power. Energy Procedia 2017, 142, 3590–3596. [Google Scholar] [CrossRef]
- Beneš, O.; Konings, R.; Wurzer, S.; Sierig, M.; Dockendorf, A. A DSC study of the NaNO3–KNO3 system using an innovative encapsulation technique. Thermochim. Acta 2010, 509, 62–66. [Google Scholar] [CrossRef]
- Ercole, D.; Manca, O.; Vafai, K. An investigation of thermal characteristics of eutectic molten salt-based nanofluids. Int. Commun. Heat Mass Transf. 2017, 87, 98–104. [Google Scholar] [CrossRef]
- Olivares, R.I.; Edwards, W. LiNO3–NaNO3–KNO3 salt for thermal energy storage: Thermal stability evaluation in different atmospheres. Thermochim. Acta 2013, 560, 34–42. [Google Scholar] [CrossRef]
- Villada, C.; Bonk, A.; Bauer, T.; Bolívar, F. High-temperature stability of nitrate/nitrite molten salt mixtures under different atmospheres. Appl. Energy 2018, 226, 107–115. [Google Scholar] [CrossRef]
- Zhang, P.; Cheng, J.; Jin, Y.; An, X. Evaluation of thermal physical properties of molten nitrate salts with low melting temperature. Sol. Energy Mater. Sol. Cells 2018, 176, 36–41. [Google Scholar] [CrossRef]
- Mohammad, M.B.; Brooks, G.A.; Rhamdhani, M.A. Thermal analysis of molten ternary lithium-sodium-potassium nitrates. Renew. Energy 2017, 104, 76–87. [Google Scholar] [CrossRef]
- Dudda, B.; Shin, D. Effect of nanoparticle dispersion on specific heat capacity of a binary nitrate salt eutectic for concentrated solar power applications. Int. J. Therm. Sci. 2013, 69, 37–42. [Google Scholar] [CrossRef]
- Bauer, T.; Bonk, A. Semi-empirical Density Estimations for Binary, Ternary and Multicomponent Alkali Nitrate–Nitrite Molten Salt Mixtures. Int. J. Thermophys. 2018, 39. [Google Scholar] [CrossRef]
- López-González, D.; Valverde, J.L.; Sánchez, P.; Sanchez-Silva, L. Characterization of different heat transfer fluids and degradation study by using a pilot plant device operating at real conditions. Energy 2013, 54, 240–250. [Google Scholar] [CrossRef]
- Roget, F.; Favotto, C.; Rogez, J. Study of the KNO3–LiNO3 and KNO3–NaNO3–LiNO3 eutectics as phase change materials for thermal storage in a low-temperature solar power plant. Sol. Energy 2013, 95, 155–169. [Google Scholar] [CrossRef]
