Combustion Characteristics of 0.5 MW Class Oxy-Fuel FGR (Flue Gas Recirculation) Boiler for CO2 Capture
Abstract
:1. Introduction
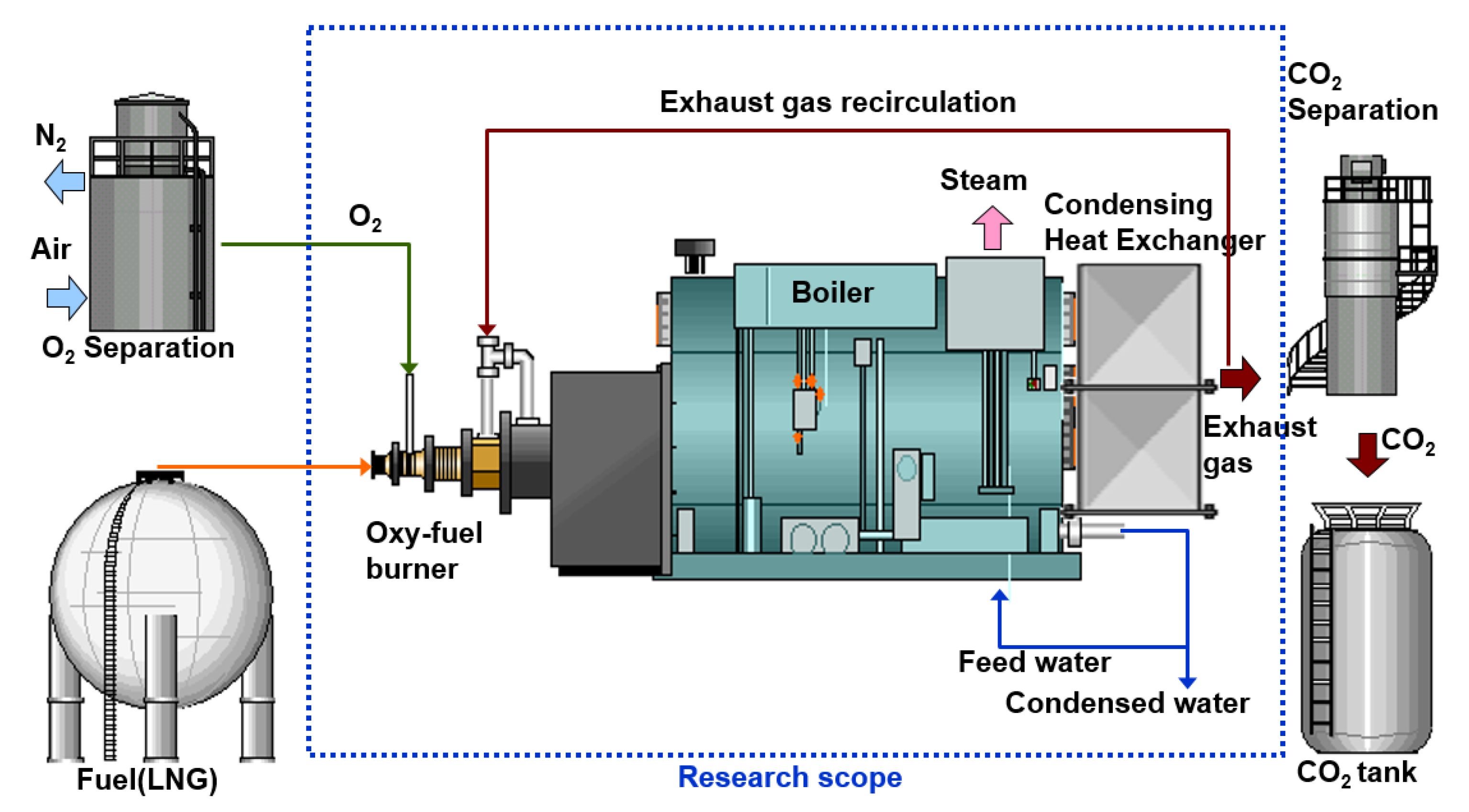
2. Experimental Setup and Methods
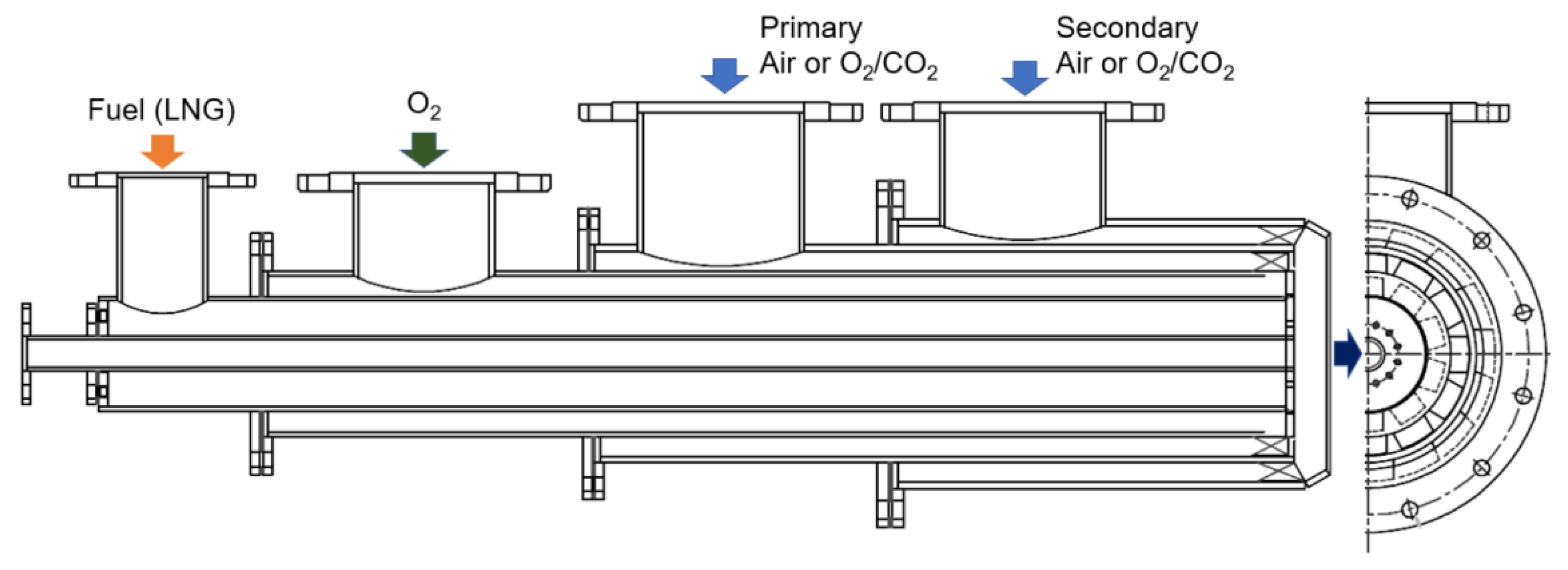
2.1. Oxy-Fuel, Air, and FGR Convertible Burner
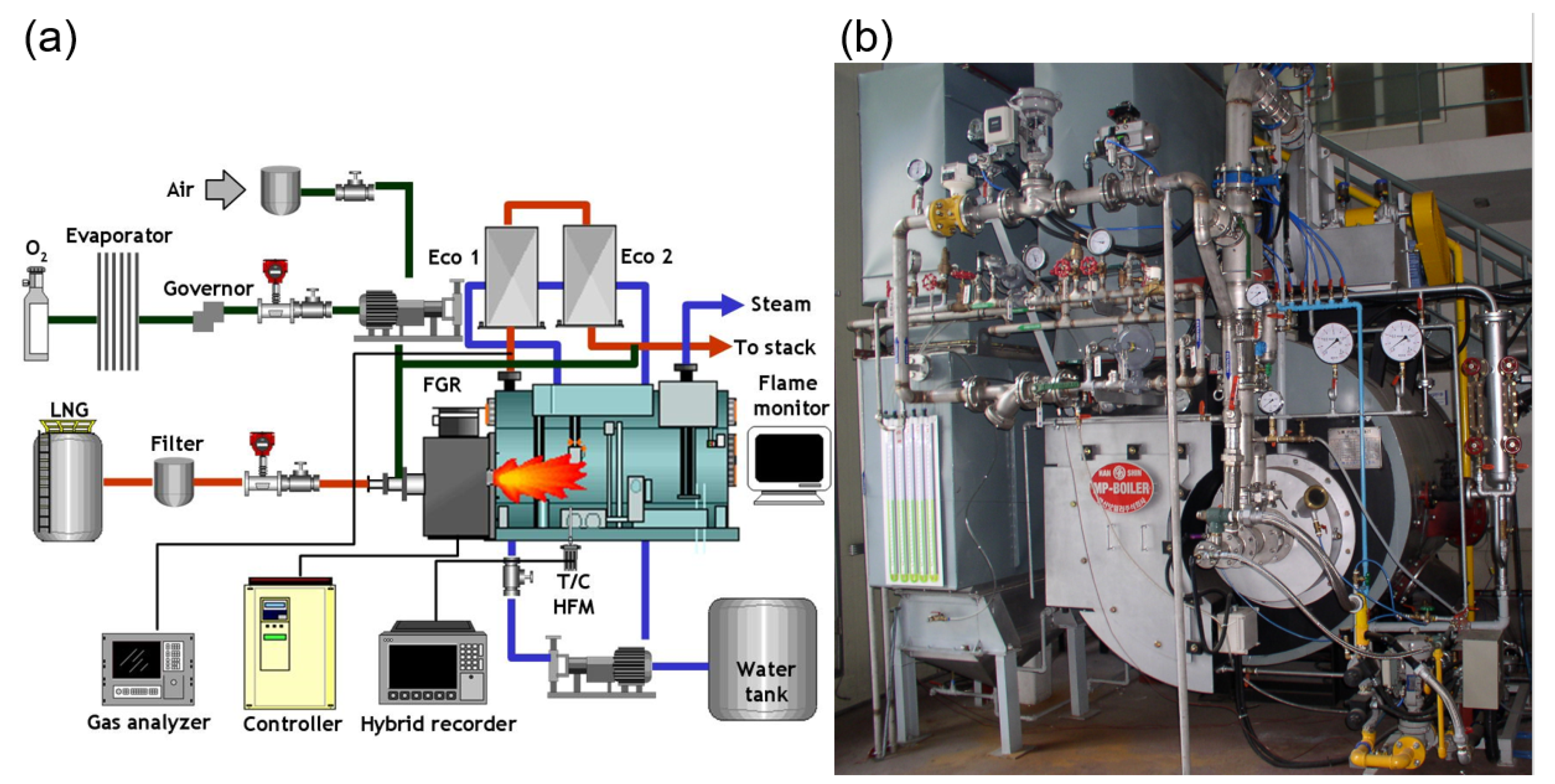
2.2. Oxy-Fuel Boiler and Instrumentation
3. Results and Discussion
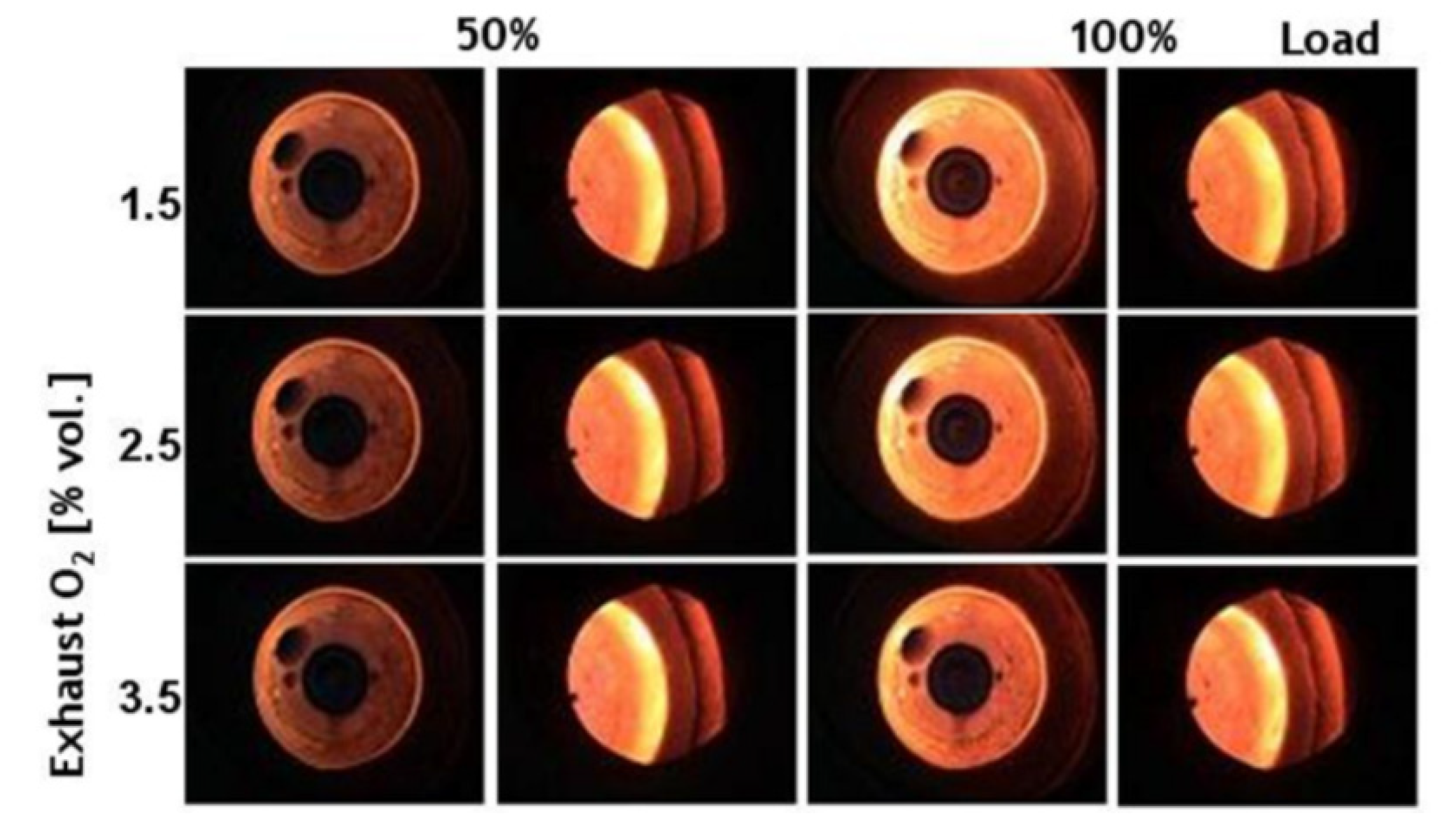
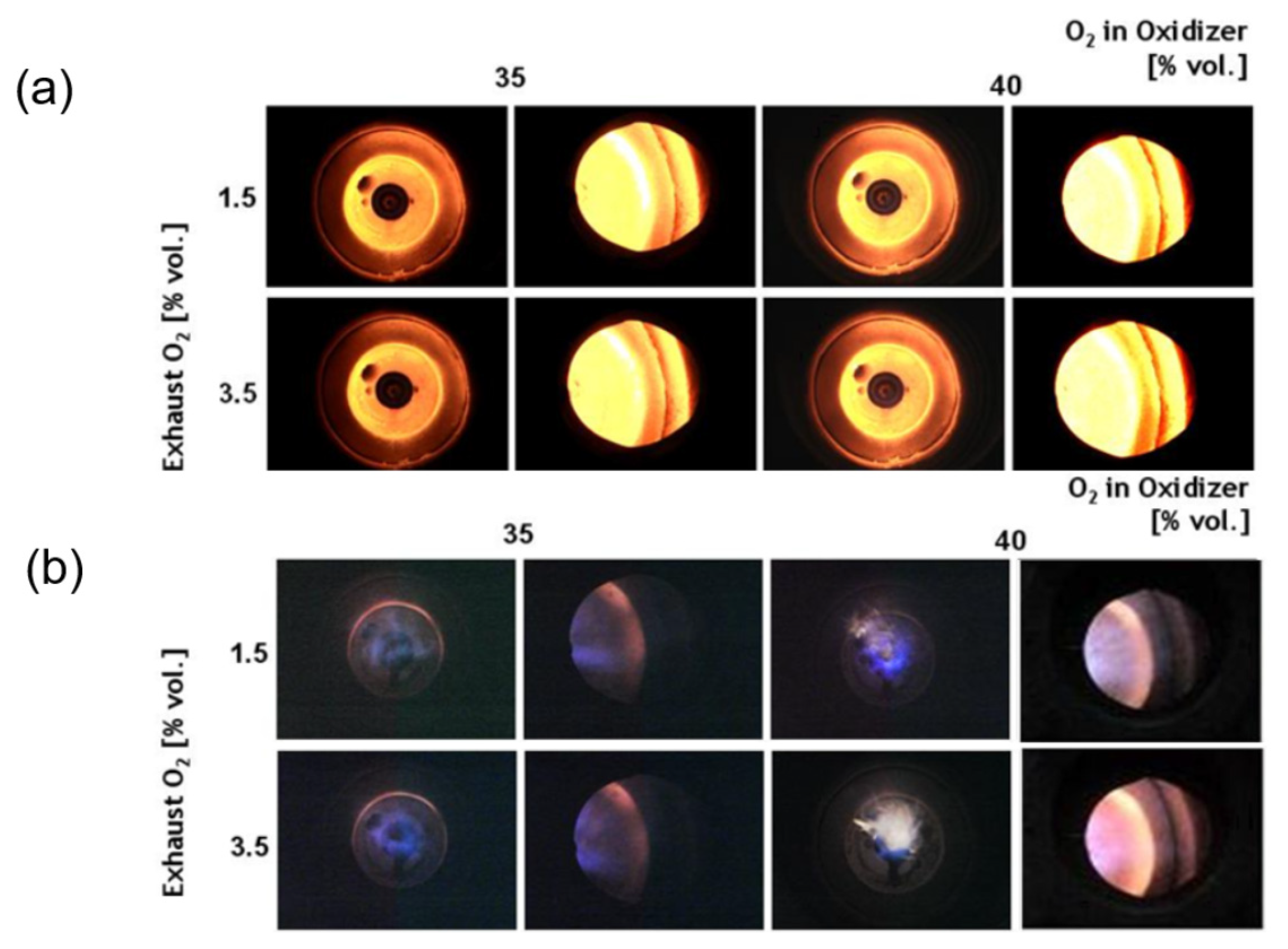
3.1. Flame
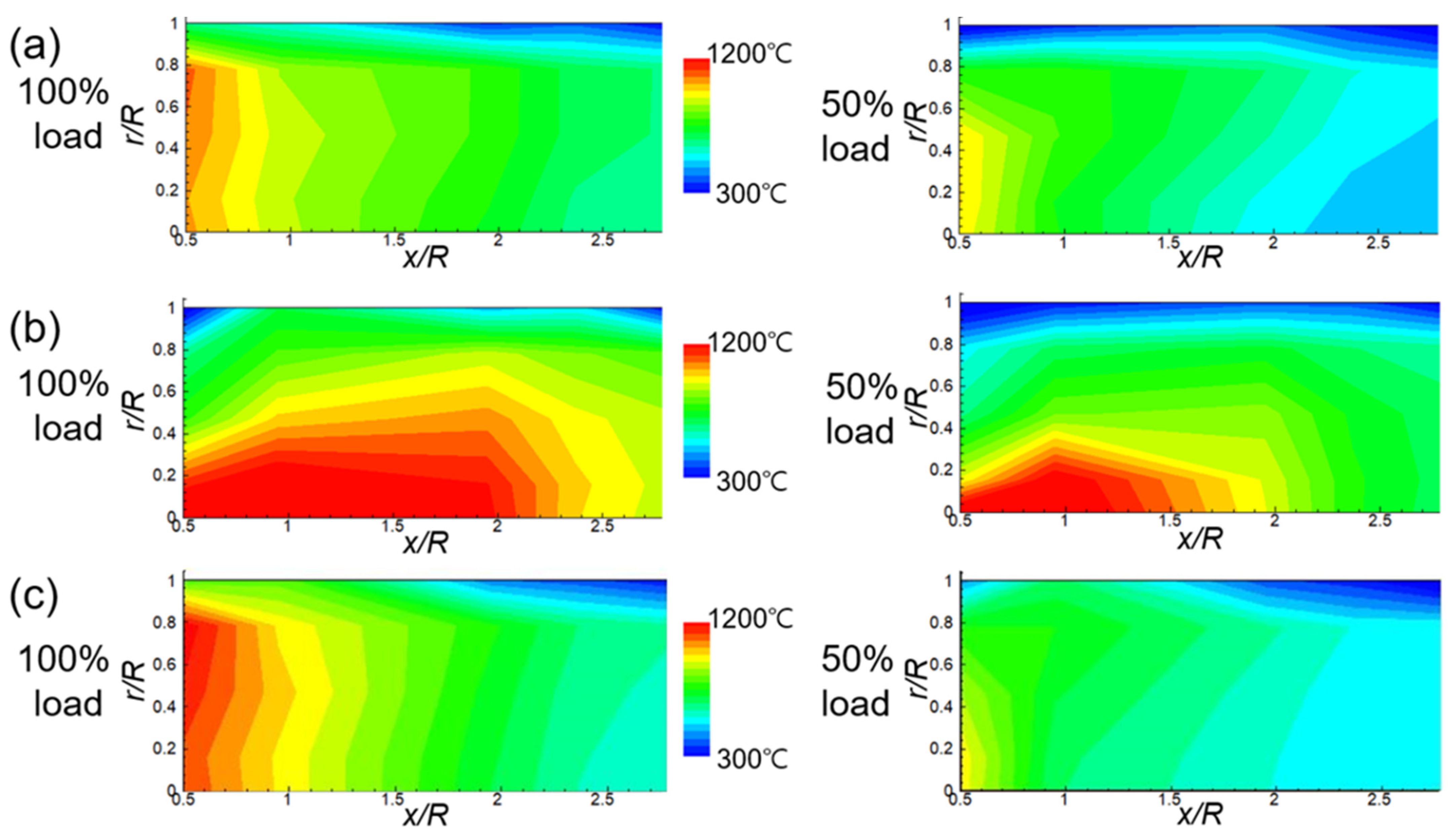
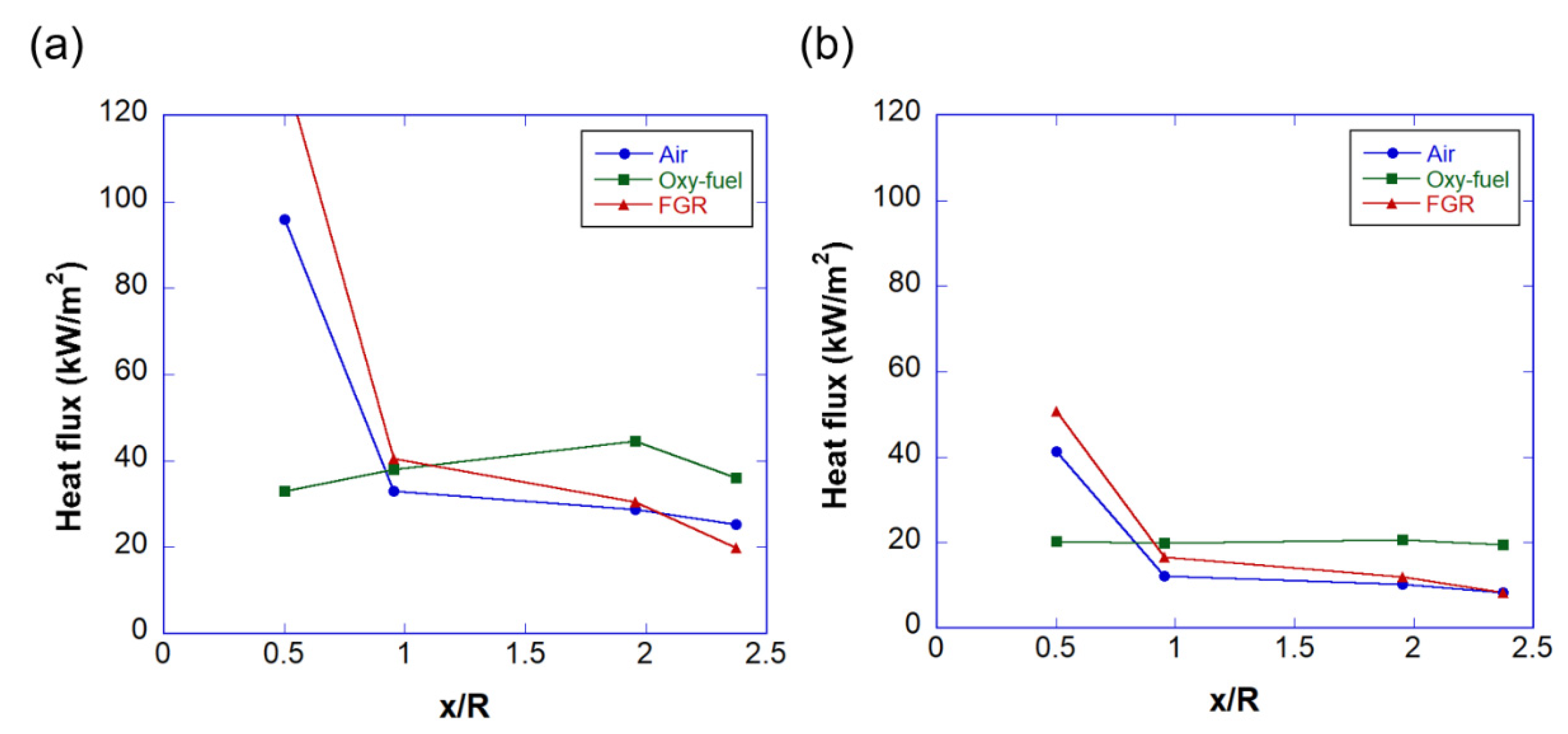
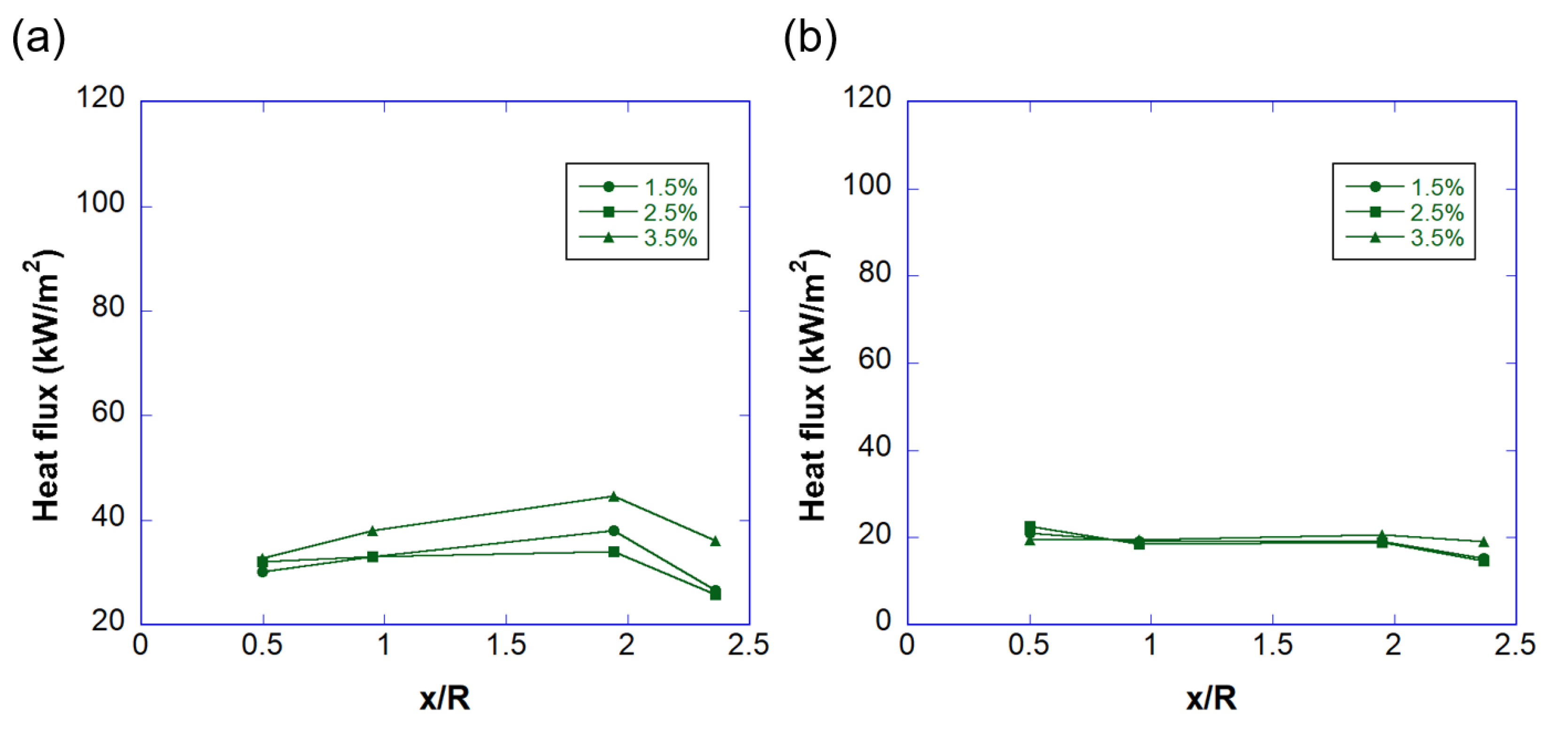
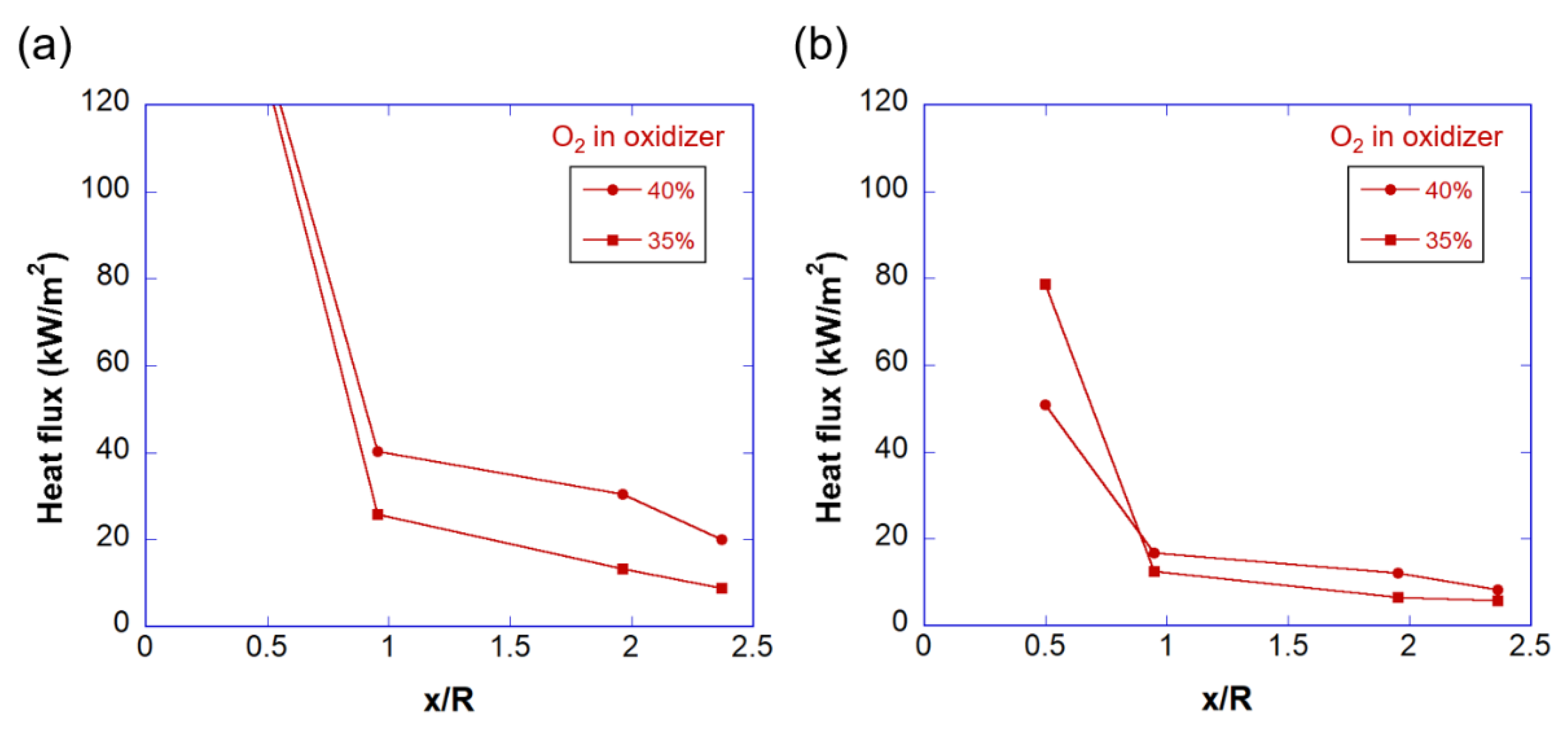
3.2. Temperature and Heat Flux
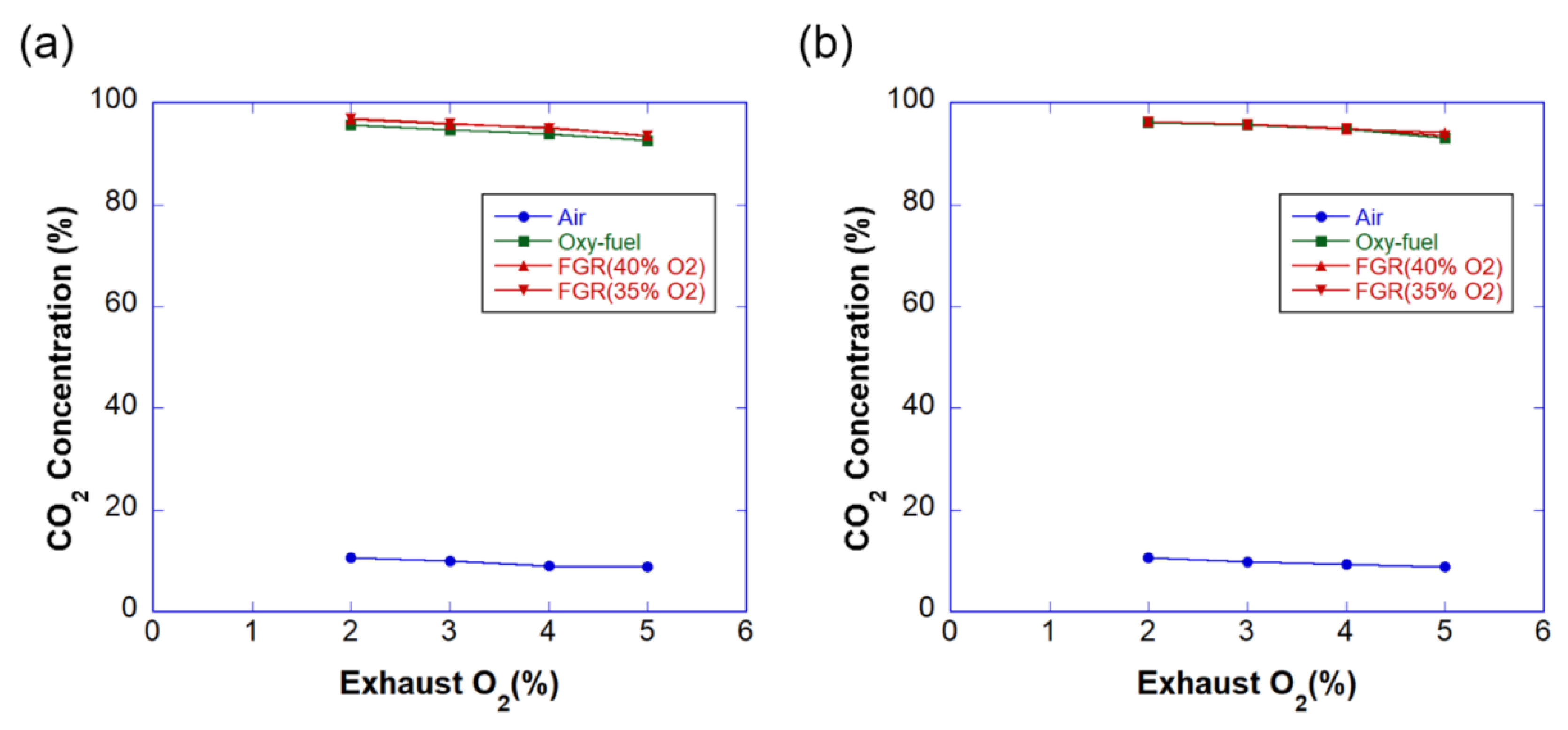
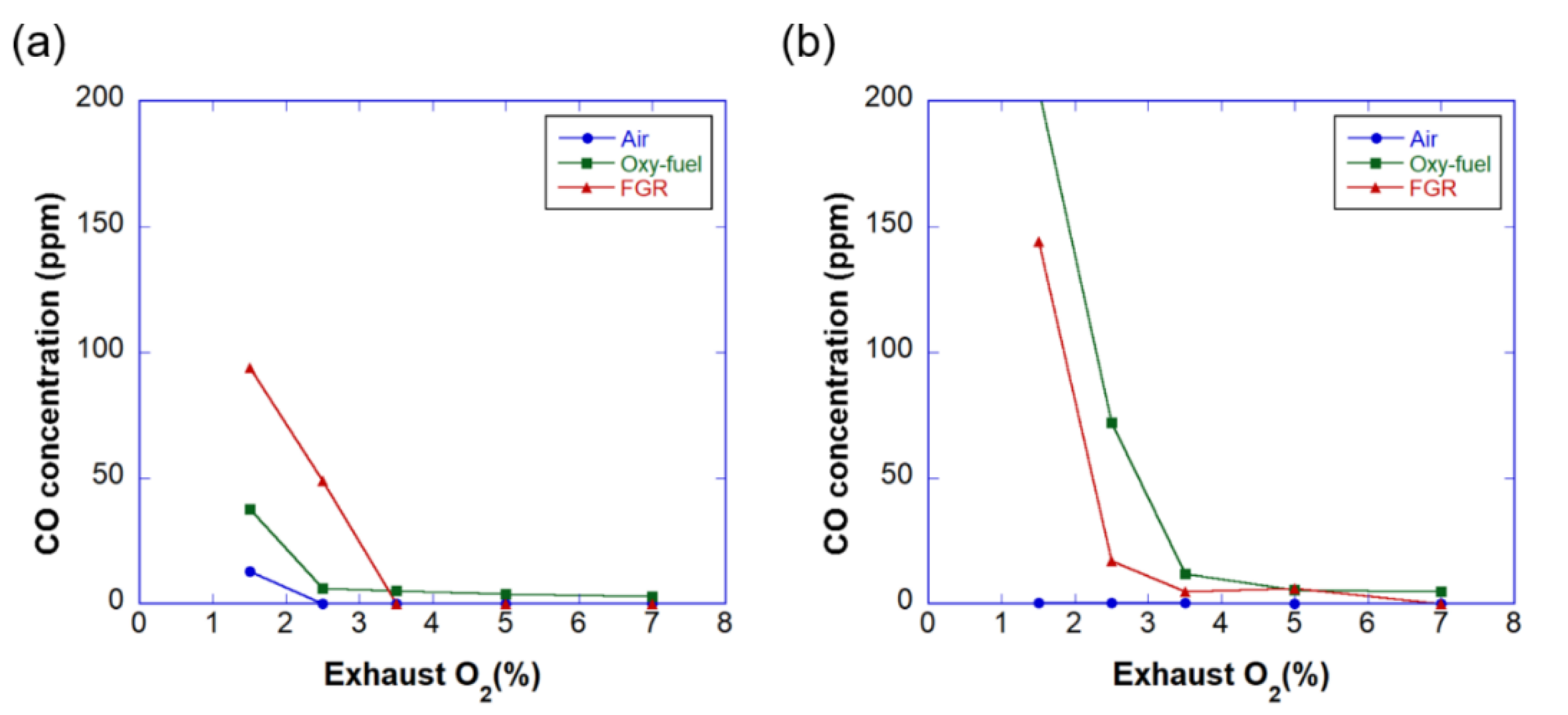
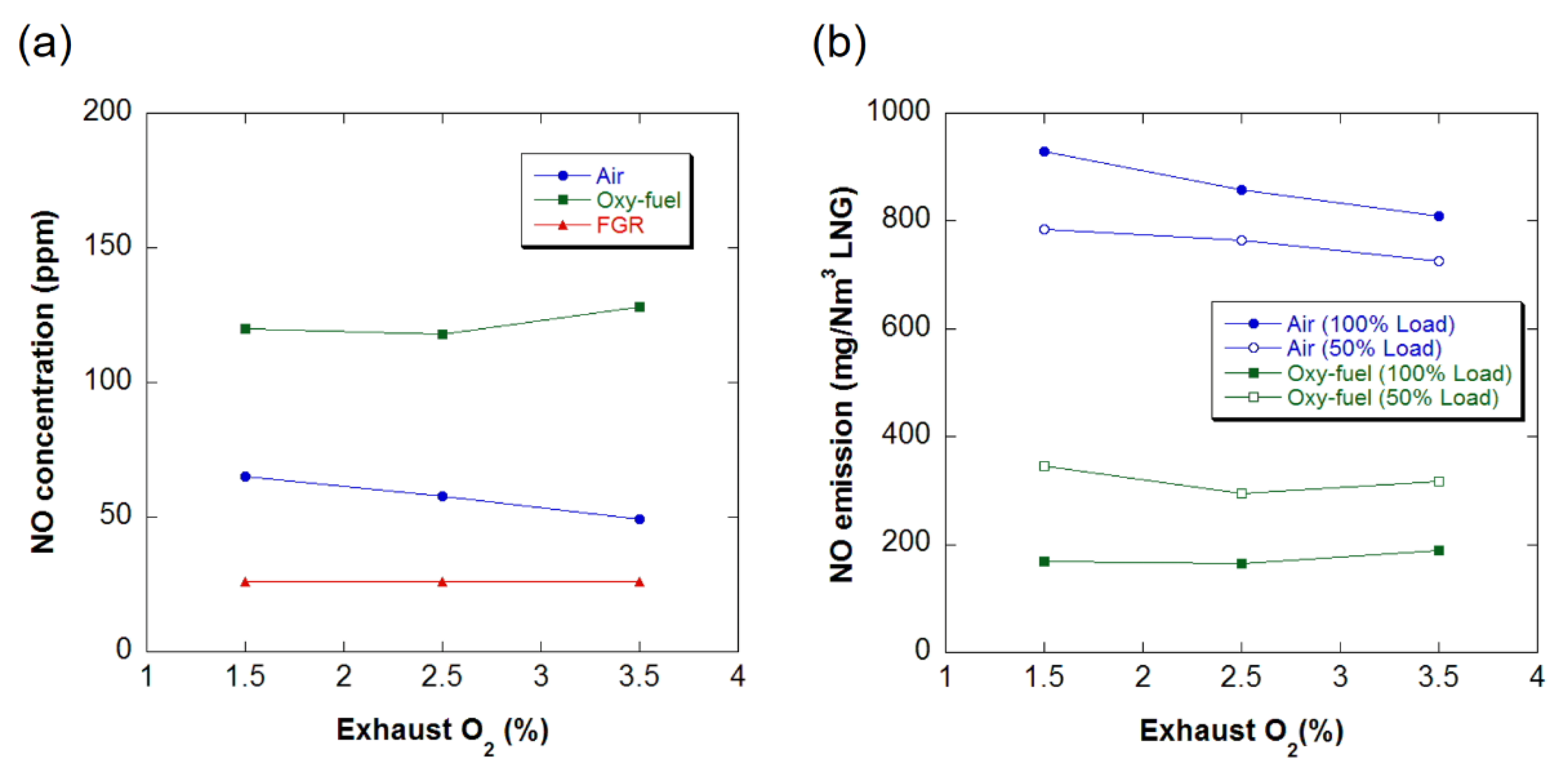
3.3. Emissions
4. Conclusions
- In oxy-natural-gas combustion without FGR, the flame temperature increases, and the gas radiation becomes stronger. Consequently, the extended flame of the coaxial combustor can heat the boiler combustion chamber designed for the air combustion flow rate.
