Influence of Sulfate Ions on the Combined Application of Modified Water and Polymer Flooding—Rheology and Oil Recovery
Abstract
1. Introduction
2. Overall Methodology and Approach
- Brine preparation and optimization: Two distinct groups of brines were prepared, including sulfate concentration changes, with one group keeping similar TDS and the second varying hardness.
- Polymer preparation and comprehensive rheological characterization: Polymer solutions were prepared, mainly with a viscosity ratio of 2 between oil and polymer. Subsequently, solutions were characterized in a detailed manner using steady-shear viscosity, elongational viscosity measurements, first normal stress differences (N1) and oscillatory (the latter two as attempts).
- Mechanical degradation of polymer solutions: This step allowed for understanding and defining any possible degradation prior to the core face and to draw better conclusions on polymer performance.
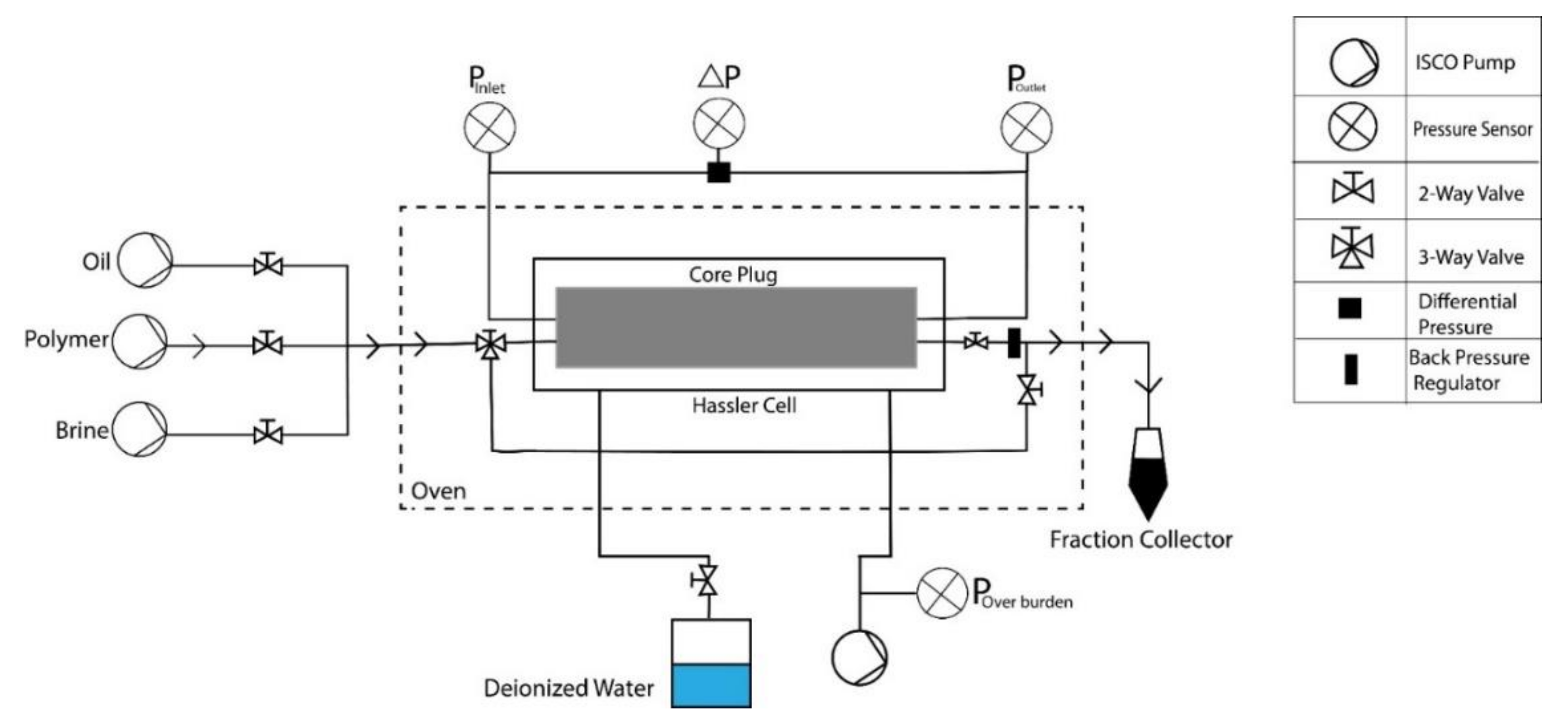
- Two-phase core flooding experiments: Core plugs were saturated to initial water saturation (Swi) using the porous plate method, before each experiment took place. Subsequently, different chemical slugs were injected to observe pressure response and define oil recovery.
3. Materials and Methods
3.1. Fluids and Chemicals
3.1.1. Brine Composition
3.1.2. Polymer Solutions
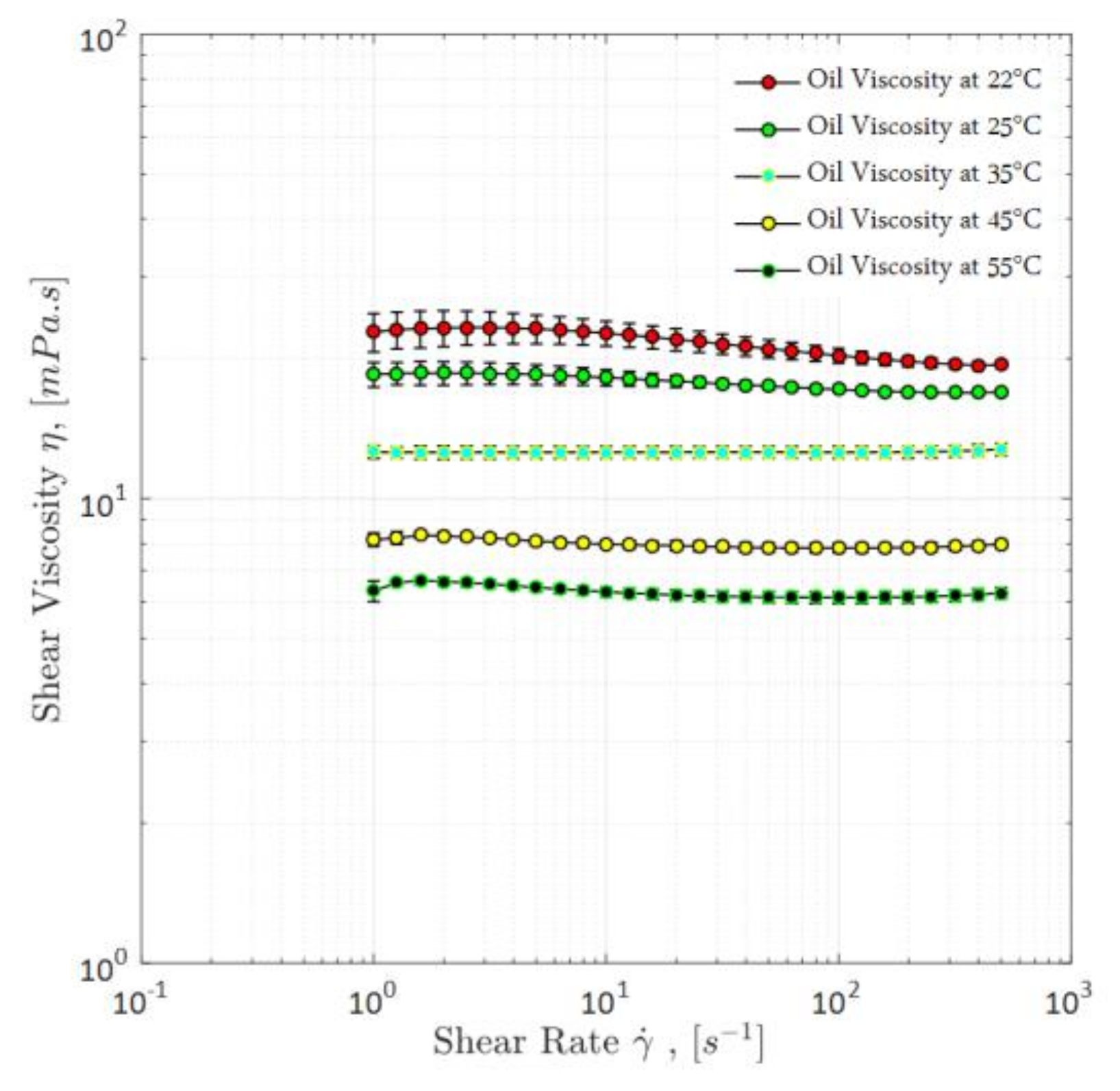
3.1.3. Oil
3.2. Porous Media
- Water saturation: Vacuum saturation was used for initial water saturation at 100% brine.
- Permeability to water (brine): SSW brine was injected at three injection rates (0.5, 1.0 and 0.5 mL/min).
- Oil saturation to initial water saturation (Swi): Oil saturation of core plugs was performed using a porous plate with a maximum injection pressure of 8.0 bar.
- Core ageing for wettability alteration: All cores were aged at 45 °C (core flooding experiments’ temperature) for three weeks prior to each core flood experiment, in order to establish attachment of polar components on the core. Skauge et al. [57] observed good wettability alteration within 3 weeks of ageing at 50 °C.
- Two-phase flooding experiments: The workflow/sequence adopted for the core flooding experiments is listed in Table 4. Bump-rate injection was performed to avoid capillary end effects before tertiary mode flooding (except core Bent 5.1).
