Investigation of Opto-Electronic Properties and Stability of Mixed-Cation Mixed-Halide Perovskite Materials with Machine-Learning Implementation
Abstract
:1. Introduction
2. Mixed-Cation Mixed-Halide Perovskites
3. Methods
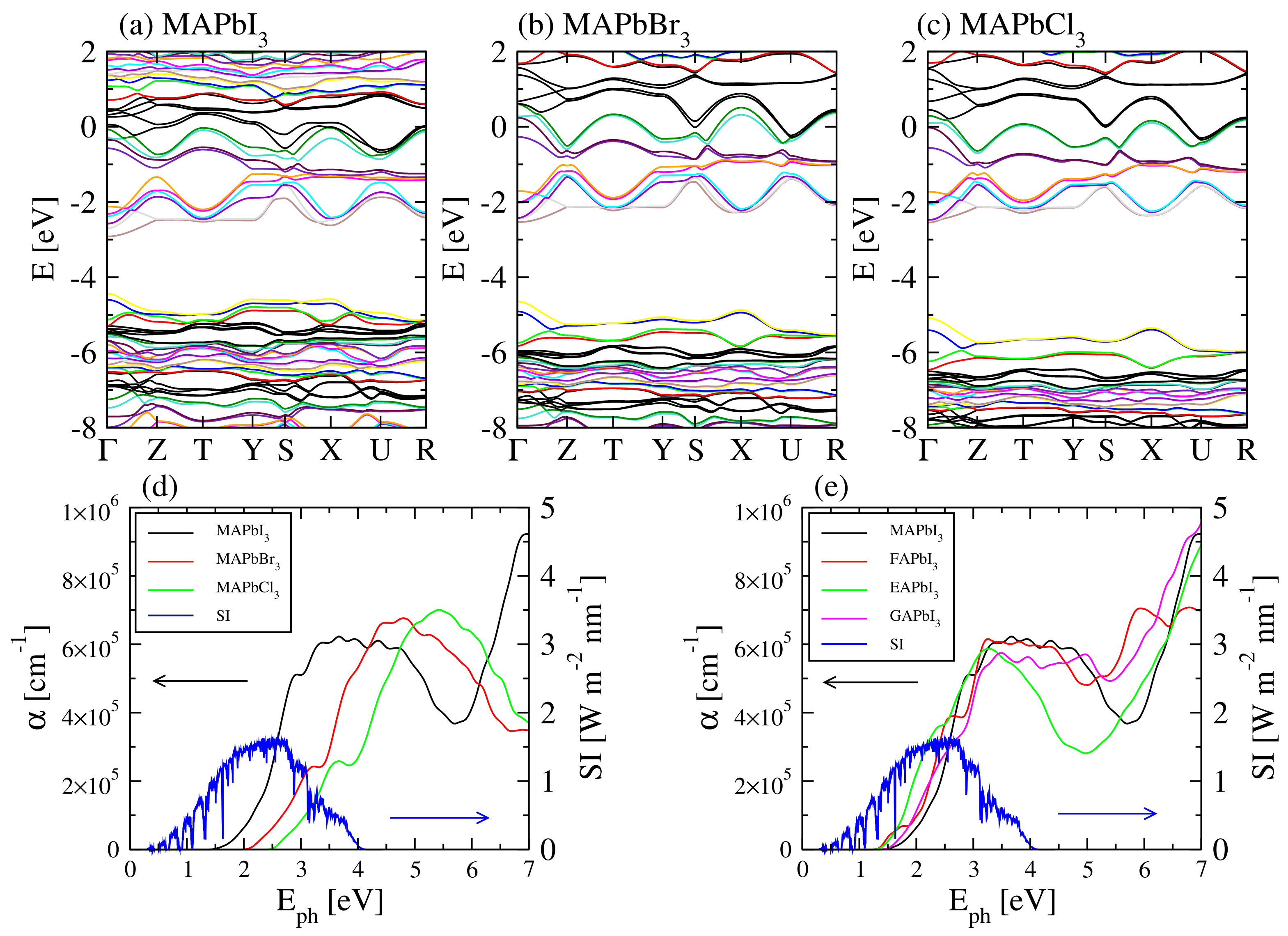
3.1. Density Functional Theory Calculations
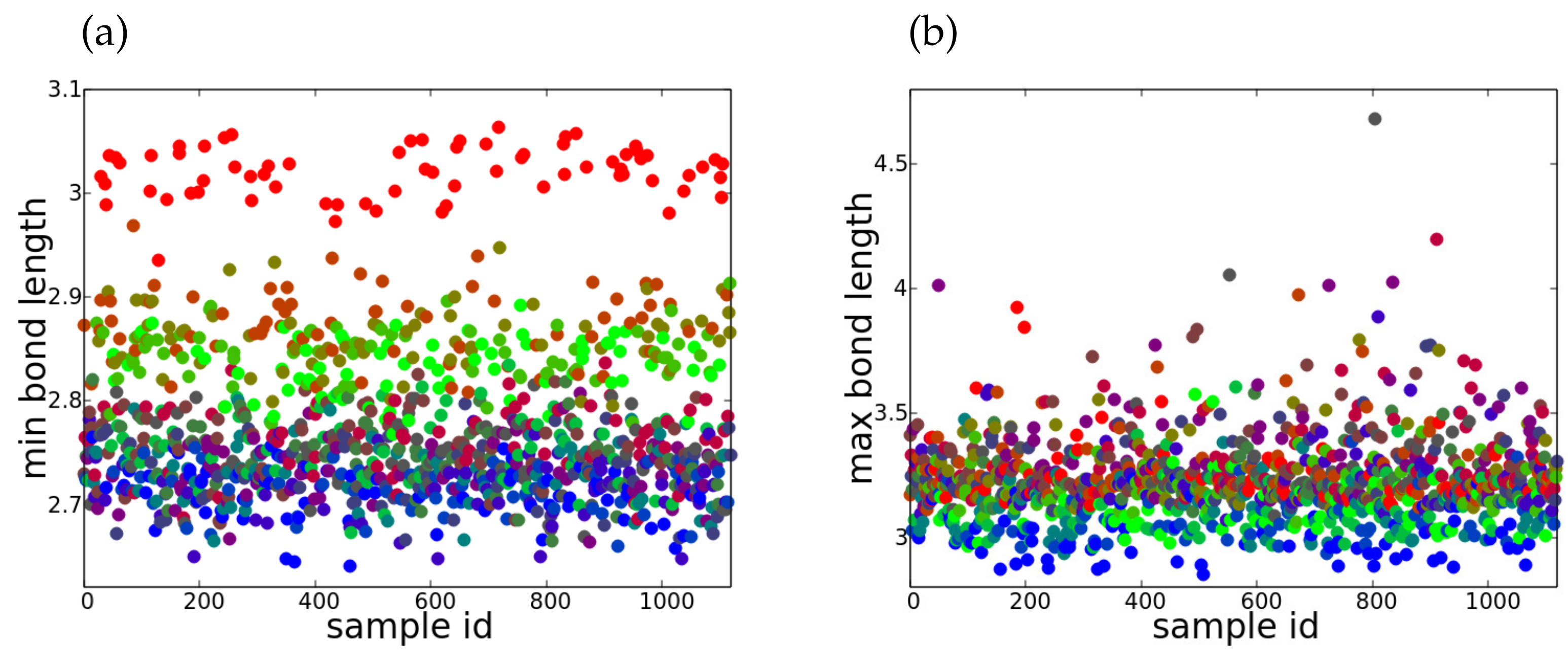
3.2. Assessment of 3D Perovskite Structures, Opto-Electronic Properties and Stability
3.3. Machine-Learning Models
4. Results
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qiu, L.; Ono, L.K.; Qi, Y. Advances and challenges to the commercialization of organic–inorganic halide perovskite solar cell technology. Mater. Today Energy 2018, 7, 169–189. [Google Scholar] [CrossRef]
- Aranda, C.A.; Caliò, L.; Salado, M. Toward Commercialization of Stable Devices: An Overview on Encapsulation of Hybrid Organic-Inorganic Perovskite Solar Cells. Crystals 2021, 11, 519. [Google Scholar] [CrossRef]
- Kundu, S.; Kelly, T.L. In situ studies of the degradation mechanisms of perovskite solar cells. EcoMat 2020, 2, e12025. [Google Scholar] [CrossRef]
- Bryant, D.; Aristidou, N.; Pont, S.; Sanchez-Molina, I.; Chotchunangatchaval, T.; Wheeler, S.; Durrant, J.R.; Haque, S.A. Light and oxygen induced degradation limits the operational stability of methylammonium lead triiodide perovskite solar cells. Energy Environ. Sci. 2016, 9, 1655–1660. [Google Scholar] [CrossRef] [Green Version]
- Aristidou, N.; Eames, C.; Sanchez-Molina, I.; Bu, X.; Kosco, J.; Islam, M.S.; Haque, S.A. Fast oxygen diffusion and iodide defects mediate oxygen-induced degradation of perovskite solar cells. Nat. Commun. 2017, 8, 15218. [Google Scholar] [CrossRef]
- Yang, J.; Yuan, Z.; Liu, X.; Braun, S.; Li, Y.; Tang, J.; Gao, F.; Duan, C.; Fahlman, M.; Bao, Q. Oxygen- and Water-Induced Energetics Degradation in Organometal Halide Perovskites. ACS Appl. Mater. Interfaces 2018, 10, 16225–16230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Li, H.; Fu, J.; Liang, T.; Ma, W. Recent Progress on the Stability of Perovskite Solar Cells in a Humid Environment. J. Phys. Chem. C 2020, 124, 27251–27266. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, T.; Li, G.; Zhao, Y. Mixed cation hybrid lead halide perovskites with enhanced performance and stability. J. Mater. Chem. A 2017, 5, 11450–11461. [Google Scholar] [CrossRef]
- Li, Q.; Zhao, Y.; Zhou, W.; Han, Z.; Fu, R.; Lin, F.; Yu, D.; Zhao, Q. Halogen Engineering for Operationally Stable Perovskite Solar Cells via Sequential Deposition. Adv. Energy Mater. 2019, 9, 1902239. [Google Scholar] [CrossRef]
- Yang, L.; Xiao, Y.; Han, G.; Chang, Y.; Zhang, Y.; Hou, W.; Lin, J.Y.; Li, H. Enhanced stability and efficiency of perovskite solar cells via bifunctional group passivation with thiosalicylic acid. Org. Electron. 2020, 81, 105681. [Google Scholar] [CrossRef]
- Stoumpos, C.C.; Cao, D.H.; Clark, D.J.; Young, J.; Rondinelli, J.M.; Jang, J.I.; Hupp, J.T.; Kanatzidis, M.G. Ruddlesden–Popper Hybrid Lead Iodide Perovskite 2D Homologous Semiconductors. Chem. Mater. 2016, 28, 2852–2867. [Google Scholar] [CrossRef]
- Ortiz-Cervantes, C.; Carmona-Monroy, P.; Solis-Ibarra, D. Two-Dimensional Halide Perovskites in Solar Cells: 2D or not 2D? ChemSusChem 2019, 12, 1560–1575. [Google Scholar] [CrossRef] [PubMed]
- Meggiolaro, D.; De Angelis, F. First-Principles Modeling of Defects in Lead Halide Perovskites: Best Practices and Open Issues. ACS Energy Lett. 2018, 3, 2206–2222. [Google Scholar] [CrossRef] [Green Version]
- Brakkee, R.; Williams, R.M. Minimizing Defect States in Lead Halide Perovskite Solar Cell Materials. Appl. Sci. 2020, 10, 3061. [Google Scholar] [CrossRef]
