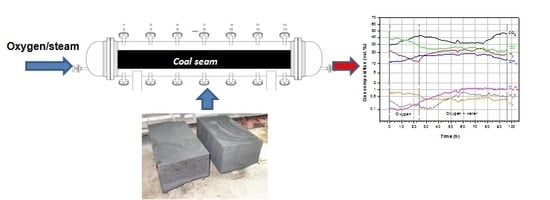
Large-scale Experimental Investigations to Evaluate the Feasibility of Producing Methane-Rich Gas (SNG) through Underground Coal Gasification Process. Effect of Coal Rank and Gasification Pressure
Abstract
1. Introduction
2. Materials and Methods
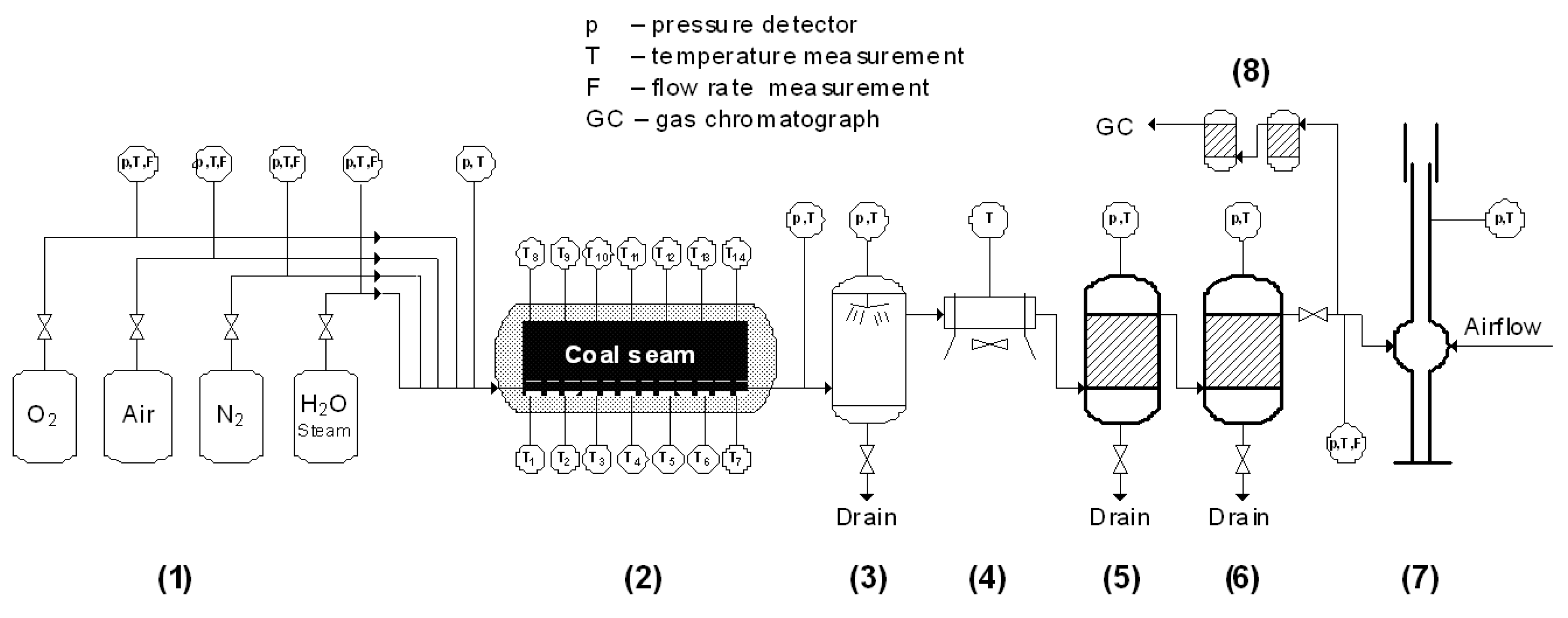
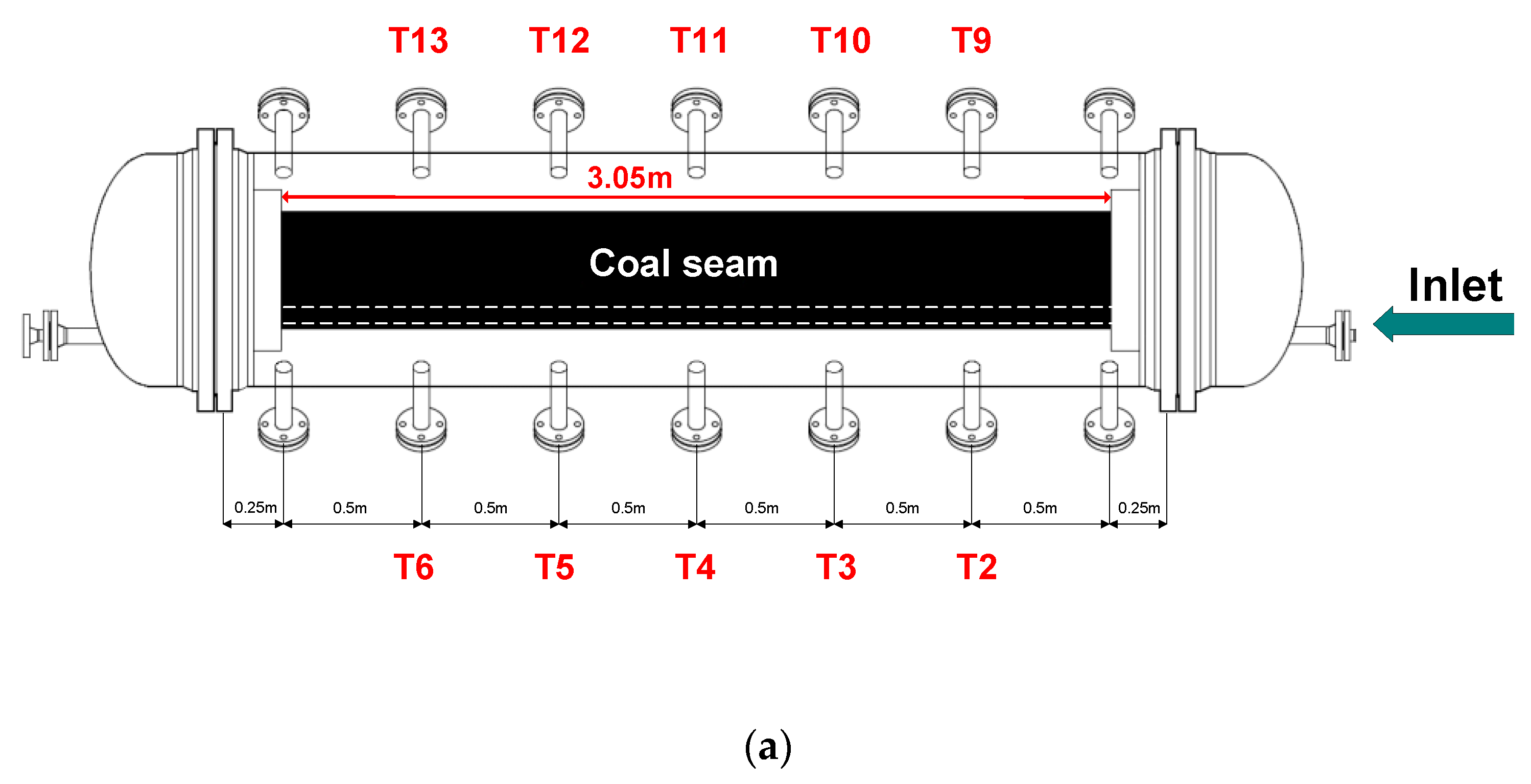
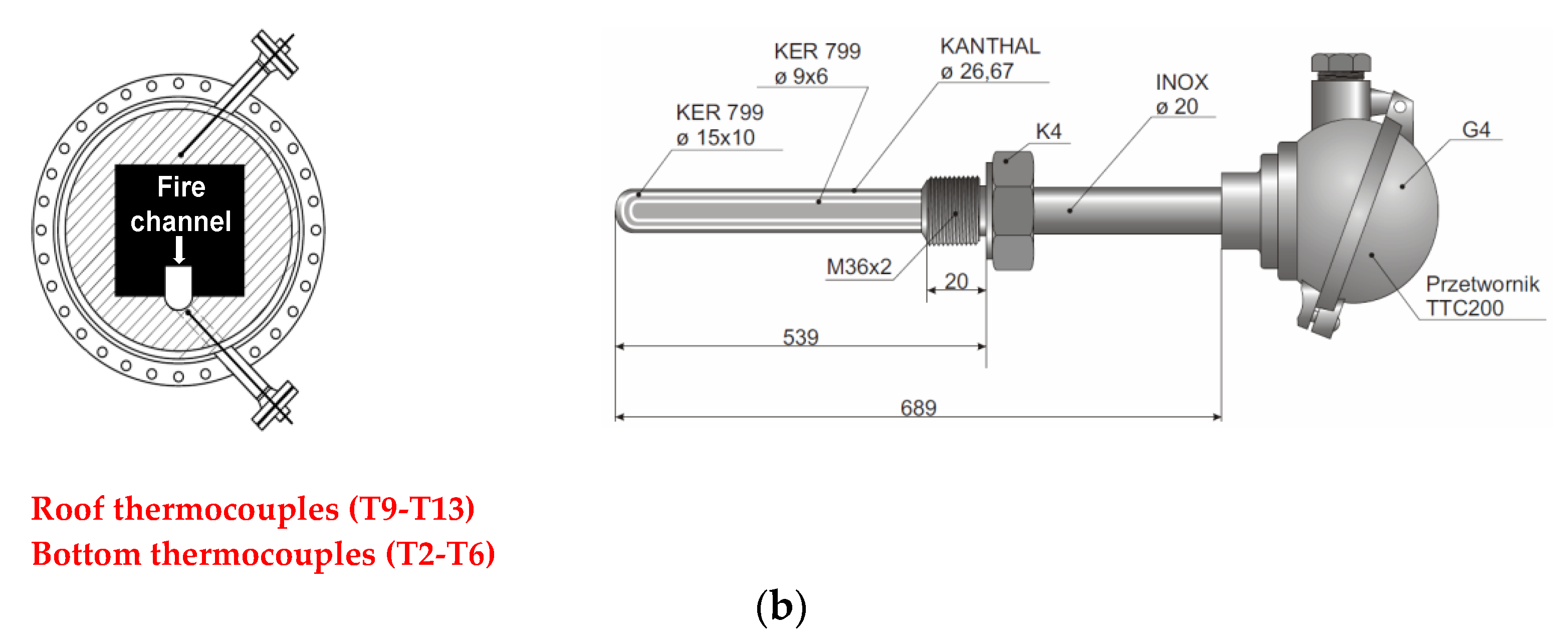
2.1. Description of the UCG Test Stand
2.2. Coal Samples and Preparation of the Artificial Seam