Mechanical Degradation of Polymer—Before Entering the Plug Face
3.3. Fluids Investigation
3.3.1. e-VROC®—Extensional Viscometer-Rheometer on a Chip
3.3.2. Rheological Measurements
4. Results and Discussion
4.1. Comprehensive Rheological Evaluation
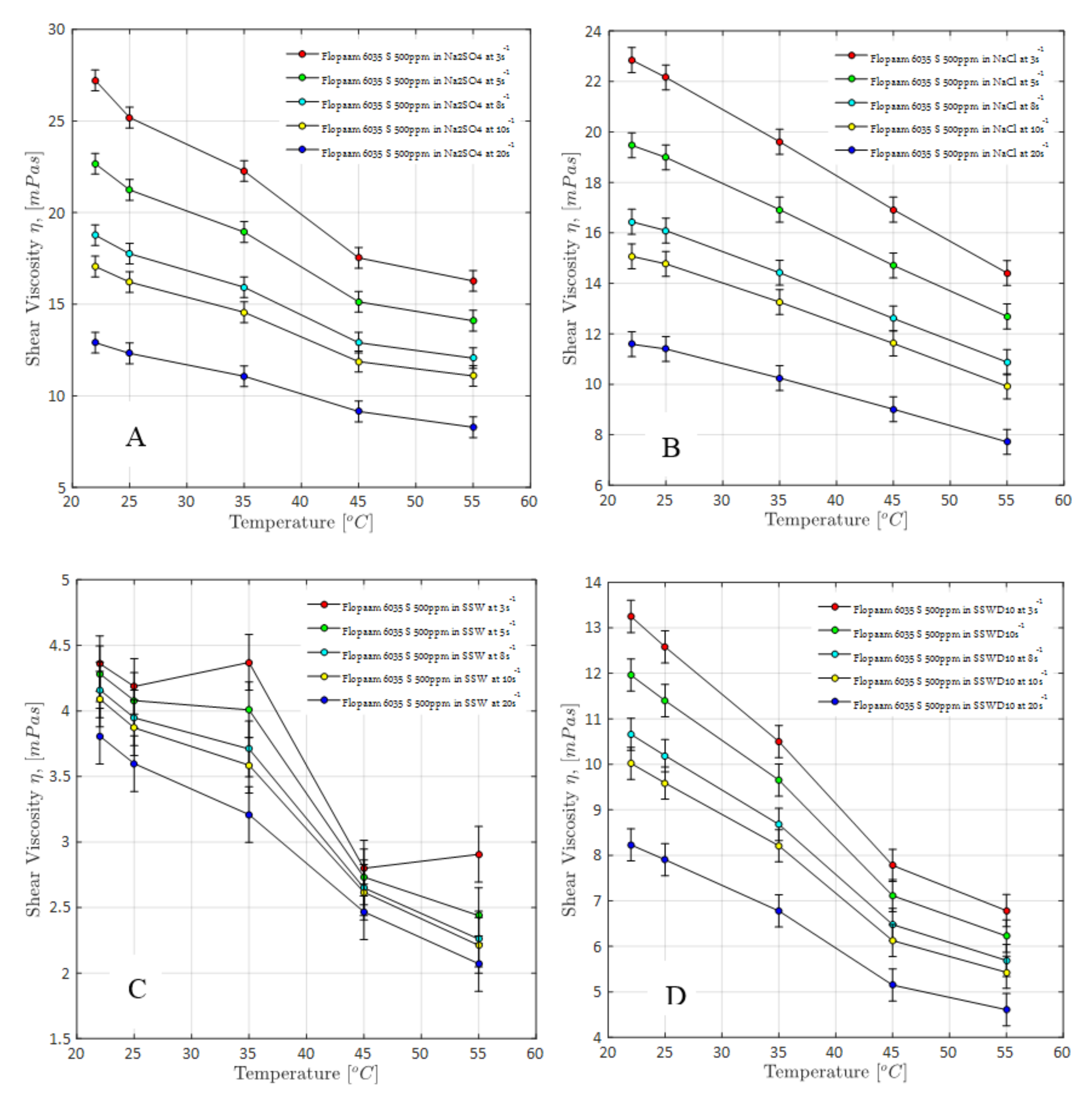
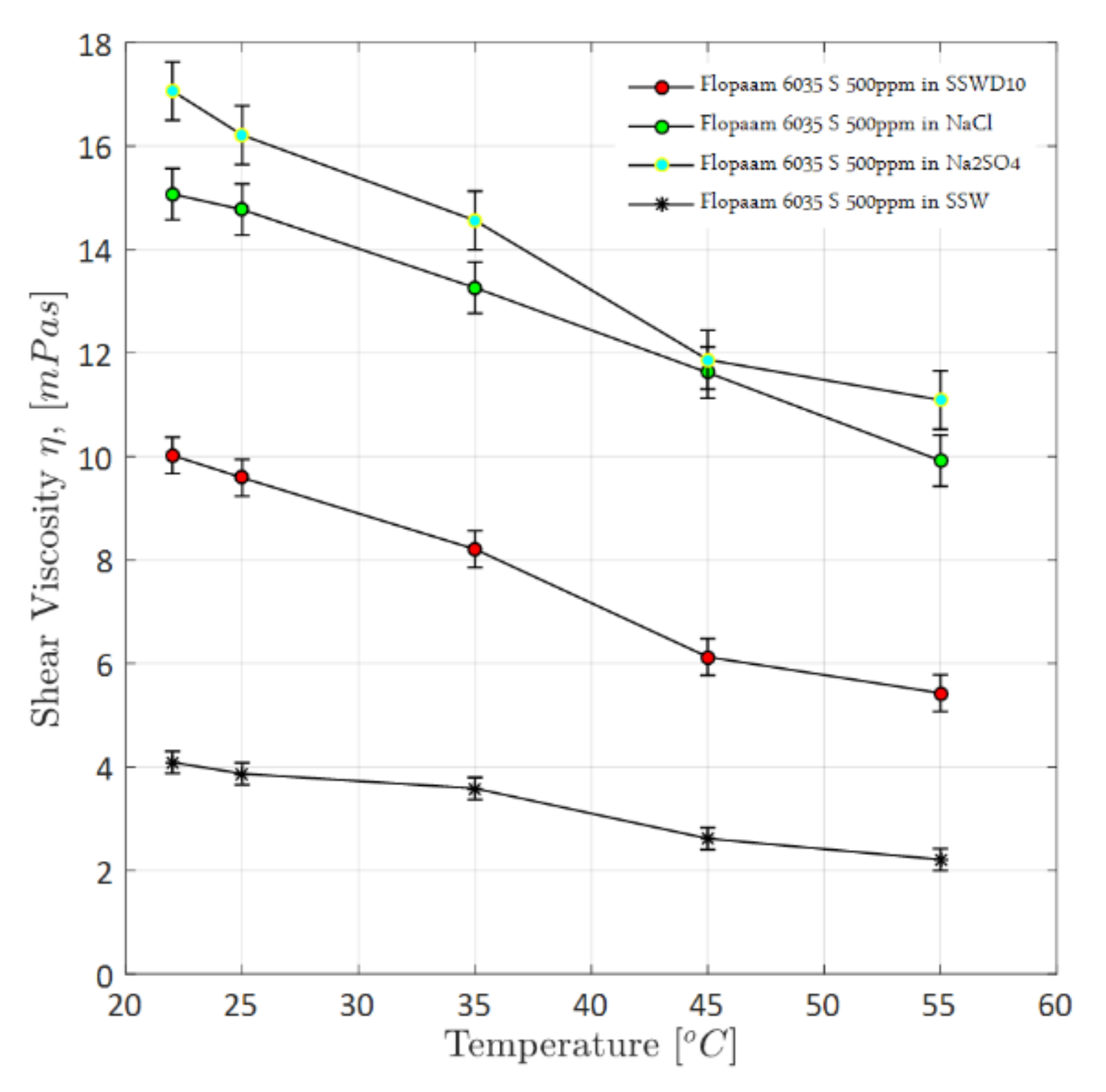
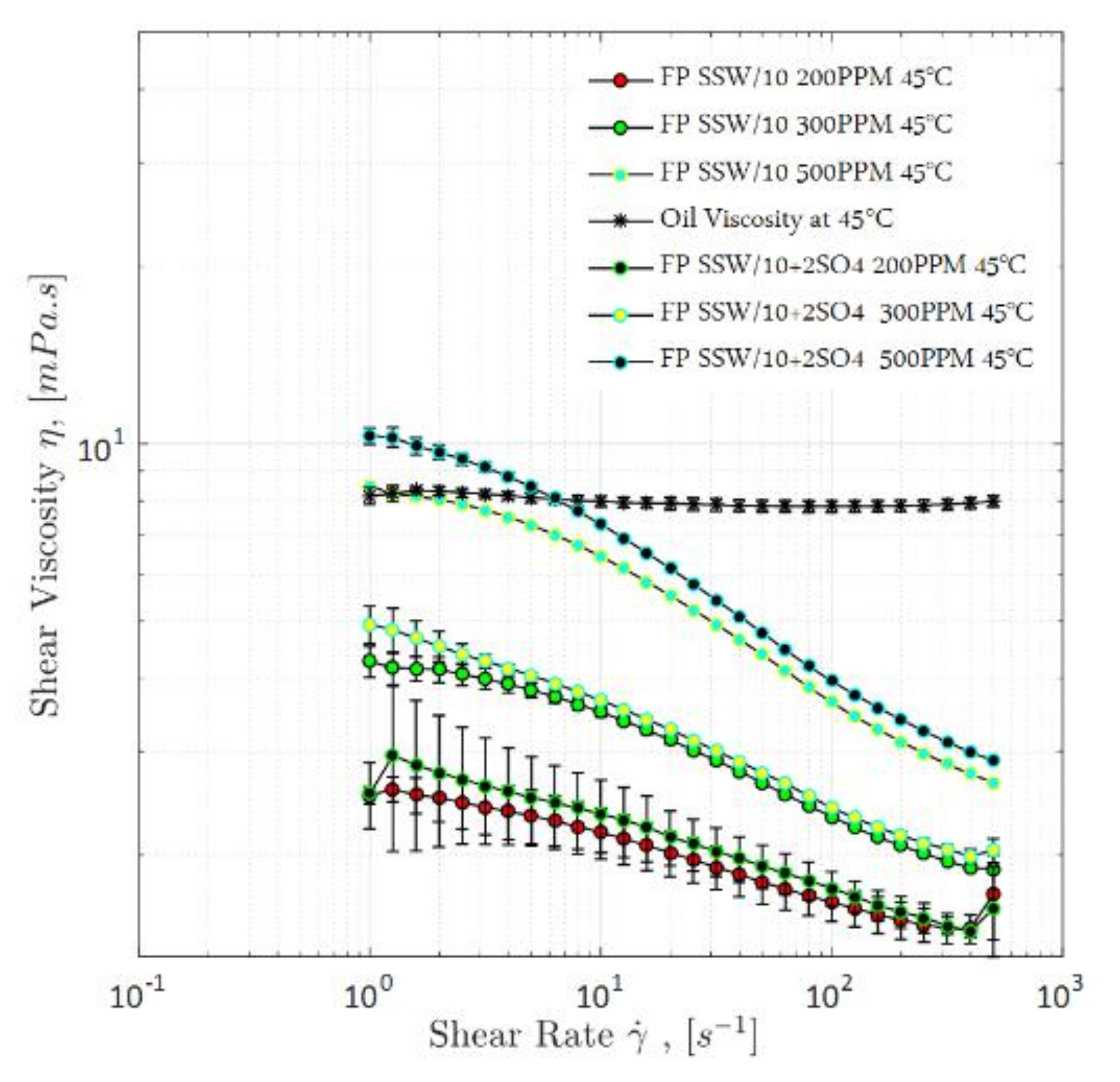
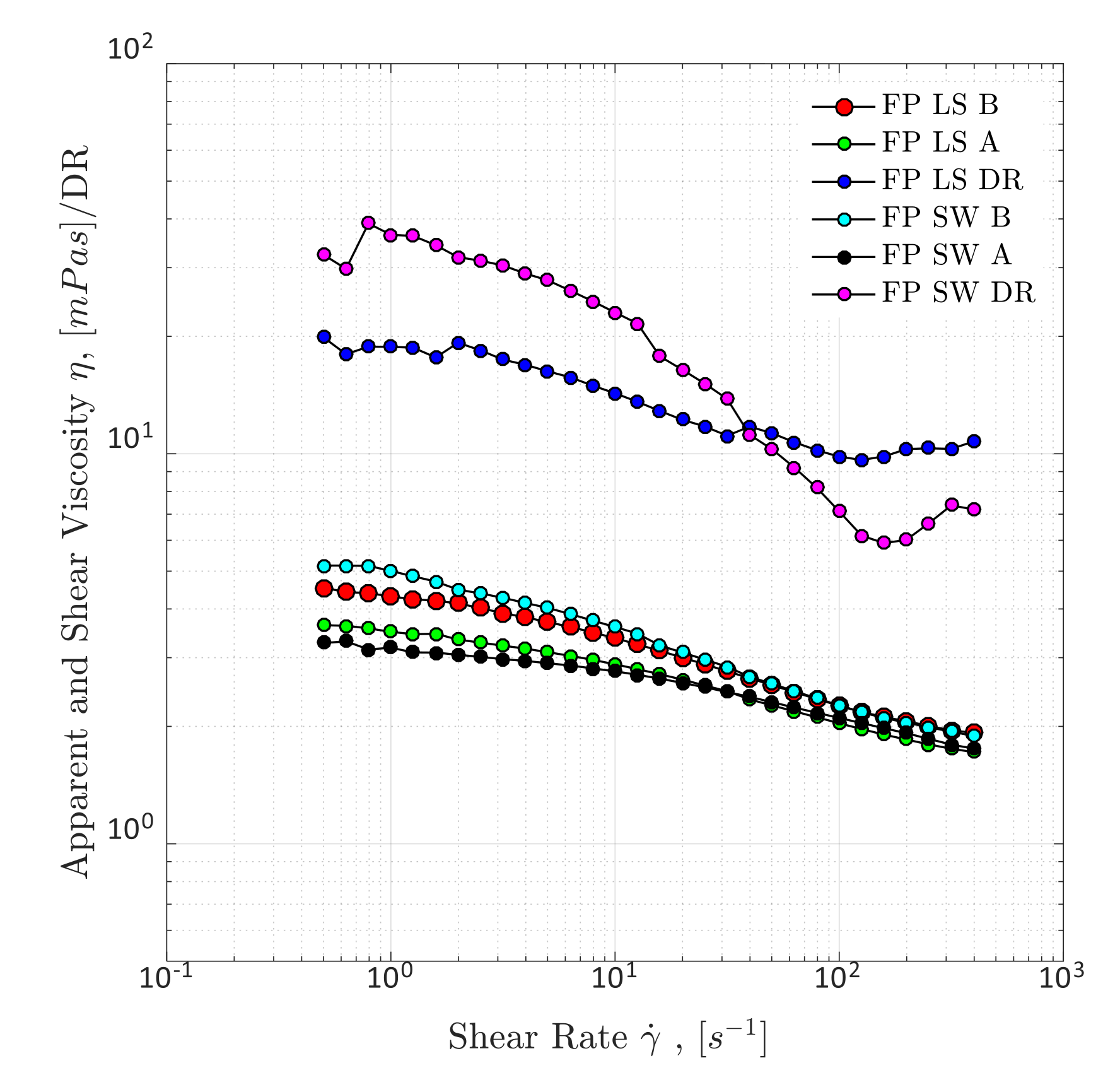
4.1.1. Steady-Shear Rheology (Shear Viscosity)
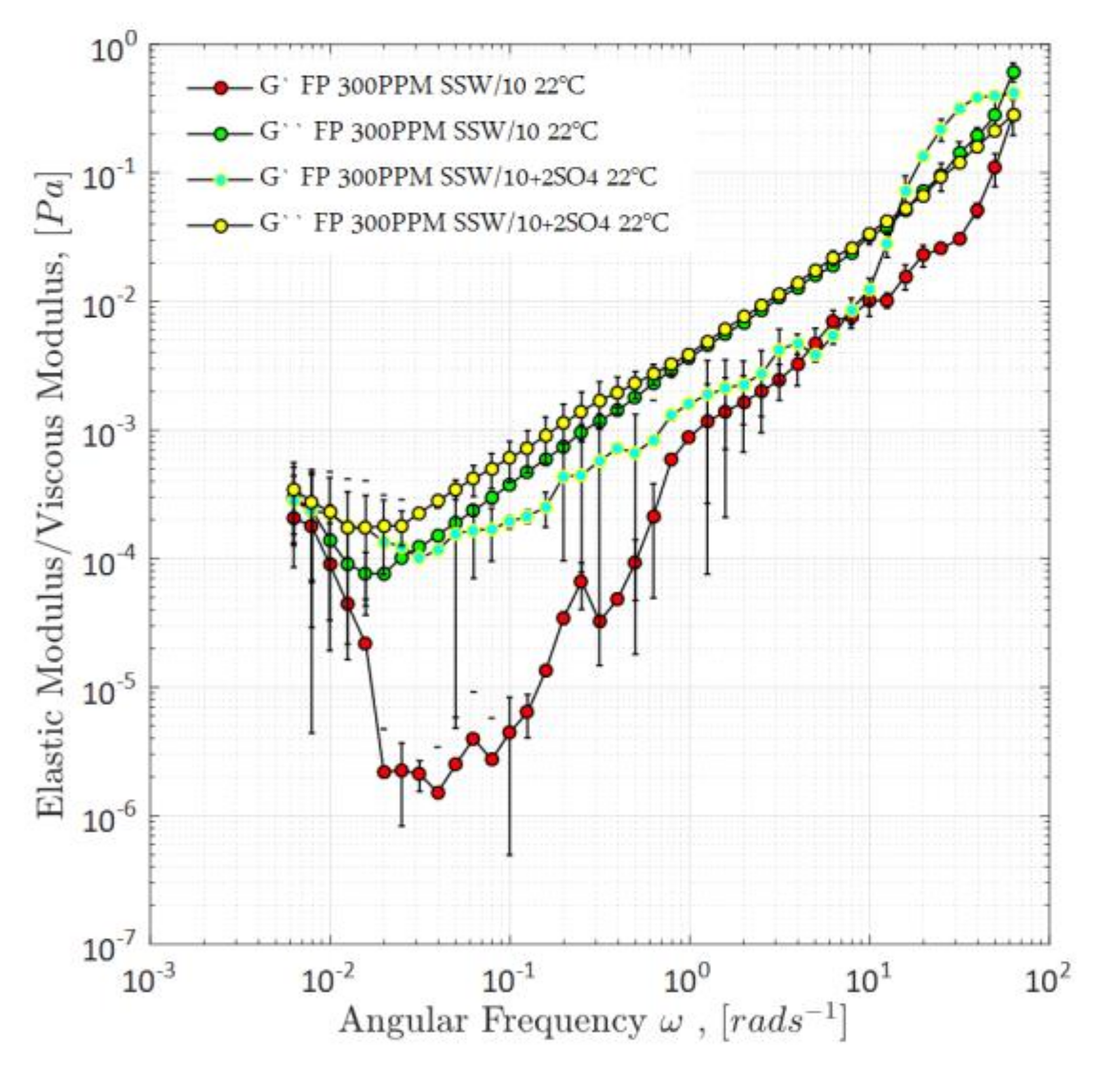
4.1.2. Viscoelasticity Evaluations—Small-Amplitude Oscillatory Shear and First Normal Stress Difference, N1
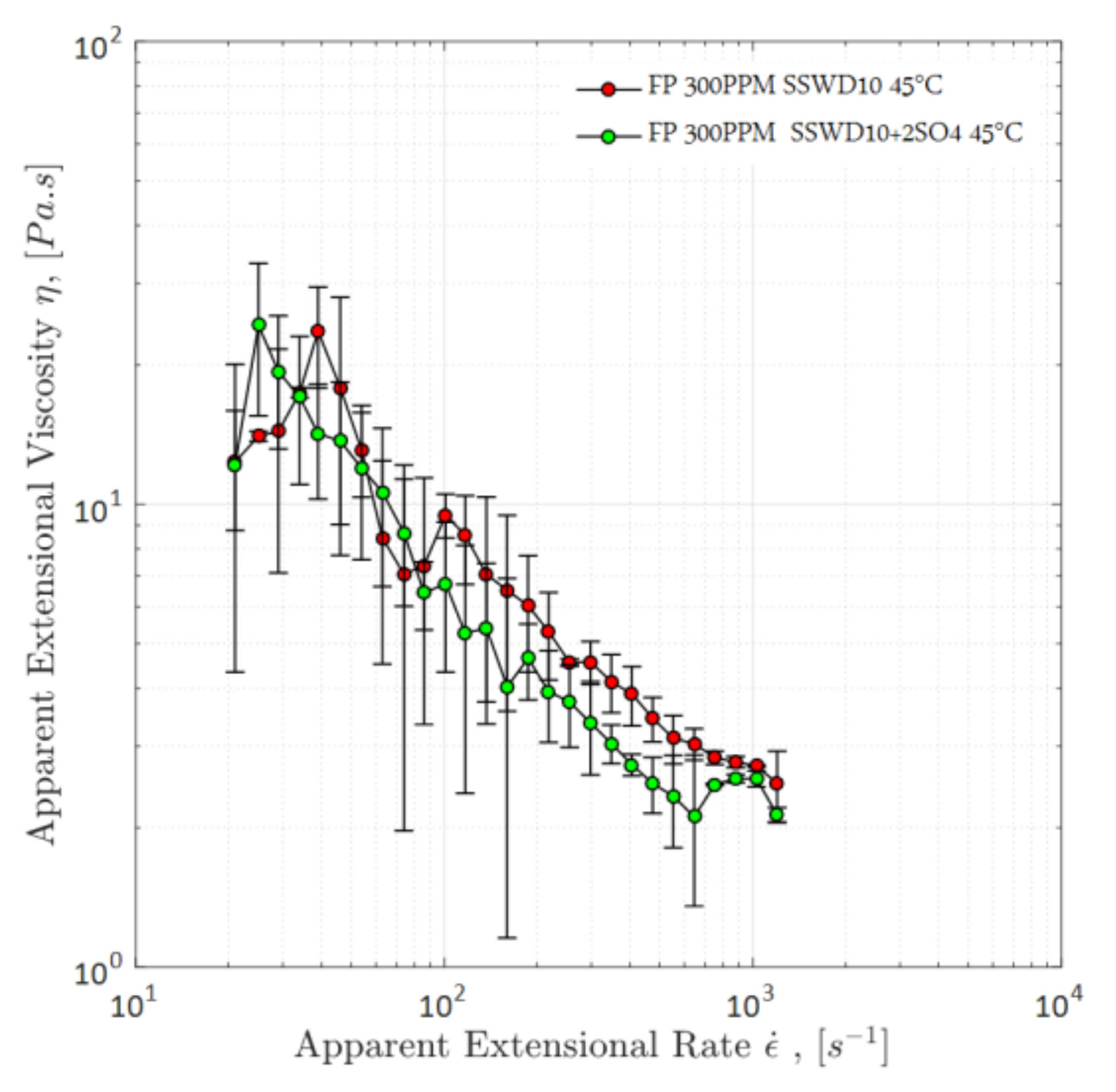
4.1.3. Apparent Elongational Viscosity
- The measurement errors, as described with error bars: At lower apparent extensional rates, both solutions exhibited a wider range of measurement errors compared to higher apparent extensional rates. One reason for the errors could be that the elongational measurement is not unique, and there is no device that can reproduce the same behavior of fluids. Moreover, at lower extensional rates, the sensitivity of measurement is higher compared to higher rates. The error bar indicates that the polymer prepared in SW is more sensitive than the polymer solution in DSSW (wider range of error bar).
- The measurement response: Nearly the same elongational viscosity was observed for both solutions, considering the range of error bars between apparent extensional rates of 10–100 s−1. At high extensional rates, the DSSW solution (without sulfates) had slightly higher elasticity compared to SW, but both exhibited a decline in elastic response between apparent extensional rates of 100–1000 s−1. For flow through porous media, the extensional rate for the low to medium range is more important compared to the high rates in Figure 8. Furthermore, the elongational response of a fluid can be different while flowing through porous media (core plug). Whereas flow through porous media fluid experiences series of converging-diverging geometries; for this measurement, only one hyperbolic shape converging-diverging geometry was used. Elongational response of fluid became stronger with multiple contraction and elongation of polymer molecules.
4.1.4. Overall Observations on Rheological Evaluations
4.1.5. Polymer Stability Analysis
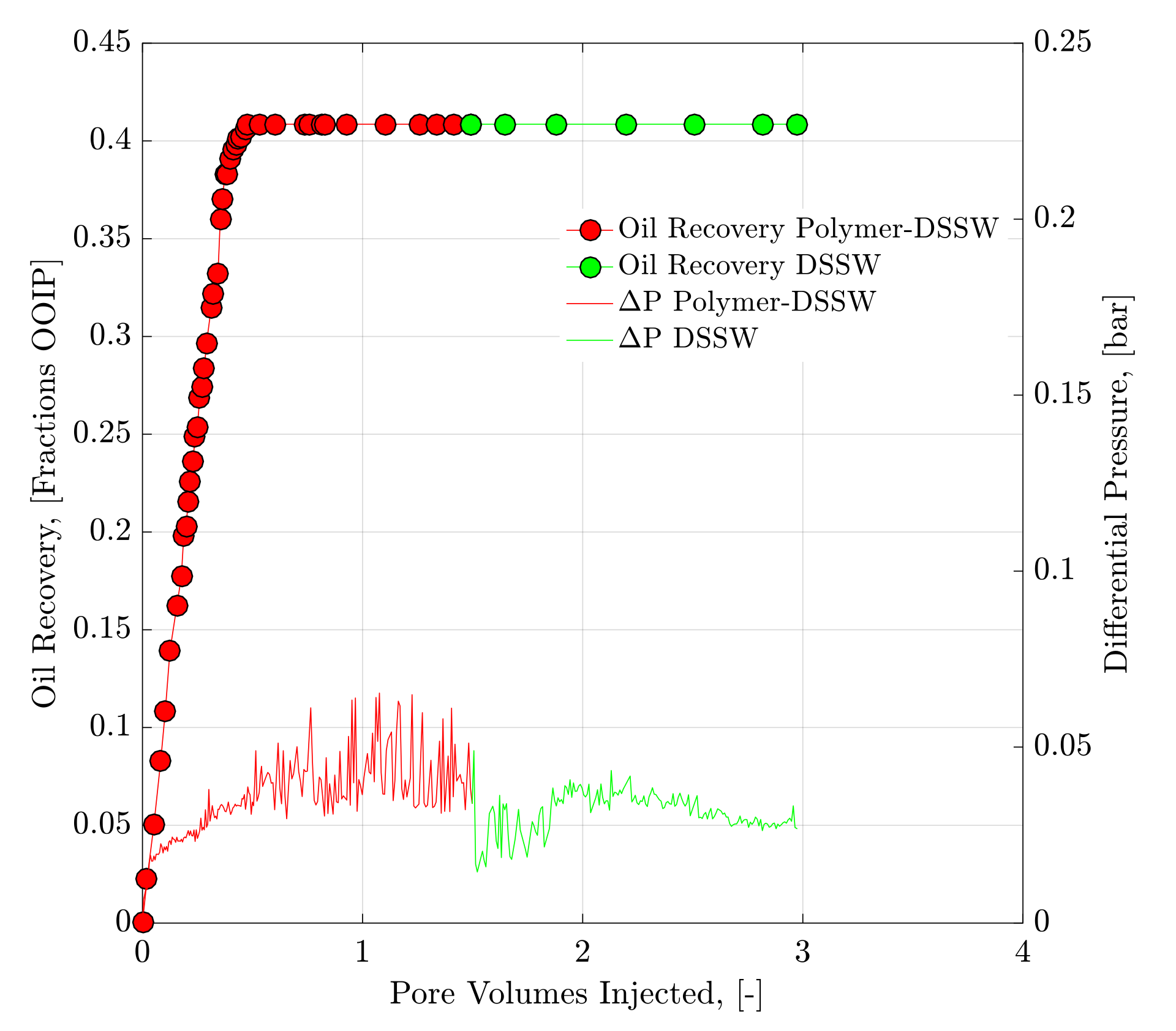
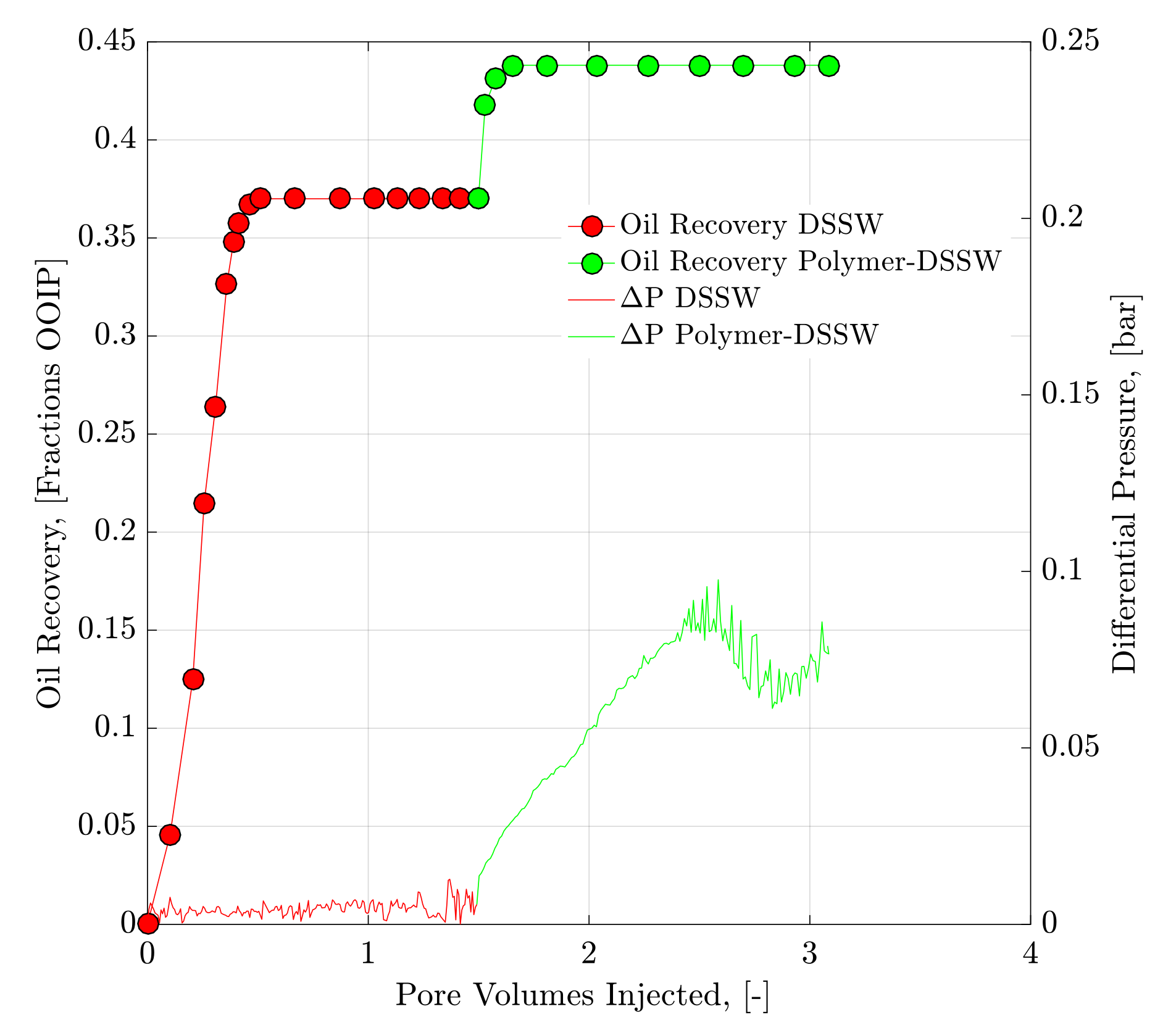
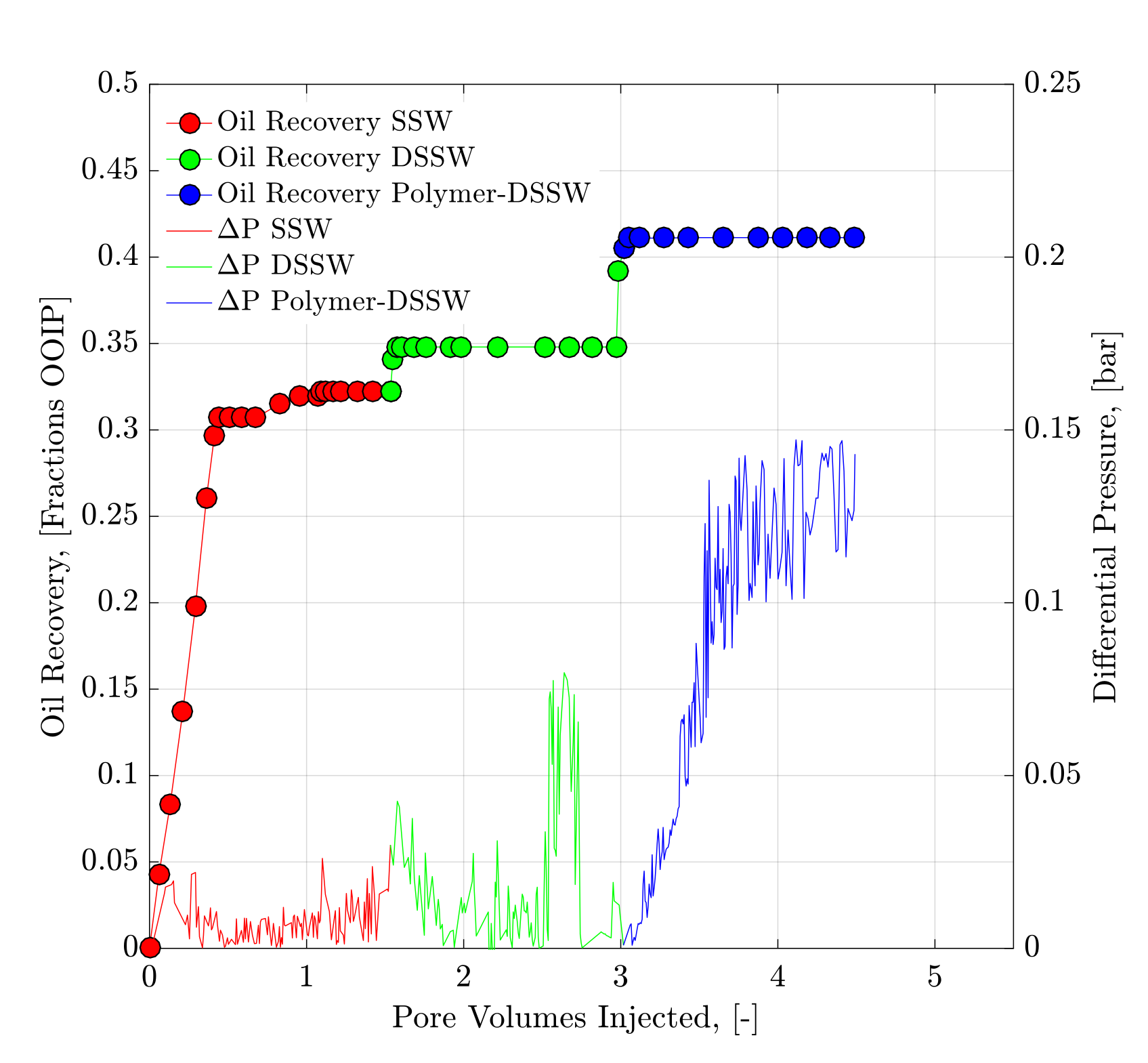
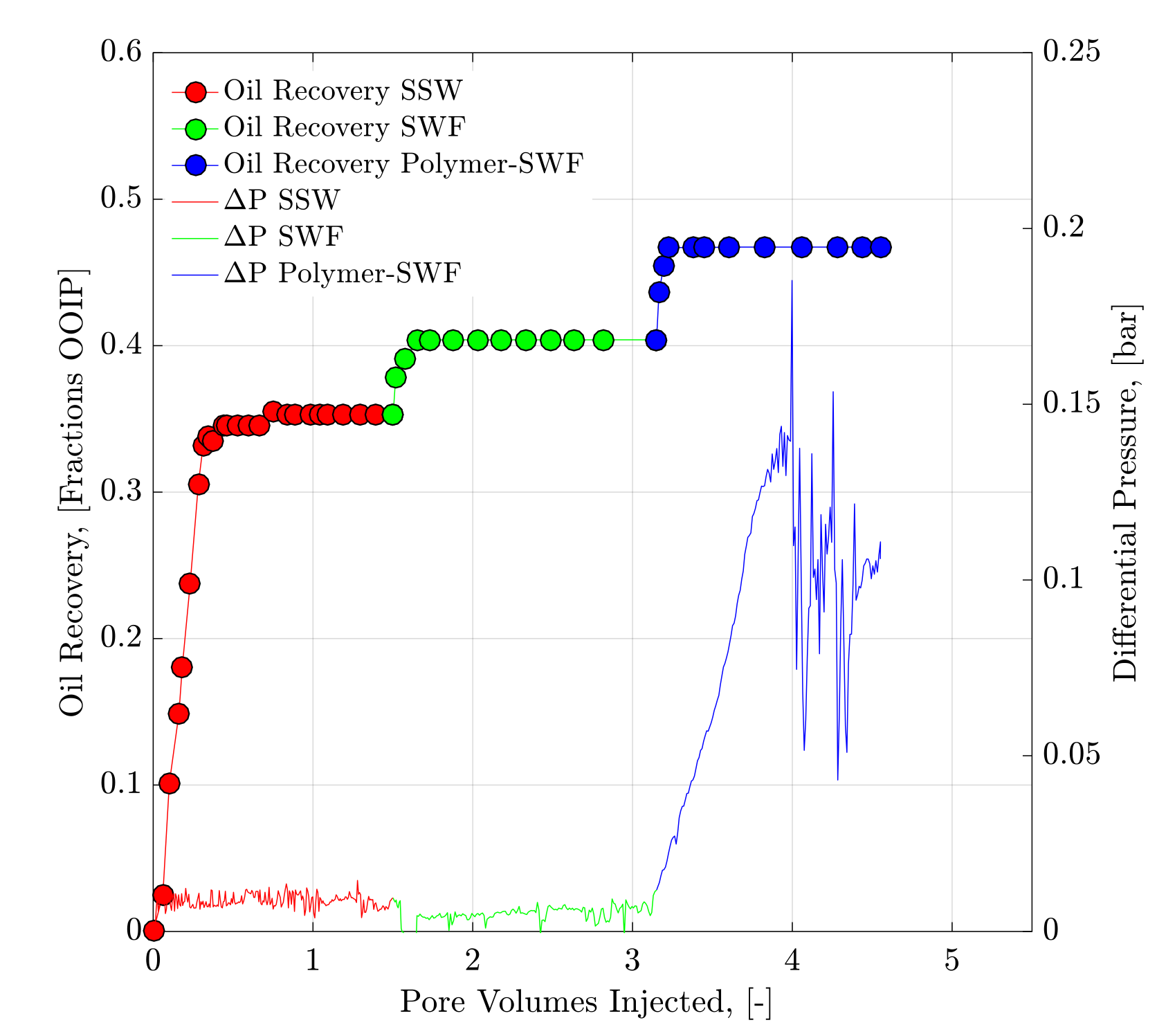
4.2. Core Flood Experiments and Oil Recovery Observations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations/Nomenclature
| A | After |
| B | Before |
| DR | Degradation Rate |
| EOR | Enhanced oil recovery |
| FP | Flopaam 6035 S |
| DSSW | Ten-times diluted synthetic seawater |
| DSSWF | Ten-times diluted synthetic seawater flood |
| OIIP | Oil initial in place |
| Polymer-DSSW | Polymer solution prepared in ten-times diluted synthetic seawater |
| Polymer-SW | Polymer solution prepared in sulfate-modified water |
| SR | Shear Rate |
| SSW | Synthetic sea water |
| SW | Smart water |
| SWF | Smart water flood |
| TDS | Total dissolved solids |
| ΔΡ | Pressure drop across the core |
| kb | Permeability to brine |
| kg | Permeability to gas |
References