- In oxy-natural-gas FGR combustion, an O2 concentration exceeding the oxy-coal concentration was required for flame stabilization owing to the effect of moisture. In the boiler used in this study, a stable flame was obtained above 32% O2.
- While flame delay was observed at a partial load in FGR combustion, it was not observed in the other combustion modes.
- If the airtightness of the system is maintained, the concentration of CO2 in the exhaust gas of industrial boilers can be increased by more than 90% through oxy-fuel combustion. The emission of NOx can be considerably reduced by implementing the coaxial burner.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| FGR | flue gas recirculation |
| PC | pulverized coal |
| r | radial coordinate at the combustion chamber [m] |
| R | radius of the combustion chamber [m] |
| x | streamwise coordinate from the burner [m] |
References
- Nemitallah, M.A.; Habib, M.A.; Badr, H.M.; Said, S.A.; Jamal, A.; Ben-Mansour, R.; Mokheimer, E.M.A.; Mezghani, K. Oxy-fuel combustion technology: Current status, applications, and trends. Int. J. Energy Res. 2017, 41, 1670–1708. [Google Scholar] [CrossRef]
- Shirmohammadi, R.; Aslani, A.; Ghasempour, R. Challenges of carbon capture technologies deployment in developing countries. Sustain. Energy Technol. Assessm. 2020, 42, 100837. [Google Scholar] [CrossRef]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; Hallett, J.P.; et al. Carbon capture and storage (CCS): The way forward. Energy Environ. Sci. 2018, 11, 1062–1176. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Xie, J.; Cai, W.; Wang, R. Thermodynamic effects of cycling carbon injectivity in shale reservoirs. J. Petrol. Sci. Eng. 2020, 195, 107717. [Google Scholar] [CrossRef]
- Davarpanah, A.; Mirshekari, B. Experimental investigation and mathematical modeling of gas diffusivity by carbon dioxide and methane kinetic adsorption. Ind. Eng. Chem. Res. 2019, 58, 12392–12400. [Google Scholar] [CrossRef]
- Markewitz, P.; Leitner, W.; Linssen, J.; Zapp, P.; Müller, T.; Schreiber, A. Worldwide innovations in the development of carbon capture technologies and the utilization of CO2. Energy Environ. Sci. 2013, 5, 7281–7385. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.B.; Xu, M.X.; Li, Y.B.; Wu, J.H.; Shen, J.C.; Liao, H. Experimental research on the process of compression and purification of CO2 in oxy-fuel combustion. Appl. Energy 2020, 259, 114123. [Google Scholar] [CrossRef]