2.3. Experimental Campaign and Test Procedure
2.4. Calculations
Gas calorific value
- Gas yield
- 2.
- Average gas production rate
- 3.
- Energy in process gas
- 4.
- Average reactor power.
- 5.
- Gasification rate
- 6.
- Energy efficiency
3. Results and Discussion
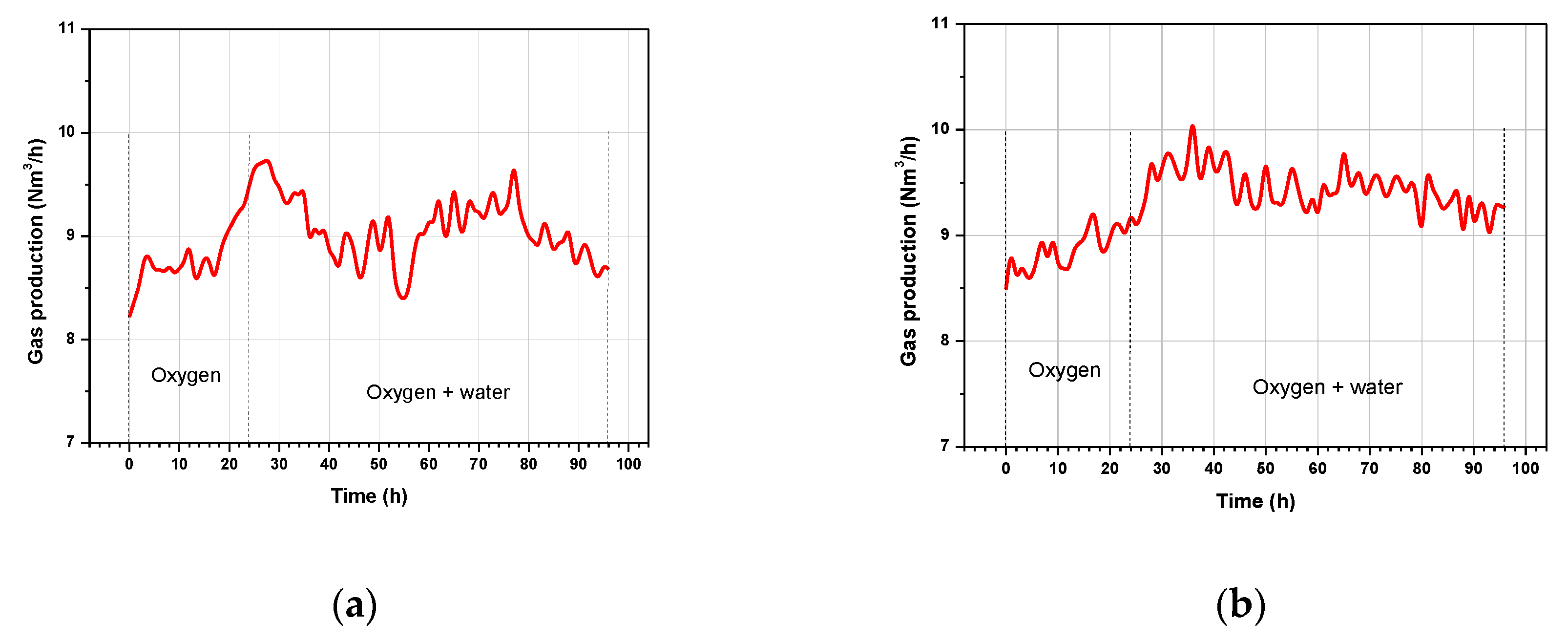
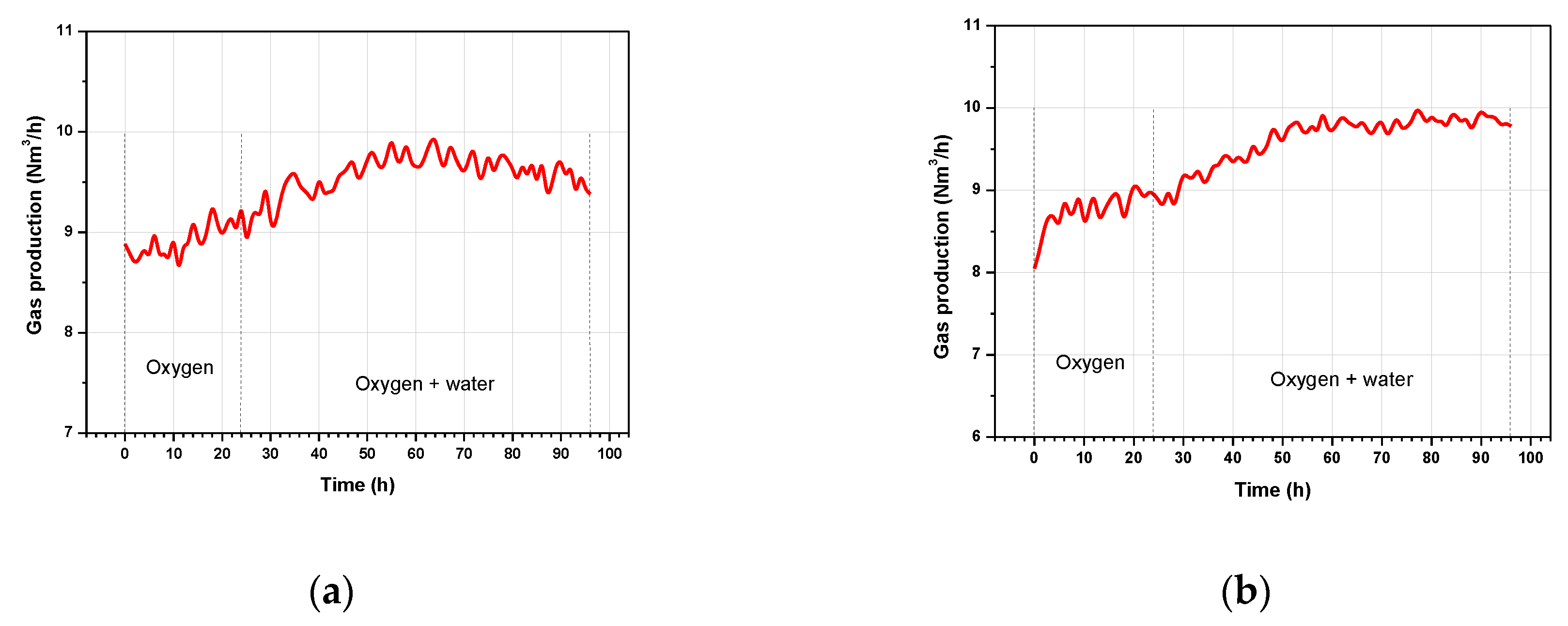
3.1. Gas Production Rates
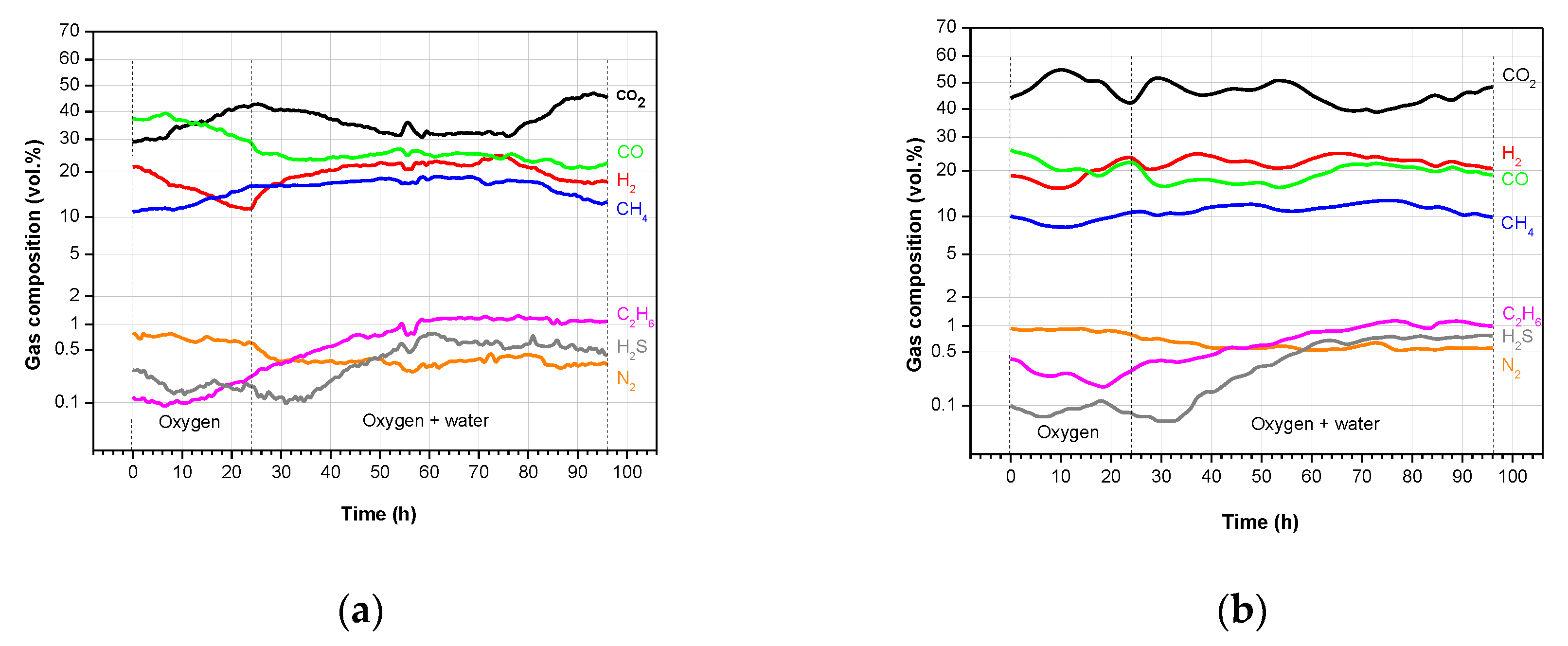
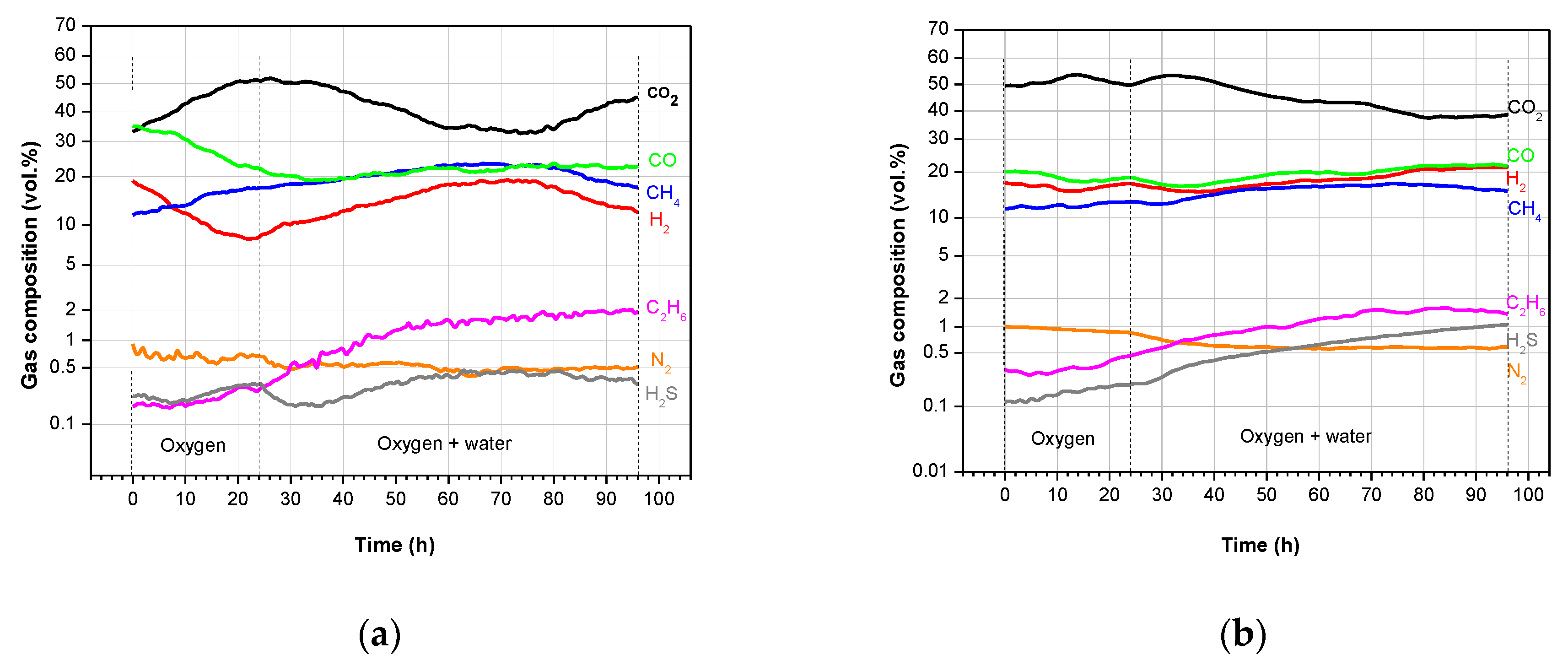
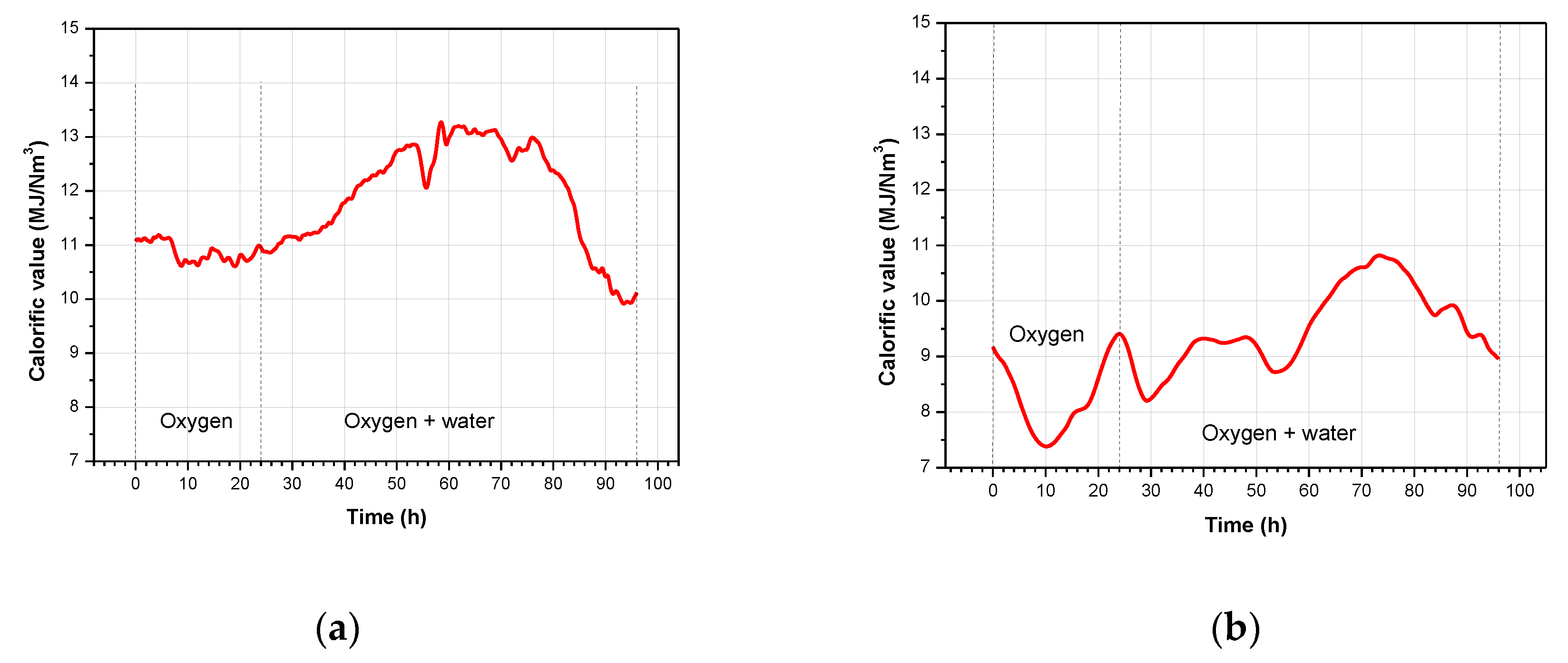
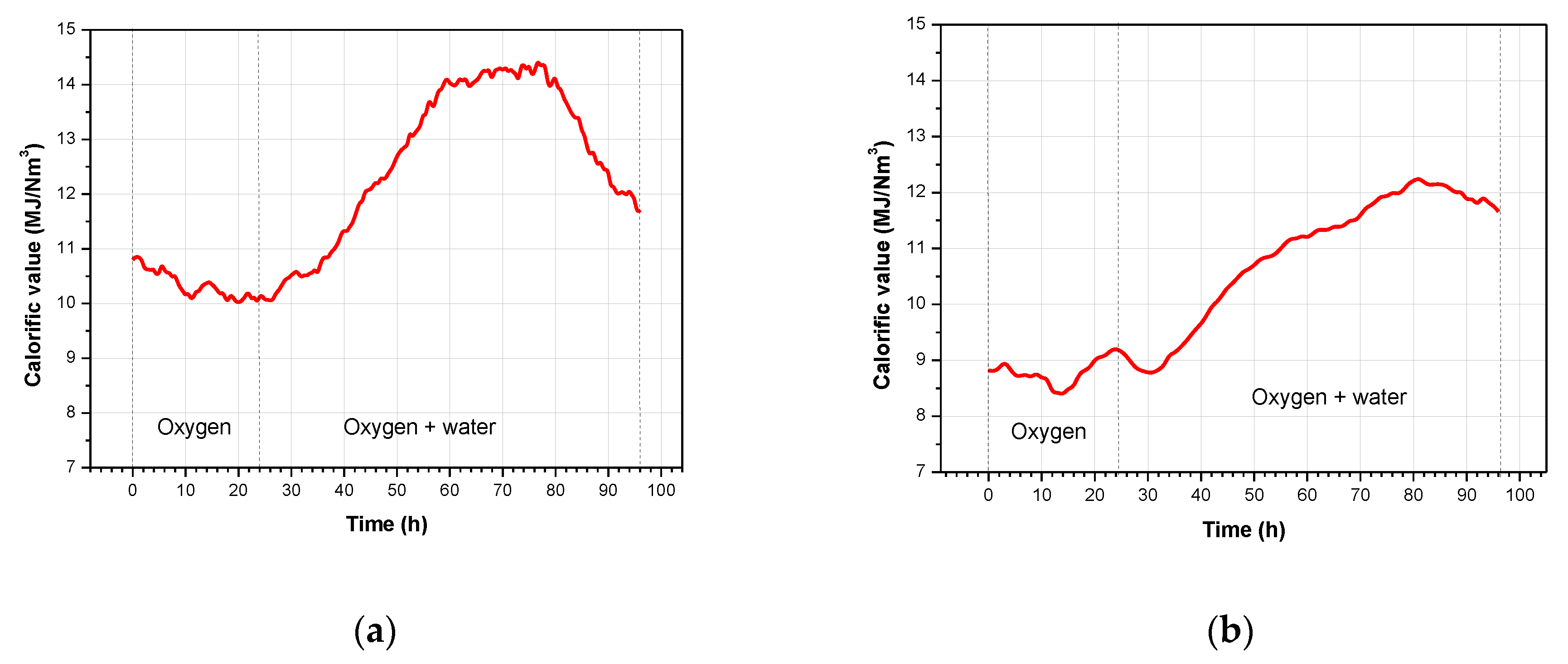