- Roknuzzaman, M.; Zhang, C.; Ostrikov, K.K.; Du, A.; Wang, H.; Wang, L.; Tesfamichael, T. Electronic and optical properties of lead-free hybrid double perovskites for photovoltaic and optoelectronic applications. Sci. Rep. 2019, 9, 718. [Google Scholar] [CrossRef] [Green Version]
- Maiti, A.; Chatterjee, S.; Peedikakkandy, L.; Pal, A.J. Defects and Their Passivation in Hybrid Halide Perovskites toward Solar Cell Applications. Sol. RRL 2020, 4, 2000505. [Google Scholar] [CrossRef]
- Tao, S.X.; Cao, X.; Bobbert, P.A. Accurate and efficient band gap predictions of metal halide perovskites using the DFT-1/2 method: GW accuracy with DFT expense. Sci. Rep. 2017, 7, 14386. [Google Scholar] [CrossRef]
- Frost, J.M.; Butler, K.T.; Brivio, F.; Hendon, C.H.; van Schilfgaarde, M.; Walsh, A. Atomistic Origins of High-Performance in Hybrid Halide Perovskite Solar Cells. Nano Lett. 2014, 14, 2584–2590. [Google Scholar] [CrossRef] [Green Version]
- Tenuta, E.; Zheng, C.; Rubel, O. Thermodynamic origin of instability in hybrid halide perovskites. Sci. Rep. 2016, 6, 37654. [Google Scholar] [CrossRef] [Green Version]
- Ivanov, I.; Steparuk, A.; Bolyachkina, M.; Tsvetkov, D.; Safronov, A.; Zuev, A. Thermodynamics of formation of hybrid perovskite-type methylammonium lead halides. J. Chem. Thermodyn. 2018, 116, 253–258. [Google Scholar] [CrossRef]
- Juarez-Perez, E.J.; Ono, L.K.; Uriarte, I.; Cocinero, E.J.; Qi, Y. Degradation Mechanism and Relative Stability of Methylammonium Halide Based Perovskites Analyzed on the Basis of Acid–Base Theory. ACS Appl. Mater. Interfaces 2019, 11, 12586–12593. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.Y.; Chen, S.; Xu, P.; Xiang, H.; Gong, X.G.; Walsh, A.; Wei, S.H. Intrinsic Instability of the Hybrid Halide Perovskite Semiconductor CH3NH3PbI3. Chin. Phys. Lett. 2018, 35, 036104. [Google Scholar] [CrossRef] [Green Version]
- Senocrate, A.; Kim, G.Y.; Grätzel, M.; Maier, J. Thermochemical Stability of Hybrid Halide Perovskites. ACS Energy Lett. 2019, 4, 2859–2870. [Google Scholar] [CrossRef] [Green Version]
- Ciccioli, A.; Latini, A. Thermodynamics and the Intrinsic Stability of Lead Halide Perovskites CH3NH3PbX3. J. Phys. Chem. Lett. 2018, 9, 3756–3765. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xu, Q.; Sun, Q.; Hou, Z.; Yin, W.J. Thermodynamic Stability Landscape of Halide Double Perovskites via High-Throughput Computing and Machine Learning. Adv. Funct. Mater. 2019, 29, 1807280. [Google Scholar] [CrossRef]
- Park, H.; Ali, A.; Mall, R.; Bensmail, H.; Sanvito, S.; El-Mellouhi, F. Data-driven enhancement of cubic phase stability in mixed-cation perovskites. Mach. Learn. Sci. Technol. 2021, 2, 025030. [Google Scholar] [CrossRef]
- Wu, T.; Wang, J. Deep Mining Stable and Nontoxic Hybrid Organic–Inorganic Perovskites for Photovoltaics via Progressive Machine Learning. ACS Appl. Mater. Interfaces 2020, 12, 57821–57831. [Google Scholar] [CrossRef]
- Le Corre, V.M.; Sherkar, T.S.; Koopmans, M.; Koster, L.J.A. Identification of the dominant recombination process for perovskite solar cells based on machine learning. Cell Rep. Phys. Sci. 2021, 2, 100346. [Google Scholar] [CrossRef]
- Workman, M.; Zhi Chen, D.; Musa, S.M. Machine Learning for Predicting Perovskite Solar Cell Opto-Electronic Properties. In Proceedings of the 2020 International Conference on Data Analytics for Business and Industry: Way Towards a Sustainable Economy (ICDABI), Sakheer, Bahrain, 26–27 October 2020; pp. 1–5. [Google Scholar] [CrossRef]
- Tao, Q.; Xu, P.; Li, M.; Lu, W. Machine learning for perovskite materials design and discovery. Npj Comput. Mater. 2021, 7, 23. [Google Scholar] [CrossRef]
- Bidikoudi, M.; Kymakis, E. Novel approaches and scalability prospects of copper based hole transporting materials for planar perovskite solar cells. J. Mater. Chem. C 2019, 7, 13680–13708. [Google Scholar] [CrossRef] [Green Version]