- Shirmohammadi, R.; Aslani, A.; Ghasempour, R.; Romeo, L.M.; Petrakopoulou, F. Process design and thermoeconomic evaluation of a CO2 liquefaction process driven by waste exhaust heat recovery for an industrial CO2 capture and utilization plant. J. Therm. Anal. Calorim. 2021. [Google Scholar] [CrossRef]
- Stanger, R.; Wall, T.; Spörl, R.; Paneru, M.; Grathwohl, S.; Weidmann, M.; Scheffknecht, G.; McDonald, D.; Myöhänen, K.; Ritvanen, J.; et al. Oxy-fuel combustion for CO2 capture in power plants. Int. J. Greenhouse Gas Control 2015, 40, 55–125. [Google Scholar] [CrossRef]
- Geem, Z.W.; Kim, J.H. Optimal energy mix with renewable portfolio standards in Korea. Sustainability 2016, 8, 423. [Google Scholar] [CrossRef] [Green Version]
- Park, N.-B.; Park, S.Y.; Kim, J.J.; Choi, D.G.; Yun, B.Y.; Hong, J.C. Technical and economic potential of highly efficient boiler technologies in the Korean industrial sector. Energy 2017, 121, 884–891. [Google Scholar] [CrossRef]
- Ahn, J.; Kim, H.J.; Choi, K.S. Combustion characteristics of oxy-fuel burners for CO2 capturing boilers. J. Therm. Sci. Tech. 2009, 4, 408–413. [Google Scholar] [CrossRef] [Green Version]
- Ahn, J.; Kim, H.J.; Choi, K.S. Oxy-fuel combustion boiler for CO2 capturing: 50kW-class model test. J. Mech. Sci. Tech. 2010, 24, 2135–2141. [Google Scholar] [CrossRef]