3.2. Product Gas Composition and Gas Calorific Value
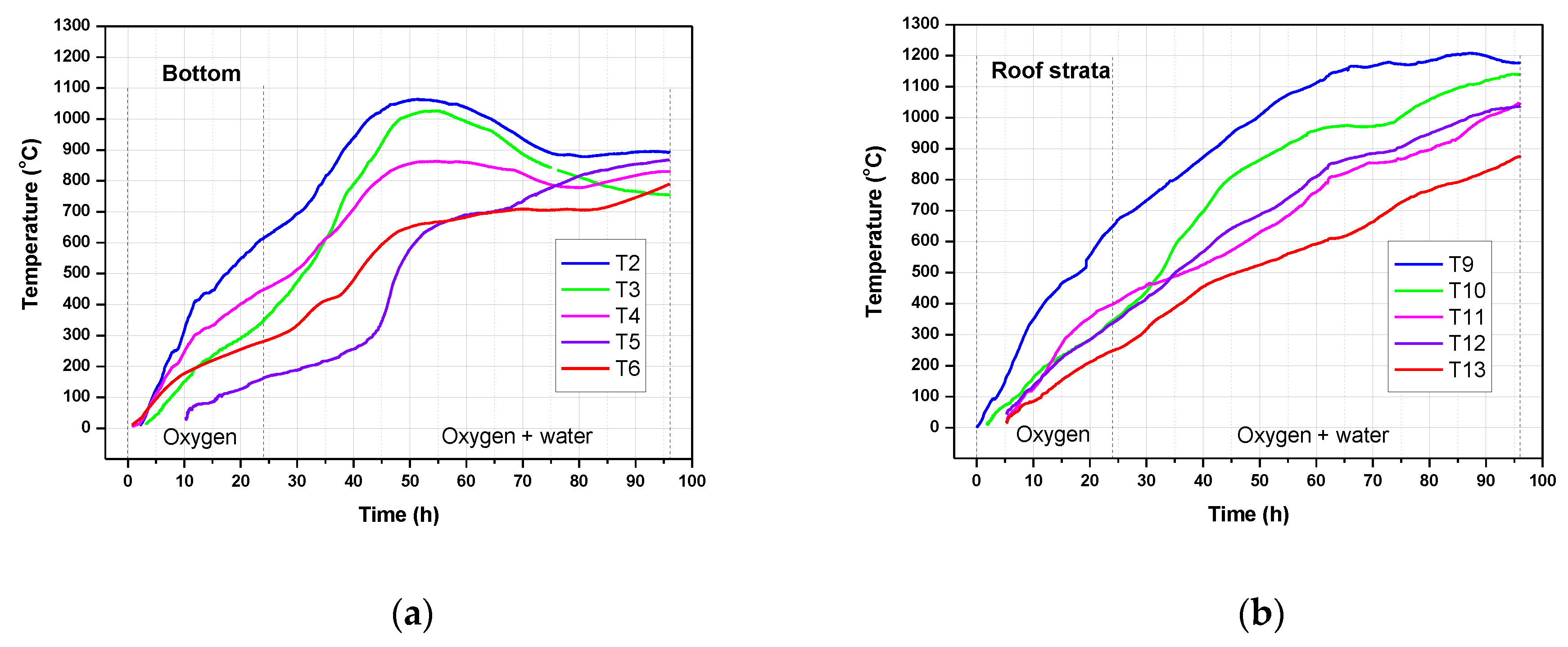
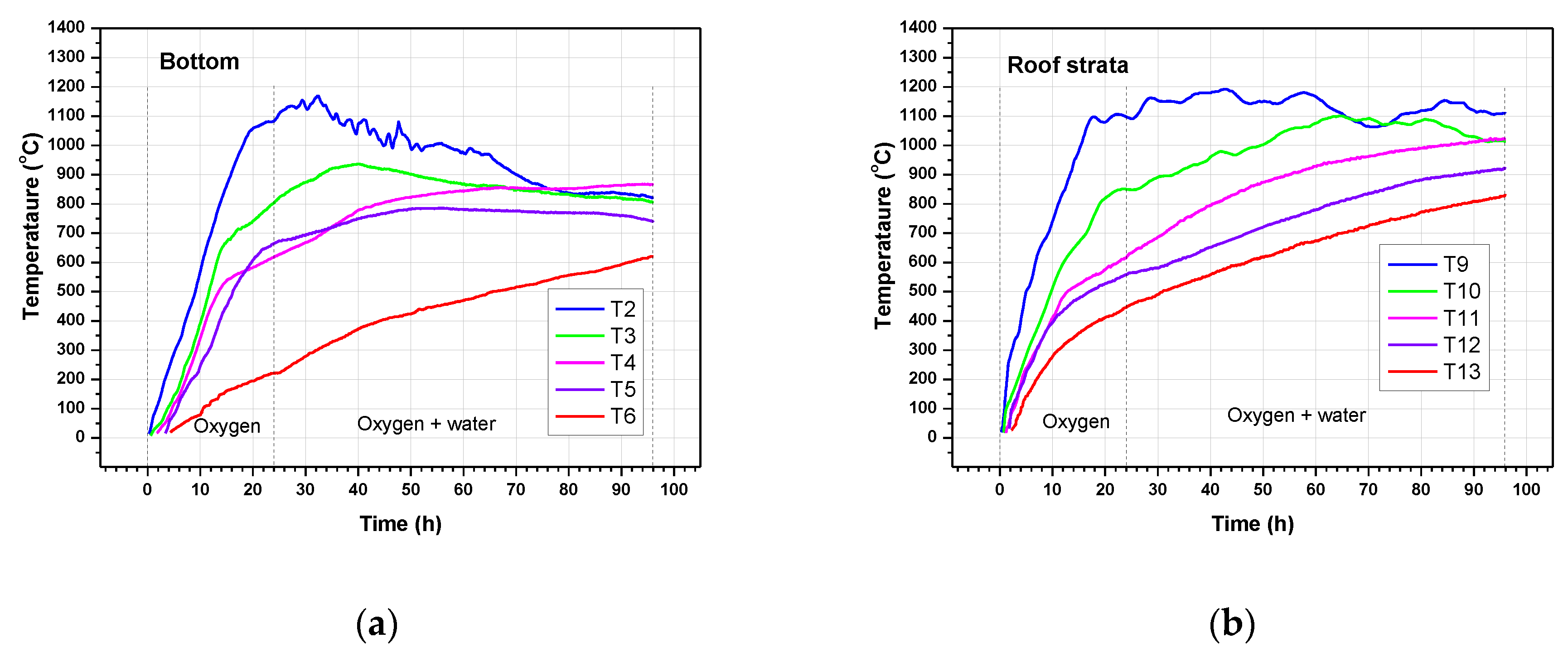
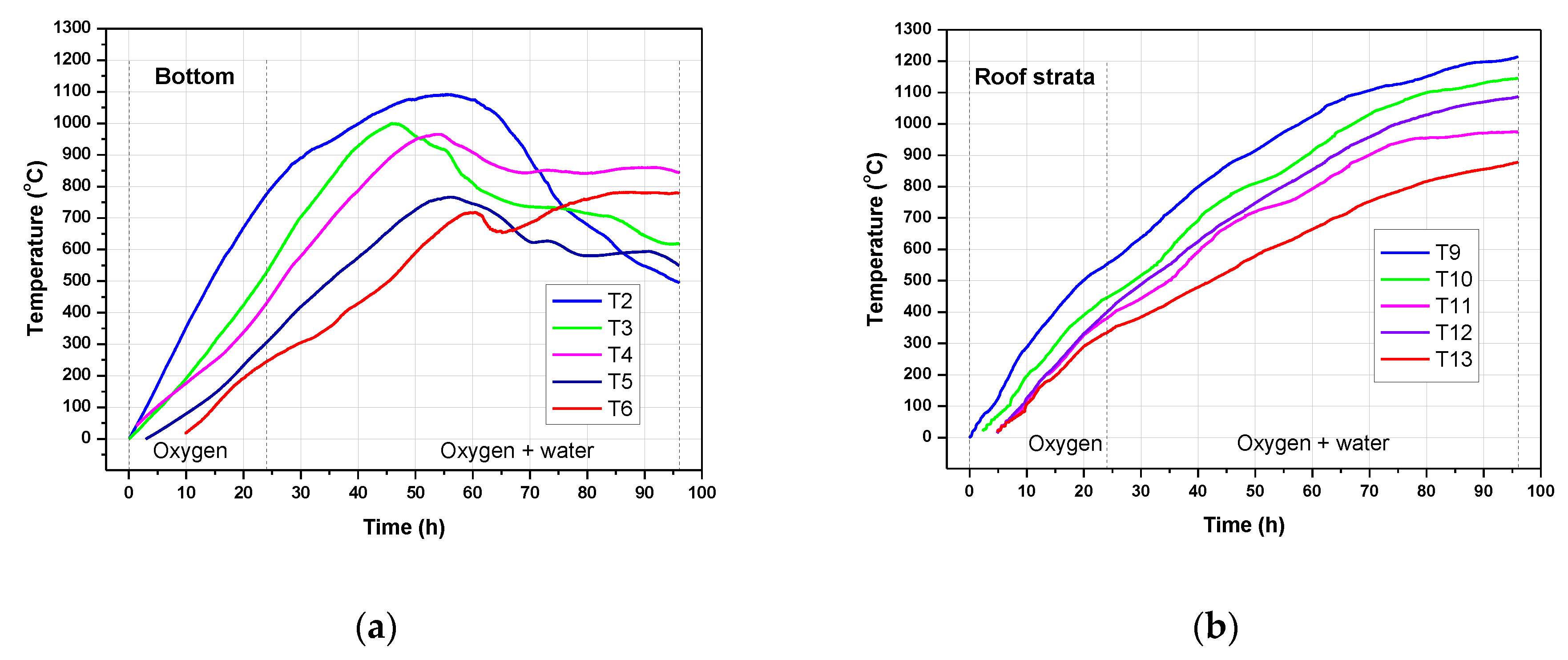
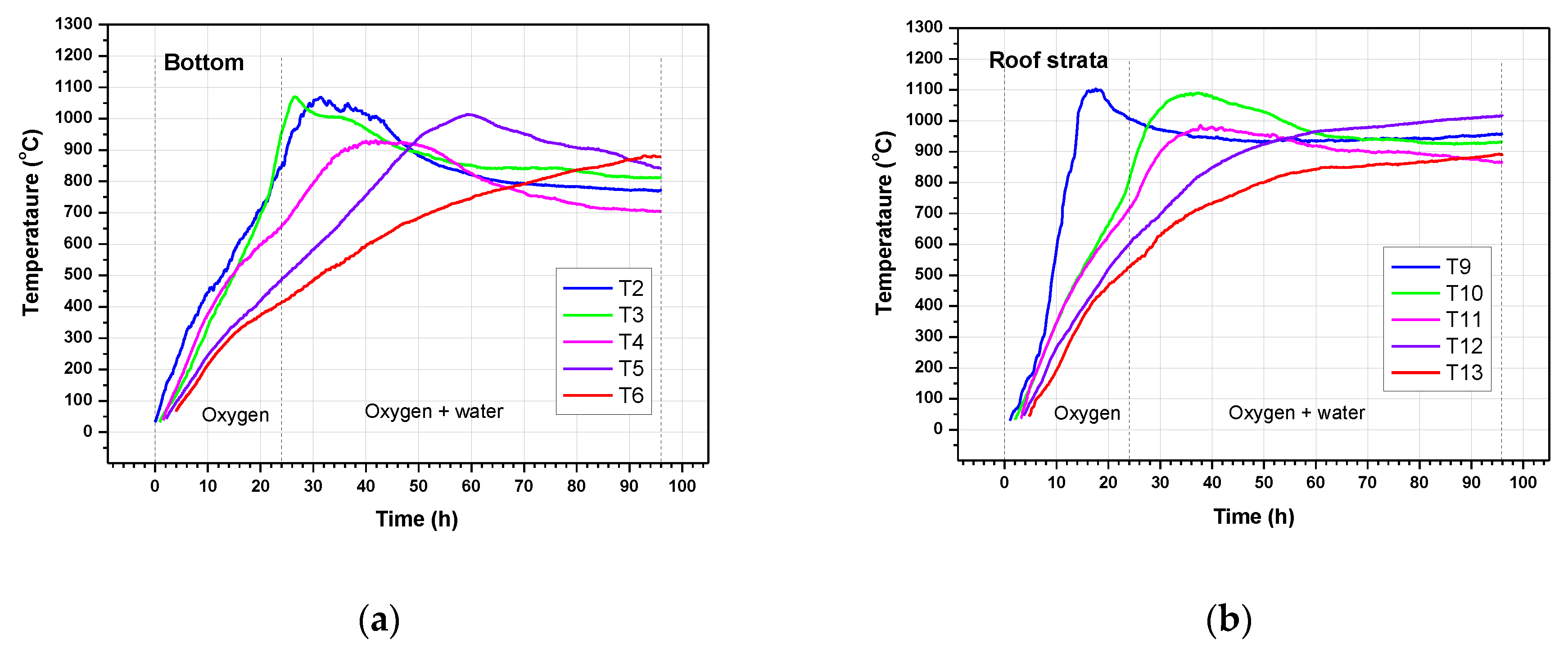
3.3. Temperature Profiles
3.4. Process Balance Calculations
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- International Energy Agency. World Energy Outlook 2019; IEA: Paris, UK, 2019; Available online: https://www.iea.org/reports/world-energy-outlook-2019 (accessed on 13 November 2019).