- Martin, J.C. The Effects of Clay on the Displacement of Heavy Oil by Water. Venez. Ann. Meet. 1959. [Google Scholar] [CrossRef]
- Yousef, A.A.; Al-Saleh, S.; Al-Jawfi, M.S. Smart WaterFlooding for Carbonate Reservoirs: Salinity and Role of Ions. In Proceedings of the SPE Middle East Oil and Gas Show and Conference, Manama, Bahrain, 25–28 September 2011. [Google Scholar] [CrossRef]
- Nasralla, R.A.; Nasr-El-Din, H.A. Coreflood Study of Low Salinity Water Injection in Sandstone Reservoirs. SPE/DGS Saudi Arabia Sect. Tech. Symp. Exhib. 2011. [Google Scholar] [CrossRef]
- Shiran, B.S.; Skauge, A. Wettability and Oil Recovery by Low Salinity Injection. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar] [CrossRef]
- Suman, Y.K.; Shirif, E.; Ibrahim, H.; Ala-Ktiwi, A. Evaluation of Low Saline “Smart Water” Enhanced Oil Recovery in Light Oil Reservoirs. World J. Eng. Technol. 2014, 2, 13–22. [Google Scholar] [CrossRef]
- Bagci, S.; Kok, M.V.; Türksoy, U.; Baǧci, S. Effect of Brine Composition on oil Recovery by Waterflooding. Pet. Sci. Technol. 2001, 19, 359–372. [Google Scholar] [CrossRef]
- Ligthelm, D.J.; Gronsveld, J.; Hofman, J.; Brussee, N.; Marcelis, F.; Van Der Linde, H. Novel Waterflooding Strategy By Manipulation Of Injection Brine Composition. In Proceedings of the EUROPEC/EAGE Conference and Exhibition; Society of Petroleum Engineers (SPE), Amsterdam, The Netherlands, 8–11 June 2009. [Google Scholar] [CrossRef]
- Morrow, N.; Buckley, J. Improved Oil Recovery by Low-Salinity Waterflooding. J. Pet. Technol. 2011, 63, 106–112. [Google Scholar] [CrossRef]
- Webb, K.J.; Black, C.J.J.; Tjetland, G. A Laboratory Study Investigating Methods for Improving Oil Recovery in Carbonates. In Proceedings of the International Petroleum Technology Conference, Doha, Qatar, 21–23 November 2005. [Google Scholar] [CrossRef]
- Lager, A.; Webb, K.; Black, C. Impact of Brine Chemistry on Oil Recovery. In Proceedings of the IOR 2007—14th European Symposium on Improved Oil Recovery, Cairo, Egypt, 22–24 April 2007; EAGE Publications: Houten, The Netherlands, 2007. [Google Scholar]
- Awolayo, A.; Sarma, H.; Alsumaiti, A.M. A Laboratory Study of Ionic Effect of Smart Water for Enhancing Oil Recovery in Carbonate Reservoirs. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 31 March–2 April 2014. [Google Scholar] [CrossRef]
- Fathi, S.J.; Austad, T.; Strand, S. Water-Based Enhanced Oil recovery (EOR) by “Smart Water” in Carbonate Reservoirs. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar] [CrossRef]
- Zahid, A.; Stenby, E.H.; Shapiro, A.A. Smart Waterflooding (High Sal/Low Sal) in Carbonate Reservoirs. In Proceedings of the SPE Europec/EAGE Annual Conference, Copenhagen, Denmark, 4–7 June 2012. [Google Scholar] [CrossRef]
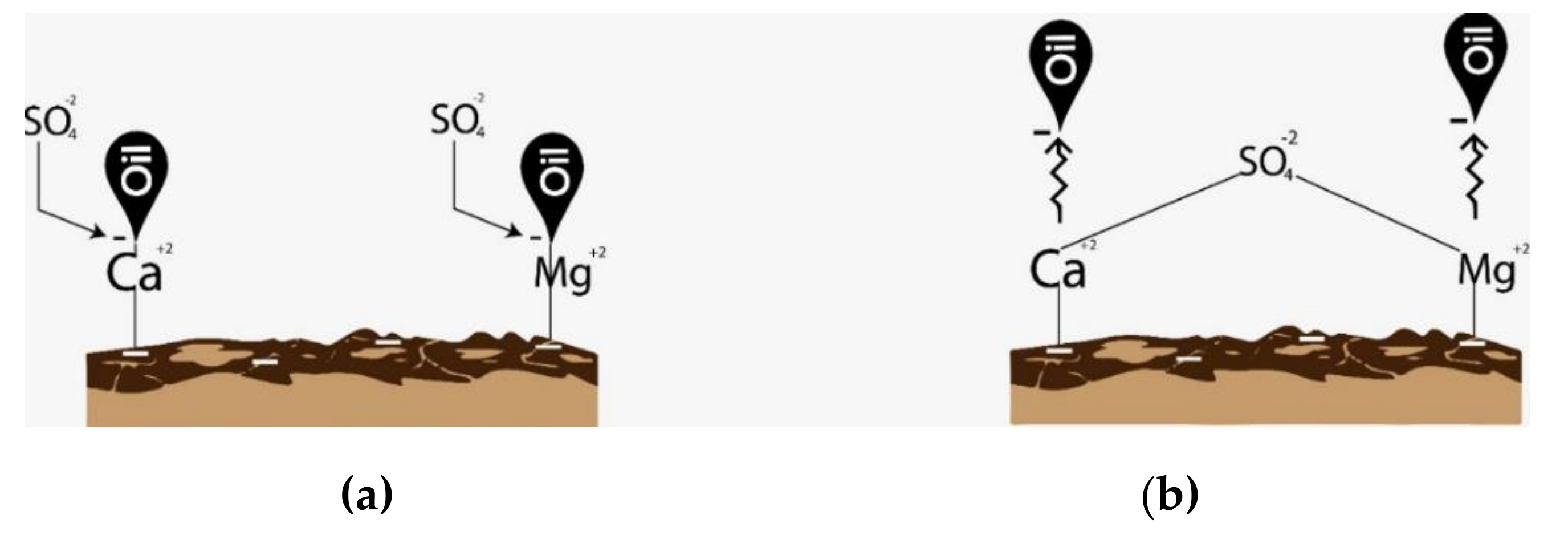
- Zhang, P.; Tweheyo, M.T.; Austad, T. Wettability alteration and improved oil recovery by spontaneous imbibition of seawater into chalk: Impact of the potential determining ions Ca2+, Mg2+, and SO42−. Colloids Surf. A Physicochem. Eng. Asp. 2007, 301, 199–208. [Google Scholar] [CrossRef]
- Zhang, P.; Tweheyo, M.T.; Austad, T. Wettability Alteration and Improved Oil Recovery in Chalk: The Effect of Calcium in the Presence of Sulfate. Energy Fuels 2006, 20, 2056–2062. [Google Scholar] [CrossRef]