- Lu-Lu, C.; Xia, C.; Yu-Ting, W.; Xin, W.; Chong-Fang, C. Experimental study of thermophysical properties and thermal stability of quaternary nitrate molten salts for thermal energy storage. Sol. Energy Mater. Sol. Cells 2019, 190, 12–19. [Google Scholar]
- Wang, T.; Mantha, D.; Reddy, R.G. Novel low melting point quaternary eutectic system for solar thermal energy storage. Appl. Energy 2013, 102, 1422–1429. [Google Scholar] [CrossRef]
- Jin, Y.; Cheng, J.; An, X.; Su, T.; Zhang, P.; Li, Z. Accurate viscosity measurement of nitrates/nitrites salts for concentrated solar power. Sol. Energy 2016, 137, 385–392. [Google Scholar] [CrossRef]
- Bradshaw, R.W.; Siegel, N.P. Molten Nitrate Salt Development for Thermal Energy Storage in Parabolic Trough Solar Power Systems. In Proceedings of the ASME 2008 2nd International Conference on Energy Sustainability, Jacksonville, FL, USA, 10–14 August 2008; Volume 2, pp. 631–637. [Google Scholar]
- Lappalainen, J.; Hakkarainen, E.; Sihvonen, T.; Rodríguez-García, M.M.; Alopaeus, V. Modelling a molten salt thermal energy system—A validation study. Appl. Energy 2019, 233–234, 126–145. [Google Scholar] [CrossRef]
- Ping, W.; Harrowell, P.; Byrne, N.; Angell, C.A. Composition dependence of the solid state transitions in NaNO3/KNO3 mixtures. Thermochim. Acta 2009, 486, 27–31. [Google Scholar] [CrossRef]
- Gomez, J.C.; Calvet, N.; Starace, A.; Glatzmaier, G.C. Ca(NO3)2—NaNO3—KNO3 Molten Salt Mixtures for Direct Thermal Energy Storage Systems in Parabolic Trough Plants. J. Sol. Energy Eng. 2013, 135, 021016. [Google Scholar] [CrossRef]
- Ni, H.; Wu, J.; Sun, Z.; Lu, G.; Yu, J. Insight into the viscosity enhancement ability of Ca(NO3)2 on the binary molten nitrate salt: A molecular dynamics simulation study. Chem. Eng. J. 2019, 377, 120029. [Google Scholar] [CrossRef]
- Li, Y.; Wu, Y.-T.; Lu, Y.; Ma, C.-F. Novel low melting point binary nitrates for thermal energy storage applications. In Proceedings of the Second Thermal and Fluids Engineering Conference, Las Vegas, NV, USA, 2–5 April 2017. [Google Scholar]
- González-Roubaud, E.; Pérez-Osorio, D.; Prieto, C. Review of commercial thermal energy storage in concentrated solar power plants: Steam vs. molten salts. Renew. Sustain. Energy Rev. 2017, 80, 133–148. [Google Scholar] [CrossRef]