- Jia, B.; Tsau, J.S.; Barati, R. A review of the current progress of CO2 injection EOR and carbon storage in shale oil reservoirs. Fuel 2019, 236, 404–427. [Google Scholar] [CrossRef]
- Couch, G. Underground Coal Gasification; CCC/151; IEA Clean Coal Centre: London, UK, 2009; p. 129. [Google Scholar]
- Bhutto, A.W.; Bazmi, A.A.; Zahedi, G. Underground coal gasification: From fundamentals to applications. Prog. Energy Combust. Sci. 2013, 39, 189–214. [Google Scholar] [CrossRef]
- Shafirovich, E.; Varma, A. Underground coal gasification: A brief review of current status. Ind. Eng. Res. 2009, 48, 7865–7875. [Google Scholar] [CrossRef]
- Gregg, D.W.; Edgar, T.F. Underground coal gasification. AIChE J. 1978, 24, 753–781. [Google Scholar] [CrossRef]
- Burton, E.; Friedmann, J.; Upadhye, R. Best Practices in Underground Coal Gasification; Contract No. W-7405-Eng-48; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2005. [Google Scholar]
- Fei, M. Underground coal gasification (UCG): A new trend of supply-side economics of fossil fuels. Nat. Gas Ind. B 2016, 3, 312–322. [Google Scholar]
- Nakaten, N.; Schlüter, R.; Azzam, R.; Kempka, T. Development of a techno-economic model for dynamic calculation of cost of electricity, energy demand and CO2 emissions of an integrated UCG-CCS process. Energy 2014, 66, 779–790. [Google Scholar] [CrossRef]
- Maev, S.; Blinderman, M.S.; Gruber, G.P. Underground coal gasification (UCG) to products: Designs, efficiencies, and economics. Undergr. Coal Gasif. Combust. 2018, 435–468. [Google Scholar] [CrossRef]
- Burchart-Korol, D.; Krawczyk, P.; Czaplicka-Kolarz, K.; Smoliński, A. Eco-efficiency of underground coal gasification (UCG) for electricity production. Fuel 2016, 173, 239–246. [Google Scholar] [CrossRef]
- Perkins, G. Underground Coal Gasification. Part I: Field demonstrations and process performance. Progr. Energy Comb. Sci. 2018, 67, 158–187. [Google Scholar] [CrossRef]
- Kapusta, K.; Stańczyk, K. Development conditions and limitations of the underground coal gasification in Poland. Przem. Chem. 2009, 88, 331–338. [Google Scholar]
- Xiao, Y.; Yin, J.; Hu, Y.; Wang, J.; Yin, H.; Qi, H. Monitoring and Control in Underground Coal Gasification: Current Research Status and Future Perspective. Sustainability 2019, 11, 217. [Google Scholar] [CrossRef]
- Kapusta, K.; Wiatowski, M.; Stańczyk, K. An experimental ex-situ study of the suitability of a high moisture ortho-lignite for underground coal gasification (UCG) process. Fuel 2016, 179, 150–155. [Google Scholar] [CrossRef]
- Klebingat, S.; Kempka, T.; Schulten, M.; Azzam, R.; Fernández-Steeger, M.T. Optimization of synthesis gas heating values and tar by-product yield in underground coal gasification. Fuel 2018, 229, 248–261. [Google Scholar] [CrossRef]
- Klebingat, S.; Kempka, T.; Schulten, M.; Azzam, R.; Fernandez-Steeger, T.M. Innovative thermodynamic underground coal gasification model for coupled synthesis gas quality and tar production analyses. Fuel 2016, 183, 680–686. [Google Scholar] [CrossRef]
- Perkins, G. Underground coal gasification—Part II: Fundamental phenomena and modeling. Prog. Energy Combust. Sci. 2018, 67, 234–274. [Google Scholar] [CrossRef]
- Zagoršcak, R.; An, N.; Palange, R.; Green, M.; Krishnan, M.; Thomas, H.R. Underground coal gasification—A numerical approach to study the formation of syngas and its reactive transport in the surrounding strata. Fuel 2019, 253, 349–360. [Google Scholar] [CrossRef]
- Perkins, G.; Sahajwalla, V. A Mathematical Model for the Chemical Reaction of a Semi-infinite Block of Coal in Underground Coal Gasification. Energy Fuels 2005, 19, 1679–1692. [Google Scholar] [CrossRef]
- Stańczyk, K.; Smoliński, A.; Kapusta, K.; Wiatowski, M.; Świądrowski, J.; Kotyrba, A.; Rogut, J. Dynamic experimental simulation of hydrogen oriented underground gasification of lignite. Fuel 2010, 89, 3307–3314. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, X.; Liu, S.; Yu, L.; Zhang, W. Field test of large–scale hydrogen manufacturing from underground coal gasification (UCG). Int. J. Hydrog. Energy 2008, 33, 1275–1285. [Google Scholar] [CrossRef]
- Liu, S.Q.; Wang, Y.Y.; Zhao, K.; Yang, N. Enhanced-hydrogen gas production through underground gasification of lignite. Min. Sci. Technol. 2009, 19, 384–394. [Google Scholar] [CrossRef]
- Yang, L.; Ding, R. The fundamental researches on hydrogen manufacturing from underground coal gasification. Resour. Mater. 1990, 12, 27–32. [Google Scholar]
- Stańczyk, K.; Kapusta, K.; Wiatowski, M.; Świądrowski, J.; Smoliński, A.; Rogut, J.; Kotyrba, A. Experimental simulation of hard coal underground gasification for hydrogen production. Fuel 2012, 91, 40–50. [Google Scholar] [CrossRef]
- Wiatowski, M.; Stańczyk, K.; Świądrowski, J.; Kapusta, K.; Cybulski, K.; Krause, E.; Grabowski, J.; Rogut, J.; Howaniec, N.; Smoliński, A. Semi-technical underground coal gasification (UCG) using the shaft method in Experimental Mine “Barbara”. Fuel 2012, 99, 170–179. [Google Scholar] [CrossRef]
- Wiatowski, M.; Kapusta, K.; Świądrowski, J.; Cybulski, K.; Ludwik-Pardała, M.; Grabowski, J.; Stańczyk, K. Technological aspects of underground coal gasification in the Experimental “Barbara” Mine. Fuel 2015, 159, 454–462. [Google Scholar] [CrossRef]
