High Reynold’s Number Turbulent Model for Micro-Channel Cold Plate Using Reverse Engineering Approach for Water-Cooled Battery in Electric Vehicles
Abstract
1. Introduction
2. State of the Art
3. Experimental Studies
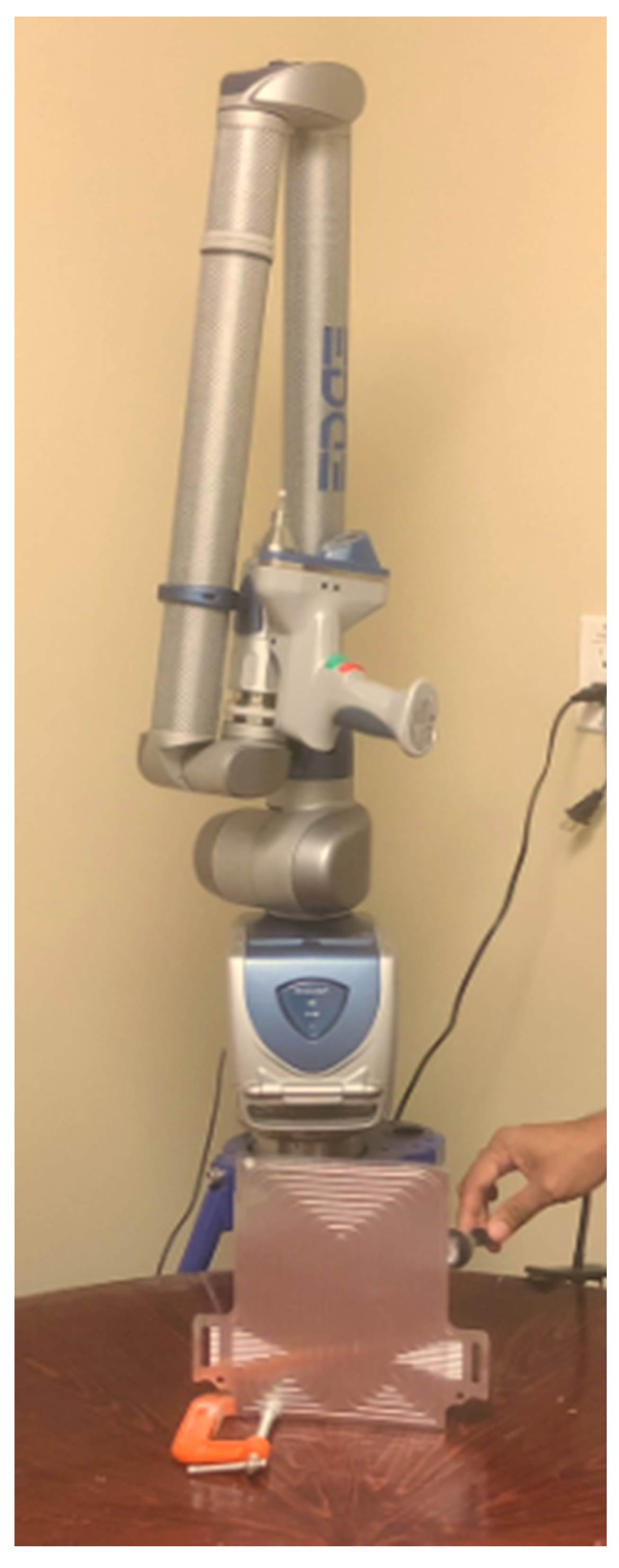
3.1. Reverse Engineering
3.2. Laser Scanning
3.3. Battery Description, Experimental Set-up and Heat Flux Locations
3.4. Test Plan
4. Cold Plate Cooling System Modeling
4.1. Governing Equations
4.2. CFD Modeling Details Using STAR CCM+
4.3. Meshing in DEP MeshWorks 8.0
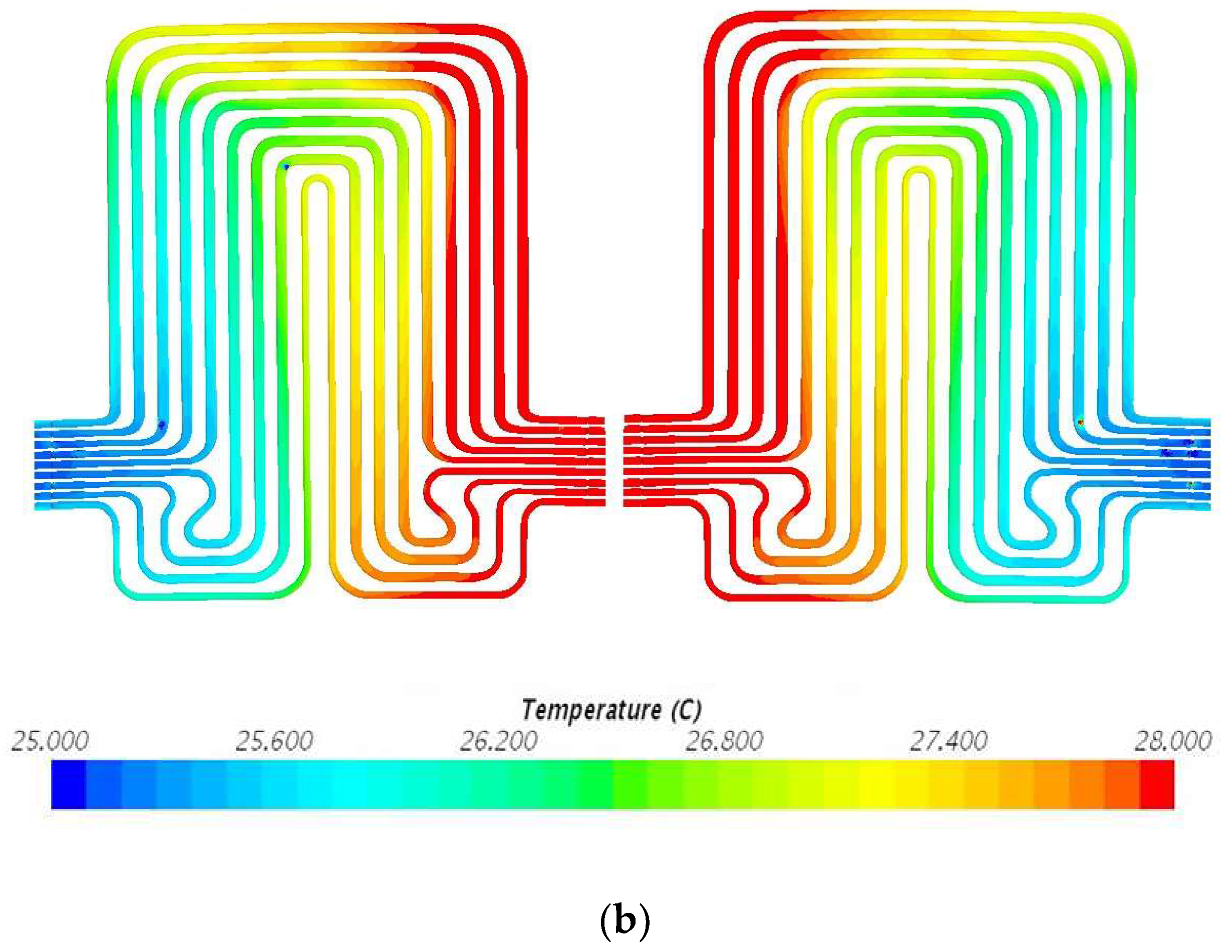
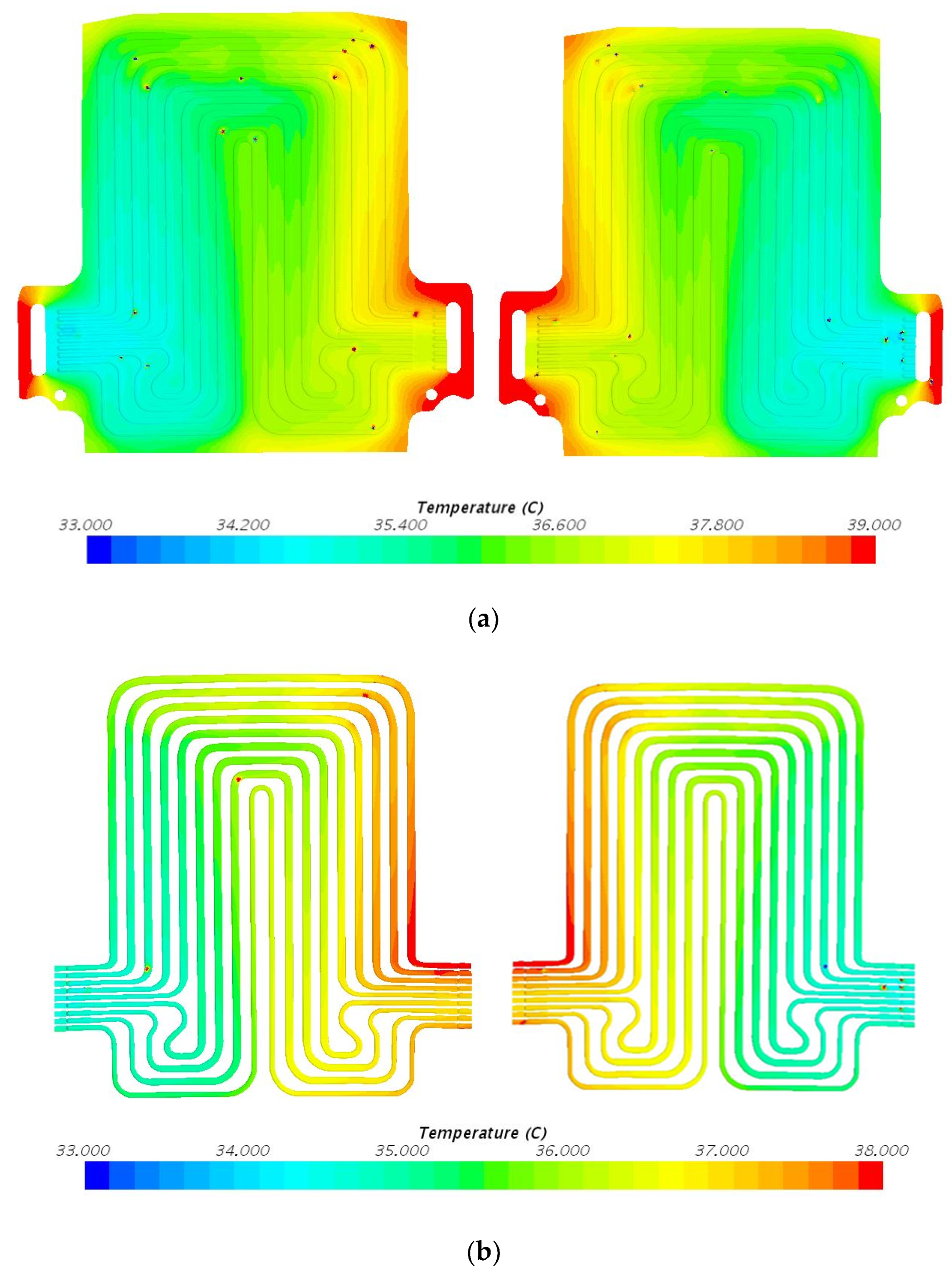
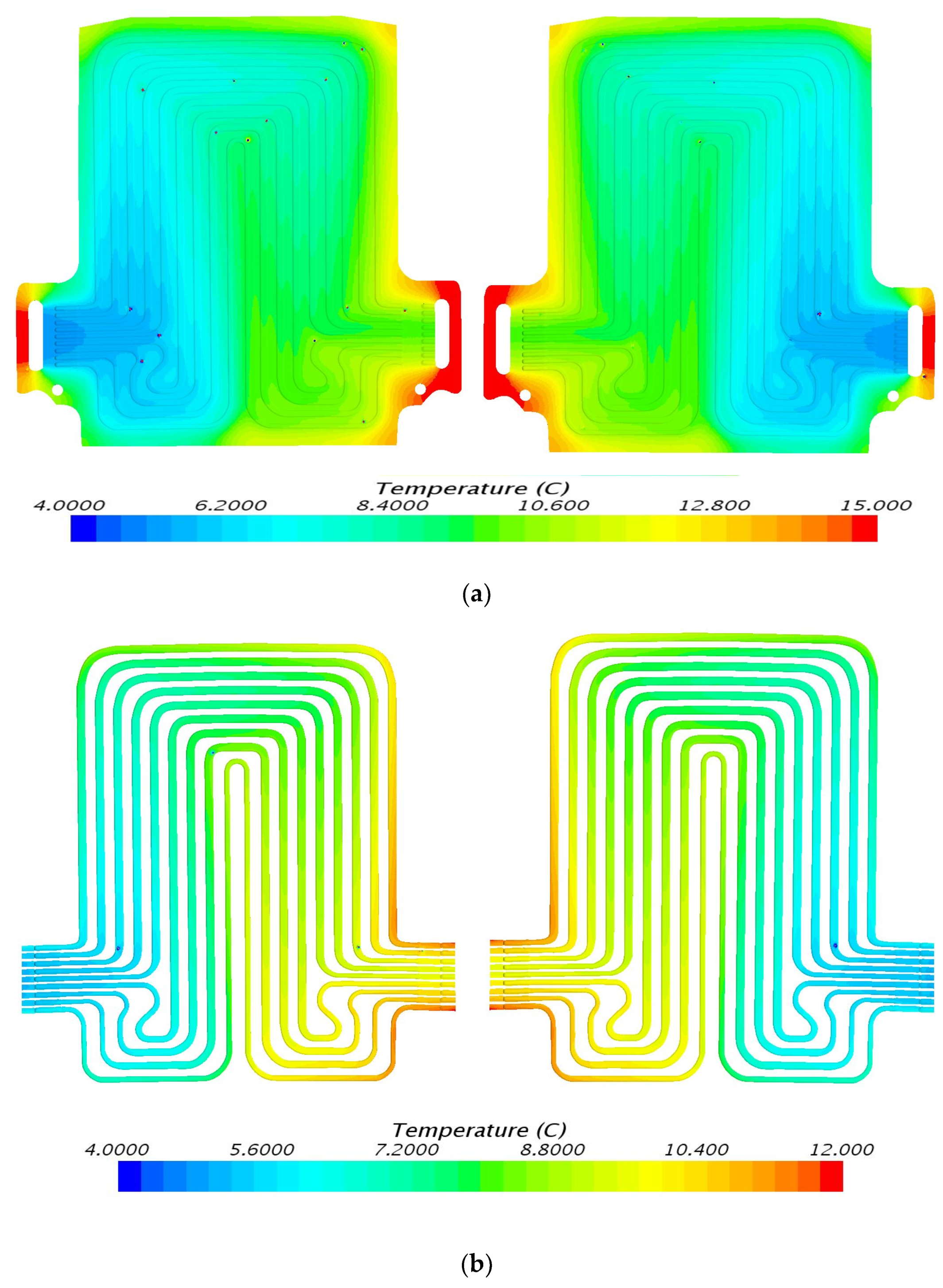
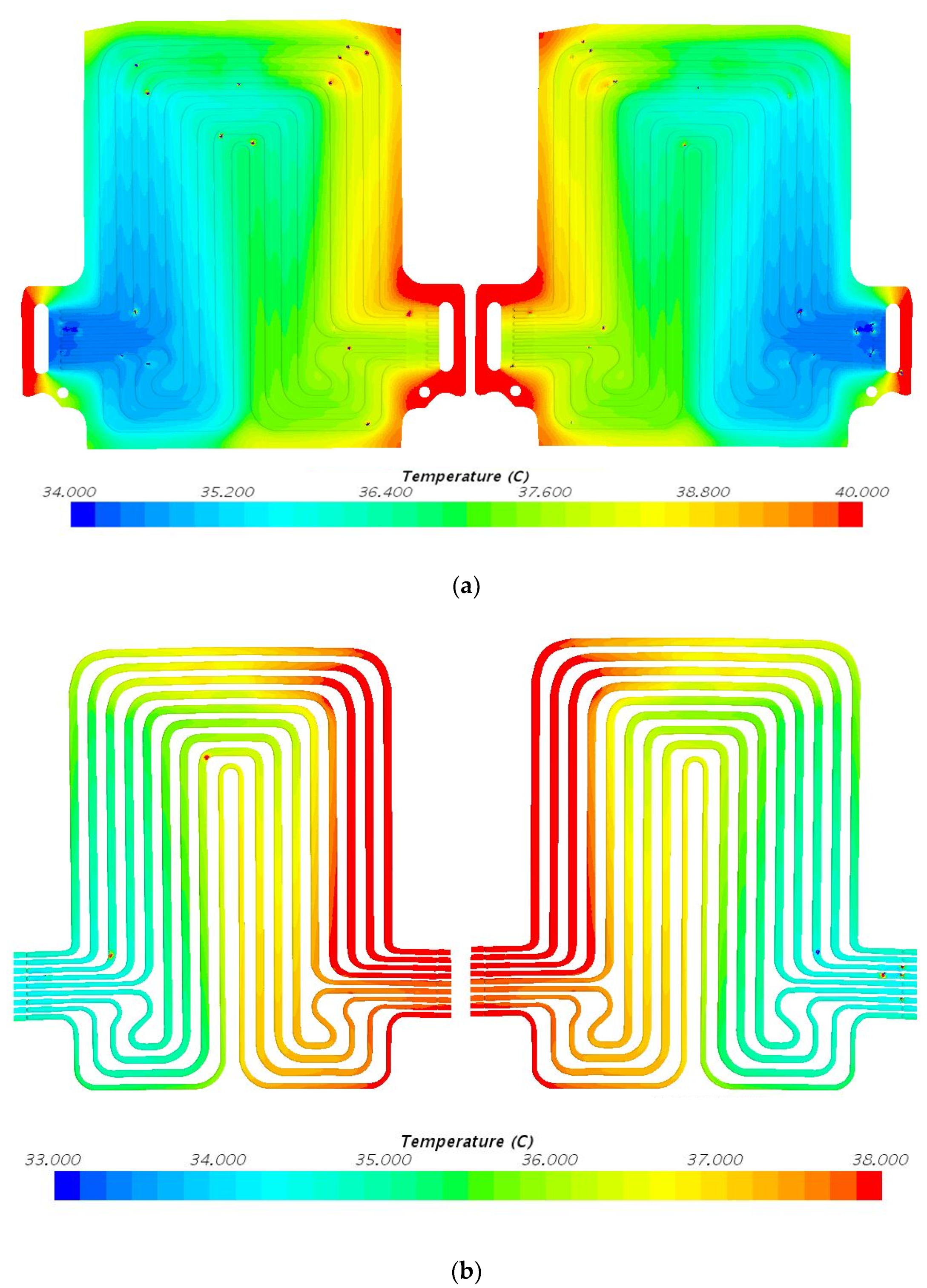
5. Results and Discussion
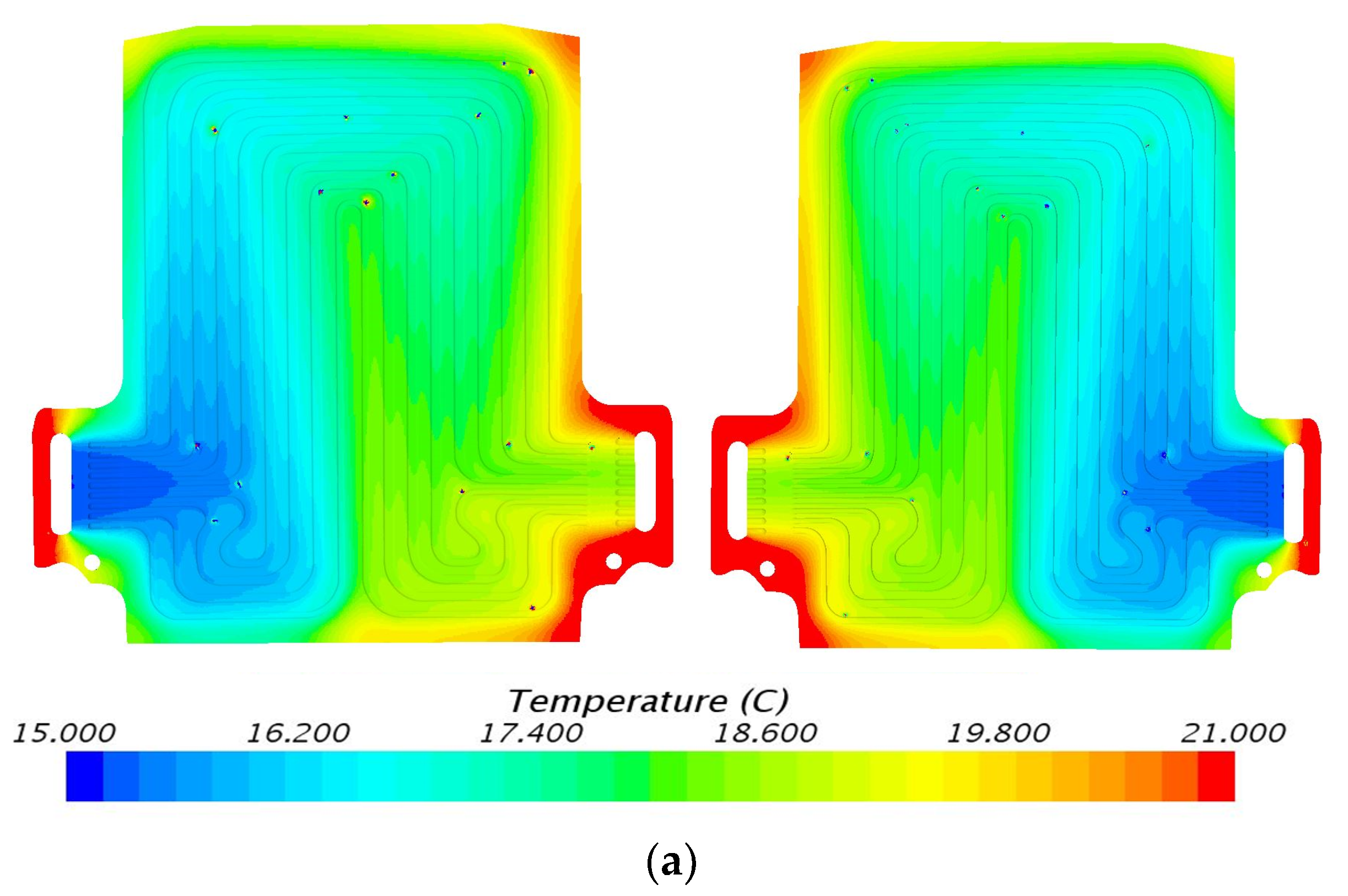
5.1. Temperature Contours at 3C (60 A) Discharge
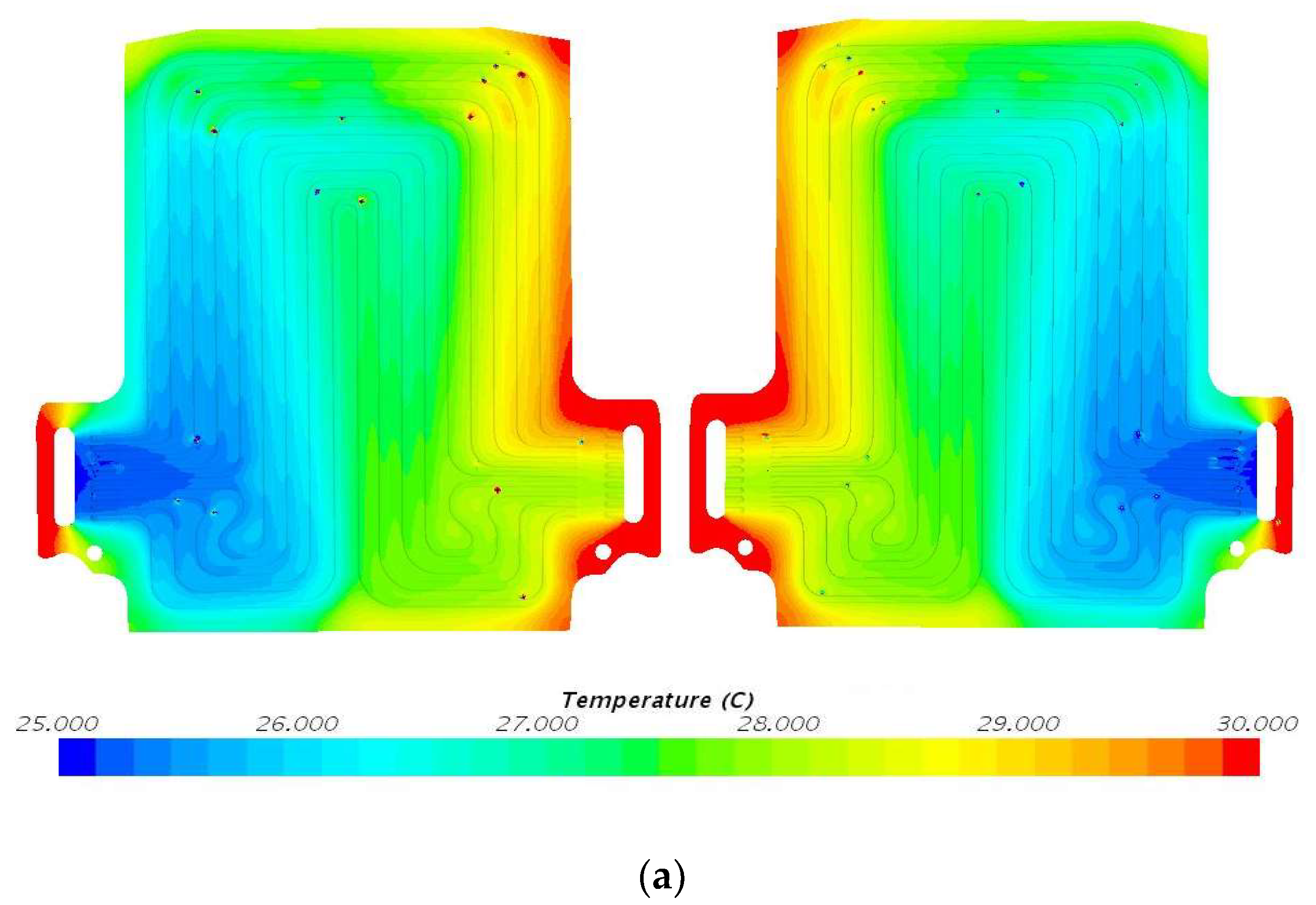
5.2. Temperature Contours at 4C (80 A) Discharge
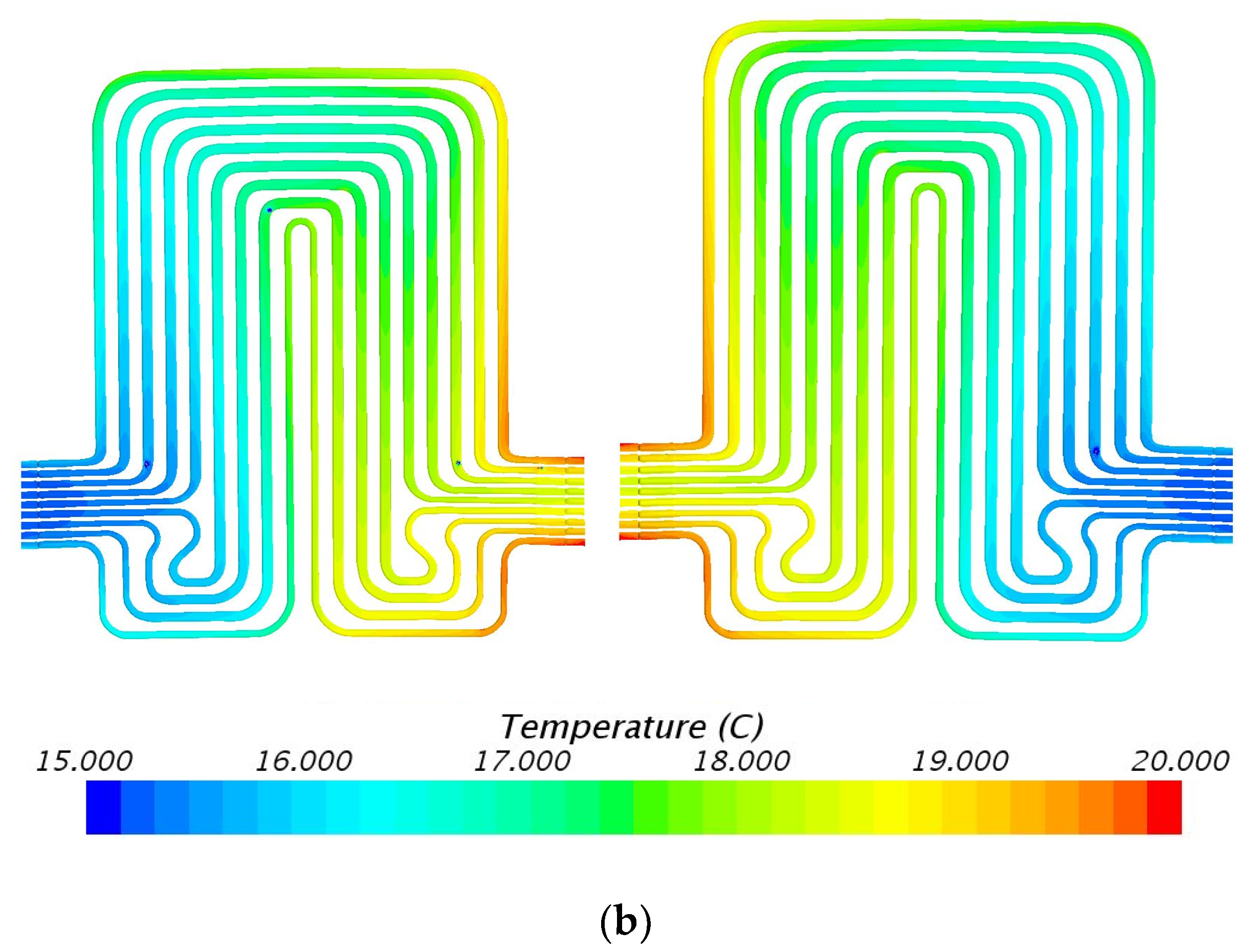
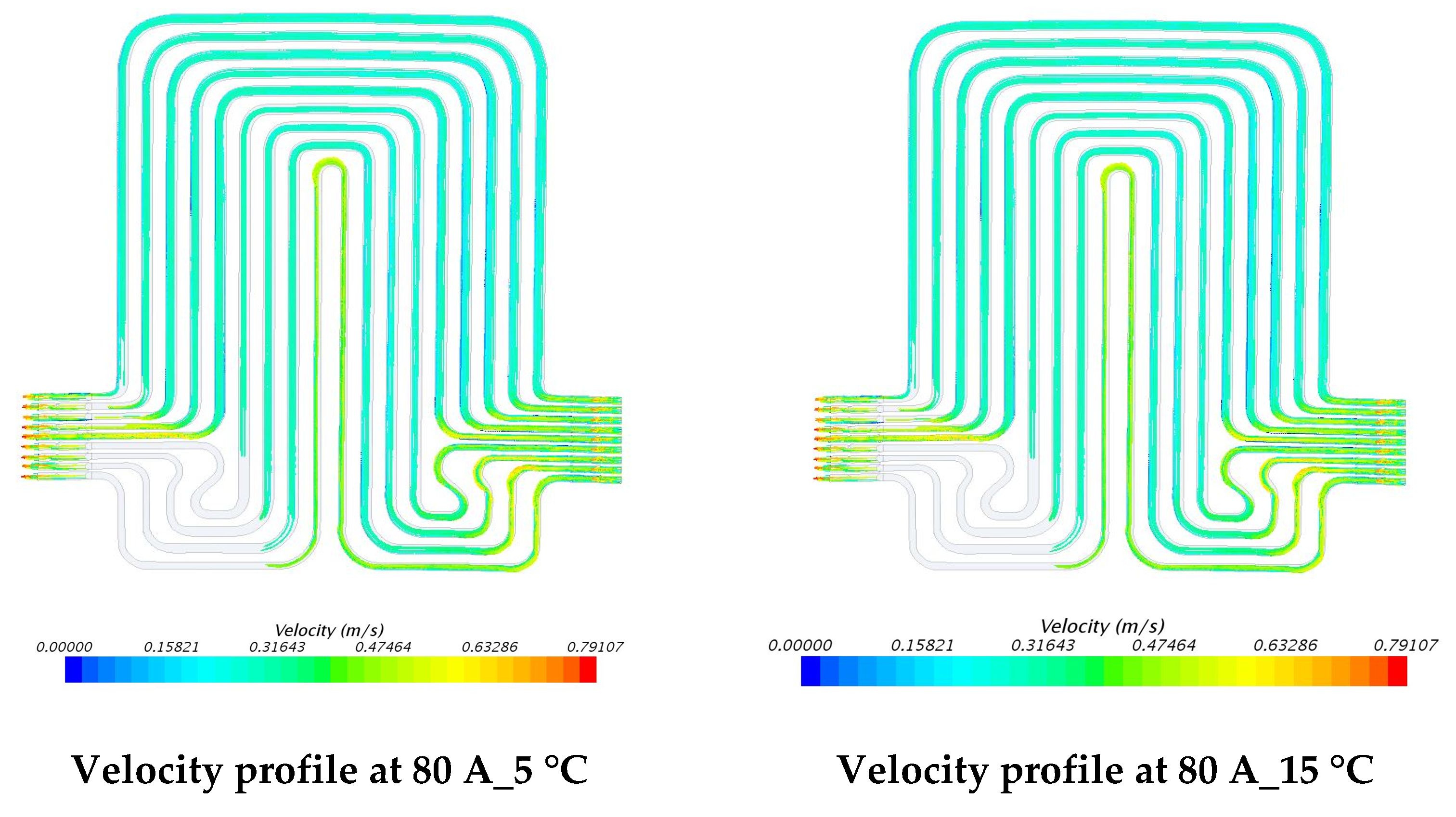
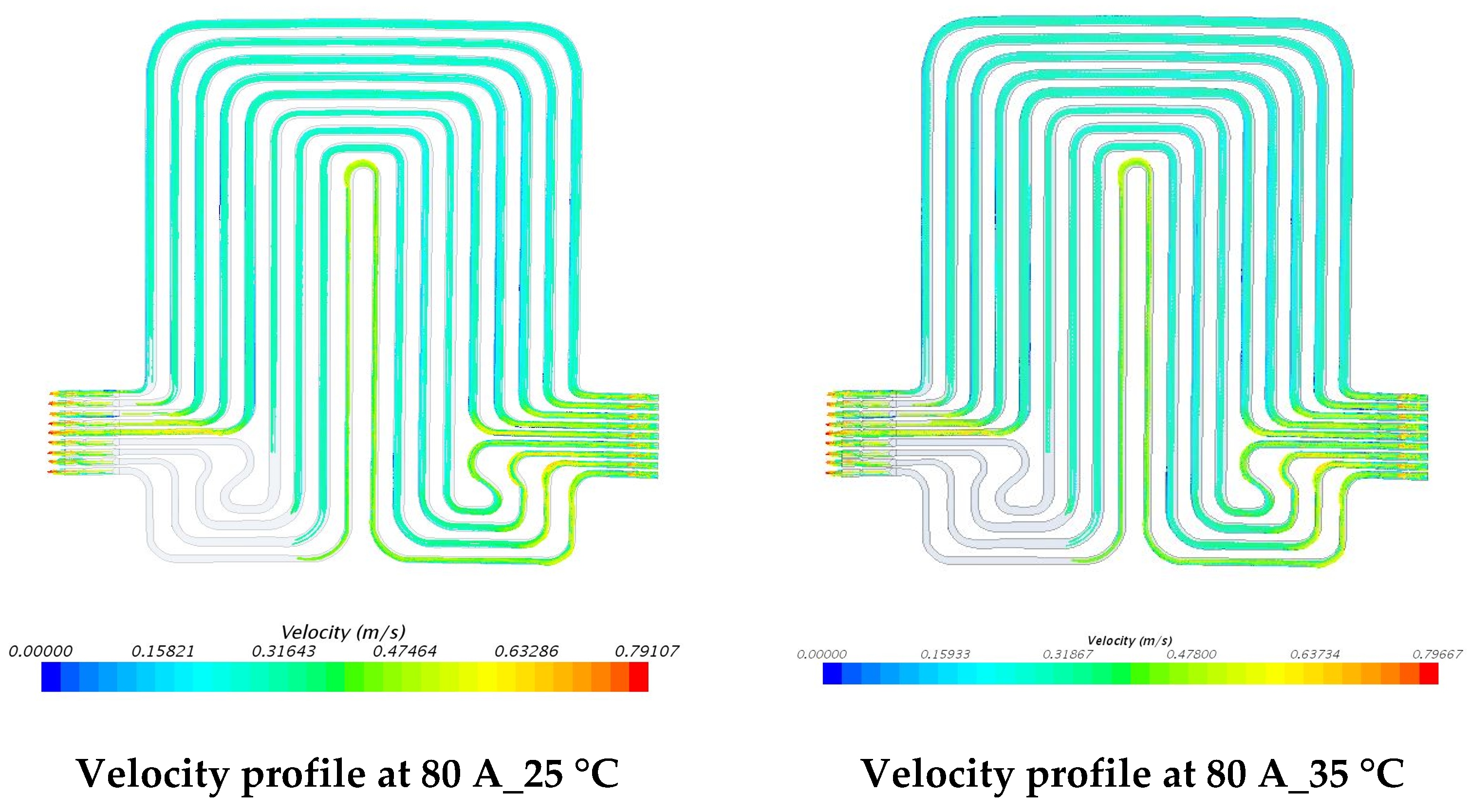
5.3. Velocity Contours at 3C (60 A) and 4C (80 A) Discharge
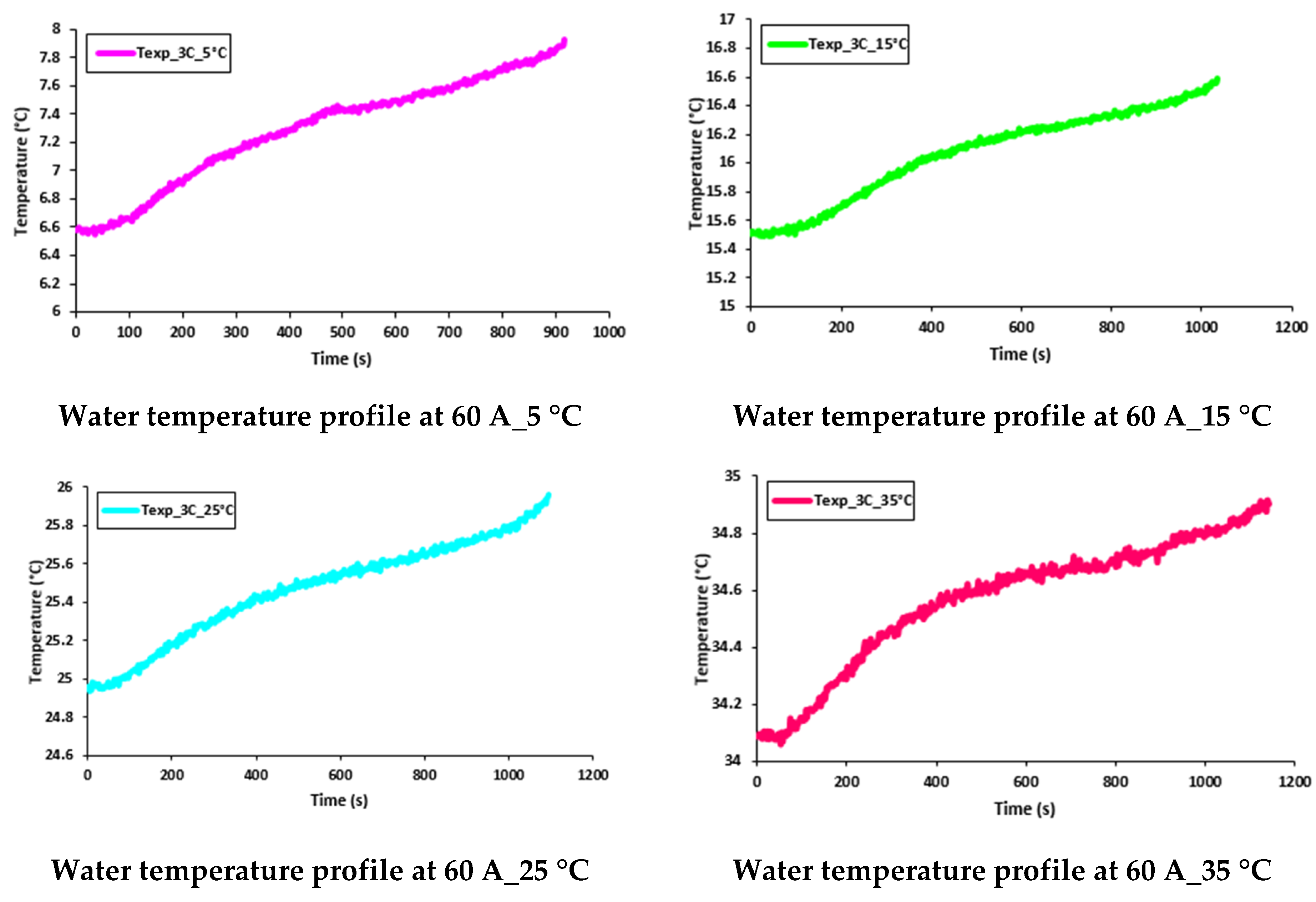
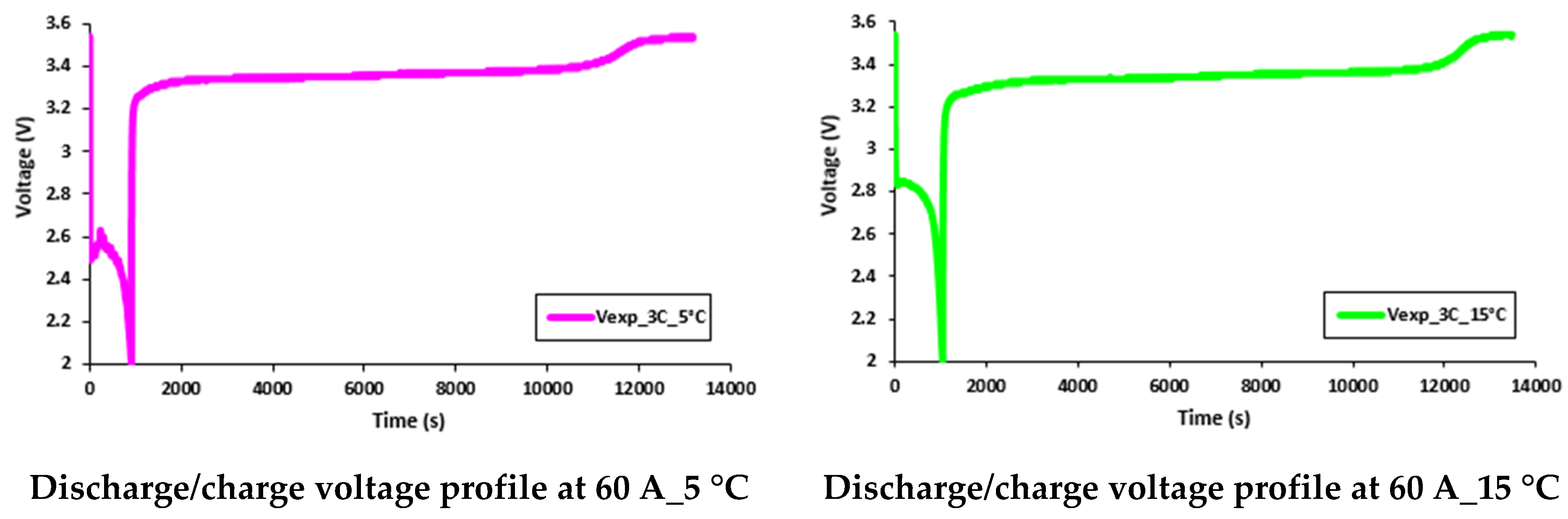
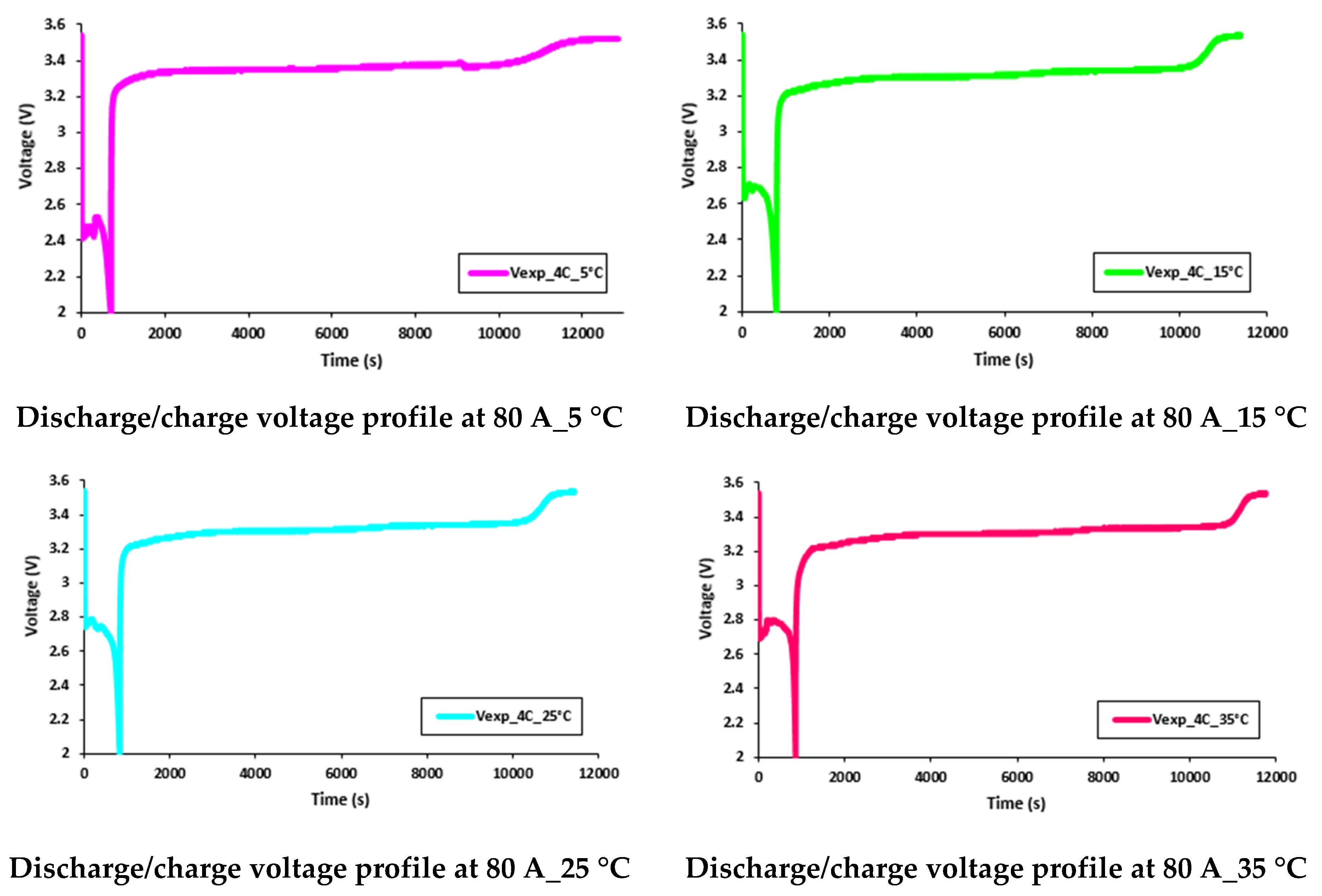
5.4. Transient Temperature Profiles of Water Flow and Voltage Distributions
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| constant | |
| constant for model | |
| C | cell potential or cell voltage (V) |
| I | current (A) |