- Kim, E.B.; Akhtar, M.S.; Shin, H.S.; Ameen, S.; Nazeeruddin, M.K. A review on two-dimensional (2D) and 2D-3D multidimensional perovskite solar cells: Perovskites structures, stability, and photovoltaic performances. J. Photochem. Photobiol. C Photochem. Rev. 2021, 48, 100405. [Google Scholar] [CrossRef]
- Gholipour, S.; Ali, A.M.; Correa-Baena, J.P.; Turren-Cruz, S.H.; Tajabadi, F.; Tress, W.; Taghavinia, N.; Grätzel, M.; Abate, A.; De Angelis, F.; et al. Globularity-Selected Large Molecules for a New Generation of Multication Perovskites. Adv. Mater. 2017, 29, 1702005. [Google Scholar] [CrossRef]
- Kieslich, G.; Sun, S.; Cheetham, A.K. Solid-state principles applied to organic–inorganic perovskites: New tricks for an old dog. Chem. Sci. 2014, 5, 4712–4715. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. Sect. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Raifuku, I.; Chiang, Y.H.; Hou, C.H.; Li, M.H.; Lin, C.F.; Lin, P.Y.; Shyue, J.J.; Chen, P. Formamide iodide: A new cation additive for inhibiting δ-phase formation of formamidinium lead iodide perovskite. Mater. Adv. 2021, 2, 2272–2277. [Google Scholar] [CrossRef]
- D’Annibale, A.; Panetta, R.; Tarquini, O.; Colapietro, M.; Quaranta, S.; Cassetta, A.; Barba, L.; Chita, G.; Latini, A. Synthesis, physico-chemical characterization and structure of the elusive hydroxylammonium lead iodide perovskite NH3OHPbI3. Dalton Trans. 2019, 48, 5397–5407. [Google Scholar] [CrossRef]
- Akbulatov, A.F.; Frolova, L.A.; Anokhin, D.V.; Gerasimov, K.L.; Dremova, N.N.; Troshin, P.A. Hydrazinium-loaded perovskite solar cells with enhanced performance and stability. J. Mater. Chem. A 2016, 4, 18378–18382. [Google Scholar] [CrossRef]
- Han, Q.; Bae, S.H.; Sun, P.; Hsieh, Y.T.; Yang, Y.M.; Rim, Y.S.; Zhao, H.; Chen, Q.; Shi, W.; Li, G.; et al. Single Crystal Formamidinium Lead Iodide (FAPbI3): Insight into the Structural, Optical, and Electrical Properties. Adv. Mater. 2016, 28, 2253–2258. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Dai, N.; Lan, Y. Solution Route to Single-Crystalline Ethylammonium Lead Halide Microstructures. ChemistrySelect 2019, 4, 2174–2180. [Google Scholar] [CrossRef]
- Kim, Y.; Bae, C.; Jung, H.S.; Shin, H. Enhanced stability of guanidinium-based organic-inorganic hybrid lead triiodides in resistance switching. APL Mater. 2019, 7, 081107. [Google Scholar] [CrossRef] [Green Version]
- Becker, M.; Wark, M. Organic Cation Substitution in Hybrid Perovskite CH3NH3PbI3 with Hydroxylammonium (NH3OH+): A First-Principles Study. J. Phys. Chem. C 2018, 122, 3548–3557. [Google Scholar] [CrossRef]
- Li, W.G.; Rao, H.S.; Chen, B.X.; Wang, X.D.; Kuang, D.B. A formamidinium–methylammonium lead iodide perovskite single crystal exhibiting exceptional optoelectronic properties and long-term stability. J. Mater. Chem. A 2017, 5, 19431–19438. [Google Scholar] [CrossRef]
- Xiao, Z.; Wang, Q.; Wu, X.; Wu, Y.; Ren, J.; Xiong, Z.; Yang, X. Efficient light-emitting devices based on mixed-cation lead halide perovskites. Org. Electron. 2020, 77, 105546. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, T.; Li, G.; Xu, F.; Wang, T.; Li, Y.; Yang, Y.; Zhao, Y. A mixed-cation lead iodide MA1-xEAxPbI3 absorber for perovskite solar cells. J. Energy Chem. 2018, 27, 215–218. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Li, Q.; Wu, K. Ethylammonium as an alternative cation for efficient perovskite solar cells from first-principles calculations. RSC Adv. 2019, 9, 7356–7361. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.T.; Liu, E.E.; Xu, A.F.; Yang, L.W.; Chen, J.Y.; Xu, G. Ethylammonium Lead Iodide Formation in MAPbI3 Precursor Solutions by DMF Decomposition and Organic Cation Exchange Reaction. Crystals 2020, 10, 162. [Google Scholar] [CrossRef] [Green Version]