- Gomari, K.R.; Hamouda, A.A.; Denoyel, R. Influence of sulfate ions on the interaction between fatty acids and calcite surface. Colloids Surf. A Physicochem. Eng. Asp. 2006, 287, 29–35. [Google Scholar] [CrossRef]
- Karoussi, O.; Hamouda, A.A. Imbibition of Sulfate and Magnesium Ions into Carbonate Rocks at Elevated Temperatures and Their Influence on Wettability Alteration and Oil Recovery. Energy Fuels 2007, 21, 2138–2146. [Google Scholar] [CrossRef]
- Garcia-Olvera, G.; Alvarado, V.; Alvarado, V. Interfacial rheological insights of sulfate-enriched smart-water at low and high-salinity in carbonates. Fuel 2017, 207, 402–412. [Google Scholar] [CrossRef]
- Bin Merdhah, A.B.; Yassin, A.A.M. Laboratory Study and Prediction of Calcium Sulphate at High-Salinity Formation Water. Open Pet. Eng. J. 2008, 1, 62–73. [Google Scholar] [CrossRef][Green Version]
- Yuan, M. Effect of Temperature on Barium Sulfate Scale Inhibition of Diethylene Triamine Penta (Methylene Phosphonic Acid). In Advances in Crystal Growth Inhibition Technologies; Springer Science and Business Media LLC: Boston, MA, USA, 2005; pp. 151–163. [Google Scholar] [CrossRef]
- Ghosh, B.; Sun, L.; Osisanya, S. Smart-Water EOR Made Smarter A Laboratory Development. In Proceedings of the International Petroleum Technology Conference, Bangkok, Thailand, 14–16 November 2016. [Google Scholar] [CrossRef]
- Andersen, K.; Halvorsen, E.; Sælensminde, T.; Østbye, N. Water Management in a Closed Loop—Problems and Solutions at Brage Field. In Proceedings of the SPE European Petroleum Conference, Paris, France, 24–25 October 2000. [Google Scholar] [CrossRef]
- Graham, G.; Boak, L.; Hobden, C. Examination of the Effect of Generically Different Scale Inhibitor Species (PPCA and DETPMP) on the Adherence and Growth of Barium Sulphate Scale on Metal Surfaces. Int. Symp. Oilfield Scale 2001. [Google Scholar] [CrossRef]
- Luo, H.; Al Shalabi, P.E.W.; Delshad, M.; Panthi, K.; Sepehrnoori, K. A Robust Geochemical Simulator to Model Improved-Oil-Recovery Methods. SPE J. 2016, 21, 55–73. [Google Scholar] [CrossRef]
- Shiran, B.S.; Skauge, A. Enhanced Oil Recovery (EOR) by Combined Low Salinity Water/Polymer Flooding. Energy Fuels 2013, 27, 1223–1235. [Google Scholar] [CrossRef]
- Sheng, J.J. Critical review of low-salinity waterflooding. J. Pet. Sci. Eng. 2014, 120, 216–224. [Google Scholar] [CrossRef]
- Sheng, J.J.; Leonhardt, B.; Azri, N. Status of Polymer-Flooding Technology. J. Can. Pet. Technol. 2015, 54, 116–126. [Google Scholar] [CrossRef]
- Shiran, B.S.; Skauge, A. Similarities and Differences of Low Salinity Polymer and Low Salinity LPS (Linked Polymer Solutions) for Enhanced Oil Recovery. J. Dispers. Sci. Technol. 2014, 35, 1656–1664. [Google Scholar] [CrossRef]
- Sorbie, K.S.; Phil, K.S.S.D. Polymer-Improved Oil Recovery; Springer: Dordrecht, The Netherlands, 1991; ISBN 978-94-011-3044-8. [Google Scholar]
- Tahir, M.; Hincapie, R.E.; Be, M.; Ganzer, L. Experimental Evaluation of Polymer Viscoelasticity during Flow in Porous Media: Elongational and Shear Analysis. In Proceedings of the SPE Europec Featured at 79th EAGE Conference and Exhibition, Paris, France, 12–15 June 2017. [Google Scholar] [CrossRef]