| turbulence kinetic energy generation due to the mean velocity gradients | |
| turbulence kinetic energy generation due to buoyancy | |
| turbulent kinetic energy (J) | |
| characteristic dimension (m) | |
| pressure (Pa) | |
| Prandtl number | |
| Turbulent Prandtl number | |
| Re | Reynold’s number |
| user-defined source terms | |
| t | time (s) |
| T | temperature (°C or K) |
| V | speed (m/s) |
| average velocity (m/s) | |
| mean fluid velocity (m/s) | |
| y+ | enhanced wall treatment |
| turbulent eddy frequency (1/s) | |
| the contribution of the fluctuating dilatation in compressible turbulence to the overall dissipation rate | |
| Greek Symbols | |
| kinematic fluid viscosity (m2/s) | |
| Reynold’s stress | |
| density (kg/m3) | |
| dynamic fluid viscosity (Ns/m2) | |
| gradient operator | |
| Turbulent Prandtl numbers for and | |
| Subscripts | |
| sim | simulated |
| act | actual |
| Superscripts | |
| ° | degree |
| + | Related to wall treatment |
| Acronyms | |
| BCS | Battery cooling system |
| BEV | Battery electric vehicle |
| BTMS | Battery thermal management system |
| C | Capacity |
| CC | Constant-current |
| CV | Constant-voltage |
| CT | Computed tomography |
| CAD | Computer aided design |
| CFD | Computational fluid dynamics |
| DEP | Detroit Engineered Products, Inc. |
| EV | Electric vehicle |
| ESS | Energy storage system |
| FE | Finite element |
| HEV | Hybrid electric vehicle |
| HFS | Heat flux sensor |