- Jodlowski, A.D.; Roldán-Carmona, C.; Grancini, G.; Salado, M.; Ralaiarisoa, M.; Ahmad, S.; Koch, N.; Camacho, L.; de Miguel, G.; Nazeeruddin, M.K. Large guanidinium cation mixed with methylammonium in lead iodide perovskites for 19% efficient solar cells. Nat. Energy 2017, 2, 972–979. [Google Scholar] [CrossRef] [Green Version]
- Tsarev, S.; Boldyreva, A.G.; Luchkin, S.Y.; Elshobaki, M.; Afanasov, M.I.; Stevenson, K.J.; Troshin, P.A. Hydrazinium-assisted stabilisation of methylammonium tin iodide for lead-free perovskite solar cells. J. Mater. Chem. A 2018, 6, 21389–21395. [Google Scholar] [CrossRef]
- Yu, S.; Liu, H.; Wang, S.; Zhu, H.; Dong, X.; Li, X. Hydrazinium cation mixed FAPbI3-based perovskite with 1D/3D hybrid dimension structure for efficient and stable solar cells. Chem. Eng. J. 2021, 403, 125724. [Google Scholar] [CrossRef]
- Saliba, M.; Matsui, T.; Seo, J.Y.; Domanski, K.; Correa-Baena, J.P.; Nazeeruddin, M.K.; Zakeeruddin, S.M.; Tress, W.; Abate, A.; Hagfeldt, A.; et al. Cesium-containing triple cation perovskite solar cells: Improved stability, reproducibility and high efficiency. Energy Environ. Sci. 2016, 9, 1989–1997. [Google Scholar] [CrossRef] [Green Version]
- Stoddard, R.J.; Rajagopal, A.; Palmer, R.L.; Braly, I.L.; Jen, A.K.Y.; Hillhouse, H.W. Enhancing Defect Tolerance and Phase Stability of High-Bandgap Perovskites via Guanidinium Alloying. ACS Energy Lett. 2018, 3, 1261–1268. [Google Scholar] [CrossRef]
- Boziki, A.; Mladenović, M.; Grätzel, M.; Rothlisberger, U. Why choosing the right partner is important: Stabilization of ternary CsyGUAxFA(1-y-x)PbI3 perovskites. Phys. Chem. Chem. Phys. 2020, 22, 20880–20890. [Google Scholar] [CrossRef]
- Ono, L.K.; Juarez-Perez, E.J.; Qi, Y. Progress on Perovskite Materials and Solar Cells with Mixed Cations and Halide Anions. ACS Appl. Mater. Interfaces 2017, 9, 30197–30246. [Google Scholar] [CrossRef] [Green Version]
- Zarick, H.F.; Soetan, N.; Erwin, W.R.; Bardhan, R. Mixed halide hybrid perovskites: A paradigm shift in photovoltaics. J. Mater. Chem. A 2018, 6, 5507–5537. [Google Scholar] [CrossRef]
- McGovern, L.; Futscher, M.H.; Muscarella, L.A.; Ehrler, B. Understanding the Stability of MAPbBr3 versus MAPbI3: Suppression of Methylammonium Migration and Reduction of Halide Migration. J. Phys. Chem. Lett. 2020, 11, 7127–7132. [Google Scholar] [CrossRef] [PubMed]
- Pistor, P.; Burwig, T.; Brzuska, C.; Weber, B.; Fränzel, W. Thermal stability and miscibility of co-evaporated methyl ammonium lead halide (MAPbX3, X = I, Br, Cl) thin films analysed by in situ X-ray diffraction. J. Mater. Chem. A 2018, 6, 11496–11506. [Google Scholar] [CrossRef]
- Knight, A.J.; Borchert, J.; Oliver, R.D.J.; Patel, J.B.; Radaelli, P.G.; Snaith, H.J.; Johnston, M.B.; Herz, L.M. Halide Segregation in Mixed-Halide Perovskites: Influence of A-Site Cations. ACS Energy Lett. 2021, 6, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Soler, J.M.; Artacho, E.; Gale, J.D.; Garcia, A.; Junquera, J.; Ordejon, P.; Sanchez-Portal, D. The SIESTA method for ab initio order- N materials simulation. J. Phys. Condens. Matter 2002, 14, 2745. [Google Scholar] [CrossRef] [Green Version]
- Ceperley, D.M.; Alder, B.J. Ground State of the Electron Gas by a Stochastic Method. Phys. Rev. Lett. 1980, 45, 566–569. [Google Scholar] [CrossRef] [Green Version]
- Latini, A.; Gigli, G.; Ciccioli, A. A study on the nature of the thermal decomposition of methylammonium lead iodide perovskite, CH3NH3PbI3: An attempt to rationalise contradictory experimental results. Sustain. Energy Fuels 2017, 1, 1351–1357. [Google Scholar] [CrossRef]
- Scikit-Learn. Available online: https://scikit-learn.org (accessed on 15 July 2021).