- Gil, A.; Medrano, M.; Martorell, I.; Lázaro, A.; Dolado, P.; Zalba, B.; Cabeza, L.F. State of the art on high temperature thermal energy storage for power generation. Part 1—Concepts, materials and modellization. Renew. Sustain. Energy Rev. 2010, 14, 31–55. [Google Scholar] [CrossRef]
- Paulik, J.; Paulik, F.; Arnold, M. Thermogravimetric examination of the dehydration of calcium nitrate tetrahydrate under quasiisothermal and quasi-isobaric conditions. J. Therm. Anal. Calorim. 1983, 27, 409–418. [Google Scholar] [CrossRef]
- Wang, T.; Mantha, D.; Reddy, R.G. Thermal stability of the eutectic composition in LiNO3–NaNO3–KNO3 ternary system used for thermal energy storage. Sol. Energy Mater. Sol. Cells 2012, 100, 162–168. [Google Scholar] [CrossRef]
- Coscia, K.; Nelle, S.; Elliott, T.; Mohapatra, S.; Oztekin, A.; Neti, S. Thermophysical Properties of LiNO3–NaNO3–KNO3 Mixtures for Use in Concentrated Solar Power. J. Sol. Energy Eng. 2013, 135, 034506. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Gutierrez, A.; Barreneche, C.; Ushak, S.; Fernandez, A.I.; Fernádez, A.I.; Grágeda, M. Lithium in thermal energy storage: A state-of-the-art review. Renew. Sustain. Energy Rev. 2015, 42, 1106–1112. [Google Scholar] [CrossRef] [Green Version]
- Wang, T. High themal energy storage density molten salts for parabolic trough solar power generation. In Metallurgical and Materials Engineering; University of Alabama: Tuscaloosa, AL, USA, 2011. [Google Scholar]
- Fernández, A.G.; Muñoz-Sánchez, B.; Nieto-Maestre, J.; García-Romero, A. High temperature corrosion behavior on molten nitrate salt-based nanofluids for CSP plants. Renew. Energy 2019, 130, 902–909. [Google Scholar] [CrossRef]
- Fernández, A.G.; Cabeza, L.F. Molten salt corrosion mechanisms of nitrate based thermal energy storage materials for concentrated solar power plants: A review. Sol. Energy Mater. Sol. Cells 2019, 194, 160–165. [Google Scholar] [CrossRef]
- Ruiz-Cabañas, F.J.; Prieto, C.; Osuna, R.; Madina, V.; Fernández, A.I.; Cabeza, L.F. Corrosion testing device for in-situ corrosion characterization in operational molten salts storage tanks: A516 Gr70 carbon steel performance under molten salts exposure. Sol. Energy Mater. Sol. Cells 2016, 157, 383–392. [Google Scholar] [CrossRef]