| HPBTMS | Heat pipe battery thermal management system |
| LIB | Lithium-ion battery |
| LiFePO4 | Lithium iron phosphate |
| LED | Light emitting diode |
| MeshWorks | Popular proprietary software package used for CAD and mesh generation |
| MWCNT | multi-walled carbon nanotubes |
| PC | Personal computer |
| PCM | Phase change material |
| PHEV | Plug-In hybrid electric vehicle |
| RE | Reverse engineering |
| RANS | Reynolds-Averaged Navier-Stokes |
| STAR CCM+ | Simulation of Turbulent flow in Arbitrary Regions-Computational Continuum Mechanics + (C++ based) |
| TMS | Thermal management system |
| 1D | One-dimensional |
| 2D | Two-dimensional |
| 3D | Three-dimensional |
| 18650 | IFR 18650 cylindrical valence cells (“I” stands for Li-ion rechargeable, “F” stands for the element “Fe” which is Iron, “R” indicates that the cell shape is round, 18650 means 18 mm diameter and 650 means 65 mm height) |
| 25S2P | 25 series 2 parallel |
References
- Walter, M.; Kravchyk, K.V.; Böfer, C.; Widmer, R.; Kovalenko, M.V. Polypyrenes as High-Performance Cathode Materials for Aluminum Batteries. Adv. Mater. 2018, 30, 1705644. [Google Scholar] [CrossRef] [PubMed]
- Angell, M.; Pan, C.-J.; Rong, Y.; Yuan, C.; Lin, M.-C.; Hwang, B.-J.; Dai, H. High Coulombic efficiency aluminum-ion battery using an AlCl3-urea ionic liquid analog electrolyte. Proc. Natl. Acad. Sci. USA 2017, 114, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.J.; Huang, Q.; Kintner-Meyer, M.C.; Zhang, J.