- Ehyaei, M.A.; Ahmadi, A.; Rosen, M.A.; Davarpanah, A. Thermodynamic optimization of a geothermal power plant with a genetic algorithm in two stages. Processes 2020, 8, 1277. [Google Scholar] [CrossRef]
- Ahn, J.; Kim, H.J. Combustion and heat transfer characteristics at the combustion chamber of an oxy-fuel boiler for CO2 capturing: A numerical simulation. Progress CFD 2011, 11, 210–215. [Google Scholar]
- Marcano, N.; Recourt, P.; Tsiava, R.; Laurent, J.; Lethier, S.; Deveaux, M.; Bouvarel, A.; Quet, J.P. Oxy-combustion at Lacq CCS pilot plant: Preliminary analysis of burner-boiler performance. In Proceedings of the 2nd Oxyfuel Combustion Conference (OCC2), Yeppoon, Australia, 11–16 September 2011. [Google Scholar]
- Burchhardt, U. Übersicht zu Tests und Ergebnissen der Oxyfuel-Forschungsanlage Schwarze Pumpe. In Proceedings of the Public Final Project Meeting of the German Research Project “ADECOS-Komponenten”, Cottbus, Germany, 7–8 May 2014. [Google Scholar]
- Rehfeldt, S.; Schiffer, F.P.; Weckes, P.; Bergins, C.; Tigges, K.D. Oxy-fuel combustion with Hitachi’s DST—Burner at Vattenfall’s 30MWth pilot plant at Schwarze Pumpe. In Proceedings of the 2nd Oxyfuel Combustion Conference (OCC2), Yeppoon, Australia, 11–16 September 2011. [Google Scholar]