- Tahir, M.; Hincapie, R.E. Coexistence of Shear and Elongational Components of Flow Paths through Porous Media during Polymer-Flooding Applications. In Proceedings of the 77th EAGE Conference and Exhibition, Madrid, Spain, 1–4 June 2015. [Google Scholar]
- Vermolen, E.C.; Van Haasterecht, M.J.; Masalmeh, S.K.; Faber, M.J.; Boersma, D.M.; Gruenenfelder, M.A. Pushing the envelope for polymer flooding towards high-temperature and high-salinity reservoirs with polyacrylamide based ter-polymers. In Proceedings of the SPE Middle East Oil and Gas Show and Conference, Manama, Bahrain, 25–28 September 2011. [Google Scholar] [CrossRef]
- Be, M.; Hincapie, R.E.; Rock, A.; Gaol, C.L.; Tahir, M.; Ganzer, L. Comprehensive Evaluation of the EOR Polymer Viscoelastic Phenomenon at Low Reynolds Number. In Proceedings of the SPE Europec Featured at 79th EAGE Conference and Exhibition, Paris, France, 12–15 June 2017. [Google Scholar] [CrossRef]
- Elhajjaji, R.R.; Hincapie, R.E.; Tahir, M.; Rock, A.; Wegner, J.; Ganzer, L. Systematic Study of Viscoelastic Properties During Polymer-Surfactant Flooding in Porous Media (Russian). In Proceedings of the SPE Russian Petroleum Technology Conference and Exhibition, Moscow, Russia, 24–26 October 2016. [Google Scholar] [CrossRef]
- Hincapie, R.E. Pore-Scale Investigation of the Viscoelastic Phenomenon during Enhanced Oil Recovery (EOR) Polymer Flooding through Porous Media; Papierflieger: Clausthal-Zellerfeld, Germany, 2016. [Google Scholar]
- Hincapie, R.E.; Duffy, J.; O’Grady, C.; Ganzer, L. An Approach to Determine Polymer Viscoelasticity Under Flow Through Porous Media by Combining Complementary Rheological Techniques. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 11–13 August 2015. [Google Scholar]
- Garcia-Olvera, G.; Alvarado, V. The Potential of Sulfate as Optimizer of Crude Oil-Water Interfacial Rheology to Increase Oil Recovery During Smart Water Injection in Carbonates. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 11–13 April 2016. [Google Scholar] [CrossRef]
- Skauge, A.; Shiran, B. Low Salinity Polymer Flooding. In Proceedings of the IOR 2013—17th European Symposium on Improved Oil Recovery, Saint Petersburg, Russia, 16–18 April 2013. [Google Scholar] [CrossRef]
- Sohrabi, M.; Mahzari, P.; Farzaneh, S.A.; Mills, J.R.; Tsolis, P.; Ireland, S. Novel Insights into Mechanisms of Oil Recovery by Use of Low-Salinity-Water Injection. SPE J. 2017, 22, 407–416. [Google Scholar] [CrossRef]
- Graham, G.M.; Boak, L.S.; Hobden, C.M. Examination of the Effect of Generically Different Scale Inhibitor Species (PPCA and DETPMP) on the Adherence and Growth of Barium Sulphate Scale on Metal Surfaces. In International Symposium on Oilfield Scale, Aberdeen, UK, 30–31 January 2001; Society of Petroleum Engineers: Amsterdam, The Netherlands, 2001. [Google Scholar] [CrossRef]
- Wei, B.; Romero-Zerón, L.; Rodrigue, D. Oil displacement mechanisms of viscoelastic polymers in enhanced oil recovery (EOR): A review. J. Pet. Explor. Prod. Technol. 2013, 4, 113–121. [Google Scholar] [CrossRef]
- Al-Saedi, H.; Flori, R.E.; Mortadha, A. Investigation of Smart Water Flooding in Sandstone Reservoirs: Experimental and Simulation Study Part 2; Missouri University of Science and Technology: Rolla, MO, USA, 2018. [Google Scholar] [CrossRef]