-G.; Reed, D.M.; Sprenkle, V.L.; Viswanathan, V.V.; Choi, D. Lifecycle comparison of selected Li-ion battery chemistries under grid and electric vehicle duty cycle combinations. J. Power Sources 2018, 380, 185–193. [Google Scholar] [CrossRef]
- Panchal, S.; Mathewson, S.; Fraser, R.; Culham, R.; Fowler, M. Measurement of Temperature Gradient (dT/dy) and Temperature Response (dT/dt) of a Prismatic Lithium-Ion Pouch Cell with LiFePO4 Cathode Material. SAE Tech. Pap. Ser. 2017, 1. [Google Scholar]
- Panchal, S.; Rashid, M.; Long, F.; Mathew, M.; Fraser, R.; Fowler, M. Degradation Testing and Modeling of 200 Ah LiFePO4 Battery. SAE Tech. Pap. Ser. 2018. [Google Scholar]
- Panchal, S. Impact of Vehicle Charge and Discharge Cycles on the Thermal Characteristics of Lithium-ion Batteries; University of Waterloo: Waterloo, ON, Canada, 2014. [Google Scholar]
- Panchal, S. Experimental Investigation and Modeling of Lithium-ion Battery Cells and Packs for Electric Vehicles; University of Ontario Institute of Technology: Oshawa, ON, Canada, 2016. [Google Scholar]
- Al-Zareer, M.; Dincer, I.; Rosen, M.A. Heat and mass transfer modeling and assessment of a new battery cooling system. Int. J. Heat Mass Transf. 2018, 126, 765–778. [Google Scholar] [CrossRef]
- Waldmann, T.; Wilka, M.; Kasper, M.; Fleischhammer, M.; Wohlfahrt-Mehrens, M. Temperature dependent ageing mechanisms in Lithium-ion batteries—A Post-Mortem study. J. Power Sources 2014, 262, 129–135. [Google Scholar] [CrossRef]