- Sturgeon, D. Oxy-coal burner technology development. In Proceedings of the 2nd Oxyfuel Combustion Conference (OCC2), Yeppoon, Australia, 11–16 September 2011. [Google Scholar]
- Choi, S.; Kim, T.Y.; Kim, H.K.; Koo, J.; Kim, J.S.; Kwon, O.C. Properties of inverse non-premixed pure O2/CH4 coflow flames in a model combustor. Energy 2015, 93, 1105–1115. [Google Scholar] [CrossRef]
- Kim, H.K.; Kim, Y.; Lee, S.M.; Ahn, K.Y. Studies on combustion characteristics and flame length of turbulent oxy-fuel flames. Energy Fuel 2007, 21, 1459–1467. [Google Scholar] [CrossRef]
- Oh, J.; Noh, D. Flame characteristics of a non-premixed oxy-fuel jet in a lab-scale furnace. Energy 2015, 81, 328–345. [Google Scholar] [CrossRef]
- Becker, L.G.; Kosaka, H.; Böhm, B.; Knappstein, R.; Habermehl, M.; Kneer, R.; Janicka, J.; Dreizler, A. Experimental investigation of flame stabilization inside the quarl of an oxyfuel swirl burner. Fuel 2017, 201, 124–135. [Google Scholar] [CrossRef]
- Hees, J.; Zabrodiec, D.; Massmeyer, A.; Hatzfeld, O.; Kneer, R. Experimental investigation into the influence of the oxygen concentration on a pulverized coal swirl flame in oxy-fuel atmosphere. Fuel 2019, 240, 64–74. [Google Scholar] [CrossRef]