- Mohamed, M.I.; Alvarado, V. Smart Water Flooding in Berea Sandstone at Low Temperature: Is Wettability Alteration the Sole Mechanism at Play? In Proceedings of the SPE Annual Technical Conference and Exhibition, San Antonio, TX, USA, 9–11 October 2017. [Google Scholar] [CrossRef]
- Verruto, V.J.; Kilpatrick, P.K. Water-in-Model Oil Emulsions Studied by Small-Angle Neutron Scattering: Interfacial Film Thickness and Composition. Langmuir 2008, 24, 12807–12822. [Google Scholar] [CrossRef]
- Hybrid Enhanced Oil Recovery using Smart Waterflooding; Gulf Professional Publishing: Houston, TX, USA, 2019; ISBN 9780128172988.
- Torrijos, I.D.P.; Puntervold, T.; Strand, S.; Austad, T.; Bleivik, T.H.; Abdullah, H.I. An experimental study of the low salinity Smart Water—Polymer hybrid EOR effect in sandstone material. J. Pet. Sci. Eng. 2018, 164, 219–229. [Google Scholar] [CrossRef]
- Shabib-Asl, A.; Ayoub, M.A.; Elraies, K.A. A new hybrid technique using low salinity water injection and foam flooding for enhanced oil recovery in sandstone rock. J. Pet. Sci. Eng. 2019, 174, 716–728. [Google Scholar] [CrossRef]
- Kilybay, A.; Ghosh, B.; Thomas, N.C.; Sulemana, N.T. Hybrid EOR Technology: Carbonated Water-Smart Water Flood Improved Recovery in Oil Wet Carbonate Formation: Part-II. In Proceedings of the SPE Oil and Gas India Conference and Exhibition, Mumbai, India, 4–6 April 2017. [Google Scholar] [CrossRef]
- Xu, X.; Saeedi, A. Evaluation and Optimization Study on a Hybrid EOR Technique Named as Chemical-Alternating-Foam Floods. Oil Gas Sci. Technol. Rev. de l’IFP 2017, 72, 1. [Google Scholar] [CrossRef][Green Version]
- Almansour, A.O.; AlQuraishi, A.A.; AlHussinan, S.N.; Alyami, H.Q. Efficiency of enhanced oil recovery using polymer-augmented low salinity flooding. J. Pet. Explor. Prod. Technol. 2017, 7, 1149–1158. [Google Scholar] [CrossRef]
- Kozaki, C. Efficiency of Low Salinity Polymer Flooding in Sandstone Cores. Master’s Thesis, The University of Texas at Austin, Austin, TX, USA, 2012. [Google Scholar]
- Mohammadi, H.; Jerauld, G. Mechanistic Modeling of the Benefit of Combining Polymer with Low Salinity Water for Enhanced Oil Recovery. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 14–18 April 2012. [Google Scholar] [CrossRef]
- Pourafshary, P.; Moradpour, N. Hybrid EOR Methods Utilizing Low-Salinity Water. Enhanc. Oil Recovery Process. New Technol. 2019. [Google Scholar] [CrossRef]
- AlSofi, A.M.; Wang, J.; AlBoqmi, A.M.; Alotaibi, M.B.; Ayirala, S.; Alyousef, A. Smart Water Synergy with Chemical EOR for a Slightly Viscous Arabian Heavy Reservoir. In Proceedings of the SPE Heavy Oil Conference and Exhibition, Kuwait City, Kuwait, 6–8 December 2016. [Google Scholar] [CrossRef]
- Tabary, R.; Douarche, F.; Bazin, B.; Lemouzy, P.M.; Moreau, P.; Morvan, M. Design of a Surfactant/Polymer Process in a Hard Brine Context: A Case Study Applied to Bramberge Reservoir. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar] [CrossRef]
- Tay, A.; Oukhemanou, F.; Wartenberg, N.; Moreau, P.; Guillon, V.; Delbos, A.; Tabary, R. Adsorption Inhibitors: A New Route to Mitigate Adsorption in Chemical Enhanced Oil Recovery. In Proceedings of the SPE Asia Pacific Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 11–13 August 2015. [Google Scholar] [CrossRef]