- Tran, M.-K.; Fowler, M. A Review of Lithium-Ion Battery Fault Diagnostic Algorithms: Current Progress and Future Challenges. Algorithms 2020, 13, 62. [Google Scholar] [CrossRef]
- Panchal, S.; Mathewson, S.; Fraser, R.; Culham, R.; Fowler, M. Experimental Measurements of Thermal Characteristics of LiFePO4 Battery. SAE Tech. Pap. Ser. 2015. [Google Scholar]
- Panchal, S.; Mathewson, S.; Fraser, R.; Culham, R.; Fowler, M. Thermal Management of Lithium-Ion Pouch Cell with Indirect Liquid Cooling using Dual Cold Plates Approach. SAE Int. J. Altern. Powertrains 2015, 4, 293–307. [Google Scholar] [CrossRef]
- Ramadass, P.; Haran, B.; White, R.; Popov, B. Capacity fade of Sony 18650 cells cycled at elevated temperatures: Part II. Capacity fade analysis. J. Power Sources 2002, 112, 614–620. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fowler, M.; Fraser, R. Uneven temperature and voltage distributions due to rapid discharge rates and different boundary conditions for series-connected LiFePO 4 batteries. Int. Commun. Heat Mass Transf. 2017, 81, 210–217. [Google Scholar] [CrossRef]
- Uddin, A.I.; Ku, J. Design and Simulation of Lithium-Ion Battery Thermal Management System for Mild Hybrid Vehicle Application. SAE Tech. Pap. Ser. 2015. [Google Scholar]