- Massmeyer, A.; Zabrodiec, D.; Hees, J.; Kreitzberg, T.; Hatzfeld, O.; Kneer, R. Flame pattern analysis for 60kWth flames under conventional air-fired and oxy-fuel conditions for two different types of coal. Fuel 2020, 271, 117457. [Google Scholar] [CrossRef]
- Silva, R.C.; Krautz, H.J. Combustion measurements of type-1 pulverized coal flames operating under oxy-fired conditions. Fuel Process Tech. 2018, 171, 232–247. [Google Scholar] [CrossRef]
- Toftegaard, M.B.; Brix, J.; Jensen, P.A.; Glarborg, P.; Jensen, A.D. Oxyfuel combustion of solid fuels. Prog. Energy Combust. Sci. 2010, 36, 581–625. [Google Scholar] [CrossRef]
- Toporov, D.; Bocian, P.; Heil, P.; Kellermann, A.; Stadler, H.; Tschunko, S.; Förster, M.; Kneer, R. Detailed investigation of a pulverized fuel swirl flame in CO2/O2 atmosphere. Combust. Flame 2008, 155, 605–618. [Google Scholar] [CrossRef]
- Andersson, K.; Normann, F.; Johnsson, F.; Leckner, B. NO emission during oxy-fuel combustion of lignite. Ind. Eng. Chem. Res. 2008, 47, 1835–1845. [Google Scholar] [CrossRef]