- Skauge, T.; Djurhuus, K.; Reichenbach-Klinke, R. Visualization of Heavy Oil Mobilization by Associative Polymer. In Proceedings of the SPE Europec featured at 80th EAGE Conference and Exhibition, Copenhagen, Denmark, 11–14 June 2018. [Google Scholar] [CrossRef]
- Severs, E.T.; Austin, J.M. Flow Properties of Vinyl Chloride Resin Plastisols. Ind. Eng. Chem. 1954, 6, 2369–2375. [Google Scholar] [CrossRef]
- Wang, D.; Wang, G.; Xia, H. Large Scale High Visco-Elastic Fluid Flooding in the Field Achieves High Recoveries. In Proceedings of the SPE Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 19–21 July 2011. [Google Scholar] [CrossRef]
- Yin, H.; Wang, D.; Zhong, H. Study on Flow Behavoirs of Viscoelastic Polymer Solution in Micropore with Dead End. In Proceedings of the SPE Annual Technical Conference and Exhibition, San Antonio, TX, USA, 24–27 September 2006. [Google Scholar] [CrossRef]
- Demin, W.; Jiecheng, C.; Huifen, X.; Qun, L.; Jingping, S. Viscous-Elastic Fluids Can Mobilize Oil Remaining after Water-Flood by Force Parallel to the Oil-Water Interface. In Proceedings of the SPE Asia Pacific Improved Oil Recovery Conference, Kuala Lumpur, Malaysia, 6–9 October 2001. [Google Scholar] [CrossRef]
- Tahir, M.; Hincapie, R.E.; Foedisch, H.; Abdullah, H.; Ganzer, L. Impact of Sulphates Presence During Application of Smart Water Flooding Combined with Polymer Flooding. In Proceedings of the SPE Europec Featured at 80th EAGE Conference and Exhibition, Copenhagen, Denmark, 11–14 June 2018. [Google Scholar] [CrossRef]
- Tahir, M.; Hincapie, R.E.; Foedisch, H.; Strobel, G.-J.; Ganzer, L. Potential Benefits of Fluid Optimization for Combined Smart-Water and Polymer Flooding: Impact on Remaining Oil Saturation. In Proceedings of the SPE Russian Petroleum Technology Conference, Moscow, Russia, 22–24 October 2019. [Google Scholar] [CrossRef]

| Chemical Formula | Composition (g/L) | |||
|---|---|---|---|---|
| SSW* | SSW/10 | (NaCl)5 | (Na2SO4)5 | |
| NaCl | 23.9744 | 2.3974 | 5.0000 | - |
| KCl | 0.8009 | 0.0801 | - | - |
| CaCl₂ | 1.1077 | 0.1108 | - | - |
| MgCl₂·6H₂O | 11.0412 | 1.1042 | - | - |
| SrCl₂·6H₂O | 0.0268 | 0.0027 | - | - |
| Na₂SO₄ | 3.9332 | 0.3933 | - | 5.0000 |
| NaHCO₃ | 0.2758 | 0.0275 | - | - |
| TDS** | 41.1596 | 4.1160 | 5.0000 | 5.0000 |
| Hardness (R+1) | 0.1300 | 0.1300 | 0 | 0 |
| Chemical Formula | Composition (g/L) | |||
|---|---|---|---|---|
| Brine 1 | Brine 2 | Brine 3 (DSSW*) | Brine 4 (SW) | |
| SSW | SSW + 2SO4 | SSW/10 | (SSW + 2SO4)/10 | |
| Nacl | 23.9744 | 23.9744 | 2.3974 | 2.3974 |
| KCl | 0.8009 | 0.8009 | 0.0801 | 0.0800 |
| CaCl₂ | 1.1077 | 1.1077 | 0.1108 | 0.1107 |
| MgCl₂·6H₂O | 11.0412 | 11.0412 | 1.1041 | 1.1041 |
| SrCl₂·6H₂O | 0.0268 | 0.0268 | 0.0027 | 0.0026 |
| Na₂SO₄ | 3.9332 | 7.8664 | 0.3933 | 0.7866 |
| NaHCO₃ | 0.2754 | 0.2754 | 0.0275 | 0.0275 |
| TDS | 41.1596 | 45.0928 | 4.1156 | 4.5092 |
| Hardness (R+1) | 0.1300 | 0.1150 | 0.1300 | 0.1150 |
| pH @ 22 °C | 5.8300 | - | 5.7800 | 5.2800 |
| IFT (mN/m) with Oil @ 22 °C | - | - | 9.4000 | 8.8700 |
| Core | L | D | phi, Φ | kg | kb | PV | Swc | Soi |
|---|---|---|---|---|---|---|---|---|