- Wang, C.H.; Lin, T.; Huang, J.T.; Rao, Z.H. Temperature response of a high power lithium-ion battery subjected to high current discharge. Mater. Res. Innov. 2015, 19, S2–S156. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Experimental and theoretical investigation of temperature distributions in a prismatic lithium-ion battery. Int. J. Therm. Sci. 2016, 99, 204–212. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Thermal modeling and validation of temperature distributions in a prismatic lithium-ion battery at different discharge rates and varying boundary conditions. Appl. Therm. Eng. 2016, 96, 190–199. [Google Scholar] [CrossRef]
- Patil, M.S.; Panchal, S.; Kim, N.; Lee, M.-Y. Cooling Performance Characteristics of 20 Ah Lithium-Ion Pouch Cell with Cold Plates along Both Surfaces. Energies 2018, 11, 2550. [Google Scholar] [CrossRef]
- He, F.; Ma, L. Thermal Management in Hybrid Power Systems Using Cylindrical and Prismatic Battery Cells. Heat Transf. Eng. 2015, 37, 581–590. [Google Scholar] [CrossRef]
- Giuliano, M.R.; Prasad, A.K.; Advani, S. Experimental study of an air-cooled thermal management system for high capacity lithium–titanate batteries. J. Power Sources 2012, 216, 345–352. [Google Scholar] [CrossRef]
- Jin, L.W.; Lee, P.; Kong, X.; Fan, Y.; Chou, S. Ultra-thin minichannel LCP for EV battery thermal management. Appl. Energy 2014, 113, 1786–1794. [Google Scholar] [CrossRef]
- Rao, Z.; Wang, S. A review of power battery thermal energy management. Renew. Sustain. Energy Rev. 2011, 15, 4554–4571. [Google Scholar] [CrossRef]