- Jia, B.; Tsau, J.S.; Barati, R.G. Investigation of Shale-Gas-Production Behavior: Evaluation of the Effects of Multiple Physics on the Matrix. SPE Reserv. Eval. Eng. 2019, 23, 13. [Google Scholar] [CrossRef]
- Wiatowski, M.; Kapusta, K.; Ludwik-Pardała, M.; Stańczyk, K. Ex-situ experimental simulation of hard coal underground gasification at elevated pressure. Fuel 2016, 184, 401–408. [Google Scholar] [CrossRef]
- Wiatowski, M.; Kapusta, K.; Stańczyk, K.; Stańczyk, K. Efficiency assessment of underground gasification of orthoand meta-lignite: High-pressure ex situ experimental simulations. Fuel 2019, 236, 221–227. [Google Scholar] [CrossRef]
- Barady, B.H.G.; Brown, E.T. Rock Mechanics for Underground Mining, 2nd ed.; Chapman and Hall: London, UK, 1993. [Google Scholar]
- Beath, A.; Craig, S.; Littleboy, A.; Mark, R.; Mallett, C. Underground Coal Gasification: Evaluating Environmental Barriers; Exploration and Mining Report P2004/5; CSIRO Exploration and Mining: Canberra, Australia, 2004. [Google Scholar]

| No. | Parameter | Coal Sample | |
|---|---|---|---|
| “Six Feet” Semi-Anthracite | “Wesoła” Hard Coal | ||
| As Received | |||
| 1 | Total Moisture Wtr, % | 1.15 ± 0.40 | 3.60 ± 0.40 |
| 2 | Ash Atr, % | 4.61 ± 0.30 | 8.74 ± 40 |
| 3 | Volatiles Vr, % | 9.92 ± 0.12 | 27.67 ± 0.50 |
| 4 | Total Sulphur Str, % | 1.55 ± 0.04 | 0.31 ± 0.02 |
| 5 | Calorific Value Qir, kJ/kg | 33,416 ± 220 | 28,798 ± 200 |
| Analytical | |||
| 6 | Moisture Wa, % | 0.84 ± 0.30 | 2.18 ± 0.27 |
| 7 | Ash Aa, % | 4.62 ± 0.30 | 8.87 ± 0.63 |
| 8 | Volatiles Va, % | 9.95 ± 0.13 | 28.08 ± 0.92 |
| 9 | Heat of Combustion Qsa, kJ/kg | 34,414 ± 228 | 30,317 ± 161 |
| 10 | Calorific Value Qia, kJ/kg | 33,527 ± 221 | 29,258 ± 201 |
| 11 | Total Sulphur Sa, % | 1.55 ± 0.04 | 0.31 ± 0.08 |
| 12 | Carbon Cta, % | 87.31 ± 0.66 | 75.35 ± 1.13 |
| 13 | Hydrogen Hta, % | 3.97 ± 0.28 | 4.61 ± 0.40 |
| 14 | Nitrogen Na, % | 1.29 ± 0.12 | 1.20 ± 0.22 |
| 15 | Oxygen Oda, % | 0.50 ± 0.05 | 7.65 ± 0.1 |
| 16 | Specific Gravity, g/cm3 | 1.35 ± 0.028 | 1.40 ± 0.018 |
| Parameter | Experiment No. | |||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| Coal Type | semi-anthracite | semi-anthracite | bituminous | bituminous |
| Origin | “Six Feet” deposit (South Wales, UK) | “Six Feet” deposit (South Wales, UK) | “Wesoła” coal (Upper Silesia, Poland) | “Wesoła” coal (Upper Silesia, Poland) |
| Gasification Reagent | O2/H2O | O2/H2O | O2/H2O | O2/H2O |
| Gasification Pressure, Bar | 20 | 40 | 20 | 40 |
| Experiment Duration, h | 96 | 96 | 96 | 96 |
| Gas Production Parameter | Gasification Experiment | |||
|---|---|---|---|---|
| “Six Feet” Semi-anthracite 20 bar | “Wesoła” Hard Coal 20 bar | “Six Feet” Semi-anthracite 40 bar | Wesoła” Hard Coal 40 bar | |
| Average Gas Production Rate, Nm3/h | 9.0 | 9.3 | 9.4 | 9.4 |
| Gas Yield, Nm3/kg of Coal Consumed | 1.98 | 1.77 | 1.98 | 1.70 |
| Gasification Experiment | Average Process Gas Concentration, %vol. | Q, MJ/Nm3 | ||||||
|---|---|---|---|---|---|---|---|---|
| CO2 | N2 | H2 | CH4 | CO | C2H6 | H2S | ||
| “Six Feet” Semi-anthracite 20 bar | 36.3 | 0.4 | 19.2 | 15.8 | 27.2 | 0.69 | 0.38 | 11.7 |
| “Wesoła” Hard Coal 20 bar | 46.3 | 0.7 | 21.6 | 10.9 | 19.5 | 0.64 | 0.37 | 9.2 |
| “Six Feet” Semi-anthracite 40 bar | 41.6 | 0.6 | 14.1 | 19.1 | 23.2 | 1.05 | 0.32 | 12.1 |
| “Wesoła” Hard Coal 40 bar | 46.1 | 0.7 | 17.7 | 14.8 | 19.3 | 0.94 | 0.51 | 10.4 |
| Gasification Experiment | Total Gas Yield, Nm3 | Energy in Gas, MJ | Average Reactor Power, kW | Coal Gasified, kg | Gasification Rate, kg/h | Energy Efficiency, % |
|---|---|---|---|---|---|---|
| “Six feet” Semi-anthracite 20 bar | 864 | 10,117.5 | 29.3 | 436.1 | 4.5 | 69.7 |
| “Wesoła” Hard Coal 20 bar | 896 | 8243.2 | 23.9 | 504.0 | 5.3 | 56.8 |
| “Six feet” Semi-anthracite 40 bar | 903 | 10,890.2 | 31.5 | 455.5 | 4.7 | 71.6 |
| “Wesoła” Hard Coal 40 bar | 903 | 9364.1 | 27.1 | 530.2 | 5.6 | 60.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kapusta, K.; Wiatowski, M.; Stańczyk, K.; Zagorščak, R.; Thomas, H.R. Large-scale Experimental Investigations to Evaluate the Feasibility of Producing Methane-Rich Gas (SNG) through Underground Coal Gasification Process. Effect of Coal Rank and Gasification Pressure. Energies 2020, 13, 1334. https://doi.org/10.3390/en13061334
Kapusta K, Wiatowski M, Stańczyk K, Zagorščak R, Thomas HR. Large-scale Experimental Investigations to Evaluate the Feasibility of Producing Methane-Rich Gas (SNG) through Underground Coal Gasification Process. Effect of Coal Rank and Gasification Pressure. Energies. 2020; 13(6):1334. https://doi.org/10.3390/en13061334
Chicago/Turabian StyleKapusta, Krzysztof, Marian Wiatowski, Krzysztof Stańczyk, Renato Zagorščak, and Hywel Rhys Thomas. 2020. "Large-scale Experimental Investigations to Evaluate the Feasibility of Producing Methane-Rich Gas (SNG) through Underground Coal Gasification Process. Effect of Coal Rank and Gasification Pressure" Energies 13, no. 6: 1334. https://doi.org/10.3390/en13061334
APA StyleKapusta, K., Wiatowski, M., Stańczyk, K., Zagorščak, R., & Thomas, H. R. (2020). Large-scale Experimental Investigations to Evaluate the Feasibility of Producing Methane-Rich Gas (SNG) through Underground Coal Gasification Process. Effect of Coal Rank and Gasification Pressure. Energies, 13(6), 1334. https://doi.org/10.3390/en13061334