| mm | mm | % | mD | mD | ml | % | % | |
| Bent 5.1 | 60.10 | 29.55 | 23.33 | 2510 | 1684 | 9.61 | 18.15 | 81.85 |
| Bent 5.2 | 59.95 | 29.55 | 23.69 | 2714 | 1964 | 9.74 | 24.60 | 75.40 |
| Bent 5.3 | 60.10 | 29.50 | 23.54 | 2835 | 1976 | 9.67 | 24.60 | 75.40 |
| Bent 5.4 | 60.00 | 29.55 | 24.10 | 2848 | 1608 | 9.91 | 20.60 | 79.40 |
| Bent 5.5 | 60.05 | 29.55 | 24.10 | 3029 | 2114 | 9.92 | 20.70 | 79.30 |
| Mean | 60.04 | 29.54 | 23.75 | 2787 | 1869 | 9.77 | 21.73 | 78.27 |
| No. | Core | Secondary | Bump-Rate | Tertiary | Quaternary |
|---|---|---|---|---|---|
| 1 | Bent 5.1 | Polymer-DSSW | - | DSSWF | |
| 2 | Bent 5.2 | DSSWF | Bump-Rate | Polymer-DSSW | |
| 3 | Bent 5.3 | SWF | Bump-Rate | Polymer-SW | |
| 4 | Bent 5.4 | SSW | Bump-Rate | DSSWF | Polymer-DSSW |
| 5 | Bent 5.5 | SSW | Bump-Rate | SWF | Polymer-SW |
| Q (mL/min) | 0.10 | 0.33 | 0.15 | 0.15 | |
| Q (ft/day) | 1 | 3.3 | 1.5 | 1.5 | |
| Experimental Description | Name | Φ (%) | PV (cm3) | k, Brine (md) | k, gas (md) | Soi (%) | Swc (%) | Additional RF (OIIP) (%) | Total RF (OIIP) EOR (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Secondary Mode 45 °C | ||||||||||
| 1 | Polymer-DSSW | Bent 5.1 | 23.33 | 9.61 | 1684 | 2510 | 81.85 | 18.15 | - | 40.85 |
| 2 | DSSWF | Bent 5.2 | 23.69 | 9.74 | 1964 | 2714 | 75.40 | 24.60 | - | 36.90 |
| 3 | SWF | Bent 5.3 | 23.54 | 9.67 | 1976 | 2835 | 75.40 | 24.60 | - | 37.87 |
| 4 | SSW | Bent 5.4 | 24.10 | 9.91 | 1608 | 2848 | 79.40 | 20.60 | - | 32.22 |
| 5 | SSW | Bent 5.5 | 24.10 | 9.92 | 2114 | 3029 | 79.30 | 20.70 | - | 35.27 |
| Tertiary Mode 45 °C | ||||||||||
| 1 | DSSWF | Bent 5.1 | 23.33 | 9.61 | 1684 | 2510 | 81.85 | 18.15 | 0 | 40.85 |
| 2 | Polymer-DSSW | Bent 5.2 | 23.69 | 9.74 | 1964 | 2714 | 75.40 | 24.60 | 6.90 | 43.80 |
| 3 | Polymer-SW | Bent 5.3 | 23.54 | 9.67 | 1976 | 2835 | 75.40 | 24.60 | 9.60 | 47.47 |
| 4 | DSSWF | Bent 5.4 | 24.10 | 9.91 | 1608 | 2848 | 79.40 | 20.60 | 2.50 | 34.72 |
| 5 | SWF | Bent 5.5 | 24.10 | 9.92 | 2114 | 3029 | 79.30 | 20.70 | 5.00 | 40.27 |
| Post-tertiary (Quaternary) Mode 45 °C | ||||||||||
| 4 | Polymer-DSSW | Bent 5.4 | 24.10 | 9.91 | 1608 | 2848 | 79.40 | 20.60 | 6.30 | 41.02 |
| 5 | Polymer-SW | Bent 5.5 | 24.10 | 9.92 | 2114 | 3029 | 79.30 | 20.70 | 6.40 | 46.67 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tahir, M.; Hincapie, R.E.; Ganzer, L. Influence of Sulfate Ions on the Combined Application of Modified Water and Polymer Flooding—Rheology and Oil Recovery. Energies 2020, 13, 2356. https://doi.org/10.3390/en13092356
Tahir M, Hincapie RE, Ganzer L. Influence of Sulfate Ions on the Combined Application of Modified Water and Polymer Flooding—Rheology and Oil Recovery. Energies. 2020; 13(9):2356. https://doi.org/10.3390/en13092356
Chicago/Turabian StyleTahir, Muhammad, Rafael E. Hincapie, and Leonhard Ganzer. 2020. "Influence of Sulfate Ions on the Combined Application of Modified Water and Polymer Flooding—Rheology and Oil Recovery" Energies 13, no. 9: 2356. https://doi.org/10.3390/en13092356
APA StyleTahir, M., Hincapie, R. E., & Ganzer, L. (2020). Influence of Sulfate Ions on the Combined Application of Modified Water and Polymer Flooding—Rheology and Oil Recovery. Energies, 13(9), 2356. https://doi.org/10.3390/en13092356