- Audigié, P.; Encinas-Sánchez, V.; Juez-Lorenzo, M.; Rodríguez, S.; Gutiérrez, M.; Perez-Trujillo, F.J.; Agüero, A. High temperature molten salt corrosion behavior of aluminide and nickel-aluminide coatings for heat storage in concentrated solar power plants. Surf. Coat. Technol. 2018, 349, 1148–1157. [Google Scholar] [CrossRef]
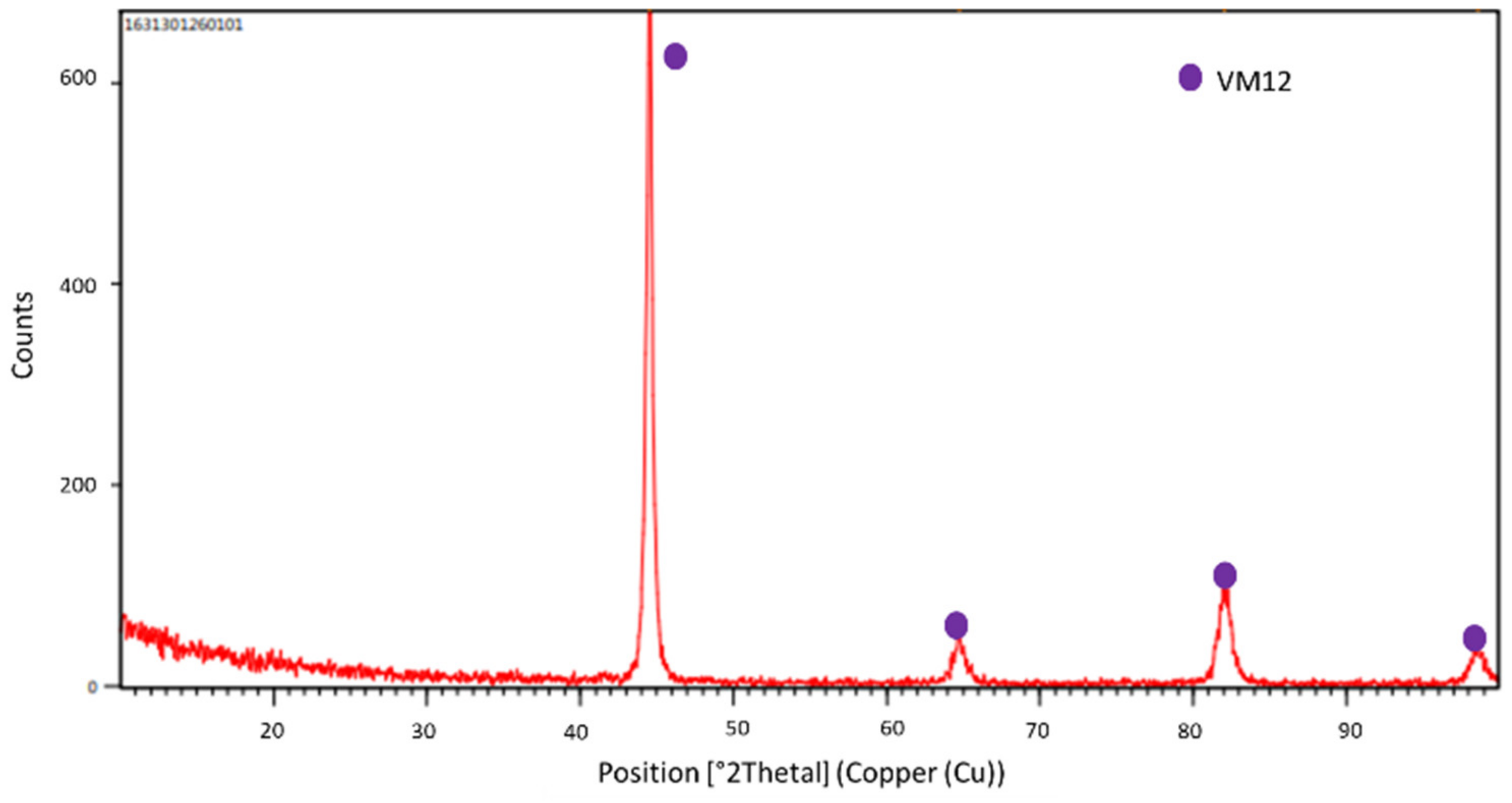

| Chemicals | Purity (%) | Cl− (ppm) | SO4−2 (ppm) | NO2− (ppm) | CO3−2 (ppm) | Mg (ppm) |
|---|---|---|---|---|---|---|
| KNO3 | 99–100 | 10 | 100 | ≤5 | - | ≤10 |
| Ca(NO3)2 | 98 | 52 | 364 | - | - | ≤5 |
| NaNO3 | 99.5 | 250 | 50 | 50 | 400 | ≤5 |
| LiNO3 | 98 | 50 | 500 | - | 50 | 10 |
| Molten Salt Composition (wt.%) | Melting Point (°C) | Stability Limit (°C) | Spec. Heat Capacity (J/(g°C)) | Density (g/cm3) | Viscosity (cP) | Energetic Density (MJ/m3) |
|---|---|---|---|---|---|---|
| 60% NaNO3-40% KNO3 | 227 | 597 | 1.55 (500 °C) | 1.930 (250 °C) 1.854 (350 °C) 1.726 (450 °C) 1.705 (500 °C) | 1.12 (500 °C) | 721 (500 °C) |
| 46% NaNO3-19% Ca(NO3)2-35% LiNO3 | 176 | 575 | 1.62 | 1.923 (250 °C) 1.817 (350 °C) 1.718 (450 °C) 1.647 (500 °C) | 2.93 (500 °C) | 861 (500 °C) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Martin, G.; Lasanta, M.I.; de Miguel, M.T.; Sánchez, A.I.; Pérez-Trujillo, F.J. Corrosion Behavior of VM12-SHC Steel in Contact with Solar Salt and Ternary Molten Salt in Accelerated Fluid Conditions. Energies 2021, 14, 5903. https://doi.org/10.3390/en14185903
García-Martin G, Lasanta MI, de Miguel MT, Sánchez AI, Pérez-Trujillo FJ. Corrosion Behavior of VM12-SHC Steel in Contact with Solar Salt and Ternary Molten Salt in Accelerated Fluid Conditions. Energies. 2021; 14(18):5903. https://doi.org/10.3390/en14185903
Chicago/Turabian StyleGarcía-Martin, Gustavo, María I. Lasanta, María T. de Miguel, Andre Illana Sánchez, and Francisco J. Pérez-Trujillo. 2021. "Corrosion Behavior of VM12-SHC Steel in Contact with Solar Salt and Ternary Molten Salt in Accelerated Fluid Conditions" Energies 14, no. 18: 5903. https://doi.org/10.3390/en14185903
APA StyleGarcía-Martin, G., Lasanta, M. I., de Miguel, M. T., Sánchez, A. I., & Pérez-Trujillo, F. J. (2021). Corrosion Behavior of VM12-SHC Steel in Contact with Solar Salt and Ternary Molten Salt in Accelerated Fluid Conditions. Energies, 14(18), 5903. https://doi.org/10.3390/en14185903