- TensorFlow. Available online: https://www.tensorflow.org/ (accessed on 15 July 2021).
- Keras. Available online: https://keras.io/ (accessed on 15 July 2021).
- Brunetti, B.; Cavallo, C.; Ciccioli, A.; Gigli, G.; Latini, A. On the Thermal and Thermodynamic (In)Stability of Methylammonium Lead Halide Perovskites. Sci. Rep. 2016, 6, 31896. [Google Scholar] [CrossRef] [PubMed]
- Valenzano, V.; Cesari, A.; Balzano, F.; Milella, A.; Fracassi, F.; Listorti, A.; Gigli, G.; Rizzo, A.; Uccello-Barretta, G.; Colella, S. Methylammonium-formamidinium reactivity in aged organometal halide perovskite inks. Cell Rep. Phys. Sci. 2021, 2, 100432. [Google Scholar] [CrossRef]
- Nazarenko, O.; Kotyrba, M.R.; Yakunin, S.; Aebli, M.; Rainò, G.; Benin, B.M.; Wörle, M.; Kovalenko, M.V. Guanidinium-Formamidinium Lead Iodide: A Layered Perovskite-Related Compound with Red Luminescence at Room Temperature. J. Am. Chem. Soc. 2018, 140, 3850–3853. [Google Scholar] [CrossRef] [PubMed]
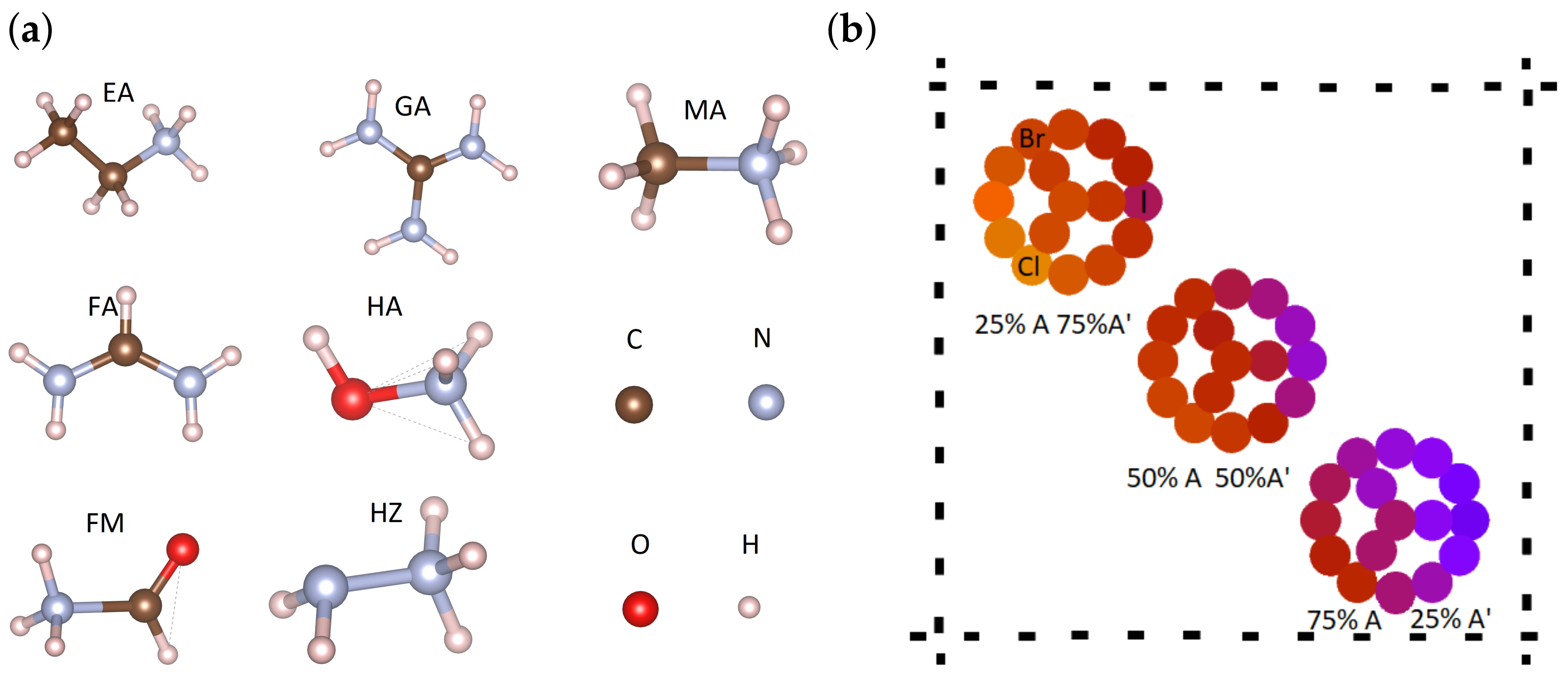
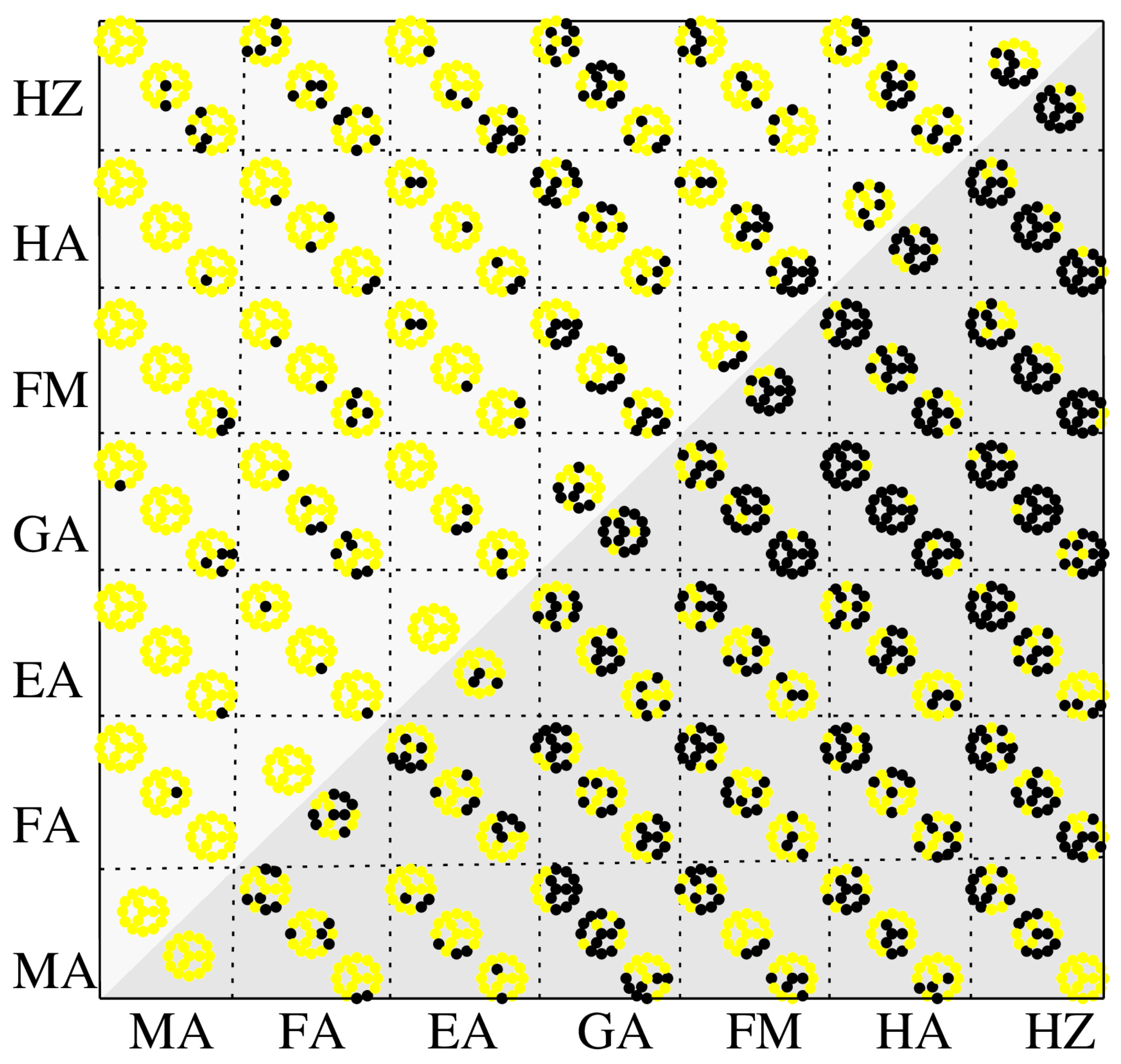

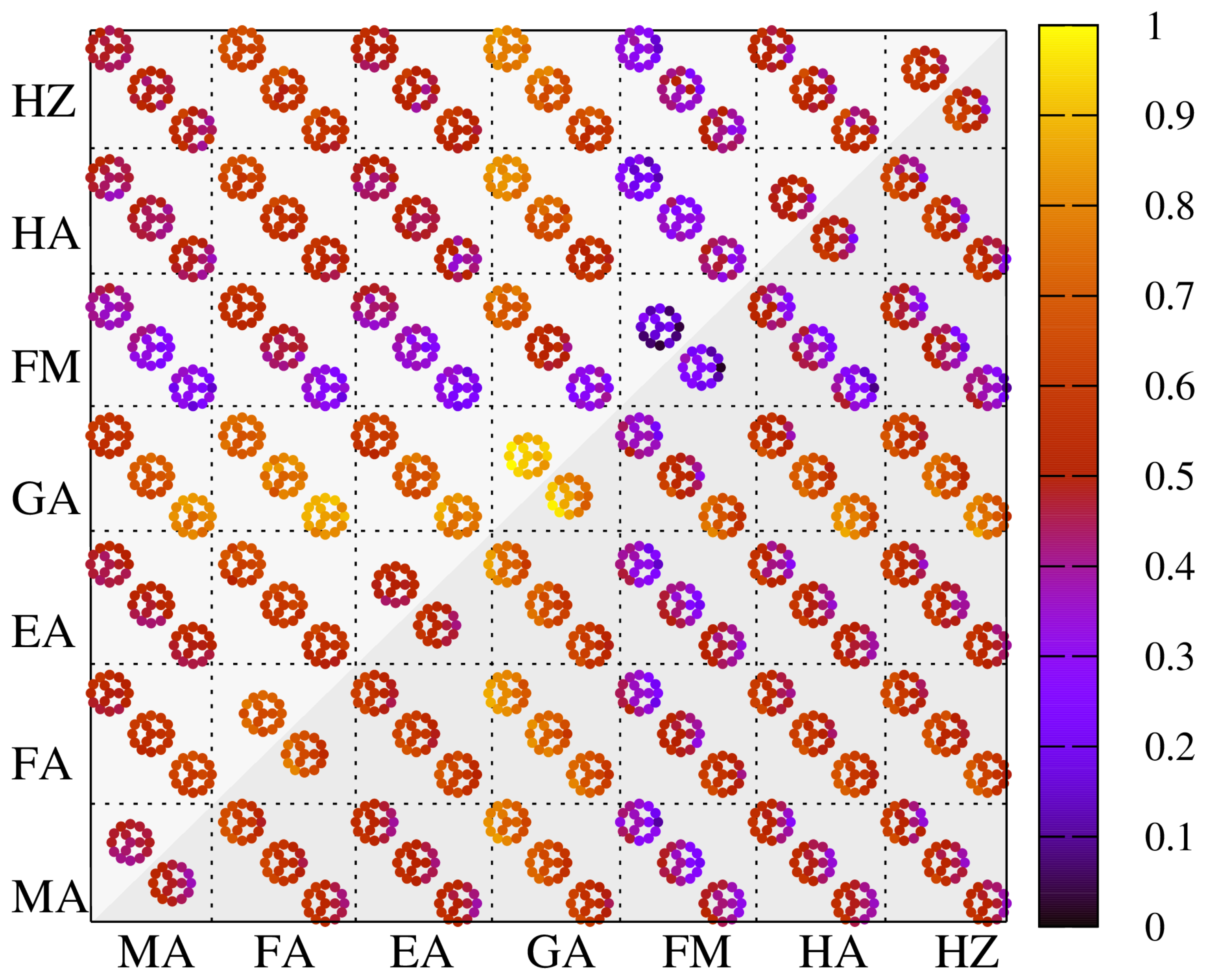
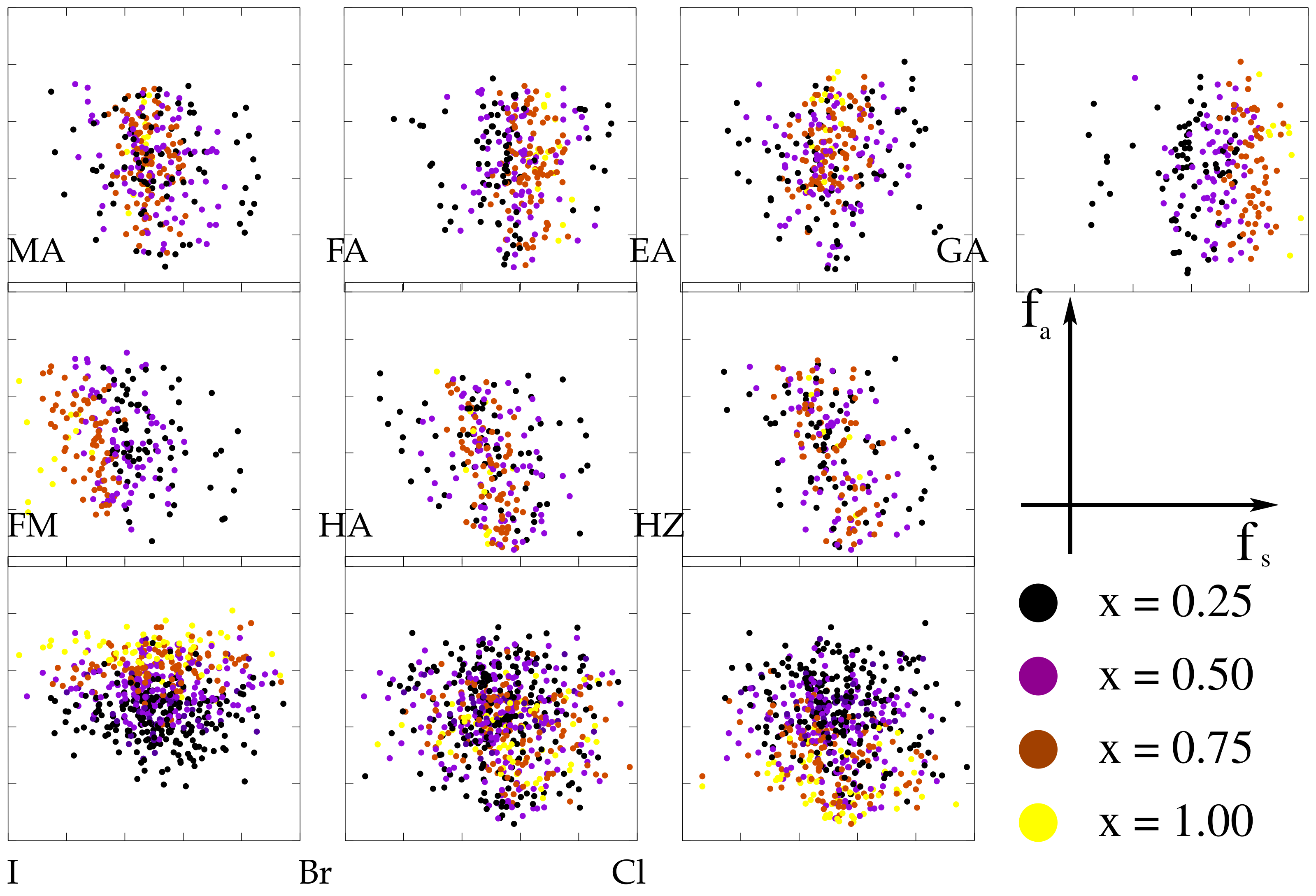