- Lemmon, E.W.; Jacobsen, R.T. Viscosity and Thermal Conductivity Equations for Nitrogen, Oxygen, Argon, and Air. Int. J. Thermophys. 2004, 25, 21–69. [Google Scholar] [CrossRef]
- Park, H. A design of air flow configuration for cooling lithium ion battery in hybrid electric vehicles. J. Power Sources 2013, 239, 30–36. [Google Scholar] [CrossRef]
- Fan, L.-W.; Khodadadi, J.; Pesaran, A. A parametric study on thermal management of an air-cooled lithium-ion battery module for plug-in hybrid electric vehicles. J. Power Sources 2013, 238, 301–312. [Google Scholar] [CrossRef]
- Kelly, K.J.; Mihalic, M.; Zolot, M. Battery usage and thermal performance of the Toyota Prius and Honda Insight during chassis dynamometer testing. In Proceedings of the Seventeenth Annual Battery Conference on Applications and Advances (Cat. No.02TH8576), Long Beach, CA, USA, 18 January 2002; Institute of Electrical and Electronics Engineers (IEEE): Piscataway, NJ, USA, 2002; pp. 247–252. [Google Scholar]
- Berdichevsky, G.; Kelty, K.; Straubel, J.B.; Toomre, E. The Tesla Roadster Battery System. 2007. Available online: http://www.batterypoweronline.com (accessed on 21 July 2019).
- What Is the Best Electric Vehicle Battery Cooling System? 2017. Available online: https://avidtp.com/what-is-the-best-cooling-system-for-electric-vehicle-battery-packs/ (accessed on 29 June 2019).