- Gaber, C.; Schluckner, C.; Wachter, P.; Demuth, M.; Hochenauer, C. Experimental study on the influence of the nitrogen concentration in the oxidizer on NOx and CO emissions during the oxy-fuel combustion of natural gas. Energy 2021, 214, 118905. [Google Scholar] [CrossRef]
- Kather, A.; Scheffknecht, G. Review: The oxy-coal process with cryogenic oxygen supply. Naturwissenschaften 2009, 96, 993–1010. [Google Scholar] [CrossRef] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, J.; Kim, H.-J. Combustion Characteristics of 0.5 MW Class Oxy-Fuel FGR (Flue Gas Recirculation) Boiler for CO2 Capture. Energies 2021, 14, 4333. https://doi.org/10.3390/en14144333
Ahn J, Kim H-J. Combustion Characteristics of 0.5 MW Class Oxy-Fuel FGR (Flue Gas Recirculation) Boiler for CO2 Capture. Energies. 2021; 14(14):4333. https://doi.org/10.3390/en14144333
Chicago/Turabian StyleAhn, Joon, and Hyouck-Ju Kim. 2021. "Combustion Characteristics of 0.5 MW Class Oxy-Fuel FGR (Flue Gas Recirculation) Boiler for CO2 Capture" Energies 14, no. 14: 4333. https://doi.org/10.3390/en14144333
APA StyleAhn, J., & Kim, H.-J. (2021). Combustion Characteristics of 0.5 MW Class Oxy-Fuel FGR (Flue Gas Recirculation) Boiler for CO2 Capture. Energies, 14(14), 4333. https://doi.org/10.3390/en14144333