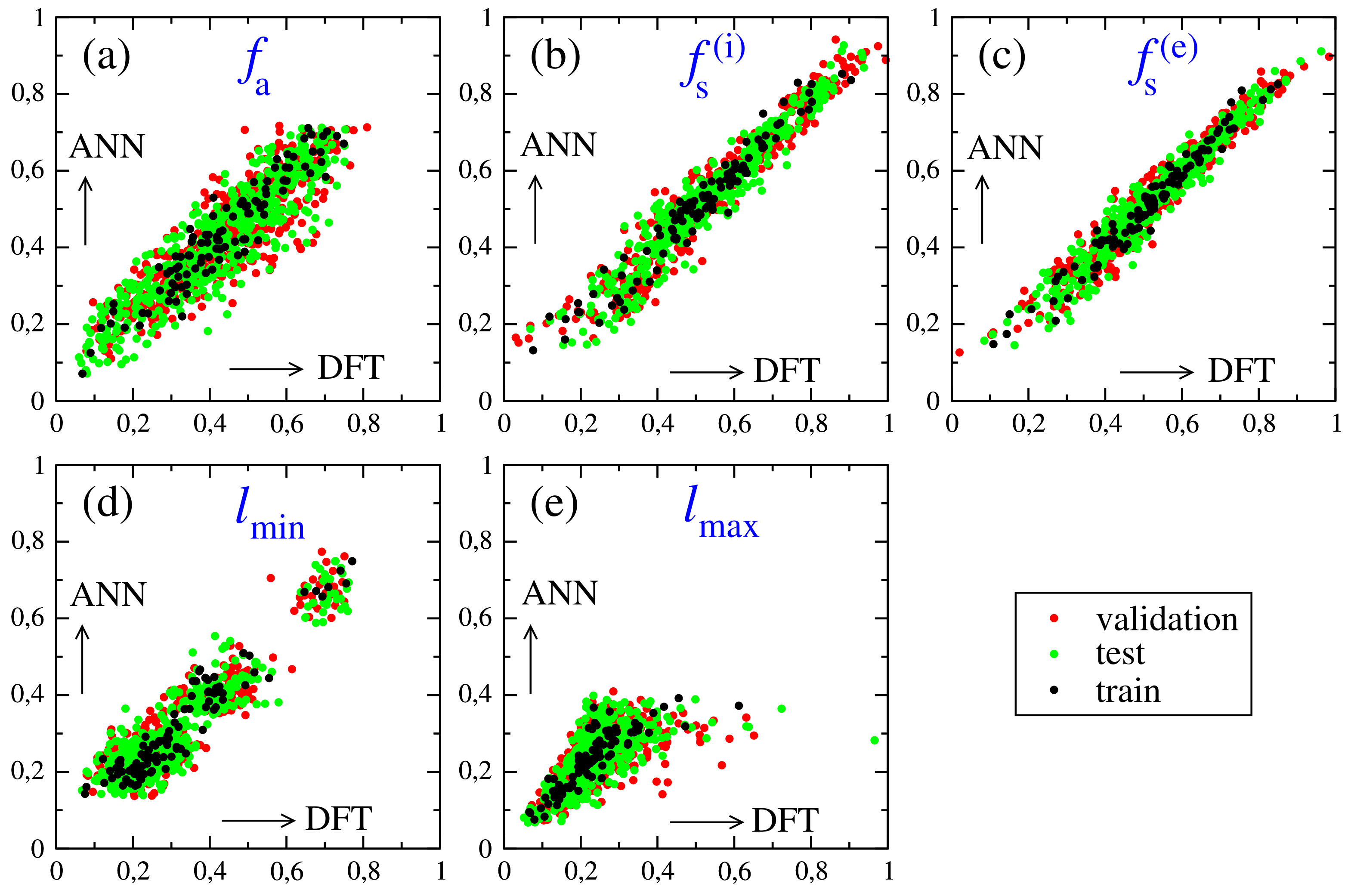
| A-Cations | MA | FA | EA | GA | FM | HA | HZ |
|---|---|---|---|---|---|---|---|
| Formula | CHNH | CH(NH) | (CHCH)NH | (NH)C | NHCOH | NHOH | NHNH |
| 3D? | yes | yes | yes | yes | no | no | no |
| 217 | 253 | 274 | 278 | 254 | 216 | 217 | |
| (I) | 0.91 | 0.99 | 1.03 | 1.04 | 0.99 | 0.91 | 0.91 |
| (Br) | 0.93 | 1.01 | 1.06 | 1.06 | 1.01 | 0.92 | 0.93 |
| (Cl) | 0.94 | 1.02 | 1.07 | 1.08 | 1.03 | 0.94 | 0.94 |
| Absorption Index () | Stability Index () | Both and |
|---|---|---|
| EAGAPbI | GAPbBrCl | EAGAPbI |
| and | and | and |
| GAPbICl | GAPbIBrCl | GAPbICl |
| and | and | and |
| EAPbI | GAPbCl | EAGAPbIBr |
| and | and | and |
| EAGAPbI | GAPbI | GAHZPbI |
| and | and | and |
| FAEAPbICl | GAPbIBr | EAGAPbIBr |
| and | and | and |
| EAGAPbIBr | FAGAPbIBr | FAGAPbI |
| and | and | and |
| EAPbIBr | FAGAPbIBr | EAGAPbIBr |
| and | and | and |
| EAGAPbI | FAGAPbI | EAGAPbI |
| and | and | and |
| GAHZPbI | FAGAPbBrCl | FAPbI |
| and | and | and |
| GAFMPbI | EAGAPbBrCl | EAGAPbIBr |
| and | and | and |
| Training: | 50 Samples | 100 Samples | 200 Samples |