- Samba, A.; Omar, N.; Gualous, H.; Firouz, Y.; Bossche, P.V.D.; Van Mierlo, J.; Boubekeur, T.I. Development of an Advanced Two-Dimensional Thermal Model for Large size Lithium-ion Pouch Cells. Electrochim. Acta 2014, 117, 246–254. [Google Scholar] [CrossRef]
- Li, G.; Li, S.-P. Physics-Based CFD Simulation of Lithium-Ion Battery under the FUDS Driving Cycle. ECS Trans. 2015, 64, 1–14. [Google Scholar] [CrossRef]
- Vyroubal, P.; Kazda, T.; Maxa, J.; Vondrák, J. Analysis of Temperature Field in Lithium Ion Battery by Discharging. ECS Trans. 2015, 70, 269–273. [Google Scholar] [CrossRef]
- Yeow, K.; Teng, H.; Thelliez, M.; Tan, E. Thermal Analysis of a Li-ion Battery System with Indirect Liquid Cooling Using Finite Element Analysis Approach. SAE Int. J. Altern. Powertrains 2012, 1, 65–78. [Google Scholar] [CrossRef]
- Al Hallaj, S.; Selman, J.; King, S.W.; Kern, R.S.; Benjamin, M.C.; Barnak, J.P.; Nemanich, R.J.; Davis, R.F. A Novel Thermal Management System for Electric Vehicle Batteries Using Phase-Change Material. J. Electrochem. Soc. 2000, 147, 3231. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, T.; Jiang, S.B.; Xu, H.G.; Zhang, Y.N. Modelling and Simulation of Pouch Lithium-Ion Battery Thermal Management Using Cold Plate. Int. J. Simul. Model. 2018, 17, 498–511. [Google Scholar] [CrossRef]
- Omkar, D.; Vijaykumar, P. Development of Phase Change Material/Cooling Plate Coupled Battery Thermal Management System Using CFD. Int. J. Res. Eng. Appl. Manag. 2018, 4, 1–6. [Google Scholar]
- Chen, D.; Jiang, J.; Kim, G.-H.; Yang, C.; Pesaran, A. Comparison of different cooling methods for lithium ion battery cells. Appl. Therm. Eng. 2016, 94, 846–854. [Google Scholar] [CrossRef]
- Lu, Z.; Meng, X.; Wei, L.; Hu, W.; Zhang, L.; Jin, L.W. Thermal Management of Densely-packed EV Battery with Forced Air Cooling Strategies. Energy Procedia 2016, 88, 682–688. [Google Scholar] [CrossRef]
- Qian, Z.; Li, Y.; Rao, Z. Thermal performance of lithium-ion battery thermal management system by using mini-channel cooling. Energy Convers. Manag. 2016, 126, 622–631. [Google Scholar] [CrossRef]
- Jarrett, A.; Kim, I.Y. Influence of operating conditions on the optimum design of electric vehicle battery cooling plates. J. Power Sources 2014, 245, 644–655. [Google Scholar] [CrossRef]
- Zou, D.; Ma, X.; Liu, X.; Zheng, P.; Hu, Y. Thermal performance enhancement of composite phase change materials (PCM) using graphene and carbon nanotubes as additives for the potential application in lithium-ion power battery. Int. J. Heat Mass Transf. 2018, 120, 33–41. [Google Scholar] [CrossRef]
- Greco, A.; Cao, D.; Jiang, X.; Yang, H. A theoretical and computational study of lithium-ion battery thermal management for electric vehicles using heat pipes. J. Power Sources 2014, 257, 344–355. [Google Scholar] [CrossRef]
- Liang, J.; Gan, Y.; Li, Y. Investigation on the thermal performance of a battery thermal management system using heat pipe under different ambient temperatures. Energy Convers. Manag. 2018, 155, 1–9. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, G.; Li, X.; Huang, J.; Wang, Z.; Lv, Y.; Meng, L.; Situ, W.; Rao, M. Experimental examination of large capacity liFePO4 battery pack at high temperature and rapid discharge using novel liquid cooling strategy. Int. J. Energy Res. 2017, 42, 1172–1182. [Google Scholar] [CrossRef]
- Panchal, S.; Khasow, R.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Numerical modeling and experimental investigation of a prismatic battery subjected to water cooling. Numer. Heat Transf. Part A Appl. 2017, 93, 1–12. [Google Scholar] [CrossRef]
- Panchal, S.; Khasow, R.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Thermal design and simulation of mini-channel cold plate for water cooled large sized prismatic lithium-ion battery. Appl. Therm. Eng. 2017, 122, 80–90. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Design and simulation of a lithium-ion battery at large C-rates and varying boundary conditions through heat flux distributions. Measurement 2018, 116, 382–390. [Google Scholar] [CrossRef]











| Specification | Value | Unit |
|---|---|---|
| Material for electrolyte | Carbonate based | - |
| Material for anode | Graphite | - |
| Material for cathode | LiFePO4 | - |
| Voltage (nominal) | 3.3 | V |
| Dimensions | 7.25 (t) × 160 (w) × 227 (h) | mm |
| Capacity of the cell (nominal) | 20 | Ah |
| Discharge power | 1200 | W |
| Energy (nominal) | 65 | Wh |
| Specific energy | 131 | Wh/kg |
| Energy density | 247 | Wh/L |
| Operating temperature | −30 to 55 | °C |
| Mass of the cell | 496 | g |
| Specific power | 2400 | W/kg |
| Maximum discharge | 300 | A |
| Internal resistance | 0.5 | mΩ |
| Volume | 0.263 | L |
| Storage temperature | −40 to 60 | °C |
| Number of cycles | Min. 300, approx. 2000 | Cycles |
| Maximum charge | 300 | A |
| Working Fluid | Operating Temperature (°C) | Charge Current | Discharge Current |
|---|---|---|---|
| Water | 5 | 20 A | 60 A, 80 A |
| 15 | 20 A | 60 A, 80 A | |
| 25 | 20 A | 60 A, 80 A | |
| 35 | 20 A | 60 A, 80 A |
| Working Fluid | Working Temperature (°C) | Difference between Experimental and Simulated Values | Water Inlet and Outlet Temperature (°C) | |||
|---|---|---|---|---|---|---|
| 60 A | 80 A | |||||
| Inlet | Outlet | Inlet | Outlet | |||
| Water | 5 | Experimental (°C) | 5.7391 | 7.9307 | 5.1435 | 7.7029 |
| Simulated (°C) | 7.93 | 9.35 | 7.70 | 10.25 | ||
| Difference (%) | 38.17 | 17.90 | 49.70 | 33.07 | ||
| 15 | Experimental (°C) | 15.1377 | 16.7696 | 15.0906 | 16.9376 | |
| Simulated (°C) | 16.76 | 18.84 | 16.93 | 19.75 | ||
| Difference (%) | 10.72 | 12.35 | 12.55 | 16.60 | ||
| 25 | Experimental (°C) | 25.0992 | 25.9614 | 25.0984 | 26.3445 | |
| Simulated (°C) | 25.96 | 28.47 | 26.34 | 29.3 | ||
| Difference (%) | 3.43 | 9.66 | 4.95 | 11.22 | ||
| 35 | Experimental (°C) | 34.4912 | 34.9092 | 34.2555 | 35.2637 | |
| Simulated (°C) | 34.90 | 37.29 | 35.26 | 38.12 | ||
| Difference (%) | 1.19 | 6.82 | 2.93 | 8.10 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panchal, S.; Gudlanarva, K.; Tran, M.-K.; Fraser, R.; Fowler, M. High Reynold’s Number Turbulent Model for Micro-Channel Cold Plate Using Reverse Engineering Approach for Water-Cooled Battery in Electric Vehicles. Energies 2020, 13, 1638. https://doi.org/10.3390/en13071638
Panchal S, Gudlanarva K, Tran M-K, Fraser R, Fowler M. High Reynold’s Number Turbulent Model for Micro-Channel Cold Plate Using Reverse Engineering Approach for Water-Cooled Battery in Electric Vehicles. Energies. 2020; 13(7):1638. https://doi.org/10.3390/en13071638
Chicago/Turabian StylePanchal, Satyam, Krishna Gudlanarva, Manh-Kien Tran, Roydon Fraser, and Michael Fowler. 2020. "High Reynold’s Number Turbulent Model for Micro-Channel Cold Plate Using Reverse Engineering Approach for Water-Cooled Battery in Electric Vehicles" Energies 13, no. 7: 1638. https://doi.org/10.3390/en13071638
APA StylePanchal, S., Gudlanarva, K., Tran, M.-K., Fraser, R., & Fowler, M. (2020). High Reynold’s Number Turbulent Model for Micro-Channel Cold Plate Using Reverse Engineering Approach for Water-Cooled Battery in Electric Vehicles. Energies, 13(7), 1638. https://doi.org/10.3390/en13071638