|---|---|---|---|
| Data: | Method: min. max. av. | Method: min. max. av. | Method: min. max. av. |
| MLS: 0.69 0.77 0.74 | MLS: 0.74 0.79 0.77 | MLS: 0.77 0.79 0.78 | |
| RF : 0.61 0.75 0.69 | RF : 0.70 0.76 0.73 | RF : 0.73 0.77 0.75 | |
| ANN: 0.82 | |||
| MLS: 0.90 0.91 0.91 | MLS: 0.92 0.93 0.92 | MLS: 0.92 0.93 0.92 | |
| RF : 0.78 0.85 0.82 | RF : 0.85 0.90 0.88 | RF : 0.89 0.92 0.90 | |
| ANN: | ANN: 0.92 | ||
| MLS: 0.92 0.93 0.93 | MLS: 0.93 0.94 0.94 | MLS: 0.94 0.94 0.94 | |
| RF : 0.70 0.81 0.77 | RF : 0.81 0.87 0.85 | RF : 0.87 0.91 0.89 | |
| ANN: | ANN: 0.95 | ||
| MLS: 0.43 0.56 0.48 | MLS: 0.51 0.60 0.56 | MLS: 0.55 0.60 0.58 | |
| RF : 0.81 0.87 0.84 | RF : 0.83 0.86 0.85 | RF : 0.84 0.86 0.85 | |
| ANN: 0.84 | |||
| MLS: 0.12 0.31 0.23 | MLS: 0.11 0.33 0.28 | MLS: 0.27 0.38 0.32 | |
| RF : 0.16 0.37 0.31 | RF : 0.17 0.42 0.33 | RF : 0.20 0.42 0.35 | |
| ANN: 0.43 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filipoiu, N.; Mitran, T.L.; Anghel, D.V.; Florea, M.; Pintilie, I.; Manolescu, A.; Nemnes, G.A. Investigation of Opto-Electronic Properties and Stability of Mixed-Cation Mixed-Halide Perovskite Materials with Machine-Learning Implementation. Energies 2021, 14, 5431. https://doi.org/10.3390/en14175431
Filipoiu N, Mitran TL, Anghel DV, Florea M, Pintilie I, Manolescu A, Nemnes GA. Investigation of Opto-Electronic Properties and Stability of Mixed-Cation Mixed-Halide Perovskite Materials with Machine-Learning Implementation. Energies. 2021; 14(17):5431. https://doi.org/10.3390/en14175431
Chicago/Turabian StyleFilipoiu, Nicolae, Tudor Luca Mitran, Dragos Victor Anghel, Mihaela Florea, Ioana Pintilie, Andrei Manolescu, and George Alexandru Nemnes. 2021. "Investigation of Opto-Electronic Properties and Stability of Mixed-Cation Mixed-Halide Perovskite Materials with Machine-Learning Implementation" Energies 14, no. 17: 5431. https://doi.org/10.3390/en14175431
APA StyleFilipoiu, N., Mitran, T. L., Anghel, D. V., Florea, M., Pintilie, I., Manolescu, A., & Nemnes, G. A. (2021). Investigation of Opto-Electronic Properties and Stability of Mixed-Cation Mixed-Halide Perovskite Materials with Machine-Learning Implementation. Energies, 14(17), 5431. https://doi.org/10.3390/en14175431








